As filed with the Securities and Exchange Commission on October 29, 2024
Registration No. 333-281459
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
__________________________
Amendment No. 3 to
FORM S-4
REGISTRATION STATEMENT
UNDER
THE SECURITIES ACT OF 1933
__________________________
PIERIS PHARMACEUTICALS, INC.
(Exact name of co-registrant as specified in its charter)
__________________________
|
Nevada |
2834 |
30-0784346 |
|
(State or jurisdiction |
(Primary Standard Industrial |
(I.R.S. Employer |
|
of incorporation) |
Classification Code Number) |
Identification Number) |
225 Franklin Street, 26th Floor
Boston, Massachusetts 02110
(857) 246-8998
(Address, including zip code, and telephone number, including area code, of registrant’s principal executive offices)
Pieris Pharmaceuticals, Inc.
225 Franklin Street, 26th Floor
Boston, Massachusetts 02110
(857) 246-8998
(Name, address, including zip code, and telephone number, including area code, of agent for service)
__________________________
Copies to:
|
Christopher S. Miller, Esq. Joseph Walsh, Esq. Troutman Pepper Hamilton Sanders LLP Berwyn, Pennsylvania 19312 (610) 640-7800 |
William C. Hicks, Esq. Marc D. Mantell, Esq. Jeffrey D. Cohan, Esq. Mintz, Levin, Cohn, Ferris, Glovsky and Popeo, P.C. One Financial Center Boston, Massachusetts 02111 |
|
(617) 542-6000 |
__________________________
Approximate date of commencement of proposed sale to the public: As soon as practicable after the effective date of this registration statement and the satisfaction or waiver of all other conditions under the Merger Agreement described herein.
If the securities being registered on this Form are being offered in connection with the formation of a holding company and there is compliance with General Instruction G, check the following box: ☐
If this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
If this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
|
Large accelerated filer |
☐ |
Accelerated filer |
☐ |
|||
|
Non-accelerated filer |
☒ |
Smaller reporting company |
☒ |
|||
|
Emerging growth company |
☐ |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 7(a)(2)(B) of the Securities Act. ☐
If applicable, place an X in the box to designate the appropriate rule provision relied upon in conducting this transaction:
Exchange Act Rule 13e-4(i) (Cross-Border Issuer Tender Offer) ☐
Exchange Act Rule 14d-1(d) (Cross-Border Third-Party Tender Offer) ☐
__________________________
The Registrant hereby amends this registration statement on such date or dates as may be necessary to delay its effective date until the Registrant shall file a further amendment which specifically states that this registration statement shall thereafter become effective in accordance with Section 8(a) of the Securities Act of 1933, as amended, or until the registration statement shall become effective on such date as the Securities and Exchange Commission, acting pursuant to said Section 8(a), may determine.
The information in this proxy statement/prospectus is not complete and may be changed. These securities may not be sold until the registration statement filed with the Securities and Exchange Commission is effective. This proxy statement/prospectus is not an offer to sell and it is not soliciting an offer to buy these securities in any jurisdiction where the offer or sale is not permitted.
SUBJECT TO COMPLETION, DATED October 29, 2024
PROPOSED MERGER
YOUR VOTE IS VERY IMPORTANT
_____________________________________________
To the Stockholders of Pieris Pharmaceuticals, Inc. and Palvella Therapeutics, Inc.,
Pieris Pharmaceuticals, Inc., a Nevada corporation, or “Pieris”, and Palvella Therapeutics, Inc., a Delaware corporation, or “Palvella”, entered into an Agreement and Plan of Merger, or the “Merger Agreement”, on July 23, 2024, pursuant to which, subject to the terms and conditions thereof, Polo Merger Sub, Inc., or “Merger Sub”, a Delaware corporation and a wholly owned subsidiary of Pieris, will merge with and into Palvella, with Palvella surviving as a wholly owned subsidiary of Pieris, and the surviving corporation of the merger, which transaction is referred to herein as the “Merger”.
Immediately prior to the effective time of the Merger, or the “Effective Time” each share of Palvella capital stock will be converted into the right to receive a number of shares of Pieris common stock equal to the exchange ratio described in more detail in the section titled “The Merger—Exchange Ratio” beginning on page 152 of the accompanying proxy statement/prospectus. Based on Pieris’ and Palvella’s capitalization as of July 23, 2024, the date the Merger Agreement was executed, the exchange ratio is estimated to be equal to approximately 0.315478222 shares of Pieris common stock for each share of Palvella capital stock. The final exchange ratio is subject to adjustment prior to closing of the Merger based upon Pieris’ net cash at as of the date immediately preceding the closing of the Merger.
In connection with the Merger, each option to purchase Palvella common stock, whether vested or unvested, and all stock or equity-related plans, agreements or arrangements, each and collectively a “Palvella Stock Plan”, that is outstanding immediately prior to the Effective Time will be assumed by Pieris and will become an option to acquire, on the same terms and conditions as were applicable to such Palvella stock option immediately prior to the Effective Time, a number of shares of Pieris common stock equal to the number of shares of Palvella common stock subject to the unexercised portion of the Palvella stock option immediately prior to the Effective Time, multiplied by the exchange ratio (rounded down to the nearest whole share number), with an exercise price per share for the options equal to the exercise price per share of such Palvella stock option immediately prior to the Effective Time divided by the exchange ratio (rounded up to the nearest whole cent). Such assumed options will continue to be governed by the terms and conditions of the Palvella Stock Plan.
Immediately after the Merger, but without giving effect to the PIPE Financing (as defined below), Pieris securityholders as of immediately prior to the Merger are expected to own approximately 18% of the outstanding shares of capital stock of the combined company and former Palvella securityholders, are expected to own approximately 82% of the outstanding shares of capital stock of the combined company, in each case, on a fully-diluted basis, calculated using the treasury stock method, subject to certain assumptions, including, but not limited to, a valuation for Pieris equal to $21 million, provided, that (a) if Pieris’ net cash as of the closing is greater than $11 million, then Pieris’ valuation will be adjusted upwards on a dollar-for-dollar basis by the difference of: (i) Pieris’ net cash, minus (ii) $11 million, and (b) if Pieris’ net cash is less than $11 million, then Pieris’ valuation will be adjusted downwards on a dollar-for-dollar basis by the difference of: (i) $11 million, minus (ii) Pieris’ net cash, as further described in the Merger Agreement.
Certain investors (including the holders of outstanding convertible notes issued by Palvella), or the “PIPE Investors” have agreed to subscribe for and purchase (in cash or by cancellation of convertible notes) an aggregate of approximately 3,154,241 of shares of Pieris common stock, at a price per share equal to $13.7299, multiplied by (x) 0.315478222 divided by (y) the exchange ratio, or the “Purchase Price”, subject to adjustment as set forth in the securities purchase agreement, by and among Pieris and certain investors, or the “Purchase Agreement”, and/or in lieu of Pieris common stock to certain purchasers who so choose, pre-funded warrants, or the Pre-Funded Warrants, to purchase up to 2,592,585 shares of the combined company common stock, at a purchase price per Pre-Funded Warrant equal to the Purchase Price minus $0.001, referred to as the “PIPE Financing”, which is expected to close immediately following the closing of the Merger. The gross proceeds from the PIPE Financing are expected to be approximately $78.9 million, consisting of approximately $60.0 million in cash and the conversion of approximately $18.9 million of principal and interest payable under the outstanding convertible notes issued by Palvella, before paying estimated expenses. The closing of the PIPE Financing is conditioned upon the closing of the Merger, as well as certain other conditions.
Additionally, immediately following the PIPE Financing, the Pieris securityholders as of immediately prior to the Merger are expected to hold approximately 10.47% of the shares of combined company capital stock, the Palvella securityholders as of immediately prior to the Merger are expected to hold approximately 50.18% of the shares of combined company capital stock, and the PIPE Investors are expected to hold approximately 39.35% of the shares of combined company capital stock, in each case, on a fully diluted basis, subject to certain assumptions.
Shares of Pieris common stock are currently listed on The Nasdaq Capital Market under the symbol “PIRS.” On , 2024, the last trading day before the date of the accompanying proxy statement/prospectus, the closing sale price of Pieris common stock was $ per share. Palvella intends to file a listing application for the combined company with The Nasdaq Stock Market Inc., or “Nasdaq”. After completion of the Merger, the combined company will be renamed “Palvella Therapeutics, Inc.” and, assuming approval of the initial listing application, the common stock of the combined company will trade on The Nasdaq Capital Market under the symbol “PVLA.” However, Nasdaq’s determination may not be known at the time stockholders are asked to vote on the Merger.
Pieris stockholders are cordially invited to attend the special meeting of Pieris stockholders, or the Pieris special meeting. Pieris is holding the Pieris special meeting, on Wednesday, December 11, 2024, at 9:00 a.m. Eastern Time, unless postponed or adjourned to a later date, in order to obtain the stockholder approvals necessary to complete the Merger and related matters. The Pieris special meeting will be held entirely online. Pieris stockholders will be able to attend and participate in the Pieris special meeting online by visiting www.virtualshareholdermeeting.com/PIRS2024SM, where they will be able to listen to the meeting live, submit questions and vote. At the Pieris special meeting, Pieris will ask its stockholders:
|
1. |
To approve the issuance of shares of common stock of Pieris pursuant to the terms of the Merger Agreement and the Purchase Agreement (as each may be amended from time to time), copies of which are attached as Annex A and Annex B, respectively, to the accompanying proxy statement/prospectus, and the change of control of Pieris pursuant to the Merger, for purposes of Nasdaq Listing Rules 5635(a), (b) and (d); |
|
2. |
To approve an amendment to the amended and restated articles of incorporation of Pieris to increase the number of authorized shares of Pieris common stock from 3,750,000 shares to 200,000,000 shares; |
|
3. |
To approve the adoption of the Palvella Therapeutics, Inc. 2024 Equity Incentive Plan (as defined in this proxy statement/prospectus); |
|
4. |
To ratify the Pieris board of directors’ approval of an amendment to the amended and restated articles of incorporation of Pieris to change the name of the corporation from “Pieris Pharmaceuticals, Inc.” to “Palvella Therapeutics, Inc.”; and |
|
5. |
To vote upon an adjournment of the Pieris special meeting, if necessary, to solicit additional proxies if there are not sufficient votes in favor of Proposal Nos. 1 and 2. |
As described in the accompanying proxy statement/prospectus, certain Pieris stockholders who in the aggregate owned approximately 19.6% of the outstanding shares Pieris common stock as of July 23, 2024, and certain Palvella stockholders who in the aggregate owned approximately 64.3% of the outstanding shares of Palvella voting capital stock as of July 23, 2024, are parties to stockholder support agreements with Pieris and Palvella, respectively, whereby such stockholders have agreed, subject to the terms of the support agreements, to vote in favor of the approval of the transactions contemplated therein, including, with respect to Palvella stockholders, adoption of the Merger Agreement and approval of the Merger and, with respect to such Pieris stockholders, approval of the issuance of Pieris common stock in the Merger pursuant to the Merger Agreement, and the approval and ratification, respectively, of the amendments to the amended and restated articles of incorporation of Pieris to increase the number of authorized shares and change the name of the corporation from “Pieris Pharmaceuticals, Inc.” to “Palvella Therapeutics, Inc.” Following the effectiveness of this Registration Statement on Form S-4 of which the accompanying proxy statement/prospectus is a part and pursuant to the Merger Agreement, Palvella stockholders holding a sufficient number of shares of Palvella voting capital stock to adopt the Merger Agreement and approve the Merger and related transactions will be asked to execute written consents providing for such adoption and approval.
After careful consideration, each of the Pieris and Palvella boards of directors has approved the Merger Agreement and has determined that it is advisable to consummate the Merger and the other transactions contemplated by the Merger Agreement. The Pieris board of directors has approved the proposals described in the accompanying proxy statement/prospectus and recommends that its stockholders vote “FOR” the proposals described in the accompanying proxy statement/prospectus.
_____________________________________________
More information about Pieris, Palvella, the Merger Agreement and transactions contemplated thereby and the foregoing proposals is contained in the accompanying proxy statement/prospectus. Pieris urges you to read the accompanying proxy statement/prospectus carefully and in its entirety. IN PARTICULAR, YOU SHOULD CAREFULLY CONSIDER THE MATTERS DISCUSSED UNDER “RISK FACTORS” BEGINNING ON PAGE 25 OF THE ACCOMPANYING PROXY STATEMENT/PROSPECTUS.
Each of Pieris and Palvella are excited about the opportunities the Merger brings to Pieris and Palvella stockholders and thank you for your consideration and continued support.
Sincerely,
|
Stephen S. Yoder |
Wesley H. Kaupinen |
|
Chief Executive Officer |
Chief Executive Officer |
|
Pieris Pharmaceuticals, Inc. |
Palvella Therapeutics, Inc. |
Neither the Securities and Exchange Commission, or “SEC”, nor any state securities commission has approved or disapproved of these securities or passed upon the adequacy or accuracy of the accompanying proxy statement/prospectus. Any representation to the contrary is a criminal offense.
The accompanying proxy statement/prospectus is dated , and is first being mailed to Pieris stockholders on or about , .
PIERIS PHARMACEUTICALS, INC.
225 Franklin Street, 26th Floor
Boston, Massachusetts 02110
(857) 246-8998
NOTICE OF SPECIAL MEETING OF STOCKHOLDERS
To the stockholders of Pieris Pharmaceuticals, Inc.:
NOTICE IS HEREBY GIVEN that the Pieris special meeting will be held on Wednesday, December 11, 2024, at 9:00 a.m. Eastern Time, unless postponed or adjourned to a later date. The Pieris special meeting will be held entirely online. You will be able to attend and participate in the Pieris special meeting online by visiting www.virtualshareholdermeeting.com/PIRS2024SM, where you will be able to listen to the meeting live, submit questions and vote.
The Pieris special meeting will be held for the following purposes:
|
1. |
To approve the issuance of shares of common stock of Pieris pursuant to the terms of the Merger Agreement and the Purchase Agreement (as each may be amended from time to time), copies of which are attached as Annex A and Annex B, respectively, to the accompanying proxy statement/prospectus, and the change of control of Pieris pursuant to the Merger, for purposes of Nasdaq Listing Rules 5635(a), (b) and (d); |
|
2. |
To approve an amendment to the amended and restated articles of incorporation of Pieris to increase the number of authorized shares of Pieris common stock from 3,750,000 shares to 200,000,000 shares; |
|
3. |
To approve the adoption of the Palvella Therapeutics, Inc. 2024 Equity Incentive Plan (as defined in this proxy statement/prospectus); |
|
4. |
To ratify the Pieris board of directors’ approval of an amendment to the amended and restated articles of incorporation of Pieris to change the name of the corporation from “Pieris Pharmaceuticals, Inc.” to “Palvella Therapeutics, Inc.”; and |
|
5. |
To vote upon an adjournment of the Pieris special meeting, if necessary, to solicit additional proxies if there are not sufficient votes in favor of Proposal Nos. 1 and 2. |
|
Record Date: |
The Pieris board of directors has fixed the close of business on October 28, 2024 as the record date for the determination of stockholders entitled to notice of, and to vote at, the Pieris special meeting and any adjournment or postponement thereof. Only holders of record of shares of Pieris common stock and Series F Preferred Stock at the close of business on the record date are entitled to notice of, and to vote at, the Pieris special meeting. At the close of business on the record date, there were 1,320,240 shares of Pieris common stock issued and outstanding and one share of Series F Preferred Stock issued and outstanding. Each share of Pieris common stock entitles the holder thereof to one vote on each matter submitted for stockholder approval. Each share of Series F Preferred Stock entitles the holder thereof to 25,000,000 votes on Proposal No. 2, which are cast in the same proportion as shares of Pieris common stock are voted “for” and “against” Proposal No. 2, excluding any shares of common stock that are not voted “for” or “against” the Authorized Share Increase for any reason, including, without limitation, any abstentions or broker non-votes. |
Your vote is important. Assuming a quorum is present (i) the affirmative vote of the holders of a majority in voting power of the outstanding shares of Pieris common stock and preferred stock, voting as a single class, entitled to vote thereon is required for approval of Proposal No. 2 and (ii) the affirmative vote of the holders of a majority of the votes cast on the proposal by the holders of shares of Pieris common stock present or represented at the meeting (with abstentions and broker non-votes not considered as votes cast) is required for approval of Proposal Nos. 1, 3, 4 and 5. Approval of Proposal Nos. 1 and 2 is a condition to the completion of the Merger. Therefore, the Merger cannot be consummated without the approval of Proposal Nos. 1 and 2.
Pieris requests that you sign and return the enclosed proxy or submit a proxy to vote online, by telephone or by mail before the special meeting date to ensure that your shares will be represented at the Pieris special meeting, even if you plan to virtually attend. You may change or revoke your proxy at any time before it is voted at the Pieris special meeting.
THE PIERIS BOARD OF DIRECTORS HAS DETERMINED AND BELIEVES THAT EACH OF THE PROPOSALS OUTLINED ABOVE IS FAIR TO, IN THE BEST INTERESTS OF, AND ADVISABLE TO PIERIS AND ITS STOCKHOLDERS AND HAS APPROVED EACH SUCH PROPOSAL. THE PIERIS BOARD OF DIRECTORS RECOMMENDS THAT PIERIS STOCKHOLDERS VOTE “FOR” EACH SUCH PROPOSAL.
|
Important Notice Regarding the Availability of Proxy Materials for the Stockholders’ Meeting to Be Held on Wednesday, December 11, 2024 at 9:00 a.m. Eastern Time via the Internet The proxy statement/prospectus and annual report to stockholders are available at www.virtualshareholdermeeting.com/PIRS2024SM. |
By Order of the Pieris board of directors,
|
Stephen S. Yoder |
|
President and Chief Executive Officer |
|
Pieris Pharmaceuticals, Inc. |
Boston, Massachusetts
, 2024
REFERENCES TO ADDITIONAL INFORMATION
This proxy statement/prospectus incorporates important business and financial information about Pieris that is not included in or delivered with this document. You may obtain this information without charge through the SEC website (www.sec.gov) or upon your written or oral request by contacting the Corporate Secretary of Pieris Pharmaceuticals, Inc., 225 Franklin Street, 26th Floor, Boston, Massachusetts 02110, by calling (857) 246-8998 or via email to info@pieris.com.
You may also request this information from our proxy solicitor, Alliance Advisors LLC, or “Alliance Advisors”, using the following contact information:
Alliance Advisors LLC
200 Broadacres Drive, 3rd Floor
Bloomfield, New Jersey 07003
833-782-7190 (toll free)
PIRS@allianceadvisors.com (Email)
To ensure timely delivery of these documents, any request should be made no later than to receive them before the Pieris special meeting.
For additional details about where you can find information about Pieris, please see the section titled “Where You Can Find More Information” beginning on page 336 of this proxy statement/prospectus.
Page
|
PROPOSAL NO. 5: APPROVAL OF POSSIBLE ADJOURNMENT OF THE PIERIS SPECIAL MEETING |
|
QUESTIONS AND ANSWERS ABOUT THE MERGER
The following section provides answers to frequently asked questions about the Merger. This section, however, provides only summary information. For a more complete response to these questions and for additional information, please refer to the cross-referenced sections. You should carefully read this entire proxy statement/prospectus, including each of the annexes.
Q: What is the Merger?
A: Pieris and Palvella have entered into the Merger Agreement, dated as of July 23, 2024, a copy of which is attached as Annex A to this proxy statement/prospectus. The Merger Agreement contains the terms and conditions of the proposed business combination of Pieris and Palvella. Pursuant to the Merger Agreement, Merger Sub a Delaware corporation and a wholly-owned subsidiary of Pieris, will merge with and into Palvella, with Palvella surviving as a wholly owned subsidiary of Pieris. After the completion of the Merger, Pieris will change its corporate name to “Palvella Therapeutics, Inc.” The combined company following the Merger is referred to herein as the “combined company”.
Immediately prior to the Effective Time, each share of Palvella capital stock will be converted into the right to receive a number of shares of Pieris common stock equal to the Exchange Ratio described in more detail in the section titled “The Merger—Exchange Ratio” beginning on page 152 of the accompanying proxy statement/prospectus. Based on Pieris’ and Palvella’s capitalization as of July 23, 2024, the date the Merger Agreement was executed, the exchange ratio is estimated to be equal to approximately 0.315478222 shares of Pieris common stock for each share of Palvella capital stock. The final exchange ratio is subject to adjustment prior to closing of the Merger based upon Pieris’ net cash as of the date immediately preceding the closing of the Merger.
In connection with the Merger, each stock option granted under the Palvella Stock Plan, that is outstanding immediately prior to the Effective Time will be assumed by Pieris and will become an option to acquire, on the same terms and conditions as were applicable to such Palvella stock option immediately prior to the Effective Time, a number of shares of Pieris common stock equal to the number of shares of Palvella common stock subject to the unexercised portion of the Palvella stock option immediately prior to the Effective Time, multiplied by the exchange ratio (rounded down to the nearest whole share number), with an exercise price per share for the options equal to the exercise price per share of such Palvella stock option immediately prior to the Effective Time divided by the exchange ratio (rounded up to the nearest whole cent). Such assumed options will continue to be governed by the terms and conditions of the Palvella Stock Plan.
Each share of Pieris common stock and option to purchase Pieris common stock that is issued and outstanding at the Effective Time will remain issued and outstanding and such shares and options will be unaffected by the Merger. Immediately after the Merger, but without giving effect to the PIPE Financing, Pieris securityholders as of immediately prior to the Merger are expected to own approximately 18% of the outstanding shares of capital stock of the combined company and former Palvella securityholders, are expected to own approximately 82% of the outstanding shares of capital stock of the combined company, in each case, on a fully-diluted basis, calculated using the treasury stock method, subject to certain assumptions, including, but not limited to, a valuation for Pieris equal to $21 million, provided, that (a) if Pieris’ net cash as of the closing is greater than $11 million, then Pieris’ valuation will be adjusted upwards on a dollar-for-dollar basis by the difference of: (i) Pieris’ net cash, minus (ii) $11 million, and (b) if Pieris’ net cash is less than $11 million, then Pieris’ valuation will be adjusted downwards on a dollar-for-dollar basis by the difference of: (i) $11 million, minus (ii) Pieris’ net cash, as further described in the Merger Agreement.
The PIPE Investors have agreed to subscribe for and purchase (in cash or by cancellation of convertible notes) an aggregate of approximately 3,154,241 of shares of Pieris common stock, at a price per share equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, and/or in lieu of Pieris common stock to certain purchasers who so choose, Pre-Funded Warrants to purchase up to 2,592,585 shares of the combined company common stock, at a purchase price per Pre-Funded Warrant equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, minus $0.001, referred to as the “PIPE Financing”, which is expected to close immediately following the closing of the Merger. The gross proceeds from the PIPE Financing are expected to be approximately $78.9 million, consisting of approximately $60.0 million in cash and the conversion of approximately $18.9 million of principal and interest payable under the outstanding convertible notes issued by Palvella, before paying estimated expenses. The closing of the PIPE Financing is conditioned upon the closing of the Merger as well as certain other conditions.
Additionally, immediately following the PIPE Financing, the Pieris securityholders as of immediately prior to the Merger are expected to hold approximately 10.47% of the shares of combined company capital stock, the Palvella securityholders as of immediately prior to the Merger are expected to hold approximately 50.18% of the shares of combined company capital stock, and the PIPE Investors are expected to hold approximately 39.35% of the shares of combined company capital stock, in each case, on a fully diluted basis, subject to certain assumptions.
Q: Why are the two companies proposing to merge?
A: Pieris’ and Palvella’s management believe that combining the two companies will result in a company with a robust pipeline, strong leadership team and substantial capital resources, positioning it to potentially become a leading company focused on developing and, if approved, commercializing novel therapies to treat patients suffering from rare skin diseases for which there are no U.S. Food and Drug Administration, or “FDA,” approved therapies. ”. For a more complete description of the reasons for the Merger, please see the sections titled “The Merger—Pieris Reasons for the Merger” and “The Merger—Palvella Reasons for the Merger” beginning on pages 135 and 137, respectively, of this proxy statement/prospectus.
Q: Why am I receiving this proxy statement/prospectus?
A: You are receiving this proxy statement/prospectus because you have been identified as a stockholder of Pieris as of the close of business on October 28, 2024, or the “record date”, and you are entitled to vote at the Pieris special meeting to approve the matters set forth herein. This document serves as:
|
● |
a proxy statement of Pieris used to solicit proxies for the Pieris special meeting to vote on the matters set forth herein; and |
|
● |
a prospectus of Pieris used to offer shares of Pieris common stock in exchange for shares of Palvella capital stock, including those shares of Palvella common stock to be issued upon conversion of Palvella’s stock options. |
Q: What is the PIPE Financing?
A: On July 23, 2024, Pieris entered into the Purchase Agreement with the PIPE Investors, pursuant to which the investors have agreed to subscribe for and purchase (in cash or by cancellation of convertible notes) an aggregate of approximately 3,154,241 of shares of Pieris common stock, at a price per share equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, and/or in lieu of Pieris common stock to certain purchasers who so choose, Pre-Funded Warrants to purchase up to 2,592,585 shares of the combined company common stock, at a purchase price per Pre-Funded Warrant equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, minus $0.001, referred to as the “PIPE Financing”, which is expected to close immediately following the closing of the Merger. The gross proceeds from the PIPE Financing are expected to be approximately $78.9 million, consisting of approximately $60.0 million in cash and the conversion of approximately $18.9 million of principal and interest payable under the outstanding convertible notes issued by Palvella, before paying estimated expenses. The closing of the PIPE Financing is conditioned upon the closing of the Merger as well as certain other conditions.
Additionally, immediately following the PIPE Financing, the Pieris securityholders as of immediately prior to the Merger are expected to hold approximately 10.47% of the shares of combined company capital stock, the Palvella securityholders as of immediately prior to the Merger are expected to hold approximately 50.18% of the shares of combined company capital stock, and the PIPE Investors are expected to hold approximately 39.35% of the shares of combined company capital stock, in each case, on a fully diluted basis, subject to certain assumptions.
Q: What proposals will be voted on at the Pieris special meeting the approval of which are conditions to the closing of the Merger?
A: Pursuant to the terms of the Merger Agreement, the following proposals must be approved by the requisite stockholder vote at the Pieris special meeting in order for the Merger to close:
|
● |
Proposal No. 1, to approve the issuance of shares of common stock of Pieris pursuant to the terms of the Merger Agreement and the Purchase Agreement (as each may be amended from time to time), copies of which are attached as Annex A and Annex B, respectively, to the accompanying proxy statement/prospectus, and the change of control of Pieris pursuant to the Merger for purposes of The Nasdaq , Listing Rules 5635(a), (b) and (d), or “Proposal No. 1”; and |
|
● |
Proposal No. 2, to approve an amendment to the amended and restated articles of incorporation of Pieris to increase the number of authorized shares of Pieris common stock from 3,750,000 shares to 200,000,000 shares, or “Proposal No. 2” or the “Authorized Shares Increase Proposal”. |
Approval of each of Proposal Nos. 1 and 2 is a condition to the completion of the Merger. Therefore, the Merger cannot be consummated without the approval of Proposal Nos. 1 and 2.
In addition to the requirement of obtaining Pieris stockholder approval, each of the other closing conditions set forth in the Merger Agreement must be satisfied or waived. For a more complete description of the closing conditions under the Merger Agreement, please see the section titled “The Merger Agreement—Conditions to the Completion of the Merger” beginning on page 172 of this proxy statement/prospectus.
Q: What proposals are to be voted on at the Pieris special meeting, other than Proposal Nos. 1 and 2?
A: At the Pieris special meeting, the holders of Pieris common stock will also be asked to consider the following proposals:
|
● |
Proposal No. 3, to approve the adoption of the Palvella Therapeutics, Inc. 2024 Equity Incentive Plan, or “Proposal No. 3”; |
|
● |
Proposal No. 4, to ratify the Pieris board of directors’ approval of an amendment to the amended and restated articles of incorporation of Pieris to change the name of the corporation from “Pieris Pharmaceuticals, Inc.” to “Palvella Therapeutics, Inc.”, or “Proposal No. 4”; and |
|
● |
Proposal No. 5, to vote upon an adjournment of the Pieris special meeting, if necessary, to solicit additional proxies if there are not sufficient votes in favor of Proposal Nos. 1 and 2, or “Proposal No. 5”. |
The approval of Proposal Nos. 3, 4 and 5 are not a condition to the Merger. Such proposals, together with Proposal Nos. 1 and 2, are referred to collectively in this proxy statement/prospectus as the “Proposals”. Pursuant to Section 78.390(8) of the NRS, no stockholder approval is required in connection with the amendment to Pieris’ amended and restated articles of incorporation to change the corporation’s name as described above. However, the Pieris board of directors believes it is important to receive the views of its stockholders in connection with the proposed amendment, and seeks ratification of the Pieris board of directors’ approval of the amendment in Proposal No. 4.
The presence, by attending online or being represented by proxy, at the Pieris special meeting of the holders of a majority in voting power of the shares of Pieris capital stock outstanding and entitled to vote at the Pieris special meeting is necessary to constitute a quorum at the meeting for the purpose of approving the Proposals.
Q: What stockholder votes are required to approve the proposals at the Pieris special meeting?
A: Assuming a quorum is present, the affirmative vote of the holders of a majority of the votes cast on the proposal by the holders of shares of Pieris common stock present or represented at the meeting is required for approval of Proposal Nos. 1, 3, 4 and 5. Abstentions and broker non-votes, if any, are not considered votes cast and therefore will have no effect on the outcome of Proposal Nos. 1, 3, 4 and 5.
Assuming a quorum is present, the affirmative vote of the holders of a majority in voting power of the outstanding shares of Pieris common stock and preferred stock, voting as a single class, entitled to vote at the Pieris special meeting is required for approval of Proposal No. 2. Abstentions and broker non-votes, if any, will have the same effect as “AGAINST” votes on Proposal No. 2, but will not be considered as votes cast “AGAINST” for the purposes of determining the proportion of Series F Preferred stock votes that will be cast “FOR” and “AGAINST”.
As of October 28, 2024, the directors and certain executive officers of Pieris owned or controlled approximately 0.07% of the outstanding shares of Pieris common stock entitled to vote at the Pieris special meeting. As of October 28, 2024, the Pieris stockholders that are party to support agreements, including the directors and certain executive officers of Pieris, owned an aggregate of 258,496 shares of Pieris common stock representing approximately 19.6% of the outstanding shares of Pieris common stock. Pursuant to the support agreements, these stockholders, including the directors and certain executive officers of Pieris, have agreed to vote all shares of Pieris common stock owned by them as of the record date in favor of Proposal Nos. 1, 2, 3 and 4.
Q: How many votes can be cast by all stockholders?
A: As of October 28, 2024, a total of 1,320,240 shares of Pieris common stock were outstanding, all of which are entitled to vote with respect to all matters to be acted upon at the Pieris special meeting. Each stockholder of record is entitled to one vote for each share of our common stock held by such stockholder.
There was one share of our Series F Preferred Stock, par value $0.001 per share, or the “Series F Preferred Stock”, authorized and outstanding on the record date. The Series F Preferred Stock is designed to ensure the Authorized Shares Increase Proposal will be approved if the holders of a majority of the Pieris common stock actually vote in favor of the Authorized Shares Increase Proposal. To that end, the Series F Preferred Stock entitles and requires the holder thereof, as such, to (i) vote together with the holders of Pieris common stock as a single class on the Authorized Shares Increase Proposal and (ii) cast 25,000,000 votes, in person or by proxy, on such Authorized Shares Increase Proposal in a manner that is proportionate to the manner in which all shares of Pieris common stock are voted FOR and AGAINST the Authorized Shares Increase Proposal, excluding any shares of common stock that are not voted “FOR” or “AGAINST” such proposal for any reason, including, without limitation, any abstentions or broker non-votes. As an example, if 50.5% of the shares of Pieris common stock that are voted either “FOR” or “AGAINST” the Authorized Shares Increase Proposal are voted FOR the Authorized Shares Increase Proposal, 50.5% of the votes cast by the holder of the Series F Preferred Stock will be cast as votes FOR the Authorized Shares Increase Proposal. Abstentions and broker non-votes, if any, will have the same effect as “AGAINST” votes on the Authorized Shares Increase Proposal, but will not be considered as votes cast “AGAINST” for the purposes of determining the proportion of Series F Preferred stock votes that will be cast “FOR” and “AGAINST”.
Q: How is a quorum reached?
A: Pieris’ amended and restated bylaws, as amended, or the “amended and restated bylaws”, provide that the holders of a majority of the voting power of all of the shares of capital stock of the corporation entitled to vote at the meeting, present or represented by proxy, will constitute a quorum for the transaction of business at the Pieris special meeting.
Under Nevada law, abstentions and “broker non-votes” (shares held by a broker or nominee that are represented at the Pieris special meeting, but with respect to which the broker or nominee is not instructed by the beneficial owner of such shares to vote on the particular proposal) are counted as present for purposes of determining whether a quorum is present at the Pieris special meeting.
For purposes of determining the existence of a quorum at the Pieris special meeting, only the voting power represented by the shares of Pieris common stock outstanding on the record date will be counted as present for purposes of determining whether a quorum is present at the Pieris special meeting. The voting power represented by the one outstanding share of Series F Preferred Stock will not be counted as present for purposes of determining whether a quorum is present at the Pieris special meeting.
If, for any reason, a quorum is not present, the meeting may be adjourned until a quorum is obtained.
Q: What will be the impact of the Series F Preferred Stock on the Authorized Shares Increase Proposal?
A: On August 7, 2024, the Pieris board of directors authorized the designation of one (1) share of Series F Preferred Stock to ensure the Authorized Shares Increase Proposal will be approved if a majority of votes cast by the Pieris stockholders at the Pieris special meeting are voted in favor of the Authorized Shares Increase Proposal. On August 7, 2024, Pieris also entered into the Subscription and Investment Representation Agreement (the “Preferred Subscription Agreement”) with James Geraghty, Chairman of the Pieris board of directors, pursuant to which Pieris agreed to issue and sell one (1) share of Series F Preferred Stock for $1.00 cash.
Under Nevada law, the affirmative vote of the holders of a majority in voting power of the outstanding shares of Pieris common stock and preferred stock, voting as a single class, entitled to vote thereon is required to approve the Authorized Shares Increase Proposal. The Series F Preferred Stock was issued to Mr. Geraghty to address the increasing challenge faced by many corporations in obtaining stockholder approval on proposals which require a majority in voting power of a company’s outstanding shares, given that many outstanding shares of public companies are held by brokers and other record holders in “street name.” Recently, several large brokerage firms have eliminated discretionary voting, which has the effect of decreasing the number of shares represented at stockholder meetings. As a result, public companies with significant retail stockholders are finding it difficult to obtain stockholder participation sufficient to achieve a majority in voting power of the outstanding shares. At the Pieris 2023 annual meeting of stockholders, only 34% of Pieris’ common stock was voted FOR or AGAINST the non-discretionary matters being voted upon at the special meeting. By issuing the Series F Preferred Stock to Mr. Geraghty, the Pieris board of directors aimed to address the challenge of stockholder proposals failing due to the higher majority in voting power of the outstanding shares voting threshold, despite a majority of the votes cast at the special meeting being voted in favor of the proposal. Additionally, the Pieris board of directors assessed (a) the likelihood that the Authorized Shares Increase Proposal will be treated as non-routine under Rule 452 of the New York Stock Exchange and be excluded from the list of proposals on which a broker may vote shares held in “street name” in the absence of stockholder voting instructions and (b) that the Authorized Shares Increase Proposal is a condition to completion of the Merger. Consequently, the Pieris board of directors considered issuance of the Series F Preferred Stock to Mr. Geraghty necessary to achieve the required vote on the Authorized Shares Increase Proposal.
The Series F Preferred Stock requires the holder thereof, as such, to (i) vote together with the holders of Pieris common stock on the Authorized Shares Increase Proposal and (ii) cast a portion of the 25,000,000 votes of the Series F Preferred Stock, in person or by proxy, FOR the Authorized Shares Increase Proposal in a manner that is proportionate to the percentage of shares of Pieris common stock that are voted FOR the Authorized Shares Increase Proposal as compared with the total shares of Pieris common stock that are voted FOR and AGAINST the Authorized Shares Increase Proposal. For the avoidance of doubt, the Series F Preferred Stock votes on a proportionate basis to the shares voted FOR and AGAINST the Authorized Shares Increase Proposal and does not reflect any shares of common stock that are not voted “FOR” or “AGAINST” such proposal for any reason, including, without limitation, any abstentions or broker non-votes. As an example, if 50.5% of the shares of Pieris common stock that are voted either “FOR” or “AGAINST” the Authorized Shares Increase Proposal are voted FOR the Authorized Shares Increase Proposal, 50.5% of the votes cast by the holder of the Series F Preferred Stock will be cast as votes FOR the Authorized Shares Increase Proposal. Abstentions and broker non-votes, if any, will have the same effect as “AGAINST” votes on the Authorized Shares Increase Proposal, but will not be considered as votes cast as “AGAINST” for the purposes of determining the proportion of Series F Preferred Stock votes that will be cast “FOR” and “AGAINST”.
Below is an illustration of the significant impact of the Series F Preferred Stock on the vote needed to pass the Authorized Shares Increase Proposal. Given that the Authorized Shares Increase Proposal will likely be treated as non-routine under Rule 452 of the New York Stock Exchange and be excluded from the list of proposals on which a broker may vote shares held in “street name” in the absence of stockholder voting instructions, there will be a significant impact on the likelihood of achieving the vote needed to pass the Authorized Shares Increase Proposal if Pieris common stockholders abstain from voting on or decline to provide voting instructions with respect to the Authorized Shares Increase Proposal.
The below scenarios both assume that (i) forty-five percent of the common stock vote FOR or AGAINST the Authorized Shares Increase Proposal, which we believe is a realistic amount based on the historical vote total from Pieris’ prior stockholder proposal votes on non-discretionary matters at its previous two annual meetings of stockholders, which range from 34% to 43% of the common stock, (ii) a minimum of 258,496 shares of Pieris common stock held by Pieris stockholders that are party to a support agreement, representing approximately 19.58% of the outstanding shares of Pieris common stock as of October 28, 2024, vote FOR the Authorized Shares Increase Proposal, which they are required to do in accordance with the support agreement, and (iii) a minimum quorum of a majority of the voting power of Pieris common stock is present at the Pieris special meeting (which includes shares constituting broker non-votes, if any, which will be treated as shares present for the purpose of determining the presence of a quorum).
WITHOUT SERIES F PREFERRED: Assuming only 45% of Pieris common stock (594,108 shares) votes FOR or AGAINST the Authorized Shares Increase Proposal without Series F Preferred Stock:
|
Common Stockholders voting FOR |
Votes FOR |
Votes FOR as % of Outstanding Voting Power FN1 |
Votes AGAINST |
Votes AGAINST as % of Outstanding Voting Power |
Abstain/No Voting Instructions |
Abstain % of Outstanding Voting Power |
Pass/Fail |
|
45% of votes cast* |
267,349 |
20.25% |
326,759 |
24.75% |
726,132 |
55% |
FAIL (20.25% of Outstanding Voting Power) |
|
55% of votes cast* |
326,759 |
24.75% |
267,349 |
20.25% |
726,132 |
55% |
FAIL (24.75% of Outstanding Voting Power) |
|
95% of votes cast* |
564,403 |
42.75% |
29,705 |
2.25% |
726,132 |
55% |
FAIL (42.75% of Outstanding Voting Power) |
FN1: There are 1,320,240 shares of common stock votes comprising total outstanding voting power.
* Includes 258,496 shares of Pieris common stock held by Pieris stockholders that are party to a support agreement.
WITH SERIES F PREFERRED: Assuming only 45% of Pieris common stock (594,108 shares) vote FOR or AGAINST the Authorized Shares Increase Proposal with Series F Preferred Stock:
|
Common Stockholders voting FOR |
Votes FORFN1 |
Votes FOR as % of Outstanding Voting Power FN2 |
Votes AGAINSTFN3 |
Votes AGAINST as % of Outstanding Voting PowerFN2 |
Abstain/No Voting Instructions |
Abstain % of Outstanding Voting PowerFN2 |
Pass/Fail |
|
45% of votes cast* |
11,517,349 |
43.76% |
14,076,759 |
53.48% |
726,132 |
2.84% |
FAIL (43.76% of Outstanding Voting Power) |
|
55% of votes cast* |
14,076,759 |
53.48% |
11,517,349 |
43.76% |
726,132 |
2.84% |
PASS (53.48% of Outstanding Voting Power) |
|
95% of votes cast* |
24,314,403 |
92.38% |
1,279,705 |
4.86% |
726,132 |
2.84% |
PASS (92.38% of Outstanding Voting Power) |
FN1: Reflects the sum of the votes cast by holders of Pieris common stock FOR the Authorized Shares Increase Proposal, plus the number of votes of the Series F Preferred Stock cast FOR the Authorized Shares Increase Proposal. The number of votes of the Series F Preferred Stock cast FOR the Authorized Shares Increase Proposal is equal to the product of (a) 25,000,000 multiplied by (b) a fraction, whereby (x) the numerator is the total number of common stock votes FOR the Authorized Shares Increase Proposal, and (y) the denominator is the sum of (i) the total number of common stock votes FOR the Authorized Shares Increase Proposal, plus (ii) the total number of common stock votes AGAINST the Authorized Shares Increase Proposal.
FN2: There are 26,320,240 votes comprising total outstanding voting power, including 1,320,240 votes of Pieris common stock and 25,000,000 votes of Series F Preferred Stock.
FN3: Reflects the sum of the votes cast by holders of Pieris common stock AGAINST the Authorized Shares Increase Proposal, plus the number of votes of the Series F Preferred Stock cast AGAINST the Authorized Shares Increase Proposal. The number of votes of the Series F Preferred Stock cast AGAINST the Authorized Shares Increase Proposal is equal to the product of (a) 25,000,000 multiplied by (b) a fraction, whereby (x) the numerator is the total number of common stock votes AGAINST the Authorized Shares Increase Proposal, and (y) the denominator is the sum of (i) the total number of common stock votes FOR the Authorized Shares Increase Proposal, plus (ii) the total number of common stock votes AGAINST the Authorized Shares Increase Proposal.
Q: What will Pieris Stockholders, Pieris Option Holders and Pieris Warrant Holders receive in the Merger?
A: At the Effective Time, Pieris stockholders will continue to own and hold their existing shares of Pieris common stock and Pieris preferred stock, as applicable.
All options to purchase Pieris common stock that are issued and outstanding at the Effective Time will remain in effect pursuant to their terms and will be unaffected by the Merger.
The terms governing any Pieris warrants outstanding as of immediately prior to the Effective Time will remain in full force and effect following the Closing of the Merger.
In addition, at or prior to the effective time of the Merger, Pieris, a rights agent and a CVR holder representative will enter into a Contingent Value Rights Agreement, or CVR Agreement, pursuant to which Pieris stockholders as of the close of business on the last business day prior to the day on which the Effective Time occurs will receive a dividend of one contingent value right, or CVR, for each outstanding share of Pieris common stock held by such stockholder, or each share of common stock underlying Pieris preferred stock outstanding and entitled to receive such dividend in accordance with the terms of such Pieris preferred stock held by such stockholder, as applicable, on such date. Furthermore, to the extent a holder of a Pieris warrant outstanding as of the date of the CVR Agreement exercises such Pieris warrant after the date of the CVR Agreement, a number of CVRs equal to the number of shares of Pieris common stock issued to such holder in connection with such exercise shall be issued to such holder, subject to and in accordance with the terms and conditions of such Pieris Warrant, as applicable.
Each CVR will represent the contractual right to receive cash payments (net of certain expenses and taxes) upon the receipt by Pieris or any of its affiliates of certain payments under strategic partner agreements, including existing collaboration agreements pursuant to which Pieris may be entitled to milestones and royalties in the future, and other out-licensing agreements for certain of Pieris’ legacy assets, and upon the receipt of certain research and development tax credits in favor of Pieris or any of its affiliates, if any, in each case as set forth in, and subject to and in accordance with the terms and conditions of, the CVR Agreement. Pursuant to the terms of the CVR Agreement, holders of CVRs would be entitled to cash payments (net of certain expenses and taxes) upon the receipt by Pieris or any of its affiliates of certain payments under any license, collaboration or similar agreement involving any legacy assets of Pieris entered into prior to the effective date of the CVR Agreement. Pieris retains the ability under the Merger Agreement to seek to enter into such agreements related to its legacy assets and intellectual property, including PRS-400 and PRS-220, though currently does not have any such agreements in place and does not have any plans to divest any material assets. Pieris continues to explore potential transactions for PRS-400 and PRS-220. Management has determined that there is no value associated with the CVRs as the likelihood of any payments received in connection with Pieris’ legacy assets is remote. Unless earlier terminated by the representative of the CVR holders and the combined company, the CVR Agreement will terminate on the date on which no strategic partner agreement is in effect and no payments are payable to Pieris or any of its affiliates under any Strategic Partner Agreement.
The contingent payments in respect of the CVRs, if they become payable, will become payable to a rights agent for subsequent distribution to the holders of the CVRs. In the event that no such proceeds are received, holders of the CVRs will not receive any payment pursuant to the CVR Agreement. There can be no assurance that holders of CVRs will receive any amounts with respect thereto. The right to the contingent payments contemplated by the CVR Agreement is a contractual right only and will not be transferable, except in the limited circumstances specified in the CVR Agreement. The CVRs will not be evidenced by a certificate or any other instrument and will not be registered with the SEC. The CVRs will not have any voting or dividend rights and will not represent any equity or ownership interest in Pieris or any of its affiliates. No interest will accrue on any amounts payable in respect of the CVRs.
The foregoing description of the CVR Agreement does not purport to be complete and is qualified in its entirety by the full text of the form of CVR Agreement, which is included in Annex D to this proxy statement/prospectus.
Q: What will Palvella stockholders and option holders receive in the Merger?
A: Palvella stockholders will receive shares of Pieris common stock, and Palvella option holders will receive options to purchase Pieris common stock. Applying the exchange ratio, immediately after the Merger, but without giving effect to the PIPE Financing, Pieris securityholders as of immediately prior to the Merger are expected to own approximately 18% of the outstanding shares of capital stock of the combined company and former Palvella securityholders, are expected to own approximately 82% of the outstanding shares of capital stock of the combined company, in each case, on a fully-diluted basis, calculated using the treasury stock method, subject to certain assumptions, including, but not limited to, a valuation for Pieris equal to $21 million, provided, that (a) if Pieris’ net cash as of the closing is greater than $11 million, then Pieris’ valuation will be adjusted upwards on a dollar-for-dollar basis by the difference of: (i) Pieris’ net cash, minus (ii) $11 million, and (b) if Pieris’ net cash is less than $11 million, then Pieris’ valuation will be adjusted downwards on a dollar-for-dollar basis by the difference of: (i) $11 million, minus (ii) Pieris’ net cash, as further described in the Merger Agreement.
The PIPE Investors, have agreed to subscribe for and purchase (in cash or by cancellation of convertible notes) an aggregate of approximately 3,154,241 of shares of Pieris common stock, at the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, and/or in lieu of Pieris common stock to certain purchasers who so choose, Pre-Funded Warrants to purchase up to 2,592,585 shares of the combined company common stock, at a purchase price per Pre-Funded Warrant equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, minus $0.001, referred to as the “PIPE Financing”, which is expected to close immediately following the closing of the Merger. The gross proceeds from the PIPE Financing are expected to be approximately $78.9 million, consisting of approximately $60.0 million in cash and the conversion of approximately $18.9 million of principal and interest payable under the outstanding convertible notes issued by Palvella, before paying estimated expenses. The closing of the PIPE Financing is conditioned upon the closing of the Merger as well as certain other conditions.
Additionally, immediately following the PIPE Financing, the Pieris securityholders as of immediately prior to the Merger are expected to hold approximately 10.47% of the shares of combined company capital stock, the Palvella securityholders as of immediately prior to the Merger are expected to hold approximately 50.18% of the shares of combined company capital stock, and the PIPE Investors are expected to hold approximately 39.35% of the shares of combined company capital stock, in each case, on a fully diluted basis, subject to certain assumptions.
In connection with the Merger, each stock option granted under the Palvella Stock Plan, that is outstanding immediately prior to the Effective Time will be assumed by Pieris and will become an option to acquire, on the same terms and conditions as were applicable to such Palvella stock option immediately prior to the Effective Time, a number of shares of Pieris common stock equal to the number of shares of Palvella common stock subject to the unexercised portion of the Palvella stock option immediately prior to the Effective Time, multiplied by the exchange ratio (rounded down to the nearest whole share number), with an exercise price per share for the options equal to the exercise price per share of such Palvella stock option immediately prior to the Effective Time divided by the exchange ratio (rounded up to the nearest whole cent). Such assumed options will continue to be governed by the terms and conditions of the Palvella Stock Plan.
For a more complete description of what Palvella stockholders and option holders will receive in the Merger, please see the sections titled “The Merger—Merger Consideration” and “The Merger—Exchange Ratio” beginning on pages 151 and 152, respectively, of this proxy statement/prospectus. For a description of the effect of the PIPE Financing on Pieris’ and Palvella’s current securityholders, please see the section titled “Agreements Related to the Merger—Securities Purchase Agreement” beginning on page 180 of this proxy statement/prospectus.
Q: Will the common stock of the combined company trade on an exchange?
A: Shares of Pieris common stock are currently listed on The Nasdaq Capital Market under the symbol “PIRS.” On , 2024, the last trading day before the date of this proxy statement/prospectus, the closing sale price of Pieris common stock was $ per share. Palvella intends to file a listing application for the combined company with Nasdaq. After completion of the Merger, the combined company will be renamed “Palvella Therapeutics, Inc.” and, assuming approval of the application for continued listing, the common stock of the combined company will trade on The Nasdaq Capital Market under the symbol “PVLA.” However, Nasdaq’s determination may not be known at the time stockholders are asked to vote on the Merger. Pieris has agreed to cause the shares of Pieris common stock to be issued in connection with the Merger to be approved for listing on Nasdaq at or prior to the Effective Time. In addition, under the Merger Agreement, each of Pieris’ and Palvella’s obligation to complete the Merger is subject to the satisfaction or waiver by each of the parties, at or prior to the Merger, of various conditions, including that the shares of Pieris common stock to be issued in the Merger have been approved for listing on Nasdaq, subject to notice of issuance, as of the closing of the Merger. The terms of the Merger Agreement permit that this condition may be waived by agreement between Pieris and Palvella, without recirculation or re-solicitation of this proxy statement/prospectus.
Q: Who will be the directors of the combined company following the Merger?
A: Immediately following the Merger, the combined company’s board of directors will be composed of five members, consisting of (i) one director appointed by Pieris, namely Christopher Kiritsy, a current member of the Pieris board of directors and (ii) four directors appointed by Palvella, namely Wesley H. Kaupinen, Todd C. Davis, George M. Jenkins and Tadd S. Wessel.
Q: Who will be the executive officers of the combined company immediately following the Merger?
A: Immediately following the Merger, the executive management team of the combined company is expected to consist of members of the Palvella executive management team prior to the Merger, including:
|
Name |
Title |
|
|
Wesley H. Kaupinen |
Chief Executive Officer |
|
|
Matthew Korenberg |
Chief Financial Officer |
|
|
Kathleen Goin |
Chief Operating Officer |
|
|
Jeffrey Martini, Ph.D. |
Chief Scientific Officer | |
| Kathleen A. McGowan | Vice President, Finance and Operations |
Q: As a Pieris stockholder, how does the Pieris board of directors recommend that I vote?
A: After careful consideration, the Pieris board of directors recommends that Pieris stockholders vote “FOR” all of the Proposals.
Q: What risks should I consider in deciding whether to vote in favor of the Merger?
A: You should carefully review the section titled “Risk Factors” beginning on page 25 of this proxy statement/prospectus and the annexes attached hereto and documents incorporated by reference herein, which set forth certain risks and uncertainties related to the Merger, risks and uncertainties to which the combined company’s business will be subject, and risks and uncertainties to which each of Pieris and Palvella, as independent companies, are subject.
Q: When do you expect the Merger to be consummated?
A: The Merger is anticipated to close promptly after the Pieris special meeting scheduled to be held on Wednesday, December 11, 2024, but the exact timing cannot be predicted. For more information, please see the section titled “The Merger Agreement—Conditions to the Completion of the Merger” beginning on page 172 of this proxy statement/prospectus.
Q: What do I need to do now?
A: Pieris urges you to read this proxy statement/prospectus carefully, including the annexes attached hereto and the documents incorporated by reference, and to consider how the Merger affects you.
If you are a Pieris stockholder of record, you may vote or provide your proxy instructions in one of four different ways:
|
● |
you can attend the Pieris special meeting online and vote online during the Pieris special meeting; |
|
● |
you can mail your signed proxy card in the enclosed return envelope; |
|
● |
you can provide your proxy instructions via telephone by following the instructions on your proxy card; or |
|
● |
you can provide your proxy instructions via the internet by following the instructions on your proxy card. |
Your signed proxy card, telephonic proxy instructions, or internet proxy instructions must be received by December 10, 2024 at 11:59 p.m. Eastern Time to be counted.
If you hold your shares in “street name” (as described below), you may provide your proxy instructions via telephone or the internet by following the instructions on your vote instruction form. Please provide your proxy instructions only once, unless you are revoking a previously delivered proxy instruction, and as soon as possible so that your shares can be voted at the Pieris special meeting.
Q: What happens if I do not return a proxy card or otherwise vote or provide proxy instructions, as applicable?
A: If you are a Pieris stockholder, the failure to return your proxy card or otherwise vote or provide proxy instructions will reduce the aggregate number of votes required to approve Proposal Nos. 1, 3, 4 and 5. Also, your shares will not be counted for purposes of determining whether a quorum is present at the Pieris special meeting unless your broker has, and exercises, discretionary authority to vote on certain matters.
Q: May I attend the Pieris special meeting and vote in person?
A: The Pieris special meeting will be held entirely online. Stockholders of record as of the close of business on October 28, 2024 will be able to attend and participate in the Pieris special meeting online by accessing www.virtualshareholdermeeting.com/PIRS2024SM. To join the Pieris special meeting, you will need to have your 16-digit control number which is included on your Notice of Internet Availability of Proxy Materials and your proxy card. If your shares are held in “street name,” you should contact your bank, broker or other nominee to obtain your 16-digit control number or otherwise vote through your bank, broker or other nominee.
Q: Who counts the votes?
A: , or , will be engaged as Pieris’ independent agent to tabulate stockholder votes, which Pieris refers to as the inspector of election. If you are a stockholder of record of Pieris common stock or preferred stock, your executed proxy card is returned directly to for tabulation.
Q: If my shares of Pieris common stock are held in “street name” by my broker, will my broker vote my shares for me?
A: Unless your broker has discretionary authority to vote on certain matters, your broker will not be able to vote your shares of Pieris common stock on matters requiring discretionary authority without instructions from you. If you do not give instructions to your broker, your broker can vote your Pieris shares with respect to “discretionary,” routine items but not with respect to “non-discretionary,” non-routine items. Discretionary items are proposals considered routine under Rule 452 of the New York Stock Exchange on which your broker may vote shares held in “street name” in the absence of your voting instructions. With respect to non-routine items for which you do not give your broker instructions, your Pieris shares will be treated as broker non-votes. Proposal Nos. 1, 2 and 3 at the Pieris special meeting will be non-routine. It is anticipated that Proposal Nos. 4 and 5 will be routine. To make sure that your vote is counted, you should instruct your broker to vote your shares, following the procedures provided by your broker.
Q: What are broker non-votes and do they count for determining a quorum?
A: Generally, broker non-votes occur when there is at least one discretionary and one non-discretionary proposal to be voted on at the meeting and shares held by a broker in “street name” for a beneficial owner are voted on at least one “routine” proposal but not voted with respect to a particular proposal because the broker (i) has not received voting instructions from the beneficial owner for that proposal or (ii) lacks discretionary voting power to vote those shares for that proposal. A broker is entitled to vote shares held for a beneficial owner on routine matters without instructions from the beneficial owner of those shares. On the other hand, absent instructions from the beneficial owner of such shares, a broker is not entitled to vote shares held for a beneficial owner on non-routine matters.
Broker non-votes, if any, will be treated as shares present for the purpose of determining the presence of a quorum for the transaction of business at the Pieris special meeting. Broker non-votes will not be treated as votes cast for or against non-routine proposals. It is anticipated that Proposal Nos. 1, 2 and 3 will be non-routine. It is anticipated that Proposal Nos. 4 and 5 will be routine. Broker non-votes, if any, will not have any effect with respect to the outcome of Proposal Nos. 1, 3, 4 and 5, but will have the effect of a vote AGAINST Proposal No. 2.
Q: May I change my vote after I have submitted a proxy or provided proxy instructions?
A: Pieris stockholders of record, unless such stockholder’s vote is subject to a support agreement, may change their vote at any time before their proxy is voted at the Pieris special meeting in one of four ways:
|
● |
you may submit another properly completed proxy with a later date by mail or via the internet; |
|
● |
you can provide your proxy instructions via telephone at a later date; |
|
● |
you may send a written notice that you are revoking your proxy to Pieris’ Corporate Secretary at 225 Franklin St., 26th Floor, Boston MA 02110, Attn: Pieris Pharmaceuticals, Inc., Corporate Secretary; or |
|
● |
you may attend the Pieris special meeting online and vote by following the instructions at www.virtualshareholdermeeting.com/PIRS2024SM. Simply attending the Pieris special meeting will not, by itself, revoke your proxy. |
Your signed proxy card, telephonic proxy instructions, internet proxy instructions, or written notice must be received by December 10, 2024 at 11:59 p.m. Eastern Time to be counted.
If a Pieris stockholder who owns Pieris shares in “street name” has instructed a broker to vote its shares of Pieris common stock, the stockholder must follow directions received from its broker to change those instructions.
Q: Who is paying for this proxy solicitation?
A: Pieris and Palvella will share equally the cost of printing and filing of this proxy statement/prospectus and the proxy card. Arrangements will also be made with brokerage firms and other custodians, nominees and fiduciaries who are record holders of Pieris common stock for the forwarding of solicitation materials to the beneficial owners of Pieris common stock. Pieris will reimburse these brokers, custodians, nominees and fiduciaries for the reasonable out-of-pocket expenses they incur in connection with the forwarding of solicitation materials. Pieris will retain Alliance Advisors to assist it in soliciting proxies using the means referred to above. Pieris will pay the fees of Alliance Advisors, which Pieris expects to be approximately $50,000, plus reimbursement of out-of-pocket expenses.
Q: What are the material U.S. federal income tax consequences of the Merger to Pieris stockholders?
A: Pieris stockholders will not sell, exchange or dispose of any shares of Pieris common stock in the Merger. Thus, there will be no material U.S. federal income tax consequences to Pieris stockholders upon consummation of the Merger.
Q: What are the material U.S. federal income tax consequences of the Merger to Palvella U.S. holders?
A: Subject to the limitations and qualifications described in the section titled “The Merger—Material U.S. Federal Income Tax Consequences of the Merger,” in the opinion of Mintz, Levin, Cohn, Ferris, Glovsky and Popeo, P.C., or “Mintz”, and Troutman Pepper Hamilton Sanders LLP, or “Troutman”, the Merger will qualify as a “reorganization” within the meaning of Section 368(a) of the Internal Revenue Code of 1986, as amended, or the “Code”, and a Palvella U.S. holder will not recognize gain or loss for U.S. federal income tax purposes upon the receipt of shares of Pieris common stock in exchange for shares of Palvella capital stock in the Merger, except with respect to cash received in lieu of a fractional share of Pieris common stock. For a more detailed discussion of the material U.S. federal income tax consequences of the Merger, see “The Merger—Material U.S. Federal Income Tax Consequences of the Merger” beginning on page 155 of this proxy statement/prospectus.
Q: What are the material U.S. federal income tax consequences of the receipt of contingent value rights to Pieris stockholders?
A: The U.S. federal income tax treatment of Pieris stockholders’ receipt of the CVRs is unclear. Pieris intends to take the position that the fair market value of the CVRs cannot be reasonably ascertained on the date of the issuance of the CVRs and, accordingly, the issuance of the CVRs constitutes an “open transaction.” Accordingly, absent a change in law requiring otherwise after the date of the CVR Agreement, Pieris will not report the issuance of the CVRs to Pieris stockholders as a current distribution and will instead report each future payment (if any) on the CVRs as a distribution by Pieris for U.S. federal income tax purposes, with each such payment being reported as a dividend to the extent of Pieris’s current or accumulated earnings and profits in the year in which such payment is made. This position may be challenged by the Internal Revenue Service, or IRS, in which case holders of Pieris common stock could be required to recognize taxable income in respect of the CVRs without the corresponding receipt of cash. See the section titled “Agreements Related to the Merger—CVR Agreement—Material U.S. Federal Income Tax Consequences of the Receipt of CVRs” beginning on page 182 of this proxy statement/prospectus for a more complete description of the material U.S. federal income tax consequences of the receipt of CVRs to Pieris stockholders, including possible alternative treatments.
The tax consequences to you of the receipt of CVRs are subject to substantial uncertainty and may also depend on your particular facts and circumstances. Please consult your tax advisors as to the specific tax consequences to you.
Q: Who can help answer my questions?
A: If you are a Pieris stockholder and would like additional copies of this proxy statement/prospectus without charge, you may (1) visit www.ProxyVote.com, (2) call 1-800-579-1639 or (3) send an email to sendmaterial@proxyvote.com.
If you are a Pieris stockholder and have questions about the Merger, including the procedures for voting your shares, you should contact:
Alliance Advisors LLC
200 Broadacres Drive, 3rd Floor
Bloomfield, New Jersey 07003
833-782-7190 (toll free)
PIRS@allianceadvisors.com (Email)
This summary highlights selected information from this proxy statement/prospectus and may not contain all of the information that is important to you. To better understand the Merger and the proposals being considered at the Pieris special meeting, you should read this entire proxy statement/prospectus carefully, including the Merger Agreement and the other annexes to which you are referred in this proxy statement/prospectus and the documents incorporated by reference herein. For more information, please see the section titled “Where You Can Find More Information” beginning on page 336 of this proxy statement/prospectus.
Pieris Pharmaceuticals, Inc.
225 Franklin Street, 26th Floor
Boston, Massachusetts 02110
(857) 246-8998
Pieris is a biopharmaceutical company that discovered and developed Anticalin-based drugs to target validated disease pathways in unique and transformative ways. Pieris’ clinical pipeline consists of immuno-oncology, or “IO”, programs partnered with several major multi-national pharmaceutical companies.
On July 18, 2023, Pieris announced its intention to explore engaging in one or more strategic transactions, including mergers, reverse mergers, acquisitions, other business combinations or sales of assets, or other strategic transactions. This decision was related to events that impacted Pieris’ inhaled respiratory franchise in connection with AstraZeneca's discontinuation of the Phase 2a study for elarekibep, formerly PRS-060, an inhaled IL-4Rα antagonist Anticalin protein to treat uncontrolled asthma.
Also on July 18, 2023, the Pieris board of directors approved a reduction in Pieris’ workforce by approximately 70%. Since July of 2023, and through June 30, 2024, Pieris took additional steps to reduce its operating footprint including terminating its remaining lease obligations in Germany and winding down its proprietary inhaled respiratory programs. Pieris also has opted out and terminated programs where possible to reduce operating costs. Further reductions in the workforce have occurred based upon these actions. As a result, Pieris recognized approximately $7.5 million of severance costs and other related termination benefits in 2023 as the service period to earn such benefits was considered complete. Approximately $2.4 million of the termination benefits were paid in 2023. Pieris expects approximately $4.3 million of the termination benefits to be paid through the end of 2024, with the remainder of termination benefits to be paid in 2025.
On March 27, 2024, Pieris announced the implementation of a new strategy along with relevant cost-saving measures that are expected to extend its cash runway into at least 2027, while maximizing its ability to capture the potential milestones from its partnered 4-1BB bispecific Mabcalin® protein IO assets. Pieris may be entitled to aggregate milestones of up to approximately $15.0 million upon first patient dosed in the Phase 2 trials for SGN-BB228 and BOS-342, which are both currently in Phase 1 clinical development, and up to approximately $40.0 million upon first patient dosed in pivotal clinical trials for SGN-BB228 and BOS-342. To support this new strategy, Pieris discontinued all of its research and development, efforts, and implemented a workforce reduction that impacted additional employees and the executive leadership team.
On July 23, 2024, Pieris, Merger Sub and Palvella entered into the Merger Agreement, pursuant to which, among other things, and subject to the satisfaction or waiver of the conditions set forth in the Merger Agreement, Merger Sub will merge with and into Palvella, with Palvella continuing as our wholly owned subsidiary and the surviving corporation of the Merger. If the Merger is completed, the business of Palvella will continue as the business of the combined company, which will focus on developing Palvella’s product candidates, and it is anticipated that the combined company will not continue to develop any of Pieris’ legacy product candidates.
Palvella Therapeutics, Inc.
125 Strafford Avenue, Suite 360
Wayne, Pennsylvania 19087
Telephone: (484) 253-1461
Palvella is a clinical-stage biopharmaceutical company whose vision is to become the leading rare disease biopharmaceutical company focused on developing and, if approved, commercializing novel therapies to treat patients suffering from serious, rare genetic skin diseases, for which there are no FDA approved therapies. Palvella intends to leverage its versatile QTORIN™ platform to treat these patients. The QTORIN platform is designed to generate new therapies that penetrate the deep layers of the skin to locally treat a broad spectrum of rare, genetic skin diseases. Palvella’s lead product candidate, QTORIN 3.9% rapamycin anhydrous topical gel, or “QTORIN rapamycin”, is in clinical development for two of these diseases: microcystic lymphatic malformations, or “microcystic LM”, and cutaneous venous malformations. QTORIN rapamycin contains the active pharmaceutical ingredient rapamycin, also known as sirolimus, which is an inhibitor of mammalian target of rapamycin, or “mTOR”, a kinase that plays a key role in cell growth and proliferation. The QTORIN platform is novel and has only generated one program to date, QTORIN rapamycin, and clinical evidence to support this candidate is preliminary and limited at this time.
Palvella currently has one ongoing clinical trial and one clinical trial planned to start in the fourth quarter of 2024, both of which are currently or will be conducted in the United States. Palvella’s ongoing trial, SELVA, is a Phase 3 Baseline-Controlled Study Evaluating the Safety and Efficacy of QTORIN rapamycin in the Treatment of Microcystic LM. Palvella previously announced topline Phase 2 clinical trial results from the multi-center, open-label study of 12 subjects receiving QTORIN™ rapamycin once-daily for 12-weeks. The Phase 2 clinical trial featured multiple pre-specified efficacy assessments, including clinician and patient global impression assessments as well as assessments of individual clinical manifestations that are important disease burdens for individuals living with microcystic LMs. All participants in the Phase 2 clinical trial demonstrated improvements on the Clinician Global Impression of Change scale, with all participants in the study rated as either "Much Improved" (n=7, 58%) or "Very Much Improved" (n=5, 42%) after 12-weeks of treatment compared to the pre-treatment baseline period. Palvella expects to report top-line data for the Phase 3 study in approximately 40 participants with microcystic LM in the first quarter of 2026.
A baseline-controlled study is a clinical study in which the patient's condition during treatment is compared with their condition before treatment. In such studies, participants serve as their own control. In a placebo-controlled study, patients are randomized prior to treatment to receive either study drug or matching placebo and to determine how the efficacy of the treatment compares to placebo. Baseline-controlled studies are appropriate when the effects are dramatic, occur rapidly following treatment, and are unlikely to have occurred spontaneously (e.g., general anesthesia, cardioversion, measurable tumor shrinkage).
Microcystic LM is a serious, chronically debilitating, and lifelong disease of the lymphatic system characterized by lymphorrhea and acute cellulitis. It is estimated that there are more than 30,000 diagnosed patients in the United States with microcystic LM. The specific pathophysiology of microcystic LM is the result of somatic activating mutations in primarily Phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha, or “PIK3CA” which result in increased activation of the PI3K/mTOR pathway and subsequent lymphatic hyperplasia.
Palvella has received Breakthrough Therapy Designation, Fast Track Designation, and Orphan Drug Designation from the FDA for QTORIN rapamycin for the treatment of microcystic LM. Palvella has also received Fast Track Designation from the FDA for the treatment of venous malformations.
There are no FDA-approved therapies currently indicated for either microcystic LM or cutaneous venous malformations. If approved for the treatment of microcystic LM or cutaneous venous malformations, Palvella believes QTORIN rapamycin has the potential to become the standard of care for these diseases.
Palvella also has a planned study for cutaneous venous malformations, a Phase 2 Baseline-Controlled Study Evaluating the Safety and Efficacy of QTORIN rapamycin for the Treatment of Cutaneous Venous Malformations expected to start in the fourth quarter of 2024. Cutaneous venous malformations are a serious disease with a high unmet need characterized by dysregulated growth of malformed veins impacting the skin, causing functional impairment and deformity. It is estimated that there are more than 75,000 patients in the United States with cutaneous venous malformations. This Phase 2 baseline-controlled clinical trial is expected to be in approximately 15 participants in this patient population and Palvella expects to report top-line data in the fourth quarter of 2025. The trial is covered by the same Investigational New Drug Application, or “IND,” submitted to the FDA by Palvella for QTORIN rapamycin.
Palvella also has additional preclinical research programs based on Palvella’s QTORIN platform for the treatment of serious, rare genetic skin diseases for which Palvella believes there are significant unmet needs. As Palvella plans to expand its pipeline into new rare skin diseases, it plans to generate new product candidates with its QTORIN platform. Despite Palvella’s intentions with respect to its QTORIN platform, Palvella’s business carries substantial risks. The QTORIN platform is novel and has only generated one program to date, QTORIN rapamycin, and clinical evidence to support this candidate is preliminary and limited at this time.
Palvella currently plans to pursue marketing approval for QTORIN rapamycin for several indications in the U.S. through a Section 505(b)(2) NDA and will be relying on the listed drug. RAPAMUNE, a previously approved drug for organ rejection prophylactic. A Section 505(b)(2) NDA enables the applicant in certain circumstances to rely, in part, on the FDA’s prior findings in approving a similar product or published literature in support of its application. Section 505(b)(2) permits the submission of an NDA where at least some of the information required for approval comes from studies not conducted by, or for, the applicant and for which the applicant has not obtained a right of reference. If the Section 505(b)(2) applicant can establish that reliance on the FDA’s prior findings of safety and/or effectiveness is scientifically appropriate, it may eliminate the need to conduct certain preclinical or clinical studies of the new product. The FDA may also require companies to perform additional studies or measurements to support the change from the approved product. The FDA may then approve the new product candidate for all, or some, of the indications for which the listed product has been approved, as well as for any new indication sought by the Section 505(b)(2) applicant. There can be no assurance that the FDA will agree with Palvella’s use of the Section 505(b)(2) NDA pathway to seek approval for its product candidates in various indications.
Additionally, a number of factors could delay or prevent regulatory approval for Palvella’s product candidates, including, but not limited to, the need to conduct additional trials, the acceptability of the clinical evidence presented to regulatory agencies such as the baseline-controlled Phase 3 study, issues with our QTORIN platform which has not yet been approved in any product, and the efficacy endpoints utilized in the clinical studies. See the sections entitled “Palvella’s Business” and “Risk Factors––Risks Related to Palvella––Risks Related to the Discovery, Development, Regulatory Approval and Commercialization of Palvella’s Product Candidates” in this proxy statement/prospectus for more information.
As a novel platform, Palvella’s QTORIN platform may never result in a product candidate that receives regulatory approval. Negative results in the development of QTORIN rapamycin for either the treatment of microcystic LM or cutaneous venous malformations may also impact Palvella’s ability to obtain regulatory approval for other product candidates which Palvella expects to develop based on its QTORIN platform. “Risk Factors––Risks Related to Palvella––Risks Related to the Discovery, Development, Regulatory Approval and Commercialization of Palvella’s Product Candidates” in this proxy statement/prospectus for more information.
Polo Merger Sub, Inc.
c/o Pieris Pharmaceuticals, Inc.
225 Franklin Street, 26th Floor
Boston, Massachusetts 02110
(857) 246-8998
Merger Sub is a wholly-owned subsidiary of Pieris and was formed solely for the purpose of carrying out the Merger.
If the Merger is completed, Merger Sub will merge with and into Palvella, with Palvella surviving the Merger as a wholly-owned subsidiary of Pieris.
Subject to the terms and conditions of the Merger Agreement, at the closing of the Merger, each then outstanding share of Palvella capital stock will be converted into the right to receive a number of shares of Pieris common stock (subject to the payment of cash in lieu of fractional shares) calculated in accordance with the exchange ratio set forth in the Merger Agreement. Based on Pieris’ and Palvella’s capitalization as of July 23, 2024, the date the Merger Agreement was executed, the exchange ratio is estimated to be equal to approximately 0.315478222 shares of Pieris common stock for each share of Palvella capital stock. The final exchange ratio is subject to adjustment prior to closing of the Merger based upon Pieris’ net cash as of the date immediately preceding the closing of the Merger.
Additionally, subject to the terms and conditions of the Merger Agreement, at the Effective Time, each stock option granted under the Palvella Stock Plan that is outstanding immediately prior to the Effective Time will be assumed by Pieris and will become an option to acquire, on the same terms and conditions as were applicable to such Palvella stock option immediately prior to the Effective Time, a number of shares of Pieris common stock equal to the number of shares of Palvella common stock subject to the unexercised portion of the Palvella stock option immediately prior to the Effective Time, multiplied by the exchange ratio (rounded down to the nearest whole share number), with an exercise price per share for the stock options equal to the exercise price per share of such Palvella stock option immediately prior to the Effective Time divided by the exchange ratio (rounded up to the nearest whole cent). Such assumed stock options will continue to be governed by the terms and conditions of the Palvella Stock Plan.
Under the exchange ratio formula in the Merger Agreement, upon the closing of the Merger, but without giving effect to the PIPE Financing, on a pro forma basis and based upon the number of shares of Pieris common stock expected to be issued in the Merger, Pieris securityholders as of immediately prior to the Merger are expected to own approximately 18% of the outstanding shares of capital stock of the combined company, and former Palvella stockholders are expected to own approximately 82% of the outstanding shares of capital stock of the combined company, in each case, on a fully-diluted basis, calculated using the treasury stock method, subject to certain assumptions, including, but not limited to, a valuation for Pieris equal to $21 million, provided, that (a) if Pieris’ net cash as of the closing is greater than $11 million, then Pieris’ valuation will be adjusted upwards on a dollar-for-dollar basis by the difference of: (i) Pieris’ net cash, minus (ii) $11 million, and (b) if Pieris’ net cash is less than $11 million, then Pieris’ valuation will be adjusted downwards on a dollar-for-dollar basis by the difference of: (i) $11 million, minus (ii) Pieris’ net cash, as further described in the Merger Agreement. For purposes of calculating the exchange ratio, shares of Pieris common stock underlying Pieris stock options outstanding as of immediately prior to the closing of the Merger with an exercise price of less than the volume weighted average closing trading price of a share of Pieris common stock on The Nasdaq Capital Market for the five consecutive trading days ending five trading days immediately prior to the closing of the Merger will be deemed to be outstanding and shares of Palvella common stock underlying Palvella stock options, warrants and other derivative securities outstanding as of immediately prior to the closing of the Merger with an exercise price of less than the volume weighted average closing trading price of a share of Pieris common stock on The Nasdaq Capital Market for the five consecutive trading days ending five trading days immediately prior to the closing of the Merger will be deemed to be outstanding, and, in each case, such shares will be calculated using the treasury stock method. The provisions for calculating the exchange ratio assume a valuation for Palvella equal to $95 million and a valuation for Pieris equal to $21 million, in each case as further described in the Merger Agreement.
Each option to purchase shares of Pieris common stock that is outstanding immediately prior to the Effective Time, whether vested or unvested, will survive the closing and remain outstanding in accordance with its terms.
For a more complete description of the Merger and the exchange ratio please see the section titled “The Merger” beginning on page 122 in this proxy statement/prospectus.
The Merger will be completed as promptly as practicable after all of the conditions to completion of the Merger are satisfied or waived, including the adoption of the Merger Agreement by the Palvella stockholders and the approval by the Pieris stockholders of the issuance of Pieris common stock pursuant to the terms of the Merger Agreement. Pieris and Palvella are working to complete the Merger as quickly as practicable. The Merger is anticipated to close promptly after the Pieris special meeting scheduled to be held on Wednesday, December 11, 2024 at 9:00 a.m. Eastern Time. However, Pieris and Palvella cannot predict the exact timing of the completion of the Merger because it is subject to the satisfaction of various conditions. After completion of the Merger, assuming that Pieris receives the required stockholder approval, Pieris will be renamed “Palvella Therapeutics, Inc.”
In connection with the foregoing and concurrently with the execution of the Merger Agreement, Pieris entered into the Purchase Agreement with the PIPE Investors, pursuant to which the PIPE Investors have agreed to subscribe for and purchase (in cash or by cancellation of convertible notes), and Pieris has agreed to issue and sell to the PIPE Investors, an aggregate of approximately 3,154,241 of shares of Pieris common stock at a price per share equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, and/or in lieu of Pieris common stock to certain purchasers who so choose, Pre-Funded Warrants to purchase up to 2,592,585 shares of the combined company common stock at a purchase price per Pre-Funded Warrant equal to the Purchase Price minus $0.001. The gross proceeds from the PIPE Financing are expected to be approximately $78.9 million, consisting of approximately $60.0 million in cash and the conversion of approximately $18.9 million of principal and interest payable under the outstanding convertible notes issued by Palvella, before paying estimated expenses. The closing of the PIPE Financing is conditioned upon the closing of the Merger as well as certain other conditions.
Additionally, immediately following the PIPE Financing, the Pieris securityholders as of immediately prior to the Merger are expected to hold approximately 10.47% of the shares of combined company capital stock, the Palvella securityholders as of immediately prior to the Merger are expected to hold approximately 50.18% of the shares of combined company capital stock, and the PIPE Investors are expected to hold approximately 39.35% of the shares of combined company capital stock, in each case, on a fully diluted basis, subject to certain assumptions.
The Pieris common stock and the Pre-Funded Warrants to be issued pursuant to the Purchase Agreement will not be registered under the Securities Act of 1933, as amended, or the “Securities Act”, in reliance upon certain exemptions under the Securities Act. At or prior to the closing of the PIPE Financing, Pieris will enter into a registration rights agreement, or the “Registration Rights Agreement” with the PIPE Investors pursuant to which the PIPE Investors will be entitled to certain resale registration rights with respect to shares of Pieris common stock issued to the PIPE Investors and any shares of Pieris common stock issued upon exercise of the Pre-Funded Warrants. Pursuant to the Registration Rights Agreement, Pieris will be required to prepare and file a resale registration statement on Form S-1 with the SEC within 30 days following the closing of the PIPE Financing. Pieris shall use its commercially reasonable efforts to cause such resale registration statement to be declared effective by the SEC within 90 days following the closing of the PIPE Financing (or within 120 days following the PIPE Financing if the SEC reviews such resale registration statement). The PIPE Financing is contingent upon, among other things, the closing of the Merger.
During the course of its evaluation of the Merger Agreement and the transactions contemplated by the Merger Agreement, the Pieris board of directors held numerous meetings, consulted with Pieris’ senior management, legal counsel and financial advisor, and reviewed and assessed a significant amount of information. In reaching its decision to approve the Merger Agreement and the transactions contemplated by the Merger Agreement, the Pieris board of directors considered numerous factors that it viewed as supporting its decision to approve the Merger Agreement. Several factors considered by the Pieris board of directors included:
|
● |
the financial condition and prospects of Pieris and the risks associated with continuing to operate Pieris on a stand-alone basis, particularly in light of the decision to winddown its proprietary preclinical and clinical programs, including cinrebafusp alfa, PRS-220, and PRS-400, and reduce its workforce, and the termination of various partnerships, including its partnerships with AstraZeneca AB, or “AstraZeneca”, in October 2023 and Les Laboratoires Servier and Institut de Recherches Internationales Servier, or “Servier”, which Pieris was notified would be terminated in December 2024; |
|
● |
that the Pieris board of directors, with the assistance of Pieris’ financial advisor, undertook a comprehensive and thorough process of reviewing and evaluating multiple potential strategic alternatives and merger partner candidates to identify the opportunity that would, in the view of the Pieris board of directors, create the most value for Pieris stockholders; and |
|
● |
the Pieris board of directors’ belief, after a thorough review of strategic alternatives and discussions with Pieris’ senior management, financial advisors and legal counsel, that the Merger is more favorable to Pieris stockholders than the potential value that might have resulted from other strategic alternatives available to Pieris, including continuing to operate Pieris on a stand-alone basis or conducting a dissolution and liquidation of Pieris and distributing any available cash to its stockholders. |
For additional information, please see the section titled “The Merger—Pieris Reasons for the Merger” beginning on page 135 of this proxy statement/prospectus.
Palvella’s Reasons for the Merger
The Palvella board of directors has unanimously approved the Merger Agreement, the Merger and the transactions contemplated thereby. The Palvella board of directors reviewed several factors in reaching its decision and believes that the Merger Agreement, the Merger and the transactions contemplated thereby are advisable and fair to, and in the best interests of Palvella and its stockholders. Several factors considered by the Palvella board of directors included:
|
● |
the Merger will provide Palvella current stockholders with greater liquidity by owning publicly-traded stock; |
|
● |
the potential increased access to sources of capital and a broader range of investors to support the development of its product candidates following consummation of the merger compared to if Palvella continued to operate as a privately held company; |
|
● |
the PIPE Financing will generate capital resources to fund the combined company; and |
|
● |
the Palvella board of directors’ belief that no alternatives to the Merger were reasonably likely to create greater value for Palvella stockholders, after reviewing the various financing and other strategic options to enhance stockholder value that were considered by the Palvella board of directors and the likelihood of achieving any alternative transaction compared to the likelihood of completing the Merger. |
For additional information, please see the section titled “The Merger—Palvella Reasons for the Merger” beginning on page 137 of this proxy statement/prospectus.
Recommendation of the Pieris Board of Directors
|
● |
the Pieris board of directors has determined and believes that it is fair to, in the best interests of, and advisable to, Pieris and its stockholders to approve the issuance of shares of common stock of Pieris pursuant to the terms of the Merger Agreement and the Purchase Agreement (as each may be amended from time to time), a copy of which is attached as Annex A and Annex B and the change of control of Pieris pursuant to the Merger, for purposes of Nasdaq Listing Rules 5635(a), (b) and (d), as described in this proxy statement/prospectus. The Pieris board of directors recommends that Pieris stockholders vote “FOR” Proposal No. 1; |
|
● |
the Pieris board of directors has determined and believes that it is fair to, in the best interests of, and advisable to, Pieris and its stockholders to approve an amendment to the amended and restated articles of incorporation of Pieris to increase the number of authorized shares of Pieris common stock from 3,750,000 shares to 200,000,000 shares. The Pieris board of directors recommends that Pieris stockholders vote “FOR” Proposal No. 2; |
|
● |
the Pieris board of directors has determined and believes that it is fair to, in the best interests of, and advisable to, Pieris and its stockholders to approve the adoption of the Palvella Therapeutics, Inc. 2024 Equity Incentive Plan, as described in this proxy statement/prospectus. The Pieris board of directors recommends that Pieris stockholders vote “FOR” Proposal No. 3; |
|
● |
the Pieris board of directors has determined and believes that it is fair to, in the best interests of, and advisable to, Pieris and its stockholders to ratify the Pieris board of directors’ approval of an amendment to the amended and restated articles of incorporation of Pieris to change the name of the corporation from “Pieris Pharmaceuticals, Inc.” to “Palvella Therapeutics, Inc.” The Pieris board of directors recommends that Pieris stockholders vote “FOR” Proposal No. 4; and |
|
● |
the Pieris board of directors has determined and believes that it is fair to, in the best interests of, and advisable to, Pieris and its stockholders to approve the adjournment of the Pieris special meeting, if necessary, to solicit additional proxies if there are not sufficient votes in favor of Proposal Nos. 1 and 2. The Pieris board of directors recommends that Pieris stockholders vote “FOR” Proposal No. 5. |
Opinion of Pieris’ Financial Advisor
In connection with the Merger, Stifel, Nicolaus & Company, Incorporated, or Stifel, delivered a written opinion, dated July 23, 2024, or the “Stifel Opinion”, to the Pieris board of directors as to the fairness, from a financial point of view and as of the date of such opinion, of the Aggregate Merger Consideration (as defined in the Stifel Opinion) to be paid by Pieris to the holders of Palvella capital stock in the Merger pursuant to the Merger Agreement. The full text of the Stifel Opinion, which describes the procedures followed, assumptions made, matters considered, and qualifications and limitations on the review undertaken by Stifel in connection with the Stifel Opinion, is attached as Annex C to this document.
The Stifel Opinion was for the information of, and was directed to, the Pieris board of directors (in its capacity as such) for its information and assistance in connection with its consideration of the financial terms of the Merger. The Stifel Opinion addressed only the fairness, from a financial point of view, to Pieris of the Aggregate Merger Consideration to be paid by Pieris to the holders of Palvella capital stock in the Merger. It did not address the underlying business decision of the Pieris board of directors or Pieris to proceed with or effect the Merger or constitute a recommendation to the Pieris board of directors in connection with the Merger or any other matter, and it does not constitute a recommendation to any stockholder of Pieris or any stockholder of any other entity as to how to vote in connection with the Merger or as to any other action that a stockholder should take with respect to the Merger.
The full text of the Stifel Opinion should be read carefully in its entirety for a description of the procedures followed, the assumptions made, the matters considered, and the qualifications and limitations upon the review undertaken by Stifel in connection with its Opinion.
Overview of the Merger Agreement and Agreements Related to the Merger Agreement
Merger Consideration
At the Effective Time, upon the terms and subject to the conditions set forth in the Merger Agreement each outstanding share of Palvella capital stock (excluding dissenting shares) will be automatically converted solely into the right to receive a number of shares of Pieris common stock equal to the exchange ratio described in more detail below.
Immediately after the Merger, but without giving effect to the PIPE Financing, Pieris securityholders as of immediately prior to the Merger are expected to own approximately 18% of the outstanding shares of capital stock of the combined company and former Palvella securityholders, are expected to own approximately 82% of the outstanding shares of capital stock of the combined company, in each case, on a fully-diluted basis, calculated using the treasury stock method, subject to certain assumptions, including, but not limited to, a valuation for Pieris equal to $21 million, provided, that (a) if Pieris’ net cash as of the closing is greater than $11 million, then Pieris’ valuation will be adjusted upwards on a dollar-for-dollar basis by the difference of: (i) Pieris’ net cash, minus (ii) $11 million, and (b) if Pieris’ net cash is less than $11 million, then Pieris’ valuation will be adjusted downwards on a dollar-for-dollar basis by the difference of: (i) $11 million, minus (ii) Pieris’ net cash, as further described in the Merger Agreement.
The PIPE Investors have agreed to subscribe for and purchase (in cash or by cancellation of convertible notes) an aggregate of approximately 3,154,241 of shares of Pieris common stock, at a price per share equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, and/or in lieu of Pieris common stock to certain purchasers who so choose, Pre-Funded Warrants to purchase up to 2,592,585 shares of the combined company common stock, at a purchase price per Pre-Funded Warrant equal to the Purchase Price minus $0.001, which is expected to close immediately following the closing of the Merger. The gross proceeds from the PIPE Financing are expected to be approximately $78.9 million, consisting of approximately $60.0 million in cash and the conversion of approximately $18.9 million of principal and interest payable under the outstanding convertible notes issued by Palvella, before paying estimated expenses. The closing of the PIPE Financing is conditioned upon the closing of the Merger, as well as certain other conditions.
Additionally, immediately following the PIPE Financing, the Pieris securityholders as of immediately prior to the Merger are expected to hold approximately 10.47% of the shares of combined company capital stock, the Palvella securityholders as of immediately prior to the Merger are expected to hold approximately 50.18% of the shares of combined company capital stock, and the PIPE Investors are expected to hold approximately 39.35% of the shares of combined company capital stock, in each case, on a fully diluted basis, subject to certain assumptions.
Treatment of Palvella Options
Under the terms of the Merger Agreement, each stock option granted under the Palvella Stock Plan that is outstanding immediately prior to the Effective Time will be assumed by Pieris and will become an option to acquire, on the same terms and conditions as were applicable to such Palvella stock option immediately prior to the Effective Time, a number of shares of Pieris common stock equal to the number of shares of Palvella common stock subject to the unexercised portion of the Palvella stock option immediately prior to the Effective Time, multiplied by the exchange ratio (rounded down to the nearest whole share number), with an exercise price per share for the options equal to the exercise price per share of such Palvella stock option immediately prior to the Effective Time divided by the exchange ratio (rounded up to the nearest whole cent). Such assumed options will continue to be governed by the terms and conditions of the Palvella Stock Plan, including the adjustment and change in control provisions contained therein.
Treatment of Pieris Common Stock and Pieris Options
Each share of Pieris common stock and option to purchase Pieris common stock that is issued and outstanding at the Effective Time will remain issued and outstanding and such shares and options will be unaffected by the Merger.
Conditions to the Completion of the Merger
To complete the Merger, Pieris stockholders must approve Proposal No. 1, Proposal No. 2 and Proposal No. 4 and Palvella stockholders must adopt the Merger Agreement. Additionally, each of the other closing conditions set forth in the Merger Agreement must be satisfied or waived (including the closing of the transactions contemplated by the PIPE Financing).
Non-Solicitation
The Merger Agreement contains “non-solicitation” provisions, pursuant to which, subject to specified exceptions, each of Pieris and Palvella has agreed that neither it nor its subsidiaries will, and each of Pieris and Palvella will cause its respective directors, officers, employees, and consultants not to, and will instruct their respective attorneys, and financial advisors not to, directly or indirectly:
|
● |
solicit, seek or initiate or knowingly take any action to facilitate or encourage any offers, inquiries or the making of any proposal or offer that constitutes, or would reasonably be expected to lead to, any Acquisition Proposal (as defined in the section of this proxy statement/prospectus titled “The Merger Agreement—Non-Solicitation”); |
|
● |
enter into, continue or otherwise participate or engage in any discussions or negotiations regarding any Acquisition Proposal, or furnish to any person any non-public information or afford any person other than Pieris or Palvella, as applicable, access to such party’s property, books or records (except pursuant to a request by a governmental entity) in connection with any offers, inquiries or the making of any proposal or offer that constitutes, or would reasonably be expected to lead to, any Acquisition Proposal; |
|
● |
take any action to make the provisions of any “fair price”, “business combination” or “control share acquisition” statute or other similar statute or regulation inapplicable to any transactions contemplated by an Acquisition Proposal; or |
|
● |
publicly propose to do any of the foregoing. |
Board Recommendation Change
Subject to specified exceptions described in the Merger Agreement, Pieris has agreed that the Pieris board of directors (and any committee thereof) may not take any of the following actions, each of which are referred to in this proxy statement/prospectus as a Pieris board of directors recommendation change:
|
● |
fail to include its recommendation to the Pieris stockholders in connection with the solicitation of their approval of the share issuance and the amendment to Pieris’ amended and restated articles of incorporation to increase the number of authorized shares of Pieris common stock and ratification of the Pieris board of directors’ approval of the amendment to Pieris’ amended and restated articles of incorporation to change the name of Pieris to “Palvella Therapeutics, Inc.” at the Pieris special meeting in this proxy statement/prospectus or shall have withdrawn or modified such recommendation in a manner adverse to Palvella; |
|
● |
withhold, withdraw or modify (or publicly propose to withhold, withdraw or modify) the approval or recommendation of the Pieris board of directors with respect to the share issuance or the amendment to Pieris’ amended and restated articles of incorporation to increase the number of authorized shares of Pieris common stock and the amendment to Pieris’ amended and restated articles of incorporation to change the name of Pieris to “Palvella Therapeutics, Inc.”; |
|
● |
after the receipt by Pieris of an Acquisition Proposal and Palvella’s subsequent request in writing that the Pieris board of directors reconfirm its recommendation to the Pieris stockholders to solicit their approval of the required Pieris voting proposals at the Pieris special meeting, fail to reconfirm its recommendation within ten business days after its receipt of Palvella’s request; |
|
● |
fail to recommend against acceptance of a tender offer within ten business days after commencement; or |
|
● |
propose publicly to approve, endorse, adopt or recommend, or have approved, endorsed, adopted, or recommended any Acquisition Proposal. |
Subject to specified exceptions described in the Merger Agreement, Palvella agreed that the Palvella board of directors may not take any of the following actions, each of which are referred to in this proxy statement/prospectus as a Palvella board of directors recommendation change:
|
● |
withhold, withdraw or modify (or publicly propose to withhold, withdraw or modify) the approval or recommendation of the Palvella board of directors with respect to the Merger; |
|
● |
fail to recommend against acceptance of a tender offer within ten business days after commencement; or |
|
● |
propose publicly to approve, endorse, adopt or recommend, or have approved, endorsed, adopted, or recommended any Acquisition Proposal. |
Termination of the Merger Agreement
Either Pieris or Palvella may terminate the Merger Agreement under certain circumstances, which would prevent the Merger from being consummated.
Termination Fee
If the Merger Agreement is terminated under specified circumstances, Pieris will be required to pay Palvella a termination fee of $1.0 million. If the Merger Agreement is terminated under certain specified circumstances, Palvella will be required to pay Pieris a termination fee of $2.0 million.
Support Agreements
In order to induce Pieris to enter into the Merger Agreement, certain Palvella stockholders have entered into support agreements with Pieris pursuant to which, among other things, each such stockholder has agreed, solely in his, her or its capacity as a Palvella stockholder, to vote all of his, her or its shares of Palvella capital stock in favor of the adoption of the Merger Agreement. These Palvella stockholders also agreed to vote against any competing Acquisition Proposal with respect to Palvella.
As of October 28, 2024, the Palvella stockholders that are party to a support agreement with Pieris owned an aggregate of 13,160,619 shares of Palvella capital stock, representing approximately 64.3% of the outstanding shares of Palvella voting capital stock on an as converted to common stock basis. These stockholders include (i) all executive officers and directors of Palvella and their affiliates which own shares of Palvella capital stock and (ii) certain stockholders owning 5% or more of the outstanding shares of Palvella capital stock. Following the effectiveness of the Registration Statement on Form S-4 of which this proxy statement/prospectus is a part and pursuant to the Merger Agreement, Palvella stockholders holding a sufficient number of shares of Palvella capital stock to adopt the Merger Agreement and approve the Merger and related transactions will be asked to execute written consents providing for such adoption and approval. Petrichor Opportunities Fund I LP and Petrichor Opportunities Fund I Intermediate LP are parties to the support agreement and are affiliates of Tadd S. Wessel, a member of the Palvella board of directors.
In addition, in order to induce Palvella to enter into the Merger Agreement, certain Pieris stockholders have entered into support agreements with Palvella pursuant to which, among other things, each such stockholder has agreed to vote all shares of Pieris common stock owned by him, her, or it as of the record date in favor of Proposals Nos. 1, 2, 3 and 4. These Pieris stockholders also agreed to vote against any competing Acquisition Proposal with respect to Pieris.
As of October 28, 2024, the Pieris stockholders that are party to a support agreement owned an aggregate of 258,496 shares of Pieris common stock representing approximately 19.6% of the outstanding shares of Pieris common stock. These stockholders include certain executive officers and directors of Pieris and Biotechnology Value Fund, L.P. and/or its affiliates (collectively, “BVF”), a stockholder of Pieris holding a significant portion of the outstanding shares of Pieris common stock.
Lock-Up Agreements
Certain of Palvella’s and Pieris’ executive officers, directors and stockholders have entered into lock-up agreements, pursuant to which such parties have agreed not to, except in limited circumstances, offer, pledge, sell, contract to sell, sell any option to purchase, purchase any option or contract to sell, grant any option, right or warrant to purchase, or otherwise transfer or dispose of, any shares of Pieris common stock, until 180 days after the Effective Time.
The Palvella stockholders who have executed lock-up agreements as of October 28, 2024 owned, in the aggregate, approximately 63.3% of the shares of Palvella’s outstanding capital stock. The Pieris stockholders who have executed lock-up agreements as of October 28, 2024 owned, in the aggregate, approximately 19.6% of the shares of Pieris’ outstanding common stock.
Securities Purchase Agreement
On July 23, 2024, in connection with and concurrently with the execution of the Merger Agreement, Pieris entered into the Purchase Agreement with the PIPE Investors, pursuant to which such PIPE Investors have agreed to subscribe for and purchase (in cash or by cancellation of convertible notes), and Pieris has agreed to issue and sell to the PIPE Investors, an aggregate of approximately 3,154,241 of shares of Pieris common stock at a price per share equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, and/or in lieu of Pieris common stock to certain purchasers who so choose, Pre-Funded Warrants to purchase up to 2,592,585 shares of the combined company common stock at a purchase price per Pre-Funded Warrant equal to the Purchase Price minus $0.001, which is expected to be consummated immediately following the closing of the Merger. The closing of the PIPE Financing is conditioned upon the closing of the Merger, as well as certain other conditions. Pieris’ and Palvella’s obligations to consummate the Merger are conditioned upon the closing of the PIPE Financing immediately following the closing of the Merger.
Management Following the Merger
Effective as of the closing of the Merger, the combined company’s executive officers are expected to be:
|
Name |
Title |
|
|
Wesley H. Kaupinen |
President and Chief Executive Officer |
|
|
Matthew Korenberg |
Chief Financial Officer |
|
|
Kathleen Goin |
Chief Operating Officer |
|
|
Jeffrey Martini, Ph.D. |
Chief Scientific Officer |
|
| Kathleen A. McGowan | Vice President, Finance and Operations |
Interests of Certain Directors, Officers and Affiliates of Pieris and Palvella
Interests of Pieris
|
● |
In considering the recommendation of the Pieris board of directors with respect to approving the Merger, Pieris stockholders should be aware that Pieris’ executive officers and directors may have interests in the Merger that are different from, or in addition to, the interests of Pieris stockholders generally. Interests of the directors and executive officers may be different from or in addition to the interests of the stockholders for the following reasons, among others: As of October 28, 2024, Pieris’ current directors and executive officers beneficially owned, in the aggregate, approximately 0.07% of the shares of Pieris capital stock, which for purposes of this subsection excludes any Pieris shares issuable upon exercise or settlement of Pieris stock options held by such individual. Each of Pieris’ officers, directors and affiliated stockholders have also entered into a support agreement in connection with the Merger. For a more detailed discussion of the support agreements, please see the section titled “Agreements Related to the Merger—Support Agreements” beginning on page 179 of this proxy statement/prospectus. |
|
● |
one of Pieris’ directors is expected to become a director in the combined company following closing of the Merger. |
These interests are discussed in more detail in the sections titled “The Merger—Interests of Pieris Directors and Executive Officers in the Merger,” “The Merger Agreement—Indemnification and Insurance for Directors and Officers” and “Pieris Executive and Director Compensation” beginning on pages 148, 171, and 187, respectively, of this proxy statement/prospectus.
Interests of Palvella
|
● |
in considering the recommendation of the Palvella board of directors with respect to approving the Merger, stockholders should be aware that Palvella’s directors and executive officers may have interests in the Merger that are different from, or in addition to, the interests of Palvella stockholders generally. Interests of the directors and executive officers may be different from or in addition to the interests of the stockholders for the following reasons, among others: As of October 28, 2024, Palvella’s current directors and executive officers beneficially owned, in the aggregate, approximately 28.53% of the shares of Palvella capital stock, which for purposes of this subsection excludes any Palvella shares issuable upon exercise or settlement of Palvella stock options held by such individual. Each of Palvella’s officers, directors and affiliated stockholders have also entered into a support agreement in connection with the Merger. For a more detailed discussion of the support agreements, please see the section titled “Agreements Related to the Merger—Support Agreements” beginning on page 179 of this proxy statement/prospectus; and |
|
● |
all of Palvella’s executive officers and four of the six of Palvella directors are expected to become the executive officers and directors of the combined company upon the closing of the Merger. |
These interests are discussed in more detail in the sections titled “The Merger—Interests of Palvella Directors and Executive Officers in the Merger,” “The Merger Agreement—Indemnification and Insurance for Directors and Officers” and “Palvella Executive and Director Compensation” beginning on pages 150, 171, and 201, respectively, of this proxy statement/prospectus.
Each of Palvella’s directors and executive officers have also entered into a lock-up agreement in connection with the Merger. For a more detailed discussion of the lock-up agreements, please see the section titled “Agreements Related to the Merger—Lock-Up Agreements” beginning on page 180, respectively, of this proxy statement/prospectus.
Material U.S. Federal Income Tax Consequences of the Merger
Subject to the qualifications and limitations set forth in the section titled “The Merger—Material U.S. Federal Income Tax Consequences of the Merger,” in the opinion of Mintz and Troutman, the Merger will qualify as a “reorganization” within the meaning of Section 368(a) of the Code, and a Palvella U.S. holder will not recognize gain or loss for U.S. federal income tax purposes upon the receipt of shares of Pieris common stock in exchange for shares of Palvella capital stock in the Merger, except with respect to cash received in lieu of a fractional share of Pieris common stock.
However, Palvella has not sought and does not intend to seek a ruling from the IRS regarding the intended tax treatment of the Merger and, even though an opinion of counsel has been sought and obtained by Palvella, such opinion is not binding upon the IRS or a court. Consequently, there can be no assurance that the IRS will not challenge the intended tax treatment of the Merger and, if challenged, that a court would not sustain the IRS’ position.
If the Merger does not qualify as a reorganization within the meaning of Section 368(a) of the Code, then each Palvella U.S. holder would recognize gain or loss upon the exchange of shares of Palvella capital stock for Pieris common stock in the Merger equal to the difference between the fair market value of the shares of Pieris common stock received in exchange for the shares of Palvella capital stock (plus any cash received in lieu of a fractional share) and such Palvella U.S. holder’s adjusted tax basis in the shares of Palvella capital stock surrendered.
Because the Pieris stockholders will not sell, exchange or dispose of any shares of Pieris common stock in the Merger, there will be no material U.S. federal income tax consequences to Pieris stockholders upon consummation of the Merger.
See the section titled “The Merger—Material U.S. Federal Income Tax Consequences of the Merger” beginning on page 155 of this proxy statement/prospectus for a more complete description of the material U.S. federal income tax consequences of the Merger to Palvella U.S. holders.
Material U.S. Federal Income Tax Consequences of Receipt of CVRs
The U.S. federal income tax treatment of the Pieris stockholders’ receipt of the CVRs is unclear. Absent a change in law requiring otherwise after the date of the CVR Agreement, Pieris will not report the issuance of the CVRs to Pieris stockholders as a current distribution and will report each payment (if any) on the CVRs as a distribution by Pieris for U.S. federal income tax purposes. This position may be challenged by the IRS in which case holders of Pieris common stock could be required to recognize taxable income in respect of the CVRs without the corresponding receipt of cash. See the section titled “Agreements Related to the Merger—CVR Agreement—Material U.S. Federal Income Tax Consequences of the Receipt of CVRs” beginning on page 182 of this proxy statement/prospectus for a more complete description of the material U.S. federal income tax consequences of the receipt of CVRs to Pieris stockholders, including possible alternative treatments.
Both Pieris and Palvella are subject to various risks associated with their businesses and their industries. In addition, the Merger, including the possibility that the Merger may not be completed, poses a number of risks to each company and its respective securityholders, including the following risks:
Risks Related to the Merger
|
● |
the exchange ratio will not be adjusted based on the market price of Pieris common stock, so the merger consideration at the closing may have a greater or lesser value than at the time the Merger Agreement was signed; |
|
● |
failure to complete the Merger may result in either Pieris or Palvella paying a termination fee to the other party, which could harm the common stock price of Pieris and future business and operations of each company; |
|
● |
if the conditions to the Merger are not satisfied or waived, the Merger may not occur; and |
|
● |
at the time of the Merger, Pieris will be a shell company due to its expected nominal operations and assets following the discontinuation of its historical research and development efforts. As a result, the combined company will be subject to more stringent reporting requirements, offering limitations and resale restrictions. |
Risks Related to Pieris
|
● |
Pieris has incurred significant losses since its inception and anticipates that it will continue to incur losses for the foreseeable future. Pieris currently has no product revenues and no approved products and will rely on its partnered IO programs to generate revenue; |
|
● |
if the Merger is not completed, Pieris will reconsider its strategic alternatives, including operating its business, dissolving and liquidating its assets, or pursuing another strategic transaction. Pieris’ future capital requirements depend on many factors, and adequate additional financing may not be available to it on acceptable terms, or at all; |
Risks Related to Palvella
|
● |
Palvella has incurred significant operating losses since inception and anticipates that it will continue to incur significant operating losses for at least the next several years. Palvella may never achieve or maintain profitability; |
|
● |
Palvella has never generated revenue from product sales and may never achieve or maintain profitability; |
|
● |
Palvella’s recurring losses from operations and financial condition raise substantial doubt about its ability to continue as a going concern; |
|
● |
Palvella will likely require substantial additional funding to finance its operations, which may cause dilution to its stockholders, and a failure to obtain this necessary funding when needed on acceptable terms, or at all, could force it to delay, limit, reduce or terminate its product development, commercialization efforts or other operations; |
|
● |
Palvella’s future success is substantially dependent on the successful clinical development, regulatory approval and commercialization of QTORIN rapamycin, which is in later stages of development than its other product candidates; |
|
● |
Palvella may be unable to obtain regulatory approval for its product candidates under applicable regulatory requirements. The denial or delay of any such approval would adversely impact its potential to generate revenue, its business and its results of operations; |
|
● |
the rare genetic skin diseases Palvella is currently targeting have no FDA-approved therapies, which subjects the design and execution of its clinical development program to complexities and known and unknown risks, including those related to novel and/or subjective clinical endpoints and varying patient population characteristics; |
|
● |
Palvella’s lead product candidates are based on its QTORIN platform and it is highly dependent on the successful development of this novel and unproven technology. |
|
● |
Palvella may be unable to obtain Orphan Drug Designation for certain of its product candidates and, even if it obtains such designation, it may not be able to realize the benefits of such designation, including potential marketing exclusivity of its product candidates, if approved; |
|
● |
Palvella’s development and commercialization strategy for its product candidates depends, in part, on published scientific literature and the FDA’s prior findings regarding the safety and efficacy of rapamycin. If Palvella is not able to pursue this strategy, it may be delayed in receiving regulatory authority approval; |
|
● |
Fast Track Designation granted for QTORIN rapamycin for the treatment of microcystic LM and, if granted, for any of Palvella’s other product candidates by the FDA may not lead to a faster development or regulatory review or approval process and does not increase the likelihood that its product candidates will receive marketing approval; |
|
● |
even if QTORIN rapamycin or any future product candidates receive marketing approval, they may fail to achieve market acceptance by physicians, patients, third-party payors or others in the medical community necessary for commercial success; |
|
● |
Palvella currently relies on contract manufacturer organizations, or “CMOs”, to manufacture preclinical and clinical supplies of its product candidates and will rely on CMOs for the commercial supplies of any approved product candidate. The loss of these manufacturers, or their failure to provide Palvella with sufficient quantities at acceptable quality levels or prices, or at all, would materially and adversely affect its business; and |
|
● |
Palvella may not be able to obtain, maintain or enforce patent rights that cover its product candidates and technologies that are of sufficient breadth to prevent third parties from competing against it. |
Risks Related to the Combined Company
|
● |
the market price of the combined company common stock is expected to be volatile, and the market price of the common stock may drop following the Merger; |
|
● |
the combined company will need substantial additional funding, in addition to the funds from the PIPE Financing before it can complete the development of its product candidates. If the combined company is unable to obtain such additional capital on favorable terms, on a timely basis or at all, it would be forced to delay, reduce or eliminate its product development and clinical programs and may not have the capital required to otherwise operate its business; and |
|
● |
the combined company will incur additional costs and increased demands upon management as a result of complying with the laws and regulations affecting public companies. |
These risks and other risks are discussed in greater detail under the section titled “Risk Factors” beginning on page 25 of this proxy statement/prospectus. Pieris and Palvella both encourage you to read and consider all of these risks carefully.
Palvella intends to file a listing application for the combined company common stock with Nasdaq following completion of the Merger. After completion of the Merger, the combined company will be renamed “Palvella Therapeutics, Inc.” and, assuming approval of the application for continued listing, the common stock of the combined company will trade on The Nasdaq Capital Market under the symbol “PVLA.” However, Nasdaq’s determination may not be known at the time stockholders are asked to vote on the Merger. In addition, under the Merger Agreement, each of Pieris’ and Palvella’s obligation to complete the Merger is subject to the satisfaction or waiver by each of the parties, at or prior to the Merger, of various conditions, including that the shares of Pieris common stock to be issued in the Merger have been approved for listing on Nasdaq as of the closing of the Merger.
Anticipated Accounting Treatment
The Merger is expected to be accounted for as a reverse recapitalization in accordance with U.S. generally accepted accounting principles, or “GAAP”. Under this method of accounting, Pieris, which is the legal acquirer, will be treated as the “acquired” company for financial reporting purposes, and Palvella will be treated as the accounting acquirer. This determination will be primarily due to Pieris being determined to be a shell company at the time of the Merger, immediately following the dividend of the CVR and following the discontinuation of historical research and development efforts, because it will not meet the GAAP definition of a business, will not have more than nominal assets, and will not have more than nominal operations. Further, it is expected that, immediately following the Merger, Palvella’s stockholders will have a majority of the voting power of the combined company, Palvella will control four of five seats on the board of directors of the combined company, and Palvella’s senior management will comprise all of the senior management of the combined company. Accordingly, for accounting purposes, the Merger will be treated as the equivalent of a capital transaction in which Palvella is issuing stock for the net assets of Pieris. The net assets of Pieris will be stated at historical cost, with no goodwill or other intangible assets recorded. See the section titled “Unaudited Pro Forma Condensed Combined Financial Information” beginning on page 299 of this proxy statement/prospectus for additional information.
Appraisal Rights and Dissenters’ Rights
Holders of Pieris common stock are not entitled to dissenter’s or appraisal rights under Nevada law in connection with the Merger. Holders of Palvella capital stock are entitled to appraisal rights in connection with the Merger under Delaware law. See the section titled “Appraisal Rights and Dissenters’ Rights” on page 159 of this proxy statement/prospectus for additional information.
Comparison of Stockholder Rights
Pieris is incorporated under the laws of the State of Nevada and Palvella is incorporated under the laws of the State of Delaware. Accordingly, the rights of the stockholders Pieris are currently, and will continue to be, governed by the Nevada Revised Statutes, or the “NRS”, and the rights of the stockholders of Palvella are currently governed by the Delaware General Corporation Law, or the “DGCL”. If the Merger is completed, Palvella stockholders will become Pieris stockholders, and their rights will be governed by Nevada law, the amended and restated bylaws of Pieris and the amended and restated articles of incorporation of Pieris, as may be further amended by Proposal No. 2 and Proposal No. 4 if approved or ratified, as the case may be, by the Pieris stockholders at the Pieris special meeting. The rights of Pieris stockholders contained in its amended and restated articles of incorporation and amended and restated bylaws of Pieris differ from the rights of Palvella stockholders under Palvella’s third amended and restated certificate of incorporation and bylaws of Palvella, as more fully described under the section titled “Comparison of Rights of Holders of Pieris Capital Stock and Palvella Capital Stock” beginning on page 313 of this proxy statement/prospectus.
MARKET PRICE AND DIVIDEND INFORMATION
Market Price Information
Pieris common stock is currently listed on The Nasdaq Capital Market under the symbol “PIRS.”
The closing price of Pieris common stock on July 23, 2024, the last trading day prior to the public announcement of the Merger, was $7.98 per share and the closing price of Pieris common stock on October 28, 2024 was $17.00 per share, in each case as reported on The Nasdaq Capital Market.
Because the market price of Pieris common stock is subject to fluctuation, the market value of the shares of Pieris common stock that Palvella stockholders will be entitled to receive in the Merger may increase or decrease.
Palvella is a private company and its shares of common stock and preferred stock are not publicly traded.
Assuming approval of Proposal Nos. 1 and 2 and successful application for initial listing with The Nasdaq Capital Market, Pieris and Palvella anticipate that the common stock of the combined company, which will be renamed to Palvella Therapeutics, Inc., will be listed on The Nasdaq Capital Market following the closing of the Merger under the trading symbol “PVLA.”
As of October 28, 2024, the record date for the Pieris special meeting, there were approximately 30 holders of record of Pieris common stock and five holders of record of Pieris preferred stock. As of October 28, 2024, Palvella had 16 holders of record of Palvella common stock and 77 holders of record of Palvella preferred stock. For detailed information regarding the beneficial ownership of certain Pieris and Palvella stockholders, see the sections titled “Principal Stockholders of Pieris” and “Principal Stockholders of Palvella” on pages 328 and 331, respectively, of this proxy statement/prospectus.
Dividends
Pieris has never declared or paid cash dividends on its capital stock and does not anticipate paying any cash dividends in the foreseeable future. Palvella has never paid or declared any cash dividends on its capital stock and does not anticipate paying any cash dividends in the foreseeable future. Palvella intends to retain all available funds and any future earnings for use in the operation of its business and does not anticipate paying any cash dividends on its capital stock in the foreseeable future. Notwithstanding the foregoing, any determination to pay cash dividends subsequent to the Merger will be at the discretion of the combined company’s board of directors and will depend upon a number of factors, including the combined company’s results of operations, financial condition, future prospects, contractual restrictions, restrictions imposed by applicable law and other factors the combined company’s board of directors deems relevant.
The combined company will be faced with a market environment that cannot be predicted and that involves significant risks, many of which will be beyond its control. In addition to the other information contained or incorporated by reference in this proxy statement/prospectus, you should carefully consider the material risks described below before deciding how to vote your shares of Pieris common stock. You should also read and consider the other information in this proxy statement/prospectus and additional information about Pieris set forth in its Annual Report on Form 10-K for the fiscal year ended December 31, 2023, which is filed with the SEC as such risks may be updated or supplemented in its subsequent Quarterly Reports on Form 10-Q or Current Reports on Form 8-K, each of which is incorporated by reference into this proxy statement/prospectus. Please see the section titled “Where You Can Find More Information” beginning on page 336 of this proxy statement/prospectus for further information regarding the documents incorporated by reference into this proxy statement/prospectus.
Risks Related to the Strategic Transactions
The exchange ratio will not be adjusted based on the market price of Pieris common stock as the Exchange Ratio depends on the Pieris net cash as of the date immediately preceding the closing of the Merger and not the market price of Pieris common stock, so the merger consideration at the closing may have a greater or lesser value than at the time the Merger Agreement was signed.
At the Effective Time, outstanding shares of Palvella capital stock will be converted into shares of Pieris common stock. Immediately after the Merger, but without giving effect to the PIPE Financing, applying the exchange ratio, Pieris securityholders immediately before the Merger are expected to own approximately 18% of the outstanding shares of capital stock of the combined company and the former Palvella securityholders immediately before the Merger are expected to own approximately 82% of the outstanding share of capital stock of the combined company, in each case, on a fully-diluted basis, calculated using the treasury stock method, subject to certain assumptions, including, but not limited to, a valuation for Pieris equal to $21 million, provided, that (a) if Pieris’ net cash as of the closing is greater than $11 million, then Pieris’ valuation will be adjusted upwards on a dollar-for-dollar basis by the difference of: (i) Pieris’ net cash, minus (ii) $11 million, and (b) if Pieris’ net cash is less than $11 million, then Pieris’ valuation will be adjusted downwards on a dollar-for-dollar basis by the difference of: (i) $11 million, minus (ii) Pieris’ net cash, as further described in the Merger Agreement.
Immediately following the PIPE Financing, the Pieris securityholders as of immediately prior to the Merger are expected to hold approximately 10.47% of the shares of combined company capital stock, the Palvella securityholders as of immediately prior to the Merger are expected to hold approximately 50.18% of the shares of combined company capital stock, and the PIPE Investors are expected to hold approximately 39.35% of the shares of combined company capital stock, in each case, on a fully diluted basis, subject to certain assumptions.
Any changes in the market price of Pieris stock before the completion of the Merger will not affect the number of shares Palvella stockholders will be entitled to receive pursuant to the Merger Agreement. Therefore, if before the completion of the Merger, the market price of Pieris common stock increases from the market price on the date of the Merger Agreement, then Palvella stockholders could receive merger consideration with substantially more value for their shares of Palvella capital stock than the parties had negotiated when they established the exchange ratio. Similarly, if before the completion of the Merger the market price of Pieris common stock declines from the market price on the date of the Merger Agreement, then Palvella stockholders could receive merger consideration with substantially lower value. The Merger Agreement does not include a price-based termination right.
Failure to complete the Merger may result in either Pieris or Palvella paying a termination fee to the other party, which could harm the common stock price of Pieris and future business and operations of each company.
If the Merger is not completed, Pieris and Palvella are subject to the following risks:
|
● |
if the Merger Agreement is terminated under certain specified circumstances, Pieris will be required to pay Palvella a termination fee of $1 million; |
|
● |
if the Merger Agreement is terminated under certain other specified circumstances, Palvella will be required to pay Pieris a termination fee of $2 million; |
|
● |
the price of Pieris common stock may decline and could fluctuate significantly; and |
|
● |
costs related to the Merger for Pieris, such as financial advisor, legal and accounting fees, which Pieris estimates will total approximately $2.05 million, $1.5 million, and $0.1 million, respectively, a majority of which must be paid even if the Merger is not completed. |
If the Merger Agreement is terminated and the Pieris board of directors or the Palvella board of directors determines to seek another business combination, there can be no assurance that either Pieris or Palvella will be able to find a partner with whom a business combination would yield greater benefits than the benefits to be provided under the Merger Agreement.
If the conditions to the Merger are not satisfied or waived, the Merger may not occur.
The completion of the Merger is not assured. Even if the Merger Agreement is adopted by the stockholders of Palvella and the Merger and the issuance of Pieris common stock pursuant thereto are approved by the stockholders of Pieris, specified conditions must be satisfied or waived to complete the Merger. These conditions are set forth in the Merger Agreement and described in the section titled “The Merger Agreement—Conditions to the Completion of the Merger” beginning on page 172 of this proxy statement/prospectus. Pieris and Palvella cannot assure you that all of the conditions to the consummation of the Merger will be satisfied or waived. If the conditions are not satisfied or waived, the Merger may not occur or the closing may be delayed, and Pieris and Palvella each may lose some or all of the intended benefits of the Merger.
Transfers of the combined company’s securities utilizing Rule 144 of the Securities Act may be limited.
A significant portion of the combined company’s securities will be restricted from immediate resale. Holders should be aware that transfers of the combined company’s securities pursuant to Rule 144 may be limited as Rule 144 is not available, subject to certain exceptions, for the resale of securities initially issued by shell companies (other than business combination related shell companies) or issuers that have been at any time previously a shell company. At the time of the Merger, Pieris will be a shell company due to its expected nominal operations and assets, immediately following the dividend of the CVR and following the discontinuation of its historical research and development efforts. Pieris anticipates that following the consummation of the Merger, the combined company will no longer be a shell company. As a result, Pieris anticipates that holders will not be able to sell their restricted combined company securities pursuant to Rule 144 without registration until one year after the combined company files the Current Report on Form 8-K following the closing that includes the required Form 10 information that reflects that the combined company is no longer a shell company.
At the time of the Merger, Pieris will be a shell company due to its expected nominal operations and assets following the discontinuation of historical research and development efforts. As a result, the combined company will be subject to more stringent reporting requirements, offering limitations and resale restrictions.
According to SEC guidance, the requirements applicable to reporting shell company business combinations apply to any company that sells or otherwise disposes of its historical assets or operations in connection with or as part of a plan to combine with a non-shell private company in order to convert the private company into a public one. At the time of the Merger, Pieris will be a shell company immediately following the dividend of the CVR, because it will not have more than nominal operations and assets following the discontinuation of historical research and development efforts. Further, it is expected that, immediately following the Merger, Palvella’s stockholders will have a majority of the voting power of the combined company, Palvella will control four of five seats on the board of directors of the combined company, and Palvella’s senior management will comprise all of the senior management of the combined company. As such, Pieris will become a shell company upon consummation of the Merger, and its Merger with Palvella would be subject to the requirements applicable to shell company business combinations.
The requirements applicable to shell company business combinations are as follows:
|
● |
the combined company will need to file a Form 8-K to report the Form 10 type information after closing with the SEC reflecting its status as an entity that is not a shell company; |
|
● |
Pieris is not, and the combined company will not be, eligible to use a Form S-3 until 12 full calendar months after closing; |
|
● |
the combined company will need to wait at least 60 calendar days after closing to file a Form S-8 for any equity plans or awards; |
|
● |
the combined company will be an “ineligible issuer” for three years following the closing, which will prevent the combined company from (i) incorporating by reference in its Form S-1 filings, (ii) using a free writing prospectus, or (iii) taking advantage of well-known seasoned issuer status despite its public float; |
|
● |
investors who (i) are affiliates of Palvella at the time the Merger is submitted for the vote or consent of Palvella stockholders, (ii) receive securities of the combined company in the Merger (i.e., Rule 145(c) securities) and (iii) publicly offer or sell such securities, will be deemed to be engaged in a distribution of such securities, and therefore to be underwriters with respect to resales of those securities, and accordingly such securities may not be included in the Form S-1 resale registration statement anticipated to be filed after the closing of the Merger unless such securities are sold only in a fixed price offering in which such investors are named as underwriters in the prospectus; |
|
● |
Rule 144(i)(2) will limit the ability to publicly resell Rule 145(c) securities per Rule 145(d), as well as any other “restricted” or “control” securities of the combined company per Rule 144 (i.e., holders of restricted securities and any affiliates of the public company are also affected) until one year after the Form 10 information is filed with the SEC. |
The foregoing SEC requirements would increase the combined company’s time and cost of raising capital, offering stock under equity plans, and complying with securities laws. Further, such requirements will add burdensome restrictions on the resale of combined company shares by affiliates of Palvella and any holders of “restricted” or “control” securities.
Palvella or Pieris may waive one or more of the conditions to the Merger without re-soliciting stockholder approval.
Certain conditions to Palvella’s or Pieris’ obligations to complete the Merger may be waived, in whole or in part, to the extent permitted by law, either unilaterally or by agreement of Palvella and Pieris. In the event of a waiver of a condition, the Pieris board of directors will evaluate the materiality of any such waiver to determine whether amendment of this proxy statement/prospectus and re-solicitation of proxies is necessary.
In the event that the Pieris board of directors, in its own reasonable discretion, determines any such waiver is not significant enough to require re-solicitation of its stockholders, it will have the discretion to complete the Merger without seeking further stockholder approval, which decision may have a material adverse effect on the Pieris stockholders. For example, if Palvella and Pieris agree to waive the requirement that the Nasdaq application be accepted for listing prior to the consummation of the Merger, and their respective boards of directors elected to proceed with the closing of the Merger, Nasdaq may notify the combined company of its determination to delist the company’s securities based upon the failure to satisfy the initial inclusion criteria. The combined company may appeal the determination to a hearings panel, which will stay the delisting action pending a panel decision. If the combined company does not appeal the determination, its common stock will be delisted.
For more information about the conditions to the completion of the Merger, see the section titled “The Merger Agreement—Conditions to the Completion of the Merger.”
Palvella or Pieris may waive one or more of the conditions to the Merger.
Palvella or Pieris may agree to waive, in whole or in part, some of the conditions to each party’s obligations to complete the Merger, to the extent permitted by applicable law. For example, it is a condition to Palvella’s obligations to close the Merger that certain of Pieris’ representations and warranties are true and correct in all respects as of the closing of the Merger, except where the failure of such representants and warranties to be true and correct, individually or in the aggregate, has not had and is not reasonably likely to have a material adverse effect with respect to Pieris. However, if the Palvella board of directors determines that it is in the best interest of the stockholders of Palvella to waive any such breach, then the Palvella board of directors may elect to waive that condition and consummate the Merger, which decision may have an adverse effect on the stockholders of the combined company following the Merger. For example, if such a breach was the result of a material adverse effect with respect to Pieris, the market could react negatively to such information, which may cause a substantial decline in the price of the common stock of the combined company following the Merger.
Notwithstanding the foregoing, certain closing conditions may not be waived due to applicable law or otherwise. The following closing conditions may not be waived: receipt of the requisite stockholder approvals; the effectiveness of the Registration Statement on Form S-4 of which this proxy statement/prospectus forms a part; and the absence of any order or injunction that has the effect of prohibiting the consummation of the Merger; See the section “The Merger Agreement—Conditions to the Completion of the Merger” for further information.
The Merger may be completed even though a material adverse effect may result from the announcement of the Merger, industry-wide changes or other causes.
In general, neither Pieris nor Palvella is obligated to complete the Merger if there is a material adverse effect affecting the other party between July 23, 2024, the date of the Merger Agreement, and the closing of the Merger. However, certain types of changes are excluded from the concept of a “material adverse effect.” Such exclusions include but are not limited to changes in general economic or market conditions, industry wide changes, changes in GAAP, changes in laws, rules or regulations of general applicability or interpretations thereof, natural disasters, epidemics, pandemics or other disease outbreaks, outbreaks of major hostilities or acts of terrorism, changes resulting from the announcement or pendency of the Merger, and failures to meet internal guidance, budgets, plans or forecasts. Therefore, if any of these events were to occur impacting Pieris or Palvella, the other party would still be obliged to consummate the closing of the Merger. If any such adverse changes occur and Pieris and Palvella consummate the closing of the Merger, the stock price of the combined company may suffer. This in turn may reduce the value of the Merger to the stockholders of Pieris, Palvella or both. Additionally, if there is a material adverse effect, the PIPE Investors will not be obligated to consummate the purchase of shares of Pieris common stock in connection with the PIPE Financing, which, in turn, may reduce the value of the Merger to the stockholders of Pieris, Palvella, or both. For a more complete discussion of what constitutes a material adverse effect on Pieris or Palvella, see the section titled “The Merger Agreement—Conditions to Completion of the Merger” beginning on page 172 of this proxy statement/prospectus.
If Pieris and Palvella complete the Merger and the PIPE Financing closes, the combined company will need to raise additional capital by issuing equity securities or additional debt or through licensing arrangements, which may cause significant dilution to the combined company’s stockholders or restrict the combined company’s operations.
On July 23, 2024, Pieris entered into the Purchase Agreement with the PIPE Investors, pursuant to which, among other things, the PIPE Investors agreed to subscribe for and purchase (either for cash or in exchange for the termination and cancellation of outstanding convertible notes issued by Palvella) an aggregate of approximately 3,154,241 of shares of Pieris common stock at a price per share equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, and/or in lieu of Pieris common stock to certain purchasers who so choose, Pre-Funded Warrants to purchase up to 2,592,585 shares of the combined company common stock at a purchase price per Pre-Funded Warrant equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, minus $0.001, which is expected to close immediately following the closing of the Merger. The closing of the PIPE financing is conditioned upon the closing of the Merger.
Even if the PIPE Financing closes as expected, the combined company may need to raise additional capital in the future. Additional financing may not be available to the combined company when it is needed or may not be available on favorable terms. To the extent that the combined company raises additional capital by issuing equity securities, such financing will cause additional dilution to all securityholders of the combined company, including Pieris’ pre-Merger securityholders and Palvella’s former securityholders. It is also possible that the terms of any new equity securities may have preferences over the combined company common stock. Any debt financing the combined company enters into may involve covenants that restrict its operations. These restrictive covenants may include limitations on additional borrowing and specific restrictions on the use of the combined company’s assets, as well as prohibitions on its ability to create liens, pay dividends, redeem its stock or make investments. In addition, if the combined company raises additional funds through licensing arrangements, it may be necessary to grant licenses on terms that are not favorable to the combined company.
Some Pieris and Palvella directors and executive officers may have interests in the Merger that are different from yours and that may influence them to support or approve the Merger without regard to your interests.
Directors and executive officers of Pieris and Palvella may have interests in the Merger that are different from, or in addition to, the interests of other Pieris stockholders generally. These interests with respect to Pieris’ directors and executive officers may include, among others, that certain of Pieris’ executives are entitled to, in connection with a qualifying termination of employment, accelerated vesting of options and restricted stock units with respect to Pieris common stock and the payment of severance, that certain of Pieris’ executives are entitled to the extension of the applicable executive’s post-termination exercise period with respect to their options in the event of the executive’s continued employment through the closing of the Merger, and that all of Pieris’ directors and executive officers are entitled to certain indemnification and liability insurance coverage pursuant to the terms of the Merger Agreement. In addition, a current member of the Pieris board of directors, who is expected to be Christopher Kiritsy, is expected to continue as a director of the combined company after the Effective Time, and, following the closing of the Merger, will be eligible to be compensated as a non-employee director of the combined company pursuant to the Pieris non-employee director compensation policy that is expected to remain in place following the Effective Time. These interests with respect to Palvella’s directors and executive officers may include, among others, that certain of Palvella’s directors and executive officers have options, subject to vesting, to purchase shares of Palvella common stock which, after the Effective Time, will be converted into and become options to purchase shares of the common stock of the combined company; Palvella’s executive officers are expected to continue as executive officers of the combined company after the Effective Time; and all of Palvella’s directors and executive officers are entitled to certain indemnification and liability insurance coverage pursuant to the terms of the Merger Agreement. Further, four of the current members of the Palvella board of directors are expected to continue as directors of the combined company after the Effective Time, and, following the closing of the Merger, will be eligible to be compensated as non-employee directors of the combined company pursuant to the Pieris non-employee director compensation policy that is expected to remain in place following the Effective Time. The directors and executive officers own options to purchase the shares of their respective companies.
The Pieris board of directors considered that (i) BVF is a stockholder of Pieris, (ii) BVF is a stockholder of Palvella, and (iii) BVF agreed to participate in the PIPE Financing.
The Palvella board of directors considered that (i) Todd C. Davis, a director of Palvella, is the Chief Executive Officer Ligand Pharmaceuticals, Inc. or “Ligand”, a stockholder of Palvella, and (A) Ligand will receive proceeds as a result of the Merger akin to other stockholders of Palvella, (B) Mr. Davis and Ligand agreed to participate in the PIPE Financing, and (C) Mr. Davis will be appointed to the combined company’s board of directors in connection with the Merger; (ii) Tadd Wessel, a director of Palvella, is the founder and Managing Partner of Petrichor Healthcare Capital Management, or “Petrichor”, a stockholder of Palvella, and (A) Petrichor will receive proceeds as a result of the Merger akin to other stockholders of Palvella, (B) Petrichor agreed to participate in the PIPE Financing, and (C) Mr. Wessel will be appointed to the combined company’s board of directors in connection with the Merger, (iii) (A) Aditya Asokan, Ph.D., a director of Palvella, is affiliated with Samsara BioCapital L.P., or “Samsara”, a stockholder of Palvella and (B) Samsara will receive proceeds as a result of the Merger akin to other stockholders of Palvella; and (iv) each of Wesley H. Kaupinen, Chief Executive Officer and Director of Palvella, and George Jenkins, a director of Palvella, are stockholders of Palvella and (A) will receive proceeds as a result of the Merger akin to other stockholders of Palvella, (B) agreed to participate in the PIPE Financing and (C) will be appointed to the combined company’s board of directors.
The Pieris and Palvella boards of directors were aware of and considered those interests, among other things, in reaching their decisions to approve and adopt the Merger Agreement, approve the Merger, and recommend the approval of the Merger Agreement and certain related matters to Pieris and Palvella stockholders. These interests, among other factors, may have influenced the directors and executive officers of Pieris and Palvella to support or approve the Merger.
For more information regarding the interests of Pieris and Palvella directors and executive officers in the Merger, please see the sections titled “The Merger—Interests of Pieris Directors and Executive Officers in the Merger” beginning on page 148 and “The Merger—Interests of Palvella Directors and Executive Officers in the Merger” beginning on page 150 of this proxy statement/prospectus.
Pieris stockholders may potentially not receive any payment on the CVRs and the CVRs may otherwise expire valueless.
The Merger Agreement contemplates that, prior to the Effective Time, Pieris will declare a dividend to each holder of record of shares of Pieris common stock and preferred stock entitled to receive a dividend in accordance with the terms of such preferred stock of the right to receive one CVR for each outstanding share of Pieris common stock held by such stockholder, or share of common stock underlying such preferred stock, held by such stockholder, on such date, and enter into a CVR Agreement with a rights agent and a CVR holder representative. Each CVR will represent the contractual right to receive payments upon the receipt of payments by Pieris or any of its affiliates under certain strategic partner agreements, including existing collaboration agreements pursuant to which Pieris may be entitled to milestones and royalties in the future and other outlicensing agreements for certain of Pieris’ legacy assets, and upon the receipt of certain research and development tax credits in favor of Pieris or any of its affiliates, in each case as set forth in, and subject to and in accordance with the terms and conditions of, the CVR Agreement.
Pieris may not receive proceeds under the strategic partner agreements or other CVR events set forth in the CVR Agreement. If such events are not achieved for any reason within the time periods specified in the CVR Agreement, no payments will be made under the CVRs, and the CVRs will expire valueless.
Pieris stockholders may not realize a benefit from the Merger commensurate with the ownership dilution they will experience in connection with the Merger.
If the combined company is unable to realize the full strategic and financial benefits currently anticipated from the Merger, Pieris stockholders will have experienced substantial dilution of their ownership interests without receiving any commensurate benefit, or only receiving part of the commensurate benefit to the extent the combined company is able to realize only part of the strategic and financial benefits currently anticipated from the Merger.
If the Merger is not completed, Pieris’ stock price may fluctuate significantly.
The market price of Pieris common stock is subject to significant fluctuations. During the 12-month period ended October 28, 2024, the closing sales price of Pieris common stock on The Nasdaq Capital Market ranged from a high of $22.21 on November 7, 2023 to a low of $6.53 on July 7, 2024. Market prices for securities of pharmaceutical, biotechnology and other life science companies have historically been particularly volatile. In addition, the market price of Pieris common stock will likely be volatile based on whether stockholders and other investors believe that Pieris can complete the Merger or otherwise raise additional capital to support Pieris’ operations if the Merger is not consummated and another strategic transaction cannot be identified, negotiated and consummated in a timely manner, if at all. The volatility of the market price of Pieris common stock is exacerbated by low trading volume. Additional factors that may cause the market price of Pieris common stock to fluctuate include:
|
● |
the initiation of, material developments in, or conclusion of litigation to enforce or defend its intellectual property rights or defend against claims involving the intellectual property rights of others; |
|
● |
the entry into, or termination of, key agreements, including commercial partner agreements; |
|
● |
announcements by commercial partners or competitors of new commercial products, clinical progress or lack thereof, significant contracts, commercial relationships or capital commitments; |
|
● |
the introduction of technological innovations or new therapies that compete with its future products; |
|
● |
the loss of key employees; |
|
● |
future sales of its common stock; |
|
● |
general and industry-specific economic conditions that may affect its research and development expenditures; |
|
● |
the failure to meet industry analyst expectations; and |
|
● |
period-to-period fluctuations in financial results. |
Moreover, the stock markets in general have experienced substantial volatility that has often been unrelated to the operating performance of individual companies. These broad market fluctuations may also adversely affect the trading price of Pieris common stock. In the past, following periods of volatility in the market price of a company’s securities, stockholders have often instituted class action securities litigation against such companies.
Pieris and Palvella securityholders will have a reduced ownership and voting interest in, and will exercise less influence over the management of, the combined company following the completion of the Merger as compared to their current ownership and voting interests in the respective companies.
After the completion of the Merger, the current stockholders of Pieris and Palvella will own a smaller percentage of the combined company than their ownership of their respective companies prior to the Merger. Immediately after the Merger, but without giving effect to the PIPE Financing, Pieris securityholders as of immediately prior to the Merger are expected to own approximately 18% of the outstanding shares of capital stock of the combined company and former Palvella securityholders, are expected to own approximately 82% of the outstanding shares of capital stock of the combined company, in each case, on a fully-diluted basis, calculated using the treasury stock method, subject to certain assumptions, including, but not limited to, a valuation for Pieris equal to $21 million, provided, that (a) if Pieris’ net cash as of the closing is greater than $11 million, then Pieris’ valuation will be adjusted upwards on a dollar-for-dollar basis by the difference of: (i) Pieris’ net cash, minus (ii) $11 million, and (b) if Pieris’ net cash is less than $11 million, then Pieris’ valuation will be adjusted downwards on a dollar-for-dollar basis by the difference of: (i) $11 million, minus (ii) Pieris’ net cash, as further described in the Merger Agreement.
Immediately following the PIPE Financing, the Pieris securityholders as of immediately prior to the Merger are expected to hold approximately 10.47% of the shares of combined company capital stock, the Palvella securityholders as of immediately prior to the Merger are expected to hold approximately 50.18% of the shares of combined company capital stock, and the PIPE Investors are expected to hold approximately 39.35% of the shares of combined company capital stock, in each case, on a fully diluted basis, subject to certain assumptions.
During the pendency of the Merger, Pieris and Palvella may not be able to enter into a business combination with another party on more favorable terms because of restrictions in the Merger Agreement, which could adversely affect their respective business prospects.
Covenants in the Merger Agreement impede the ability of Pieris and Palvella to make acquisitions during the pendency of the Merger, subject to specified exceptions. As a result, if the Merger is not completed, the parties may be at a disadvantage to their competitors during that period. In addition, while the Merger Agreement is in effect, each party is generally prohibited from soliciting, proposing, seeking, initiating or knowingly encouraging, facilitating or supporting any inquiries, indications of interest, proposals or offers that constitute or may reasonably be expected to lead to certain transactions involving a third party, including a merger, sale of assets or other business combination, subject to specified exceptions. Any such transactions could be favorable to such party’s stockholders, but the parties may be unable to pursue them. For more information, see the section titled “The Merger Agreement—Non-Solicitation” beginning on page 168 of this proxy statement/prospectus.
Certain provisions of the Merger Agreement may discourage third parties from submitting competing proposals, including proposals that may be superior to the transactions contemplated by the Merger Agreement.
The terms of the Merger Agreement prohibit each of Pieris and Palvella from soliciting competing proposals or cooperating with persons making unsolicited takeover proposals, except in limited circumstances as described in further detail in the section titled “The Merger Agreement—Non-Solicitation.” In addition, if the Merger Agreement is terminated under specified circumstances, Pieris would be required to pay Palvella a termination fee of $1.0 million and Palvella will be required to pay Pieris a termination fee of $2.0 million. This termination fee may discourage third parties from submitting competing proposals to Pieris or its stockholders, and may cause the Pieris board of directors to be less inclined to recommend a competing proposal.
Because the lack of a public market for Palvella capital stock makes it difficult to evaluate the fair market value of Palvella capital stock, Pieris may pay more than the fair market value of Palvella capital stock and/or the stockholders of Palvella may receive consideration in the Merger that is less than the fair market value of Palvella capital stock.
The outstanding capital stock of Palvella is privately held and is not traded in any public market. The lack of a public market makes it difficult to determine the fair market value of Palvella capital stock. Because the percentage of Pieris equity to be issued to Palvella stockholders was determined based on negotiations between the parties, it is possible that the value of the Pieris common stock to be received by Palvella stockholders will be less than the fair market value of Palvella capital stock, or Pieris may pay more than the aggregate fair market value for Palvella capital stock.
The Merger may not qualify as a “reorganization” within the meaning of Section 368(a) of the Code, resulting in recognition of taxable gain or loss by Palvella stockholders in respect of their Palvella capital stock.
As discussed more fully under the section entitled “The Merger—Material U.S. Federal Income Tax Consequences of the Merger,” and subject to the qualifications and limitations set forth therein, in the opinion of Mintz and Troutman, the Merger will qualify as a “reorganization” within the meaning of Section 368(a) of the Code. Assuming the Merger so qualifies, a Palvella U.S. holder will not recognize gain or loss for U.S. federal income tax purposes upon the receipt of shares of Pieris common stock in exchange for shares of Palvella capital stock in the Merger, except with respect to cash received in lieu of a fractional share of Pieris common stock.
However, none of the parties to the Merger Agreement have sought and do not intend to seek a ruling from the IRS regarding the intended tax treatment of the Merger and, even though an opinion of counsel has been sought and obtained by Pieris and Palvella, such opinions are not binding upon the IRS or a court. Consequently, there can be no assurance that the IRS will not challenge the intended tax treatment of the Merger and, if challenged, that a court would not sustain the IRS’ position. In the event that the Merger does not qualify as a “reorganization” within the meaning of Section 368(a) of the Code, each Palvella U.S. holder would recognize gain or loss upon the exchange of shares of Palvella common stock for Pieris common stock in the Merger equal to the difference between the fair market value of the shares of Pieris common stock received in exchange for the shares of Palvella capital stock (plus any cash received in lieu of a fractional share) and such Palvella U.S. holder’s adjusted tax basis in the shares of Palvella capital stock surrendered. Each Palvella stockholder is urged to consult with his, her or its own tax advisor with respect to the tax consequences of the Merger.
The U.S. federal income tax treatment of the CVRs is unclear, and there can be no assurance that the IRS would not assert, or that a court would not sustain, a position that could result in adverse U.S. federal income tax consequences to holders of the CVRs.
The U.S. federal income tax treatment of the CVRs is unclear. There is no legal authority directly addressing the U.S. federal income tax treatment of the receipt of, and payments on, the CVRs, and there can be no assurance that the IRS would not assert, or that a court would not sustain, a position that could result in adverse U.S. federal income tax consequences to holders of the CVRs. As discussed in the section titled “Agreements Related to the Merger—CVR Agreement—Material U.S. Federal Income Tax Consequences of the Receipt of CVRs,” Pieris intends to take the position that the fair market value of the CVR cannot be reasonably ascertained on the date of the issuance of the CVRs and, accordingly, the issuance of the CVRs constitutes an “open transaction”. Accordingly, absent a change in law requiring otherwise after the date of the CVR Agreement, Pieris will not report to Pieris stockholders as a current distribution of property with respect to its stock and will instead report each future cash payment (if any) on the CVRs as a distribution by Pieris for U.S. federal income tax purposes, with each such payment being reported as a dividend to the extent of Pieris’ current or accumulated earnings and profits in the year in which such payment is made. This position may be challenged by the IRS, in which case holders of Pieris common stock could be required to recognize taxable income in respect of the CVRs at the time such CVRs are issued, without the corresponding receipt of cash as described further in the section titled “Agreements Related to the Merger—CVR Agreement—Material U.S. Federal Income Tax Consequences of the Receipt of CVRs” beginning on page 182 of this proxy statement/prospectus.
Lawsuits could delay or prevent the Merger.
Putative stockholder complaints, including stockholder class action complaints, and other complaints may be filed against Pieris, Palvella, and/or their respective boards of directors in connection with the transactions contemplated by the Merger Agreement.
The outcome of litigation is uncertain, and Pieris, Palvella, and/or their respective boards of directors may not be successful in defending against any such claims. Any such lawsuits that have been or may be filed against Pieris, Palvella, and/or their respective boards of directors could delay or prevent the Merger from becoming effective or from becoming effective within the intended timeframe, divert the attention of Pieris’ and/or Palvella’s management and employees from their respective day-to-day businesses and otherwise adversely affect their respective financial conditions.
The financial projections for Palvella included in the section entitled “The Merger—Certain Unaudited Financial Projections”, which were considered by the Pieris board of directors in evaluating the Merger and used by Pieris’ financial advisor in rendering the Stifel Opinion and performing its related financial analyses, reflect numerous variables, estimates and assumptions and are inherently uncertain. If any of these variables, estimates and assumptions prove to be wrong, such as the assumptions relating to the approval of Palvella’s product candidates, the actual results for the combined company’s business may be materially different from the results reflected in the financial projections.
As further described below in the section entitled “The Merger—Certain Unaudited Financial Projections”, in connection with the Pieris board of directors’ evaluation of the Merger, preliminary internal financial forecasts for Palvella were prepared by the management of Palvella and provided to the management of Pieris, for use by Pieris’ financial advisor, Stifel, in connection with the rendering of the Stifel Opinion and performing its related financial analyses, as described below under “The Merger—Opinion of Pieris’ Financial Advisor”. The financial forecasts and financial projections reflect numerous variables, estimates, and forecasts made by Palvella’s management at the time the initial financial forecasts were prepared by Palvella and approved by Palvella. If any of these variables, estimates and assumptions prove to be wrong, the actual results for the combined company’s business may differ materially from the results reflected in the financial projections.
The estimated probabilities of success included in the financial projections take into account a range of potential outcomes, including outcomes in which product candidates fail to achieve commercial launch due to commercial and regulatory uncertainty (including failure to obtain regulatory authorization to market the applicable product candidate), as well as economic and portfolio management decisions and competition, and these assumptions, including those with respect to regulatory approval and probability of success more broadly, are inherently uncertain and could prove inaccurate. If one or more of the Palvella product candidates do not receive marketing authorization when anticipated, for the indications anticipated, or at all, or the other assumptions reflected in the estimates as to probability of success prove untrue, the actual results of the combined company’s business will differ materially from the results reflected in the financial projections. For example, while the financial projections reflect the probability of success assessments described below in the section entitled “The Merger—Certain Unaudited Financial Projections” for each of Palvella’s product candidates, if one or both of these product candidates are not approved then actual results will differ materially, including the potential for one or both of these product candidates to generate no revenue at all.
In addition, the financial projections cover a significant period of time. This extended period was used in light of the anticipated timing for regulatory approval and the initiation of commercial sales of the Palvella product candidates. However, the risks and uncertainties regarding the financial projections, including the potential for adverse developments such as delays in obtaining or failure to obtain regulatory approvals or additional competition or changes in the competitive or regulatory landscape, increase with each successive year and the likelihood that the actual results will differ materially from the projected results increases with each successive year. The financial projections also do not reflect general business, economic, market and financial conditions and any changes in any of these conditions over the period of the projections could result in the actual results differing materially from the results reflected in the financial projections.
If the Merger is not completed, Pieris will reconsider its strategic alternatives, including dissolving and liquidating its assets, pursuing another strategic transaction, or operating its business. If the Merger is not completed, Pieris will face various risks related to its financial condition and need for capital; its ability to execute on alternative strategies; its third party agreements, licenses, and collaborations; its intellectual property; regulatory and compliance matters; and its status as a public company, all as further discussed in the Risk Factors, including this subsection titled “—Risks Related to Pieris.”
Risks Related to Pieris’ Corporate Strategy
Pieris’ business strategy depends substantially upon its ability to receive future contingent milestone and royalty payments.
Pieris’ business strategy depends substantially upon its ability to receive future milestone and royalty payments from Pfizer, Inc. (formerly Seagen), or “Pfizer”, and BP Asset XII, Inc., or “Boston Pharmaceuticals”. On March 27, 2024, Pieris announced a strategy to maximize its ability to capture the potential milestones from licensing and collaboration agreements with its partners, including Pfizer and Boston Pharmaceuticals, while maintaining the capability to consider other strategic options. The Pieris board of directors implemented a series of measures designed to extend its cash runway into at least 2027 and maximize its ability to capture the potential milestone payments. These measures included discontinuing research and development activities and conducting further workforce reduction that affect additional employees and the executive leadership team. Pieris does not have any ongoing research or development activities.
Any failure to achieve the potential future milestones or a perception that the milestones may not be achieved will materially and adversely affect Pieris and the value of the common stock.
Even if some or all of the milestones or royalties set forth in the Pfizer Agreement or Boston Pharmaceuticals Agreement are achieved, it may take significantly longer than Pieris anticipates and could require Pieris to raise additional funding in order to maintain its ability to receive payment for such milestones.
Achievement of the milestones set forth in the Pfizer Agreement and the Boston Pharmaceuticals Agreement are not guaranteed and there is significant risk that some or all of such milestones will not be achieved when anticipated, if at all. If achievement of the milestones is delayed beyond what Pieris currently anticipates, it could require Pieris to raise additional funds in order to maintain its ability to receive payment for the potential future achievement of such milestones. Sources of funds may not be available or, if available, may not be available on terms satisfactory to Pieris. Raising additional funds could be dilutive or otherwise disadvantageous to Pieris stockholders. Any delay in receipt of the potential benefit to Pieris or its stockholders resulting from achievement of such milestones, in addition to any additional uncertainty as to whether such milestones will be achieved at all, would materially and adversely affect Pieris and the value of the common stock.
Time and costs associated with winding down Pieris’ research and development activities and any return of cash to stockholders may be significant.
There are significant costs associated with winding down Pieris’ normal historic operations, such as separation of employees, termination of contracts and engagement of external consultants, all of which have and may in the future reduce its cash resources. Additionally, if the Pieris board of directors decides to issue any cash dividends to its stockholders in the future, Pieris may incur third party costs associated with the distribution of such dividends, all of which would reduce its cash resources.
If some or all of Pieris’ partners terminate its partnerships for which Pieris may be entitled to milestone payments, the Pieris board of directors may decide to pursue a dissolution and liquidation. In such an event, the amount of cash available for distribution to Pieris stockholders will depend heavily on the timing of such liquidation, as well as the amount of cash that will need to be reserved for commitments and contingent liabilities.
There can be no assurance or guarantee that Pieris will realize all or some of the milestone payments in connection with its licensing and collaboration agreements, and in the event Pieris’ partners terminate their respective licensing and collaboration agreements, the Pieris board of directors may decide to pursue a dissolution and liquidate. In such an event, the amount of cash available for distribution to Pieris stockholders will depend heavily on the timing of such decision and, with the passage of time the amount of cash available for distribution will be reduced as Pieris continues to fund its operations. In addition, if the Pieris board of directors were to approve and recommend, and Pieris stockholders were to approve, a dissolution and liquidation, Pieris would be required under Nevada corporate law to pay its outstanding debts and other obligations prior to making any distributions in liquidation to its stockholders. As a result of this requirement, a portion of Pieris’ assets may need to be reserved pending the resolution of such obligations and the timing of any such resolution is uncertain. In addition, Pieris may be subject to litigation or other claims related to a dissolution and liquidation. If a dissolution and liquidation were pursued, the Pieris board of directors, in consultation with its advisors, would need to evaluate these matters and make a determination about a reasonable amount to reserve. Accordingly, holders of Pieris common stock could lose all or a significant portion of their investment in the event of a liquidation, dissolution or winding up.
Pieris may rely on external consultants for the execution of its business strategy.
In connection with Pieris’ March 27, 2024 announcement, Pieris disclosed an additional reduction in force impacting additional employees and its executive leadership team, which was implemented in the third quarter of 2024. Pieris expects that as the number of employees is reduced, Pieris may become reliant on a limited number of external consultants for the operation of Pieris, any of whom may terminate their consultancy with Pieris at any time. The loss of some or all of Pieris’ consultants could delay or inhibit its ability to run its operations or consummate any divestitures of its remaining assets or could interfere with its ability to receive and distribute any potential milestones from Pfizer, Boston Pharmaceuticals, or any other future partner.
While Pieris has announced that it remains open to considering other strategic opportunities that might arise, there is no assurance that Pieris would be successful in pursuing any such strategic opportunities.
Since Pieris announced in July 2023 that it intends to explore engaging in one or more strategic transactions, Pieris’ strategic review process has included a focus on enhancing stockholder value, which includes the maximization of potential milestone payments Pieris is eligible to receive. Pieris’ management and the Pieris board of directors evaluated a broad spectrum of potential options, including asset in-licensing, out-licensing, royalty monetization, strategic transactions (including reverse mergers, strategic mergers, and sale), and liquidation. With the assistance of Pieris’ financial advisor, Stifel, more than 500 companies were contacted regarding a strategic transaction, and Pieris underwent a robust process to identify and negotiate with a select number of final candidates. Pieris entered into extended exclusivity with one party contemplating a strategic merger, which centered on that party’s interest in developing its clinical-stage asset cinrebafusp alfa, but after extensive diligence and negotiations, that counterparty was unable to secure adequate capitalization and offer acceptable terms. On March 27, 2024, Pieris announced a strategy that would maximize its ability to capture potential milestones from its licensing and collaboration agreements while maintaining the capability to consider other strategic opportunities, which Pieris believes offers the best opportunity to enhance stockholder value. Despite remaining open to considering other strategic opportunities that might arise, there can be no assurance that Pieris will be successful in pursuing any opportunity or that any opportunity, if pursued, will be completed on attractive terms or at all. Pieris may rely on the support of consultants and external advisors to assist in the review of strategic opportunities which may be costly. Additionally, there can be no assurance that any particular course of action, strategy to capture potential milestones or other strategy, business arrangement or transaction, or series of transactions, will be successfully pursued, consummated or lead to increased stockholder value. Such other strategies, business arrangement or transaction, or series of transactions could lead to increased costs, dilution to its existing stockholders’ percentage of ownership, or assumption of debt and liabilities.
Pieris may be treated as a "public shell" company under the Nasdaq rules which could have negative consequences, including potential Nasdaq delisting of its common stock.
Pieris common stock is currently listed on The Nasdaq Capital Market and it has no current plans to delist its common stock from Nasdaq. At the time of the Merger, Pieris will be a shell company due to its expected nominal operations and assets following the discontinuation of historical research and development efforts. Pieris anticipates that following the consummation of the Merger, the combined company will no longer be a shell company. As a result, Pieris may be treated as a “public shell” company under the Nasdaq rules. Although Nasdaq evaluates whether a listed company is a public shell company based on a facts and circumstances determination, a Nasdaq-listed company with no or nominal operations and either no or nominal assets, assets consisting solely of cash and cash equivalents, or assets consisting of any amount of cash and cash equivalents and nominal other assets is generally considered to be a public shell company. Listed companies determined to be public shell companies by Nasdaq may be subject to delisting proceedings or additional and more stringent listing criteria.
In addition, among other requirements, a minimum $1.00 per share bid price requirement for continued inclusion on the Nasdaq pursuant to Nasdaq Listing Rule 5550(a)(2), or the “Bid Price Requirement”. The closing bid price for Pieris common stock must remain at or above $1.00 per share to comply with the Bid Price Requirement for continued listing. As previously disclosed, on May 15, 2023, Pieris received a deficiency letter, or the “Notice”, from the Nasdaq Listing Qualifications Department, or the “Staff”, notifying Pieris that because the closing bid price of its common stock had fallen below $1.00 per share for 30 consecutive business days, Pieris no longer met the Bid Price Requirement.
Pursuant to Nasdaq Listing Rule 5810(c)(3)(A), or the “Compliance Period Rule”, Pieris had an initial period of 180 calendar days, or until November 13, 2023, or the “Compliance Date”, to regain compliance with the Bid Price Requirement. To regain compliance, the closing bid price of Pieris common stock had to meet or exceed $1.00 per share for a minimum of ten consecutive business days as required under the Compliance Period Rule (unless the Staff exercises its discretion to extend this ten-day period pursuant to Nasdaq Listing Rule 5810(c)(3)(H)). However, if during the compliance period Pieris common stock had a closing bid price of $0.10 or less for ten consecutive trading days, Nasdaq will issue a Staff Delisting Determination with the potential opportunity for Pieris to appeal that determination.
Since the closing bid price of Pieris common stock did not meet or exceed $1.00 per share for a minimum of 10 consecutive business days prior to the Compliance Date, Pieris requested an additional 180 calendar day compliance period on November 6, 2023 in which to regain compliance, in which Pieris provided written notice to Nasdaq of Pieris’ intention to cure the deficiency during the second compliance period, by effecting a reverse stock split, if necessary. On November 14, 2023, Pieris received a second notice from Nasdaq providing it with the additional 180 calendar days to regain compliance. If the Staff had concluded that Pieris was not able to cure the deficiency, or if Pieris did not regain compliance with the Bid Price Requirement within such additional 180 calendar day compliance period, the Staff would have provided written notification to Pieris that its common stock would have been subject to delisting. At that time, Pieris would have had the ability to appeal the Staff’s delisting determination to a Nasdaq Listing Qualifications Panel, or the “Panel”. However, the Pieris board of directors approved a reverse stock split of Pieris’ authorized, issued and outstanding shares of common stock at a ratio of 1-for-80, effective on Monday, April 22, 2024. Pieris common stock began trading on a reverse split-adjusted basis under the Pieris’ existing trading symbol, “PIRS,” at the market open on April 23, 2024. As a result, Pieris common stock traded above $1.00 for ten consecutive trading days and Pieris regained compliance with the Bid Price Requirement.
If Pieris common stock is delisted from Nasdaq, whether because Nasdaq determines it is a “public shell”, Pieris fails to maintain compliance with the Bid Price Requirement, or otherwise, or if in the future Pieris determines to delist its common stock, Pieris would expect that such securities would qualify for trading over-the-counter, or “OTC”, in the United States on a market colloquially referred to as the “Pink Sheets.” Securities quoted OTC are generally subject to lesser requirements than securities listed for trading on a U.S. national stock exchange, such as Nasdaq, including reduced corporate governance and public reporting standards. If Nasdaq should delist Pieris common stock from trading, or if in the future Pieris determines to delist its common stock, a reduction in some or all of the following may occur, each of which could have a material adverse effect on holders of its common stock: the liquidity of its common stock; the market price of the common stock; the number of institutional and general investors that will consider investing in the common stock; the number of investors in general that will consider investing in the common stock; the number of market makers in Pieris common stock; the availability of information concerning the trading prices and volume of the common stock; and the number of broker-dealers willing to execute trades in Pieris common stock. In addition to the foregoing, there are certain consequences under the Securities Act of being a public shell company, including the unavailability of Rule 144 thereunder for the resale of restricted securities and the inability to utilize Form S-8 for the registration of employee benefit plan securities.
Pieris may become involved in litigation, including securities class action litigation, that could divert management’s attention and harm Pieris’ business, and insurance coverage may not be sufficient to cover all costs and damages.
In the past, litigation, including securities class action litigation, has often followed certain significant business transactions, such as the sale of a company or announcement of any other strategic transaction, or the announcement of negative events, such as negative results from clinical trials. These events may also result in investigations by the SEC. Pieris may be exposed to such litigation even if no wrongdoing occurred. Litigation is usually expensive and diverts management’s attention and resources, which could adversely affect Pieris’ business and cash resources and its ability to execute its strategy to capture potential milestones from licensing and collaboration agreements while maintaining the ability to consider other strategic opportunities.
Any potential changes to Pieris’ leadership structure as a result of its workforce reduction and restructuring could adversely affect its business.
As a result of Pieris’ decision to conduct a workforce reduction and additional restructuring, Pieris may implement changes to its leadership and governance structure. Any personnel transition that may result could be difficult and inherently cause some loss of institutional knowledge and skills, which could negatively affect Pieris’ results of operations and financial condition. Pieris’ ability to execute its business strategies may be adversely affected by the uncertainty associated with these transitions and changes to leadership and governance structures, and the time and attention of the Pieris board of directors and management dedicated to such changes and transitions could disrupt Pieris’ business. Further, Pieris cannot guarantee that it will not face other transitions in the future. Although Pieris generally enters into employment agreements with its executives, its executive officers may terminate their employment relationship with Pieris at any time, and Pieris cannot ensure that it will be able to retain the services of any of them. Pieris’ leadership’s knowledge of its business and industry could be difficult to replace, and management turnover could negatively affect its business, growth, financial conditions, results of operations and cash flows.
Pieris may not be successful in identifying and implementing any strategic opportunities and any strategic opportunities that Pieris may consider and consummate in the future could have negative consequences.
On March 27, 2024, Pieris announced the implementation of a new strategy along with relevant cost-saving measures that are expected to extend its cash runway into at least 2027, while maximizing its ability to capture the potential milestones from its partnered 4-1BB bispecific Mabcalin protein IO assets, and allowing Pieris to consider, from time-to-time, other strategic opportunities that it believes may enhance stockholder value. If Pieris does consider or explore any strategic opportunities, there can be no assurances that any particular course of action, business arrangement or transaction, or series of transactions, will be pursued, successfully consummated or lead to increased stockholder value. Furthermore, if Pieris considers any other strategic opportunities, the process of considering such strategic opportunities may be very costly, time-consuming and complex and Pieris has incurred, and may in the future incur, significant costs related to this evaluation, such as legal and accounting fees and expenses and other related charges. Pieris may also incur additional unanticipated expenses in connection with this process. A considerable portion of these costs will be incurred regardless of whether any such course of action is implemented or transaction is completed. Any such expenses will decrease the remaining cash available for use in Pieris’ business.
Further, despite remaining open to considering strategic options and transactions that might arise, there can be no assurance that Pieris will be successful in pursuing any transaction or that any transaction, if pursued, will be completed on attractive terms or at all. Additionally, there can be no assurances that any particular course of action, strategy to capture potential milestones or other strategy, business arrangement or transaction, or series of transactions, will be successfully pursued, consummated or lead to increased stockholder value.
In addition, potential counterparties in a strategic opportunity involving Pieris may place minimal or no value on its assets and its public listing. Pieris may also not be able to adequately limit or avoid future liabilities which may impair the value of any potential transaction or present additional challenges on consummating a potential strategic opportunity. In addition, any strategic opportunities, including business combination or other transactions, that Pieris may consider and consummate in the future could have a variety of negative consequences and Pieris may implement a course of action or consummate a transaction that yields unexpected results that adversely affects its business and decreases the remaining cash available for use in its business or the execution of its strategy. Any potential transaction resulting from a strategic opportunity would be dependent on a number of factors that may be beyond Pieris’ control, including, among other things, market conditions, industry trends, the interest of third parties in a potential transaction with Pieris, maintaining its Nasdaq listing, obtaining stockholder approval and the availability of financing to third parties in a potential transaction with Pieris on reasonable terms. Any failure of such potential transaction to achieve the anticipated results could significantly impair Pieris’ ability to continue executing its current strategy or consider future strategic opportunities. In addition, speculation regarding any developments related to the consideration of strategic opportunities and perceived uncertainties related to the future of Pieris could cause its stock price to fluctuate significantly.
Even if Pieris identifies and successfully consummates any strategic opportunity, including, but not limited to, any partnership, acquisition, merger, business combination and/or divestiture, Pieris may fail to realize all of the anticipated benefits of such opportunity, those benefits may take longer to realize than expected, or Pieris may encounter integration difficulties.
Pieris’ ability to realize the anticipated benefits of any potential business combination or any other result from any potential strategic opportunity Pieris considers and decides to pursue, are highly uncertain. Any anticipated benefits will depend on a number of factors, including Pieris’ ability to integrate with any future business partner, its ability to obtain value for its existing programs, if divested, and its ability to generate future stockholder value from existing programs Pieris may continue to pursue. The process may be disruptive to Pieris’ business and the expected benefits may not be achieved within the anticipated time frame, or at all. The failure to meet the challenges involved and to realize the anticipated benefits of any potential transaction could adversely affect Pieris’ business and financial condition.
If Pieris identifies and is successful in consummating a strategic opportunity, it may be exposed to other operational and financial risks.
If Pieris identifies and decides to pursue a strategic opportunity, the negotiation and consummation of any such opportunity will require significant time on the part of Pieris’ management, and the diversion of management’s attention may disrupt its business.
Additionally, Pieris’ ability to consummate a strategic opportunity depends in part on its ability to retain certain of its remaining personnel. If Pieris is unable to successfully retain certain of its remaining personnel, Pieris is at risk of a disruption to its exploration and consummation of a strategic alternative, as well as business operations.
The negotiation and consummation of any such opportunity may also require more time or greater cash resources than Pieris anticipates and expose Pieris to other operational and financial risks, including:
|
● |
increased near-term and long-term expenditures; |
|
● |
exposure to unknown liabilities; |
|
● |
higher than expected acquisition or integration costs; |
|
● |
incurrence of substantial debt or dilutive issuances of equity securities to fund future operations; |
|
● |
write-downs of assets or goodwill or incurrence of non-recurring, impairment or other charges; |
|
● |
increased amortization expenses; |
|
● |
difficulty and cost in combining the operations and personnel of any acquired business with Pieris’ operations and personnel; |
|
● |
impairment of relationships with key suppliers or customers of any acquired business due to changes in management and ownership; |
|
● |
inability to retain key employees of Pieris or any acquired business; and |
|
● |
possibility of future litigation. |
Any of the foregoing risks could have a material adverse effect on Pieris’ business, financial condition and prospects.
Risks Related to Pieris’ Business, Financial Position, Capital Requirements, Managing Its Growth and Other Legal Compliance Matters
Pieris has incurred significant losses since its inception and anticipates that it will continue to incur losses for the foreseeable future. Pieris currently has no product revenues and no approved products and will rely on its partnered IO programs to generate revenue.
Pieris was historically a clinical-stage biopharmaceutical company. To date, Pieris has not generated any commercial sales revenue, is not profitable, and has incurred losses since its inception in 2001. For the years ended December 31, 2023 and 2022, Pieris reported net losses of $24.5 million and $33.3 million, respectively. As of June 30, 2024, Pieris had an accumulated deficit of $323.4 million. Pieris expects to continue to incur losses for the foreseeable future.
Pieris has collaborations with Pfizer and Boston Pharmaceuticals in IO. Pieris’ IO partnered programs include SGN-BB228 (PRS-346) in partnership with Pfizer, and BOS-342 (PRS-342) in partnership with Boston Pharmaceuticals, which are currently both in Phase 1 studies.
In July 2023, AstraZeneca notified Pieris of its intention to terminate the license and collaboration agreement, or the “AstraZeneca Collaboration Agreement” and on-exclusive Anticalin platform technology license agreement, or the “AstraZeneca Platform License”, which terminations became effective October 15, 2023. AstraZeneca’s decision to terminate these agreements was based on non-clinical safety findings in a 13-week toxicology study of elarekibep in non-human primates. If Pieris’ research and development efforts, including preclinical studies or clinical trials for any of its partnered drug candidates fail or produce unsuccessful results and those drug candidates do not gain regulatory approval, or if any of its drug candidates, if approved, fail to achieve market acceptance, Pieris may never become profitable. In addition, the failure of one drug candidate or program may have an adverse impact on other drug candidates and programs within Pieris’ class of Anticalin-based therapies. Even if Pieris achieves profitability in the future, Pieris may not be able to sustain profitability in subsequent periods. Pieris’ prior losses, combined with expected future losses, have had and will continue to have an adverse effect on its stockholders’ equity and working capital.
On June 28, 2024, Servier provided Pieris with a written notice of termination of the License and Collaboration Agreement between Servier, Pieris, and Pieris Pharmaceuticals GmbH, dated January 4, 2017, as subsequently amended, or the “Collaboration Agreement”. The Non-Exclusive Anticalin® Platform Technology License Agreement, between Servier, Pieris, and Pieris Pharmaceuticals GmbH, dated January 4, 2017, or the “Non-Exclusive License Agreement”, terminates upon termination of the Collaboration Agreement. The Collaboration Agreement and Non-Exclusive License Agreement will terminate effective December 27, 2024, or 180 days from the date on which Servier notified Pieris of its intent to terminate the agreements. Pursuant to these agreements, Servier and Pieris agreed to collaborate on the research, development, and commercialization of Anticalin-based therapeutics as part of Pieris’ IO franchise, including S095012 (formerly, PRS-344), a 4-1BB/PD-L1 bispecific Mabcalin® protein that was being developed worldwide by Servier. With this notice, Servier will discontinue and cease dosing in the Phase 1 clinical study of S095012. Servier's decision to terminate the agreements was based on a potential safety concern in the S095012 Phase 1 clinical studies. Pieris does not intend to pursue any further development of S095012.
On March 27, 2024, Pieris announced a strategy to maximize its ability to collect potential milestones from its licensing and collaboration agreements while maintaining the ability to consider other strategic opportunities. Pieris is not developing and commercializing products and does not anticipate seeking to develop any new products with any of its existing cash or any future milestone payments it may receive. Pieris’ failure to achieve these potential milestone payments would depress the value of Pieris. A decline in the value of Pieris could also cause its stockholders to lose all or part of their investment.
In the event Pieris determines to pursue any future product development efforts, Pieris will need substantial additional funding to continue its operations. In that case, if Pieris is unable to raise capital when needed, Pieris would be forced to delay, reduce, or eliminate such product development programs or commercialization efforts.
Pieris’ operations have consumed substantial amounts of cash since its inception. Although Pieris is not developing any drug product candidates and does not have any current plans to do so, if Pieris determines to pursue any future product development efforts, Pieris expects that it would incur significant research and development expenses and will need substantial additional funding.
Furthermore, Pieris expects to continue to incur additional costs associated with operating as a public company. Pieris may also encounter unforeseen expenses, difficulties, complications, delays and other unknown factors that may increase its capital needs and/or cause it to spend its cash resources faster than it expects.
To date, Pieris has financed its operations through a mix of equity investments from private and public investors, the incurrence of debt, grant funding and the receipt of up-front and milestone payments due under its various collaboration agreements, and Pieris may require additional financing to fund operations in order to execute its current business strategy and maintain its ability to receive some or all of the milestones from its IO partnered programs. Additional funding may not be available when or in the amounts needed, on acceptable terms, or at all.
As of the filing of this proxy statement/prospectus, Pieris will be subject to the SEC general instructions of Form S-3 known as the “baby shelf rules.” Under these instructions, the amount of funds Pieris can raise through primary public offerings of securities in any 12-month period using its registration statement on Form S-3 is limited to one-third of the aggregate market value of the shares of its common stock held by non-affiliates. Therefore, Pieris will be limited in the amount of proceeds it is able to raise by selling shares of its common stock using its Form S-3, including under the ATM Program under a sales agreement with Jefferies LLC, as amended, pursuant to which Pieris may offer and sell shares of Pieris common stock, from time to time, up to an aggregate amount of gross sales proceeds of $75.0 million, the “ATM Program”, until such time as its public float exceeds $75 million. Furthermore, if Pieris is deemed to be a shell company, the baby shelf rules, and therefore its Form S-3, would not be available to Pieris.
Pieris’ ability to secure additional funding could be significantly impacted by a multitude of events that are beyond its control, including, but not limited to, changes in the macroeconomic environment and other events affecting the stock market, including the availability of research and other information, favorable or unfavorable, published by securities or industry analysts and news agencies.
Raising capital through the sale of equity or securities convertible into equity would result in dilution to Pieris’ then-existing stockholders, which could be significant depending on the price at which Pieris may be able to sell its securities. If Pieris raises additional capital through the incurrence of indebtedness, Pieris would likely become subject to covenants restricting its business activities, and holders of debt instruments may have rights and privileges senior to those of its equity investors. In addition, servicing the interest and principal repayment obligations under debt facilities could divert funds that would otherwise be available to support research and development, clinical or commercialization activities.
If Pieris obtains capital through collaborative or licensing arrangements, these arrangements could require Pieris to relinquish rights to its Anticalin-based technology or drug candidates and could result in receipt of only a portion of the revenues associated with the potential commercialization of its partnered drug candidates.
Pieris’ international operations pose currency risks, which may adversely affect its operating results and net income.
Due to Pieris’ operations outside of the United States, Pieris is exposed to market risk related to changes in foreign currency exchange rates. Changes in the relative values of currencies occur regularly and, in some instances, could materially adversely affect Pieris’ business, its financial condition, the results of its operations or its cash flows. Pieris’ operating results may be affected by volatility in currency exchange rates and its ability to effectively manage its currency transaction risks. Pieris’ reporting currency is the U.S. dollar, however, 81% of its operating expenses and all of its revenues are recorded in non-U.S. entities. As such, Pieris’ financial statements are translated for reporting purposes as follows: (1) asset and liability accounts at year-end rates, (2) income statement accounts at weighted average exchange rates for the year and (3) stockholders’ equity accounts at historical rates. Corresponding translation gains or losses are recorded in stockholders’ equity.
Pieris incurs currency transaction risks whenever it enters into either a purchase or a sale transaction using a currency other than the euro, its functional currency, particularly in its arrangements for the purchase of supplies or licensing and collaboration agreements with partners outside of the United States. In such cases, Pieris may suffer an exchange loss because it does not currently engage in currency swaps or other currency hedging strategies to address this risk.
Pieris does not manage its foreign currency exposure in a manner that would eliminate the effects of changes in foreign exchange rates. Therefore, changes in exchange rates between these foreign currencies and the U.S. dollar will affect its revenues and expenses and could result in exchange losses in any given reporting period.
Given the volatility of exchange rates, Pieris can give no assurance that it will be able to effectively manage its currency transaction risks or that any volatility in currency exchange rates will not have an adverse effect on its results of operations.
Pieris is subject to anti-corruption laws, as well as export control laws, customs laws, sanctions laws and other laws governing its operations. If Pieris fails to comply with these laws, it could be subject to civil or criminal penalties, other remedial measures and legal expenses, which could adversely affect its business, results of operations and financial condition.
Pieris’ operations are subject to anti-corruption laws, including the Foreign Corrupt Practices Act, or “FCPA”, and other anti-corruption laws that apply in countries where Pieris does business and may do business in the future. The FCPA and these other laws generally prohibit Pieris, its officers, and its employees and intermediaries from bribing, being bribed or making other prohibited payments to government officials or other persons to obtain or retain business or gain some other business advantage. Pieris may participate in collaborations and relationships with third parties whose actions could potentially subject Pieris to liability under the FCPA or local anti-corruption laws.
There is no assurance that Pieris will be completely effective in ensuring its compliance with all applicable anti-corruption laws, including the FCPA, or other legal requirements, including Trade Control laws. If Pieris is not in compliance with the FCPA and other anti-corruption laws or Trade Control laws, it may be subject to criminal and civil penalties, disgorgement and other sanctions and remedial measures, and legal expenses, which could have an adverse impact on its business, financial condition, results of operations and liquidity. Likewise, any investigation of any potential violations of the FCPA, other anti-corruption laws or Trade Control laws by U.S. or other authorities could also have an adverse impact on Pieris’ reputation, business, results of operations and financial condition.
If Pieris fails to comply with environmental, health and safety laws and regulations that apply to it, Pieris could become subject to fines or penalties or incur costs that could harm its business.
Pieris is subject to numerous environmental, health and safety laws and regulations, including those governing laboratory procedures and the handling, use, storage, treatment and disposal of hazardous materials and wastes. Pieris’ operations have historically involved and may continue to involve the use of hazardous and flammable materials, including chemicals and biological materials. Pieris’ operations have historically involved and may continue to also produce hazardous waste products. Pieris generally contracts with third parties for the disposal of any hazardous materials and wastes. The use of these materials in Pieris’ business could result in contamination or injury, which could cause damage for which Pieris may be responsible but may not have sufficient resources to pay. Pieris also could incur significant costs associated with civil or criminal fines and penalties for failure to comply with these laws and regulations, which it may not be able to afford.
Although Pieris maintain workers’ compensation insurance for its operations in Germany to cover it for costs and expenses it may incur due to injuries to its employees resulting from the use of hazardous materials, this insurance may not provide adequate coverage against potential liabilities. Pieris does not maintain insurance for environmental liability or toxic tort claims that may be asserted against it in connection with its storage or disposal of biological, hazardous or radioactive materials.
In addition, Pieris may incur substantial costs in order to comply with current or future environmental, health and safety laws and regulations applicable to it. These current or future laws and regulations may impair Pieris’ research, development or production efforts or impact the research activities it pursues, particularly with respect to research involving human subjects or animal testing. Pieris’ failure to comply with these laws and regulations also may result in substantial fines, penalties or other sanctions, which could cause its financial condition to suffer.
Pieris may be limited in its use of its net operating loss carryforwards.
As of December 31, 2023, Pieris had net operating loss, or “NOL”, carryforwards for U.S. federal income tax purposes of $43.4 million and NOL carryforwards for state income tax purposes of $46.7 million. Tax loss carryforwards that were generated prior to December 31, 2017 expire through 2037; U.S. federal tax loss carryforwards generated after that date do not expire. State loss carryforwards expire starting in 2035. In the United States, utilization of the NOL carryforwards may be subject to a substantial annual limitation under Section 382 of the Code, and similar state provisions due to ownership change limitations that have occurred previously or that could occur in the future. These ownership changes may limit the amount of NOL carryforwards that can be utilized annually to offset future taxable income and tax, respectively. If Pieris were to lose the benefits of these loss carryforwards, its future earnings and cash resources would be materially and adversely affected. Pieris completed a Section 382 study through December 31, 2020. Based on the study, Pieris underwent an ownership change for Section 382 purposes which occurred in February 2018. As a result of the ownership change, Pieris’ NOL and tax credit carryforwards as of the ownership change dates are subject to limitation under Section 382; however, these limitations are not expected to result in any of the impacted NOL and tax credit carryforwards to expire unutilized. Any NOLs or tax credits generated after the February 2018 change are not subject to this annual limitation. However, subsequent ownership changes, as defined by Section 382, may potentially further limit the amount of NOL and tax credit carryforwards that could be utilized to offset future taxable income and tax.
As of December 31, 2023, Pieris had German corporate income tax and trade tax NOL carryforwards of approximately $187.6 million and $183.7 million, respectively, which may be used to reduce its future taxable income in Germany. Under current German laws, tax loss carryforwards may only be used to offset any relevant later assessment period (calendar year) by $1.2 million plus 60% of the exceeding taxable income and trade profit of such period and do not expire. In addition, certain transactions, including transfers of shares or interest in the loss holding entity, may result in the partial or total forfeiture of tax losses existing at that date. Partial or total forfeiture of tax losses may further occur in corporate reorganizations of the loss holding entity.
Pieris’ business and operations would suffer in the event of system failures, and its operations are vulnerable to interruption by natural disasters, terrorist activity, power loss, adverse public health events and other events beyond its control, the occurrence of which could materially harm its business and drug development efforts.
Despite the implementation of security measures, Pieris’ internal computer systems and those of its contractors and consultants are vulnerable to damage from computer viruses, hacking, ransomware, cyber-attacks, unauthorized access as well as telecommunication and electrical failures. Pieris’ information technology and other internal infrastructure systems, including corporate firewalls, servers, leased lines and connection to the Internet, face the risk of systemic failure that could materially disrupt its operations. Although Pieris has invested significant resources to enhance the security of its computer systems, there can be no assurances it will not experience unauthorized intrusions into its computer systems, or those of its contract research organizations, or “CROs”, vendors, contractors and consultants, that it will successfully detect future unauthorized intrusions in a timely manner or that future unauthorized intrusions will not result in material adverse effects on its financial condition, reputation or business prospects.
While Pieris has not experienced any such material system failure, accident or security breach to date, if such an event were to occur and cause interruptions in its operations, it could result in a material impact on Pieris’ business. In addition, certain data security breaches must be reported to affected individuals and the government, and in some cases to the media, under provisions of the federal Health Insurance Portability and Accountability Act of 1996, or “HIPAA”, as amended by the Health Information Technology for Economic and Clinical Health Act, or the “HITECH Act”, other U.S. federal and state law and requirements of non-U.S. jurisdictions, and financial penalties may also apply. If any disruption or security breach resulted in a loss of or damage to Pieris’ data or applications, or inappropriate disclosure of confidential or proprietary information, Pieris could incur liability.
Pieris is also vulnerable to accidents, electrical blackouts, labor strikes, terrorist activities, war, natural disasters, adverse public health events and other events beyond its control, and Pieris has not undertaken a systematic analysis of the potential consequences to its business as a result of all of such events and do not have an applicable recovery plan in place. Any disruption to Pieris’ operations or the operations of its collaborators from these kinds of events would likely impact its operating results and financial condition.
Although Pieris carries insurance to protect it against some losses or damages resulting from certain types of disasters, the extent of that insurance is limited in scope and amount, and Pieris cannot assure you that its insurance coverage will be sufficient to satisfy any damages and losses. Any business interruption may have a material adverse effect on Pieris’ business, financial position, results of operations, and prospects.
Pieris’ failure to comply with data protection laws and regulations could lead to government enforcement actions, private litigation and/or adverse publicity and could negatively affect its operating results and business.
Pieris is subject to data protection laws and regulations that address privacy and data security. The legislative and regulatory landscape for data protection continues to evolve, and there has been an increasing focus on privacy and data security issues with the potential to affect its business. In the United States, numerous federal and state laws and regulations, including state data breach notification laws, state health information privacy laws and federal and state consumer protection laws govern the collection, use, disclosure and protection of health-related and other personal information. Failure to comply with data protection laws and regulations could result in government enforcement actions, which could include civil or criminal penalties, private litigation and/or adverse publicity and could negatively affect its operating results and business. For example, California enacted the California Consumer Privacy Act, or “CCPA”, which went into effect in January 2020. The CCPA creates data privacy obligations for covered businesses and establishes data privacy rights for California residents, including the right to opt out of certain disclosures of their information. The CCPA provides for civil penalties for violations, as well as a private right of action for data breaches. Additionally, California voters passed the California Privacy Rights Act, or “CPRA”, which became effective January 1, 2023. The CPRA significantly amends the CCPA, and imposes additional data protection obligations on covered businesses, including additional consumer rights processes, limitations on data uses, new audit requirements for higher risk data, and opt outs for certain uses of sensitive personal information. The CPRA also established a new regulatory authority, the California Privacy Protection Agency, which is tasked with enacting new regulations under the CPRA and will have expanded enforcement authority, which may result in increased privacy and information security enforcement in California. In addition to California, more U.S. states are enacting similar consumer privacy legislation, increasing compliance complexity and increasing risks of failures to comply. As of 2023, Virginia, Colorado, Connecticut and Utah enacted similar comprehensive data protection laws. Additional consumer privacy laws have also been enacted in Delaware, Indiana, Iowa, Montana, New Jersey, Oregon, Tennessee, and Texas, which laws will take effect over the next three years.
Numerous other countries have, or are developing, laws governing the collection, use and transmission of personal information as well. For example, the European Union’s General Data Protection Regulation, or “GDPR”, which took effect in 2018, imposed a broad data protection framework that expanded the scope of data protection law across the European Union and European Economic Area, or “EEA”, and can apply to non-EU entities that process, or control the processing of, personal data relating to individuals located in the EEA, including clinical trial data. The GDPR sets out a number of requirements that must be complied with when handling the personal data of EEA-based data subjects, including: providing expanded disclosures about how their personal data will be used; higher standards for organizations to demonstrate that they have obtained valid consent or have another legal basis to justify their data processing activities; the obligation to appoint data protection officers in certain circumstances; new rights for individuals to be “forgotten” and rights to data portability; an expansion of data subject rights with respect to access and control over their personal data (e.g., access requests); requirements for demonstrating compliance through policies, procedures, training and audit; and a new mandatory data breach reporting and notification regime. In particular, medical or health data, genetic data and biometric data are all classified as “special category” data under the GDPR and are therefore subject to additional compliance obligations. Further, EEA member states have a broad right to impose additional conditions—including restrictions—on these data categories in connection with permitted derogations from the requirements of the GDPR mainly in regard to specific processing situations (including special category data and processing for scientific or statistical purposes). Pieris is subject to the GDPR and the German federal data privacy law, the Bundesdatenschutzgesetz, and it is subject to the regulatory authority of the Bavarian data protection authority, the BayLDA. As the EU states continue to reframe their national legislation to harmonize with the GDPR, Pieris will monitor compliance with all relevant EU member states’ laws and regulations, including where permitted derogation from the GDPR are introduced.
Pieris is also subject to evolving EU laws on data export since it transfers data to countries outside of the EEA, including the United States and United Kingdom, to itself or third parties. The GDPR only permits exports of data outside of the EEA where there is a suitable data transfer mechanism in place to safeguard personal data (e.g., the EU Commission approved Standard Contractual Clauses). On July 16, 2020, the Court of Justice of the EU, or the “CJEU”, issued a landmark opinion in the case Maximilian Schrems vs. Facebook (Case C-311/18) (Schrems II). This decision calls into question certain data transfer mechanisms as between the EU member states and the United States. The CJEU is the highest court in Europe and the Schrems II decision heightens the burden on data importers to assess U.S. national security laws on their business and to evaluate risks of potential fines and penalties and/or data transfers from the EU being halted. On July 10, 2023, the EU Commission adopted an adequacy decision for a new mechanism for transferring data from the EU to the United States – the EU-US Data Privacy Framework, or the “Framework”. The Framework provides EU individuals with several new rights, including the right to obtain access to their data, or obtain correction or deletion of incorrect or unlawfully handled data. The adequacy decision followed the signing of an executive order introducing new binding safeguards to address the points raised in the Schrems II decision. Notably, the new obligations were geared to ensure that data can be accessed by U.S. intelligence agencies only to the extent necessary and proportionate and to establish an independent and impartial redress mechanism to handle complaints from Europeans concerning the collection of their data for national security purposes. The EU Commission will continually review developments in the United States along with its adequacy decision. Adequacy decisions can be adapted or even withdrawn in the event of developments affecting the level of protection in the applicable jurisdiction. Future actions of EU data protection authorities are difficult to predict. Reliance on the Framework to enable cross-border transfers without certain contractual and other representations is dependent upon certification to the Framework, which Pieris has not yet done.
If Pieris has to rely on third parties to carry out services for it, including processing personal data on its behalf, Pieris is required under GDPR to enter into contractual arrangements to help ensure that these third parties only process such data according to Pieris’ instructions and have sufficient security measures in place. Any security breach or non-compliance with its contractual terms or breach of applicable law by such third parties could result in enforcement actions, litigation, fines and penalties or adverse publicity and could harm consumer confidence in Pieris, which would have an adverse impact on its reputation and business. Any contractual arrangements requiring the processing of personal data from the EEA to Pieris in the United States will require greater scrutiny and assessments as required under Schrems II and may have an adverse impact on cross-border transfers of personal data, or increase costs of compliance. The GDPR provides an enforcement authority to impose large penalties for noncompliance, including the potential for fines of up to €20 million or 4% of total global turnover from the preceding fiscal year of the noncompliant company, whichever is greater.
Applicable data privacy and data protection laws may conflict with each other, and by complying with the laws or regulations of one jurisdiction, Pieris cannot be assured of compliance with the laws or regulations of another jurisdiction. Despite Pieris’ efforts, it may not have fully complied in the past and may not in the future. That could require Pieris to incur significant expenses, which could significantly affect its business. Failure to comply with data protection laws may expose Pieris to risk of enforcement actions taken by data protection authorities or other regulatory agencies, private rights of action in some jurisdictions, and potential significant fines and penalties if it is found to be non-compliant.
Furthermore, the number of government investigations related to data security incidents and privacy violations continues to increase and government investigations typically require significant resources and generate negative publicity, which could harm Pieris’ business and reputation.
U.S. tax legislation and future changes to applicable U.S. or foreign tax laws and regulations may have a material adverse effect on Pieris’ business, financial condition and results of operations.
Pieris is subject to income and other taxes in the United States and foreign jurisdictions. Changes in laws and policy relating to taxes or trade may have an adverse effect on its business, financial condition and results of operations. This proxy statement/prospectus does not discuss any such tax legislation or changes to tax laws and regulations, or the manner in which it might affect Pieris or purchasers of its securities. Pieris urges its investors to consult with their own legal and tax advisors with respect to such legislation and the potential tax consequences of investing in its securities.
Pieris is also subject to different tax regulations in each of the jurisdictions where it conducts its business or where its management is located. Pieris expects the scope and extent of regulation in the jurisdictions in which it conducts its business, or where its management is located, as well as regulatory oversight and supervision, to generally continue to increase. Generally, future changes in applicable U.S. or foreign tax laws and regulations, or their interpretation and application could have an adverse effect on Pieris’ business, financial conditions and results of operations.
Pieris could be subject to product liability lawsuits based on the use of its drug candidates in clinical testing or, if obtained, following its products’ marketing approval and commercialization. If product liability lawsuits are brought against Pieris, it may incur substantial liabilities.
Pieris could be subject to product liability lawsuits if any drug candidate it develops allegedly causes injury or is found to be otherwise unsuitable for human use during product testing, manufacturing, marketing or sale. Any such product liability claim may include allegations of defects in manufacturing, defects in design, a failure to warn of dangers inherent in the product, negligence, strict liability and a breach of warranties. Claims could also be asserted under state consumer protection acts. If Pieris cannot successfully defend itself against these claims, it will incur substantial liabilities. Even successful defense would require significant financial and management resources.
Regardless of merit or eventual outcome, liability claims may result in, among other things, reduced resources of Pieris’ management to pursue its business strategy, injury to its reputation and significant negative media attention, significant costs to defend the related litigation, substantial monetary awards to clinical trial participants or patients, and increased insurance costs.
While Pieris currently carries insurance in an amount and on terms and conditions that are customary for similarly situated companies and that are satisfactory to the Pieris board of directors, its insurance coverage may not reimburse Pieris or may not be sufficient to reimburse Pieris for any expenses or losses it may suffer.
Risks Related to the Development and Commercialization of Pieris’ Drug Candidates
Although Pieris has in the past depended heavily on the success of its drug candidates and programs, it does not have any product candidates currently in active development. Future clinical trials, if any, may not be successful and Pieris cannot be certain that it will receive regulatory approvals or be able to successfully commercialize its products even if it receives regulatory approvals.
Pieris currently has no products that are approved for commercial sale, and do not have plans to independently develop any drug product candidates. All of Pieris’ IO drug candidates are being developed in partnership with its collaborators. Accordingly, Pieris’ business is currently substantially dependent on the successful development, clinical testing, regulatory approval and commercialization of its partnered programs, which may never occur. For example, in July 2023, AstraZeneca notified Pieris of its intention to terminate the AstraZeneca Collaboration Agreement and the AstraZeneca Platform License, which terminations became effective October 15, 2023. AstraZeneca’s decision to terminate these agreements was based on non-clinical safety findings in a 13-week toxicology study of elarekibep in non-human primates. Additionally, on June 28, 2024, Servier provided Pieris with a written notice of termination of the Collaboration Agreement. The Non-Exclusive License Agreement with Servier terminates upon termination of the Collaboration Agreement. The Collaboration Agreement and Non-Exclusive License Agreement will terminate effective December 27, 2024 and Servier will discontinue and cease dosing in the Phase 1 clinical study of S095012. Servier's decision to terminate the agreements was based on a potential safety concern in the S095012 Phase 1 clinical studies. Pieris intends to review the safety data from the S095012 Phase 1 clinical study to understand the implications of the data. Pieris does not intend to pursue any further development of S095012.
In order for Pieris to achieve potential milestones or royalties from its partnered programs, its partners must complete some or all of the following activities, any one of which may not be successfully completed:
|
● |
conduct additional preclinical and clinical development with successful outcomes; |
|
● |
manage preclinical, manufacturing and clinical activities; |
|
● |
obtain regulatory approval from the FDA and other comparable foreign regulatory authorities; |
|
● |
establish manufacturing relationships for the clinical and post-approval supply of the applicable drug candidate in compliance with all regulatory requirements; |
|
● |
build a commercial sales and marketing team, either internally or by contract with third parties; |
|
● |
establish and maintain patent and trade secret protection or regulatory exclusivity for Pieris’ product candidates; |
|
● |
develop and implement marketing strategies for successful commercial launch of Pieris’ product candidates, if and when approved; |
|
● |
secure acceptance of Pieris’ products, if and when approved, by patients, from the relevant medical communities and from third-party payors; |
|
● |
compete effectively with other therapies; |
|
● |
establish and maintain adequate health care coverage and reimbursement; |
|
● |
ensure continued compliance with any post-marketing requirements imposed by regulatory authorities, including any required post-marketing clinical trials or the elements of any post-marketing risk evaluation and mitigation strategy, or “REMS”, that may be required by the FDA or comparable requirements in other jurisdictions to ensure the benefits of the product outweigh its risks; |
|
● |
maintain continued acceptable safety profile of the product candidates following approval; and |
|
● |
invest significant additional cash in each of the above activities. |
If Pieris’ partners are unable to address one or more of these factors in a timely manner or at all, there could be significant delays in the successful commercialization of, or an inability to successfully commercialize, Pieris’ product candidates, which would materially harm its business. If regulatory approvals are not received for one or more of Pieris’ product candidates, Pieris may not be able to continue its operations. Even if Pieris’ partners successfully obtain regulatory approvals to manufacture and market its product candidates, revenues will be dependent, in part, upon the size of the markets in the territories for which regulatory approval is obtained and where there are commercial rights, competitors’ products in the same markets, market acceptance, and other factors. If the markets for patient subsets that are targeted are not as significant as Pieris estimates, significant revenues may not be generated from sales of such products, if approved.
Clinical testing of Pieris’ IO partnered programs is ongoing, while clinical testing for other IO programs, for example the preclinical programs with Pfizer, have not yet commenced, and the results of any future clinical trials or preclinical studies of these programs, if unsuccessful, could lead to abandonment of the development of those drug candidates. If studies of these drug candidates produce unsuccessful results and its partners are forced or elect to cease their development, Pieris’ business and prospects could be substantially harmed.
Preclinical and clinical testing of drug candidates that has been conducted to date or will be conducted in the future may not have been or may not be performed in compliance with applicable regulatory requirements, which could lead to increased costs or material delays for their further development.
Given the complexity, as well as the uncertainty inherent in preclinical and other nonclinical studies and clinical trials, and because of Pieris’ limited operating experience, Pieris may discover that its own development activities are not in compliance with applicable regulatory requirements or are otherwise deficient, and therefore, determine that the development of its drug candidates on the basis of those trials and studies is not warranted or will be delayed.
Pieris has entered into license, partnership and option arrangements, such as with Pfizer and Boston Pharmaceuticals, relating to certain drug candidates and Pieris may continue to do so in the future. Under some of these arrangements, the development of some of those drug candidates has been, or in the future may be, conducted wholly by such partners or third parties with which the partners contract. As a result, Pieris has not been or may not be closely involved with or have any control over those development activities. Although some of Pieris’ partners have provided information regarding those drug candidates and the related studies conducted to date, including data that has been included in Pieris’ Annual Reports on Form 10-K, Pieris has not received and may not receive in the future, comprehensive information regarding all of those development activities, including the raw data from certain studies that have been conducted, information regarding the design, procedural implementation and structure and information regarding the manufacture of the drug candidates used in the studies. Because Pieris may have limited or no input on the development of these drug candidates, Pieris may discover that all or certain elements of the trials and studies its partners have performed have not been, or may not in the future be, in compliance with applicable regulatory standards or have otherwise been or may be deficient, and that advancement of the development of these drug candidates on the basis of those trials and studies is not warranted.
Further, the majority of Pieris’ development activities for each of its drug candidates, and any future clinical trials, have been, or may in the future be conducted in whole or in part outside of the United States, including in Europe, Australia or Asia. Pieris’ partners may also conduct future development activities in other countries or regions. As a result, although those studies may meet the standards of applicable foreign regulatory bodies, the structure and design of those clinical trials and preclinical studies may not meet applicable FDA requirements and also may not meet the requirements of the applicable regulatory authorities in other foreign countries in which Pieris desires to pursue marketing approval.
If the studies conducted by Pieris or its partners or collaborators do not comply with applicable regulatory requirements or are otherwise not eligible for continued development in the United States or abroad, then new studies may be required in order to progress the development of its drug candidates. Pieris’ partners may not have the funding or other resources to conduct or complete these additional studies, which would severely delay or prevent the development plans for these drug candidates and their commercialization. Any such deficiency and delay in the development of these drug candidates could significantly harm Pieris’ business plans, product revenues and prospects.
Clinical drug development involves a lengthy and expensive process with uncertain outcomes, clinical trials are difficult to design and implement, and any of Pieris’ partners' clinical trials could produce unsuccessful results or fail at any stage in the process.
Clinical trials conducted on humans are expensive and can take many years to complete, and outcomes are inherently uncertain. Failure can occur at any time during the process. Additionally, any positive results of preclinical studies and early clinical trials of a drug candidate may not be predictive of the results of later-stage clinical trials, such that drug candidates may reach later stages of clinical trials and fail to show the desired safety and efficacy traits despite having shown indications of those traits in preclinical studies and early-stage clinical trials. A number of companies in the biopharmaceutical industry have suffered significant setbacks in advanced clinical trials due to lack of efficacy or adverse safety profiles, notwithstanding promising results in earlier phases of the trials. Therefore, the results of any ongoing or future clinical trials Pieris’ partners conduct may not be successful.
For example, in June 2023, AstraZeneca communicated to Pieris its decision to discontinue and cease dosing in the ongoing clinical studies of elarekibep. This decision was based on lung findings from a non-clinical 13-week GLP toxicology study with dry powder inhaler-formulated elarekibep, which did not support long-term use and progression to later-stage development. The 13-week non-human primates study included three active dose cohorts. AstraZeneca concluded that there were no clinical observations across any of the doses but that there were respiratory tract pathology findings. These findings included inflammation-mediated lung tissue damage, which did not appear to be dose related. AstraZeneca’s decision was made independent of any data from the Phase 2a study. In July 2023, AstraZeneca notified Pieris of its intention to terminate the AstraZeneca Collaboration Agreement and the AstraZeneca Platform License, which terminations became effective October 15, 2023. AstraZeneca’s decision to terminate these agreements was based on non-clinical safety findings in a 13-week toxicology study of elarekibep in non-human primates.
Additionally, on June 28, 2024, Servier provided Pieris with a written notice of termination of the Collaboration Agreement. The Non-Exclusive License Agreement with Servier terminates upon termination of the Collaboration Agreement. The Collaboration Agreement and Non-Exclusive License Agreement will terminate effective December 27, 2024 and Servier will discontinue and cease dosing in the Phase 1 clinical study of S095012. Servier's decision to terminate the agreements was based on a potential safety concern in the S095012 Phase 1 clinical studies. Pieris intends to review the safety data from the S095012 Phase 1 clinical study to understand the implications of the data. Pieris does not intend to pursue any further development of S095012.
Clinical trials may also be delayed, suspended or prematurely terminated for a variety of other reasons, such as:
|
● |
delay or failure in reaching agreement with the FDA or a comparable foreign regulatory authority on a trial design; |
|
● |
delay or failure in obtaining authorization to commence a trial, including approval from the appropriate institutional review board, or “IRB” to conduct testing of a candidate on human subjects, or inability to comply with conditions imposed by a regulatory authority regarding the scope or design of a clinical trial; |
|
● |
inability, delay or failure in identifying and maintaining a sufficient number of trial sites, many of which may already be engaged in other clinical programs; |
|
● |
delay or failure in recruiting and enrolling suitable volunteers or patients to participate in a trial; |
|
● |
delay or failure in developing and validating companion diagnostics, if they are deemed necessary, on a timely basis; |
|
● |
failure of trial participants to complete a trial or return for post-treatment follow-up; |
|
● |
inability to monitor trial participants adequately during or after treatment; |
|
● |
clinical sites and investigators deviating from trial protocols, failing to conduct the trial in accordance with regulatory requirements or dropping out of a trial; |
|
● |
failure to initiate or delay of or inability to complete a clinical trial as a result of a clinical hold imposed by the FDA or comparable foreign regulatory authority due to observed safety findings or other reasons; |
|
● |
negative or inconclusive results in Pieris’ clinical trials, and a decision to or regulators’ requirement that additional non-clinical studies or clinical trials be conducted or that one or more of its partnered product development programs be abandoned; or |
|
● |
inability to manufacture sufficient quantities of a drug candidate of acceptable quality for use in clinical trials. |
Further, Pieris’ partners may also encounter delays if a clinical trial is suspended or terminated by Pieris, by any IRB or ethics committee, by a Data Safety Monitoring Board, or by the FDA, European Medicines Agency, or “EMA”, the Medicines and Healthcare Products Regulatory Agency, or “MHRA”, or other regulatory authority. A suspension or termination may occur due to a number of factors, including failure to conduct the clinical trial in accordance with regulatory requirements, inspection of the clinical trial operations or trial site by the FDA, EMA, MHRA or other regulatory authorities, exposing participants to health risks caused by unforeseen safety issues or adverse side effects, development of previously unseen safety issues, failure to demonstrate a benefit from using a drug candidate or changes in governmental regulations or administrative actions. Pieris cannot predict with any certainty the schedule for commencement or completion of any currently ongoing, planned or future clinical trials.
If Pieris’ partners experience delays in the commencement or completion of, or suspension or termination of, any clinical trial for its drug candidates, the commercial prospects of the drug candidate could be harmed, and ability to and ability to realize milestones or royalties. The occurrence of any of these events could harm Pieris’ business, financial condition, results of operations and prospects significantly.
Risks Related to Pieris’ Dependence on Third Parties
Disagreements with respect to the commercial terms of Pieris’ sales, licensing, purchase or manufacturing agreements may limit its commercial success.
The rights and obligations of the partners to which Pieris may license its Anticalin-based technology are governed by the licensing and collaboration agreements Pieris enters into with those partners. Pieris’ collaborators may include assumptions, understandings or agreements that are not included in Pieris’ agreements with them, or that are inaccurately or incompletely represented by their terms. In addition, key terms in such agreements may be misunderstood or contested, even when Pieris and the other party previously believed that they both had a mutual understanding of such terms.
Any differences in interpretation or misunderstandings between Pieris and other parties may result in substantial costs, and may negatively impact its revenues and operating results. Partners may fail to develop the drug candidates with the diligence or under the timeline or in the manner Pieris anticipated, and results may differ from the terms upon which Pieris had agreed. Resolution of these problems may entail costly and lengthy litigation or dispute resolution procedures. In addition, there is no guarantee that Pieris will prevail in any such dispute or, if Pieris does prevail, that any remedy it receives, whether legal or otherwise, will adequately redress the harm Pieris has suffered. The delays and costs associated with such disputes may themselves harm Pieris’ business and reputation.
Pieris depends on third parties and may to continue to license or collaborate with third parties, and events involving these strategic partners or any future collaboration could delay or prevent development or commercialization of drug products.
Pieris’ business strategy, along with its short- and long-term operating results, depend in part on its ability to execute on existing strategic collaborations and to license or partner with new strategic partners. Pieris has entered into and may in the future to enter into collaborative arrangements with both U.S.-based and foreign pharmaceutical and drug development companies, which will lead or otherwise collaborate with Pieris or assist it in the development, manufacturing and marketing of its drug products. Pieris believes collaborations allow Pieris to leverage its resources and technologies and Pieris may derive some revenues from research and development fees, license fees, milestone payments, and royalties from its collaborative partners.
Pieris’ prospects, therefore, may depend to some extent upon its ability to attract and retain collaborative partners. Pieris has limited control over the amount and timing of resources that its current collaborators or any future collaborators devote to its collaborations or potential products, in particular with respect to its collaborations with Boston Pharmaceuticals for the development of BOS-342, with Pfizer for the development of SGN-BB228 and other programs. These collaborators may breach or terminate their agreements with Pieris or otherwise fail to conduct their collaborative activities successfully and in a timely and reasonable manner. Termination of these agreements or reduction or elimination of Pieris’ rights under these agreements may result in Pieris having to negotiate new, amended or reinstated agreements with less favorable terms, or cause Pieris to lose its rights under these agreements, including its rights to important intellectual property or technology. Further, Pieris’ collaborators may not develop or commercialize products that arise out of its collaborative arrangements or devote sufficient resources to the development, manufacturing, marketing or sale of these products. In addition, Pieris’ collaborative partners may have the right to guide strategy regarding prosecution of relevant patent applications, abandon research projects and/or terminate applicable agreements, including funding obligations, prior to or upon the expiration of the agreed-upon research terms.
Pieris’ collaborators may also decide to terminate these agreements based on findings in its clinical trials. For example, in July 2023, AstraZeneca notified Pieris of its intention to terminate the AstraZeneca Collaboration Agreement and the AstraZeneca Platform License, effective October 15, 2023. AstraZeneca’s decision to terminate the AstraZeneca Agreements was based on non-clinical safety findings in a 13-week toxicology study of elarekibep in non-human primates.
Additionally, on June 28, 2024, Servier provided Pieris with a written notice of termination of the Collaboration Agreement. The Non-Exclusive License Agreement with Servier terminates upon termination of the Collaboration Agreement. The Collaboration Agreement and Non-Exclusive License Agreement will terminate effective December 27, 2024 and Servier will discontinue and cease dosing in the Phase 1 clinical study of S095012. Servier's decision to terminate the agreements was based on a potential safety concern in the S095012 Phase 1 clinical studies. Pieris intends to review the safety data from the S095012 Phase 1 clinical study to understand the implications of the data. Pieris does not intend to pursue any further development of S095012.
By entering into such collaborations, Pieris may forego opportunities to collaborate with other third parties who do not wish to be associated with its existing third-party strategic partners. In the event of termination of a collaboration agreement, termination negotiations may result in less than favorable terms.
There can be no assurance that Pieris will be successful in establishing collaborative arrangements on acceptable terms or at all, that collaborative partners will not terminate funding before the completion of projects, that Pieris’ collaborative arrangements will result in successful product commercialization, or that Pieris will derive any revenues from such arrangements. Potential collaborators may reject collaborations based upon their assessment of Pieris’ financial, regulatory or intellectual property position and its internal capabilities. Additionally, the negotiation, documentation and implementation of collaborative arrangements are complex and time-consuming. Any discussions with potential collaborators may not lead to new collaborations on favorable terms and may have the potential to provide collaborators with access to Pieris’ key intellectual property rights.
Pieris’ success depends in part on the efforts of its current and possible future collaborators, who will likely have substantial control and discretion over the continued development and commercialization of drug candidates that are the subject of its collaborations.
Pieris’ current collaborators and future collaborators will have significant discretion in determining the effort and amount of resources that they dedicate to its collaborations. Pieris’ collaborators may determine not to proceed with clinical development or commercialization of a particular drug candidate for a number of reasons that are beyond Pieris’ control, even under circumstances where it might have continued such a program, currently including BOS-342 and SGN-BB228. In addition, Pieris’ rights to receive milestone payments and royalties from its collaborators will depend in part on its collaborators’ abilities to establish the safety and efficacy of its drug candidates, obtain regulatory approvals and achieve market acceptance of products developed from its drug candidates. Pieris may also depend on its collaborators to manufacture clinical scale quantities of some of its drug candidates and, possibly, for commercial scale manufacture, distribution, marketing and sales. Pieris’ collaborators may not be successful in manufacturing its drug candidates or successfully commercializing them.
Pieris faces additional risks in connection with its existing and future collaborations, including the following:
|
● |
Pieris’ collaborators may develop and commercialize, either alone or with others, products that are similar to or competitive with the products that are the subject of the collaboration with Pieris; |
|
● |
Pieris’ collaborators may underfund, not commit sufficient resources to, or conduct in an unsatisfactory manner the development, testing, marketing, distribution or sale of Pieris’ drug candidates; |
|
● |
Pieris’ collaborators may not properly maintain or defend its intellectual property rights or utilize Pieris’ proprietary information in such a way as to invite litigation that could jeopardize or potentially invalidate Pieris’ intellectual property or proprietary information or expose Pieris to potential liability; |
|
● |
Pieris’ collaborators may encounter conflicts of interest, changes in business strategy or other business issues that could adversely affect their willingness or ability to fulfill their obligations to Pieris (for example, pharmaceutical and biotechnology companies historically have re-evaluated their priorities following mergers and consolidations, which have been common in recent years in these industries); |
|
● |
Pieris does not control the conduct and communications of Pieris’ collaborators, and, thus, Pieris is subject to the risk that their actions may negatively impact Pieris’ reputation and potentially harm Pieris’ business; |
|
● |
disputes may arise between Pieris and its collaborators delaying or terminating the research, development, manufacture or commercialization of its drug candidates, resulting in significant litigation or arbitration that could be time-consuming and expensive, or causing collaborators to act in their own self-interest and not in the interest of Pieris stockholders; |
|
● |
Pieris might not have the financial or human resources to meet its obligations or take advantage of its rights under the terms of its existing and future collaborations; and |
|
● |
Pieris’ existing collaborators may exercise their respective rights to terminate their collaborations with Pieris without cause, in which event, Pieris does not currently expect to be able to complete development and commercialization of such drug candidates on its own. |
Pieris’ collaborative relationships may not produce the financial benefits that it is anticipating, which could cause its business to suffer.
Part of Pieris’ strategy is to partner with, or out-license selective products to, other pharmaceutical companies in order to mitigate the cost of developing a drug through clinical trials to commercialization. For example, in July 2023, AstraZeneca notified Pieris of its intention to terminate the AstraZeneca Collaboration Agreement and the AstraZeneca Platform License, effective October 15, 2023. AstraZeneca’s decision to terminate the AstraZeneca Agreements was based on non-clinical safety findings in a 13-week toxicology study of elarekibep in non-human primates. Additionally, on June 28, 2024, Servier provided Pieris with a written notice of termination of the Collaboration Agreement. The Non-Exclusive License Agreement with Servier terminates upon termination of the Collaboration Agreement. The Collaboration Agreement and Non-Exclusive License Agreement will terminate effective December 27, 2024 and Servier will discontinue and cease dosing in the Phase 1 clinical study of S095012. Servier's decision to terminate the agreements was based on a potential safety concern in the S095012 Phase 1 clinical studies. Pieris intends to review the safety data from the S095012 Phase 1 clinical study to understand the implications of the data. Pieris does not intend to pursue any further development of S095012. If Pieris’ collaboration with other similar partners is not successful, its future revenues and business will be harmed.
Pieris may not receive any further milestone, royalty or license payments under its current collaborations.
Although Pieris has received upfront, milestone and other payments to date under its current drug development collaborations, it may not receive any royalty payments or additional license and milestone fees under such agreements. In general, Pieris’ receipt of milestone, royalty or license payments depends on many factors, including whether its collaborators want and are able to continue to pursue potential drug candidates, intellectual property issues, the approval of biosimilars, unforeseen complications in the development or commercialization process, and the ultimate commercial success of the drugs.
Risks Related to Pieris’ Intellectual Property
If Pieris breaches any of the agreements under which it licenses from third parties the intellectual property rights or commercialization rights to its drug candidates, particularly its license agreements with TUM and Kelun, Pieris could lose license rights that are important to its business and its operations could be materially harmed.
Pieris in-licenses significant intellectual property related to its Anticalin platforms from Technische Universität München, or “TUM”. Under the terms of the research and licensing agreement by and between Pieris and TUM, or the “TUM License”, TUM assigns to Pieris certain materials and records resulting from the research. Pieris retains rights to inventions made by its employees, and TUM assigns to Pieris all inventions made under the agreement jointly by Pieris’ employees and TUM personnel, provided that Pieris’ employees have made a certain inventive contribution. With respect to all other inventions made in the course of the research, TUM grants to Pieris worldwide exclusive license rights under patents and patent applications claiming such inventions. TUM retains rights to practice these inventions for research and teaching purposes. Pieris bears the costs of filing, prosecuting and maintaining the patents assigned or licensed to Pieris under the TUM License.
As consideration for the assignments and licenses, Pieris is obliged to pay milestone payments to TUM on development of its proprietary products claimed by patents assigned or licensed to Pieris by TUM. Pieris is also obliged to pay low single-digit royalties, including annual minimum royalties, on the sales of such products. Should Pieris grant licenses or sublicenses to those patents to third parties, Pieris is obliged to pay to TUM certain undisclosed fees as a function of out-licensing revenues in connection with those patents, or “Out-License Fees”, where such Out-License Fees are creditable against annual license payments to TUM. Pieris’ payment obligations are reduced by its proportionate contribution to a joint invention. Payment obligations terminate on expiration or annulment of the last patent covered by the TUM License that covers a proprietary product or is sublicensed, as applicable.
Pieris and TUM initiated discussions in the second quarter of 2018, and may enter into an amendment to the license agreement in the future, to clarify, expand and restructure the TUM License, including the parties’ obligations under such license agreement. The contemplated amendment relates to revised commercial terms. Pieris recorded the probable expected impact of the amendment in research and development expense in 2019, although the final expense could be different than what Pieris currently has recorded.
In connection with Pieris’ efforts to develop multispecific Anticalin-based proteins designed to engage immunomodulatory targets, during the second quarter of 2017, Pieris entered into a license and transfer agreement, or the “Kelun Agreement” with Sichuan Kelun-Biotech Biopharmaceutical Co. Ltd., or “Kelun”. Under the Kelun Agreement, Kelun has granted to Pieris a non-exclusive worldwide license (with the right to sublicense) under certain intellectual property owned or controlled by Kelun to research, develop, manufacture and commercialize bi- and multi-specific fusion proteins that include an antibody developed by Kelun specific for an undisclosed target and one or more Anticalin proteins.
In addition to the TUM License and the Kelun Agreement, Pieris has other in-license agreements and may seek to enter into additional agreements with other third parties in the future granting similar license rights with respect to other potential drug candidates. If Pieris fails to comply with any of the conditions or obligations or otherwise breach the terms of the TUM License, the Kelun Agreement or any future license agreement Pieris may enter on which its business or drug candidates are dependent, TUM, Kelun or other licensors may have the right to terminate the applicable agreement in whole or in part and thereby extinguish its rights to the licensed technology and intellectual property and/or any rights Pieris has acquired to develop and commercialize certain drug candidates, including, with respect to the TUM License and Kelun Agreement, its Anticalin-based drug therapies. Under the TUM License, Pieris can terminate the licenses to any or all licensed patents upon specified advance notice to TUM. TUM may terminate the license provisions of the agreement only for cause. Termination of the TUM License does not terminate Pieris’ rights in patents assigned to it but would terminate its rights to patents licensed to it under the agreement. The loss of the rights licensed to Pieris under its license agreement with TUM or Kelun Agreement, or any future license agreement that it may enter granting it rights on which its business or drug candidates are dependent, would eliminate its ability to further develop the applicable drug candidates and may materially harm its business, prospects, financial condition and results of operations.
If Pieris’ efforts to protect the proprietary nature of the intellectual property related to its technologies are not adequate, Pieris may not be able to compete effectively and its business could be harmed.
Pieris relies upon a combination of patents, trade secret protection and confidentiality agreements, or “CDA” to protect the intellectual property related to its technologies. Any disclosure to, or misappropriation by, third parties of Pieris’ proprietary information could enable competitors to quickly duplicate or surpass its technological achievements, thus eroding any competitive advantage Pieris may derive from the proprietary information.
The strength of patents in the biotechnology and pharmaceutical fields can be uncertain and involve complex legal and scientific questions. No consistent policy regarding the breadth of claims allowed in patents has emerged to date in the United States. Accordingly, Pieris cannot predict the breadth of claims that may be allowed or enforced, or that the scope of any patent rights could provide a sufficient degree of protection that could permit Pieris to gain or keep its competitive advantage with respect to these products and technologies. For example, Pieris cannot predict:
|
● |
the degree and range of protection any patents will afford Pieris against competitors, including whether third parties will find ways to make, use, sell, offer to sell or import competitive products without infringing its patents; |
|
● |
if and when patents will be issued; |
|
● |
how laws in the various jurisdictions, such as the U.S. Patent and Trademark Officer, or “USPTO”, or the European Patent Office, or the “EPO”, will change, thus affecting Pieris’ ability to obtain patents or maintain and enforce existing patents; |
|
● |
whether or not others will obtain patents claiming inventions similar to those covered by Pieris’ patents and patent applications; or |
|
● |
whether Pieris will need to initiate litigation or administrative proceedings (for example, at the USPTO or the EPO) in connection with patent rights, which may be costly whether Pieris wins or loses. |
As a result, the patent applications Pieris owns or license may fail to result in issued patents in the United States or in foreign countries. Third parties may challenge the validity, enforceability or scope of any issued patents Pieris owns or licenses or any applications that may issue as patents in the future, which may result in those patents being narrowed, invalidated or held unenforceable. Even if they are unchallenged, Pieris’ patents and patent applications may not adequately protect its intellectual property or prevent others from developing similar products that do not fall within the scope of its patents. If the breadth or strength of protection provided by the patents Pieris holds or pursues is threatened, its ability to commercialize any drug candidates with technology protected by those patents could be threatened. Further, if Pieris encounters delays in its clinical trials, the period of time during which Pieris would have patent protection for any covered drug candidates that obtain regulatory approval would be reduced. Since patent applications in the United States and most other countries are confidential for a period of time after filing, Pieris cannot be certain at the time of filing that it is the first to file any patent application related to its drug candidates.
While patent term extensions under the Hatch-Waxman Act in the United States and under supplementary protection certificates in Europe may be available to extend its patent exclusivity for its drug candidates, the applicable patents may not meet the specified conditions for eligibility for any such term extension and, even if eligible, Pieris may not be able to obtain any such term extension. Further, because filing, prosecuting, defending and enforcing patents in multiple jurisdictions can be expensive, Pieris may elect to pursue patent protection relating to its drug candidates in only certain jurisdictions. As a result, competitors would be permitted to use Pieris’ technologies in jurisdictions where it has not obtained patent protection to develop their own products, any of which could compete with its drug candidates.
In addition to the protection afforded by patents, Pieris seeks to rely on trade secret protection and CDAs to protect proprietary know-how that is not patentable, processes for which patents are difficult to enforce and any other elements of its discovery platform and drug development processes that involve proprietary know-how, information or technology that is not covered by patents or not amenable to patent protection. Although Pieris requires all of its employees and certain consultants, third parties and advisors to assign inventions to it, they may refuse to assign the inventions which could create delay or risk assignment of inventions. Pieris also requires all of its employees, consultants, advisors and any third parties who have access to its proprietary know-how, information or technology to enter into CDAs, its trade secrets and other proprietary information may be disclosed or competitors may otherwise gain access to such information or independently develop or reverse engineer substantially equivalent information. Further, the laws of some foreign countries do not protect proprietary rights to the same extent or in the same manner as the laws of the United States. As a result, Pieris may encounter significant difficulty in protecting and defending its intellectual property both in the United States and abroad. If Pieris is unable to prevent material disclosure of the trade secrets and other intellectual property related to its technologies to third parties, Pieris may not be able to establish or maintain the competitive advantage that it believes is provided by such intellectual property, adversely affecting its market position and business and operational results.
Claims that Pieris or its partners infringe the intellectual property rights of others may prevent or delay drug discovery and development efforts.
Pieris’ partnered drug candidates may infringe or be accused of infringing a patent or other form of intellectual property under which Pieris does not hold a license or other rights. Third parties may assert that Pieris is employing their proprietary technology without authorization.
There may be third-party patents of which Pieris is currently unaware with claims that cover the use or manufacture of its drug candidates or the practice of its related methods. Because patent applications can take many years to issue, there may be currently pending patent applications that may later result in issued patents that Pieris’ partnered drug candidates may infringe. In addition, third parties may obtain patents in the future and claim that use of such drug candidates infringes upon one or more claims of these patents. If Pieris’ partnered drug candidates infringe the patents or other intellectual property rights of third parties, the holders of such intellectual property rights may be able to block the ability to commercialize such drug candidates unless Pieris or its partners obtain a license under the intellectual property rights or until any applicable patents expire or are determined to be invalid or unenforceable.
Defense of any intellectual property infringement claims against Pieris, regardless of their merit, would involve substantial litigation expense and would be a significant diversion of resources from its business. In the event of a successful claim of infringement against Pieris, it may have to pay substantial damages, obtain one or more licenses from third parties, limit its business to avoid the infringing activities, pay royalties and/or redesign its infringing drug candidates or alter related formulations, processes, methods or other technologies, any or all of which may be impossible or require substantial time and monetary expenditure. Further, if Pieris were to seek a license from the third-party holder of any applicable intellectual property rights, Pieris may not be able to obtain the applicable license rights when needed or on reasonable terms, or at all. Some of Pieris’ competitors may be able to sustain the costs of complex patent litigation or proceeding more effectively than Pieris due to their substantially greater resources. The occurrence of any of the above events could cause Pieris’ business to materially suffer.
The patent protection covering some of Pieris’ drug candidates may be dependent on third parties, who may not effectively maintain that protection.
While Pieris expects the right to fully prosecute any patents covering drug candidates Pieris may in-license from third-party owners, there may be instances when the prosecution and maintenance of issued patents and pending patent applications that cover its drug candidates remain controlled by its licensors. Similarly, some of Pieris’ future licensing partners may retain the right, or may seek the rights, to prosecute patents covering the drug candidates Pieris licenses to them and Pieris may grant such rights to those partners for business reasons. If such third parties fail to appropriately maintain that patent protection, Pieris may not be able to prevent competitors from developing and selling competing products or practicing competing methods and its ability to generate revenue from any commercialization of the affected drug candidates may suffer.
Pieris may be involved in lawsuits to protect or enforce its patents or the patents of its licensors, which could be expensive, time-consuming and unsuccessful.
Competitors may infringe Pieris’ patents or the patents of its current or potential licensors. To attempt to stop infringement or unauthorized use, Pieris may need to enforce one or more of its patents, which can distract its management and divert its limited time and resources. Pieris’ standing to enforce such patents may sometimes be dependent on the licensor joining such suit, and a licensor's failure to join such suit may prevent Pieris from enforcing the patent. If Pieris pursues any litigation, a court may decide that a patent of Pieris’ or any of its licensors’ is not valid or is unenforceable or may refuse to stop the other party from using the relevant technology on the grounds that its patents do not cover the technology in question. Further, the legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents, which could reduce the likelihood of success of, or the amount of damages that could be awarded resulting from, any infringement proceeding Pieris pursues in any such jurisdiction. An adverse result in any infringement litigation or defense proceedings could put one or more of Pieris’ patents at risk of being invalidated, held unenforceable, or interpreted narrowly and could put its patent applications at risk of not issuing, which could limit its ability to exclude competitors from directly competing with Pieris in those jurisdictions.
Interference proceedings may also be provoked or suggested by third parties, or brought by the USPTO or at its foreign counterparts (such as the EPO), to determine the priority of inventions with respect to Pieris’ patents or patent applications or those of its licensors. An unfavorable outcome could require Pieris to cease using the related technology or to attempt to license rights to use it from the prevailing party. Pieris’ business could be harmed if the prevailing party does not offer Pieris a license on commercially reasonable terms, or at all.
Litigation or interference proceedings may fail and, even if successful, may result in substantial costs and distract Pieris’ management and other employees.
If Pieris is unsuccessful in obtaining or maintaining patent protection for intellectual property in development, its business and competitive position would be harmed.
Pieris may continue to seek patent protection of its technology and for its drug candidates. Patent prosecution is a challenging process and is not assured of success. If Pieris is unable to secure patent protection for its technology and drug candidates, its business may be adversely impacted.
Furthermore, issued patents and pending applications require regular maintenance. Failure to maintain Pieris’ portfolio may result in loss of rights that may adversely impact its intellectual property rights, such as rendering issued patents unenforceable or terminating pending applications prematurely.
In addition, under the European Union regulation on classification, labeling and packaging of substances and mixtures, and under other regulations in the United States or other countries related to the clinical development of Pieris’ drug candidates (including, for example, submissions to regulatory authorities such as the FDA and EMA, as well as submissions related to obtaining a non-proprietary, or international nonproprietary names, or “INN”, and United States adopted name, or “USAN”, name for Pieris’ clinical drug candidates to the World Health Organization, and USAN Council), Pieris or its partners may be required to publicly disclose the composition of its proprietary products or substances, which may facilitate infringement or avoidance of its intellectual property by third parties and may potentially reduce the margin Pieris is able to charge for its products by allowing competitors to more accurately determine its production costs. Future development of these regulations may have a further negative impact on Pieris’ revenues and a substantial negative impact on its business.
If Pieris is unable to protect the confidentiality of its trade secrets, its business and competitive position would be harmed.
In addition to seeking patents for Pieris’ Anticalin-brand technology and some of its drug candidates, Pieris also relies on trade secrets, including unpatented know-how, technology and other proprietary information, to maintain its competitive position. Pieris currently, and expects in the future to continue to, seek to protect these trade secrets, in part by entering into CDAs with parties who have access to them, such as its employees, collaborators, CMOs, consultants, advisors, investigators and other third parties. Pieris also enters into confidentiality and invention or patent assignment agreements with its employees and consultants. Despite these efforts, any of these parties may breach the agreements and disclose Pieris’ proprietary information, including its trade secrets, and Pieris may not be able to obtain adequate remedies for any such disclosure. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive and time-consuming, and the outcome is unpredictable. In addition, some courts inside and outside the United States are less willing or unwilling to protect trade secrets. If any of Pieris’ trade secrets were to be lawfully obtained or independently developed by a competitor, Pieris would have no right to prevent them, or those to whom they disclose the trade secrets, from using that technology or information to compete with Pieris. If any of Pieris trade secrets were to be disclosed to or independently developed by a competitor, its competitive position would be harmed.
Risks Related to Pieris’ Personnel
If Pieris is not able to retain highly qualified personnel, it may not be able to successfully implement its business objectives.
Pieris continues to rely on a limited number of employees and may rely on external consultants in the future for the operation of Pieris. Any of these employees or external consultants may terminate their relationship with Pieris at any time. Pieris may not be able to attract and retain consultants on acceptable terms given the competition among numerous pharmaceutical and biotechnology companies for similar personnel. Any future consultant or advisor may be employed by employers other than Pieris and may have commitments under consulting or advisory contracts with other entities that may limit their availability to Pieris. Pieris does not maintain “key person” insurance on any of its employees or consultants.
In addition, Pieris’ industry tends to experience a high rate of turnover of management personnel and its employees are generally able to terminate their relationships with it on short notice. Pursuant to German employment law, Pieris’ employment arrangements with employees of Pieris Pharmaceuticals GmbH are governed by employment contracts, which provide certain defined terms for either party to terminate the employment relationship.
Furthermore, to the extent Pieris pursues any strategic opportunities, Pieris’ ability to consummate such opportunities depends upon its ability to retain its employees and consultants required to consummate such a transaction, the loss of whose services may adversely impact the ability to consummate such transaction.
Pieris may be subject to labor claims brought by its employees against Pieris.
In the United States, an employment relationship with no specified duration is presumed to be employment “at-will” and the employer or employee may terminate the employment relationship at any time, with or without cause, except for public policy reasons including discrimination, participating in union activity, or refusing to carry out an activity that violates the law.
In contrast, in Germany, there is no analogous doctrine of “employment at will.” By law, German employees must have written employment contracts that reflect the key aspects of the employment relationship. Pieris’ relations between German employers and employees are extensively regulated under German labor and employment laws and regulations. Employment relationships may be terminated for cause without observing the ordinary notice period. If terminated without cause, the applicable ordinary notice period must be observed. German employees have special protection against dismissals provided the employee has been employed by a company for more than six months and such company employs more than ten full-time employees.
German employment termination law is regulated by various codes, in particular the Kündigungsschutzgesetz, or the German Termination Protection Act, and is intended to give the employee maximum protection against unfair dismissal, including among other things:
|
● |
the employer must observe the applicable notice period, which is ordinarily determined by law (between four weeks and seven months, depending upon the length of employment, though it is possible for the notice period to be two weeks, if a probationary period, lasting up to the first six months of employment, is agreed upon), if a longer period is not otherwise agreed by the parties, and has to deliver a written notice of termination to the employee; |
|
● |
for companies with more than 10 full-time employees, the German Termination Protection Act generally restricts termination of employment if the employee has been employed for more than six months, wherein the employee may be terminated only for a particular reason, including certain behavioral or personal reasons relating to the employee or certain developments relating to the business of the employer, such as a business restructuring which reduces the number of employee positions; |
|
● |
special termination protection against unlawful dismissal applies to several other groups of employees, such as an employee that is an officially acknowledged handicapped person, an employee who was appointed as a company’s data protection officer or as a member of the works council of a company, if any, an employee on maternity leave or a pregnant employee (in these cases, approval of various German authorities is required prior to termination but usually very difficult to obtain); and |
|
● |
if a company engages in a mass layoff, which is deemed to occur when the employer intends to dismiss a large percentage of its employees during a thirty-calendar day period, prior written notification to the German employment office is required. |
In July 2023, Pieris conducted a reduction in force that impacted 70% of its employees, and in March 2024, Pieris announced additional measures that would result in a further reduction in workforce that was implemented in the third quarter of 2024. In this regard, if Pieris downsizes for any reason and fails to adhere to the complex requirements articulated by the employee protection law, Pieris could face legal actions brought by affected employees or former employees, and, as a result, Pieris may incur operational or financial losses and divert the attention of its executive officers from managing its business.
Pieris may be subject to claims by third parties asserting that its employees or Pieris has misappropriated their intellectual property or claiming ownership of what Pieris regards as its own intellectual property.
Many of Pieris’ employees were previously employed at universities or other biotechnology or pharmaceutical companies, including its competitors or potential competitors. Pieris may be subject to claims that these employees or Pieris has used or disclosed intellectual property, including trade secrets or other proprietary information, of any such employee’s former employers. Litigation may be necessary to defend against any such claims.
In addition, while it is Pieris’ policy to require its employees and contractors, who may be involved in the development of intellectual property, to execute agreements assigning such intellectual property to Pieris, Pieris may be unsuccessful in executing such an agreement with each party who contributes to the development of intellectual property that Pieris regards as its own. Further, the terms of such assignment agreements may be breached and Pieris may not be able to successfully enforce their terms, which may force Pieris to bring claims against third parties, or defend claims they may bring against Pieris, to determine the ownership of intellectual property rights Pieris may regard and treat as its own.
Pieris’ employees, independent contractors, principal investigators, CROs, consultants, or vendors may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements, which could cause Pieris’ business to suffer.
Pieris is exposed to the risk that its employees, independent contractors, principal investigators, CROs, consultants, or vendors may engage in fraudulent or other illegal activity. Misconduct by any of these parties could include intentional, reckless, and/or negligent conduct that may include failures to comply with FDA, MHRA, EMA or other foreign jurisdiction regulations, provide accurate information to the FDA, MHRA, EMA or their comparable foreign equivalents, comply with manufacturing standards Pieris has established, comply with federal, state and international healthcare fraud and abuse laws and regulations as they may become applicable to Pieris’ operations, report financial information or data accurately or disclose unauthorized activities to Pieris. Employee and other third-party misconduct could also involve the improper use of information obtained in the course of clinical trials, which could result in regulatory sanctions and serious harm to Pieris’ reputation. It is not always possible to identify and deter misconduct by Pieris’ employees and other third parties, and the precautions and procedures Pieris currently takes or may establish in the future as its operations and employee and third-party base expand to detect and prevent this type of activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting Pieris from governmental investigations or other actions or lawsuits stemming from a failure to be in compliance with such laws or regulations. If any such actions are instituted against Pieris, and Pieris is not successful in defending itself or asserting its rights, those actions could have a significant impact on its business and results of operations, including the imposition of civil, criminal and administrative penalties, damages, monetary fines, possible exclusion from participation in Medicare, Medicaid, and other federal healthcare programs, contractual damages, reputational harm, diminished potential profits and future earnings, and curtailment of its operations.
Certain of Pieris’ employees and their inventions are subject to German law.
Many of Pieris’ employees work in Germany and are subject to German employment law. Ideas, developments, discoveries and inventions made by such employees and consultants are subject to the provisions of the Gesetz über Arbeitnehmererfindungen, or the “German Act on Employees’ Inventions”, which regulates the ownership of, and compensation for, inventions made by employees. Pieris has experienced disputes and faces the risk that disputes may occur in the future between Pieris and such employees or ex-employees pertaining to alleged non-adherence to the provisions of this act. Such disputes can be costly to defend and take up Pieris’ management’s time and efforts whether it prevails or not. In addition, under the German Act on Employees’ Inventions, certain employees retained rights to patents they invented or co-invented prior to 2009. Although most of these employees have subsequently assigned their interest in these patents to Pieris, there is a risk that the compensation Pieris provides to them may be deemed insufficient and Pieris may be required under German law to increase the compensation due to such employees for the use of the patents. In those cases where employees have not assigned their interests to Pieris, Pieris may need to pay compensation for the use of those patents. If Pieris is required to pay additional compensation or face other disputes under the German Act on Employees’ Inventions, its results of operations could be adversely affected.
Risks Related to the Ownership of Pieris Common Stock
Pieris’ share price is volatile and may be influenced by numerous factors, some of which are beyond its control.
Market prices for shares of biotechnology companies such as Pieris’ are often volatile. Thus, the quoted price of Pieris common stock has been, and is likely to continue to be, highly volatile and could be subject to wide fluctuations in response to various factors, some of which are beyond its control. In addition to the factors discussed in this “Risk Factors” section and elsewhere in this proxy statement/prospectus, these factors include:
|
● |
the timing and amount of potential milestone payments that Pieris may receive from Pfizer and Boston Pharmaceuticals; |
|
● |
additions or departures of key scientific or management personnel; |
|
● |
changes in laws or regulations applicable to Pieris’ drug candidates, including without limitation clinical trial requirements for approvals; |
|
● |
the perception of the pharmaceutical and biotechnology industry by the public, legislatures, regulators and the investment community; |
|
● |
disputes or other developments relating to patents and other proprietary rights and Pieris’ ability to obtain patent protection for its drug candidates; |
|
● |
significant lawsuits, including patent and stockholder class action litigation; |
|
● |
Pieris’ potential inability to maintain the listing of its common stock on The Nasdaq Capital Market; |
|
● |
actual or anticipated variations in quarterly operating results; |
|
● |
failure to meet or exceed the estimates and projections of the investment community; |
|
● |
overall performance of the equity markets and other factors that may be unrelated to Pieris’ operating performance or the operating performance of its competitors, including changes in market valuations of similar companies; |
|
● |
conditions or trends in the biotechnology and biopharmaceutical industries; |
|
● |
introduction of new products by Pieris’ competitors; |
|
● |
announcements of significant acquisitions, strategic partnerships, joint ventures or capital commitments by Pieris or its competitors; |
|
● |
issuances of debt or equity securities; |
|
● |
execution, cost and timing of Pieris’ reduction in force and operations; |
|
● |
sales of Pieris common stock by Pieris or its stockholders in the future, or the perception that such sales could occur; |
|
● |
trading volume of Pieris common stock; |
|
● |
ineffectiveness of Pieris’ internal control over financial reporting or disclosure controls and procedures; |
|
● |
general political and economic conditions; |
|
● |
effects of natural or man-made catastrophic events; and |
|
● |
other events or factors, many of which are beyond Pieris’ control. |
In addition, the stock market in general, and the stocks of biotechnology companies in particular, have experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of these companies. Broad market and industry factors may negatively affect the market price of Pieris common stock, regardless of its actual operating performance. Furthermore, other biotechnology companies or Pieris’ competitors’ programs could have positive or negative results that impact their stock prices and their results or stock fluctuations could have a positive or negative impact on Pieris’ stock price regardless of whether such impact is direct or not. The realization of any of the above risks or any of a broad range of other risks, including those described in these “Risk Factors”, could have a dramatic and material adverse impact on the market price of Pieris common stock.
Pieris has broad discretion in how it uses its cash, cash equivalents and investments, including the net proceeds from its collaborations, public and private securities offerings, and may not use these financial resources effectively, which could affect its results of operations and cause its stock price to decline.
Pieris’ management has considerable discretion in the application of its cash, cash equivalents and investments, including the fees and milestone payments from its collaborations and the net proceeds of its securities offerings. As a result, investors will be relying upon management’s judgment with only limited information about Pieris’ specific intentions for the use of the cash, cash equivalents and investments. Pieris may use the cash, cash equivalents and investments for purposes that do not yield a significant return or any return at all for its stockholders. In addition, pending their use, Pieris may invest the financial resources from its collaborations and securities offerings in a manner that does not produce income or that loses value. Pieris may also use the cash to pay out dividends to stockholders if it determines that there is sufficient cash and investments to achieve its near and long-term objectives.
If securities or industry analysts do not publish, or cease publishing, research or publish inaccurate or unfavorable research about Pieris’ business or its market, or if they change their recommendations regarding its stock adversely, its stock price and any trading volume could decline.
The trading market for Pieris common stock will depend in part on the research and reports that securities or industry analysts publish about Pieris or its business. Pieris does not have any control over these analysts. If only a few securities or industry analysts commence coverage of Pieris, the trading price for Pieris’ stock would likely be negatively affected and there can be no assurance that analysts will provide favorable coverage. If securities or industry analysts who initiate coverage downgrade Pieris’ stock or publish inaccurate or unfavorable research about its business or its market, its stock price would likely decline. If one or more of these analysts cease coverage of Pieris’ company or fail to publish reports on Pieris regularly, demand for Pieris’ stock could decrease, which might cause its stock price and any trading volume to decline.
If Pieris fails to maintain proper and effective internal controls, its ability to produce accurate and timely financial statements could be impaired, which could harm its operating results, its ability to operate its business and investors’ views of Pieris.
Pursuant to Section 404 of the Sarbanes-Oxley Act of 2002, or “Section 404”, Pieris is required to furnish a report by its management on its internal control over financial reporting. However, as Pieris remains a smaller reporting company with less than $100 million in revenue, Pieris is not currently required to include an attestation report on internal control over financial reporting issued by its independent registered public accounting firm.
If Pieris cannot favorably assess the effectiveness of its internal controls over financial reporting, investor confidence and, in turn, its stock price could be materially adversely affected.
Ensuring that Pieris has adequate internal financial and accounting controls and procedures in place so that it can produce accurate financial statements on a timely basis is a costly and time-consuming effort that will need to be evaluated frequently. Pieris’ failure to maintain the effectiveness of its internal controls in accordance with the requirements of the Sarbanes-Oxley Act could have a material adverse effect on the tradability of its common stock, which in turn would negatively impact its business. Pieris could lose investor confidence in the accuracy and completeness of its financial reports, which could have an adverse effect on the price of its common stock. In addition, if Pieris’ efforts to comply with new or changed laws, regulations and standards differ from the activities intended by regulatory or governing bodies due to ambiguities related to practice, regulatory authorities may initiate legal proceedings against Pieris and its business may be harmed.
Shares of Pieris common stock that have not been registered under federal securities laws are subject to resale restrictions imposed by Rule 144 of the Securities Act, including those set forth in Rule 144(i) which apply to a former “shell company.”
Pieris was previously deemed a “shell company” under applicable SEC rules and regulations, prior to the reverse merger transaction in which Pieris became a public company, because it had no or nominal operations and either no or nominal assets, assets consisting solely of cash and cash equivalents, or assets consisting of any amount of cash and cash equivalents and nominal other assets. Sales of the restricted securities of a former shell company, such as Pieris, are not permitted pursuant to Rule 144 of the Securities Act, unless at the time of a proposed sale, (i) Pieris is subject to the reporting requirements of Section 13 or 15(d) of the Exchange Act; and (ii) Pieris has filed all reports and other materials required to be filed by Section 13 or 15(d) of the Exchange Act, as applicable, during the preceding twelve months, other than current reports on Form 8-K. Additionally, Pieris’ previous status as a shell company could also limit its use of its securities to pay for any acquisitions it may seek to pursue in the future. The lack of liquidity of its securities as a result of the inability to sell under Rule 144 for a longer period of time than a non-former shell company could cause the market price of Pieris’ securities to decline.
If Pieris issues additional shares of its capital stock in the future, its existing stockholders will be diluted.
Pieris’ amended and restated articles of incorporation authorize the issuance of up to 3,750,000 shares of its common stock and up to 10,000,000 shares of preferred stock with the terms, limitations, voting rights, relative rights and preferences and variations of each series that the Pieris board of directors may determine from time to time. Possible business and financial uses for Pieris’ authorized capital stock include, without limitation, equity financing, future stock splits, acquiring other companies, businesses or products in exchange for shares of Pieris capital stock, issuing shares of its capital stock to partners or other collaborators in connection with strategic alliances, attracting and retaining employees by the issuance of additional securities under its equity compensation plan, or other transactions and corporate purposes that the Pieris board of directors deems are in the interests of Pieris. Furthermore, issuances of shares of Pieris capital stock could have the effect of delaying or preventing changes in control or Pieris’ management. Any future issuances of shares of Pieris capital stock may not be made on favorable terms or at all, they may have rights, preferences and privileges that are superior to those of Pieris common stock and may have an adverse effect on its business or the trading price of its common stock. The issuance of any additional shares of Pieris common stock will reduce the book value per share and may contribute to a reduction in the market price of the outstanding shares of its common stock. Additionally, any such issuance will reduce the proportionate ownership and voting power of all of Pieris’ current stockholders.
Sales of a substantial number of shares of Pieris common stock in the public market, or the perception that such sales could occur, could cause its stock price to fall.
If Pieris’ existing stockholders sell, or indicate an intention to sell, substantial amounts of its common stock in the public market, the trading price of its common stock could decline. As of October 28, 2024, a total of 1,320,240 shares of Pieris common stock were outstanding. Any sales of those shares or any perception in the market that such sales may occur could cause the trading price of Pieris common stock to decline.
In addition, shares of Pieris common stock that are either subject to outstanding options or reserved for future issuance under Pieris’ equity incentive plan, or issuable upon the conversion of its outstanding preferred stock or upon the exercise of its outstanding warrants, will be eligible for sale in the public market to the extent permitted by the provisions of applicable vesting schedules and/or terms of such securities. If these additional shares of common stock are sold, or if it is perceived that they will be sold, in the public market, the trading price of Pieris common stock could decline.
The resale of shares covered by Pieris’ effective resale registration statements could adversely affect the market price of its common stock in the public market, which result would in turn negatively affect its ability to raise additional equity capital.
The sale, or availability for sale, of Pieris’ common stock in the public market may adversely affect the prevailing market price of its common stock and may impair its ability to raise additional equity capital. Pursuant to registration statements filed with the SEC, Pieris previously registered for resale shares of its common stock, which included all of the shares of its common stock issued in its private placements and in connection with the closing of the reverse merger transaction in which Pieris became a public company. For example, in March 2021, Pieris registered for resale 46,328 shares of common stock in connection with a private placement transaction with Pfizer, and 44,804 shares of common stock in connection with a private placement transaction with AstraZeneca, in both cases on a pre-split basis. The resale registration statements permit the resale of these shares at any time without restriction.
The resale of a substantial number of shares of Pieris common stock in the public market could adversely affect the market price for Pieris common stock and make it more difficult for investors to sell shares of its common stock at times and prices that investors feel are appropriate. Furthermore, because there are a large number of shares registered pursuant to the resale registration statements, Pieris may continue to offer shares covered by the resale registration statements for a significant period of time, the precise duration of which cannot be predicted. Accordingly, the adverse market and price pressures resulting from an offering pursuant to the resale registration statement may continue for an extended period of time and continued negative pressure on the market price of Pieris common stock could have a material adverse effect on its ability to raise additional equity capital, if Pieris seeks to do so in the future.
Future sales and issuances of Pieris common stock or rights to purchase common stock, including pursuant to its equity incentive plans or otherwise, could result in dilution of the percentage ownership of its stockholders and could cause its stock price to fall.
If Pieris seeks to raise capital in the future, Pieris may sell common stock, convertible securities or other equity securities in one or more transactions at prices and in a manner, in which Pieris may determine from time to time. If Pieris sells common stock, convertible securities or other equity securities in more than one transaction, investors in a prior transaction may be materially diluted. Additionally, new investors could gain rights, preferences and privileges senior to those of existing holders of Pieris common stock. Further, any future sales of Pieris common stock by Pieris or resales of its common stock by its existing stockholders could cause the market price of its common stock to decline.
As of June 30, 2024, there were 319,992 shares reserved for future issuance under Pieris’ equity compensation plans, and 135,941 shares reserved for issuance upon the exercise of outstanding equity awards. Pursuant to Pieris’ 2023 Employee Stock Purchase Plan, Pieris is authorized to sell 9,375 shares to its employees. Any future grants of options, warrants or other securities exercisable or convertible into Pieris common stock, or the exercise or conversion of such shares, and any sales of such shares in the market, could have an adverse effect on the market price of its common stock.
Anti-takeover provisions in Pieris’ organizational documents could delay or prevent a change of control.
Certain provisions of Pieris’ amended and restated articles of incorporation and amended and restated bylaws may have an anti-takeover effect and may delay, defer or prevent a merger, acquisition, tender offer, takeover attempt or other change of control transaction that a stockholder might consider to be in its interests, including attempts that might result in a premium over the market price for the shares held by Pieris stockholders.
These provisions provide, among other things:
|
● |
a classified board of directors with staggered three-year terms; |
|
● |
the ability of the Pieris board of directors to issue one or more series of preferred stock with voting or other rights or preferences that could have the effect of impeding the success of an attempt to acquire Pieris or otherwise effect a change of control; |
|
● |
advance notice for nominations of directors by stockholders and for stockholders to include matters to be considered at stockholder meetings; |
|
● |
certain limitations on convening special stockholder meetings and the prohibition of stockholder action by written consent; and |
|
● |
directors may only be removed for cause and only by the affirmative vote of the holders of at least 80% of the voting power of all of the then-outstanding shares of Pieris capital stock entitled to vote at an election of directors, voting together as a single class. |
These anti-takeover provisions, including those noted above, could make it more difficult for a third party to acquire Pieris, even if the third party’s offer may be considered beneficial by many of Pieris stockholders. As a result, Pieris stockholders may be limited in their ability to obtain a premium for their shares.
Pieris may incur significant costs from class action litigation due to its expected stock volatility.
Pieris’ stock price may fluctuate for many reasons, including as a result of public announcements regarding Pieris’ strategic updates or the development efforts of current or future collaborators or competitors, the addition or departure of its key personnel, variations in its quarterly operating results and changes in market valuations of biopharmaceutical and biotechnology companies.
This risk is especially relevant to Pieris because biopharmaceutical and biotechnology companies have experienced significant stock price volatility in recent years. When the market price of a stock has been volatile, as Pieris’ stock price may be, holders of that stock have occasionally brought securities class action litigation against it that issued the stock. If any of the Pieris stockholders were to bring a lawsuit of this type against Pieris, even if the lawsuit was without merit, it could result in substantial costs incurred defending the lawsuit and diversion of the time, attention and resources of the Pieris board of directors and management, which could significantly harm its profitability and reputation.
Pieris’ amended and restated articles of incorporation designates the Eighth Judicial District Court of Clark County, Nevada, as the sole and exclusive forum for certain types of actions and proceedings that may be initiated by its stockholders, and therefore limit its stockholders’ ability to choose a forum for disputes with Pieris or its directors, officers, employees or agents.
Pieris’ amended and restated articles of incorporation provide that, to the fullest extent permitted by law, and unless Pieris consents to the selection of an alternative forum, the Eighth Judicial District Court of Clark County, Nevada shall be the sole and exclusive forum for any (i) derivative action or proceeding brought in the name or right of the corporation or on its behalf, (ii) action asserting a claim of breach of a fiduciary duty owed by any of its directors, officers, employees or agents to the corporation or any of its stockholders, (iii) any action arising or asserting a claim arising pursuant to any provision of Chapters 78 or 92A of the NRS or any provision of its amended and restated articles of incorporation or amended and restated bylaws, (iv) any action to interpret, apply, enforce or determine the validity of its articles of incorporation or bylaws or (v) any action asserting a claim governed by the internal affairs doctrine. Pieris’ amended and restated articles of incorporation further provide that any person purchasing or otherwise acquiring any interest in shares of its capital stock shall be deemed, to the fullest extent permitted by law, to have notice of and consented to the foregoing provision.
Choice-of-forum provisions of the type and scope included in Pieris’ amended and restated articles of incorporation are expressly permitted by Section 78.046 of the NRS, but application of these choice-of-forum provisions may be limited in some instances by law. Section 27 of the Exchange Act establishes exclusive federal jurisdiction over all suits brought to enforce any duty or liability created by the Exchange Act or the rules and regulations thereunder and therefore the choice-of-forum provision would not apply to actions arising under, or brought to enforce a duty or liability created by, the Exchange Act or any other claim for which the U.S. federal courts have exclusive jurisdiction. However, Section 22 of the Securities Act provides that federal and state courts have concurrent jurisdiction over all suits brought to enforce any duty or liability created by the Securities Act or the rules and regulations thereunder, and the choice-of-forum provision would apply to actions arising under, or brought to enforce a duty or liability created by, the Securities Act. To the extent the text of the choice-of-forum provision in Pieris’ amended and restated articles of incorporation purports to restrict the courts in which claims arising under the Securities Act may be brought, there remains some uncertainty as to whether a court would enforce such a provision. We note that the choice-of-forum provision does not relieve Pieris of its duties to comply with the federal securities laws and the rules and regulations thereunder, and Pieris’ stockholders will not be deemed to have waived compliance with these laws, rules and regulations.
Pieris believes the choice-of-forum provision in its amended and restated articles of incorporation will help provide for the orderly, efficient and cost-effective resolution of the types of legal issues affecting Pieris, as identified in the choice-of-forum provision, by designating courts located in the State of Nevada (Pieris’ state of incorporation) as the exclusive forum for cases involving such issues. However, this provision may limit a stockholder’s ability to bring a claim in a judicial forum that it believes to be favorable for disputes with Pieris or its directors, officers, employees or agents, which may discourage such actions against Pieris and its directors, officers, employees and agents, and could also increase the costs of stockholders in connection with bringing a claim and resolving such matters. If a court were to find the choice-of-forum provision in Pieris’ amended and restated articles of incorporation inapplicable to, or unenforceable in respect of, one or more of the specified types of actions or proceedings, Pieris may incur additional costs associated with resolving such matters in other jurisdictions, which could adversely affect its business, financial condition or results of operations.
The elimination of personal liability of Pieris’ directors and officers under Nevada law and the existence of indemnification rights held by its directors, officers and employees may result in substantial expenses.
Pieris’ amended and restated articles of incorporation eliminate, to the furthest extent permitted under Nevada law, the personal liability of its directors and officers to Pieris, its stockholders and creditors for damages as a result of any act or failure to act in his or her capacity as a director or officer. Further, Pieris’ amended and restated articles of incorporation, its amended and restated bylaws and individual indemnification agreements that Pieris has entered with each of its directors and officers provide that it is obligated to indemnify, subject to certain exceptions, each of its directors or officers to the fullest extent authorized by Nevada law and, subject to certain conditions, to advance the expenses incurred by any director or officer in defending any action, suit or proceeding prior to its final disposition. Those indemnification obligations could expose Pieris to substantial expenditures to cover the cost of settlement or damage awards against its directors or officers, which Pieris may be unable to afford. Further, those provisions and resulting costs may discourage Pieris or its stockholders from bringing a lawsuit against any of its current or former directors or officers for such damages, even if such actions might otherwise benefit its stockholders.
The Pieris board of directors may, at their sole discretion, elect to pay cash dividends on its capital stock.
Pieris has never declared or paid any cash dividends on its common stock. Pieris may pay out dividends to stockholders if it is determined that there is sufficient cash and investments to achieve its near and long-term objectives. Pieris may choose to retain all future earnings to fund strategic opportunities in the future. Any future payment of cash dividends will be at the discretion of the Pieris board of directors and will depend on, among other things, its earnings, financial condition, capital requirements, level of indebtedness, statutory and contractual restrictions applying to the payment of dividends and other considerations that the Pieris board of directors deems relevant.
Pieris can issue and has issued shares of preferred stock, which may adversely affect the rights of holders of its common stock.
Pieris’ amended and restated articles of incorporation authorizes it to issue up to 10,000,000 shares of preferred stock with designations, rights, and preferences determined from time-to-time by the Pieris board of directors. Accordingly, the Pieris board of directors is empowered, without stockholder approval, to issue preferred stock with dividend, liquidation, conversion, voting or other rights superior to those of holders of Pieris common stock. For example, an issuance of shares of preferred stock could:
|
● |
adversely affect the voting power of the holders of Pieris common stock; |
|
● |
make it more difficult for a third party to gain control of Pieris; |
|
● |
discourage bids for Pieris common stock at a premium; |
|
● |
limit or eliminate any payments that the holders of Pieris common stock could expect to receive upon its liquidation; or |
|
● |
otherwise adversely affect the market price or Pieris common stock. |
Pieris has in the past issued, and Pieris may at any time in the future issue, shares of preferred stock. In connection with Pieris’ June 2016 private placement, Pieris issued 4,963 shares of its Series A convertible preferred stock to certain affiliates of BVF each share of which is convertible into 13.34 shares of Pieris common stock, subject to certain ownership restrictions. In January 2019, Pieris entered into an exchange agreement with BVF to exchange 62,500 shares, on a post-split basis, of its common stock previously held by BVF for 5,000 shares of its Series B convertible preferred stock, each share of which is convertible into 13.34 shares of its common stock, subject to certain ownership restrictions. In connection with Pieris’ November 2019 private placement, Pieris issued 3,522 shares of its Series C convertible preferred stock to certain affiliates of BVF each share of which is convertible into 13.34 shares of its common stock, subject to certain ownership restrictions. In March 2020, Pieris entered into another exchange agreement with BVF to exchange 37,500 shares, on a post-split basis, of its common stock previously held by BVF for 3,000 shares of its Series D convertible preferred stock, each share of which is convertible into 13.34 shares of its common stock, subject to certain ownership restrictions. In May 2021, Pieris entered into another exchange agreement with BVF to exchange 62,500 shares, on a post-split basis, of its common stock previously held by BVF for 5,000 shares of its Series E convertible preferred stock, each share of which is convertible into 13.34 shares of its common stock, subject to certain ownership restrictions. If the holders of Pieris’ shares of preferred stock convert their shares into common stock, existing holders of Pieris common stock will experience dilution. On August 7, 2024, Pieris entered into a Subscription and Investor Representation Agreement with Mr. James Geraghty, the chairman of the Pieris board of directors, to sell to one share of Pieris’ Series F Preferred Stock to Mr. Geraghty, which must be voted on the Authorized Stock Increase Proposal in the same proportion as shares of Pieris common stock are voted “for” and “against” such proposal, excluding any shares of common stock that are not voted “for” and “against” the proposal for any reason, including, without limitation, any abstentions or broker non-votes, and is subject to certain restrictions and redemption terms.
Requirements associated with being a public company have increased Pieris’ costs significantly and have diverted significant company resources and management attention.
As a public company, Pieris is subject to the reporting requirements of the Exchange Act and the Sarbanes-Oxley Act. The Exchange Act requires the filing of annual, quarterly and current reports with respect to a public company’s business and financial condition. The Sarbanes-Oxley Act requires, among other things, that a public company establish and maintain effective internal control over financial reporting. Pieris’ management and other personnel need to devote a substantial amount of time to these compliance initiatives. Moreover, these rules and regulations have increased and will continue to negatively impact Pieris’ legal and financial compliance costs and will make some activities more time-consuming and costly.
Having availed itself of scaled disclosure available to smaller reporting companies, Pieris cannot be certain if such reduced disclosure will make its common stock less attractive to investors.
Under Rule 12b-2 of the Exchange Act, a “smaller reporting company” is a company that is not an investment company, an asset-backed issuer or a majority-owned subsidiary of a parent company that is not a smaller reporting company, and had a public float of less than $250 million as of the last business day of its most recently completed second fiscal quarter or, if such public float is less than $700 million, had annual revenues of less than $100 million during the most recently completed fiscal year. Smaller reporting companies are permitted to provide simplified executive compensation disclosure in their filings; and they have certain other decreased disclosure obligations in their SEC filings, including, among other things, only being required to provide two years of audited financial statements in annual reports. Pieris qualifies as a smaller reporting company. For as long as Pieris continues to be a smaller reporting company, it expects that it will take advantage of the reduced disclosure obligations available to it as a result of those respective classifications. Decreased disclosure in Pieris’ SEC filings as a result of Pieris having availed itself of scaled disclosure may make it harder for investors to analyze its results of operations and financial prospects.
Risks Related to Palvella’s Limited Operating History, Financial Position and Capital Requirements
Palvella has incurred significant operating losses since inception and anticipates that it will continue to incur significant operating losses for at least the next several years. Palvella may never achieve or maintain profitability.
Since inception in September 2015, Palvella has incurred significant operating losses and has never generated any revenue. Palvella’s operating loss for the years ended December 31, 2023 and 2022 was $11.9 million and $18.0 million, respectively, and for the six months ended June 30, 2024 was $4.7 million. As of June 30, 2024, Palvella had an accumulated deficit of $83.0 million. Palvella expects to continue to incur significant operating losses for at least the next several years, and it may never achieve or sustain profitability. Since inception, Palvella has devoted substantially all of its efforts to identifying, researching and conducting preclinical and clinical activities for its product candidates, developing its QTORIN platform, organizing and staffing its company, business planning, raising capital and establishing its intellectual property portfolio. Palvella has never obtained regulatory approval for, or commercialized, any products. Palvella anticipates that its expenses will increase substantially for the foreseeable future if, and as, it:
|
● |
seeks regulatory approval for QTORIN rapamycin for the treatment of microcystic LM and any other product candidates that successfully complete clinical trials; |
|
● |
continues clinical development of its product candidates, including its ongoing Phase 3 clinical trial for QTORIN rapamycin in patients with microcystic LM, and its planned Phase 2 clinical trial for QTORIN rapamycin in patients with cutaneous venous malformations; |
|
● |
continues preclinical development of its product candidates, including QTORIN rapamycin for other mTOR-driven skin diseases; |
|
● |
establishes a specialized commercial organization in the United States to commercialize any product candidate for which Palvella obtains marketing approval; |
|
● |
initiates and continues relationships with suppliers and manufacturers and has commercial quantities of its product candidates manufactured at acceptable cost and quality levels and in compliance with the FDA and other regulatory requirements; |
|
● |
initiates additional clinical trials and preclinical studies for its other product candidates; |
|
● |
seeks to identify and develop or in-license additional product candidates; |
|
● |
incurs additional costs associated with operating as a public company, which will require it to add operational, financial, and management information systems and personnel, including personnel to support product development, any future commercialization efforts, and its transition to a public company; |
|
● |
expands its infrastructure and facilities to accommodate its growing employee base, including adding equipment and physical infrastructure to support its research and development; and |
|
● |
maintains, expands and protects its intellectual property portfolio. |
Palvella may never succeed in any or all of these activities and, even if it does, it may never generate revenue.
Palvella has never generated revenue from product sales and may never achieve or maintain profitability.
To become and remain profitable, Palvella must succeed in developing and eventually commercializing products that generate significant revenue. This will require Palvella to be successful in a range of challenging activities, including completing preclinical studies and clinical trials, obtaining regulatory approval, procuring commercial-scale manufacturing, and marketing and selling any products for which it obtains regulatory approval. Palvella has never obtained regulatory approval, procured commercial-scale manufacturing or marketed any product, and it may never succeed in these activities. Even if Palvella does obtain regulatory approval for and begins commercializing QTORIN rapamycin for microcystic LM, cutaneous venous malformations, or any other indication or any future product candidates, its ability to become profitable will be dependent upon, in part and among other things, the size of the markets in the territories for which it gains regulatory approval, the number of competitors in such markets, the accepted price for any such product candidate and the degree of market acceptance it achieves.
Even if it does achieve profitability, Palvella may not be able to sustain or increase profitability on a quarterly or annual basis. Palvella’s failure to become and remain profitable would decrease the value of its company and could impair its ability to raise capital, maintain its research and development efforts, expand its business or continue its operations. A decline in the value of its company also could cause you to lose all or part of its investment.
Palvella’s limited operating history may make it difficult to evaluate its business to date and its future viability.
Palvella is a late clinical stage biopharmaceutical company with a limited operating history. Palvella has no products approved for commercial sale and has not generated any revenue from product sales. As an organization, Palvella has limited experience successfully completing pivotal clinical trials, and has not yet demonstrated an ability to obtain marketing approval, manufacture a commercial-scale product or arrange for a third party to do so on its behalf, or conduct sales and marketing activities necessary for successful product commercialization. Consequently, Palvella has little or no meaningful operations upon which to evaluate its business, and predictions about its future success or viability may not be as accurate as it could be if it had a longer operating history or a history of successfully developing and commercializing pharmaceutical products.
Palvella’s ability to generate product revenue and become profitable depends upon its ability to successfully complete the development of, and obtain the necessary regulatory approvals for, its product candidates. Even if Palvella receives regulatory approval for any product candidate, it does not know when or if such product candidate will generate product revenue. Palvella may encounter unforeseen expenses, difficulties, complications, delays and other known or unknown factors in achieving its business objectives, including with respect to its technology and product candidates. In the future, Palvella will need to transition from a company with a research and development focus to a company capable of supporting commercial activities. Palvella may also need to secure strategic collaborations with partners in order to commercialize any approved product candidates outside of the U.S. market. Palvella may not be successful in making such a transition or in securing such strategic collaborations.
Palvella’s recurring losses from operations and financial condition raise substantial doubt about its ability to continue as a going concern.
Palvella’s recurring losses from operations and financial condition raise substantial doubt about its ability to continue as a going concern. In its financial statements for the year ended December 31, 2023, Palvella concluded that its recurring losses from operations and need for additional financing to fund future operations raise substantial doubt about its ability to continue as a going concern. Similarly, its independent registered public accounting firm included an explanatory paragraph in its report on its financial statements for the year ended December 31, 2023 with respect to this uncertainty. Palvella’s ability to continue as a going concern will require it to obtain additional funding. If Palvella is unable to obtain sufficient funding, its business, prospects, financial condition and results of operations will be materially and adversely affected, and it may be unable to continue as a going concern. If Palvella is unable to raise capital when needed or on acceptable terms, it would be forced to delay, limit, reduce or terminate its product development or future commercialization efforts of one or more of its product candidates, or may be forced to reduce or terminate its operations. If Palvella is unable to continue as a going concern, it may have to liquidate its assets and may receive less than the value at which those assets are carried on its audited financial statements, and it is likely that investors will lose all or a part of their investment. In future required quarterly assessments, Palvella may again conclude that there is substantial doubt about its ability to continue as a going concern, and future reports from its independent registered public accounting firm may also contain statements expressing substantial doubt about its ability to continue as a going concern. If Palvella seeks additional financing to fund its business activities in the future and there remains substantial doubt about its ability to continue as a going concern, investors or other financing sources may be unwilling to provide additional funding to it on commercially reasonable terms, if at all. Based on its current business plans, Palvella believes that the anticipated cash and cash equivalents after the merger will be sufficient for it to fund its operating expenses and capital expenditure requirements through at least the next 33 months. This estimate is based on certain significant assumptions, which are uncertain and may turn out to be incorrect.
Palvella will likely require substantial additional funding to finance its operations, which may cause dilution to its stockholders, and a failure to obtain this necessary funding when needed on acceptable terms, or at all, could force it to delay, limit, reduce or terminate its product development, commercialization efforts or other operations.
As of June 30, 2024, Palvella had cash and cash equivalents of $14.5 million. Based upon its current operating plan, Palvella believes that the anticipated cash and cash equivalents after the closing of the merger and the PIPE Financing will be sufficient to fund its planned operations through in to the second half of 2027. Palvella has based this estimate on assumptions that may prove to be wrong, and it could exhaust its available capital resources sooner than it expects. To finance its operations beyond that point it may need to raise additional capital, which cannot be assured. Its operating plans may change as a result of many factors currently unknown to Palvella, and it may need to seek additional funds sooner than planned, through public or private equity or debt financings or other sources, such as strategic collaborations. To the extent that Palvella raises additional capital by issuing equity securities, its existing stockholders may experience substantial dilution, and the terms of these securities may include liquidation or other preferences that could harm the rights of a common stockholder. Any agreements for future debt or preferred equity financings, if available, may involve covenants limiting or restricting its ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends. If Palvella raises additional funds through collaborations, strategic alliances or marketing, distribution or licensing arrangements with third parties, it may have to relinquish valuable rights to its technologies, future revenue streams, research programs or product candidates, or grant licenses on terms that may not be favorable to it. Palvella may seek additional capital due to favorable market conditions or strategic considerations even if it believes Palvella has sufficient funds for its current or future operating plans. In addition, Palvella may seek additional capital due to favorable market conditions or strategic considerations even if it believes Palvella has sufficient funds for its current or future operating plans.
Palvella’s future capital requirements depend on many factors, including, but not limited to:
|
● |
the scope, progress, timing, results and costs of researching and developing its lead product candidates or any future product candidates, and conducting preclinical studies and clinical trials; |
|
● |
the number and scope of clinical programs it decides to pursue; |
|
● |
the cost, timing and outcome of seeking regulatory approvals of its product candidates; |
|
● |
the cost of manufacturing its product candidates and any products it commercializes, including costs associated with building out its supply chain; |
|
● |
the cost of commercialization activities if any of its product candidates are approved for sale, including marketing, sales and distribution costs; |
|
● |
its ability to establish and maintain strategic collaborations, licensing or other arrangements and the financial terms of any such agreements that Palvella may enter into; |
|
● |
the timing, receipt and amount of sales of any future approved products, if any; |
|
● |
the timing and amount of milestone or royalty payments due to Ligand, under Ligand Agreements (as defined under the section titled “Palvalla’s Business - Ligand Development Funding Agreement” beginning on page 245 of this proxy statement/prospectus )”, or under similar arrangements with any future collaboration or licensing partners; |
|
● |
the expenses needed to attract and retain skilled personnel; |
|
● |
its need to implement additional internal systems and infrastructure, including financial and reporting systems; and |
|
● |
the costs involved in preparing, filing, prosecuting, maintaining, defending and enforcing its intellectual property portfolio. |
Adequate additional funds may not be available when Palvella needs them, on terms that are acceptable to it, or at all. If adequate funds are not available to Palvella on a timely basis or on attractive terms, it may be required to reduce its workforce, delay, limit, reduce or terminate its research and development activities, preclinical studies, clinical trials or other development activities and future commercialization efforts, or grant rights to develop and market product candidates that it would otherwise develop and market itself. In addition, attempting to secure additional financing may divert the time and attention of its management from daily activities and distract from its research and development efforts.
Palvella’s development funding agreement with Ligand obligates it to make certain milestone payments, some of which will be triggered prior to its commercialization of any of its product candidates.
Certain of the milestone payments payable by Palvella in connection with the Ligand Agreements are due upon events that will occur prior to its planned commercialization of its lead product candidate, QTORIN rapamycin. Accordingly, Palvella may be required to make payments in an aggregate amount of up to $5.0 million prior to the time at which it is able to generate revenue, if any, from sales of QTORIN rapamycin for any indication, if approved. There can be no assurance that Palvella will have the funds necessary to make such payments, or be able to raise such funds when needed, on terms acceptable to it, or at all. In order to make the required payments when due, Palvella may be required to divert its capital resources by delaying, limiting, reducing or terminating its product development or future commercialization efforts, or it may have to grant rights to develop and market product candidates that it would otherwise develop and market itself. If Palvella is required to raise funds but is unable to do so, or if it is unable to otherwise maintain sufficient liquidity to make its payment obligations if and when they become due, it may be in material breach of the Ligand Agreements, and Ligand may seek legal action or remedies against Palvella (including by seeking to terminate the Ligand Agreements), which would harm its business, financial condition, results of operations and prospects. If Palvella is able to raise funds, it may not be able to do so on terms that are favorable to it, and its existing stockholders may experience substantial dilution, it may agree to certain covenants limiting or restricting its ability to take specific actions, or it may have to relinquish valuable rights to its technologies, future revenue streams, research programs or product candidates.
Palvella’s ability to utilize its NOL carryforwards and certain other tax attributes may be limited.
Palvella’s federal NOL carryforwards may be unavailable to offset future taxable income because of restrictions under U.S. tax law. Palvella’s federal NOL carryforwards generated in tax years beginning after December 31, 2017 may be carried forward indefinitely, but for taxable years beginning after December 31, 2020, the deductibility of such federal NOL carryforwards is limited to 80% of Palvella’s current year taxable income. It is uncertain if and to what extent limitations under state law may differ. As of December 31, 2023, Palvella had federal NOL carryforwards of approximately $36.7 million, which are available to reduce future federal taxable purposes and have an indefinite carryforward. Palvella has NOLs for state income tax purposes of $37.6 million, which are available to reduce future state taxable income through 2038.
In addition, as noted above, under Sections 382 and 383 of the Code, if a corporation undergoes an “ownership change” (generally defined as a cumulative change in the corporation’s ownership by “5-percent shareholders” that exceeds 50 percentage points over a rolling three-year period), the corporation’s ability to use its pre-change NOL carryforwards and certain other pre-change tax attributes to offset its post-change taxable income may be limited. Similar rules may apply under state tax laws. Palvella may have experienced such ownership changes in the past, and it may experience ownership changes in the future as a result of the Merger and the PIPE financing or subsequent shifts in its stock ownership. Palvella has not conducted any studies to determine annual limitations, if any, that could result from such changes in ownership. There is also a risk that due to regulatory changes, such as suspensions on the use of NOL carryforwards, or other unforeseen reasons, Palvella’s existing NOL carryforwards could expire or otherwise be unavailable to offset future income tax liabilities. Because Palvella’s ability to utilize its NOL carryforwards and certain other tax attributes could be limited as described above, Palvella may not be able to utilize a material portion of its NOL carryforwards and certain other tax attributes, which could have a material adverse effect on its cash flows and results of operations.
Risks Related to the Discovery, Development, Regulatory Approval and Commercialization of Palvella’s Product Candidates
Clinical drug development is a lengthy, complex and expensive process, with an uncertain outcome. Palvella may incur additional costs or experience delays in completing, or ultimately be unable to complete, the development of its product candidates.
Palvella’s lead product candidate, QTORIN rapamycin, is in clinical development and the risk of failure is high. It is impossible to predict when or if any of Palvella’s product candidates will prove effective and safe in humans or will receive regulatory approval. Before obtaining marketing approval from regulatory authorities for the sale of any product candidate, Palvella must complete preclinical development and then conduct extensive clinical trials to demonstrate the safety and efficacy of its product candidates in humans. Clinical testing is expensive, difficult to design and implement, can take many years to complete and is inherently uncertain as to outcome. Clinical trial failure may result from a multitude of factors including flaws in trial design, carryover effect, dose selection, placebo effect, patient enrollment criteria and failure to demonstrate favorable safety or efficacy, and failure in clinical trials can occur at any stage. For example, Palvella’s Phase 2b clinical trial of QTORIN rapamycin in patients with Gorlin Syndrome and Phase 3 clinical trials of QTORIN rapamycin in patients with pachyonychia congenita failed to meet their respective primary endpoints.
Palvella is currently conducting its Phase 3 clinical trial of QTORIN rapamycin for the treatment of microcystic LM, for which it expects to report top-line data in the first quarter of 2026. Palvella is also planning to conduct a Phase 2 clinical trial of QTORIN rapamycin for the treatment of cutaneous venous malformations, for which it expects to report top-line data in the fourth quarter of 2025. Palvella’s other programs under evaluation for the treatment of other serious, rare genetic skin diseases and other genetic diseases are in early-stage preclinical development.
Palvella may experience numerous unforeseen events that could delay or prevent its ability to receive marketing approval for its product candidates, including:
|
● |
regulators or IRBs may not authorize it or its investigators to commence a clinical trial or conduct a clinical trial at a prospective trial site, or may halt or suspend an ongoing trial; |
|
● |
clinical trials of its product candidates may fail to show safety, efficacy or an acceptable benefit-risk profile, produce negative or inconclusive results, and Palvella may decide, or regulators may require it, to conduct additional nonclinical studies or clinical trials or abandon drug development programs; |
|
● |
the design of any of its clinical trials may be flawed, and those flaws may not become apparent until such clinical trial is well advanced or completed; |
|
● |
regulators may not agree with its selection of novel endpoints or other key clinical trial design features, such as choice of control, used in its clinical evaluation of its rare disease product candidates; for example, the FDA has commented that a placebo-controlled trial or additional trials assessing different clinical endpoints may be required to assess the efficacy of QTORIN rapamycin for the treatment of microcystic LM; |
|
● |
its third-party contractors may fail to comply with regulatory requirements or meet their contractual obligations to Palvella in a timely manner, or at all; |
|
● |
the number of subjects required for clinical trials of its product candidate may be larger than anticipated, enrollment in the clinical trials for its product candidates may be slower than Palvella anticipates, it may be difficult to identify and enroll suitable participants given the small patient populations of the diseases it is targeting, or participants may drop out of these clinical trials or fail to return for post-treatment follow-up at a higher rate than it anticipates; |
|
● |
its product candidates may have undesirable side effects or other unexpected characteristics, causing it or its investigators, regulators or IRBs to suspend or terminate the trials; |
|
● |
Palvella may experience delays in reaching, or fail to reach, agreement on acceptable clinical trial contracts or clinical trial protocols with prospective trial sites or prospective CROs, the terms of which can be subject to extensive negotiation and may vary significantly among different trial sites and CROs; the cost of clinical trials of its product candidates may be greater than Palvella anticipates, particularly if the FDA or other equivalent foreign regulatory authorities require post-marketing studies and/or a patient registry; and |
|
● |
the supply or quality of its product candidates or other materials necessary to conduct clinical trials of its product candidates may be insufficient or inadequate. |
If Palvella is required to conduct additional clinical trials or other testing of its product candidates beyond those that currently contemplated, if it is unable to successfully complete clinical trials of its product candidates or other testing in a timely manner, if the results of these trials or tests are not positive or are only modestly positive or if there are safety concerns, Palvella may incur unplanned costs, be delayed in seeking and obtaining regulatory approval, if it receives such approval at all, receive more limited or restrictive regulatory approval, be subject to additional post-marketing testing requirements. If Palvella experiences any delays in the completion of, or termination of, any clinical trial of its product candidates, the commercial prospects of its product candidates will be harmed, and its ability to generate product revenues from any of these product candidates may not be successful. In addition, any delays in completing its clinical trials will increase its costs, slow down its product candidate development process and jeopardize its ability to receive regulatory approval and commence product sales and generate revenues. Any of these occurrences could materially adversely affect its business, financial condition, results of operations and prospects.
Palvella’s future success is substantially dependent on the successful clinical development, regulatory approval and commercialization of QTORIN rapamycin, which is in later stages of development than its other product candidates.
Palvella currently has no products that are approved for commercial sale. Palvella is developing its lead product candidate, QTORIN rapamycin, for the treatment of two rare genetic skin diseases. Palvella is currently evaluating QTORIN rapamycin in patients with microcystic LM in its Phase 3 clinical trial. Palvella is also developing QTORIN rapamycin for patients with cutaneous venous malformations. Additionally, Palvella is developing other research-stage product candidates, but these product candidates are in earlier stages of development. Palvella expects that a substantial portion of its efforts and expenditures over the next few years will be devoted to the continued clinical evaluation of QTORIN rapamycin and the commercialization of this product candidate for the treatment of microcystic LM, following regulatory approval, if received. Accordingly, the success of its business, including its ability to finance its company and generate any revenue in the future, will primarily depend on the successful development, regulatory approval and commercialization of QTORIN rapamycin.
The clinical and commercial success of QTORIN rapamycin and any future product candidates will depend on many factors, including the following:
|
● |
the ability to raise any additional required capital on acceptable terms, or at all; |
|
● |
timely completion of its preclinical studies and clinical trials, which may be significantly slower or cost more than Palvella currently anticipates and may depend substantially upon the performance of certain third-party contractors; |
|
● |
the ability to demonstrate the safety, efficacy and acceptable benefit-risk profile of its product candidates to the satisfaction of the FDA and equivalent foreign regulatory authorities; |
|
● |
delays in developing and testing, or inability to develop and test, any clinical outcome assessments to the extent necessary for the FDA and equivalent foreign regulatory authorities to agree to their use as endpoints utilized in a clinical trial to support labeling claims; |
|
● |
the prevalence, duration and severity of potential side effects or other safety issues experienced with its product candidates, if any, or experienced by competitors who are developing topical rapamycin (also known as sirolimus) products or who are targeting the same indications in the rare genetic skin diseases space; |
|
● |
the timely receipt of necessary marketing approvals from the FDA and equivalent foreign regulatory authorities and, if granted, completion of any required post-marketing studies or trials and available funding to perform any such studies or trials; |
|
● |
the ability of any CMO, upon which Palvella relies to manufacture clinical and commercial supplies of its product candidates or any future product candidates to remain in good standing with relevant regulatory authorities and to develop, validate and maintain commercially viable manufacturing processes that are compliant with current good manufacturing practices, or “cGMP”; |
|
● |
its ability to successfully develop a targeted rare disease commercial strategy and thereafter establish sales, marketing and distribution capabilities to launch and commercialize its product candidates in the United States and internationally, if approved for marketing, reimbursement, sale and distribution in such countries and territories; |
|
● |
acceptance by physicians, payors and patients of the benefits, safety and efficacy of its product candidates or any future product candidates, if approved; |
|
● |
obtainment and maintenance of coverage, adequate pricing and adequate reimbursement from third-party payors, including government payors; |
|
● |
its ability to retain subjects who have enrolled in a clinical study but may be prone to withdraw due to the rigors of the clinical trial, lack of efficacy, side effects, personal issues or loss of interest; |
|
● |
the size of the potential markets for its rare disease product candidates, if approved; and |
|
● |
its ability to establish and enforce intellectual property rights in and to its product candidates or any future product candidates. |
Even if Palvella completes clinical testing and receives approval from the FDA or applicable foreign agencies for QTORIN rapamycin, the FDA or the equivalent foreign regulatory authorities may grant approval or other marketing authorization contingent on the performance of costly additional clinical trials, including post- market clinical trials, or impose restrictions on the product’s distribution in the form of a REMS. The FDA or the equivalent foreign regulatory authority may also approve QTORIN rapamycin for a more limited indication or a narrower patient population than Palvella originally requested. In addition, the FDA or the equivalent foreign regulatory authority may not approve QTORIN rapamycin with the labeling that Palvella believes is necessary or desirable, or may approve it with labeling that includes warnings or precautions or limitations of use that may not be desirable, for the successful commercialization of QTORIN rapamycin.
The factors outlined above, many of which are beyond its control, could cause Palvella to experience significant delays or affect its ability to obtain regulatory approvals or commercialize QTORIN rapamycin. If Palvella is unable to obtain regulatory approval and successfully commercialize its product candidates, its financial position will be materially adversely affected, and Palvella may not be able to generate sufficient revenue to continue its business.
Palvella may be unable to obtain regulatory approval for its product candidates under applicable regulatory requirements. The denial or delay of any such approval would adversely impact its potential to generate revenue, its business and its results of operations.
The research, testing, manufacturing, labeling, licensure, sale, marketing and distribution of biopharmaceutical products are subject to extensive regulation by the FDA and comparable regulatory authorities in the United States and other countries, and such regulations differ from country to country. Palvella is not permitted to market its product candidates in any jurisdiction until it receives the requisite marketing approval from the applicable regulatory authorities of such jurisdictions. To gain approval to market its product candidates, Palvella must provide the FDA and foreign regulatory authorities with preclinical, manufacturing and clinical data that adequately demonstrate the safety and efficacy of the product for the intended indication applied for in the applicable regulatory filing. The approval process is typically lengthy and expensive, and approval is never certain.
Palvella expects to report top-line data from its Phase 3 trial of QTORIN rapamycin for the treatment of microcystic LM in the first quarter of 2026. Data are often susceptible to varying interpretations and analyses, and many companies that have believed their product candidates performed satisfactorily in clinical trials have nonetheless failed to obtain marketing approval of their drugs.
The FDA or any foreign regulatory authorities can delay, limit or deny approval of QTORIN rapamycin for the treatment of microcystic LM or any future product candidates for many additional reasons, including:
|
● |
the FDA or other equivalent foreign regulatory authorities may disagree with the number, design, size, conduct or implementation of Palvella’s clinical trials; |
|
● |
Palvella’s inability to demonstrate to the satisfaction of the FDA or the equivalent foreign regulatory authority that any of its product candidates are safe and effective for the requested indication; |
|
● |
the results of Palvella’s clinical trials may not meet the level of statistical significance or clinical meaningfulness or establish an acceptable benefit-risk profile required by the FDA or other equivalent foreign regulatory authorities for marketing approval; |
|
● |
the FDA or other equivalent foreign regulatory authorities may not accept data generated from Palvella’s clinical trial sites; |
|
● |
the FDA or other equivalent foreign regulatory authorities may find the chemistry, manufacturing and controls, or “CMC”, data insufficient to support the quality of Palvella’s product candidates; |
|
● |
the FDA or other equivalent foreign regulatory authorities may identify deficiencies in the manufacturing processes or facilities of Palvella’s CMOs; |
|
● |
the FDA or other equivalent foreign regulatory authorities may disagree with Palvella’s assessment that the delivery device component associated with its QTORIN platform is a Class I device exempt from premarket notification requirements as well as Quality System Regulation; |
|
● |
the FDA or equivalent foreign regulatory authorities may not approve the formulation, dosing, labeling or specifications; or |
|
● |
the potential for approval policies or regulations of the FDA or the equivalent foreign regulatory authorities to significantly change in a manner rendering Palvella’s data insufficient for approval or invalidated. |
Any of these factors, many of which are beyond Palvella’s control, may result in its failing to obtain regulatory approval to market any of its product candidates, which could materially adversely affect its business, financial condition, results of operations and prospects.
Success in preclinical studies or earlier clinical trials may not be indicative of results in future clinical trials.
Results from preclinical studies or early clinical trials are not necessarily predictive of future clinical trial results and are not necessarily indicative of final results. Palvella’s product candidates may fail to show the desired characteristics in clinical development sufficient to obtain regulatory approval, despite positive results in preclinical studies or having successfully advanced through earlier clinical trials. The results of nonclinical studies and early clinical trials of QTORIN rapamycin or any future product candidates may not be predictive of the results of later-stage clinical trials. Differences in trial design between early-stage clinical trials and later-stage clinical trials make it difficult to extrapolate the results of earlier clinical trials to later clinical trials. For example, Palvella announced topline results from its Phase 2 study of QTORIN rapamycin in patients with microcystic LM where, as is common in Phase 2 studies, efficacy was evaluated as secondary endpoints without multiplicity adjustment or statistical analyses, and the results from this study may not be predictive of results in its ongoing Phase 3 study in microcystic LM where a single hypothesis will be tested as the primary endpoint. Many companies in the pharmaceutical and biotechnology industries have suffered significant setbacks in late-stage clinical trials even after achieving promising results in preclinical testing and earlier-stage clinical trials. Moreover, even though Palvella is using and plans to use the same formulation of QTORIN rapamycin to support multiple investigational development programs in multiple product candidates, it cannot be certain that any success Palvella has with respect to the development of QTORIN rapamycin for the treatment of microcystic LM or for the treatment of cutaneous venous malformations will lead to the successful development of additional product candidates.
In addition, the design of a pivotal clinical trial can determine whether its results will support approval of a product and flaws in the design of a clinical trial may not become apparent until the clinical trial is well advanced or completed. Palvella has limited experience in designing and conducting clinical trials and it may be unable to successfully design and execute a clinical trial to support regulatory approval.
Palvella is currently conducting a Phase 3 clinical trial of QTORIN rapamycin for the treatment of microcystic LM, for which it expects to report top-line data in the first quarter of 2026. Even if the trial design in Palvella’s Phase 3 clinical trial of QTORIN rapamycin for the treatment of microcystic LM proves successful, Palvella may be unable to duplicate these results in other clinical trials it may conduct. Additionally, even if the FDA or other regulatory authorities accept the novel clinical endpoints Palvella establishes in connection with its Phase 3 trial in microcystic LM, there are no assurances that the FDA or other regulatory authorities will find the efficacy endpoints Palvella proposes in its future pivotal clinical trials to be sufficiently developed and tested and clinically meaningful, or that its product candidates will achieve the pre-specified endpoints in future pivotal clinical trials to a degree of statistical significance. For example, the FDA has commented that a placebo-controlled trial or additional trials assessing different clinical endpoints may be required to assess the efficacy of QTORIN rapamycin for the treatment of microcystic LM.
The rare genetic skin diseases Palvella is currently targeting have no FDA-approved therapies, which subjects the design and execution of its clinical development program to complexities and known and unknown risks, including those related to novel and/or subjective clinical endpoints and varying patient population characteristics.
There are currently no FDA-approved therapies indicated for the treatment of microcystic LM or cutaneous venous malformations. Palvella has concentrated its current research and development efforts on developing effective therapies for these indications, in addition to other rare genetic skin diseases and rare genetic conditions in other disease areas, and its future success depends on the success of this approach. The clinical trial requirements of the FDA and other comparable regulatory agencies and the criteria these regulators use to determine the safety and efficacy of any product candidate vary substantially according to the type, complexity, novelty and intended use and market of the potential product. Given the nature of the genetic skin diseases Palvella is targeting, the design and execution of its clinical development program is subject to both known and unknown risks.
As with QTORIN rapamycin for the treatment microcystic LM or cutaneous venous malformations and any future product candidates that may require Palvella to use new or novel endpoints or methodologies, the FDA or other regulatory authorities may not consider the endpoints of its clinical trials to provide clinically meaningful results. Even if applicable regulatory authorities do not object to its proposed endpoints in an earlier-stage clinical trial, such regulatory authorities may require evaluation of additional or different clinical endpoints in later-stage clinical trials or may not accept the clinical endpoints evaluated in later-stage clinical trials. For example, while the primary endpoint in the Phase 3 clinical trial of QTORIN rapamycin for the treatment of microcystic LM employs a dynamic assessment that uses a comparative rating scale, which was also assessed as one of several efficacy endpoints in the Phase 2 study in microcystic LM, the FDA has recommended that primary efficacy in the treatment of microcystic LM be evaluated on a static multicomponent assessment scale but recommended that Palvella provide a rationale for selecting the comparative rating scale should Palvella proceed with a comparative rating scale. If the FDA does not agree with Palvella’s primary endpoint, the FDA may instead consider the Phase 3 clinical trial’s key secondary endpoint, which is a static multicomponent assessment scale, as pivotal to assessing efficacy, if alpha-protected. Alternatively, the FDA may consider the study to not be adequate and well-controlled and could request additional clinical trials to assess a static multicomponent assessment scale as the primary endpoint. As a result, the design and conduct of its ongoing clinical trials and any future product candidates may take longer, be more costly or be less effective.
Further, different results may be achieved depending upon the characteristics of the population enrolled in its studies and which analysis population is used to analyze results. As a result, there is no guarantee that its clinical trials will produce statistically significant results with respect to subject-reported outcomes, and there can be no guarantee that the characteristics of the population enrolled in its clinical trials does not adversely impact the results reported for such trials.
Any delays in, or the denial of, approval of any of its product candidates resulting from its inability to establish effective trial designs for rare genetic skin diseases could materially adversely affect its business, financial condition, results of operations and prospects.
Palvella’s lead product candidates are based on its QTORIN platform and it is highly dependent on the successful development of this novel and unproven technology.
Palvella’s proprietary QTORIN platform was developed over several years of research to overcome inherent challenges, including chemical stability, skin penetration and skin distribution, with topical delivery of mTOR inhibitors, such as rapamycin and other therapeutic agents. QTORIN is an anhydrous gel comprising excipients intentionally selected in a ratio designed to achieve drug stability at room temperature and enable cutaneous distribution of therapeutics levels of cargoes into the target cells in the basal layer of the epidermis and to the dermis. Palvella’s product candidate for the treatment of microcystic LM and the treatment of cutaneous venous malformations leverages QTORIN as a mechanism of delivery of a 3.9% concentration of rapamycin to treat the applicable disease.
QTORIN is the platform for Palvella’s current clinical-stage product candidates and for other research-stage product candidates in its pipeline, and accordingly, its future success depends in significant part on the successful development of this novel technology. Negative results in the development of QTORIN rapamycin for either the treatment of microcystic LM or cutaneous venous malformations may also impact its ability to obtain regulatory approval for other product candidates which Palvella expects to develop based on its QTORIN platform, either at all or within anticipated timeframes because, although Palvella may be targeting different indications, the underlying technology platform is the same for each product candidate and there may be commonalities in the manufacturing and development processes. Accordingly, a failure in any one QTORIN-based program may decrease trust in its technology and affect its ability to conduct clinical programs for other QTORIN-based product candidates.
Palvella has not yet succeeded and may not succeed in completing clinical development of or obtaining regulatory approval for any of its product candidates using QTORIN. As a result, it is more difficult for it to predict whether the application of its QTORIN platform, or any similar or competitive platforms, will result in the development and marketing approval of any products. Any developmental problems Palvella experiences in the future related to its QTORIN platform or any of its research programs may cause significant delays or unanticipated costs or may prevent the development of a commercially viable product. Any of these factors may prevent Palvella from completing its preclinical studies or clinical trials or commercializing any product candidates it may develop on a timely or profitable basis, if at all, which could materially adversely affect its business, financial condition, results of operations and prospects.
Palvella may find it difficult to enroll subjects in its clinical trials, which could delay or prevent it from proceeding with clinical trials of its product candidates.
Identifying and qualifying subjects to participate in clinical trials of its product candidates is critical to Palvella’s success. The timing of its clinical trials depends on its ability to recruit subjects to participate, as well as the completion of required follow-up periods. Patients may be unwilling to participate in its clinical trials because of negative publicity from adverse events related to the biotechnology or pharmaceutical fields, competitive clinical trials for similar patient populations, the existence of current treatments or for other reasons outside of Palvella’s control. The timeline for recruiting subjects, conducting studies and obtaining regulatory approval of its product candidates may be delayed, which could result in increased costs, delays in advancing its product candidates, delays in testing the effectiveness of its product candidates or termination of the clinical trials altogether.
Importantly, the indications that Palvella is currently targeting and may in the future target are rare diseases, which may limit the pool of subjects that may be enrolled in its ongoing or planned clinical trials. To the extent Palvella’s clinical trials are limited to specific genotypes, the population of eligible trial participants is even further limited. Microcystic LM affects an estimated greater than 30,000 diagnosed patients in the United States. cutaneous venous malformations affects an estimated greater than 75,000 people in the United States. Some of the other diseases Palvella intends to target have similarly limited patient populations. Palvella expects to rely in part on its relationships with patient advocacy groups to assist in identifying eligible patients, and any deterioration of those relationships could impede its ability to successfully enroll patients in its clinical trials. Palvella may not be able to initiate or continue clinical trials for its product candidates if it is unable to locate and enroll a sufficient number of eligible subjects to participate in these trials as required by the FDA or similar regulatory authorities outside of the United States.
Subject enrollment and trial completion are affected by numerous factors, including the:
|
● |
size and nature of the target population and the process for identifying subjects; |
|
● |
design of the trial protocol; |
|
● |
eligibility and exclusion criteria for the trial; |
|
● |
safety profile, to date, of the product candidate under study; |
|
● |
perceived risks and benefits of the product candidate under study; |
|
● |
availability of competing therapies and clinical trials for similar product candidates or targeting subjects meeting our trial eligibility criteria; |
|
● |
severity of the disease under investigation; |
|
● |
availability of genetic testing for potential patients; |
|
● |
proximity and availability of clinical trial sites for prospective subjects; |
|
● |
ability to obtain and maintain subject consent; |
|
● |
risk that enrolled subjects will drop out before completion of the trial; |
|
● |
patient referral practices of physicians; and |
|
● |
ability to monitor subjects adequately during and after treatment. |
If Palvella has difficulty enrolling a sufficient number of subjects to conduct its clinical trials as planned, it may need to delay, limit or terminate ongoing or planned clinical trials, any of which would have an adverse effect on its business, financial condition, results of operations and prospects.
Palvella may be unable to obtain Orphan Drug Designation for certain of its product candidates and, even if it obtains such designation, it may not be able to realize the benefits of such designation, including potential marketing exclusivity of its product candidates, if approved.
Regulatory authorities in some jurisdictions, including the United States and other major markets, may designate drugs intended to treat conditions or diseases affecting relatively small patient populations as orphan drugs. Under the Orphan Drug Act of 1983, the FDA may designate a product candidate as an orphan drug if it is intended to treat a rare disease or condition, which is generally defined as having a patient population of fewer than 200,000 individuals in the United States, or a patient population greater than 200,000 in the United States where there is no reasonable expectation that the cost of developing the drug will be recovered from sales in the United States. In the European Union, the EMA Committee for Orphan Medicinal Products grants Orphan Drug Designation to promote the development of products that are intended for the diagnosis, prevention or treatment of a life-threatening or chronically debilitating disease affecting not more than five in 10,000 persons. Additionally, Orphan Drug Designation is granted by the EMA for products intended for the diagnosis, prevention or treatment of a life-threatening, seriously debilitating or serious and chronic disease and when, without incentives, it is unlikely that sales of the drug in the European Union would be sufficient to justify the necessary investment in developing the drug or biologic product.
Palvella has received Orphan Drug Designation for QTORIN rapamycin for the treatment of microcystic LM from the FDA and EMA. If Palvella requests Orphan Drug Designation or the foreign equivalent for any of its other or future product candidates, there can be no assurances that the FDA or foreign regulatory authorities will grant any of its product candidates such designation. Additionally, the designation of any of its product candidates as an orphan product does not mean that any regulatory agency will accelerate regulatory review of, or ultimately approve, that product candidate, nor does it limit the ability of any regulatory agency to grant Orphan Drug Designation to product candidates of other companies that treat the same indications as its product candidates prior to its product candidates receiving exclusive marketing approval.
Generally, if a product candidate with an Orphan Drug Designation receives the first marketing approval for the indication for which it has such designation, the product is entitled to a period of marketing exclusivity, which precludes the FDA or foreign regulatory authorities from approving another marketing application for a product that constitutes the same drug treating the same indication for that marketing exclusivity period, except in limited circumstances. If another sponsor receives such approval before Palvella does (regardless of its Orphan Drug Designation), Palvella will be precluded from receiving marketing approval for its product for the applicable exclusivity period unless FDA concludes that its drug is not the same drug or is clinically superior in that it is shown to be safer, more effective or makes a major contribution to patient care. The applicable exclusivity period is seven years in the United States and 10 years in the European Union. The exclusivity period in the European Union can be reduced to six years if a product no longer meets the criteria for Orphan Drug Designation or if the product is sufficiently profitable so that market exclusivity is no longer justified. Orphan drug exclusivity may be revoked if any regulatory agency determines that the request for designation was materially defective or if the manufacturer is unable to assure sufficient quantity of the product to meet the needs of patients with the rare disease or condition.
Even if Palvella obtains orphan drug exclusivity for a product candidate, that exclusivity may not effectively protect the product candidate from competition because different drugs can be approved for the same condition in the United States. Even after an orphan drug is approved, the FDA may subsequently approve another drug for the same disease if the FDA concludes that the latter drug is not the same drug or is clinically superior. In the European Union, marketing authorization may be granted to a similar medicinal product for the same orphan indication if the second applicant can establish in its application that its medicinal product, although similar to the orphan medicinal product already authorized, is safer, more effective or otherwise clinically superior; if the holder of the marketing authorization for the original orphan medicinal product consents to a second orphan medicinal product application; or if the holder of the marketing authorization for the original orphan medicinal product cannot supply sufficient quantities of orphan medicinal product.
Palvella’s inability to obtain Orphan Drug Designation or the foreign equivalent for its product candidates, or to realize the benefits of such designation, could have an adverse effect on its business, financial condition, results of operations and prospects.
Palvella is targeting rare genetic skin diseases, and the small patient populations associated with such diseases present additional risks with respect to clinical development, regulatory approvals and commercialization of product candidates.
Palvella’s approach of targeting genetic skin diseases present risks related to the clinical development, regulatory approval and commercialization of its product candidates, including the following:
|
● |
it may be difficult to establish safety and efficacy in these types of patient populations given there is less known of the natural history of the disease; |
|
● |
Palvella expects to face challenges with respect to patient enrollment in its clinical trials, as described above; |
|
● |
small sample sizes in its clinical trials suggest that Palvella faces the risk of substantial variability in the results of its trials, and so the outcome of nonclinical testing and early clinical trials is less likely to be predictive of the success of later-stage clinical trials; |
|
● |
following approval of its product candidates, if any, pricing and level of reimbursement may not be sufficient to offset costs of development, manufacturing, marketing, and commercialization; and |
|
● |
market size is a significant variable in disease indications classified as rare. Palvella’s projections of both the number of people who have these diseases, as well as the subset of people with these diseases who have the potential to benefit from treatment with product candidates it may develop, are based on estimates. These estimates have been derived from a variety of sources, including scientific literature, surveys of clinics, patient advocacy groups or market research. These estimates may prove to be incorrect and new studies may change the estimated incidence or prevalence of these diseases. The number of patients in the United States, Europe and elsewhere may turn out to be lower than expected, and patients may not be amenable to treatment with its product candidates Palvella may develop or may become increasingly difficult to identify or gain access to. Accordingly, even if Palvella obtains significant market share for its product candidates, because the potential target populations are small, Palvella may never achieve profitability without obtaining regulatory approval for additional indications. |
Adverse developments with respect to any of the foregoing could result in significant changes in Palvella’s business plan and have a material adverse effect on its business, financial condition, results of operations, and prospects.
Palvella’s development and commercialization strategy for its product candidates depends, in part, on published scientific literature and the FDA’s prior findings regarding the safety and efficacy of rapamycin. If Palvella is not able to pursue this strategy, it may be delayed in receiving regulatory authority approval.
The Hatch-Waxman Amendments added Section 505(b)(2) to the U.S. Federal Food, Drug, and Cosmetic Act, or “FDCA”. Section 505(b)(2) permits the submission of an NDA, where at least some of the information required for approval comes from investigations that were not conducted by or for the applicant and for which the applicant has not obtained a right of reference or use from the person by or for whom the investigations were conducted. The FDA interprets Section 505(b)(2) of the FDCA, for purposes of approving an NDA, to permit the applicant to rely, in part, upon published literature and/or the FDA’s previous findings of safety and efficacy for an approved product. The FDA also requires companies to perform additional clinical trials or measurements to support any deviation from the previously approved product and to justify that it is scientifically appropriate to rely on the applicable published literature or referenced product, referred to as bridging. The FDA may then approve the new product candidate for all or some of the indications for which the referenced product has been approved, as well as for any new indication sought by the Section 505(b)(2) applicant, if such approval is supported by study data. The labeling, however, may be required to include all or some of the limitations, contraindications, warnings or precautions or restrictions on use included in the reference product’s labeling, including a boxed warning, or may require additional limitations, contraindications, warnings or precautions or restrictions on use.
Palvella currently plans to pursue marketing approval for QTORIN rapamycin for several indications in the United States through Section 505(b)(2) NDAs and will be completing bridging analyses comparing QTORIN rapamycin to the approved oral rapamycin product, a previously approved organ rejection prophylactic, prior to NDA submission. If the FDA disagrees with Palvella’s conclusions regarding the appropriateness of its reliance on the FDA’s prior findings of safety and efficacy for the approved oral rapamycin productor on published literature, or if Palvella is not otherwise able to bridge to the listed drug or published literature to demonstrate that its reliance is scientifically appropriate, Palvella could be required to conduct additional nonclinical toxicology, clinical safety or efficacy trials, or other studies to support its NDA, which could lead to unanticipated costs and delays or to the termination of its development programs. For example, while Palvella plans to bridge QTORIN rapamycin and the approved oral rapamycin product based on cross-study comparison between pharmacokinetic data from the prescribing information for the approved product, the FDA recommends that bridging to support an NDA for the treatment of microcystic LM be done in a relative bioavailability study comparing the pharmacokinetics of a topical product applied under maximal use conditions and the approved oral drug. The planned cross study analysis allows for comparison of systemic pharmacokinetic parameters, key criteria for assessing the applicability of safety findings from the listed drug, which are a result of systemic exposure from the oral formulation. If the FDA does not agree with Palvella’s pharmacokinetic approach, Palvella may need to conduct a relative bioavailability study, which compares direct assessment of pharmacokinetics of both products administered under similar conditions. For example, FDA may request different specific criteria for comparisons that cannot be evaluated based on limitations in the pharmacokinetic data available in the prescribing information of the approved drug. If Palvella is unable to obtain approval for its product candidates through the Section 505(b)(2) NDA process, it may be required to pursue the more expensive and time consuming 505(b)(1) approval process, which consists of full reports of investigations of safety and effectiveness conducted by or for the applicant. Even if Palvella is allowed to pursue the Section 505(b)(2) regulatory pathway to FDA approval, Palvella cannot assure you that its product candidates will receive the requisite approvals for commercialization.
The validity, scope and enforceability of any patents that Palvella may list in the Orange Book that cover QTORIN rapamycin, if approved by the FDA for any indication, can be challenged by competitors.
If QTORIN rapamycin is approved by the FDA for any indication, one or more third parties may challenge the patents covering QTORIN rapamycin with respect to such indication, which could result in the invalidation of, or render unenforceable, some or all of the relevant patent claims or a finding of non-infringement. For example, if a third party files an Abbreviated New Drug Application, or “ANDA”, for a generic drug bioequivalent to its QTORIN rapamycin, and relies in whole or in part on studies conducted by or for Palvella, the third party will be required to certify to the FDA that either: (1) there is no patent information listed in the FDA’s Orange Book with respect to its NDA for the applicable approved drug candidate; (2) the patents listed in the Orange Book have expired; (3) the listed patents have not expired, but will expire on a particular date and approval is sought after patent expiration; or (4) the listed patents are invalid or will not be infringed by the manufacture, use or sale of the third party’s generic drug. Alternatively, a third party that files an ANDA for a generic drug bioequivalent to QTORIN rapamycin may elect to submit a “section viii” statement certifying that its proposed label does not contain (or carves out) any language regarding the patented method of use rather than certify to a listed method of use patent. This section viii statement does not require notice to the patent holder or NDA owner. A certification that the new drug will not infringe the Orange Book-listed patents for the applicable approved drug candidate, or that such patents are invalid, is called a paragraph IV certification. If the third party submits a paragraph IV certification to the FDA, a notice of the paragraph IV certification must also be sent to Palvella once the third party’s ANDA is accepted for filing by the FDA. Palvella may then initiate a lawsuit to defend the patents identified in the notice. The filing of a patent infringement lawsuit within 45 days of receipt of the notice automatically prevents the FDA from approving the third party’s ANDA until the earliest of 30 months or the date on which the patent expires, the lawsuit is settled, or the court reaches a decision in the infringement lawsuit in favor of the third party. If Palvella does not file a patent infringement lawsuit within the required 45-day period, the third party’s ANDA will not be subject to the 30-month stay of FDA approval. Litigation or other proceedings to enforce or defend intellectual property rights are often very complex in nature, may be very expensive and time-consuming, may divert its management’s attention from its core business, and may result in unfavorable results that could limit its ability to prevent third parties from competing with its product candidates.
Other companies could receive FDA approval for a topical rapamycin product before Palvella receives FDA approval for QTORIN rapamycin for microcystic LM or cutaneous venous malformations, and thus could be granted regulatory exclusivity that could significantly delay Palvella’s ability to receive approval for and commercialize its QTORIN rapamycin and therefore dramatically reduce its market potential.
Other companies may submit a Section 505(b)(2) NDA and receive approval for a topical rapamycin product candidate prior to the approval of Palvella’s NDA for QTORIN rapamycin for the treatment of microcystic LM or for other indications Palvella is pursuing or may pursue in the future. The first approved Section 505(b)(2) product for a particular condition of use or change to a marketed product, such as a new formulation for a previously approved product, may be granted three-year exclusivity if one or more clinical studies, other than bioavailability or bioequivalence studies, was essential to the approval of the application and was conducted/sponsored by the applicant. The grant of three-year exclusivity can delay the FDA’s approval of other Section 505(b)(2) applicants for the same condition of use or change to the drug product, such as the first approval of a topical formulation of rapamycin, that was granted exclusivity, regardless of the date of submission of each NDA.
Palvella believes that other companies are developing topical rapamycin products. In order to obtain regulatory approval with a Section 505(b)(2) NDA, other companies would have to sponsor or conduct new clinical investigations (other than bioavailability studies) that are essential to approval of the application, as well as conduct the required bridging studies. If the FDA approves another company’s Section 505(b)(2) NDA for its topical rapamycin product, even for another indication, and grants the other company three-year exclusivity before Palvella receives approval for QTORIN rapamycin for the treatment of microcystic LM, the FDA may be precluded from approving any NDA Palvella may submit with respect to QTORIN rapamycin until after that three-year exclusivity period has expired unless Palvella pursues the more expensive and time consuming 505(b)(1) approval process, which would likely require that Palvella sponsors or conducts additional nonclinical and/or clinical studies. For example, upon approval of a Section 505(b)(2) NDA for the treatment of facial angiofibroma associated with tuberous sclerosis, Hyftor, a topical gel product containing sirolimus (also known as rapamycin), received three years of new product exclusivity. If another rapamycin topical product were to receive three-year exclusivity for a condition of use that overlaps with QTORIN rapamycin, approval of QTORIN rapamycin would be delayed until the expiration of such exclusivity.
It is also not uncommon for a sponsor of an approved product to file a citizen petition with the FDA seeking to delay approval of, or impose additional approval requirements for, pending competing products. If successful, such petitions can significantly delay, or even prevent, the approval of the new product. However, even if the FDA ultimately denies such a petition, the FDA may substantially delay approval while it considers and responds to the petition.
Any such delay could dramatically reduce its expected market potential for its QTORIN rapamycin for any disease indication and could materially adversely affect its business, financial condition, results of operations and prospects.
Serious adverse or unacceptable side effects may be identified during the development of its product candidates, which could prevent or delay regulatory approval and commercialization, increase its costs or necessitate the abandonment or limitation of the development of some of its product candidates.
As Palvella continues its development of its product candidates and initiate additional preclinical studies or clinical trials of these or future product candidates, if any, serious adverse events, unacceptable levels of toxicity, undesirable side effects or unexpected characteristics may emerge. Although Palvella has designed QTORIN rapamycin for topical application and in a manner that it believes will not result in systematic absorption, systematic exposure to rapamycin, the active ingredient in its lead product candidate, at levels consistent with the approved oral dosage form, is known to result in significant adverse reactions, including peripheral edema, hypertriglyceridemia, hypertension, hypercholesterolemia, creatinine increases, abdominal pain, diarrhea, headache, fever, urinary tract infection, anemia, nausea, arthralgia, pain and thrombocytopenia. Investigators may attribute infectious diseases occurring during clinical trials of QTORIN rapamycin to suspected or possible immunosuppression, based on the systematic mechanism of action of rapamycin. Further, Palvella has conducted and continues to conduct open-label studies of QTORIN rapamycin and, without a concurrent control arm, adverse events may be attributed to QTORIN rapamycin that may be a result of background disease or other external factors. Other active pharmaceutical ingredients Palvella selects for its product candidates may have similar adverse event profiles. The emergence of any such serious adverse events, unacceptable levels of toxicity, undesirable side effects or unexpected characteristics could cause difficulty recruiting and retaining participants for its trials or it may abandon these product candidates, institute burdensome monitoring programs or limit their development to more narrow uses, less frequent dosing, lower potency levels or subpopulations in which the serious adverse events, unacceptable levels of toxicity, undesirable side effects or other characteristics are less prevalent, less severe or more acceptable from a risk/benefit perspective. The FDA or an IRB, or equivalent foreign regulatory authorities, may also require that Palvella suspend, discontinue, or limit its clinical trials based on safety information or that there is inadequate prospect of treatment benefit. Such findings could further result in regulatory authorities failing to provide marketing authorization for its product candidates. Many product candidates that initially showed promise in early-stage testing have later been found to cause side effects that prevented further development of the product candidate.
Additionally, if one or more of its product candidates receives marketing approval, and Palvella or others identify undesirable side effects caused by such products, a number of potentially significant negative consequences could result, including:
|
● |
regulatory authorities may withdraw approvals of such product; |
|
● |
regulatory authorities may require additional warnings, precautions, or limitations of use in the labeling; |
|
● |
Palvella may be required to create a medication guide outlining the risks of such side effects for distribution to patients; |
|
● |
Palvella may be required to implement a REMS; |
|
● |
Palvella may be required to conduct additional clinical trials as post-marketing requirements; |
|
● |
Palvella could be sued and held liable for harm caused to patients; and |
|
● |
its reputation and physician or patient acceptance of its products may suffer. |
Any of these events could prevent Palvella from achieving or maintaining market acceptance of the particular product candidate, if approved, which could materially adversely affect its business, financial condition, results of operations and prospects.
Fast Track Designation granted for QTORIN rapamycin for the treatment of microcystic LM and, if granted, for any of Palvella’s other product candidates by the FDA may not lead to a faster development or regulatory review or approval process and does not increase the likelihood that its product candidates will receive marketing approval.
Palvella was granted Fast Track Designation by the FDA for QTORIN rapamycin for the treatment of microcystic LM and may seek such designation for QTORIN rapamycin for other indications, and for any other product candidates. If a drug is intended for the treatment of a serious or life-threatening disease and the drug demonstrates the potential to address unmet medical needs for this disease the drug sponsor may apply for Fast Track Designation. The FDA has broad discretion whether or not to grant this designation. Even if Palvella believes a particular product candidate is eligible for this designation, it cannot assure you that the FDA would decide to grant it. Even if Palvella does receive Fast Track Designation, as Palvella has for QTORIN rapamycin for the treatment of microcystic LM, it may not experience a faster development process, review or approval compared to conventional FDA procedures. The FDA may withdraw Fast Track Designation if it believes that the designation is no longer supported by data from its clinical development programs. Many drugs that have received Fast Track Designation have failed to obtain approval.
Breakthrough Therapy Designation granted for QTORIN rapamycin for the treatment of microystic LM and, if granted, for any of Palvella’s other product candidates by the FDA may not lead to a faster development or regulatory review or approval process and does not increase the likelihood that its product candidates will receive marketing approval.
Palvella was granted Breakthrough Therapy Designation by the FDA for QTORIN rapamycin for the treatment of microcystic LM and may seek such designation for QTORIN rapamycin for other indications, and for any other product candidates. A breakthrough therapy is defined as a drug that is intended, alone or in combination with one or more other drugs, to treat a serious or life-threatening disease or condition, and preliminary clinical evidence indicates that the drug may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. For drugs that have been designated as breakthrough therapies, interaction and communication between the FDA and the sponsor of the trial can help to identify the most efficient path for clinical development while minimizing the number of patients placed in ineffective control regimens. Drugs designated as breakthrough therapies by the FDA are also eligible for priority review if supported by clinical data at the time of the submission of the NDA.
Designation as a breakthrough therapy is at the discretion of the FDA. Accordingly, even if Palvella believes that one of its product candidates meets the criteria for designation as a breakthrough therapy, the FDA may disagree and instead determine not to make such designation. In any event, the receipt of a Breakthrough Therapy Designation for a drug may not result in a faster development process, review, or approval compared to drugs considered for approval under conventional FDA procedures and it would not assure ultimate approval by the FDA. In addition, even if one or more of Palvella’s product candidates qualify as breakthrough therapies, the FDA may later decide that the product candidate no longer meets the conditions for designation.
Interim, top-line or preliminary data from its clinical trials that Palvella announces or publishes from time to time may change as more patient data become available and are subject to audit and verification procedures that could result in material changes in the final data.
From time to time, Palvella may publish interim, top-line or preliminary data from its clinical trials. Interim data from clinical trials that Palvella may complete are subject to the risk that one or more of the clinical outcomes may materially change as patient enrollment continues and more patient data become available. Preliminary or top-line data also remain subject to audit and verification procedures that may result in the final data being materially different from the preliminary or top-line data Palvella previously published. For example, Palvella expects to report top-line data from its Phase 3 clinical trial for the treatment of microcystic LM in the first quarter of 2026, but any such data may change following further auditing. As a result, preliminary and top-line data should be viewed with caution until the final data are available. If the interim, top-line, or preliminary data that Palvella reports differ from actual results, or if others, including regulatory authorities, disagree with the conclusions reached, its ability to obtain approval for and commercialize its product candidates, its business, operating results, prospects or financial condition may be harmed. In addition, the information Palvella chooses to publicly disclose regarding a particular clinical trial is based on what is typically extensive information, and you or others may not agree with what Palvella determines is material or otherwise appropriate information to include in its disclosure.
Palvella may expend its limited resources to pursue a particular product candidate or indication and fail to capitalize on product candidates or indications that may be more profitable or for which there is a greater likelihood of success.
Because Palvella has limited financial and managerial resources, it focuses on research programs and product candidates that it identifies for specific indications. As a result, Palvella may forego or delay pursuit of opportunities with other product candidates or for other indications that later prove to have greater commercial potential. Palvella’s resource allocation decisions may cause it to fail to capitalize on viable commercial products or profitable market opportunities. Palvella’s spending on current and future research and development programs and product candidates for specific indications may not yield any commercially viable products. If Palvella does not accurately evaluate the commercial potential or target market for a particular product candidate, it may relinquish valuable rights to that product candidate through collaboration, licensing or other royalty arrangements in cases in which it would have been more advantageous for it to retain sole development and commercialization rights to such product candidate. Failure to allocate resources or capitalize on strategies in a successful manner will have an adverse impact on its business.
Palvella may choose not to continue developing or commercializing any of its product candidates at any time during development or after approval, which would reduce or eliminate its potential return on investment for those product candidates.
At any time, Palvella may decide to discontinue the development or commercialization of any of its product candidates during or after approval for a variety of reasons, including the appearance of new technologies that render its product obsolete, competition from a competing product or changes in or inability to comply with applicable regulatory requirements. If Palvella terminates a program in which Palvella has invested significant resources, it will not receive any return on its investment and it will have missed the opportunity to allocate those resources to potentially more productive uses.
If Palvella seeks to market any product candidates in its pipeline in countries other than the United States, it will need to comply with the regulations of each country in which it seeks to market its products. Additionally, although its trials are currently being conducted in the U.S., Palvella may conduct clinical trials for its product candidates at clinical trial sites outside the U.S. and the FDA and equivalent foreign regulatory authorities may not accept data from such sites.
None of its product candidates are currently approved for sale by any government authority. If Palvella fails to comply with regulatory requirements in any market it decides to enter, or to obtain and maintain required approvals, or if regulatory approvals in the relevant markets are delayed, its target market will be reduced and its ability to realize the full market potential of its product candidates, if approved, will be harmed. Marketing approval in one jurisdiction, including the United States, does not ensure marketing approval in another, but a failure or delay in obtaining marketing approval in one jurisdiction may have a negative effect on the regulatory process in others. Failure to obtain a marketing approval in countries in which Palvella seeks to market its products or any delay or setback in obtaining such approval would impair its ability to develop foreign markets for any of its products. Approval procedures vary among jurisdictions and can involve requirements and administrative review periods different from those in the United States, including additional nonclinical studies or clinical trials as clinical trials conducted in one jurisdiction may not be accepted by regulatory authorities in other jurisdictions. Obtaining foreign regulatory approvals and compliance with foreign regulatory requirements could result in significant delays, difficulties and costs for Palvella and could delay or prevent the introduction of its products in certain countries.
Additionally, although its trials are currently being conducted in the U.S., Palvella may in the future choose to conduct one or more of its clinical trials at clinical trial sites outside the United States, including in Canada and Europe. Although the FDA or equivalent foreign regulatory authority may accept data from clinical trial sites conducted outside the United States or the applicable jurisdiction, acceptance of such study data by the FDA or equivalent foreign regulatory authority may be subject to certain conditions. Where data from foreign clinical trial sites are intended to serve as the basis for marketing approval in the United States, the FDA will not approve the application on the basis of foreign data alone unless those data are applicable to the U.S. population and U.S. medical practice; the site study conduct was performed by clinical investigators of recognized competence; and the data are considered valid without the need for an on-site inspection by the FDA or, if the FDA considers such an inspection to be necessary, the FDA is able to validate the data through an on-site inspection or other appropriate means. Many foreign regulatory authorities have similar requirements. In addition, such foreign studies would be subject to the applicable local laws of the foreign jurisdictions where the studies are conducted. There can be no assurance the FDA or equivalent foreign regulatory authority will accept data from trials conducted outside of the United States or the applicable jurisdiction. If the FDA or equivalent foreign regulatory authority does not accept such data, it would likely result in the need for additional trials, which would be costly and time- consuming and delay aspects of its business plan.
Governments outside of the United States tend to impose strict price controls, which may adversely affect Palvella’s revenue, if any.
In some countries, particularly the countries of the European Union, the pricing of prescription pharmaceuticals is subject to governmental control. In these countries, pricing negotiations with governmental authorities can take considerable time after the receipt of marketing approval for a drug. In addition, there can be considerable pressure by governments and other stakeholders on prices and reimbursement levels, including as part of cost containment measures. Political, economic and regulatory developments may further complicate pricing negotiations. To obtain coverage and reimbursement or pricing approval in some countries, Palvella may be required to conduct a clinical trial that compares the cost-effectiveness of its product candidate to other available procedures. If reimbursement of its product candidates is unavailable or limited in scope or amount, or if pricing is set at unsatisfactory levels, it could have a material adverse effect on its business, financial condition, results of operations, and prospects.
Risks Related to Commercialization of Palvella’s Product Candidates, if Approved
Even if QTORIN rapamycin or any future product candidates receive marketing approval, they may fail to achieve market acceptance by physicians, patients, third-party payors or others in the medical community necessary for commercial success.
Even if QTORIN rapamycin for the treatment of microcystic LM or for the treatment of cutaneous venous malformations or any future product candidates receive marketing approval, they may nonetheless fail to gain sufficient market acceptance by physicians, patients, patient advocacy groups, third-party payors and others in the medical community. If Palvella’s product candidates do not achieve an adequate level of acceptance, it may never be able to generate adequate product revenue or become profitable. The degree of market acceptance of a product candidate, if approved for commercial sale, will depend on a number of factors, including but not limited to:
|
● |
the safety, efficacy, risk-benefit profile and potential advantages compared to alternative or existing treatments, which include, with respect to microcystic LM, surgery, sclerotherapy, laser and, cryotherapy, any of which physicians may perceive to be adequately effective or to present less risk for some or all patients; |
|
● |
the willingness of the target patient population to try new therapies and of physicians to prescribe these therapies over existing treatment alternatives; |
|
● |
support from patient advocacy groups; |
|
● |
side effects that may be attributable to Palvella’s product candidates and the difficulty of or costs associated with resolving such side effects; |
|
● |
the timing of market introduction of Palvella’s product candidates as well as competitive products; |
|
● |
the clinical indications for which a product candidate is approved; |
|
● |
restrictions on the use of Palvella’s product candidates in the labeling approved by regulatory authorities, including boxed warnings, contraindications or a REMS, which may not be required of alternative treatments and competitors’ products; |
|
● |
the potential and perceived advantages of Palvella’s product candidates over alternative treatments; |
|
● |
the effectiveness of Palvella’s sales, marketing and market access efforts; |
|
● |
the cost of treatment in relation to alternative treatments or methods of symptom management; |
|
● |
Palvella’s ability to offer its products, if approved, for sale at competitive prices; |
|
● |
the convenience and ease of administration compared to alternative treatments; |
|
● |
publicity relating to Palvella’s product candidates or those of its competitors; |
|
● |
the availability of third-party coverage and adequate reimbursement at any given price level of each of its product candidates and patients’ willingness to pay out-of-pocket in the absence of such coverage and adequate reimbursement; and |
|
● |
utilization controls imposed by third-party payors, such as prior authorizations and step edits. |
Palvella cannot assure you that its current or future product candidates, if approved, will achieve market acceptance among physicians, patients, patient advocacy groups, third-party payors or others in the dermatological community necessary for commercial success. Any failure by Palvella’s product candidates that obtain regulatory approval to achieve market acceptance or commercial success could materially adversely affect its business, financial condition, results of operations and prospects.
Palvella currently has no marketing and sales organization and has no experience as a company in commercializing products, and it may have to invest significant resources to develop these capabilities. If Palvella is unable to establish marketing and sales capabilities or enter into agreements with third parties to market and sell its product candidates if approved, it may not be able to generate product revenue.
Palvella has never commercialized a product. To achieve commercial success for any product for which Palvella obtains marketing approval, Palvella will need a sales and marketing organization. Palvella does not currently have any infrastructure for the sales, marketing, or distribution of any product, and the cost of establishing and maintaining such an organization may exceed the cost-effectiveness of doing so. In order to market QTORIN rapamycin or any other product candidate, if approved, Palvella must build its sales, distribution, marketing, managerial and other nontechnical capabilities or make arrangements with third parties to perform these services.
Palvella believes that it will be able to commercialize QTORIN rapamycin for the treatment of microcystic LM, if approved, with a specialized sales force that targets a focused subset of medical dermatologists, and is supported by sales management, medical liaisons, market access, an internal marketing group, and distribution support. There are significant expenses and risks involved with establishing Palvella’s own sales, marketing and distribution capabilities, including its ability to hire, retain and appropriately incentivize qualified individuals, provide adequate training to sales and marketing personnel, and effectively manage geographically dispersed sales and marketing teams to generate sufficient demand. Other factors that may inhibit Palvella’s efforts to commercialize its product candidates, once approved, include:
|
● |
the inability to recruit and retain adequate numbers of effective sales, marketing, coverage or reimbursement, customer service, medical affairs, and other support personnel; |
|
● |
the inability of sales personnel to obtain access to physicians or educate adequate numbers of physicians on the benefits of prescribing its product candidates; |
|
● |
its inability to maintain its collaborative relationships with patient advocacy groups and leverage those relationships to increase patient identification and outreach and the rate of new patient acceptance of its product candidates; |
|
● |
its inability to equip sales personnel with effective materials, including medical and sales literature to help them educate physicians and other healthcare providers regarding rare diseases and its future products; |
|
● |
the costs associated with training sales and marketing personnel on legal and regulatory compliance matters and monitoring their actions; |
|
● |
the clinical indications for which the products are approved and the claims that Palvella may make for the products; |
|
● |
the inability to price its products at a sufficient price point to ensure an adequate and attractive level of profitability; |
|
● |
limitations of use, contraindications, or warnings, including boxed warnings, contained in the products’ approved labeling; |
|
● |
any distribution and use restrictions imposed by the FDA or to which Palvella agrees as part of a mandatory REMS or voluntary risk management plan; |
|
● |
liability for sales or marketing personnel who fail to comply with the applicable legal and regulatory requirements; |
|
● |
the lack of complementary products to be offered by sales personnel, which may put it at a competitive disadvantage relative to companies with more extensive product lines; and |
|
● |
unforeseen costs and expenses associated with creating an independent sales and marketing organization or engaging a contract sales organization. |
If Palvella is unable to establish adequate sales, marketing, and distribution capabilities, either on its own or in collaboration with third parties, it will not be successful in commercializing any of its product candidates, if approved, and will not become profitable. Palvella may be competing with companies that currently have extensive and well-funded marketing and sales operations. Without an internal team or the support of a third party to perform marketing and sales functions, Palvella may be unable to compete successfully against these more established companies.
If Palvella enters into arrangements with third parties to perform sales, marketing and distribution services in the United States or in any markets outside of the United States, its revenues from product sales and its profitability, if any, may be lower than if it were to market, sell and distribute any products that it develops itself in all such territories. In addition, Palvella may not be successful in entering into arrangements with third parties to sell, market and distribute its product candidates, if approved, or may be unable to do so on terms that are acceptable to it. Palvella may have little control over such third parties, and any of them may fail to devote the necessary resources and attention to sell and market its products effectively. If Palvella does not establish sales, marketing and distribution capabilities successfully, either on its own or in collaboration with third parties, it will not be successful in commercializing its product candidates, if approved.
Palvella’s estimated market opportunities for its product candidates are subject to numerous uncertainties and may prove to be inaccurate. If Palvella has overestimated the size of its market opportunities, its future growth may be limited.
Palvella’s estimated addressable markets and market opportunities for its product candidates are based on a variety of inputs, including data published by third parties, including patient advocacy groups, its own market insights and internal market intelligence, and internally generated data and assumptions. Palvella has not independently verified any third-party information and cannot assure you of its accuracy or completeness. Market opportunity estimates, whether obtained or derived from third-party sources or developed internally, are subject to significant uncertainty and are based on assumptions and estimates that may not prove to be accurate. While Palvella believes its market opportunity estimates are reasonable, such information is inherently imprecise. In addition, Palvella’s assumptions and estimates of market opportunities are necessarily subject to a high degree of uncertainty and risk due to a variety of factors, including but not limited to those described in this “Risk Factors” section. If these third-party or internally generated data prove to be inaccurate or Palvella makes errors in its assumptions based on that data, its actual market may be more limited than its estimates. In addition, these inaccuracies or errors may cause Palvella to misallocate capital and other critical business resources, which could harm its business. The estimates of its addressable patient populations included in this proxy statement/prospectus should not be taken as indicative of its ability to grow its business. For more information regarding the estimates of patient populations included in this proxy statement/prospectus, see the sections titled “Market and Industry Data” and “Business—Its Pipeline.”
The size of the markets for Palvella’s product candidates have not been established and may be smaller than it estimates.
Palvella’s estimates of the annual total addressable markets for its product candidates are based on internal and third-party estimates, including, without limitation, estimated incidence and prevalence of these diseases, and estimated annual price per patient for its product candidates. While Palvella believes its assumptions and the data underlying its estimates are reasonable, these assumptions and estimates may not be correct and the conditions supporting its assumptions or estimates may change at any time, thereby reducing the predictive accuracy of these underlying factors. As a result, Palvella’s estimates of the annual total addressable market for its product candidates may prove to be incorrect. If the annual total addressable markets for its product candidates are smaller than Palvella has estimated, this may have an adverse effect on its business, financial condition, results of operations, and prospects.
If Palvella is unable to achieve and maintain coverage and adequate levels of reimbursement for any of its product candidates for which it receives regulatory approval, or any future products it may seek to commercialize, their commercial success may be severely hindered.
As to any of Palvella’s product candidates that become available by prescription only, its success will depend on the availability of coverage and adequate reimbursement for its product from third-party payors. Because there are currently no products approved for the treatment of microcystic LM, the pricing and reimbursement of its product candidates, if approved, is uncertain. Novel medical products, if covered at all, may be subject to enhanced utilization management controls designed to ensure that the products are used only when medically necessary. Such utilization management controls may discourage the prescription or use of a medical product by increasing the administrative burden associated with its prescription or creating coverage uncertainties for prescribers and patients. Moreover, Palvella’s target patient populations are small, as a result of which the pricing and third-party payor reimbursement of its product candidates, if approved, must be adequate to support commercial infrastructure. Eligibility for reimbursement does not imply that a medical product will be paid for in all cases or at a rate that covers its costs, including research, development, manufacture, sale and distribution. If Palvella is unable to obtain adequate levels of reimbursement, its ability to successfully market and sell its product candidates will be adversely affected.
Patients who are prescribed medicine for the treatment of their diseases generally rely on third-party payors to reimburse all or part of the costs associated with their prescription drugs. The availability of coverage and adequate reimbursement from governmental healthcare programs, such as Medicare and Medicaid, and private third-party payors is critical to new product acceptance. If any of Palvella’s product candidates fail to demonstrate attractive efficacy and safety profiles, they may not qualify for coverage and reimbursement. Even if Palvella obtains coverage for a given product, the resulting reimbursement payment rates might not be adequate, particularly given the small patient populations for its targeted indications, or may require co-payments that patients find unacceptably high. Patients are unlikely to use Palvella’s products unless coverage is provided and reimbursement is adequate to cover a significant portion of the cost of its products.
In addition, third-party payors, whether foreign or domestic, or governmental or commercial, are developing increasingly sophisticated methods of controlling healthcare costs. In addition, in the United States, although private third-party payors tend to follow Medicare, no uniform policy of coverage and reimbursement for drug products exists among third-party payors. Therefore, coverage and reimbursement for drug products can differ significantly from payor to payor. As a result, the coverage determination process is often a time- consuming and costly process that will require Palvella to provide scientific and clinical support for the use of its product candidates to each payor separately, with no assurance that coverage and adequate reimbursement will be obtained.
Palvella believes that future coverage and reimbursement will likely be subject to increased restrictions in both the United States and in international markets. Third-party coverage and reimbursement for any of its product candidates for which Palvella may receive regulatory approval may not be available or adequate in either the United States or international markets, which could materially adversely affect its business, financial condition, results of operations and prospects.
Palvella faces substantial competition, which may result in others discovering, developing or commercializing products before or more successfully than it does, thus rendering its products non-competitive, obsolete or reducing the size of its market.
The pharmaceutical and biotechnology industries are characterized by rapidly advancing technologies, intense competition and a strong emphasis on proprietary products. Palvella faces and will continue to face competition from many different sources, including major pharmaceutical, specialty pharmaceutical and biotechnology companies, academic institutions, government agencies and public and private research organizations that conduct research, seek patent protection and establish collaborative arrangements for research, development, manufacturing and commercialization.
In addition, some of the market demand for topical rapamycin may be satisfied by state-licensed compounding pharmacies operating under Section 503A of the FDCA. Although such pharmacies will be unable to compound any drug that is essentially a copy of QTORIN rapamycin, if approved, a compounded product would not be considered a copy of QTORIN rapamycin if there were a difference between the FDA-approved product and the compounded product that was made for an individual patient and which the prescribing practitioner determines produces a significant difference for that patient. Physicians may determine that such differences exist for some or all of their patients and may choose to prescribe compounded rapamycin because it would be a component of an FDA-approved drug product (specifically QTORIN). If the FDA-approved drug product is not commercially available and thus added to the FDA’s published drug shortage list, compounders also would be able to copy it without the necessity of noting a significant difference between the compounded formulation and the FDA-approved drug In the event compounders engage in the compounding of rapamycin products following FDA approval of QTORIN rapamycin, Palvella could be subject to significant competition from those compounded formulations.
The companies against which Palvella may compete may have significantly greater financial resources and expertise in research and development, manufacturing, preclinical testing, conducting clinical trials, obtaining regulatory approvals and marketing approved products than it does. These competitors also compete with Palvella in recruiting and retaining qualified scientific and management personnel and establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies complementary to, or necessary for, its programs. Palvella’s commercial opportunity could be reduced or eliminated if its competitors develop and commercialize products that are safer, more effective, have fewer or less severe side effects, are more convenient or are less expensive than any products that it may develop. Palvella’s competitors also may obtain FDA or other regulatory approval for their products more rapidly than it may obtain approval for itself, which could result in its competitors establishing a strong market position before it is able to enter the market. In addition, Palvella’s ability to compete may be affected in many cases by insurers or other third-party payors seeking to encourage the use of generic products. Because of Palvella’s primary focus on rare diseases, if its product candidates achieve marketing approval, it expects to seek premium pricing.
Mergers and acquisitions in the pharmaceutical and biotechnology industries may result in even more resources being concentrated among a smaller number of its competitors. Smaller and other early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. These third parties compete with Palvella in recruiting and retaining qualified scientific and management personnel, establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies complementary to, or necessary for, its programs.
If Palvella obtains approval to commercialize its product candidates outside of the United States, a variety of risks associated with international operations could materially adversely affect its business.
If any of Palvella’s product candidates are approved for commercialization, it may enter into agreements with third parties to market them on a worldwide basis or in more limited geographical regions. Palvella expects that it will be subject to additional risks related to conducting marketing and sales activities in international jurisdictions and entering into international business relationships, including:
|
● |
different regulatory requirements for approval of drugs in foreign countries; |
|
● |
the potential for so-called parallel importing, which is what happens when a local seller, faced with high or higher local prices, opts to import goods from a foreign market (with low or lower prices) rather than buying them locally; |
|
● |
challenges enforcing its contractual and intellectual property rights, especially in those foreign countries that do not respect and protect intellectual property rights to the same extent as the United States; |
|
● |
the need to seek additional patent approvals, licenses to patents held by third parties and/or face claims of infringing third-party patent rights; |
|
● |
unexpected changes in tariffs, trade barriers and regulatory requirements; |
|
● |
economic weakness, including inflation, or political instability in particular foreign economies and markets; |
|
● |
compliance with tax, employment, immigration and labor laws for employees living or traveling abroad; |
|
● |
foreign currency fluctuations, which could result in increased operating expenses and reduced revenues, and other obligations incident to doing business in another country; |
|
● |
differing reimbursement regimes and price controls in certain non-U.S. markets; |
|
● |
difficulties staffing and managing foreign operations; |
|
● |
workforce uncertainty in countries where labor unrest is more common than in the United States; |
|
● |
potential liability under the FCPA, the U.K. Bribery Act 2010 or other comparable foreign regulations; |
|
● |
production shortages resulting from any events affecting raw material supply or manufacturing capabilities abroad; and |
|
● |
business interruptions resulting from geopolitical actions, including war and terrorism, or natural disasters including pandemics, or other outbreaks of infectious disease, earthquakes, typhoons, floods and fires. |
These and other risks associated with international operations may materially adversely affect Palvella’s ability to attain or maintain profitable operations.
Risks Related to Palvella’s Business and Operations
Palvella will need to increase the size of its organization, and it may experience difficulties in executing its growth strategy and managing any growth, including with respect to any acquired businesses, therapeutic candidates or technologies.
As of October 28, 2024, Palvella had nine full-time employees. Palvella’s management and personnel, systems and facilities currently in place are not adequate to support its future growth. Palvella will need to expand its managerial, operational, finance and other resources in order to manage its operations and clinical trials, continue its development activities, commercialize its lead product candidates or any future product candidates and operate as a public company. In order to effectively execute its growth strategy, Palvella will need to identify, recruit, retain, incentivize and integrate additional employees in order to expand its ability to:
|
● |
manage its clinical trials effectively; |
|
● |
manage its internal development and operational efforts effectively while carrying out its contractual obligations to third parties; |
|
● |
continue to improve its operational, financial, management and regulatory compliance controls and reporting systems and procedures; |
|
● |
develop a marketing, sales and distribution capability; |
|
● |
manage its commercialization activities for its product candidates effectively and in a cost-effective manner; |
|
● |
establish and maintain relationships with development and commercialization partners; and |
|
● |
manage its third-party supply and manufacturing operations effectively and in a cost-effective manner, while increasing production capabilities for its current product candidates to commercial levels. |
Should Palvella in the future acquire any complementary business, therapeutic candidates or technologies, its ability to integrate and manage acquired businesses, therapeutic candidates or technologies effectively will depend upon a number of factors including the size of the acquired business, the complexity of any therapeutic candidate or technology and the resulting difficulty of integrating the acquired business’s operations, if any. Palvella’s relationship with current employees or employees of any acquired business may become impaired. Palvella may also be subject to unexpected claims and liabilities arising from such acquisitions. These claims and liabilities could be costly to defend, could be material to Palvella’s financial position and might exceed either the limitations of any applicable indemnification provisions or the financial resources of the indemnifying parties. There can also be no assurance that Palvella will be able to assess ongoing profitability and identify all actual or potential liabilities of a business, therapeutic candidate or technology prior to its acquisition. If Palvella acquires businesses, therapeutic candidates or technologies that result in assuming unforeseen liabilities in respect of which it has not obtained contractual protections or for which protection is not available, this could materially adversely affect its business, financial condition, results of operations and prospects.
Palvella’s future financial performance and its ability to commercialize its product candidates, if approved, will depend, in part, on its ability to effectively manage any future growth, and its management may also have to divert a disproportionate amount of its attention away from day-to-day activities in order to devote a substantial amount of time to managing these growth activities.
If Palvella is unable to successfully identify, recruit, retain, incentivize and integrate additional employees and otherwise expand its managerial, operational, finance and other resources, its business and operational performance will be materially and adversely affected.
If Palvella fails to attract and retain management and other key personnel, it may be unable to continue to successfully develop its current and any future product candidates, commercialize its product candidates or otherwise implement its business plan.
Palvella’s ability to compete in the highly competitive pharmaceuticals industry depends upon its ability to attract and retain highly qualified managerial, scientific, medical, sales and marketing and other personnel. Palvella is highly dependent on its management and scientific personnel, including Wesley H. Kaupinen, its Chief Executive Officer, Kathleen Goin, its Chief Operating Officer, and Jeffrey Martini Ph.D., its Chief Scientific Officer. The loss of the services of any of these individuals could impede, delay or prevent the successful development of its product candidates and otherwise negatively impact its ability to successfully implement its business plan. If Palvella loses the services of any of these individuals, it might not be able to find suitable replacements on a timely basis or at all, and its business could be harmed as a result. Palvella maintains “key person” insurance only for its Chief Executive Officer.
Palvella’s employment agreement with Mr. Kaupinen may be terminated immediately by Palvella for cause or by Mr. Kaupinen with good reason, or upon thirty days’ notice if terminated for any other reason. In order to retain valuable employees at its company, in addition to salary and cash incentives, Palvella provides stock options that vest over time. The value to employees and key consultants of stock options that vest over time will be significantly affected by movements in its stock price that are beyond its control, and may at any time be insufficient to counteract offers from other companies.
Palvella might not be able to attract or retain qualified management and other key personnel in the future due to the intense competition for qualified personnel among biotechnology, pharmaceutical and other businesses, particularly in the eastern Pennsylvania area where it is headquartered. Palvella could have difficulty attracting experienced personnel to its company and may be required to expend significant financial resources in its employee recruitment and retention efforts. Many of the other pharmaceutical companies with whom Palvella competes for qualified personnel have greater financial and other resources, different risk profiles and longer histories in the industry than Palvella does. They also may provide more diverse opportunities and better chances for career advancement. If Palvella is not able to attract and retain the necessary personnel to accomplish its business objectives, it may experience constraints that will harm its ability to implement its business strategy and achieve its business objectives.
In addition, Palvella has scientific and clinical advisors who assist it in formulating its development and clinical strategies. These advisors are not its employees and may have commitments to, or consulting or advisory contracts with, other entities that may limit their availability to Palvella. In addition, its advisors may have arrangements with other companies to assist those companies in developing products or technologies that may compete with Palvella.
Palvella faces potential product liability exposure, and if successful claims are brought against it, Palvella may incur substantial liability for its product candidates and may have to limit its commercialization if it obtains regulatory approval.
The use of Palvella’s product candidates in clinical trials, and the sale of any of its product candidates for which it obtains regulatory approval, exposes Palvella to the risk of product liability claims. Product liability claims might be brought against it by consumers, healthcare providers, pharmaceutical companies or others selling or otherwise coming into contact with its products. For example, Palvella may be sued if any product candidate it develops allegedly causes injury or is found to be otherwise unsuitable during clinical testing, manufacturing, marketing or sale. Any such product liability claims may include allegations of defects in manufacturing, defects in design, a failure to warn of dangers inherent in the product, negligence, strict liability or a breach of warranties. Claims could also be asserted under state consumer protection acts. If Palvella cannot successfully defend itself against these claims, it will incur substantial liabilities or be required to limit development or commercialization of its product candidates for which it obtains regulatory approval. Even successful defense would require significant financial and management resources. Regardless of merit or eventual outcome, product liability claims may result in:
|
● |
loss of revenue from decreased demand for its product candidates, if approved; |
|
● |
impairment of Palvella’s business reputation or financial stability; |
|
● |
costs of related litigation; |
|
● |
substantial monetary awards to patients or other claimants; |
|
● |
exhaustion of any available insurance and Palvella’s capital resources; |
|
● |
diversion of management attention; |
|
● |
withdrawal of clinical trial participants and potential termination of clinical trial sites or entire clinical programs; |
|
● |
the inability to commercialize Palvella’s product candidates, if approved; |
|
● |
significant negative media attention; |
|
● |
decrease in Palvella’s stock price; or |
|
● |
initiation of investigations, and enforcement actions by regulators; and product recalls, withdrawals, revocation of approvals, or labeling, marketing or promotional restrictions. |
Palvella currently holds $5.0 million in product liability insurance coverage in the aggregate, which may not be adequate to cover all liabilities that it may incur. Palvella may need to increase its insurance coverage as it initiates additional clinical trials. Palvella will need to further increase its insurance coverage if it commences commercialization of any of its product candidates for which it obtains marketing approval. Insurance coverage is increasingly expensive. Palvella may not be able to maintain insurance coverage at a reasonable cost or in an amount adequate to satisfy any liability that may arise. On occasion, large judgments have been awarded in class action lawsuits based on therapeutics that had unanticipated side effects. Its insurance policies also have various exclusions, and Palvella may be subject to a product liability claim for which Palvella has no coverage. A successful product liability claim or series of claims brought against Palvella could cause its stock price to fall and, if judgments exceed its insurance coverage, could decrease its cash and adversely affect its business and its prospects.
Unfavorable global economic or political conditions could adversely affect Palvella’s business, financial condition or results of operations.
Palvella’s results of operations could be adversely affected by general conditions in the global economy and in the global financial markets. A global financial crisis or a global or regional political disruption could cause extreme volatility in the capital and credit markets. A severe or prolonged economic downturn or political disruption could result in a variety of risks to its business, including weakened demand for Palvella’s lead product candidates or any future product candidates, if approved, and its ability to raise additional capital when needed on acceptable terms, if at all. A weak or declining economy or political disruption could also strain its manufacturers or suppliers, possibly resulting in supply disruption, or cause its customers to delay making payments for its services. Any of the foregoing could harm Palvella’s business and it cannot anticipate all of the ways in which the political or economic climate and financial market conditions could materially adversely affect its business, financial condition, results of operations and prospects.
Palvella’s business and operations would suffer in the event of system failures, and it faces risks related to its collection and use of data, which could result in investigations, inquiries, litigation, fines, legislative and regulatory action and negative press about its privacy and data protection practices.
Palvella’s information security systems and internal computer systems and those of its CROs and other contractors and consultants are vulnerable to damage from computer viruses, unauthorized access, natural disasters, terrorism, war, and telecommunication and electrical failures. If such an event were to occur and cause interruptions in Palvella’s operations, it could result in a material disruption of its product candidate development and, if such product candidates are approved, commercialization programs.
Additionally, Palvella’s business processes personal data, including some data related to health. When conducting clinical trials, Palvella faces risks associated with collecting trial participants’ data, especially health data, in a manner consistent with applicable laws and regulations. Palvella also faces risks inherent in handling large volumes of data and in protecting the security of such data. Palvella could be subject to attacks on its systems by outside parties or fraudulent or inappropriate behavior by its service providers or employees. Third parties may also gain access to the company’s systems using stolen or inferred credentials, computer malware, viruses, spamming, phishing attacks or other means, and may use such access to obtain personal data. Data breaches could subject Palvella to individual or consumer class action litigation and governmental investigations and proceedings by federal, state and local regulatory entities in the United States and by international regulatory entities, resulting in exposure to material civil and/or criminal liability. As Palvella’s operations and business grow, it may become subject to or affected by new or additional data protection laws and regulations and face increased scrutiny or attention from regulatory authorities, including various domestic and international privacy and security regulations.
The legislative and regulatory landscape for privacy and data protection continues to evolve. In the United States, certain states may adopt privacy and security laws and regulations that may be more stringent than applicable federal law. For example, California enacted the CCPA, which took effect on January 1, 2020. The CCPA creates individual privacy rights for California consumers and increases the privacy and security obligations of entities handling certain personal data. Palvella may also in the future be subject to data protection laws and regulations of other jurisdictions, such as the EU’s GDPR, which provides data subjects with certain rights and requires organizations to adopt technical and organizational safeguards to protect personal data. In the event that Palvella is subject to or affected by privacy and data protection laws, including the CCPA or GDPR and other domestic or international privacy and data protection laws, Palvella may expend significant resources to comply with such laws, and any liability from failure to comply with the requirements of these laws could adversely affect its financial condition.
If Palvella or its third-party contractors fail to comply with environmental, health and safety laws and regulations, it could become subject to fines or penalties or incur costs that could have a material adverse effect on the success of its business.
Palvella and its third-party contractors are subject to numerous environmental, health and safety laws and regulations, including those governing laboratory procedures and the handling, use, storage, treatment and disposal of hazardous materials and wastes. Palvella’s operations involve the use of hazardous and flammable materials, including chemicals and biological materials. Palvella’s operations also produce hazardous waste products. Palvella generally contracts with third parties for the disposal of these materials and wastes. In the event of contamination or injury resulting from its use of hazardous materials, Palvella could be held liable for any resulting damages, and any liability could exceed its resources. Palvella also could incur significant costs associated with civil or criminal fines and penalties. Furthermore, environmental laws and regulations are complex, change frequently and have tended to become more stringent. Palvella cannot predict the impact of such changes and cannot be certain of its future compliance. In addition, Palvella may incur substantial costs in order to comply with current or future environmental, health and safety laws and regulations. These current or future laws and regulations may impair its research, development or production efforts. Failure to comply with these laws and regulations also may result in substantial fines, penalties or other sanctions. Although Palvella maintains workers’ compensation insurance to cover it for costs and expenses it may incur due to injuries to its employees resulting from the use of hazardous materials or other work-related injuries, this insurance may not provide adequate coverage against potential liabilities.
Risks Related to Healthcare Laws and Other Legal Compliance Matters
Even if Palvella receives regulatory and marketing approval of its product candidates, it will be subject to extensive and ongoing regulatory obligations and continued regulatory review, which may result in significant additional expense, and it may be subject to penalties if it fails to comply with regulatory requirements or experience unanticipated problems with its product candidates.
Any regulatory approvals or other marketing authorizations Palvella obtains for its product candidates may be subject to limitations on the indicated uses for which the product may be marketed or the conditions of approval or marketing authorization or contain requirements for potentially costly post-market testing and surveillance to monitor the safety and efficacy of the product candidate. The FDA may also require a REMS as a condition of approval of Palvella’s drug product candidates, such as QTORIN rapamycin for the treatment of microcystic LM, which could include requirements for a medication guide, physician communication plans or additional elements to assure safe use, such as restricted distribution methods, patient registries and other risk minimization tools. In addition, if the FDA or an equivalent foreign regulatory authority authorizes its product candidates for marketing, the manufacturing processes, labeling, packaging, distribution, adverse event reporting, storage, advertising, promotion, import, export and recordkeeping for its product candidates will be subject to extensive and ongoing regulatory requirements. These requirements include submissions of safety and other post-marketing information and reports, registration, as well as continued compliance with cGMPs and good clinical practices, or “GCP”, requirements for any clinical trials that Palvella conducts post-approval. Later discovery of previously unknown problems with its product candidates, including adverse events of unanticipated severity or frequency, or with its CMOs or manufacturing processes, or failure to comply with regulatory requirements, may result in, among other things:
|
● |
restrictions on the marketing or manufacturing of its product candidates, withdrawal of the product from the market, or voluntary or mandatory product recalls; |
|
● |
fines, warning or untitled letters, Form 483s, or holds on clinical trials; |
|
● |
refusal by the FDA to accept new marketing applications or supplements, approve or otherwise authorize for marketing pending applications or supplements to applications filed by Palvella or suspension or revocation of approvals or other marketing authorizations; |
|
● |
product seizure or detention, or refusal to permit the import or export of its product candidates; and |
|
● |
injunctions or the imposition of civil or criminal penalties. |
The FDA’s and other regulatory authorities’ policies may change, and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of its product candidates. If Palvella is slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if Palvella is not able to maintain regulatory compliance, Palvella may lose any marketing approval that it may have obtained, and it may not achieve or sustain profitability, which could materially adversely affect its business, financial condition, results of operations and prospects.
Palvella’s product candidates, if authorized for marketing, may cause or contribute to adverse medical events that Palvella is required to report to the FDA, and if it fails to do so, it would be subject to sanctions that could harm its reputation, business, financial condition and results of operations. The discovery of serious safety issues with its product candidates, or a recall of its products either voluntarily or at the direction of the FDA or another governmental authority, if such products are marketed, could have a negative impact on Palvella.
With respect to any of its product candidates in clinical testing or approved by the FDA, Palvella will be subject to the FDA’s safety reporting requirements. The timing of its obligation to report is triggered by the date Palvella becomes aware of the adverse event as well as the nature of the event. Palvella may fail to report adverse events of which it becomes aware within the prescribed timeframe. Palvella may also fail to recognize that Palvella has become aware of a reportable adverse event, especially if it is not reported to Palvella as an adverse event or if it is an adverse event that is unexpected or removed in time from the use of the product. If Palvella fails to comply with its reporting obligations, the FDA could take action, including warning letters, untitled letters, administrative actions, criminal prosecution, imposition of civil monetary penalties, revocation of its approval or delay in approval of future products.
Palvella may choose to voluntarily recall a product if any material deficiency is found. A recall could occur as a result of an unacceptable risk to health, component failures, malfunctions, manufacturing defects, labeling or design deficiencies, packaging defects or other deficiencies or failures to comply with applicable regulations. Product defects or other errors may occur in the future. Recalls involving Palvella’s product candidates, if and when they are approved or otherwise authorized for marketing, could materially adversely affect its business, financial condition, results of operations and prospects.
Palvella will be subject to healthcare laws and regulations relating to its business, and could face substantial penalties if Palvella is determined not to have fully complied with such laws, which would have an adverse impact on its business.
Palvella’s business operations and current and future arrangements with investigators, healthcare professionals, consultants, third-party payors, customers and patients, will expose Palvella to broadly applicable fraud and abuse and other healthcare laws and regulations. These laws may constrain the business or financial arrangements and relationships through which Palvella conduct its operations, including how it researches, markets, sells and distributes any products for which it obtains marketing approval. Such laws include:
|
● |
the U.S. federal Anti-Kickback Statute, which prohibits, among other things, persons and entities from knowingly and willfully offering, paying, soliciting or receiving remuneration, directly or indirectly, overtly or covertly, in cash or in kind, to induce or reward, or in return for the purchase, leasing, ordering, recommending or arranging for the purchase, lease or order of any item or service reimbursable under Medicare, Medicaid or other federal healthcare programs. A person or entity does not need to have actual knowledge of the U.S. federal Anti-Kickback Statute or specific intent to violate it in order to have committed a violation. In addition, the government may assert that a claim including items or services resulting from a violation of the U.S. federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the civil False Claims Act; |
|
● |
U.S. federal civil and criminal false claims laws and civil monetary penalties laws, including the federal False Claims Act, which can be enforced through civil whistleblower or qui tam actions against individuals or entities, and the Federal Civil Monetary Penalties Law, which prohibit, among other things, impose criminal and civil penalties, against individuals or entities for knowingly presenting, or causing to be presented, a false claim for payment to, or approval by, the U.S. government or knowingly making, using, or causing to be made or used a false record or statement material to a false or fraudulent claim to the U.S. government. In addition, certain marketing practices, including off-label promotion, may also violate false claims laws. Moreover, the government may assert that a claim including items and services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the federal False Claims Act; |
|
● |
HIPAA and its implementing regulations, which impose criminal and civil liability for, among other things, knowingly and willfully executing, or attempting to execute, a scheme to defraud or to obtain, by means of false or fraudulent pretenses, representations or promises, any money or property owned by, or under the control or custody of, any healthcare benefit program, including private third-party payors and knowingly and willfully falsifying, concealing or covering up by trick, scheme or device, a material fact or making any materially false, fictitious or fraudulent statement in connection with the delivery of or payment for healthcare benefits, items or services. Similar to the U.S. federal Anti- Kickback Statute, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation; |
|
● |
HIPAA, as amended by the HITECH Act, and their respective implementing regulations, which also impose obligations, including mandatory contractual terms, with respect to safeguarding the privacy, security and transmission of individually identifiable health information without appropriate authorization by covered entities subject to the rule, such as health plans, healthcare clearinghouses and healthcare providers as well as their business associates and their subcontractors that perform certain services for or on their behalf involving the use or disclosure of individually identifiable health information; |
|
● |
the federal Physician Payments Sunshine Act, also known as Open Payments program, which requires certain manufacturers of drugs, devices, biologics and medical supplies for which payment is available under Medicare, Medicaid or the Children’s Health Insurance Program, with specific exceptions, to report annually to the Centers for Medicare & Medicaid Services, or “CMS”, information related to “payments or other transfers of value” made to physicians, which is defined to include doctors, dentists, optometrists, podiatrists and chiropractors, and teaching hospitals and applicable manufacturers and applicable group purchasing organizations to report annually to CMS ownership and investment interests held by physicians and their immediate family members. Applicable manufacturers are also required to report such information regarding payments and transfers of value provided, as well as ownership and investment interests held, during the previous year to physician assistants, nurse practitioners, clinical nurse specialists, certified nurse anesthetists and certified nurse-midwives; |
|
● |
state privacy laws and regulations, such as those of California, that impose restrictive requirements regulating the use and disclosure of health information and other personally identifiable information (for example, the CCPA gives California residents expanded rights to access and delete their personal information, opt out of certain personal information sharing and receive detailed information about how their personal information is used, and provides for civil penalties for violations, as well as a private right of action for data breaches that is expected to increase data breach litigation; resulting in increased compliance costs and potential liability); and |
|
● |
analogous state and non-U.S. laws and regulations, such as state anti-kickback and false claims laws, which may apply to its business practices, including, but not limited to, research, distribution, sales and marketing arrangements and claims involving healthcare items or services reimbursed by non-governmental third-party payors, including private insurers; state laws that require pharmaceutical and device companies to comply with the industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the U.S. government, or otherwise restrict payments that may be made to healthcare providers and other potential referral sources; state laws and regulations that require manufacturers to report information related to payments and other transfers of value to physicians and other healthcare providers or marketing expenditures and pricing information; certain state and local laws that require the registration of pharmaceutical sales representatives; and state and non-U.S. laws governing the privacy and security of health information in some circumstances, many of which differ from each other in significant ways and often are not preempted by HIPAA, thus complicating compliance efforts. |
Efforts to ensure that Palvella’s current and future business arrangements with third parties will comply with applicable healthcare laws and regulations will involve substantial costs. It is possible that governmental authorities may conclude that Palvella’s business practices, including its consulting arrangements with and/or ownership interests by physicians and other healthcare providers, do not comply with current or future statutes, regulations, agency guidance or case law involving applicable healthcare laws. If Palvella’s operations are found to be in violation of any of these or any other health regulatory laws that may apply to it, it may be subject to significant penalties, including the imposition of significant civil, criminal and administrative penalties, damages, monetary fines, disgorgement, individual imprisonment, possible exclusion from participation in Medicare, Medicaid and other U.S. healthcare programs, integrity oversight and reporting obligations, contractual damages, reputational harm, diminished profits and future earnings, and curtailment or restructuring of its operations, any of which could adversely affect its ability to operate its business and its results of operations. Defending against any such actions can be costly, time-consuming and may require significant financial and personnel resources. Therefore, even if Palvella is successful in defending against any such actions that may be brought against it, its business may be impaired. If any of the above occur, it could materially adversely affect Palvella’s business, financial condition, results of operations and prospects.
Disruptions at the FDA, the SEC and other government agencies or comparable regulatory authorities could hinder their ability to hire and retain key leadership and other personnel, or otherwise prevent new products and services from being developed or commercialized in a timely manner, or otherwise prevent those agencies from performing normal business functions on which Palvella’s business operations rely, which could negatively impact Palvella’s business.
The ability of the FDA or comparable foreign regulatory authorities to review and approve new products can be affected by a variety of factors, including government budget and funding levels, ability to hire and retain key personnel and accept the payment of user fees, and statutory, regulatory, and policy changes that may otherwise affect the FDA’s or comparable foreign regulatory authorities’ ability to perform routine functions. In addition, government funding of the SEC and other government agencies or comparable foreign regulatory authorities on which Palvella’s operations may rely, including those that fund research and development activities, is subject to the political process, which is inherently fluid and unpredictable. Disruptions at the FDA and other agencies may also slow the time necessary for new drugs to be reviewed and/or approved by necessary government agencies, which would harm its business. For example, over the last several years, the U.S. government has shut down several times and certain regulatory agencies, such as the FDA, have had to furlough critical FDA employees and stop critical activities. If a prolonged government shutdown occurs, or if geopolitical or global health concerns prevent the FDA or other regulatory authorities from conducting their regular inspections, reviews, or other regulatory activities, it could significantly impact the ability of the FDA to timely review and process Palvella’s regulatory submissions, which could materially adversely affect its business, financial condition, results of operations and prospects.
Healthcare reform measures may increase the difficulty and cost for Palvella to successfully commercialize its product and product candidates, if approved, and affect the prices it may obtain.
The United States and many foreign jurisdictions have enacted or proposed legislative and regulatory changes affecting the healthcare system that could restrict or regulate post-approval activities relating to Palvella’s product and product candidates, if approved, including implementing cost-containment programs to limit the growth of government-paid healthcare costs, including price controls, restrictions on reimbursement and requirements for substitution of generic products for branded prescription drugs. Palvella cannot be sure whether additional legislative changes will be enacted, or whether the FDA regulations, guidance or interpretations will be changed, or what the impact of such changes on the marketing authorizations of our product candidates, if any, may be. In addition, increased scrutiny by the U.S. Congress of the FDA’s approval process may significantly delay or prevent marketing authorization, as well as subject us to more stringent product labeling and post-marketing testing and other requirements.
In the United States, there have been and continue to be a number of legislative initiatives to contain healthcare costs, including costs of pharmaceuticals. There has been heightened governmental scrutiny over the manner in which manufacturers set prices for their products, which has resulted in several presidential executive orders, Congressional inquiries, and proposed and enacted federal and state legislation designed to, among other things, bring more transparency to product pricing, review the relationship between pricing and manufacturer patient programs, reduce the costs of drugs under Medicare and Medicaid, and reform government program reimbursement methodologies for drug products. For example, on August 2, 2011, the Budget Control Act of 2011 imposed, subject to certain temporary suspension periods, 2% reductions in Medicare payments to providers per fiscal year starting April 1, 2013 and, due to subsequent legislative amendments to the statute, will remain in effect through 2032, unless additional Congressional action is taken. In December 2020, CMS issued a final rule implementing significant manufacturer price reporting changes under the Medicaid Drug Rebate Program, including an alternative rebate calculation for line extensions that is tied to the price increases of the original drug, and Best Price reporting related to certain value-based purchasing arrangements. Under the American Rescue Plan Act of 2021, effective January 1, 2024, the statutory cap on Medicaid Drug Rebate Program rebates that manufacturers pay to state Medicaid programs is eliminated. Elimination of this cap may, in some cases, require pharmaceutical manufacturers to pay more in rebates than they receive on the sale of products.
Recently, several healthcare reform initiatives culminated in the enactment of the Inflation Reduction Act, or “IRA”, in August 2022, which, among other things, allows the Department of Health and Human Services, or “HHS”, to directly negotiate the selling price of a statutorily specified number of drugs and biologics each year that CMS reimburses under Medicare Part B and Part D. Only high-expenditure single-source drugs that have been approved for at least 7 years (11 years for single-source biologics) are eligible to be selected for negotiation by CMS, with the negotiated price taking effect two years after the selection year. Negotiations for Medicare Part D products begin in 2024 with the negotiated price taking effect in 2026, and negotiations for Medicare Part B products begin in 2026 with the negotiated price taking effect in 2028. In August 2023, HHS announced the ten Medicare Part D drugs and biologics that it selected for negotiations. HHS will announce the negotiated maximum fair prices by September 1, 2024. This price cap, which cannot exceed a statutory ceiling price, will come into effect on January 1, 2026, and will represent a significant discount from average prices to wholesalers and direct purchasers. The IRA also imposes rebates on Medicare Part D and Part B drugs whose prices have increased at a rate greater than the rate of inflation. In addition, the law eliminates the “donut hole” under Medicare Part D beginning in 2025 by significantly lowering the beneficiary maximum out-of-pocket cost and requiring manufacturers to subsidize, through a newly established manufacturer discount program, 10% of Part D enrollees’ prescription costs for brand drugs below the out-of-pocket maximum, and 20% once the out-of-pocket maximum has been reached. The IRA also extends enhanced subsidies for individuals purchasing health insurance coverage in ACA marketplaces through plan year 2025. The IRA permits the Secretary of HHS to implement many of these provisions through guidance, as opposed to regulation, for the initial years. Manufacturers that fail to comply with the IRA may be subject to various penalties, including significant civil monetary penalties. These provisions may be subject to legal challenges. For example, the provisions related to the negotiation of selling prices of high-expenditure single-source drugs and biologics have been challenged in multiple lawsuits brought by pharmaceutical manufacturers. The outcome of these lawsuits is uncertain, and some IRA drug discount provisions have not been challenged in litigation. Thus, while it is unclear how the IRA will be implemented, it will likely have a significant impact on the pharmaceutical industry and the pricing of Palvella’s product candidates, if approved.
Palvella expects that the IRA, as well as other healthcare reform measures that have and may be adopted in the future, may result in more rigorous coverage criteria and in additional downward pressure on the price that it receives for its product and product candidates, if approved, and could seriously harm its future revenues. Any reduction in reimbursement from Medicare, Medicaid, or other government programs may result in a similar reduction in payments from private payers. The implementation of cost containment measures or other healthcare reforms may prevent Palvella from being able to generate revenue, attain profitability or successfully commercialize its product and product candidates, if approved.
Any product candidates for which Palvella is able to obtain regulatory approval in the future may become subject to unfavorable pricing regulations, third-party payor coverage and reimbursement policies, or healthcare reform initiatives.
Palvella’s ability to commercialize any of its other product candidates, if approved, successfully will depend, in part, on the extent to which coverage and adequate reimbursement for these products and related treatments will be available from government payor programs at the federal and state level, including Medicare and Medicaid, private health insurers, managed care plans and other organizations. Government authorities and other third-party payors, such as private health insurers and health maintenance organizations, decide which medications they will pay for and establish reimbursement levels.
A primary trend in the U.S. healthcare industry and elsewhere is cost containment. Government authorities and other third-party payors have attempted to control costs by limiting coverage and the amount of reimbursement for particular medications or procedures. Increasingly, third-party payors are requiring that drug companies provide them with predetermined discounts from list prices and are challenging the prices charged for medical products. Coverage and reimbursement may not be available for any drug that Palvella or its collaborators commercialize and, even if these are available, the level of reimbursement for a product or procedure may not be satisfactory. Inadequate reimbursement levels may adversely affect the demand for, or the price of, any product candidate for which Palvella obtains marketing approval. Obtaining and maintaining adequate reimbursement for its product candidates, if approved, may be difficult. Palvella may be required to conduct expensive pharmacoeconomic studies to seek to justify coverage and reimbursement or the level of reimbursement relative to other therapies. If coverage and adequate reimbursement are not available or reimbursement is available only to limited levels, Palvella may not be able to successfully commercialize any of its other product candidates for which marketing approval is obtained.
As discussed above, the IRA contains substantial drug pricing reforms, including the establishment of a drug price negotiation program within the HHS t, the establishment of rebate payment requirements on manufacturers of certain drugs payable under Medicare Parts B and D to penalize price increases that outpace inflation, and the requirement for manufacturers to provide discounts on Part D drugs. Substantial penalties can be assessed for noncompliance with the drug pricing provisions. Although the IRA exempts orphan drugs that treat only one rare disease from the drug price negotiation provisions, Palvella does not know if additional drug pricing reforms could eliminate this exemption and therefore affect the prices it can charge and reimbursement it receives for its product candidates, if approved, thereby reducing its profitability. The full extent of the IRA on Palvella’s business and the pharmaceutical industry in general is not yet known.
Future efforts to reform healthcare or reduce costs under government insurance programs may result in lower reimbursement for its products and product candidates or exclusion of its products and product candidates from coverage. The cost containment measures that healthcare payors and providers are instituting and any healthcare reform could significantly reduce its revenue from the sale of any approved product candidates. Palvella cannot provide any assurances that it will be able to obtain and maintain third-party coverage or adequate reimbursement for its product candidates in whole or in part.
If any of Palvella’s product candidates are approved for marketing and Palvella is found to have improperly promoted off-label uses, or if physicians misuse its products or use its products off-label, it may become subject to prohibitions on the sale or marketing of its products, product liability claims and significant fines, penalties and sanctions, and its brand and reputation could be harmed.
The FDA and other equivalent foreign regulatory authorities strictly regulate the marketing of and promotional claims that are made about drug products. In particular, a product may not be promoted for uses or indications that are not approved by the FDA or such other equivalent foreign regulatory authorities as reflected in the product’s approved labeling. In addition, although Palvella believes its product candidates may exhibit a lower risk of side effects or more favorable tolerability profile or better symptomatic improvement than other products for the indications Palvella is studying, without head-to-head data, Palvella will be unable to make comparative claims for its product candidates, if approved. If Palvella receives regulatory approval for any of its product candidates and are found to have promoted any of its products for off-label uses, it may become subject to significant liability, which would materially harm its business. Both federal and state governments have levied large civil and criminal fines against companies for alleged improper promotion and have enjoined several companies from engaging in off-label promotion. If Palvella becomes the target of such an investigation or prosecution based on its marketing and promotional practices, it could face similar sanctions, which would materially harm its business. In addition, management’s attention could be diverted from Palvella’s business operations, significant legal expenses could be incurred, and its brand and reputation could be damaged. The FDA has also previously requested that companies enter into consent decrees or permanent injunctions under which specified promotional conduct is changed or curtailed. If Palvella is deemed by the FDA to have engaged in the promotion of its products for off-label use, it could be subject to FDA regulatory or enforcement actions, including the issuance of an untitled letter, a warning letter, injunction, seizure, civil fine or criminal penalties. It is also possible that other federal, state or foreign enforcement authorities might take action if they determine Palvella’s business activities constitute promotion of an off-label use, which could result in significant penalties, including criminal, civil or administrative penalties, damages, fines, disgorgement, exclusion from participation in government healthcare programs and the curtailment or restructuring of its operations.
Palvella cannot, however, prevent a physician from using its product candidates, once approved, as he or she may deem appropriate in his or her medical judgment even if such use falls outside of the scope of the approved indications. Physicians may also misuse Palvella’s product candidates or use improper techniques, which may lead to adverse results, side effects or injury and, potentially, subsequent product liability claims. Furthermore, the use of Palvella’s product candidates for indications other than those approved by the FDA and/or other regulatory authorities may not effectively treat such diseases which could harm its brand and reputation among both physicians and patients.
Palvella’s business activities may be subject to the FCPA and similar anti-bribery and anti-corruption laws of other countries in which Palvella operates, as well as U.S. and certain foreign export controls, trade sanctions, and import laws and regulations. Compliance with these legal requirements could limit Palvella’s ability to compete outside of the U.S. market and subject it to liability if it violates them.
If Palvella expands its operations outside of the United States, it must dedicate additional resources to comply with numerous laws and regulations in each jurisdiction in which it plans to operate. Palvella’s business activities may be subject to the FCPA and similar anti-bribery or anti-corruption laws, regulations or rules of other countries in which it operates. The FCPA generally prohibits companies and their employees and third-party intermediaries from offering, promising, giving or authorizing the provision of anything of value, either directly or indirectly, to a non-U.S. government official in order to influence official action or otherwise obtain or retain business. The FCPA also requires public companies to make and keep books and records that accurately and fairly reflect the transactions of the corporation and to devise and maintain an adequate system of internal accounting controls. Palvella’s business is heavily regulated and therefore involves significant interaction with public officials, including officials of non-U.S. governments. Additionally, in many other countries, hospitals owned and operated by the government, and doctors and other hospital employees would be considered foreign officials under the FCPA. Recently the SEC and Department of Justice have increased their FCPA enforcement activities with respect to biotechnology and pharmaceutical companies. There is no certainty that all of Palvella’s employees, agents or contractors, or those of its affiliates, will comply with all applicable laws and regulations, particularly given the high level of complexity of these laws. Violations of these laws and regulations could result in fines, criminal sanctions against Palvella, its officers or its employees, disgorgement, and other sanctions and remedial measures, and prohibitions on the conduct of its business. Any such violations could include prohibitions on Palvella’s ability to offer its products in one or more countries and could materially damage its reputation, its brand, its international activities, its ability to attract and retain employees and its business, financial condition, results of operations and prospects.
In addition, Palvella’s products and activities may be subject to U.S. and foreign export controls, trade sanctions and import laws and regulations. Governmental regulation of the import or export of Palvella’s products, or its failure to obtain any required import or export authorization for its products, when applicable, could harm its international sales and adversely affect its revenue. Compliance with applicable regulatory requirements regarding the export of Palvella’s products may create delays in the introduction of its products in international markets or, in some cases, prevent the export of its products to some countries altogether. Furthermore, U.S. export control laws and economic sanctions prohibit the shipment of certain products and services to countries, governments, and persons targeted by U.S. sanctions. If Palvella fails to comply with export and import regulations and such economic sanctions, penalties could be imposed, including fines and/or denial of certain export privileges. Moreover, any new export or import restrictions, new legislation or shifting approaches in the enforcement or scope of existing regulations, or in the countries, persons, or products targeted by such regulations, could result in decreased use of its products by, or in its decreased ability to export its products to existing or potential customers with international operations. Any decreased use of Palvella’s products or limitation on its ability to export or sell access to its products would likely adversely affect its business, financial condition, results of operations and prospects.
Risks Related to Palvella’s Reliance on Third Parties
Palvella currently relies on CMOs to manufacture preclinical and clinical supplies of its product candidates and will rely on CMOs for the commercial supplies of any approved product candidate. The loss of these manufacturers, or their failure to provide Palvella with sufficient quantities at acceptable quality levels or prices, or at all, would materially and adversely affect its business.
Palvella does not currently have nor does it plan to build or acquire the infrastructure or capability internally to manufacture supplies of its product candidates or the materials necessary to produce its product candidates for use in the conduct of its preclinical studies or clinical trials, and it lacks the internal resources and the capability to manufacture any of its product candidates on a preclinical, clinical or commercial scale. Instead, Palvella currently relies on CMOs to manufacture preclinical and clinical supplies of its product candidates and the commercial supplies of any approved product candidate. Palvella has agreements with Altasciences and PMRS, both, cGMP facilities to manufacture clinical supply of its product candidate for clinical trials and for the manufacture of commercial supply of its QTORIN rapamycin, if approved. Additionally, Palvella has agreements with Medpharm UK for the manufacture of its clinical supply of its product candidate for clinical trials. Palvella plans to enter into an agreement with another CMO for its supply of QTORIN rapamycin and qualify them as its second source of clinical and commercial supply. Please see “Business—Manufacturing” for a discussion of Palvella’s current manufacturing and supply agreements.
Reliance on CMOs entails risks to which Palvella may not be subject if it manufactured product candidates itself, including:
|
● |
the possible failure of the third party to manufacture its product candidates according to its schedule, or at all, including if its third-party contractors give greater priority to the supply of other products over its product candidates or otherwise do not satisfactorily perform according to the terms of the agreements between Palvella and them; |
|
● |
reliance on the third party for regulatory compliance and quality control and assurance and failure of the third party to comply with regulatory requirements; |
|
● |
the possibility of breach of the manufacturing agreement by the third party because of factors beyond its control (including a failure to manufacture its product candidates in accordance with its product specifications); |
|
● |
a disruption to one or more of its CMOs’ relevant operations; the possible mislabeling of clinical supplies, potentially resulting in the wrong dose amounts being supplied or active drug or vehicle not being properly identified; |
|
● |
the possibility of clinical supplies not being delivered to clinical sites on time, leading to clinical trial interruptions, or of drug supplies not being distributed to commercial vendors in a timely manner, resulting in lost sales; |
|
● |
the risk that its CMOs face financial difficulties or declare bankruptcy; and |
|
● |
the possibility of its failure to enter into agreements for manufacturing services, on commercially reasonable terms or at all, or the possibility of termination or nonrenewal of the agreement by the third party at a time that is costly or damaging to Palvella. |
Moreover, there are a limited number of manufacturers capable of producing Palvella’s product candidates, which exposes it to the risk of disruption in the supply of product candidates for its preclinical studies and clinical trials, and if approved, ultimately for commercial sale. In the case of QTORIN rapamycin which Palvella is currently developing for both the treatment of microcystic LM and the treatment of cutaneous venous malformations, there is a limited number of manufacturers that will work with an active pharmaceutical ingredient, such as rapamycin, that has immunosuppressant properties. If its third-party manufacturing agreements were to be terminated for any reason, Palvella may be unable to procure alternative manufacturers for clinical or commercial manufacture of QTORIN rapamycin, as applicable, on a timely basis or at all.
Additionally, while Palvella has entered into agreements with Altasciences and PMRS for the commercial manufacture of its product candidates, both organizations must complete a scale-up process that includes the completion of various technical and regulatory steps before it will be able to produce commercial supply of its QTORIN rapamycin. If both CMOs fail for any reason to carry out its contractual duties or otherwise fails to meet its manufacturing requirements prior to its completion of the process of qualifying its second source manufacturer, its business, financial condition, results of operations and prospects could be materially and adversely affected.
Palvella may be unable to enter into additional agreements with third-party manufacturers or suppliers or do so on favorable terms. Palvella’s anticipated reliance on a limited number of third party-manufacturers or suppliers exposes it to the following risks:
|
● |
reliance on the third party for regulatory, compliance and quality assurance; |
|
● |
reliance on the third party for product development, analytical testing and data generation to support regulatory applications; |
|
● |
operations of its third-party manufacturers or suppliers could be disrupted by conditions unrelated to our business or operations, including the bankruptcy of the manufacturer or supplier, the issuance of an FDA Form 483 notice or warning letter or other enforcement action by the FDA or other regulatory authority; |
|
● |
the possible breach of the manufacturing agreement by the third party; |
|
● |
the possible misappropriation of proprietary information, including trade secrets and know-how; |
|
● |
the possible termination or nonrenewal of the agreement by the third party at a time that is costly or inconvenient for Palvella; and |
|
● |
carrier disruptions or increased costs that are beyond Palvella’s control. |
Failure to deliver Palvella’s drugs under specified storage conditions and in a timely manner. If any of these risks materialize and impact Palvella’s CMOs’ ability to produce its product candidates, it will have no other means of producing its product candidates until the adverse impact is mitigated or Palvella or they procure alternative manufacturing facilities or sources of supply. Though Palvella carefully manage its relationships with its CMOs, there can be no assurance that Palvella will not encounter challenges or delays in the future. The loss of any of Palvella’s CMOs, or their failure to provide it with sufficient quantities at acceptable quality levels or prices, or at all, would materially and adversely affect its business.
If the manufacturers upon whom Palvella relies fail to produce its product candidates or components pursuant to the terms of contractual arrangements with Palvella or fail to comply with stringent regulations applicable to pharmaceutical manufacturers, it may face delays in the development and commercialization of, or be unable to meet demand for, its product candidates, if approved, and may lose potential revenues.
Palvella has agreements governing the activities of the CMOs which manufacturers its preclinical, clinical and commercial supply of its product candidates, and Palvella expects to enter into agreements with additional CMOs in the future, but Palvella has or will have limited influence and control over their actual performance and activities. If Palvella’s CMOs do not successfully carry out their contractual duties, meet expected deadlines or manufacture its product candidates in accordance with strictly enforced federal, state, and foreign regulations, or if there are disagreements between Palvella and such parties, clinical development or marketing approval of its product candidates could be delayed.
All manufacturers of Palvella’s product candidates and therapeutic substances must comply with cGMP requirements enforced by the FDA that are applicable to both finished products and their active components used both for clinical and commercial supply. The cGMP requirements include quality control, quality assurance, and the maintenance of records and documentation. The FDA enforces these requirements through its facilities inspection program. If the FDA determines that Palvella’s manufacturers are not in compliance with FDA laws and regulations, including those governing cGMPs, the FDA may not approve a marketing application until the deficiencies are corrected or until Palvella replaces the manufacturer in its application with a manufacturer that is in compliance. Palvella’s manufacturers will also be subject to continuing FDA and other regulatory authority inspections should Palvella receive marketing approval for any of its product candidates. Any such deviations from the regulatory requirements of the FDA or other regulatory authorities may require remedial measures that may be costly and/or time-consuming for Palvella or a third party to implement and that may include the temporary or permanent suspension of a clinical trial or commercial sales or the temporary or permanent closure of a facility.
While Palvella is ultimately responsible for the manufacture of its product candidates, other than through its contractual arrangements, it expects to have limited ability to control its manufacturers’ compliance with these regulations and standards. A failure to comply with the applicable regulatory requirements may result in regulatory enforcement actions against its manufacturers or Palvella, including fines and civil and criminal penalties. Depending on the severity of any potential regulatory action, Palvella’s clinical or commercial supply could be interrupted or limited. Any problems or delays Palvella experiences in preparing for commercial-scale manufacturing of a product candidate or component, including manufacturing validation, may result in a delay in FDA approval or commercial launch of the product candidate or may impair Palvella’s ability to manufacture commercial quantities or such quantities at an acceptable cost, which could result in the delay, prevention, or impairment of commercialization of its product candidates and could adversely affect its business.
Palvella acquires the pumps it intends to use to dispense its QTORIN rapamycin, if approved, from a sole source supplier. If Palvella experiences an interruption in supply from this supplier, its business may be harmed.
Palvella has an agreement with Nemera Le Tréport SAS, or “Nemera”, a sole source supplier, for the pumps it intends to use to dispense QTORIN rapamycin, if approved. If there is an interruption in the supply of these pumps from Nemera due to pricing, timing, availability or other issues, if Nemera does not successfully carry out its contractual duties, meet expected deadlines or supply these pumps in accordance with the terms of its agreement and with applicable federal, state, and foreign regulations, or if there are disagreements between Palvella and Nemera, clinical development, marketing approval or commercial manufacturing of its product candidates, if approved, could be delayed.
If Palvella’s agreement with Nemera is terminated or if Nemera otherwise ceases to supply the pumps Palvella intends to use to dispense QTORIN rapamycin, if approved, there is no guarantee that Palvella will find an alternative supplier for the necessary packaging materials on terms acceptable to it, or at all. As a result, Palvella would have to redesign its commercial packaging which would be subject to FDA review. This may cause delay in the commercialization of its product candidates and cause Palvella to incur additional expense. The qualification process for a new vendor could take months or even years, particularly if Palvella is unable to locate an alternative supplier that has sufficient regulatory qualifications, and any such delay in qualification could materially adversely affect Palvella’s business, financial condition, results of operations and prospects.
Palvella relies on third parties to conduct aspects of its nonclinical studies and its clinical trials. If these third parties do not successfully carry out their contractual duties or meet expected deadlines, Palvella may be unable to obtain regulatory approval for or commercialize QTORIN rapamycin for the treatment of microcystic LM or any other current or future product candidates.
Palvella does not have the ability to independently conduct nonclinical studies and clinical trials. Although its employees manage the overall conduct of its preclinical studies and clinical trials and Palvella is responsible for ensuring that each of its preclinical studies and clinical trials is conducted in accordance with applicable regulations and the investigational plan and protocol, Palvella relies on third parties, such as CROs and academic institutions, to conduct aspects of its preclinical studies and clinical trials of QTORIN rapamycin for the treatment of microcystic LM. The third parties with whom Palvella contracts for execution of its preclinical studies and clinical trials play a significant role in the conduct of these studies and trials and the subsequent collection and analysis of data. These third parties are not Palvella employees, and Palvella expect CROs, independent clinical investigators and consultants to play a significant role in the conduct of its trials and the subsequent collection and analysis of data. Nevertheless, Palvella is responsible for ensuring that each clinical trial is conducted in accordance with the applicable protocol and legal, regulatory and scientific standards, and its reliance on the investigators, CROs and other third parties does not relieve it of its regulatory responsibilities. Palvella and its CROs are required to comply with GCP requirements, which are regulations and guidelines enforced by the FDA for product candidates in clinical development. Regulatory authorities enforce these GCP requirements through periodic inspections of trial sponsors, clinical trial investigators and clinical trial sites. If Palvella or any of its CROs or clinical trial sites fail to comply with applicable GCP requirements, the data generated in its clinical trials may be deemed unreliable, and the FDA may require Palvella to perform additional clinical trials before approving it marketing applications. Palvella cannot assure you that, upon inspection, the FDA will determine that its clinical trials comply with GCPs. Palvella’s failure or the failure of third parties on whom it relies to comply with these regulations may require Palvella to stop and/or repeat clinical trials, which would delay the marketing authorization process.
In addition, except for contractual duties and obligations, Palvella has limited ability to control the amount or timing of resources that its third party CROs, investigators, and institutions devote to its programs. If any of these third parties fail to meet expected deadlines, adhere to Palvella’s clinical protocols or meet regulatory requirements, otherwise perform in a substandard manner, or terminate their engagements with Palvella, the timelines for its development programs may be extended or delayed or its development activities may be suspended or terminated. If a clinical trial site terminates for any reason, Palvella may experience the loss of follow-up information on subjects enrolled in such clinical trial unless it is able to transfer those subjects to another qualified clinical trial site, which may be difficult or impossible.
Moreover, principal investigators for Palvella’s clinical trials may serve as scientific advisors or consultants to it from time to time and receive compensation in connection with such services. Under certain circumstances, Palvella may be required to report some of these relationships to the FDA or comparable foreign regulatory authorities. The FDA or comparable foreign regulatory authority may conclude that a financial relationship between Palvella and a principal investigator has created a conflict of interest or otherwise affected interpretation of the study. The FDA or comparable foreign regulatory authority may therefore question the integrity of the data generated at the applicable clinical trial site and the utility of the clinical trial itself may be jeopardized. This could result in a delay in approval, or rejection, of Palvella’s marketing applications by the FDA or comparable foreign regulatory authority, as the case may be, and may ultimately lead to the denial of regulatory approval of its product candidates.
Palvella’s third parties may also have relationships with other commercial entities, some of which may compete with Palvella. In some cases, these third parties could terminate their agreements with Palvella without cause.
The execution of non-clinical studies and clinical trials, and the subsequent compilation and analysis of the data produced, requires coordination among various parties. In order for these functions to be carried out effectively and efficiently, it is imperative that these parties communicate and coordinate with one another. If the third parties conducting aspects of Palvella’s clinical trials fail to communicate and coordinate with one another, do not perform their contractual duties or obligations, experience work stoppages, do not meet expected deadlines, terminate their agreements with Palvella or need to be replaced, or if the quality or accuracy of the data they obtain is compromised due to the failure to adhere to Palvella’s clinical trial protocols GCPs, or for any other reason, Palvella may need to enter into new arrangements with alternative third parties, which could be difficult, costly or impossible, and its clinical trials may be extended, delayed or terminated or may need to be repeated, which would have a material adverse effect on its business.
Palvella may rely on third parties to perform many essential services for any products that it commercializes. If these third parties fail to perform as expected or to comply with legal and regulatory requirements, Palvella’s ability to commercialize its product candidates will be significantly impacted and it may be subject to regulatory sanctions.
Palvella may retain third-party service providers to perform a variety of functions related to the sale and distribution of its product candidates, if approved, key aspects of which will be out of its direct control. These service providers may provide key services related to warehousing and inventory control, distribution, customer service, accounts receivable management, and cash collection. If these third-party service providers fail to comply with applicable laws and regulations, fail to meet expected deadlines, or otherwise do not carry out their contractual duties to Palvella, or encounter physical or natural damage at their facilities, Palvella’s ability to deliver adequate product to meet commercial demand would be significantly impaired and it may be subject to regulatory enforcement action.
In addition, Palvella may engage third parties to perform various other services for it relating to pharmacovigilance and adverse event reporting, safety database management, fulfillment of requests for medical information regarding its product candidates and related services. If the quality or accuracy of the data maintained by these service providers is insufficient, or these third parties otherwise fail to comply with regulatory requirements, Palvella could be subject to regulatory sanctions.
Palvella may also contract with a third party to calculate and report pricing information mandated by various government programs. If a third party fails to timely report or adjust prices as required, or commits errors in calculating government pricing information from transactional data in Palvella’s financial records, it could impact its discount and rebate liability, and potentially subject Palvella to regulatory sanctions or False Claims Act lawsuits.
Palvella’s future commercial collaborators, as well as its independent contractors, including principal investigators, consultants, suppliers, service providers and other vendors, may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements, which could have an adverse effect on its results of operations.
Palvella is exposed to the risk that its employees and independent contractors, including principal investigators, consultants, suppliers, service providers and other vendors, may engage in misconduct or other illegal activity. Misconduct by these parties could include intentional, reckless and/or negligent conduct or other unauthorized activities that violate the laws and regulations of the FDA and other equivalent foreign regulatory authorities, including those laws that require the reporting of true, complete and accurate information to such foreign regulatory authorities; manufacturing standards; U.S. federal and state healthcare fraud and abuse, data privacy laws and other similar non-U.S. laws; or laws that require the true, complete and accurate reporting of financial information or data. Activities subject to these laws also involve the improper use or misrepresentation of information obtained in the course of clinical trials, the creation of fraudulent data in Palvella’s preclinical studies or clinical trials, or illegal misappropriation of product, which could result in regulatory sanctions and cause serious harm to its reputation. It is not always possible to identify and deter misconduct by third parties, and the precautions Palvella takes to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting it from governmental investigations or other actions or lawsuits stemming from a failure to be in compliance with such laws or regulations. In addition, Palvella is subject to the risk that a person or government could allege such fraud or other misconduct, even if none occurred. If any such actions are instituted against Palvella, and it is not successful in defending itself or asserting its rights, those actions could have a significant impact on its business and financial results, including, without limitation, the imposition of significant civil, criminal and administrative penalties, damages, monetary fines, disgorgements, integrity oversight and reporting obligations, possible exclusion from participation in Medicare, Medicaid and other U.S. healthcare programs, imprisonment, other sanctions, contractual damages, reputational harm, diminished profits and future earnings and curtailment of its operations, any of which could materially adversely affect its business, financial condition, results of operations and prospects.
Palvella intends to explore strategic collaborations with third parties for the development or commercialization of its product candidates, which collaborations may never materialize or may require that it relinquish rights to and control over the development and commercialization of its product candidates.
An element of Palvella’s business strategy includes acquiring or in-licensing technologies or product candidates for the treatment of rare genetic skin diseases. As a result, Palvella intends to periodically explore a variety of possible strategic collaborations or licenses in an effort to gain access to additional product candidates, technologies, resources or markets. While Palvella does not have a strategic collaboration in place with respect to QTORIN rapamycin and it intends to independently commercialize this product candidate in the United States, it may selectively seek collaborators to commercialize its products outside of the U.S. market.
At this time, Palvella cannot predict what form such strategic collaborations or licenses might take in the future. Palvella is likely to face significant competition in seeking appropriate strategic collaborators, and strategic collaborations and licenses can be complicated and time-consuming to negotiate and document. Palvella may not be able to negotiate strategic collaborations on acceptable terms, or at all. Palvella is unable to predict when, if ever, it will enter into any additional strategic collaborations or licenses because of the numerous risks and uncertainties associated with establishing them. Any delays in entering into new strategic collaborations or licenses related to Palvella’s product candidates could delay the development and commercialization of its product candidates in certain geographies or for certain indications, which could materially adversely affect its business, financial condition, results of operations and prospects.
Palvella’s current collaborators and future collaborations and licenses could subject it to a number of risks, including:
|
● |
it may be required to undertake the expenditure of substantial operational, financial and management resources; |
|
● |
it may be required to issue equity securities that would dilute its stockholders’ percentage ownership of its company; |
|
● |
it may be required to assume substantial actual or contingent liabilities; |
|
● |
it may not be able to control the amount and timing of resources that its strategic collaborators devote to the development or commercialization of its product candidates; |
|
● |
it may not have the right to control the preparation, filing, prosecution and maintenance of patents and patent applications covering the technology that it licenses, and it cannot always be certain that these patents and patent applications will be prepared, filed, prosecuted and maintained in a manner consistent with the best interests of its business; |
|
● |
collaborators may select indications or design clinical trials in a way that may be less successful than if it was doing so; |
|
● |
collaborators may delay clinical trials, provide insufficient funding, terminate a clinical trial or abandon a product candidate, repeat or conduct new clinical trials or require a new version of a product candidate for clinical testing; |
|
● |
collaborators may not commit adequate resources to the marketing and distribution of its product candidates, limiting its potential revenue from these products; |
|
● |
disputes may arise between Palvella and its collaborators that result in the delay or termination of the research, development or commercialization of its product candidates or that result in costly litigation or arbitration that diverts management’s attention and consumes resources; |
|
● |
collaborators may experience financial difficulties; |
|
● |
collaborators could decide to move forward with a competing product candidate developed either independently or in collaboration with others, including its competitors; and |
|
● |
collaborators could terminate the arrangement or allow it to expire, which would delay the development and may increase the cost of developing its product candidates. |
Furthermore, license agreements Palvella enters into in the future may not provide exclusive rights to use intellectual property and technology in all relevant fields of use and in all territories in which it may wish to develop or commercialize its technology and products. As a result, Palvella may not be able to prevent competitors from developing and commercializing competitive products in territories included in all of its licenses.
Additionally, if any future collaborator of Palvella is involved in a business combination, the collaborator might deemphasize or terminate development or commercialization of any product candidate licensed to it by Palvella. If one of Palvella’s collaborators terminates its agreement with it, it may find it more difficult to attract new collaborators and its reputation in the business and financial communities could be adversely affected.
Risks Related to Intellectual Property
Palvella may not be able to obtain, maintain or enforce patent rights that cover its product candidates and technologies that are of sufficient breadth to prevent third parties from competing against it.
Palvella relies upon a combination of patents, trade secret protection and CDAs to protect the intellectual property related to its product candidates, proprietary technologies and product candidate development programs. Palvella’s success depends in large part on its ability to secure and maintain patent protection in the United States and other countries with respect to its product candidates and proprietary technologies and any future product candidates. Palvella seeks to protect its proprietary position by filing or collaborating with its licensors to file patent applications in the United States and abroad related to its proprietary technologies, development programs and product candidates. The patent prosecution process is expensive and time-consuming, and Palvella may not be able to file and prosecute all necessary or desirable patent applications at a reasonable cost or in a timely manner.
It is also possible that Palvella will fail to identify patentable aspects of its research and development output before it is too late to obtain patent protection. The patent applications that Palvella owns or in-licenses may fail to result in issued patents with claims that cover its product candidates, and proprietary technology in the United States or in other foreign countries, in whole or in part. Alternately, Palvella’s existing patents and any future patents it obtains may not be sufficiently broad to prevent others from using its technology or from developing competing products and technologies. There is no assurance that all potentially relevant prior art relating to Palvella’s patents and patent applications has been found, which can prevent a patent from issuing from a pending patent application or later invalidate or narrow the scope of an issued patent. Even if patents do successfully issue, and even if such patents cover Palvella’s existing product candidates or any future product candidate, third parties may challenge their validity, enforceability or scope thereof, which may result in such patents being narrowed, invalidated, or held unenforceable. Any successful challenge to these patents or any other patents owned by or licensed to Palvella could deprive it of rights necessary for the successful commercialization of any product candidates that it may develop. Further, if Palvella encounters delays in regulatory approvals, the period of time during which it could market a product candidate under patent protection could be reduced.
If the patent applications Palvella holds or has in-licensed with respect to its development programs and product candidates fail to issue, if their validity, breadth or strength of protection is threatened, or if they fail to provide meaningful exclusivity for its existing product candidates or any future product candidate, it could dissuade companies from collaborating with Palvella to develop product candidates, encourage competitors to develop competing products or technologies and threaten its ability to commercialize future product candidates. Any such outcome could have a materially adverse effect on Palvella’s business.
The patent position of biotechnology and pharmaceutical companies is highly uncertain, involves complex legal and factual questions, and is characterized by the existence of large numbers of patents and frequent litigation based on allegations of patent or other intellectual property infringement or violation. In addition, the laws of jurisdictions outside the United States may not protect Palvella’s rights to the same extent as the laws of the United States. For example, European patent law restricts the patentability of methods of treatment of the human body more than United States law does. Changes in either the patent laws or interpretation of the patent laws in the United States and other countries may diminish the value of Palvella’s patents or narrow the scope of its patent protection. Since patent applications in the United States and other jurisdictions are confidential for a period of time after filing, Palvella cannot be certain that it was the first to file for patents covering its inventions. As a result, the issuance, scope, validity, enforceability and commercial value of its patent rights are highly uncertain. Palvella’s pending and future patent applications may not result in the issuance of patents, or may result in the issuance of patents which fail to protect its technology or products, in whole or in part, or which fail to effectively prevent others from commercializing competitive technologies and products.
The issuance of a patent is not conclusive as to Palvella’s inventorship, scope, validity or enforceability, and its owned and licensed patents may be challenged in the courts or patent offices in the United States and abroad. Such challenges may result in loss of exclusivity, patent term or freedom to operate or in patent claims being narrowed, invalidated or held unenforceable, in whole or in part, which could limit Palvella’s ability to stop others from using or commercializing similar or identical technology and products, or limit the duration of the patent protection of its technology and products. Thus, even if Palvella’s patent applications issue as patents, they may not issue in a form that will provide it with meaningful protection, prevent competitors from competing with it or otherwise provide it with any competitive advantage.
The patent rights Palvella owns covering QTORIN rapamycin are directed to specific methods of use and specific formulations of rapamycin. As a result, Palvella’s ability to prevent others from marketing products related to QTORIN rapamycin may be limited by the lack of patent protection for the active ingredient itself and other rapamycin formulations may be developed by competitors. No patent protection is available for rapamycin itself, the active ingredient in QTORIN rapamycin. As a result, competitors who develop and receive required regulatory approval for competing products using the same active ingredient as QTORIN rapamycin may market their competing products so long as they do not infringe any of the method or formulation patents owned by Palvella.
Patent terms may be inadequate to protect Palvella’s competitive position and if it does not obtain additional patent protection by issuing additional patents with longer patent terms for its product candidates, its business may be materially harmed.
Palvella’s commercial success will largely depend on its ability to obtain and maintain patent and other intellectual property in the United States and other countries with respect to its proprietary technology, product candidates and its target indications. The term of any individual patent depends on applicable law in the country where the patent is granted. In the United States, provided all maintenance fees are timely paid, a patent generally has a term of 20 years from its application filing date or earliest claimed non-provisional filing date. Extensions may be available under certain circumstances, but the life of a patent and, correspondingly, the protection it affords is limited. Palvella’s issued U.S. patents directed to methods of treating keratin hyperproliferation skin disorders with rapamycin naturally expire as late as 2032. Given the amount of time required for the development, testing and regulatory review of new product candidates, patents protecting its product candidates might expire before or shortly after such candidates begin to be commercialized.
If Palvella is unable to obtain new patents with longer expiry dates, its competitors may be able to take advantage of its investment in development and clinical trials by referencing its clinical and preclinical data to obtain approval of competing products following its patent expiration and launch their product earlier than might otherwise be the case.
Palvella may not be able to protect its intellectual property rights throughout the world.
Filing, prosecuting and defending patents on product candidates in all countries throughout the world would be prohibitively expensive, and Palvella’s intellectual property rights in some countries outside the United States can be less extensive than those in the United States or may not be pursued at all outside of the United States. For example, Palvella does not own or license any patent rights directed to QTORIN rapamycin outside of the United States Australia, China, Israel and Japan and Palvella does not own or license any patent rights directed to QTORIN with any other mTOR inhibitors outside of the United States. In addition, the laws of some foreign countries do not protect intellectual property rights to the same extent as federal and state laws in the United States. Consequently, Palvella may not be able to prevent third parties from practicing its inventions in all countries outside the United States, or from selling or importing products made using its inventions in and into the United States or other jurisdictions. Competitors may use its technologies in jurisdictions where Palvella has not obtained patent protection to develop their own products and further, may export otherwise infringing products to territories where Palvella has patent protection, but enforcement is not as strong as that in the United States. These products may compete with Palvella’s products and its patents or other intellectual property rights may not be effective or sufficient to prevent them from competing.
Many companies have encountered significant problems in protecting and defending intellectual property rights in foreign jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents and other intellectual property protection, particularly those relating to biopharmaceuticals, which could make it difficult for Palvella to stop the infringement of its patents or marketing of competing products in violation of its proprietary rights generally. In addition, certain jurisdictions do not protect to the same extent or at all inventions that constitute new methods of treatment. Proceedings to enforce its patent rights in foreign jurisdictions could result in substantial costs and divert its efforts and attention from other aspects of its business, could put its patents at risk of being invalidated or interpreted narrowly and its patent applications at risk of not issuing and could provoke third parties to assert claims against Palvella. Palvella may not prevail in any lawsuits that it initiates, and the damages or other remedies awarded, if any, may not be commercially meaningful. Accordingly, Palvella’s efforts to enforce its intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that it develops or licenses.
Many countries, including European Union countries, India, Japan and China, have compulsory licensing laws under which a patent owner may be compelled under specified circumstances to grant licenses to third parties. In those countries, Palvella may have limited remedies if patents are infringed or if Palvella is compelled to grant a license to a third party, which could materially diminish the value of those patents. This could limit Palvella’s potential revenue opportunities. Accordingly, Palvella’s efforts to enforce its intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that it develops or licenses.
Changes in U.S. patent law or the patent law of other countries or jurisdictions could diminish the value of patents in general, thereby impairing Palvella’s ability to protect its products.
Changes in either the patent laws or interpretation of the patent laws in the United States could increase the uncertainties and costs surrounding the prosecution of patent applications and the enforcement or defense of issued patents. Assuming that other requirements for patentability are met, prior to March 2013, in the United States, the first to invent the claimed invention was entitled to the patent, while outside the United States, the first to file a patent application was entitled to the patent. After March 2013, under the Leahy-Smith America Invents Act, or the “America Invents Act”, the United States transitioned to a first inventor to file system in which, assuming that other requirements for patentability are met, the first inventor to file a patent application will be entitled to the patent on an invention regardless of whether a third party was the first to invent the claimed invention. A third party that files a patent application in the USPTO after March 2013, but before Palvella could therefore be awarded a patent covering an invention of Palvella’s even if Palvella had made the invention before it was made by such third party. This will require Palvella to be cognizant of the time from invention to filing of a patent application. Since patent applications in the United States and most other countries are confidential for a period of time after filing or until issuance, Palvella cannot be certain that it or its licensors were the first to either (i) file any patent application related to its product candidates or (ii) invent any of the inventions claimed in its or its licensor’s patents or patent applications. The America Invents Act also includes a number of significant changes that affect the way patent applications are prosecuted and also may affect patent litigation. These include allowing third-party submission of prior art to the USPTO, during patent prosecution and additional procedures to attack the validity of a patent by USPTO-administered post-grant proceedings, including post-grant review, inter partes review and derivation proceedings. Because of a lower evidentiary standard in USPTO proceedings compared to the evidentiary standard in United States federal courts necessary to invalidate a patent claim, a third party could potentially provide evidence in a USPTO proceeding sufficient for the USPTO to hold a claim invalid even though the same evidence would be insufficient to invalidate the claim if first presented in a district court action. Accordingly, a third party may attempt to use the USPTO procedures to invalidate Palvella’s patent claims that would not have been invalidated if first challenged by the third party as a defendant in a district court action. Therefore, the America Invents Act and its implementation could increase the uncertainties and costs surrounding the prosecution of Palvella’s patent applications and the enforcement or defense of its issued patents, all of which could have an adverse effect on its business, financial condition, results of operations and prospects.
In addition, the U.S. Supreme Court has ruled on several patent cases in recent years, either narrowing the scope of patent protection available in certain circumstances or weakening the rights of patent owners in certain situations. In addition to increasing uncertainty with regard to Palvella’s ability to obtain patents in the future, this combination of events has created uncertainty with respect to the value of patents, once obtained. Depending on actions by the U.S. Congress, the federal courts, and the USPTO, the laws and regulations governing patents could change in unpredictable ways that could weaken Palvella’s ability to obtain new patents or to enforce patents that it owns, has licensed or might obtain in the future. Similarly, changes in patent law and regulations in other countries or jurisdictions, changes in the governmental bodies that enforce them or changes in how the relevant governmental authority enforces patent laws or regulations may weaken Palvella’s ability to obtain new patents or to enforce patents that it owns or has licensed or that it may obtain in the future.
Obtaining and maintaining Palvella’s patent protection depends on compliance with various procedural, document submission, fee payment and other requirements imposed by governmental patent agencies, and its patent protection could be reduced or eliminated for noncompliance with these requirements.
Periodic maintenance fees on any issued patent are due to be paid to the USPTO and other foreign patent agencies in several stages over the lifetime of the patent. The USPTO and various foreign national or international patent agencies require compliance with a number of procedural, documentary, fee payment and other similar provisions during the patent application process. While an inadvertent lapse can in many cases be cured by payment of a late fee or by other means in accordance with the applicable rules, there are situations in which noncompliance can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. Noncompliance events that could result in abandonment or lapse of patent rights include, but are not limited to, failure to timely file national and regional stage patent applications based on its international patent application, failure to respond to official actions within prescribed time limits, non-payment of fees and failure to properly legalize and submit formal documents. If Palvella or its licensors fail to maintain the patents and patent applications covering any of its product candidates, its competitors might be able to enter the market earlier than anticipated, which could materially adversely affect its business, financial condition, results of operations and prospects.
Palvella’s reliance on third parties requires it to share its trade secrets, which increases the possibility that a competitor will discover them or that its trade secrets will be misappropriated or disclosed.
Because Palvella expects to rely on third parties to manufacture its product candidates, and it expects to continue to collaborate with third parties on the development of its current and future product candidates, it must, at times, share trade secrets with them. Palvella may also conduct joint research and development programs that may require it to share trade secrets under the terms of collaboration or similar agreements. Palvella seeks to protect its proprietary technology in part by entering into CDAs and, if applicable, material transfer agreements, consulting agreements or other similar agreements with its advisors, employees, third-party contractors and consultants prior to beginning research or disclosing proprietary information. These agreements typically limit the rights of the third parties to use or disclose its confidential information, including its trade secrets. Despite the contractual provisions employed when working with third parties, the need to share trade secrets and other confidential information increases the risk that such trade secrets become known by Palvella’s competitors, are inadvertently incorporated into the technology of others, or are disclosed or used in violation of these agreements. Any disclosure, either intentional or unintentional, by Palvella’s employees, the employees of third parties with whom it shares its facilities or third-party consultants and vendors that it engages to perform research, clinical trials or manufacturing activities, or misappropriation by third parties (such as through a cybersecurity breach) of its trade secrets or proprietary information could enable competitors to duplicate or surpass its technological achievements, thus eroding its competitive position in its market. Further, adequate remedies may not exist in the event of unauthorized use or disclosure. Given that Palvella’s proprietary position is based, in part, on its know-how and trade secrets, a competitor’s discovery of its trade secrets or other unauthorized use or disclosure would impair its competitive position and could materially adversely affect its business, financial condition, results of operations and prospects.
In addition, these agreements typically restrict the ability of Palvella’s advisors, employees, third-party contractors and consultants to publish data potentially relating to its trade secrets, although its agreements may contain certain limited publication rights. Policing unauthorized use of Palvella’s or its licensors’ intellectual property is difficult, expensive and time-consuming, and it may be unable to determine the extent of any unauthorized use. Moreover, enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive and time-consuming, and the outcome is unpredictable. In addition, some courts inside and outside the United States are less willing or unwilling to protect trade secrets. Despite Palvella’s efforts to protect its trade secrets, its competitors may discover its trade secrets, either through breach of its agreements with third parties, independent development or publication of information by any of its third-party collaborators. A competitor’s discovery of its trade secrets would impair Palvella’s competitive position and have an adverse impact on its business.
If Palvella is unable to protect the confidentiality of its proprietary information and know-how, the value of its technology and products could be adversely affected.
Palvella may not be able to protect its proprietary information and technology adequately. Although Palvella uses reasonable efforts to protect its proprietary information, technology, and know-how, its employees, consultants, contractors, outside scientific advisors, licensors or licensees may unintentionally or willfully disclose its information to competitors. Palvella relies, in part, on non-disclosure and CDAs with its employees, consultants and other parties to protect its proprietary information, technology, and know-how. These agreements may be breached and Palvella may not have adequate remedies for any breach. Moreover, others may independently develop similar or equivalent proprietary information, and third parties may otherwise gain access to Palvella’s proprietary knowledge.
Palvella may be subject to claims challenging the inventorship or ownership of the patents and other intellectual property that it owns or licenses.
Palvella or its licensors may be subject to claims that former employees, collaborators or other third parties have an ownership interest in the patents and intellectual property that it owns or licenses or that it may own or license in the future. While it is Palvella’s policy to require its employees and contractors who may be involved in the development of intellectual property to execute agreements assigning such intellectual property to it, Palvella may be unsuccessful in executing such an agreement with each party who in fact develops intellectual property that it regards as its own; its licensors may face similar obstacles. Palvella could be subject to ownership disputes arising, for example, from conflicting obligations of consultants or others who are involved in developing its product candidates. Litigation may be necessary to defend against any claims challenging inventorship or ownership. If Palvella or its licensors fail in defending any such claims, it may have to pay monetary damages and may lose valuable intellectual property rights, such as exclusive ownership of, or right to use, intellectual property, which could materially adversely affect its business, financial condition, results of operations and prospects. Even if Palvella is successful in defending against such claims, litigation could result in substantial costs and be a distraction to management and other employees. Any of the foregoing could have a material adverse effect on Palvella’s business, financial condition, results of operations and prospects.
Palvella may become subject to claims alleging infringement of third parties’ patents or proprietary rights and/or claims seeking to invalidate its patents, which would be costly, time consuming and, if successfully asserted against it, delay or prevent the development and commercialization of its current product candidates or any future product candidates.
Palvella’s commercial success depends in part on its and its licensors avoiding infringement and other violations of the patents and proprietary rights of third parties. However, Palvella’s research, development and commercialization activities may be subject to claims that it infringes or otherwise violates patents or other intellectual property rights owned or controlled by third parties. There is a substantial amount of litigation, worldwide, involving patent and other intellectual property rights in the biotechnology and pharmaceutical industries, including patent infringement lawsuits, interferences, derivation and administrative law proceedings, inter partes review and post-grant review before the USPTO, as well as oppositions and similar processes in foreign jurisdictions. Numerous United States and foreign issued patents and pending patent applications, which are owned by third parties, exist in the fields in which Palvella and its collaborators are developing product candidates.
Palvella cannot assure you that its exploitation of QTORIN rapamycin or any future product candidate will not infringe existing or future third-party patents. Because patent applications can take many years to issue and may be confidential for 18 months or more after filing, there may be applications now pending of which Palvella is unaware and which may later result in issued patents that it may infringe by commercializing any of its product candidates, if approved. As the biotechnology and pharmaceutical industries expand and more patents are issued, and as Palvella gains greater visibility and market exposure as a public company, the risk increases that its product candidates or other business activities may be subject to claims of infringement of the patent and other proprietary rights of third parties. Third parties may assert that Palvella is infringing their patents or employing their proprietary technology without authorization. Moreover, Palvella may face claims from non-practicing entities that have no relevant product revenue and against whom its own patent portfolio may thus have no deterrent effect.
Palvella may be subject to third-party claims in the future against it or its collaborators that would cause it to incur substantial expenses and, if successful against it, could cause Palvella to pay substantial damages, including treble damages and attorney’s fees if Palvella is found to be willfully infringing a third party’s patents. Palvella may be required to indemnify future collaborators against such claims. If a patent infringement suit were brought against Palvella or its future collaborators, it or they could be forced to stop or delay research, development, manufacturing or sales of the product or product candidate that is the subject of the suit. If any third-party patents were held by a court of competent jurisdiction to cover the manufacturing process of any of its product candidates, any molecules formed during the manufacturing process or any final product itself, the holders of any such patents may be able to block Palvella’s ability to commercialize such product candidate unless it obtained a license under the applicable patents, or until such patents expire. Similarly, if any third-party patent was to be held by a court of competent jurisdiction to cover aspects of its formulations, processes for manufacture or methods of use, including combination therapy, the holders of any such patent may be able to block Palvella’s ability to develop and commercialize the applicable product candidate unless it obtained a license or until such patent expires. As a result of patent infringement claims, or in order to avoid potential claims, Palvella or its collaborators may choose to seek, or be required to seek, a license from the third party and would most likely be required to pay license fees or royalties or both. These licenses may not be available on acceptable terms, or at all. Even if Palvella or its future collaborators were able to obtain a license, the rights obtained may be nonexclusive, which would not confer a competitive advantage to Palvella from an exclusivity perspective. Ultimately, Palvella could be prevented from commercializing a product, or forced to redesign it, or to cease some aspect of its business operations if, as a result of actual or threatened patent infringement claims, it or its collaborators are unable to enter into licenses on acceptable terms to necessary third-party patent rights. Even if Palvella is successful in defending against such claims, such litigation can be expensive and time consuming to litigate and would divert management’s attention from its core business. Claims that Palvella has misappropriated the confidential information or trade secrets of third parties could have a similar negative impact on its business. Any of these events could materially adversely affect Palvella’s business, financial condition, results of operations and prospects.
Some of Palvella’s competitors may be able to sustain the costs of complex intellectual property litigation more effectively than it can because they have substantially greater resources. In addition, intellectual property litigation, regardless of its outcome, may cause negative publicity, adversely impact prospective customers, cause product shipment delays, or prohibit Palvella from manufacturing, marketing or otherwise commercializing its products, services and technology. Any uncertainties resulting from the initiation and continuation of any litigation could adversely impact Palvella’s ability to raise additional funds or otherwise materially adversely affect its business, financial condition, results of operations and prospects.
Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of Palvella’s confidential information could be compromised by disclosure during this type of litigation. There could also be public announcements of the results of hearings, motions or other interim proceedings or developments, which could adversely impact the price of Palvella common stock. If securities analysts or investors perceive these results to be negative, it could adversely impact the price of Palvella common stock. The occurrence of any of these events could materially adversely affect Palvella’s business, financial condition, results of operations and prospects.
In addition to infringement claims against Palvella, if third parties prepare and file patent applications in the United States that also claim technology similar or identical to Palvella’s, it may have to participate in interference or derivation proceedings in the USPTO, to determine which party is entitled to a patent on the disputed invention. Palvella may be subject to a third-party pre-issuance submission of prior art to the USPTO or become involved in derivation, reexamination, inter partes review, post-grant review or interference proceedings or similar opposition proceedings in the EPO or similar offices in other jurisdictions regarding its intellectual property rights with respect to its products and technology. Since patent applications are confidential for a period of time after filing, Palvella cannot be certain that it was the first to file any patent application related to its product candidates.
Palvella may not identify relevant third-party patents or may incorrectly interpret the relevance, scope or expiration of a third-party patent, which might adversely affect its ability to develop and market its product candidates.
Palvella cannot guarantee that any of its or its licensors’ patent searches or analyses, including the identification of relevant patents, the scope of patent claims or the expiration of relevant patents, are complete or thorough, nor can Palvella be certain that Palvella has identified each and every third-party patent and pending application in the United States and abroad that is or may be relevant to or necessary for the commercialization of its product candidates in any jurisdiction. Patent applications in the United States and elsewhere are not published until approximately 18 months after the earliest filing for which priority is claimed, with such earliest filing date being commonly referred to as the priority date. In addition, U.S. patent applications filed before November 29, 2000 and certain U.S. patent applications filed after that date that will not be filed outside the United States remain confidential until patents issue. Therefore, patent applications covering Palvella’s products could have been filed by others without its knowledge. Additionally, pending patent applications that have been published can, subject to certain limitations, be later amended in a manner that could cover Palvella’s product candidates or the use of its product candidates.
The scope of a patent claim is determined by an interpretation of the law, the written disclosure in a patent and the patent’s prosecution history. Palvella’s interpretation of the relevance or the scope of a patent or a pending application may be incorrect, which may negatively impact its ability to market its product candidates. Palvella may incorrectly determine that its product candidates are not covered by a third-party patent or may incorrectly predict whether a third party’s pending application will issue with claims of relevant scope. Palvella’s determination of the expiration date of any patent in the United States or abroad that it considers relevant may be incorrect, and its failure to identify and correctly interpret relevant patents, may negatively impact its ability to develop and market its product candidates.
If Palvella fails to identify and correctly interpret relevant patents, it may be subject to infringement claims. Palvella cannot guarantee that it will be able to successfully settle or otherwise resolve such infringement claims. If Palvella fails in any such dispute, in addition to being forced to pay damages, it may be temporarily or permanently prohibited from commercializing any of its product candidates that are held to be infringing. Palvella might, if possible, also be forced to redesign product candidates or services so that it no longer infringes the third-party intellectual property rights. Any of these events, even if Palvella was ultimately to prevail, could require it to divert substantial financial and management resources that it would otherwise be able to devote to its business.
Palvella may be subject to claims by third parties asserting that it, its employees or its licensors have misappropriated their intellectual property, including trade secrets, or claiming ownership of what it regards as its own intellectual property.
Many of Palvella’s employees and its licensor’s employees were previously employed at other biotechnology or pharmaceutical companies. Although Palvella and its licensors try to ensure that its employees and its licensor’s employees do not use the intellectual property rights, proprietary information or know-how of others in their work for Palvella, including by contract, it or its licensors may be subject to claims that these employees, its licensors or Palvella has used or disclosed intellectual property, including trade secrets or other proprietary information, of any such employee’s former employer. Litigation may be necessary to defend against these claims. There is no guarantee of success in defending these claims, and if Palvella fails in defending any such claims, in addition to paying monetary damages, it may lose valuable intellectual property rights, such as exclusive ownership of, or right to use, valuable intellectual property. Even if Palvella is successful, litigation could result in substantial cost and be a distraction to its management and other employees. Moreover, any such litigation or the threat thereof may adversely affect Palvella’s reputation, its ability to form strategic alliances or sublicense its rights to collaborators, engage with scientific advisors or hire employees or consultants, each of which could materially adversely affect its business, financial condition, results of operations and prospects.
In addition, while it is Palvella’s policy to require its employees and contractors who may be involved in the development of intellectual property to execute agreements assigning such intellectual property to it, Palvella may in the future be unsuccessful in executing such an agreement with each party who in fact develops intellectual property that it regards as its own. Palvella’s and their assignment agreements may not be self-executing or may be breached, and it may be forced to bring claims against third parties, or defend claims they may bring against it, to determine the ownership of what it regards as its intellectual property.
If Palvella or its licensor fail in prosecuting or defending any such claims, in addition to paying monetary damages, it may lose valuable intellectual property rights or personnel. Such intellectual property rights could be awarded to a third party, and Palvella could be required to obtain a license from such third party to commercialize its technology or products, which license may not be available on commercially reasonable terms, or at all, or such license may be non-exclusive. Even if Palvella and its licensor are successful in prosecuting or defending against such claims, litigation could result in substantial costs.
Palvella may become involved in lawsuits to protect or enforce its patents or other intellectual property or the patents of its licensors, which could be expensive and time-consuming.
Competitors may infringe, misappropriate or otherwise violate its intellectual property, including its patents or the patents of its licensors. As a result, Palvella may be required to file infringement claims or inform and cooperate with its licensors to stop third-party infringement or unauthorized use. This can be expensive, particularly for a company of Palvella’s size, and time-consuming. In addition, in an infringement proceeding, a court may decide that a patent of Palvella’s is not valid or is unenforceable, or may refuse to stop the other party from using the technology at issue on the grounds that its patent claims do not cover its technology or that the factors necessary to grant an injunction against an infringer are not satisfied. An adverse determination of any litigation or other proceedings could put one or more of Palvella’s patents at risk of being invalidated, interpreted narrowly or amended such that they do not cover its product candidates. Moreover, such adverse determinations could put Palvella’s patent applications at risk of not issuing, or issuing with limited and potentially inadequate scope to cover its product candidates or to prevent others from marketing similar products.
Interference, derivation or other proceedings brought at the USPTO may be necessary to determine the priority or patentability of inventions with respect to Palvella’s patent applications or those of its licensors or potential partners. Litigation or USPTO proceedings brought by Palvella may fail or may be invoked against it by third parties. Even if Palvella is successful, domestic or foreign litigation or USPTO or foreign patent office proceedings may result in substantial costs. Palvella may not be able, alone or with its licensors or potential partners, to prevent misappropriation of its proprietary rights, particularly in countries where the laws may not protect such rights as fully as in the United States. The initiation of a claim against a third party may also cause the third party to bring counterclaims against Palvella such as claims asserting that its patents are invalid or unenforceable.
In patent litigation in the United States, defendant counterclaims alleging invalidity or unenforceability are commonplace. Grounds for a validity challenge could be an alleged failure to meet any of several statutory requirements, including lack of novelty, obviousness, non-enablement, lack of sufficient written description, obviousness-type double patenting or lack of statutory subject matter. Grounds for an unenforceability assertion could be an allegation that someone connected with prosecution of the patent withheld relevant material information from the USPTO, or made a materially misleading statement, during prosecution. Third parties may also raise similar validity claims before the USPTO in post-grant proceedings such as ex parte reexaminations, inter partes review, or post-grant review, or oppositions or similar proceedings outside the United States, in parallel with litigation or even outside the context of litigation. The outcome following legal assertions of invalidity and unenforceability is unpredictable and could result in the revocation, cancelation, amendment or shortening of term of patents Palvella owns or licenses. Palvella cannot be certain that there is no invalidating prior art, of which it and the patent examiner were unaware during prosecution. For the patents and patent applications that Palvella has licensed, it may have limited or no right to participate in the defense of any licensed patents against challenge by a third party. If a defendant were to prevail on a legal assertion of invalidity or unenforceability, Palvella would lose at least part, and perhaps all, of any future patent protection on its current or future product candidates. Such a loss of patent protection could harm Palvella’s business. Palvella may find it impractical or undesirable to enforce its intellectual property against some third parties. For instance, Palvella is aware of third parties that have marketed topical rapamycin formulations for the treatment of skin diseases over the internet or through compound pharmacies. These parties do not appear to have regulatory approval, and Palvella has not authorized them in any way to market these products. However, to date Palvella has refrained from seeking to enforce its intellectual property rights against these third parties due to the transient nature of their activities.
Palvella may not be able to detect or prevent, alone or with its licensors, misappropriation of its intellectual property rights, particularly in countries where the laws may not protect those rights as fully as in the United States. Any litigation or other proceedings to enforce its intellectual property rights may fail, and even if successful, may result in substantial costs and distract Palvella’s management and other employees.
Even if Palvella establishes infringement, the court may decide not to grant an injunction against further infringing activity and instead award only monetary damages, which may or may not be an adequate remedy. Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation or other proceedings, there is a risk that some of its confidential information could be compromised by disclosure during this type of litigation or other proceedings. In addition, during the course of this kind of litigation or proceedings, there could be public announcements of the results of hearings, motions or other interim proceedings or developments or public access to related documents. If investors perceive these results to be negative, the market price for Palvella common stock could be significantly harmed.
Intellectual property litigation could cause Palvella to spend substantial resources and distract its personnel from their normal responsibilities, and have a harmful effect on the success of its business.
Even if resolved in Palvella’s favor, litigation or other legal proceedings relating to intellectual property claims may cause it to incur significant expenses, and could distract its technical and management personnel from their normal responsibilities. Such litigation or proceedings could substantially increase Palvella’s operating losses and reduce the resources available for development activities or any future sales, marketing or distribution activities. Palvella may not have sufficient financial or other resources to conduct such litigation or proceedings adequately. In addition, the uncertainties associated with litigation could compromise Palvella’s ability to compete in the marketplace or to raise the funds necessary to continue its clinical trials and internal research programs, or in-license needed technology or other product candidates. Some of its competitors may be able to sustain the costs of such litigation or proceedings more effectively than Palvella can because of their greater financial resources. Accordingly, despite Palvella’s efforts, it may not be able to prevent third parties from infringing upon or misappropriating its intellectual property.
Palvella may need to license additional intellectual property from third parties, and such licenses may not be available or may not be available on commercially reasonable terms.
Additional third parties, apart from Palvella’s current licensors, may hold intellectual property, including patent rights, that are important or necessary to the development of its product candidates. It may be necessary for Palvella to use the patented or proprietary technology of these third parties to commercialize its product candidates, in which case it would be required to obtain a license from these third parties on commercially reasonable terms. Such a license may not be available, or it may not be available on commercially reasonable terms, in which case Palvella’s business would be harmed. The risks described elsewhere pertaining to Palvella’s intellectual property rights also apply to the intellectual property rights that Palvella in-licenses, and any failure by Palvella or its licensors to obtain, maintain, defend and enforce these rights could harm its business. In some cases, Palvella may not have control over the prosecution, maintenance or enforcement of the patents that it licenses, and may not have sufficient ability to provide input into the patent prosecution, maintenance and defense process with respect to such patents, and its licensors may fail to take the steps that it believes are necessary or desirable in order to obtain, maintain, defend and enforce the licensed patents.
Palvella’s intellectual property agreements with third parties may be subject to disagreements over contract interpretation, which could narrow the scope of its rights to the relevant intellectual property or technology or increase its financial or other obligations to its licensors.
Certain provisions in Palvella’s intellectual property agreements may be susceptible to multiple interpretations. Disputes may arise between Palvella and any of these counterparties regarding intellectual property rights that are subject to such agreements, including, but not limited to:
|
● |
the scope of rights granted under the agreement and other interpretation-related issues; |
|
● |
whether and the extent to which its technology and processes infringe on intellectual property of the licensor that is not subject to the agreement; |
|
● |
its right to sublicense patent and other rights to third parties; |
|
● |
its diligence obligations with respect to the use of the licensed technology in relation to its development and commercialization of its product candidates, and what activities satisfy those diligence obligations; |
|
● |
the ownership of inventions and know-how resulting from the joint creation or use of intellectual property by its licensors and Palvella and its partners; |
|
● |
its right to transfer or assign its license; |
|
● |
the effects of termination; and |
|
● |
the priority of invention of patented technology. |
The resolution of any contract interpretation disagreement that may arise could affect the scope of its rights to the relevant intellectual property or technology, or affect financial or other obligations under the relevant agreement, either of which could have a material adverse effect on Palvella’s business, financial condition, results of operations and prospects.
If Palvella fails to comply with its obligations under any license, collaboration or other agreements, it may be required to pay damages and could lose intellectual property rights that are necessary or useful for developing and protecting its product candidates.
Palvella has licensed, or may in the future license, certain intellectual property rights relating to its technology and indications of interest from third parties. If Palvella materially breaches or fails to perform any provision under these license agreements, including failure to make payments to a licensor when due for royalties or milestones and failure to use commercially reasonable efforts to develop and commercialize the licensed technology, such licensors may have the right to terminate Palvella’s license agreement. If, for any reason, one or more of its agreements with such third parties is terminated or Palvella otherwise loses those rights, it could harm its business. Palvella’s license and other agreements impose, and any future collaboration agreements or license agreements it enters into are likely to impose various development, commercialization, funding, milestone, royalty, diligence, sublicensing, insurance, patent prosecution and enforcement or other obligations on Palvella. If Palvella breaches any such material obligations, or uses the intellectual property licensed to it in an unauthorized manner, it may be required to pay damages and the licensor may have the right to terminate the license or otherwise acquire intellectual property rights from Palvella, which could result in it being unable to develop, manufacture and sell products that are covered by the licensed technology, or having to negotiate new or reinstated licenses on less favorable terms, or enable a competitor to gain access to the licensed technology.
If Palvella’s trademarks and trade names are not adequately protected, then it may not be able to build name recognition in its markets of interest and its business may be adversely affected.
Palvella’s registered or unregistered trademarks or trade names may be challenged, infringed, circumvented, declared generic or conflict with third-party rights. Palvella may not be able to protect its rights to these trademarks and trade names, which it needs to build name recognition by potential partners or customers in its markets of interest. In addition, third parties may file first for Palvella’s trademarks in certain countries. If they succeeded in registering such trademarks, and if Palvella was not successful in challenging such third-party rights, it may not be able to use these trademarks to market its products in those countries. In such cases, over the long term, if Palvella is unable to establish name recognition based on its trademarks and trade names, then its marketing abilities may be impacted.
Palvella has not yet registered trademarks for a commercial trade name for its lead candidates in the United States or foreign jurisdictions and failure to secure such registrations could adversely affect its business.
Palvella has not yet registered trademarks for a commercial trade name for its lead product candidates in the United States or any foreign jurisdiction. During trademark registration proceedings, Palvella may receive rejections. Although Palvella is given an opportunity to respond to those rejections, it may be unable to overcome such rejections. In addition, in the USPTO and in comparable agencies in many foreign jurisdictions, third parties are given an opportunity to oppose pending trademark applications and to seek to cancel registered trademarks. Opposition or cancelation proceedings may be filed against Palvella’s trademarks, and its trademarks may not survive such proceedings. Moreover, any name Palvella proposes to use with its product candidates in the United States must be approved by the FDA, regardless of whether Palvella has registered it, or applied to register it, as a trademark. The FDA typically conducts a review of proposed product names, including an evaluation of potential for confusion with other product names. If the FDA objects to any of Palvella’s proposed proprietary product names, it may be required to expend significant additional resources in an effort to identify a suitable substitute name that would qualify under applicable trademark laws, not infringe the existing rights of third parties and be acceptable to the FDA.
Furthermore, QTORIN is Palvella’s proprietary name for its technology platform. Any future commercial tradename for Palvella’s lead product candidates will be subject to approval by the FDA for commercial use and will not include the QTORIN mark. Accordingly, any goodwill and recognition that Palvella has built for the name in relation to future commercial drug products may be lost.
Intellectual property rights do not necessarily address all potential threats to Palvella’s competitive advantage.
The degree of future protection afforded by Palvella’s intellectual property rights is uncertain because intellectual property rights have limitations, and may not adequately protect its business, or permit it to maintain its competitive advantage. The following examples are illustrative:
|
● |
others may be able to make products that are the same as or similar to Palvella’s product candidates, but that are not covered by the claims of the patents or other intellectual property rights that Palvella owns that it has exclusively licensed and has the right to enforce; |
|
● |
Palvella, its licensors or any collaborators might not have been the first to make the inventions covered by the issued patents or pending patent applications that it owns or licenses; |
|
● |
Palvella or its licensors might not have been the first to file patent applications covering certain of its inventions; |
|
● |
others may independently develop similar or alternative technologies or duplicate any of Palvella’s technologies without infringing its intellectual property rights; |
|
● |
it is possible that Palvella’s pending patent applications will not lead to issued patents; |
|
● |
issued patents that Palvella owns or licenses may not provide it with any competitive advantages, or may be held invalid or unenforceable as a result of legal challenges; |
|
● |
Palvella’s competitors might conduct research and development activities in the United States and other countries that provide a safe harbor from patent infringement claims for certain research and development activities, as well as in countries where it does not have patent rights, and then use the information learned from such activities to develop competitive products for sale in its major commercial markets; |
|
● |
there may be significant pressure on the U.S. government and international governmental bodies to limit the scope of patent protection both inside and outside the United States for disease treatments that prove successful, as a matter of public policy regarding worldwide health concerns; |
|
● |
countries other than the U.S. may, under certain circumstances, force Palvella or its licensors to grant a license under its patents to a competitor, thus allowing the competitor to compete with it in that jurisdiction or forcing Palvella to lower the price of its drug in that jurisdiction; |
|
● |
Palvella cannot ensure that any of its patents, or any of its pending patent applications, if issued, or those of its licensors, will include claims having a scope sufficient to protect its product candidates; |
|
● |
Palvella may not be able to generate sufficient data to support full patent applications that protect the entire breadth of developments in one or more of its programs; |
|
● |
Palvella has engaged in scientific collaborations in the past and will continue to do so in the future and its collaborators may develop adjacent or competing products that are outside the scope of its patents; |
|
● |
Palvella cannot ensure that its commercial activities or product candidates will not infringe upon the patents of others; |
|
● |
it is possible that there are unpublished applications or patent applications maintained in secrecy that may later issue with claims covering its products or technology similar to Palvella’s; |
|
● |
Palvella may not develop additional proprietary technologies that are patentable; |
|
● |
Palvella may not have sufficient time remaining on the term of its patents or the term of its marketing exclusivity to warrant commercialization of its product candidates; |
|
● |
the patents or intellectual property rights of others may harm Palvella’s business; and |
|
● |
Palvella may choose not to file a patent in order to maintain certain trade secrets or know-how, and a third party may subsequently file a patent covering such intellectual property. |
Risks Related to the Combined Company
In determining whether you should approve the issuance of shares of Pieris common stock, the change of control resulting from the Merger and other matters related to the Merger, as applicable, you should carefully read the following risk factors in addition to the risks described above.
If any of the events described in “Risks Related to Pieris” or “Risks Related to Palvella” occur, those events could cause potential benefits of the Merger not to be realized.
Following completion of the Merger, the combined company will be susceptible to many of the risks described in the sections herein entitled “Risks Related to Pieris” and “Risks Related to Palvella,” including those incorporated by reference to Pieris’s filings with the SEC. To the extent any of the events in the risks described in those sections occurs, the potential benefits of the Merger may not be realized, and the results of operations and financial condition of the combined company could be adversely affected in a material way. This could cause the market price of the combined company common stock to decline.
The market price of the combined company common stock is expected to be volatile, and the market price of the common stock may drop following the Merger.
The market price of the combined company common stock following the Merger could be subject to significant fluctuations. Some of the factors that may cause the market price of the combined company common stock to fluctuate include:
|
● |
timing and results of Investigational New Drug Applications, preclinical studies and clinical trials of the combined company’s product candidates, or those of the combined company’s competitors or the combined company’s existing or future collaborators; |
|
● |
the success of competitive products or announcements by potential competitors of their product development efforts; |
|
● |
failure to meet or exceed financial and development projections the combined company may provide to the public; |
|
● |
failure to meet or exceed the financial and development projections of the investment community; |
|
● |
if the combined company does not achieve the perceived benefits of the Merger as rapidly or to the extent anticipated by financial or industry analysts; |
|
● |
announcements of significant acquisitions, strategic collaborations, joint ventures or capital commitments by the combined company or its competitors; |
|
● |
actions taken by regulatory agencies with respect to the combined company’s product candidates, clinical studies, manufacturing process or sales and marketing terms; |
|
● |
disputes or other developments relating to proprietary rights, including patents, trade secrets, litigation matters, and the combined company’s ability to obtain patent protection for its technologies or maintain its trade secrets; |
|
● |
disputes or termination of agreements with third parties, including CMOs, supporting with the manufacturing and clinical studies of the combined company’s product candidates; |
|
● |
additions or departures of key personnel; |
|
● |
significant lawsuits, including patent or stockholder litigation; |
|
● |
if securities or industry analysts do not publish research or reports about the combined company’s business, or if they issue adverse or misleading opinions regarding its business and stock; |
|
● |
if the combined company does not achieve the perceived benefits of the Merger as rapidly or to the extent anticipated by financial or industry analysts; |
|
● |
changes in the market valuations of similar companies; |
|
● |
geo-political developments, general market or macroeconomic conditions including inflation and interest rates; |
|
● |
market conditions in the pharmaceutical and biotechnology sectors; |
|
● |
expiration of market stand-off or lock-up agreements; |
|
● |
changes in the structure of healthcare payment systems; |
|
● |
announcement of expectation of additional financing efforts; |
|
● |
sales of securities by the combined company or its securityholders in the future; |
|
● |
if the combined company fails to raise an adequate amount of capital to fund its operations and continued development of its product candidates; |
|
● |
trading volume of the combined company common stock; |
|
● |
publicity or announcements by competitors of new commercial products, clinical progress or lack thereof, significant contracts, commercial relationships or capital commitments; |
|
● |
the impact of any natural disasters or public health emergencies; |
|
● |
the introduction of technological innovations or new product candidates that compete with the products and services of the combined company; |
|
● |
changes in accounting standards, policies, guidelines, interpretations or principles; and |
|
● |
period-to-period fluctuations in the combined company’s financial results. |
Moreover, the stock markets in general have experienced substantial volatility that has often been unrelated to the operating performance of individual companies. These broad market fluctuations may also adversely affect the trading price of the combined company common stock. In addition, macroeconomic conditions, a recession, depression or other sustained adverse market event resulting from the spread of diseases or otherwise could materially and adversely affect the combined company’s business and the value of its common stock. In the past, following periods of volatility in the market price of a company’s securities, stockholders have often instituted class action securities litigation against such companies. Furthermore, market volatility may lead to increased stockholder activism if the combined company experiences a market valuation that activists believe is not reflective of its intrinsic value. Activist campaigns that contest or conflict with the combined company’s strategic direction or seek changes in the composition of its board of directors could have an adverse effect on its operating results and financial condition.
The combined company will need substantial additional funding, in addition to the funds from the PIPE Financing before it can complete the development of its product candidates. If the combined company is unable to obtain such additional capital on favorable terms, on a timely basis or at all, it would be forced to delay, reduce or eliminate its product development and clinical programs and may not have the capital required to otherwise operate its business.
Developing pharmaceutical products, including conducting preclinical studies and clinical trials, is expensive. The combined company has not generated any revenues from the commercial sale of products and will not be able to generate any product revenues until, and only if, the combined company receives approval to sell its product candidates from the FDA or other regulatory authorities. The cash expected from both Pieris and Palvella at closing, including the net proceeds of the PIPE Financing, are expected to be sufficient to fund the combined company’s operating expenses into the second half of 2027. However, as the combined company has not generated any revenue from commercial sales to date and does not expect to generate any revenue for several years, if ever, the combined company will need to raise substantial additional capital in order to fund its general corporate activities and to fund its research and development, including its currently planned clinical trials and plans for new clinical trials and product development.
The combined company may seek to raise additional funds through various potential sources, such as equity and debt financings, or through strategic collaborations and license agreements. The combined company can give no assurances that it will be able to secure such additional sources of funds to support its operations or, if such funds are available, that such additional financing will be sufficient to meet its needs. Moreover, to the extent that the combined company raises additional funds by issuing equity securities, including through the PIPE Financing, its stockholders may experience additional significant dilution and new investors could gain rights, preferences and privileges senior to the holders of common stock. Debt financing, if available, may involve restrictive covenants. To the extent that the combined company raises additional funds through collaboration and licensing arrangements, it may be necessary to relinquish some rights to its technologies or product candidates, or grant licenses on terms that may not be favorable.
Given the combined company’s capital constraints, it will need to prioritize spending on its clinical and preclinical programs. If the combined company is unable to raise sufficient funds to support its current and planned operations, it may elect to discontinue certain of its ongoing activities or programs. The combined company’s inability to raise additional funds could also prevent it from taking advantage of opportunities to pursue promising new or existing programs in the future.
The combined company’s forecasts regarding its beliefs in the sufficiency of its financial resources to support its current and planned operations are forward-looking statements and involve significant risks and uncertainties, and actual results could vary as a result of a number of factors, including the factors discussed elsewhere in this “Risk Factors” section. These estimates are based on assumptions that may prove to be wrong, and the combined company could utilize its available capital resources sooner than currently expected.
The combined company expects to incur losses for the foreseeable future and might never achieve profitability.
The combined company may never become profitable, even if the combined company is able to complete clinical development for one or more product candidates and eventually commercialize such product candidates. The combined company will need to successfully complete significant research, development, testing and regulatory compliance activities that, together with projected general and administrative expenses, is expected to result in substantial increased operating losses for at least the next several years. Even if the combined company does achieve profitability, it may not be able to sustain or increase profitability on a quarterly or annual basis.
The combined company will incur additional costs and increased demands upon management as a result of complying with the laws and regulations affecting public companies.
The combined company will incur significant legal, accounting and other expenses as a public company that Palvella did not incur as a private company, including costs associated with public company reporting obligations under the Exchange Act. The combined company’s management team will consist of the executive officers of Palvella prior to the Merger, some of whom have not previously managed and operated a public company. These executive officers and other personnel will need to devote substantial time to gaining expertise related to public company reporting requirements and compliance with applicable laws and regulations to ensure that the combined company complies with all of these requirements. Any changes the combined company makes to comply with these obligations may not be sufficient to allow it to satisfy its obligations as a public company on a timely basis, or at all. These reporting requirements, rules and regulations, coupled with the increase in potential litigation exposure associated with being a public company, could also make it more difficult for the combined company to attract and retain qualified persons to serve on the board of directors or on board committees or to serve as executive officers, or to obtain certain types of insurance, including directors’ and officers’ insurance, on acceptable terms.
If the combined company no longer qualifies as a smaller reporting company or otherwise does not qualify for applicable exemptions, the combined company will be subject to additional laws and regulations affecting public companies that will increase the combined company’s costs and the demands on management and could harm the combined company’s operating results.
The combined company will be subject to the reporting requirements of the Exchange Act, which requires, among other things, that the combined company file with the SEC, annual, quarterly and current reports with respect to the combined company’s business and financial condition, as well as other disclosure and corporate governance requirements. However, as a “smaller reporting company”, the combined company may take advantage of exemptions from various requirements such as an exemption from the requirement to have the combined company’s independent auditors attest to the combined company’s internal control over financial reporting under Section 404 and other reduced disclosure obligations regarding executive compensation in the combined company’s periodic reports and proxy statements. If the combined company no longer qualifies as a smaller reporting company or otherwise does not qualify for these exemptions, the combined company will be required to comply with these additional legal and regulatory requirements applicable to public companies and will incur significant legal, accounting and other expenses to do so. If the combined company is not able to comply with the requirements in a timely manner or at all, the combined company’s financial condition or the market price of the combined company common stock may be harmed. For example, if the combined company or its independent auditor identifies deficiencies in the combined company’s internal control over financial reporting that are deemed to be material weaknesses the combined company could face additional costs to remedy those deficiencies, the market price of the combined company’s stock could decline or the combined company could be subject to sanctions or investigations by the SEC or other regulatory authorities, which would require additional financial and management resources.
The unaudited pro forma condensed combined financial data for Pieris and Palvella included in this proxy statement/prospectus is preliminary, and the combined company’s actual financial position and operations after the Merger may differ materially from the unaudited pro forma financial data included in this proxy statement/prospectus.
The unaudited pro forma financial data for Pieris and Palvella included in this proxy statement/prospectus is presented for illustrative purposes only and is not necessarily indicative of the combined company’s actual financial condition or results of operations of future periods, or the financial condition or results of operations that would have been realized had the entities been combined during the periods presented. The unaudited pro forma financial statements have been derived from the historical financial statements of Pieris and Palvella and adjustments and assumptions have been made regarding the combined company after giving effect to the transaction. The information upon which these adjustments and assumptions have been made is preliminary, and these kinds of adjustments and assumptions are difficult to make with accuracy. Moreover, the unaudited pro forma financial statements do not reflect all costs that are expected to be incurred by the combined company in connection with the transactions or that have been incurred since the date of such unaudited pro forma financial statements. The assumptions used in preparing the unaudited pro forma financial information may not prove to be accurate, and other factors may affect the combined company’s financial condition following the transaction. For example, the exchange ratio reflected in this proxy statement/prospectus is preliminary. The final exchange ratio could differ materially from the preliminary exchange ratio used to prepare the pro forma adjustments. The combined company’s actual results and financial position after the Merger may differ materially and adversely from the unaudited pro forma financial data included in this proxy statement/prospectus. For more information see the section titled “Unaudited Pro Forma Condensed Combined Financial Information” beginning on page 299.
Pieris and Palvella are expected to incur significant transaction costs in connection with the Merger, which may be in excess of those anticipated by them.
Pieris and Palvella have incurred and are expected to continue to incur a number of non-recurring costs associated with negotiating and completing the Merger. These costs have been, and will continue to be, substantial and, in many cases, will be borne by Pieris and Palvella whether or not the Merger is completed. A substantial majority of non-recurring expenses will consist of transaction costs and include, among others, fees paid to financial, legal, accounting and other advisors, and filing fees. Pieris and Palvella will continue to assess the magnitude of these costs, and additional unanticipated costs may be incurred in connection with the Merger. While Pieris and Palvella have assumed that a certain level of expenses would be incurred, there are many factors beyond their control that could affect the total amount or the timing of the expenses. The costs described above and any unanticipated costs and expenses, many of which will be borne by Pieris and Palvella even if the Merger is not completed, could have an adverse effect on each of Pieris’ and Palvella’s business, financial condition and results of operations.
Provisions that will be in the combined company’s articles of incorporation and bylaws and provisions under Nevada law could make an acquisition of the combined company, which may be beneficial to its stockholders, more difficult and may prevent attempts by its stockholders to replace or remove its management.
Provisions that will be included in the combined company’s articles of incorporation and bylaws may discourage, delay or prevent a merger, acquisition or other change in control of the combined company that stockholders may consider favorable, including transactions in which its common stockholders might otherwise receive a premium price for their shares. These provisions could also limit the price that investors might be willing to pay in the future for shares of the combined company common stock, thereby depressing the market price of its common stock. In addition, because the combined company’s board of directors will be responsible for appointing the members of the combined company’s management team, these provisions may frustrate or prevent any attempts by the combined company’s stockholders to replace or remove its current management by making it more difficult for stockholders to replace members of the combined company’s board of directors. Among other things, these provisions:
|
● |
allow the authorized number of the combined company’s directors to be changed only by resolution of its board of directors; |
|
● |
limit the manner in which stockholders can remove directors from the combined company’s board of directors; |
|
● |
provide for advance notice requirements for stockholder proposals that can be acted on at stockholder meetings and for nominations to the combined company’s board of directors; |
|
● |
limit who may call stockholder meetings; |
|
● |
require that stockholder actions be effected at a duly called stockholders meeting, and prohibit actions by the combined company’s stockholders by written consent; |
|
● |
authorize the combined company’s board of directors to issue preferred stock without stockholder approval, which could be used to institute a “poison pill” that would work to dilute the stock ownership of a potential hostile acquirer, effectively preventing acquisitions that have not been approved by the combined company’s board of directors; and |
|
● |
require the approval of the holders of at least 80% of the votes that all combined company stockholders would be entitled to cast to amend or repeal certain provisions of the combined company’s articles of incorporation or for the combined company’s stockholders to amend the combined company’s bylaws. |
Moreover, because the combined company is incorporated in Nevada, it is governed by the provisions of NRS, which generally prohibits a person who, together with their affiliates and associates, beneficially owns 10% or more of the company’s outstanding voting stock from, among other things, merging or combining with the company for a period of two years after the date of the transaction in which the person acquired ownership of 10% or more of the company’s outstanding voting stock, unless the merger or combination, or the acquisition that causes such person to beneficially own more than 10% of the outstanding stock, is approved in a prescribed manner.
The articles of incorporation of the combined company will generally provide that the Eighth Judicial District Court of Clark County, Nevada is the exclusive forum for substantially all disputes between the combined company and its stockholders, which could limit its stockholders’ ability to obtain a favorable judicial forum for disputes with the combined company or its directors, officers or other employees.
The articles of incorporation of the combined company will provide that, to the fullest extent permitted by law, and unless the company consents in writing to the selection of an alternative forum, the Eighth Judicial District Court of Clark County, Nevada is the sole and exclusive forum for the following types of proceedings: (a) any derivative action or proceeding brought in the name or right of the combined company or on its behalf, (b) any action asserting a claim for breach of any fiduciary duty owed by any of the combined company’s directors, officers, employees or agents to the company or its stockholders, (c) any action arising or asserting a claim arising pursuant to any provision of Chapters 78 or 92A of the NRS or any provision of the articles of incorporation or bylaws, (d) any action to interpret, apply, enforce or determine the validity of the articles of incorporation or bylaws or (e) any action asserting a claim governed by the internal affairs doctrine.
Choice-of-forum provisions of the type and scope included in the articles of incorporation of the combined company are expressly permitted by Section 78.046 of the NRS, but application of these choice-of-forum provisions may be limited in some instances by law. Section 27 of the Exchange Act establishes exclusive federal jurisdiction over all suits brought to enforce any duty or liability created by the Exchange Act or the rules and regulations thereunder and therefore the choice-of-forum provision would not apply to actions arising under, or brought to enforce a duty or liability created by, the Exchange Act or any other claim for which the U.S. federal courts have exclusive jurisdiction. However, Section 22 of the Securities Act provides that federal and state courts have concurrent jurisdiction over all suits brought to enforce any duty or liability created by the Securities Act or the rules and regulations thereunder, and the choice-of-forum provision would apply to actions arising under, or brought to enforce a duty or liability created by, the Securities Act. To the extent the text of the choice-of-forum provision in the combined company’s articles of incorporation purports to restrict the courts in which claims arising under the Securities Act may be brought, there remains some uncertainty as to whether a court would enforce such a provision. We note that the choice-of-forum provision will not relieve the combined company of its duties to comply with the federal securities laws and the rules and regulations thereunder, and the combined company’s stockholders will not be deemed to have waived compliance with these laws, rules and regulations.
This exclusive forum provision may make it more expensive for stockholders to bring a claim than if the stockholders were permitted to select another jurisdiction and may limit a stockholder’s ability to bring a claim in a judicial forum that it finds favorable for disputes with the combined company or its directors, officers or other employees or stockholders, which may discourage such lawsuits against the combined company and its directors, officers and other employees and stockholders. Alternatively, if a court were to find the choice of forum provision contained in the combined company’s articles of incorporation to be inapplicable or unenforceable in an action, the combined company may incur additional costs associated with resolving such action in other jurisdictions, which could materially and adversely affect its business, financial condition and results of operations.
The combined company’s ability to utilize its NOL carryforwards and tax credit carryforwards may be subject to limitations.
The combined company’s ability to use its federal and state NOLs to offset potential future taxable income and related income taxes that would otherwise be due is dependent upon the combined company’s generation of future taxable income, and Pieris and Palvella cannot predict with certainty when, or whether, the combined company will generate sufficient taxable income to use all of its available NOLs.
Under Sections 382 and 383 of the Code and corresponding provisions of state law, if a corporation undergoes an “ownership change,” its ability to use its pre-change NOL carryforwards and other pre-change tax attributes (such as research and development tax credits) to offset its post-change income may be limited. A Section 382 “ownership change” is generally defined as a greater than 50 percentage point change (by value) in its equity ownership by certain 5% stockholders over a three-year period. Pieris may have experienced such ownership changes in the past, including as a result of its public offering of shares of common stock in July 2021, and the Merger, if completed, will result in an ownership change. Pieris may experience additional ownership changes in the future due to subsequent shifts in its stock ownership (some of which are outside of its control). Palvella may have experienced ownership changes in the past, may experience an ownership change as a result of the Merger and the PIPE Financing, and may experience ownership changes in the future due to subsequent shifts in the combined company’s stock ownership (some of which are outside of its control). Consequently, even if the combined company achieves profitability, it may not be able to utilize a material portion of Palvella’s, Pieris’ or the combined company’s NOL carryforwards and other tax attributes, which could have a material adverse effect on cash flow and results of operations. Similar provisions of state tax law may also apply to limit the combined company’s use of accumulated state tax attributes. There is also a risk that due to regulatory changes, such as suspensions on the use of NOLs, or other unforeseen reasons, the combined company’s existing NOLs could expire or otherwise be unavailable to offset future income tax liabilities. In addition, if Pieris is not deemed to continue its historic business for two years after an ownership change, the NOL carryforwards and other tax attributes may be reduced to zero ($0).
Pieris and Palvella do not anticipate that the combined company will pay any cash dividends in the foreseeable future.
The current expectation is that the combined company will retain its future earnings, if any, to fund the growth of the combined company’s business as opposed to paying dividends. As a result, capital appreciation, if any, of the common stock of the combined company will be your sole source of gain, if any, for the foreseeable future.
An active trading market for the combined company common stock may not develop and its stockholders may not be able to resell their shares of common stock for a profit, if at all.
Prior to the Merger, there had been no public market for shares of Palvella common stock. An active trading market for the combined company’s shares of common stock may never develop or be sustained. If an active market for the combined company common stock does not develop or is not sustained, it may be difficult for its stockholders to sell their shares at an attractive price or at all.
Future sales of shares by existing stockholders could cause the combined company’s stock price to decline.
If existing securityholders of Pieris and Palvella sell, or indicate an intention to sell, substantial amounts of the combined company common stock in the public market after legal restrictions on resale discussed in this proxy statement/prospectus lapse, the trading price of the common stock of the combined company could decline. Based on shares outstanding as of October 28, 2024 and, the shares expected to be issued upon completion of the Merger, the combined company is expected to have outstanding a total of approximately 7,977,703 shares of common stock immediately following the completion of the Merger, prior to giving effect to the PIPE Financing. Of the shares of common stock, approximately 1,975,865 shares will become available for sale in the public market beginning 180 days after the closing of the Merger as a result of the expiration of lock-up agreements between Pieris and Palvella on the one hand and certain securityholders of Pieris and Palvella on the other hand. All other outstanding shares of common stock, other than shares held by affiliates of the combined company, will be freely tradable, without restriction, in the public market. In addition, shares of common stock that are subject to outstanding options of Palvella will become eligible for sale in the public market to the extent permitted by the provisions of various vesting agreements and Rules 144 and 701 under the Securities Act. If these shares are sold, the trading price of the combined company common stock could decline.
After completion of the Merger, the combined company’s executive officers, directors and principal stockholders will have the ability to control or significantly influence all matters submitted to the combined company’s stockholders for approval.
Upon the completion of the Merger, and without giving effect to the PIPE Financing, it is anticipated that the combined company’s executive officers, directors and principal stockholders will, in the aggregate, beneficially own approximately 56.5% of the combined company’s outstanding shares of common stock, subject to certain assumptions, including, but not limited to, (a) Pieris’ net cash being approximately $11 million, (b) a valuation for Pieris equal to $21 million, provided, that (i) if Pieris’ net cash as of the closing is greater than $11 million, then Pieris’ valuation will be adjusted upwards on a dollar-for-dollar basis by the difference of: (A) Pieris’ net cash, minus (B) $11 million, and (ii) if Pieris’ net cash is less than $11 million, then Pieris’ valuation will be adjusted downwards on a dollar-for-dollar basis by the difference of: (x) $11 million, minus (y) Pieris’ net cash, and (c) a valuation for Palvella equal to $95 million, in each case as further described in the Merger Agreement. As a result, if these stockholders were to choose to act together, they would be able to control or significantly influence all matters submitted to the combined company’s stockholders for approval, as well as the combined company’s management and affairs. For example, these persons, if they choose to act together, would control or significantly influence the election of directors and approval of any merger, consolidation or sale of all or substantially all of the combined company’s assets. This concentration of voting power could delay or prevent an acquisition of the combined company on terms that other stockholders may desire.
The combined company may be exposed to increased litigation, including stockholder litigation, which could have an adverse effect on the combined company’s business and operations.
The combined company may be exposed to increased litigation from stockholders, suppliers and other third parties due to the combination of Pieris’ business and Palvella’s business following the Merger. Such litigation may have an adverse impact on the combined company’s business and results of operations or may cause disruptions to the combined company’s operations. In addition, in the past, stockholders have initiated class action lawsuits against biotechnology companies following periods of volatility in the market prices of these companies’ stock. Such litigation, if instituted against the combined company, could cause the combined company to incur substantial costs and divert management’s attention and resources, which could have a material adverse effect on the combined company’s business, financial condition and results of operations.
If equity research analysts do not publish research or reports, or publish unfavorable research or reports, about the combined company, its business or its market, its stock price and trading volume could decline.
The trading market for the combined company common stock will be influenced by the research and reports that equity research analysts publish about it and its business. Equity research analysts may elect not to provide research coverage of the combined company common stock after the completion of the Merger, and such lack of research coverage may adversely affect the market price of its common stock. In the event it does have equity research analyst coverage, the combined company will not have any control over the analysts or the content and opinions included in their reports. The price of the combined company common stock could decline if one or more equity research analysts downgrade its stock or issue other unfavorable commentary or research. If one or more equity research analysts ceases coverage of the combined company or fails to publish reports on it regularly, demand for its common stock could decrease, which in turn could cause its stock price or trading volume to decline.
The combined company’s internal control over financial reporting may not meet the standards required by Section 404, and failure to achieve and maintain effective internal control over financial reporting in accordance with Section 404, could have a material adverse effect on the combined company’s business and share price.
As a privately held company, Palvella was not required to evaluate its internal control over financial reporting in a manner that meets the standards of publicly traded companies required by Section 404. Following the Merger, the combined company’s management will be required to report on the effectiveness of the combined company’s internal control over financial reporting. The rules governing the standards that must be met for the combined company’s management to assess the combined company’s internal control over financial reporting are complex and require significant documentation, testing and possible remediation.
Any failure to maintain effective internal control over financial reporting could severely inhibit the combined company’s ability to accurately report its financial condition, results of operations or cash flows. If the combined company is unable to conclude that its internal control over financial reporting is effective, or if the combined company’s independent registered public accounting firm determines the combined company has a material weakness or significant deficiency in the combined company’s internal control over financial reporting once that firm begins its reporting on internal control over financial reporting, investors may lose confidence in the accuracy and completeness of the combined company’s financial reports, the market price of the combined company common stock could decline, and the combined company could be subject to sanctions or investigations by Nasdaq, the SEC or other regulatory authorities. Failure to remedy any material weakness in the combined company’s internal control over financial reporting, or to implement or maintain other effective control systems required of public companies, could also restrict the combined company’s future access to the capital markets.
The combined company will have broad discretion in the use of proceeds from the PIPE Financing (if completed) and may invest or spend the proceeds in ways with which you do not agree and in ways that may not increase the value of your investment.
The combined company will have broad discretion over the use of proceeds from the PIPE Financing (if completed). You may not agree with the combined company’s decisions, and its use of the proceeds may not yield any return on the investment. The combined company’s failure to apply the net proceeds of the PIPE Financing (if completed) effectively could compromise its ability to pursue its growth strategy and the combined company might not be able to yield a significant return, if any, on its investment of these net proceeds. You will not have the opportunity to influence the combined company’s decisions on how to use the net proceeds from the PIPE Financing (if completed).
The PIPE Financing may not be completed.
In connection with and immediately following the closing of the Merger, Pieris intends to complete the PIPE Financing with the PIPE Investors involving the sale of approximately 3,154,241 of shares of Pieris common stock, at a price per share equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, and/or in lieu of Pieris common stock to certain purchasers who so choose, Pre-Funded Warrants to purchase up to 2,592,585 shares of the combined company common stock, at a purchase price per Pre-Funded Warrant equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, minus $0.001, for gross proceeds of approximately $78.9 million, consisting of approximately $60.0 million in cash and the conversion of approximately $18.9 million of principal and interest payable under the outstanding convertible notes issued by Palvella, before paying estimated expenses. Pieris has entered into the Purchase Agreement with the PIPE Investors pursuant to which these shares and Pre-Funded Warrants will be issued immediately following the closing of the Merger. The Purchase Agreement may be terminated under certain circumstances, and, if such termination were to occur, the combined company would not receive the proceeds of the PIPE Financing. The Purchase Agreement may be terminated as follows:
|
● |
automatically if the closing of the Merger is not consummated on or prior to 5:00 p.m. New York City time on April 30, 2025; |
|
● |
automatically if the Merger Agreement is terminated without the Merger being consummated; and |
|
● |
if any closing conditions set forth thereunder are not met and have not been waived; |
Accordingly, there can be no guarantee that the PIPE Financing will occur.
The securities issued in the PIPE Financing will be issued pursuant to an exemption from the registration requirements of the Securities Act, and the resale of the combined company common stock issued in the PIPE Financing and the shares of the combined company’s common stock issuable upon exercise of the Pre-Funded Warrants will be registered pursuant to a resale registration statement. These shares will be issued at a price per share equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, and the Pre-Funded Warrants will be issued at a purchase price per Pre-Funded Warrant equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, minus $0.001. Immediately following the PIPE Financing, the Pieris securityholders as of immediately prior to the Merger are expected to hold approximately 10.47% of the shares of combined company common stock, the Palvella securityholders as of immediately prior to the Merger are expected to hold approximately 50.18% of the shares of combined company common stock, and the investors in the PIPE financing are expected to hold approximately 39.35% of the shares of combined company common stock, in each case, on a fully diluted basis, subject to certain assumptions. For more information, please see the section titled “Agreements Related to the Merger” beginning on page 179 of this proxy statement/prospectus.
CAUTIONARY STATEMENT CONCERNING FORWARD-LOOKING STATEMENTS AND MARKET AND INDUSTRY DATA
This proxy statement/prospectus/information statement and the documents incorporated by reference into this proxy statement/prospectus/information statement contain forward-looking statements related to Pieris, Palvella, the Merger and the other proposed transactions contemplated thereby that involve substantial risks and uncertainties. These forward-looking statements are based on current expectations and beliefs and involve numerous risks and uncertainties that could cause actual results to differ materially from expectations. These forward-looking statements should not be relied upon as predictions of future events, as neither Pieris nor Palvella can assure you that the events or circumstances reflected in these statements will be achieved or will occur. You can identify forward-looking statements by the use of forward-looking terminology, including “anticipates,” “target,” “believes,” “continue,” “could,” “design,” “estimates,” “expects,” “intends,” “may,” “plans,” “potentially,” “predict,” “pro forma” “seeks,” “should,” “will,” “project,” “contemplate” or the negative of these words and phrases or other variations of these words and phrases or comparable terminology.
Forward-looking statements contained in this proxy statement/prospectus include, but are not limited to, statements about:
|
● |
the strategies, prospects, plans, expectations and objectives of management of Pieris or Palvella for future operations of the combined company following the closing of the Merger; |
|
● |
the approval and closing of the Merger, including the timing of the consummation of the Merger; |
|
● |
the ability of Pieris to obtain a sufficient number of proxies and stockholder votes to approve the transaction proposal contemplated by the Merger Agreement and submitted to the Pieris stockholders at the Pieris special meeting; |
|
● |
the likelihood of the satisfaction of other conditions to the closing of the Merger and whether and when the Merger will be consummated; |
|
● |
the Exchange Ratio, and relative ownership levels as of the Effective Time; |
|
● |
the expected benefits of and potential value created by the Merger for the stockholders of Pieris and Palvella; |
|
● |
Palvella’s ability to control and correctly estimate its operating expenses and its expenses associated with the Merger; |
|
● |
Pieris’ expectations regarding its ability to fund its operating expenses and capital expenditure requirements with its cash, cash equivalents and investments; |
|
● |
the cash balances of the combined company following the Effective Time; |
|
● |
the ability to consummate the PIPE Financing and the expected benefits of the PIPE Financing, including the cash runway it is expected to provide the combined company; |
|
● |
the ability of Pieris and the combined company to maintain compliance with Nasdaq listing standards; |
|
● |
the ability of Pieris stockholders to realize any benefits from their CVR Agreements; |
|
● |
the plans, strategies and objectives of management for future operations, including the execution of integration plans and the anticipated timing of filings; |
|
● |
Pieris’ expectations regarding the reconsideration of its strategic alternatives in the event the Merger is not completed; |
|
● |
the need to hire additional personnel and the combined company’s ability to attract and retain such personnel; |
|
● |
the ability to protect and enhance the combined company’s products and intellectual property, including the extensions of existing patent terms where available, the validity of intellectual property rights held by third parties, and the combined company’s ability not to infringe, misappropriate or otherwise violate any third-party intellectual property rights; |
|
● |
any statements concerning developments and projections relating to the combined company’s competitors or industry; |
|
● |
Pieris’, Palvella’s, or the combined company’s estimates regarding expenses, future revenue, capital requirements and needs for additional financing; |
|
● |
any statements concerning Pieris’, Palvella’s, or the combined company’s financial performance; |
|
● |
any statements regarding expectations concerning Pieris’ or Palvella’s relationships and actions with third parties, including any license and collaborations with such third parties; |
|
● |
future regulatory, judicial and legislative changes in Pieris’ or Palvella’s industry in the United States, Europe, and other jurisdictions; |
|
● |
the ability of the combined company’s clinical trials to demonstrate safety and efficacy of the combined company’s product candidates, and other positive results; |
|
● |
the combined company’s ability to utilize its proprietary drug discovery platform to develop a pipeline of product candidates to address unmet needs in rare skin disease indications; |
|
● |
the outcome of clinical trials of the combined company’s product candidates, including the ability of those trials to satisfy relevant governmental or regulatory requirements; |
|
● |
the timing of availability of data from the combined company’s clinical trials; |
|
● |
the combined company’s plans to research, develop and commercialize its current and future product candidates; |
|
● |
the combined company’s ability to protect its intellectual property and proprietary technologies; |
|
● |
the combined company’s reliance on third parties, contract manufacturers, and contract research organizations; |
|
● |
the combined company’s ability to develop and advance current product candidates and programs into, and successfully complete, clinical studies; |
|
● |
the combined company’s manufacturing, commercialization, and marketing capabilities and strategy; |
|
● |
the size of the market opportunity for the combined company’s product candidates, including estimates of the number of patients who suffer from the diseases the combined company is targeting; |
|
● |
expectations regarding potential for accelerated approval or other expedited regulatory designation; |
|
● |
the combined company’s competitive position and the success of competing therapies that are or may become available; |
|
● |
estimates of the number of patients that the combined company will enroll in its clinical trials; |
|
● |
the beneficial characteristics, and the potential safety, efficacy and therapeutic effects of the combined company’s product candidates; and |
|
● |
the combined company’s ability to obtain and maintain regulatory approval of its product candidates and its expectations regarding particular lines of therapy. |
These forward-looking statements are based largely on the current expectations and projections of about Pieris’, Palvella’s, or the combined company’s business, the industry in which Pieris and Palvella operate and financial trends that Pieris and Palvella believe may affect the business, financial condition, results of operations and prospects of Pieris, Palvella, or the combined company, and these forward-looking statements are not guarantees of future performance or development. These forward-looking statements speak only as of the date of this proxy statement/prospectus and are subject to a number of risks, uncertainties and assumptions described in the section titled “Risk Factors” beginning on page 25 of this proxy statement/prospectus. Additional factors that could cause actual results to differ materially from those expressed in the forward-looking statements are discussed in reports filed with the SEC by Pieris. Please see the section titled “Where You Can Find More Information” beginning on page 336 of this proxy statement/prospectus. Because forward-looking statements are inherently subject to risks and uncertainties, some of which cannot be predicted or quantified, you should not rely on these forward-looking statements as predictions of future events. The events and circumstances reflected in these forward-looking statements may not be achieved or occur and actual results could differ materially from those projected in the forward-looking statements. Except as required by applicable law, Pieris and Palvella do not plan to publicly update or revise any forward-looking statements contained herein until after the distribution this proxy statement/prospectus, whether as a result of any new information, future events or otherwise.
In addition, statements that “Pieris and/or Palvella believe(s)” and similar statements reflect the beliefs and opinions on the relevant subject of Pieris and Palvella. These statements are based upon information available to Pieris and Palvella as of the date of this proxy statement/prospectus, and while Pieris and Palvella believe such information forms a reasonable basis for such statements, such information may be limited or incomplete, and the statements of Pieris and/or Palvella should not be read to indicate that Pieris or Palvella have conducted an exhaustive inquiry into, or review of, all potentially available relevant information. These statements are inherently uncertain and you are cautioned not to unduly rely upon these statements.
You should read this proxy statement/prospectus and the documents that are referenced in this proxy statement/prospectus and have been filed with the SEC as exhibits to the Registration Statement on Form S-4 of which this proxy statement/prospectus is a part with the understanding that the actual future results, levels of activity, performance and events and circumstances of Pieris, Palvella, or the combined company may be materially different from what Pieris or Palvella expect.
In addition, this proxy statement/prospectus includes statistical and other industry and market data that was obtained from independent industry publications and research, surveys and studies conducted by independent third parties as well as Pieris’ and Palvella’s estimates. The market data used in this proxy statement/prospectus involves a number of assumptions and limitations, and you are cautioned not to give undue weight to such data. Industry publications and third-party research, surveys and studies generally indicate that their information has been obtained from sources believed to be reliable, although they do not guarantee the accuracy or completeness of such information. Pieris’ and Palvella’s estimates include assumptions based on their respective industry knowledge, industry publications, third-party research and other surveys. While Pieris and Palvella believe that their respective internal assumptions and estimates are reasonable, no independent source has verified such assumptions or estimates.
THE SPECIAL MEETING OF PIERIS STOCKHOLDERS
The Pieris special meeting will be held on Wednesday, December 11, 2024, commencing at 9:00 a.m. Eastern Time, unless postponed or adjourned to a later date. The Pieris special meeting will be held entirely online. Pieris is sending this proxy statement/prospectus to its stockholders in connection with the solicitation of proxies by the Pieris board of directors for use at the Pieris special meeting and any adjournments or postponements of the Pieris special meeting. This proxy statement/prospectus is first being mailed to Pieris stockholders on or about .
Purposes of the Pieris Special Meeting
The purposes of the Pieris special meeting are:
1. To approve the issuance of shares of common stock of Pieris pursuant to the terms of the Merger Agreement and the Purchase Agreement (as each may be amended from time to time), a copy of which is attached as Annex A and Annex B to the accompanying proxy statement/prospectus, and the change of control of Pieris pursuant to the Merger, for purposes of Nasdaq Listing Rules 5635(a), (b) and (d);
2. To approve an amendment to the amended and restated articles of incorporation of Pieris to increase the number of authorized shares of Pieris common stock from 3,750,000 shares to 200,000,000 shares;
3. To approve the adoption of the 2024 Equity Incentive Plan;
4. To ratify the Pieris board of directors’ approval of an amendment to the amended and restated articles of incorporation of Pieris to change the name of the corporation from “Pieris Pharmaceuticals, Inc.” to “Palvella Therapeutics, Inc.”; and
5. To vote upon an adjournment of the Pieris special meeting, if necessary, to solicit additional proxies if there are not sufficient votes in favor of Proposal Nos. 1 and 2.
Approval of each of Proposal Nos. 1 and 2 is a condition to the completion of the Merger. Therefore, the Merger cannot be consummated without the approval of Proposal Nos. 1 and 2.
Recommendation of the Pieris Board of Directors
|
● |
the Pieris board of directors has determined and believes that it is fair to, in the best interests of, and advisable to, Pieris and its stockholders to approve the issuance of shares of common stock of Pieris pursuant to the terms of the Merger Agreement and the Purchase Agreement (as each may be amended from time to time), for purposes of Nasdaq Listing Rules 5635(a), (b) and (d) as described in this proxy statement/prospectus. The Pieris board of directors recommends that Pieris stockholders vote “FOR” Proposal No. 1; |
|
● |
the Pieris board of directors has determined and believes that it is fair to, in the best interests of, and advisable to, Pieris and its stockholders to approve an amendment to the amended and restated articles of incorporation of Pieris to increase the number of authorized shares of Pieris common stock from 3,750,000 shares to 200,000,000 shares. The Pieris board of directors recommends that Pieris stockholders vote “FOR” Proposal No. 2; |
|
● |
the Pieris board of directors has determined and believes that it is fair to, in the best interests of, and advisable to, Pieris and its stockholders to approve the adoption of the 2024 Equity Incentive Plan, as described in this proxy statement/prospectus. The Pieris board of directors recommends that Pieris stockholders vote “FOR” Proposal No. 3; |
|
● |
the Pieris board of directors has determined and believes that it is fair to, in the best interests of, and advisable to, Pieris and its stockholders to ratify the Pieris board of directors’ approval of an amendment to the amended and restated articles of incorporation of Pieris to change the name of the corporation from “Pieris Pharmaceuticals, Inc.” to “Palvella Therapeutics, Inc.” The Pieris board of directors recommends that Pieris stockholders vote “FOR” Proposal No. 4; and |
|
● |
the Pieris board of directors has determined and believes that it is fair to, in the best interests of, and advisable to, Pieris and its stockholders to approve the adjournment of the Pieris special meeting, if necessary, to solicit additional proxies if there are not sufficient votes in favor of Proposal Nos. 1 and 2, as described in this proxy statement/prospectus. The Pieris board of directors recommends that Pieris stockholders vote “FOR” Proposal No. 5. |
Only holders of record of Pieris common stock and Series F Preferred Stock at the close of business on the record date, which is October 28, 2024, are entitled to notice of, and to vote at, the Pieris special meeting. At the close of business on the record date, there were 1,320,240 shares of Pieris common stock issued and outstanding and one share of Series F Preferred Stock issued and outstanding. Each share of Pieris common stock entitles the holder thereof to one vote on each matter submitted for stockholder approval. Each share of Series F Preferred Stock entitles the holder thereof to 25,000,000 votes on Proposal No. 2, which are cast in the same proportion as shares of Pieris common stock are voted “for” and “against” such proposal, excluding any shares of common stock that are not voted “for” and “against” the proposal for any reason, including, without limitation, any abstentions or broker non-votes.
Voting and Revocation of Proxies
The proxy accompanying this proxy statement/prospectus is solicited on behalf of the Pieris board of directors for use at the Pieris special meeting.
If, as of the record date referred to above, your shares were registered directly in your name with the transfer agent for Pieris common stock, Computershare Trust Company, N.A., then you are a stockholder of record. Whether or not you plan to attend the Pieris special meeting online, Pieris urges you to fill out and return the proxy card or submit a proxy to vote over the telephone or on the internet as instructed below to ensure your vote is counted.
If you are a stockholder of record, the procedures for voting are as follows:
|
● |
you may vote at the Pieris special meeting by attending the Pieris special meeting online and following the instructions posted at www.virtualshareholdermeeting.com/PIRS2024SM. Even if you have submitted a proxy before the Pieris special meeting, you may still attend the Pieris special meeting and vote in person. In such case, your previously submitted proxy will be disregarded; |
|
● |
to submit your proxy using the proxy card, simply complete, sign and date the accompanying proxy card and return it promptly in the envelope provided. If you return your signed proxy card before the Pieris special meeting, Pieris will vote your shares in accordance with the proxy card; |
|
● |
to submit your proxy over the internet, follow the instructions provided on the Notice of Internet Availability or your proxy card; or |
|
● |
to submit your proxy by telephone, you may call the toll-free number found on the Notice of Internet Availability or your proxy card. |
If you are a beneficial owner of shares registered in the name of your broker, bank or other agent, you should have received a voting instruction card and voting instructions with these proxy materials from that organization rather than from Pieris. Simply complete and mail the voting instruction card to ensure that your vote is counted. To vote in person at the Pieris special meeting, you must obtain a valid proxy from your broker, bank or other agent. Follow the instructions from your broker, bank or other agent included with these proxy materials, or contact your broker, bank or other agent to request a proxy form.
Pieris provides internet proxy voting to allow you to submit your proxy to vote your shares online, with procedures designed to ensure the authenticity and correctness of your proxy vote instructions. However, please be aware that you must bear any costs associated with your internet access, such as usage charges from internet access providers and telephone companies.
If you are a beneficial owner of shares registered in the name of your broker, bank or other agent, and do not give instructions to your broker, bank or other agent, such broker, bank or agent can vote your Pieris shares with respect to “discretionary,” routine items but not with respect to “non-discretionary,” non-routine items. Discretionary items are proposals considered routine under Rule 452 of the New York Stock Exchange on which your broker may vote shares held in “street name” in the absence of your voting instructions. On non-routine items for which you do not give your broker instructions, your Pieris shares will be treated as broker non-votes. It is anticipated that Proposal Nos. 1, 2 and 3 will be non-routine. It is anticipated that Proposal Nos. 4 and 5 will be routine.
All properly executed proxies that are not revoked will be voted at the Pieris special meeting and at any adjournments or postponements of the Pieris special meeting in accordance with the instructions contained in the proxy. If a holder of Pieris common stock executes and returns a proxy and does not specify otherwise, the shares represented by that proxy will be voted “FOR” all of the Proposals in accordance with the recommendation of the Pieris board of directors.
If you are a stockholder of record of Pieris and you have not executed a support agreement, you may change your vote at any time before your proxy is voted at the Pieris special meeting in any one of the following ways:
|
● |
you may submit another properly completed proxy with a later date by mail or via the internet; |
|
● |
you can provide your proxy instructions via telephone at a later date; |
|
● |
you may send a written notice that you are revoking your proxy to Pieris’ Corporate Secretary at 225 Franklin Street, 26th Floor, Boston, Massachusetts 02110, Attn: Corporate Secretary; or |
|
● |
you may attend the Pieris special meeting online and vote by following the instructions at www.virtualshareholdermeeting.com/PIRS2024SM. Simply attending the Pieris special meeting will not, by itself, revoke your proxy. |
If your shares are held by your broker, bank or other agent, you should follow the instructions provided by them.
The presence at the Pieris special meeting, in person or represented by proxy, of the holders of a majority in voting power of the shares of Pieris capital stock issued and outstanding and entitled to vote at the Pieris special meeting, shall constitute a quorum for the transaction of business. Abstentions and broker non-votes, if any, will be counted towards a quorum.
Votes will be counted by the inspector of election appointed for the meeting, who will separately count “FOR” and “AGAINST” votes, abstentions and broker non-votes. Assuming a quorum is present, the affirmative vote of the holders of a majority of the votes cast on the proposal by the holders of shares of Pieris common stock present or represented at the meeting is required for approval of Proposal Nos. 1, 3, 4 and 5. Abstentions and broker non-votes, if any, will have no effect on the outcome of Proposal Nos. 1, 3, 4 and 5. The affirmative vote of the holders of a majority in voting power of the outstanding shares of Pieris common stock and preferred stock, voting as a single class, entitled to vote thereon is required for approval of Proposal No. 2. Abstentions and broker non-votes, if any, will have the same effect as “AGAINST” votes on Proposal No. 2, but will not be considered as votes cast “AGAINST” for the purposes of determining the proportion of Series F Preferred stock votes that will be cast “FOR” and “AGAINST”.
Proposal Nos. 1 and 2 are conditions to the completion of the Merger. Therefore, the Merger cannot be consummated without the approval of Proposal Nos. 1 and 2. The issuance of Pieris common stock in connection with the Merger will not take place unless Proposal Nos. 1 and 2 are approved by Pieris stockholders and the Merger is consummated.
As of October 28, 2024, the directors and certain executive officers of Pieris owned or controlled approximately 0.07% of the outstanding shares of Pieris common stock entitled to vote at the Pieris special meeting. As of October 28, 2024, the Pieris stockholders that are party to support agreements, including the directors and certain executive officers of Pieris, owned an aggregate of 258,496 shares of Pieris common stock representing approximately 19.6% of the outstanding shares of Pieris common stock. Pursuant to the support agreements, these stockholders, including the directors and certain executive officers of Pieris, have agreed to vote all shares of Pieris common stock owned by them as of the record date in favor of Proposal Nos. 1, 2, 3 and 4 and against any competing Acquisition Proposal.
In addition to solicitation by mail, the directors, officers, employees and agents of Pieris may solicit proxies from Pieris stockholders by personal interview, telephone, email, fax or otherwise. Pieris and Palvella will share equally the costs of printing and filing this proxy statement/prospectus and proxy card. Arrangements will also be made with brokerage firms and other custodians, nominees and fiduciaries who are record holders of Pieris common stock for the forwarding of solicitation materials to the beneficial owners of Pieris common stock. Pieris will reimburse these brokers, custodians, nominees and fiduciaries for the reasonable out of pocket expenses they incur in connection with the forwarding of solicitation materials. Pieris will retain Alliance Advisors to assist it in soliciting proxies using the means referred to above. Pieris will pay the fees of Alliance Advisors, which Pieris expects to be approximately $50,000, plus reimbursement of out-of-pocket expenses.
As of the date of this proxy statement/prospectus, the Pieris board of directors does not know of any business to be presented at the Pieris special meeting other than as set forth in the notice accompanying this proxy statement/prospectus. If any other matters should properly come before the Pieris special meeting, it is intended that the shares represented by proxies will be voted with respect to such matters in accordance with the judgment of the persons voting the proxies.
This section and the section titled “The Merger Agreement” beginning on page 163 of this proxy statement/prospectus describe the material aspects of the Merger and the Merger Agreement. While Pieris and Palvella believe that this description covers the material terms of the Merger and the Merger Agreement, it may not contain all of the information that is important to you. You should read carefully this entire proxy statement/prospectus for a more complete understanding of the Merger and the Merger Agreement and the other documents to which you are referred in this proxy statement/prospectus. See the section titled “Where You Can Find More Information” beginning on page 336 of this proxy statement/prospectus.
The following chronology summarizes the key meetings and events that led to the signing of the Merger Agreement. The following chronology does not purport to catalogue every conversation among the Pieris board of directors or committees thereof or the representatives (including management) of Pieris and other parties.
The Pieris board of directors and Pieris’ management team have regularly reviewed and discussed Pieris’ business, operating and strategic plans, both near-term and long-term, in addition to various potential partnerships and collaborations, financing transactions and other types of strategic transactions and opportunities to enhance stockholder value. As part of these efforts, the Pieris board of directors and management team have consulted outside advisors and experts, and have focused on, among other things, its strategic goals and plans, including associated opportunities and risks, its operating and financial performance and outlook, the general economy and financial markets, the overall industry condition including the therapeutic landscapes it operates in, and its strategic relationships and other options.
Pieris has historically been a clinical-stage biotechnology company that discovers and develops Anticalin® based drugs to target validated disease pathways in unique and transformative ways in both IO and respiratory disease. Proprietary to Pieris, Anticalin proteins are a novel class of therapeutics validated in the clinic and through partnerships with leading pharmaceutical companies.
Pieris’ efforts in respiratory disease included research and development collaborations with AstraZeneca, as well as fully proprietary development programs. PRS-060, or elarekibep, was a drug candidate in a Phase 2a study partnered with AstraZeneca for the treatment of asthma. Pieris’ efforts in IO included research and development collaborations with Servier, Pfizer (formerly Seagen), and Boston Pharmaceuticals, as well as fully proprietary development of its lead fully IO asset, cinrebafusp alfa, or PRS-343, which was in a Phase 2 study in 2L+ HER2-positive gastric cancer. Pieris ceased further enrollment in cinrebafusp alfa in August 2022 as part of a strategic pipeline prioritization to focus its resources on research and development targeting respiratory diseases.
At the end of 2022 and into 2023, Pieris began exploring various opportunities to continue development of cinrebafusp alfa, including partnering opportunities for cinrebafusp alfa with other pharmaceutical companies. In February 2023, a representative of Pieris reached out separately to Company A and Company B to discuss potentially partnering cinrebafusp alfa. Pieris entered into separate mutual CDAs with both parties which did not contain standstill provisions.
Between February 2023 and July 2023, Company B conducted confidential technical and scientific diligence and both parties negotiated terms to partner cinrebafusp alfa. In April 2023, Company B proposed initial terms for a transaction, and Pieris sent a counteroffer to Company B in June 2023. However, in July 2023, Company B indicated that Pieris’ proposed deal terms along with the efforts required to restart the program led them to decide to stop negotiations.
Starting in March 2023, Company A began confidential technical, operational and scientific due diligence on cinrebafusp alfa.
On June 16, 2023, Company A sent Pieris’ management proposed terms for a potential cinrebafusp alfa licensing transaction.
On June 18, 2023, at a virtual meeting, the Pieris board of directors met with Pieris’ management and members of its respiratory clinical team to discuss certain non-clinical safety findings in non-human primates related to elarekibep that had been recently provided by AstraZeneca. During this meeting, the Pieris board of directors and Pieris’ management and members of its respiratory team discussed the potential implications of the results and the timing for AstraZeneca to decide whether it would continue the ongoing Phase 2a clinical study for elarekibep. They also discussed potential strategies for Pieris going forward in light of the risk that the clinical study could be discontinued and the potential read-through to development of Pieris’ other proprietary respiratory programs.
On June 20, 2023, Pieris received notice from AstraZeneca that it would discontinue and cease dosing of elarekibep in the ongoing clinical studies in moderate-to-severe asthma due to the non-clinical safety findings in non-human primates in the toxicology study. On that same day at a virtual meeting, the Pieris board of directors, Pieris’ management, and representatives from Mintz, Pieris’ outside counsel, discussed Pieris’ strategic options as well as potential reductions to Pieris’ discretionary spending in light of AstraZeneca’s decision. These strategic options included assessing independently advancing cinrebafusp alfa through the clinic as a proprietary asset, exploring a strategic merger or reverse merger transaction, monetizing future milestone and royalty income, and in-licensing one or more assets that could reach meaningful clinical milestones within Pieris’ cash runway, among other options. Given the potential for a broader impact of the negative elarekibep non-clinical safety findings on the residual respiratory franchise, including PRS-220 and PRS-400, Pieris’ two proprietary respiratory assets, there was an initial discussion about reassessing priorities across the IO and respiratory franchises, including continuing to pursue the potential partnering options for the proprietary respiratory assets.
During the June 20, 2023 Pieris board of directors meeting, Mr. Stephen S. Yoder, the Chief Executive Officer and President of Pieris, presented on the potential spend required for Pieris to independently advance cinrebafusp alfa and to explore reverse merger opportunities. The Pieris board of directors discussed engaging an investment banking firm as Pieris’ financial advisor to assist Pieris with its consideration and exploration of various strategic options. During this meeting, a Mintz representative provided the Pieris board of directors with an overview of the Pieris board of directors’ fiduciary duties and considerations as it relates to a strategic transaction review process.
On June 21, 2023, Pieris publicly announced in a press release and a current report on Form 8-K that AstraZeneca decided to discontinue enrollment and cease dosing of patients in the ongoing clinical study of elarekibep due to the results of the non-clinical toxicology study.
On June 22, 2023, Mr. James Geraghty, Mr. Christopher Kiritsy, and Dr. Peter Kiener, members of the Pieris board of directors, met with representatives of Stifel, an investment banking firm, to discuss Stifel’s potential engagement to act as Pieris’ financial advisor in a review by Pieris of strategic transactions.
Between June 23, 2023 and June 30, 2023, Pieris’ management met with representatives of two other potential financial advisors.
On June 28, 2023, Pieris sent a counter proposal to Company A for a potential cinrebafusp alfa licensing deal.
On June 30, 2023, the Board held a virtual meeting where Pieris’ management recommended a strategic plan to the Board. Mr. Yoder recommended that Pieris should prioritize partnering cinrebafusp alfa, in light of the recent developments and potential risks with Pieris’ respiratory franchise, while exploring a reverse merger in parallel. Pieris’ management also presented other options the Company was considering including monetizing the operations of its subsidiary, Pieris Pharmaceuticals GmbH, or Pieris GmbH, and would continue seeking partnership opportunities for Pieris’ proprietary respiratory assets. Representatives of Stifel were also present at the meeting and provided an overview of the biotech reverse merger process and landscape, and discussed the potential engagement of Stifel as Pieris’ financial advisor.
On July 7, 2023, at a virtual meeting, the Pieris board of directors and Pieris’ management, with representatives of Mintz also in attendance, discussed various strategies for Pieris, including potential reverse mergers and a sale or other monetization strategy of Pieris’ German research and development capabilities. Pieris’ management also explained that discussions were ongoing to partner cinrebafusp alfa with Company A and Company B and summarized the proposals and counterproposals made by each party. Mr. Yoder presented the potential interest expressed by another publicly traded company, Company C, via a Pieris investor, to potentially leverage Pieris’ scientific capabilities through a change in control transaction. Pieris had in place a CDA with Company C which did not include any standstill provisions. During this call, the Pieris board of directors reviewed the scope of services and terms of a potential engagement with Stifel as well as materials from another potential financial advisor. The Pieris board of directors selected Stifel as Pieris’ financial advisor, which was subsequently engaged (i) as exclusive financial advisor to advise and assist in exploring various transactions for Pieris, including a reverse merger, and (ii) on a non-exclusive basis, to advise and assist in exploring strategic transactions for Pieris GmbH, including a sale of the subsidiary. Pieris’ management also recommended that the Pieris board of directors constitute a transaction committee, or the “Transaction Committee”, to provide an efficient framework to review and advise on potential transactions and make recommendations to the Pieris board of directors.
On July 10, 2023, at Pieris’ direction, Stifel began a blinded broad outreach to publicize the Pieris reverse merger opportunity to investors and private biotech companies, which outreach included 192 private biotech companies and 334 investors.
On July 11, 2023, Pieris received a proposal from Company C for an acquisition of the Company that included CVRs to Pieris stockholders for the partnered milestones and royalties that Pieris would potentially receive from existing partners and a potential spinout of Pieris GmbH. Under this proposal, Company C would acquire Pieris’ cash at year end, and support in wind down activities.
On July 12, 2023, the Pieris board of directors virtually met with Pieris' management, with Stifel and Mintz representatives also in attendance. Mr. Yoder updated the Pieris board of directors that Company A was interested in a reverse merger in lieu of a license to develop to cinrebafusp alfa, and presented the proposal from Company C that was received on July 11, 2023. Stifel representatives provided an update on the potential sale of Pieris GmbH, including the outreach conducted to date and the leading five potential buyers.
In parallel, Pieris began evaluating five inflammation and immunology, or “I&I” assets, owned by another third party, Company D, which Pieris was familiar with from prior interactions with individuals at Company D, to determine if any could be added to Pieris’ pipeline to enhance stockholder value. The initial proposal was for Company D to co-fund development of these assets. Full scientific, clinical, technical and commercial diligence was conducted by the Pieris team on the five I&I assets. Pieris also conducted diligence to determine financing requirements and cost to reach meaningful inflection points as part of development of the I&I assets.
On July 18, 2023, Pieris filed a Current Report on Form 8-K disclosing its intention to explore strategic transactions, including focusing on the execution of new or expanded partnerships to advance its therapeutics programs, with the goal of enhancing stockholder value, and disclosing that it had engaged Stifel as Pieris’ financial advisor to support these efforts. Pieris also disclosed that to preserve cash while exploring such transactions, the Pieris board of directors approved a reduction in force of Pieris’ workforce by approximately 70%. Pieris also disclosed that on July 17, 2023, AstraZeneca notified Pieris that it was terminating (i) the License and Collaboration Agreement, dated as of May 2, 2017, and subsequently amended, by and between AstraZeneca and Pieris, and (ii) the Non-Exclusive Anticalin Platform Technology License Agreement, dated as of May 2, 2017, and subsequently amended, by and among AstraZeneca, Pieris, and Pieris Pharmaceuticals GmbH, effective as of October 15, 2023.
On July 20, 2023, the Pieris board of directors virtually met with Pieris’ management, Mintz representatives, and Stifel representatives and discussed the potential strategic options, including a reverse merger, independent financing and development of internal assets such as cinrebafusp alfa, outlicensing of cinrebafusp alfa to Company A, a milestone and royalty monetization strategy, in-licensing additional assets from Company D, liquidation, and a change-in-control transaction with Company C. The Pieris board of directors continued to endorse and encourage Pieris’ management to explore the options, with the assistance of Stifel, and to enhance stockholder value under any of these transactions, and formed a transaction committee consisting of Dr. Peter Kiener, Mr. James Geraghty, and Mr. Christopher Kiritsy to review the various strategic options and report to the Pieris board of directors.
On July 30, 2023, Pieris received an updated proposal from Company C that capped the total CVR payments Pieris stockholders could achieve, and required that any cash acquired by Company C would be used to offset Pieris’ liabilities such as Pieris GmbH’s lease liability.
On August 1, 2023, process letters were sent to 53 interested parties, including Company A. The letters indicated that interested parties should submit non-binding letters of interest, or “LOIs”, by August 18, 2023 that included the proposed structure of a transaction, proposed valuation of the counterparty and pro forma equity ownership split, interest in any of Pieris’ programs, employees, and/or facilities, proposed board seats, concurrent financing needs, a detailed list of material diligence items that they require for their diligence process, relevant information on their company, public company readiness metrics, internal review, approvals and required closing conditions, external advisors, and contact information.
On August 3, 2023 and August 4, 2023, Pieris’ management, Stifel representatives, and Company C met via teleconference to discuss Company C’s July 30, 2023 proposal.
On August 9, 2023, the Transaction Committee held a virtual meeting with Pieris’ management and representatives of Stifel and Mintz were also in attendance, to discuss the ongoing transaction workstreams. Mr. Yoder explained that Pieris’ management, with the assistance of Stifel, had explored the proposal from Company C and out-licensing cinrebafusp alfa to Company A to determine if these could be more favorable for Pieris and its stockholders than a traditional reverse merger. The potential transaction with Company D for the I&I assets was modified as Company D was acquired and went through a restructuring, which made their leadership unable to pursue this co-funding strategy. Instead, Pieris’ management indicated that Pieris could pursue acquiring a limited number of I&I assets from Company D at a lower cost and develop those assets, and that it would review this option.
During the meeting with the Transaction Committee, the Stifel representatives informed the Transaction Committee that, at Pieris’ management’s direction, they communicated to Company C that the current terms proposed by Company C would not be acceptable to Pieris, in part because the proposed transaction did not include any upfront payments and would offer a limited CVR to the potential milestones and royalties under Pieris’ partnership agreements, which was potentially less valuable than if Pieris retained all rights to these potential milestones and royalties. Pieris and Company C agreed to explore a milestone and royalty monetization option and Pieris expected an updated proposal by Company C. Mr. Thomas Bures, Pieris’ Chief Financial Officer, also provided an overview of a milestone and royalty monetization strategy, including a review of the economic rationale and risk adjusted net present value of the Company’s assets. The Pieris management team aligned with the Transaction Committee on a process for evaluating this scenario as an alternative to Company C’s proposal as well as other strategic alternatives.
By August 18, 2023, the reverse merger bid due date, 12 LOIs were received. The Transaction Committee virtually met on August 24, 2023 to discuss the 12 non-binding LOIs that were received, including three additional LOIs that had been received between August 19, 2023 and August 24, 2023, proposed next steps in the reverse merger evaluation process, and business development updates. Stifel representatives reviewed preliminary information to assist with evaluating the offer quality from the candidates, the quality of the candidate’s pipeline and investor base, if there were any closing concerns, and the cash-runway post-inflection points identified by the candidates. After considering the preliminary information reviewed, Pieris’ management proposed nine of the companies advance to the next round of diligence, which the Transaction Committee agreed to advance. Stifel also provided updates on the potential sale of Pieris GmbH and indicated that there were four parties that expressed interest with only two of those having met with either Pieris management or Stifel representatives. For those six reverse merger candidates that were not advanced into the second round, the Transaction Committee, after discussions with Pieris management and Stifel representatives, determined either the proposed valuation offers were not compelling, that the product candidate pipelines were highly risky or unproven, the intellectual property portfolio was not robust or that the therapeutic area and indications were not compelling, that candidates did not have a strong investor base, or there was insufficient cash runway post-inflection points to continue to fund the company even with a concurrent PIPE financing.
By September 5, 2023, Pieris received four additional LOIs, bringing the total number of LOIs to 19. These companies were evaluated using the same criteria as the prior 15. After reviewing these additional four LOIs and comparing them to the nine candidates suggested to move forward, it was decided to advance two of the new companies to the second round, and not advance one company from the original nine, resulting in ten potential reverse merger candidates. The other two new companies did not advance to the second round because they had proposed valuation offers that were not compelling, product candidate pipelines that were highly risky or unproven, an investor base that was not attractive, and insufficient cash runway post-inflection to continue to fund the company even with a concurrent PIPE financing. The Transaction Committee decided to remove one candidate from the original nine because its offer and the company profile including its assets and investor base, were not as strong as the other second round candidates, and the Transaction Committee wanted to focus Pieris’ resources on the other 10 second round candidates.
On September 6, 2023, Pieris finalized its opportunity assessment and cost analysis for the top three I&I assets from Company D. This analysis included an assessment of feasible and commercially attractive indications, an overview of the competition and efficacy and safety benchmarks, an analysis of how the target product profiles for these assets could compete against the emerging standards of care, and market size and commercial opportunity for each. There was also a clinical analysis to determine expected development and manufacturing costs and a proposal for how funds would be secured to bring each asset to a meaningful inflection point. Discussions with Company D management were still ongoing to determine how necessary funds could be raised due to their recent acquisition and restructuring.
By September 6, 2023, Pieris had entered into CDAs with the companies that advanced into the second round. These CDAs did not contain any standstill provisions. Also on this date, two of the ten potential reverse merger candidates withdrew from the second round of the reverse merger process. One candidate withdrew from the process because it entered into exclusivity with another third party, and the second candidate withdrew because it did not have the resources to continue to consider a reverse merger.
Between September 7, 2023 and September 22, 2023, Pieris’ management, Pieris’ strategy and commercial team, and the Transaction Committee participated in management presentation calls for the eight remaining second-round candidates, including Company A. During these meetings, the management teams of the second-round candidates presented their company's pipeline and programs, management team, investor base, and product development plans. The companies also presented their previously proposed offers, including rationale for the valuation of their company and Pieris, and the proposed pro forma ownership split. Pieris’ management and the Transaction Committee asked questions to better understand each candidates’ public company readiness, cash needs and quality of their investor base, confidence in their ability to raise the necessary funds via a financing, the timeline of valuation inflection points within the existing and proposed cash runways, the risks and opportunities within their pipelines and expected size and cost of their clinical development plans and manufacturing costs for lead asset(s), commercial opportunities and risks, and partnership strategies.
On September 19, 2023, the Pieris board of directors held a virtual meeting with Pieris’ management, with the Mintz and the Stifel representatives also in attendance. Mr. Yoder updated the Pieris board of directors on the discussions with Company D, including Pieris’ management’s determination that a potential acquisition or in-licensing was no longer a strategic option due to the inability of Company D to advance discussions because of their internal priorities and third-party contractual relationships. Pieris’ management presented details and a proposed strategy to independently advancing cinrebafusp alfa, including potential costs and financing needed, and compared that to a reverse merger with Company A. Stifel discussed the progress of the potential sale of the German research and development operations and indicated that over 100 companies had been contacted with two showing confirmed interest and conducting operational and technical due diligence. Pieris’ management also discussed the initial assessment of four reverse merger candidates that had been presented to date.
Between September 19, 2023 and September 28, 2023, one of the second round candidates removed itself from the reverse merger process.
On September 29, 2023, the Transaction Committee virtually met with Pieris’ management, Mintz representatives, and Stifel representatives to review the remaining reverse merger candidates and make a recommendation to the Pieris board of directors regarding those that should advance to round three. The Stifel representatives discussed various potential concerns with certain candidates, including lack of funding or investor support, public readiness, and concerns over the clinical trials or commercial landscapes for the drug candidates being developed by those companies. The Transaction Committee determined to recommend that three candidates move forward, one of which was Company A, and discussed potentially recommending a fourth candidate to advance to round three. During this meeting, Mr. Yoder also updated the Transaction Committee on the efforts to independently develop cinrebafusp alfa, and further discussed the required financing and a plan to obtain an independent analysis of the asset and clinical development plan proposed by Pieris’ management.
On October 2, 2023, the Pieris board of directors virtually met with Pieris’ management, Mintz representatives, and Stifel representatives. The Pieris board of directors reviewed the proposed offers from the three reverse merger candidates that the Transaction Committee recommended and the one candidate the Transaction Committee considered recommending, and decided to advance these four candidates into the final round of diligence. The Pieris board of directors discussed with Pieris’ management and Stifel representatives and approved the counteroffers to be made to the candidates. The Pieris board of directors also discussed the cinrebafusp alfa independent development strategy with Pieris’ management and reviewed the proposed plans, estimated budget, and potential financing required for this strategy. The Pieris board of directors agreed that the proposed strategy should continue to be explored and requested that Pieris’ management seek feedback from key opinion leaders, potential contract research organizations that would run the proposed clinical trials, and other potential key employees on the cinrebafusp alfa clinical development plan.
On October 4, 2023, one of the third-round reverse merger candidates withdrew from the process because they decided to stay private due to prevailing market conditions. At this stage, only three third-round candidates remained.
On October 6, 2023, the Pieris board of directors virtually met with Pieris’ management and Mintz representatives, and discussed the details of the cinrebafusp alfa independent development path. Mr. Yoder presented a draft pitch deck that would be used with potential investors and discussed the proposed timeline for clinical development. Mr. Bures also discussed the operational strategy such as required financing and the employees that would need to be retained.
On October 23, 2023, Pieris received an updated counterproposal from Company A’s management team. This offer decreased the value of cinrebafusp alfa from $53 million to $15 million, decreased the value of Pieris other proprietary assets, milestone and royalty assets and intellectual property to $3 million, and added a $12 million CVR for a milestone related to cinrebafusp alfa development.
On October 26, 2023, Pieris received a term sheet offer from Company C to acquire Pieris’ royalty and milestone rights under the Servier, Pfizer, Boston Pharmaceuticals, and Genentech partnerships. The proposed terms included a $5 million upfront payment and a CVR providing Pieris stockholders with a 50% payout of any milestones earned over the course of five years from the partnerships. Pieris’ management determined that this was unacceptable as it would potentially be more valuable to retain full ownership over the milestones and royalties.
On October 31, 2023, the Transaction Committee virtually met with Pieris’ management, Mintz representatives, and Stifel representatives. The Stifel representatives discussed updates on the offer progression with Company A. The Transaction Committee considered and, then instructed Stifel to send, a proposed counteroffer to Company A, which increased the value of cinrebafusp alfa to $22 million, increased the value of Pieris’ pipeline, milestone and royalty assets and intellectual property to $6 million, increased the proposed CVR for one cinrebafusp alfa regulatory milestone to $20 million and added another CVR of $8 million for a second cinrebafusp alfa regulatory milestone. The Stifel representatives updated the Transaction Committee on the ongoing discussions with two other remaining reverse merger candidates. Mr. Yoder also updated the Transaction Committee on the ongoing efforts to finance the cinrebafusp alfa independent development strategy.
On November 2, 2023, Pieris received a counterproposal from Company A, which decreased the value of cinrebafusp alfa to $16 million, decreased the value of Pieris pipeline milestone and royalty assets and intellectual property to $5 million, and removed the CVR on the second cinrebafusp alfa regulatory milestone.
On November 6, 2023, Pieris sent a counteroffer to Company A which maintained Company A’s terms but added back in a CVR for a second $8 million regulatory milestone for cinrebafusp alfa.
On November 8, 2023, the Transaction Committee virtually met with Pieris’ management, Mintz representatives and Stifel representatives and reviewed the proposal received from Company A on November 8, 2023, which decreased the CVR on the second cinrebafusp alfa regulatory milestone to $4 million.
That same day, Stifel representatives, at the instruction of the Transaction Committee, sent a revised proposal to Company A, which increased the value of the second cinrebafusp alfa regulatory milestone to $8 million and maintained the other terms.
On November 9, 2023, the Pieris board of directors virtually met with Pieris’ management, Mintz representatives and Stifel representatives to discuss the reverse merger process and the financing efforts for the cinrebafusp alfa independent development strategy. Mr. Yoder explained that after two weeks of investor outreach to approximately 30 investors, with the assistance of Stifel and an investor relations firm, the limited number of meetings indicated that it would be unlikely to secure the additional required capital to independently advance cinrebafusp alfa. Mr. Yoder informed the Pieris board of directors that based upon the remaining three candidates, Company A was the preferred option and that Pieris should continue to assess other potential fallback options including potentially in-licensing other assets with low capital expenditure requirements and a milestone and royalty monetization strategy. Since one of the reverse merger candidates did not respond to Pieris’ counteroffer as it was believed to be exploring a separate financing and because it appeared that the other candidate’s stockholders would not commit to a financing until clinical study readouts were obtained for its internal product candidate, the Pieris board of directors agreed to enter into an exclusivity agreement with Company A based on the terms received from Company A that day that decreased the second cinrebafusp alfa regulatory milestone to $5 million.
On November 9, 2023, the terms of a non-binding proposal were agreed upon by Pieris’ management and Company A’s management. Those terms assumed a net cash of $15-20 million, a valuation of cinrebafusp alfa of $16 million, the Nasdaq listing at $12 million, and other pipeline and royalty/milestone bearing assets at $5 million. Two CVRs were also provided for cinrebafusp alfa: one for the first regulatory milestone for $16 million and another for a second regulatory milestone for $5 million. The term sheet also contemplated the consummation of a PIPE financing of $65 million to be consummated in connection with the closing of the reverse merger.
On November 20, 2023, in a virtual meeting, the Pieris board of directors discussed with Pieris’ management and Mintz and Stifel representatives the terms of exclusivity for Company A, including a carve out to permit the Company to scientifically evaluate a clinically tested small molecule asset as a backup plan for a potential acquisition. Pieris’ management presented a background of the small molecule asset, including its method of action, historical clinical data, and future clinical plans.
On November 27, 2023, Pieris and Company A signed an exclusivity letter with an exclusivity term of 45 days and initiated two-way confirmatory diligence. The two-way confirmatory diligence included review of the strengths and weaknesses of each company’s development programs, cash runway post-inflection points, public company readiness/readiness to close, clinical strategy and commercial opportunity for lead programs, and an analysis of the strengths and weaknesses of each company’s board, investor base, and management team. The exclusivity letter included various carveouts permitting Pieris’ management to out-license or sell Pieris’ respiratory assets and other proprietary technology that did not involve 4-1BB as a target, and to continue to scientifically and technically evaluate a clinically tested small molecule asset, which was a potential in case the Company A merger was not consummated. During the week of November 20, 2023, Pieris began conducting diligence on the asset included in the exclusivity carve-out, focusing on clinical feasibility, scientific rationale, commercial opportunity, and cash requirements to bring asset through the next meaningful inflection point.
On December 13, 2023, in a virtual meeting, the Pieris board of directors and Pieris’ management met, with Mintz representatives present, and discussed, among other topics, the small molecule asset carved out from the exclusivity with Company A. The Pieris board of directors encouraged Pieris’ management to continue to conduct diligence to assess if it was a viable option for Pieris should the reverse merger not occur.
On December 29, 2023, Company A’s legal counsel sent Mintz a first draft of the definitive merger agreement. Between December 29, 2023 and February 6, 2024, Mintz and Company A’s legal counsel exchanged several drafts of the definitive merger agreement and met to discuss revisions to the draft merger agreement.
Between December 21, 2023 and February 16, 2024 Pieris’ management and the Pieris board of directors received updated reports on Company A’s PIPE financing efforts.
On January 6, 2024, Pieris’ management was notified that potential the asset carved out from the exclusivity agreement was in-licensed by another company, so Pieris ceased diligence on that potential asset.
On January 8, 2024, Pieris’ management identified three additional potential back-up assets for further evaluation.
From January 12, 2024 and until February 16, 2024, Pieris and Company A continued to negotiate the definitive merger agreement under exclusivity while Company A continued its PIPE financing efforts. As part of the exclusivity extensions, Pieris negotiated an additional carve-out for Pieris’ management to evaluate two of the additional potential back-up assets identified on January 8, 2024 as potential strategic acquisitions in case the reverse merger discussions with Company A fell through. Pieris’ management began conducting secondary scientific research on these potential back-up assets.
On January 24, 2024, the Pieris board of directors, Pieris’ management, and Mintz representatives met in-person in Boston, Massachusetts to discuss an overview of the milestone and royalty monetization scenario, the feasibility of implementing this alternate scenario, and an update on Company A’s investor outreach and Pieris’ diligence process of Company A. Mr. Bures presented the preliminary value assumptions of the milestone and royalty monetization scenario, as well as the net present value assessment for Pieris’ partnered assets, demonstrating that this path forward would be a better alternative for Pieris stockholders compared with liquidation. The Pieris board of directors agreed that this should be the back-up plan for Pieris should a transaction with Company A fail.
Between January 31, 2024 and February 5, 2024, Pieris’ management team met with Company A’s management team on multiple occasions to negotiate various business terms in the draft merger agreement.
On February 1, 2024, Pieris’ management and the Transaction Committee met virtually to discuss negotiation status of the definitive merger agreement, Company A’s investor outreach progress and whether to extend exclusivity beyond February 9, 2024.
On February 8, 2024, Pieris’ management met with Company A’s management team, via telephonic meeting, to discuss their progress on their PIPE financing efforts. Company A’s management informed Pieris that they had found a lead investor and expected a full syndicate to be assembled within approximately one week once terms were provided and negotiated. The Transaction Committee subsequently met that day to discuss Company A’s investor outreach efforts and a further extension to the exclusivity agreement.
On February 9, 2024, in a virtual meeting with the Pieris board of directors and Mintz representatives, Pieris’ management provided an update on Company A’s PIPE financing progress in connection with the potential reverse merger. The Pieris board of directors approved an additional one-week exclusivity extension period until February 16, 2024, with the expectation that PIPE financing terms and a definitive merger agreement would be agreed upon by then.
On February 16, 2024, in a virtual meeting, Pieris’ management and the Pieris board of directors met, with Mintz representatives present, to discuss Company A’s inability to secure an anchor investor and provide suitable investment terms. Due to this and the overall uncertainty of a potential PIPE Financing closing with Company A, the Pieris board of directors decided to allow Company A’s exclusivity to lapse but continued driving towards a potential final merger agreement with Company A. In parallel, the Pieris board of directors instructed Pieris’ management to continue to review the corporate governance and operational aspects of the milestone and royalty monetization strategy if a transaction with Company A could not be consummated.
On February 19, 2024, a third party reached out to Pieris’ management regarding a potential reverse merger transaction with their client, and on February 20, 2024, Pieris entered into a CDA with that company, which did not have any standstill provision, with that party. With the assistance of Stifel, Pieris’ management reviewed high-level information regarding that company, including its clinical assets, cash runway, and other operational aspects of the company. On February 23, 2024, Pieris received a LOI from that company. Due to concerns regarding that company’s need for additional financing and public company readiness, that company was informed on February 28, 2024 by Pieris’ management that Pieris would not be moving forward in the process with the company.
On February 20, 2024, Mr. Yoder and the chief executive officer of another company that was introduced by Stifel, Company E, had an introductory telephone conversation to discuss a potential reverse merger. Subsequently, Pieris entered into a CDA with Company E which did not include any standstill provisions.
On February 26, 2024, in a virtual meeting with the Pieris board of directors, along with representatives of Mintz, Pieris’ management indicated that Company A was still unable to secure an anchor investor in connection with a potential PIPE and Company A continued to extend timelines that would impact both cash runway for Pieris and prolong cinrebafusp alfa’s wind down timelines. Accordingly, the Pieris board of directors concluded that a reverse merger supported by a PIPE financing with Company A was unlikely to materialize, but that licensing discussions for cinrebafusp alfa should still be explored with Company A. The Pieris board of directors and Pieris’ management also discussed the progress in preparing for the royalty and milestone monetization strategy if no alternative and superior strategy was available.
On February 27, 2024, Company E sent a confidential company overview deck to Pieris’ management.
Between March 1, 2024 and March 19, 2024, Pieris’ management met with Company E’s management team to discuss the background, merits and strategy of their product candidate portfolio and conducted scientific, technical, commercial diligence on Company E and its assets. This included engaging a third party consulting firm to review the assets and interview various scientific and regulatory experts to provide feedback on the assets and the relevant indications. On March 8, 2024, Company A’s chief executive officer informed Mr. Yoder that they were unable to secure a PIPE financing for a reverse merger transaction and, consequently, Company A’s board of directors did not authorize any type of transaction, including both merger and licensing, because the opportunity was not validated by new investors.
Also on March 8, 2024, Company E’s management team virtually presented to the Pieris board of directors on their company’s pipeline and programs, management team, investor base, and product development plans. Pieris’ management and Pieris board of directors members asked questions relating to Company E’s public company readiness, cash needs and quality of its investor base, financing capabilities and outlook, the timeline of valuation inflection points within the existing and proposed cash runways, the risks and opportunities within their pipelines, the costs of their clinical development and manufacturing plans for their lead asset, commercial opportunities and risks, and partnership strategies.
On March 13, 2024, the Pieris board of directors, Pieris’ management and representatives from Mintz and Pieris’ third-party consulting firm met in-person in Boston, Massachusetts, with some of Pieris’ management virtually present, to discuss the initial evaluation of Company E based on internal diligence and the competitive assessment conducted by the third party consulting firm. Pieris’ management reviewed Company E’s pipeline, including the value proposition, differentiation, opportunities and risks, and the continued diligence and evaluation that was planned for their lead asset before a final recommendation could be reached. Pieris’ management also presented an update on the preparatory activities being conducted for the potential milestone and royalty monetization strategy and the risks and benefits associated with this plan in comparison to liquidation. The Pieris board of directors determined that a transaction with Company A was not feasible because they could not secure PIPE financing to adequately develop cinrebafusp alfa and would not consider a licensing transaction.
On March 19, 2024, a managing director at BVF, Pieris’ largest stockholder, emailed Mr. Yoder to discuss a rare skin disease company, Palvella, that BVF was also an investor in, as a potential reverse merger candidate. Later that day, Mr. Yoder spoke with the BVF managing director and discussed the background of Palvella. The managing director subsequently introduced Mr. Yoder to the chief executive officer of Palvella, Wesley H. Kaupinen.
Also on March 19, 2024, in a virtual meeting with the Pieris board of directors, Pieris’ management and Mintz representatives discussed Pieris’ recommendation to not proceed with a transaction with Company E and to further discuss the milestone and royalty monetization plan as the preferred go-forward strategy. The Pieris board of directors agreed with the Pieris management’s recommendation regarding Company E, on the basis that there was a low likelihood of securing a PIPE financing within the desired timelines in addition to the commercial and access concerns and regulatory and technical risks for the two most advanced assets. The Pieris board of directors concluded that the monetization strategy had the potential to provide more value to Pieris stockholders versus liquidation, based upon a comparison of the two strategies presented by Pieris’ management, and would permit Pieris to explore other strategic options, such as asset out-licensing, strategic mergers, or leveraging partnership milestones for other monetization opportunities.
On March 20, 2024, Pieris’ management met with Palvella’s management team who presented a corporate overview, their product pipeline, and their interest in a potential reverse merger transaction. Following this meeting, Pieris’ management decided to recommend to the Pieris board of directors that Pieris should consider Palvella as a potential reverse merger candidate.
On March 21, 2024, Mr. Yoder had a discussion with the BVF managing director and gave an update on the potential merger and concurrent PIPE financing discussions between Palvella and Pieris, including the terms the parties were considering.
On March 22, 2024, Mr. Yoder had a call with the BVF managing director in which both BVF reiterated its continued interest in a transaction between Pieris and Palvella on the terms discussed previously. That same day Pieris entered into a CDA with Palvella, which did not include standstill provisions.
On March 25, 2024, in a virtual meeting of the Pieris board of directors with Pieris’ management, and Stifel and Mintz representatives, the Pieris board of directors considered whether to adopt and implement the milestone and royalty monetization strategy. As part of the discussion, Pieris’ management and a Mintz representative presented the strategic review process by the Company, including the reverse merger process, the potential transactions with Companies A, B, C, D, and E and the strategy of independently developing cinrebafusp alfa. The Stifel representatives and Mintz representatives reviewed the monetization strategy with other paths forward, including liquidation and dissolution, and discussed the potential impact of the strategies on maintaining the Nasdaq listing. After careful consideration, the Pieris board of directors endorsed the monetization strategy, including any reductions in force that were required, because it would permit the Company to run on a leaner operating cash burn and there were no external funding requirements to this operating model, while also preserving its ability to collect potential near-term milestones from its partnerships and allowing the Company to explore other potential strategic opportunities that might arise, such as a reverse merger with Palvella. Pieris’ management then the provided an overview of Palvella, including Palvella’s operations and product pipeline, and discussed the additional diligence that would need to be addressed prior to determining whether to consider Palvella as a reverse merger candidate as well as preliminary transaction terms, including valuation and CVRs. The Pieris board of directors authorized Pieris’ management to continue to explore a potential transaction with Palvella.
On March 26, 2024, Pieris’ management shared their first round of diligence questions with Palvella’s management team, which primarily focused on clinical, regulatory, and commercial questions regarding their lead asset. On the same day, Pieris sent Palvella an initial draft of a non-binding LOI with proposed terms based on the initial discussions between the companies. The LOI set an initial valuation of Palvella at $75 million, an initial valuation of Pieris at $25 million, which was comprised of an expected cash balance at closing of $15 million with an upward and downward collar of $1.5 million, and a value of Pieris’ Nasdaq listing at $10 million. The LOI also provided a CVR for 100% of the milestones and royalties from Pieris’ partnered programs (i.e., the Servier, Pfizer, and Boston Pharmaceuticals licensing agreements) and customary closing conditions, including approval of the transaction by the boards and stockholders of both parties and written commitment of equity investment in Palvella by certain investors. The LOI also included a net cash definition and a proposed exclusivity term of 30 days along with carveouts to permit Pieris to dispose of its legacy assets. The LOI indicated that the board of the combined company would be composed of directors from Pieris and Palvella that are generally proportional to the ownership percentage of each party of the combined company post-closing, but did not at that time specify the total number of directors from each party, and also included a 180-day lock up provision for continuing executive officers and members of the board post-transaction. It also specified that any personnel considerations, including senior board composition, of the combined company, would be determined post-closing. These were terms that Pieris’ management felt could be acceptable to its Board for a potential transaction.
On March 27, 2024, Pieris filed a current report on Form 8-K disclosing its strategy to maximize partnered milestone and royalty potential from its partnered 4-1BB bispecific Mabcalin® protein IO assets. In announcing this strategy, Pieris stated that it may still consider other strategic options.
On March 28, 2024, Palvella’s management team provided virtual data room access to Pieris’ management and the Pieris board of directors, which included confidential information regarding their assets and financials.
On April 1, 2024, Palvella’s management team provided responses to all of Pieris’ first round diligence questions via email.
On April 3, 2024, Palvella’s management team presented to the Pieris board of directors Palvella’s pipeline and clinical plans, including clinical and commercial timelines and inflection points, and the potential PIPE financing required for a transaction. The Pieris board of directors questioned Palvella’s management on a valuation analysis and several matters related to Palvella’s clinical development plan. These questions related to the reasoning for Palvella deciding to conduct a baseline-controlled study for QTORIN rapamycin in microcystic lymphatic malformations versus a randomized clinical study that contained a placebo arm, and the basis for comparative companies that were selected by Palvella as part of a potential valuation analysis that was presented. After the presentation to the Pieris board of directors, Mr. Yoder spoke with Mr. Kaupinen and discussed the additional diligence questions that came up from the Pieris board of directors call.
Between April 9 and May 14, 2024, Mr. Yoder had several discussions with Mr. Kaupinen and the BVF managing director regarding the valuation of Palvella. During these discussions, Mr. Kaupinen indicated that the valuation of Palvella that was initially proposed by Pieris was not acceptable to Palvella. After further discussions, including discussions with Mr. Kaupinen and the managing director from BVF, Mr. Kaupinen indicated a valuation of $95 million for Palvella would likely be acceptable. The BVF representative also indicated their willingness to lead a $30 million plus PIPE transaction in which BVF would commit to $15 million based on those valuation terms of Palvella and Pieris, which, for Pieris, was based on its net cash plus $10 million for its Nasdaq listing.
On April 10, 2024, in a virtual meeting of the Pieris board of directors, Pieris’ management and Stifel representatives reviewed with the Pieris board of directors the potential reverse merger opportunity with Palvella, including Palvella’s clinical trial plans for QTORIN rapamycin, its lead asset. Stifel representatives informed the Pieris board of directors that Palvella had indicated it would be willing to assign all milestone and other payments under Pieris’ strategic partnerships through CVRs. Mr. Bures then presented an estimated net cash projection through the end of the third quarter if Pieris continued to advance a potential transaction with Palvella. The Pieris board of directors instructed Pieris’ management, with the assistance of Stifel, to continue negotiating key terms, such as valuation, breakup fee, and terms for a PIPE.
On April 12, 2024, the Pieris management team shared their second round of diligence questions with Palvella’s management team, focusing primarily on chemical, manufacturing and control management, financial, and intellectual property questions.
On April 18, 2024, Palvella’s management team provided answers to all of Pieris’ second round diligence questions, via email.
On April 19, 2024, the management teams of each of Pieris and Palvella met to discuss follow up questions from the Pieris board of directors regarding Palvella’s clinical trials plans.
On April 23, 2024, in a virtual meeting with Pieris’ management and Stifel and Mintz representatives, the Pieris board of directors met to discuss the transaction terms negotiated with Palvella, including key financial terms, as well as the potential concurrent PIPE transaction. The Stifel representatives reviewed preliminary financial aspects of a potential transaction with Palvella, including potential valuations and pro-forma ownership for Pieris and Palvella, which potential valuations for Palvella were informed by the public market values of selected public companies. Pieris’ management indicated that based upon discussions to date, a $95 million valuation for Palvella would likely be the lowest the board of directors of Palvella could accept and would be a valuation BVF would likely accept to lead a PIPE transaction. The group then discussed proposed valuations, CVRs for the potential milestone and royalties under Pieris’ strategic partnership agreements, and the sufficiency of the potential PIPE and other funding being sought by Palvella. The Stifel representatives also reviewed a comparison of the estimated net cash/valuation in a reverse merger scenario to the milestone and royalty monetization strategy that Pieris was pursuing as well as the liquidation scenario. Based on this comparison, the Pieris board of directors agreed that a reverse merger could provide better value than the milestone and royalty monetization strategy or liquidation, particularly given the CVR component of the envisioned transaction. Finally, Mr. Yoder and Dr. Shane Olwill, Pieris’ Chief Development Officer, presented additional information on Palvella’s clinical trial plans in response to questions that were raised by the Pieris board of directors. The Pieris board of directors authorized the Transaction Committee to review the terms of an LOI with Palvella and approve and authorize Pieris’ management to execute and perform its obligations under the approved LOI.
On April 26, 2024, Palvella sent a revised copy of the LOI to Pieris. The LOI maintained Pieris’ valuation at $25 million assuming net cash of $15 million with an upward and downward collar of $1 million, a CVR providing Pieris pre-merger stockholders with the right to receive all milestone or royalty payments received by Pieris in connection with its partnered programs from Servier, Pfizer, and Boston Pharmaceuticals, a 30-day exclusivity term along with carveouts to permit Pieris to dispose of its legacy assets as well as a PIPE financing in an aggregate amount of no less than $30 million in gross proceeds as a closing condition, in addition to other customary terms. The parties had previously discussed the minimum PIPE proceeds that would be needed based upon the expected cash of the combined company after close and the expected capital needed to provide sufficient cash runway after certain inflection points. Palvella also revised the LOI to provide for one director from Pieris on the combined company’s board, and an undefined break-up fee provision if there was an early termination of the to-be-executed merger agreement, including in the event Pieris did not obtain stockholder approval.
On April 27, 2024, a Mintz representative shared a revised LOI with Palvella’s legal representative at Troutman. The LOI included a valuation of Palvella at $95 million, which was based upon discussions with Pieris’ board of directors and what potentially would be acceptable to Palvella. The revised LOI also provided that the PIPE financing would not include a discount greater than 20%, based upon a valuation of Palvella of $95 million, removed a break-up fee in the event Pieris did not receive stockholder approval, and maintained that the total number of board members would be agreed upon at a later time. The LOI was further negotiated over the next several days between management and representatives of Pieris and Palvella, including the break-up fee provision, the valuation of Palvella, and the number of Pieris directors that would be on the combined company’s board of directors.
On May 3, 2024, the Transaction Committee virtually met with Pieris’ management, Stifel representatives and Mintz representatives, to discuss the status of Palvella’s LOI and its key terms, including Palvella’s valuation. Mr. Yoder indicated there were ongoing discussions regarding Palvella’s valuation, and that Pieris’ management was continuing to request additional information regarding the Phase 3 clinical trial design for QTORIN rapamycin in microcystic LM, including information related to any discussions or meetings with regulatory authorities. Following the meeting, Mr. Yoder provided a similar update to the full Pieris board of directors via email.
On May 9, 2024, the Science and Technology Committee, or the “S&T Committee” of the Pieris board of directors met with Pieris’ management and Palvella’s management team to discuss Palvella’s clinical trial design for QTORIN rapamycin in microcystic LM. During this meeting, a key discussion point between the S&T Committee and Palvella was the rationale for the baseline-controlled design and potential alternative study designs, as well as the materials and communications from meetings between Palvella and regulatory authorities, which were provided to the S&T Committee prior to the meeting. While the S&T Committee continued to deliberate on the implications of the clinical trial design, it ultimately determined that Palvella should be entitled to management and discretion on the matter due to Palvella’s discussions with regulatory authorities regarding the clinical trial design, and Palvella’s review of the clinical trial design with principal investigators, counsel and regulatory advisors; accordingly, the S&T Committee continued to support advancing the proposed transaction based on the clinical trial design and plans.
On May 14, 2024, in a virtual meeting with Pieris’ management, Stifel representatives and Mintz representatives, the Transaction Committee and two other members of the Pieris board of directors met to discuss the May 9, 2024 S&T Committee meeting and the FDA and S&T committee feedback on Palvella’s planned Phase 3 clinical trial design for QTORIN rapamycin in microcystic LM. The Transaction Committee then discussed the proposed terms of the LOI, which were not impacted by the clinical trial design decisions. The Transaction Committee acknowledged the S&T Committee’s discussion and Palvella’s efforts in designing the clinical trial, and authorized management to enter into the LOI with Palvella based on the terms presented. Also on May 14, 2024, Pieris received another revision of the LOI from Palvella that included a $95 million valuation for Palvella. Pieris also agreed to having one Pieris director on the combined company’s board in light of the agreed-upon valuations of the company and expected equity allocation. This revision of the LOI also provided that certain breakup fees would be agreed upon in the Merger Agreement, without specifying the amounts, and stated that Pieris would reimburse Palvella for reasonable costs and expenses in the case that Pieris was unable to obtain stockholder approval for the merger transaction. Prior to signing, Pieris and Palvella further negotiated the breakup fee provision such that in the case that stockholders from either party do not approve the merger transaction, then such party would cover the reasonable expenses and costs incurred by the other party up to $500,000.
On May 16, 2024, Mr. Yoder executed the LOI and on May 18, 2024, Mr. Kaupinen countersigned the LOI.
On May 30, 2024, Pieris sent Palvella another set of diligence requests focusing on clinical and regulatory, financial and financing, operational, corporate, employment, privacy and data security, and intellectual property topics.
Between May 30, 2024 and July 18, 2024, Pieris’ management met weekly to discuss progress updates on the merger agreement, diligence topics, PIPE marketing, the S-4 drafting and other topics. Between May 30, 2024 and July 22, 2024, members of Pieris’ management and Mintz, together with Palvella’s management and Troutman, continued confirmatory diligence and negotiated (i) the terms of the definitive agreement, (ii) support and lockup agreements to be executed by directors, officers and certain significant stockholders of each party and (iii) the CVR. In addition to the terms noted above, the material terms negotiated were the definition of Pieris’ net cash, the definition of the exchange ratio, including the valuation of Pieris, the treatment of Palvella’s stock options in connection with the exchange ratio calculation, certain conditions to closing (including in relation to closing of the concurrent PIPE financing), Pieris’ ability to sell or license Pieris’ non-cash assets, certain offset rights to be included in the CVR Agreement to cover potential liabilities under the Pfizer and Boston Pharmaceuticals partnership agreements, and the termination fees.
On May 31, 2024, Troutman sent Pieris a diligence request list which focused on corporate governance, corporate operations, employment, privacy and data security, intellectual property, and financial and financing topics.
On June 6, 2024, Troutman sent Pieris the first draft of the Merger Agreement. The first draft provided that directors, officers, and certain stockholders of each party, who would be defined at a later time, would sign support and lock up agreements, and that the Pieris officers would resign and Palvella officers would become the officers of the combined company following the Merger. The draft also included a $25 million valuation for Pieris, based on an assumed $15 million in net cash, with an upward and downward $1 million collar, and $10 million for the Nasdaq listing. It also included an exchange ratio definition that, solely for the purposes of the calculation, included Palvella’s shares on a fully diluted basis and used the treasury stock method for options and warrants, and assumed the Pieris shares would be calculated on a fully diluted basis and would assume, solely for the purposes of calculating the exchange ratio, acceleration, exercise and conversion of outstanding options and warrants that were less than or equal to an undefined dollar value threshold. For clarity, the draft Merger Agreement did not contemplate vesting acceleration of Pieris’ stock options. The draft further included the ability for either party to terminate if there was an intervening event, which provided that if there was any event or change in circumstances that led either party’s board to assume a higher valuation for its company (without any third-party offer), then they could change their merger recommendation. The initial draft also included a closing condition that would require investors to sign subscription agreements in connection with the PIPE financing representing no less than $30 million in order for Palvella to be obligated to close the transaction.
On June 7, 2024, Mintz sent Palvella a draft CVR agreement. Between June 7, 2024 and July 21, 2024, the parties negotiated and revised the draft CVR agreement to remove the Servier partnership after Pieris received a notice of termination from Servier on June 28, 2024, and to include any licenses or other agreements involving Pieris’ legacy assets, such as PRS-400 or PRS-220, entered into by Pieris after the date of the Merger Agreement but prior to close. Other terms of the CVR that were negotiated included the mechanics of the CVR, such as the payment procedures to Pieris legacy stockholders and the process of resignation, removal, and appointment of the holder representative. Pieris further negotiated the inclusion of a security interest to the holder representative, on behalf of the Pieris legacy stockholders, in the strategic partnership agreements and payments under them, and certain audit rights of the holder representative to audit the combined company’s records and books. The parties also negotiated an initial expense fund to cover expenses in connection with the CVR, such as payment of license fees and patent maintenance fees, which was based on Pieris’ reasonable expectation of the costs associated with the strategic partnership agreements. The parties also negotiated covenants on the combined company to, among other things, require the combined company continue to maintain the, and take no action to terminate, modify, amend or take other action that would adversely affect, the strategic partnership agreements.
On June 9, 2024, Mr. Yoder and Mr. Kaupinen had a virtual meeting to discuss certain transition matters, including a process to select a Pieris-nominated director to remain on the Pieris board of directors following the anticipated close of the merger transaction.
On June 13, 2024, Mr. Kaupinen informed Mr. Yoder that a convertible note agreement was executed with certain investors to provide up to $20 million financing, which would form part of the concurrent PIPE financing for the transaction with Pieris.
On June 17, 2024, Pieris and Palvella agreed to extend the exclusivity term until July 19, 2024. Also on this date, Troutman sent Mintz and Pieris initial drafts of the certain exhibits to the Merger Agreement, including first drafts of the lock-up agreement and support agreements to be signed by the directors, officers and certain stockholders of Pieris and Palvella.
On June 19, 2024, Mintz sent Troutman revisions to the Merger Agreement which revised the calculation of Palvella’s outstanding shares, such that the Palvella’s outstanding shares for the purposes of calculating the exchange ratio would be based on common stock that are issued and outstanding prior to the Effective Time and expressed on a fully diluted and as converted basis. Based on this method, the Palvella exchange ratio, assuming a $25 million valuation for Pieris and $95 million valuation for Palvella, would have been 0.2502, and pre-Merger Palvella shareholders would have owned 79.17% of the combined company, prior to giving effect to the PIPE financing. The revisions also revised the scope of the representations and warranties for both parties and included additional representations and warranties for Palvella, revised certain covenants of both parties, and revised carveouts from Pieris’ negative covenants to expand the kinds of transactions where Pieris could dispose of its assets without Palvella’s consent, revised the net cash definition, and revised the closing conditions to include consummation of a concurrent financing in order for both parties to close the transaction. The intervening event provision was removed by Pieris as it believed this would pose a greater risk to the transaction for Pieris.
On June 26, 2024, Mintz sent Troutman comments on the lock-up agreement and support agreements.
On June 27, 2024, the Pieris board of directors virtually met with Pieris’ management, Stifel representatives and a Mintz representative to discuss updates to the progress of the Merger Agreement negotiations. Mr. Karam Hijji, acting general counsel, informed the Pieris board of directors that the parties continued to negotiate various aspects of the agreement including the share exchange ratio, representation and warranties, affirmative and negative covenants, and the closing conditions and termination provisions. Mr. Yoder also updated the Pieris board of directors on the status of Palvella’s ongoing PIPE financing efforts, as well as its impact on the potential signing date of the Merger Agreement, and the status of Palvella’s year-end audit and first quarter 2024 financial review status and its impact on the timing of the Form S-4 filing. During the meeting, Pieris’ management proposed Mr. Chris Kiritsy, a member of Pieris’ board of directors and the chair of the Pieris’ audit committee, as Pieris’ continuing director for the combined company’s board of directors due to his considerable experience in the pharmaceutical industry and his expertise in finance and corporate development. Pieris’ management indicated that Palvella’s management also supported this decision as Mr. Kiritsy’s experience would be invaluable to the combined company’s board. The Pieris board of directors agreed to propose Mr. Kiritsy as the continuing director in the combined company from Pieris.
On June 28, 2024, Troutman representatives sent Mintz representatives a revised draft of the Merger Agreement. The draft included revisions to the closing conditions, and revised certain aspects of the net cash definition, representations and warranties, and covenants. The draft also revised the exchange ratio definition to provide that Palvella’s outstanding shares, for the purposes of the exchange ratio, would be based on the treasury stock method. Based on this method, the exchange ratio, assuming a $25 million valuation for Pieris and $95 million valuation for Palvella, would have been 0.2644 and pre-Merger Palvella shareholders would have owned 80.06% of the combined company (a 0.90% difference when compared with the method proposed in the June 17, 2024 draft of the Merger Agreement), prior to giving effect to the PIPE financing. The revised draft of the Merger Agreement also provided that Pieris would adopt a new equity plan, subject to closing of the Merger, and also removed the mutual closing condition to have executed subscription agreements from investors for a PIPE financing of no less than $30 million in order for both parties to be obligated to close, instead making it only a closing condition for Palvella.
On July 9, 2024, Mintz representatives sent back a revised copy of the Merger Agreement, which included revisions to the closing conditions to include consummation of the PIPE financing in accordance with the subscription agreements as a closing condition for both parties, and revisions to the net cash definition.
On July 10, 2024, Mintz and Troutman finalized the terms of the support agreements.
On July 11, 2024, Pieris management met with Palvella’s management and a Troutman representative to discuss additional open points in the Merger Agreement, including the treatment of any Palvella stock options and the calculation of the exchange ratio. The parties agreed to use the treasury stock method in calculating the outstanding shares of both parties for the purposes of the exchange ratio calculation, but agreed that the treasury stock method would not apply to any options granted after the date of the Merger Agreement. In deciding to agree to this, Pieris took into account the limited impact on the equity allocations by utilizing the treasury stock method and its interest in continuing to advance the negotiations.
On July 13, 2024, Troutman and Mintz finalized the terms of the lock-up agreement.
On July 17, 2024, Troutman representatives sent Mintz representatives a revised copy of the Merger Agreement, which included proposed termination fees. Troutman and Mintz also determined ownership levels above which stockholders of Pieris and Palvella, respectively, would be asked to sign the lock-up and support agreements, in addition to the officers and directors of Pieris and Palvella.
On July 18, 2024, Pieris management and Palvella’s management met to discuss open items in the Merger Agreement including the calculation of Pieris’ valuation. The parties discussed Pieris’ updated net cash assumption of $11 million and agreed to remove the net cash collar, in connection with calculating the Pieris valuation for the exchange ratio, and instead agreed for a dollar-by-dollar adjustment of Pieris’ valuation. Pieris had determined that the updated net cash assumption was based upon more accurate forecasts and that the net cash collar may be less advantageous than a dollar-for-dollar adjustment.
On July 18, 2024, Palvella’s board of directors held a virtual meeting at which members of Palvella’s management and representatives of Troutman were present. During the meeting, management provided an update on the negotiation process for the Merger Agreement, the ancillary agreements and the PIPE financing. Troutman discussed with the Palvella board of directors their fiduciary duties and summarized the material terms of the Merger Agreement, the ancillary agreements and the PIPE financing. The Palvella board of directors then discussed various considerations with respect to the proposed transaction, as summarized under the section titled “The Merger—Palvella’s Reasons for the Merger” beginning on page 137 of this proxy statement/prospectus.
On July 19, 2024, Mintz representatives sent a revised copy of the Merger Agreement to Troutman, which included updates to Pieris valuation based on revised estimates of Pieris’ net cash, revision to the exchange ratio definition to include treatment of the options as agreed to by the parties in their July 11, 2024 meeting, and revisions to the termination fees to reduce termination fees that may be payable by Pieris under specified circumstances. This draft of the Merger Agreement also set the combined company board to five directors, with one of the five being a Pieris director. The board size was negotiated to align with the expected equity allocation between the Pieris and Palvella stockholders in the combined company, prior to giving effect to the PIPE financing.
Between July 19, 2024 and July 21, 2024, Pieris and Palvella negotiated the final remaining open items to the Merger Agreement, including the termination fees and agreed that under specified circumstances, Pieris may be required to pay Palvella a termination fee of $1.0 million and Palvella may be required to pay Pieris a termination fee of $2.0 million. Pieris considered the relative positions of each side and other deal precedent in agreeing to these termination fees.
On July 22, 2024, the Palvella board of directors, via unanimous written consent, approved the Merger Agreement, the ancillary documents and the transactions contemplated thereby.
On July 23, 2024, prior to the Pieris board of directors meeting, Pieris’ Audit Committee of the Pieris board of directors met to discuss the related party transaction aspect of the transaction with Palvella with respect to BVF. After review of the relevant facts, including BVF’s interest in the transaction and whether such transaction was in the best interest of Pieris stockholders, the Audit Committee approved the related party transaction. The Audit Committee took into account that BVF did not hold a seat on the Pieris board of directors and that another investor was leading the concurrent PIPE financing based on the valuation set forth in the Merger Agreement.
On July 23, 2024, the Pieris board of directors virtually met with Pieris’ management, Stifel representatives, Mintz representatives, and a representative from Pieris’ Nevada counsel, Brownstein Hyatt Farber Schreck, LLP. During this meeting, Mr. Hijji presented to the Pieris board of directors regarding the material terms of the Merger Agreement and the ancillary agreements and concurrent PIPE financing. Mintz also presented on the fiduciary duties of the Pieris board of directors. Stifel provided the Pieris board of directors with a financial presentation, which included a review of the material financial analyses performed by Stifel, and rendered its opinion to the Pieris board of directors, which opinion was initially rendered verbally and subsequently confirmed in the Stifel Opinion dated July 23, 2024, to the effect that as of such date and based upon and subject to the assumptions made, and the qualifications and limitations upon the review undertaken by Stifel in preparing the Stifel Opinion, the Aggregate Merger Consideration (as defined in such opinion) to be paid by Pieris to the holders of Palvella capital stock in the Merger pursuant to the Merger Agreement was fair to Pieris from a financial point of view. The Pieris board of directors then discussed various considerations with respect to the proposed transaction, as summarized under the section titled “Pieris Reasons for the Merger and Recommendation of the Pieris Board of Directors.” Following discussion and the presentations, the Pieris board of directors unanimously approved the Merger Agreement and the transactions contemplated by the Merger Agreement, including the PIPE Financing.
Later on July 23, 2024 Pieris and Palvella executed the Merger Agreement. On July 24, 2024, Pieris and Palvella issued a joint press release announcing the transaction.
Pieris’ Reasons for the Merger
During the course of its evaluation of the Merger Agreement and the transactions contemplated by the Merger Agreement, the Pieris board of directors held numerous meetings, consulted with Pieris’ senior management, legal counsel and financial advisor, and reviewed and assessed a significant amount of information. In reaching its decision to approve the Merger Agreement and the transactions contemplated by the Merger Agreement, the Pieris board of directors considered numerous factors that it viewed as supporting its decision to approve the Merger Agreement, including among others, the following (which are not in any relative order of importance and all of which the Pieris board of directors viewed as supporting its decision to approve the proposed transactions with Palvella):
|
● |
that the historical and current information concerning Pieris’ business, financial condition, operations and prospects, including financial projections of Pieris under various scenarios and its short- and long-term strategic objectives, and the risks associated with continuing to operate Pieris on a stand-alone basis, particularly in light of the decision to winddown its proprietary preclinical and clinical programs, including cinrebafusp alfa, PRS-220, and PRS-400, reduce its workforce, and terminate various partnerships, including its partnerships with AstraZeneca in October 2023 and Servier, which Pieris was notified would be terminated in December 2024; |
|
● |
that there are risks for Pieris associated with Pieris’ ability to attract and retain talent should Pieris continue to operate on a stand-alone basis; |
|
● |
that the Pieris board of directors, with the assistance of Pieris’ financial advisor, undertook a comprehensive and thorough process of reviewing and evaluating multiple potential strategic alternatives, including the acquisition of new assets and independent development of existing assets, and reverse merger partner candidates to identify the opportunity that would, in the view of the Pieris board of directors, create the most value for Pieris stockholders; |
|
● |
the Pieris board of directors’ belief, after a thorough review of strategic alternatives and discussions with Pieris’ senior management, financial advisors and legal counsel, that the Merger is more favorable to Pieris stockholders than the potential value that might have resulted from other strategic alternatives available to Pieris, including continuing to operate Pieris on a stand-alone basis or conducting a dissolution and liquidation of Pieris and distributing any available cash to its stockholders; |
|
● |
the Pieris board of directors’ belief that, as a result of arm’s length negotiations with Palvella, Pieris and its representatives negotiated the highest exchange ratio to which Palvella was willing to agree, and that the other terms of the Merger Agreement include the most favorable terms to Pieris in the aggregate to which Palvella was willing to agree; |
|
● |
the Pieris board of directors’ review with Pieris management of Palvella’s current development plans for its lead asset, QTORIN rapamycin to confirm the likelihood that the combined company would possess sufficient resources or have access to sufficient resources to allow the management team to focus on its plan for the continued development of Palvella’s product pipeline, including the possibility that the combined company would be able to take advantage of the potential benefits resulting from the combination of Pieris’ SEC registration and Nasdaq listing with Palvella’s business to raise additional funds in the future; |
|
● |
the Pieris board of directors’ consideration of the expected cash balances of the combined company as of the closing of the Merger resulting from the approximately $11 million of net cash (subject to certain assumptions) expected to be held by Pieris upon completion of the Merger together with the cash Palvella currently holds and the approximately $78.9 million of expected gross proceeds from the PIPE Financing; |
|
● |
the ability of Pieris stockholders to participate in the growth and value creation of the combined company following the closing of the Merger by virtue of their continued ownership of Pieris common stock; |
|
● |
the ability of Pieris stockholders to potentially realize the full value of potential milestones and royalties under Pieris’ legacy partnership agreements with Pfizer and Boston Pharmaceuticals through the CVR Agreement; |
|
● |
the Pieris board of directors’ view that the combined company will be led by an experienced senior management team from Palvella, many members of which have extensive drug development, research and development, business and regulatory expertise and a board of directors with representation from each of the current boards of directors of Palvella and Pieris; |
|
● |
the current financial market conditions and historical market prices, volatility and trading information with respect to Pieris common stock; |
|
● |
the opinion, dated July 23, 2024, of Stifel to the Pieris board of directors as to the fairness, from a financial point of view and as of the date of such opinion, of the Aggregate Merger Consideration (as defined in such opinion) to be paid by Pieris to the holders of Palvella capital stock in the Merger pursuant to the Merger Agreement, as more fully described below under the caption “The Merger—Opinion of Pieris’ Financial Advisor,” beginning on page 140 in this proxy statement/prospectus; |
|
● |
that the Pieris board of directors reviewed and considered the terms of the Merger Agreement, including the parties’ respective representations, warranties and covenants, and the conditions to their respective obligations to consummate the Merger, the issuance of shares of Pieris common stock and the other transactions contemplated by the Merger Agreement. See the section titled “The Merger Agreement” beginning on page 163 in this proxy statement/prospectus for a detailed discussion of the terms and conditions of the Merger Agreement. In particular, the Pieris board of directors considered the following: |
|
o |
the calculation of the exchange ratio used to establish the number of shares of Pieris common stock to be issued to Palvella stockholders in the Merger, subject to adjustment in accordance with the Merger Agreement based on the valuation of Pieris; |
|
o |
the ability of Pieris to continue to conduct its business, including disposing of its legacy assets; |
|
o |
the nature and number of the conditions to the parties’ obligations to consummate the transactions contemplated by the Merger Agreement and the Pieris board of directors’ belief as to the likelihood of satisfying such conditions; and |
|
o |
the provisions in the Merger Agreement that provide for the ability of the Pieris board of directors to withdraw or modify its recommendation that holders of Pieris common stock approve the issuance of shares of Pieris common stock or the Charter Amendment Proposals (as defined in the Merger Agreement) following the receipt of an alternative Acquisition Proposal that the Pieris board of directors determines in good faith (after consultation with its outside counsel and its financial advisor) is a superior proposal (as defined in the section titled “The Merger Agreement” beginning on page 163), subject to certain restrictions imposed by the Merger Agreement, including that the Pieris board of directors shall have determined in good faith (after consultation with its outside legal counsel) that the failure to take such action would be inconsistent with its fiduciary duties to Pieris stockholders under applicable law and that Palvella shall have been given an opportunity to match the superior proposal. |
|
● |
in the course of its deliberations, the Pieris board of directors also considered a variety of risks and other countervailing factors related to the Merger and other transactions contemplated by the Merger Agreement, including, among others: |
|
o |
the fact that Pieris stockholders will be sharing participation of Pieris’ upside with Palvella stockholders as part of the combined company; |
|
o |
the substantial expenses to be incurred in connection with the Merger and the other transactions contemplated by the Merger Agreement; |
|
o |
the fact that projections of future results of operations and synergies are estimates based on assumptions that may not be realized within the expected timeframe or at all; |
|
o |
the possible volatility, at least in the short term, of the trading price of Pieris common stock resulting from the announcement of the Merger Agreement; |
|
o |
the risk that the Merger might not be consummated in a timely manner or at all and the potential adverse effect of the public announcement of the Merger Agreement or on the delay or failure to complete the transactions contemplated by the Merger Agreement on Pieris’ financial position; |
|
o |
the risk that the PIPE Financing might not be consummated in a timely manner or at all; |
|
o |
the terms of the Merger Agreement, including covenants relating to (1) the two companies’ conduct of their respective businesses during the period between the signing of the Merger Agreement and the completion of the Merger and the other transactions, including the requirement that the two companies conduct business only in the ordinary course, subject to specific exceptions and (2) the restrictions on Pieris’ ability to solicit alternative transaction proposals and dispose of its legacy assets; |
|
o |
the fact that Pieris may become obligated to pay Palvella a termination fee of $1 million in certain circumstances as further discussed under the section titled “The Merger Agreement” beginning on page 163 of this proxy statement/prospectus, which could potentially deter a potential acquirer from proposing an alternative transaction that may provide value to Pieris stockholders superior to that of the proposed transactions; |
|
o |
the potential for litigation relating to the proposed transactions and the associated costs, burden and inconvenience involved in defending those proceedings; and |
|
o |
the potential conflict of interest created by the fact that Pieris’ executive officers and directors have financial or other interests in the Merger that may be different from, or in addition to, those of other stockholders, as more fully described below in “Interests of the Pieris Directors and Executive Officers in the Merger”; and |
|
● |
various other risks associated with the combined company and the Merger, including those described in the sections titled “Risk Factors” beginning on page 25 and “Cautionary Statement Concerning Forward-Looking Statements and Market and Industry Data” beginning on page 116 of this proxy statement/prospectus. |
The foregoing information and factors considered by the Pieris board of directors are not intended to be exhaustive but are believed to include all of the material factors considered by the Pieris board of directors. In view of the wide variety of factors considered in connection with its evaluation of the Merger and the other transactions contemplated by the Merger Agreement and the complexity of these matters, the Pieris board of directors did not find it useful, and did not attempt, to quantify, rank or otherwise assign relative weights to these factors. In considering the factors described above, individual members of the Pieris board of directors may have given different weight to different factors. The Pieris board of directors conducted an overall analysis of the factors described above, including thorough discussions with, and questioning of, Pieris’ management team and legal and financial advisors, and considered the factors overall to be favorable to, and to support, its determination.
Palvella Reasons for the Merger
The following discussion sets forth material factors considered by the Palvella board of directors in reaching its determination to authorize the Merger Agreement and approve the Merger. However, it may not include all of the factors considered by the Palvella board of directors. In light of the number and wide variety of factors considered in connection with its evaluation of the Merger Agreement and the Merger, the Palvella board of directors did not consider it practicable to, and did not attempt to, quantify or otherwise assign relative weights to the specific factors it considered in reaching its determination. The Palvella board of directors viewed its position and determinations as being based on all of the information available and the factors presented to and considered by it.
In the course of reaching its decision to approve the Merger, the Palvella board of directors consulted with its senior management, financial advisors and legal counsel, reviewed a significant amount of information and considered a number of factors, including, among others:
The various factors the Palvella board of directors considered that weighed positively in favor of the proposal to adopt the Merger Agreement included, among others, the following (not necessarily in order of relative importance):
|
● |
the Merger will provide Palvella current stockholders with greater liquidity by owning publicly-traded stock; |
|
● |
the potential increased access to sources of capital and a broader range of investors to support the development of its product candidates following consummation of the Merger compared to if Palvella continued to operate as a privately held company; |
|
● |
the PIPE Financing will generate capital resources to fund the combined company; |
|
● |
the Palvella board of directors’ fiduciary duties to Palvella stockholders; |
|
● |
the Palvella board of directors’ belief that no alternatives to the Merger were reasonably likely to create greater value for Palvella stockholders, after reviewing the various financing and other strategic options to enhance stockholder value that were considered by the Palvella board of directors and the likelihood of achieving any alternative transaction compared to the likelihood of completing the Merger; |
|
● |
the availability of appraisal rights under the DGCL to holders of Palvella capital stock who comply with the required procedures under the DGCL, which allow such holders to seek appraisal of the fair value of their shares of Palvella capital stock as determined by the Delaware Court of Chancery; |
|
● |
the projected financial position, operations, management, operating plans and financial projections of the combined company, including the impact of the CVR Agreement; |
|
● |
the expected cash resources of the combined company (including the ability to support the combined company’s current and planned clinical trials and operations); |
|
● |
the terms and conditions of the Merger Agreement, including the following: |
|
o |
the determination that the expected relative percentage ownership of Pieris stockholders and Palvella stockholders in the combined company was appropriate, based on the Palvella board of directors’ judgment and assessment of the approximate valuations of Pieris (including the value of the net cash Pieris is expected to provide to the combined company) and Palvella; |
|
o |
the expectation that the Merger will be treated for U.S. federal income tax purposes as a “reorganization” within the meaning of Section 368(a) of the Code and/or as a non-taxable exchange of shares of Palvella common stock and Palvella preferred stock for shares of Pieris common stock within the meaning of Section 351(a) of the Code, with the result that in the Merger the Palvella stockholders will generally not recognize taxable gain or loss for U.S. federal income tax purposes, except with respect to cash received in lieu of a fractional share of Pieris common stock, as more fully described below under the caption “The Merger—Material U.S. Federal Income Tax Consequences of the Merger,” beginning on page 155 in this proxy statement/prospectus; |
|
o |
the limited number and nature of the conditions of the obligation of Pieris to consummate the Merger; |
|
o |
the conclusion of the Palvella board of directors that the potential termination fees of $1.0 million payable by Pieris to Palvella in certain circumstances, or of $2.0 million payable by Palvella to Pieris in certain other circumstances, and the circumstances when such fees may be payable, were reasonable; |
|
o |
the belief that the other terms of the Merger Agreement, including the parties’ representations, warranties and covenants, and the conditions to their respective obligations, were reasonable in light of the entire transaction; |
|
● |
the shares of Pieris common stock issued to Palvella stockholders will be registered on a Registration Statement on Form S-4 and will become freely tradable for Palvella stockholders who are not affiliates of Palvella and who are not parties to lock-up agreements; |
|
● |
the support agreements, pursuant to which certain directors, officers and stockholders of Palvella and Pieris, respectively, have agreed, solely in their capacity as stockholders of Palvella and Pieris, respectively, to vote all of their shares of Palvella capital stock in favor of the adoption or approval, respectively, of the Merger Agreement; |
|
● |
the ability to obtain a Nasdaq listing and the change of the combined organization’s name to Palvella Therapeutics, Inc. upon the closing of the Merger; and |
|
● |
the likelihood that the Merger will be consummated on a timely basis. |
The Palvella board of directors also considered a number of uncertainties and risks in its deliberations concerning the merger and the other transactions contemplated by the Merger Agreement, including the following (not necessarily in order of relative importance):
|
● |
the possibility that the Merger might not be completed and the potential adverse effect of the public announcement of the Merger on the reputation of Palvella and the ability of Palvella to obtain financing in the future in the event the Merger is not completed; |
|
● |
the risk that future sales of common stock by existing Pieris stockholders may cause the price of Pieris common stock to fall, thus reducing the potential value of Pieris common stock received by Palvella stockholders following the Merger; |
|
● |
the exchange ratio used to establish the number of shares of Pieris common stock to be issued to Palvella stockholders in the Merger is fixed, except for adjustments due to the parties’ respective cash balances and outstanding capital stock at closing, and thus the relative percentage ownership of Pieris stockholders and Palvella stockholders in the combined organization immediately following the completion of the Merger is similarly fixed; |
|
● |
the termination fee payable by Palvella to Pieris upon the occurrence of certain events, and the potential effect of such termination fee in deterring other potential acquirers from proposing an alternative transaction that may be more advantageous to Palvella stockholders; |
|
● |
the potential reduction of Pieris’ net cash prior to the closing of the Merger; |
|
● |
the possibility that Pieris could, under certain circumstances, consider unsolicited Acquisition Proposals if superior to the Merger or change its recommendation to approve the Merger upon certain events; |
|
● |
the possibility that the Merger might not be completed in a timely manner or at all; |
|
● |
the costs involved in connection with completing the Merger, the time and effort of Palvella senior management required to complete the Merger, the related disruptions or potential disruptions to Palvella’s business operations and future prospects, including its relationships with its employees, suppliers and partners and others that do business or may do business in the future with Palvella, and related administrative challenges associated with combining the companies; |
|
● |
the additional expenses and obligations to which Palvella’s business will be subject following the Merger that Palvella has not previously been subject to, and the operational changes to Palvella’s business, in each case that may result from being a public company; |
|
● |
the fact that the representations and warranties in the Merger Agreement do not survive the closing of the Merger and the potential risk of liabilities that may arise post-closing; and |
|
● |
various other risks associated with the combined company and the Merger, including the risks described in the section titled “Risk Factors” in this proxy statement/prospectus. |
The foregoing information is not intended to be exhaustive, but summarizes the material factors considered by the Palvella board of directors in its consideration of the merger agreement and the transactions contemplated. The Palvella board of directors concluded that the benefits, advantages and opportunities of a potential transaction outweighed the uncertainties and risks described above. After considering these and other factors, the Palvella board of directors unanimously approved the merger agreement, the merger and the other transactions contemplated by the merger agreement.
Opinion of Pieris’ Financial Advisor
Pieris engaged Stifel to act as financial advisor to Pieris in connection with the Merger. Pieris selected Stifel because Stifel is a nationally recognized investment banking firm with substantial experience in transactions similar to the proposed Merger. As part of Stifel’s engagement, the Pieris board of directors requested Stifel’s opinion, as investment bankers, as to the fairness, from a financial point of view and as of the date of such opinion, of the aggregate consideration to be paid by Pieris to the holders of Palvella capital stock in the Merger pursuant to the Merger Agreement. At a meeting of the Pieris board of directors held on July 23, 2024, Stifel delivered to the Pieris board of directors its opinion, which opinion was initially rendered verbally and confirmed in a written opinion, dated July 23, 2024, which is referred to as the Stifel Opinion, to the effect that, as of the date of the Stifel Opinion and based upon and subject to the factors, considerations, qualifications, limitations and assumptions set forth therein, the Aggregate Merger Consideration (as defined in the Stifel Opinion and which was assumed to be 6,919,206 shares of Pieris common stock as more fully described below) to be paid by Pieris to the holders of Palvella capital stock in the Merger pursuant to the Merger Agreement was fair to Pieris, from a financial point of view.
The full text of the Stifel Opinion, dated July 23, 2024, is attached as Annex C to this proxy statement/prospectus and is incorporated herein by reference. This summary of the Stifel Opinion contained in this proxy statement/prospectus is qualified in its entirety by reference to the full text of the Stifel Opinion. Pieris stockholders are urged to read the Stifel Opinion carefully and in its entirety for a discussion of the procedures followed, assumptions made, matters considered, and qualifications and limitations on the review undertaken by Stifel in connection with its opinion. The Stifel Opinion speaks only as of the date of the Stifel Opinion. The Stifel Opinion was for the information of, and was directed to, the Pieris board of directors (in its capacity as such) for its information and assistance in connection with its consideration of the financial terms of the Merger. The Stifel Opinion addressed only the fairness, from a financial point of view, to Pieris of the Aggregate Merger Consideration to be paid by Pieris to the holders of Palvella capital stock in the Merger. It did not address the underlying business decision of the Pieris board of directors or Pieris to proceed with or effect the Merger or constitute a recommendation to the Pieris board of directors in connection with the Merger or any other matter, and it does not constitute a recommendation to any shareholder of Pieris or any stockholder of any other entity as to how to vote in connection with the Merger or as to any other action that a shareholder or stockholder should take with respect to the Merger.
The issuance of the Stifel Opinion was approved by Stifel’s Fairness Opinion Committee. At the direction of Pieris and without independent verification, Stifel relied upon and assumed for purposes of its analyses and the Stifel Opinion, that, based on the unadjusted Public Company Valuation provided for in the Merger Agreement (reflecting the estimated closing net cash position of Pieris provided to Stifel by Pieris) and also Public Company Outstanding Shares (as defined in the Merger Agreement) of 1,529,509 shares of Pieris common stock if the Public Company Closing Price (as defined in the Merger Agreement) were equal to the closing price per share of Pieris common stock on July 19, 2024, the aggregate consideration (i.e., the Merger Partner Merger Shares (as defined in the Merger Agreement)) to be paid by Pieris in the Merger pursuant to the Merger Agreement will be 6,919,206 shares of Pieris common stock (which assumed number of shares was referred to in the Stifel Opinion as the Aggregate Merger Consideration), representing approximately 81.8966% of the equity of Pieris, on a fully-diluted and as-converted basis (before giving effect to the PIPE financing), at the Effective Time. In addition, at the direction of Pieris and without independent verification, Stifel relied upon and assumed that, in connection with the PIPE Financing, 4,370,270 additional shares of Pieris common stock will be issued and sold for $60,000,777 in cash and 1,376,556 additional shares of Pieris common stock will be issued upon conversion of the Palvella notes.
In rendering its opinion, Stifel, among other things:
|
(i) |
discussed the Merger and related matters with Pieris and Pieris’ counsel and reviewed the financial terms of the Merger contained in an execution version of the Merger Agreement, which was the then most recent version of the Merger Agreement provided to Stifel; |
|
(ii) |
reviewed the audited consolidated financial statements of Pieris contained in its Annual Reports on Form 10-K for the three years ended December 31, 2023 and unaudited consolidated financial statements of Pieris contained in its Quarterly Report on Form 10-Q for the quarter ended March 31, 2024; |
|
(iii) |
reviewed the audited financial statements of Palvella for the year ended December 31, 2022, unaudited financial statements of Palvella for the year ended December 31, 2023 and unaudited financial statements of Palvella for the quarter ended March 31, 2024; |
|
(iv) |
reviewed and discussed with Pieris’ management certain other publicly available information concerning Pieris; |
|
(v) |
reviewed certain non-publicly available information concerning Pieris and Palvella, including the estimated closing net cash position of Pieris provided by Pieris and internal financial analyses and forecasts for Palvella prepared by its management and reflecting the probabilities of technical success determined by Palvella’s management, and held discussions with Pieris’ and Palvella’s respective senior managements regarding recent developments; |
|
(vi) |
reviewed and analyzed certain publicly available financial and stock market data relating to selected public companies that Stifel deemed relevant to its analysis of Palvella; |
|
(vii) |
reviewed and analyzed certain publicly available information concerning the terms of selected initial public offerings, or “IPOs”, that Stifel considered relevant to its analysis of Palvella; |
|
(viii) |
participated in certain discussions and negotiations between representatives of Pieris and Palvella; |
|
(ix) |
reviewed the reported prices and trading activity of Pieris common stock; |
|
(x) |
considered the results of Pieris’ strategic review process publicly announced in July 2023, including Pieris’ efforts, with Stifel’s assistance, to solicit indications of interest from selected third parties with respect to a merger or other transaction with Pieris; |
|
(xi) |
conducted such other financial studies, analyses and investigations and considered such other information as Stifel deemed necessary or appropriate for purposes of the Stifel Opinion; and |
|
(xii) |
took into account Stifel’s assessment of general economic, market and financial conditions and Stifel’s experience in other transactions, as well as Stifel’s experience in securities valuations and knowledge of Pieris’ and Palvella’s industry generally. |
Pieris did not provide Stifel with any internal financial analyses or forecasts for Pieris (other than working capital adjustments and operating and Merger-related costs estimates and other cash forecasts used to calculate the estimated closing net cash position of Pieris provided by Pieris). For purposes of evaluating the Aggregate Merger Consideration, Stifel relied on the implied value of Pieris based on the estimated closing net cash position of Pieris provided by Pieris and did not perform any financial analyses of Pieris or its non-cash assets, including, without limitation, the legacy programs and assets to which the CVRs relate. At the direction of Pieris, Stifel relied upon and assumed, without independent verification, that the value of the CVRs will be substantially equivalent to the value of the existing business of Pieris. Furthermore, Stifel did not perform any separate financial analysis of any particular class of Palvella capital stock or the Palvella notes.
In rendering the Stifel Opinion, Stifel relied upon and assumed, without independent verification, the accuracy and completeness of all of the financial and other information that was provided to Stifel by or on behalf of Pieris or Palvella, or that was otherwise reviewed by Stifel, and Stifel has not assumed any responsibility for independently verifying any of such information. With respect to the financial forecasts (including probabilities of technical success reflected therein) supplied to Stifel by Palvella, Stifel assumed, at the direction of Pieris, that they were reasonably prepared on the basis reflecting the best currently available estimates and judgments of the management of Palvella as to the future operating and financial performance of Palvella and that they provided a reasonable basis upon which Stifel could form the Stifel Opinion. Such forecasts were not prepared with the expectation of public disclosure. All such projected financial information was based on numerous variables and assumptions that are inherently uncertain, including, without limitation, factors related to general economic and competitive conditions and the widespread disruption, extraordinary uncertainty and unusual volatility arising from global tensions and political unrest, economic uncertainty, inflation, prolonged higher interest rates, the COVID-19 pandemic, and also including, without limitation, the effect of evolving governmental interventions and non-interventions. Accordingly, actual results could vary significantly from those set forth in such projected financial information. Stifel relied on this projected information without independent verification or analyses and does not in any respect assume any responsibility for the accuracy or completeness thereof.
Stifel also assumed that there were no material changes in the assets, liabilities, financial condition, results of operations, business or prospects of either Pieris or Palvella since the date of the last financial statements of each company made available to Stifel. Stifel did not make or obtain any independent evaluation, appraisal or physical inspection of either Pieris’ or Palvella’ assets or liabilities, nor was Stifel furnished with any such evaluation or appraisal. Estimates of values of companies and assets did not purport to be appraisals or necessarily reflect the prices at which companies or assets may actually be sold. Such estimates were inherently subject to uncertainty and should not be taken as Stifel’s view of the actual value of any companies or assets.
Stifel assumed, with the consent of Pieris, that there are no factors that would delay or subject to any adverse conditions any necessary regulatory or governmental approval and that all conditions to the Merger will be satisfied and not waived. In addition, Stifel assumed that the definitive Merger Agreement will not differ materially from the execution version Stifel reviewed. Stifel also assumed that the representations and warranties of the parties set forth in the Merger Agreement were and will be true and correct and that the Merger and related transactions (including, without limitation, the PIPE Financing and the issuance of the CVRs) will be consummated substantially on the terms and conditions described in the Merger Agreement, without any waiver of material terms or conditions by Pieris or any other party and without any anti-dilution or other adjustment to the Aggregate Merger Consideration (whether due to changes in the Public Company Net Cash (as defined in the Merger Agreement) or otherwise), and that obtaining any necessary regulatory approvals or satisfying any other conditions for consummation of the Merger and related transactions will not have an adverse effect on Pieris, Palvella or the Merger. Stifel assumed that the Merger and related transactions will be consummated in a manner that complies with the applicable provisions of the Securities Act, the Exchange Act, and all other applicable federal and state statutes, rules and regulations. Stifel further assumed that Pieris had relied upon the advice of its counsel, independent accountants and other advisors (other than Stifel) as to all legal, financial reporting, tax, accounting and regulatory matters with respect to Pieris, Palvella, the Merger and related transactions and the Merger Agreement.
The Stifel Opinion was limited to whether the Aggregate Merger Consideration to be paid by Pieris to the holders of Palvella capital stock was fair to Pieris, from a financial point of view, and did not address any other terms, aspects or implications of the Merger or any related transaction (including the PIPE Financing, the issuance of the CVRs and any potential sale by Pieris of some or all of its assets at or prior to the closing of the Merger, or a “Legacy Asset Disposition”, including, without limitation, the form or structure of the Merger or any related transaction, the allocation of the Aggregate Merger Consideration among the different classes of Palvella capital stock, the prices at which shares of Pieris common stock will be issued in the PIPE Financing or assets of Pieris will be sold in any Legacy Asset Disposition, any consequences of the Merger on Pieris, its shareholders, creditors or otherwise, or any terms, aspects or implications of the CVRs, the related CVR Agreements or any lock-up, voting, support, stockholder or other agreements, arrangements or understandings contemplated or entered into in connection with the Merger or any related transaction or otherwise. The Stifel Opinion also did not consider, address or include: (i) any other strategic alternatives then (or which had been or might be) contemplated by the Pieris board of directors or Pieris; (ii) the legal, tax or accounting consequences of the Merger or any related transaction on Pieris (including, without limitation, whether or not the Merger will qualify as a tax-free reorganization pursuant to Section 368 of the Code); (iii) the fairness of the amount or nature of any compensation to any of Pieris’ officers, directors or employees, or class of such persons, relative to the compensation to the holders of Pieris’ securities or relative to the Aggregate Merger Consideration; (iv) the effect of the Merger or any related transaction on, or the fairness of the consideration to be received by, holders of any class of securities of Pieris, Palvella or any other party to any transaction contemplated by the Merger Agreement; or (v) any advice or opinions provided by any other advisor to Pieris or Palvella. Furthermore, Stifel did not express any opinion as to the Merger Partner Valuation, the Public Company Valuation, the actual value of Pieris common stock and the CVRs to be issued in connection with the Merger, the PIPE Financing and the issuance of the CVRs, or the prices, trading range or volume at which Pieris common stock, the CVRs or any other securities of Pieris will trade following public announcement or consummation of the Merger or any related transaction.
The Stifel Opinion is necessarily based on economic, market, financial and other conditions as they existed on, and on the information made available to Stifel by or on behalf of Pieris, Palvella or their respective advisors, or information otherwise reviewed by Stifel, as of the date of the Stifel Opinion. It is understood that subsequent developments may affect the conclusion reached in the Stifel Opinion and that Stifel does not have any obligation to update, revise or reaffirm the Stifel Opinion. Further, as the Pieris board of directors was aware, the credit, financial and stock markets have been experiencing unusual volatility and Stifel expressed no opinion or view as to any potential effects of such volatility on Pieris, Palvella or the Merger. In addition, Stifel expressed no view or opinion as to any changes after the date of the Stifel Opinion that would result in the actual number of Merger Partner Merger Shares pursuant to the Merger Agreement being different from the amount of the Aggregate Merger Consideration that Stifel was directed to assume for purposes of its analyses and the Stifel Opinion.
The Stifel Opinion was for the information of, and directed to, the Pieris board of directors, in its capacity as such, for its information and assistance in connection with its consideration of the financial terms of the Merger. The Stifel Opinion did not constitute a recommendation to the Pieris board of directors as to how the Pieris board of directors should vote on the Merger or to any shareholder of Pieris or stockholder of Palvella as to how any such shareholder or stockholder should vote at any shareholders’ or stockholders’ meeting at which the Merger is considered, or whether or not any shareholder or stockholder should enter into a voting, shareholders’, or affiliates’ agreement with respect to the Merger, or exercise any dissenters’ or appraisal rights that may be available to such shareholder or stockholder. In addition, the Stifel Opinion did not compare the relative merits of the Merger with any other alternative transactions or business strategies which may have been available to Pieris and did not address the underlying business decision of the Pieris board of directors or Pieris to proceed with or effect the Merger.
Stifel is not a legal, tax, regulatory or bankruptcy advisor. Stifel did not consider any potential legislative or regulatory changes currently being considered or recently enacted by the United States Congress, the various federal banking agencies, the SEC, or any other regulatory bodies, or any changes in accounting methods or GAAP that may be adopted by the SEC or the Financial Accounting Standards Board, or any changes in regulatory accounting principles that may be adopted by any or all of the federal banking agencies. The Stifel Opinion was not a solvency opinion and did not in any way address the solvency or financial condition of Pieris, Palvella or any other party.
The Stifel Opinion was provided to the Pieris board of directors in connection with its evaluation of the Merger and was only one of many factors considered by the Pieris board of directors in evaluating the Merger. Neither the Stifel Opinion nor its analyses were determinative of the Aggregate Merger Consideration or of the views of the Pieris board of directors or Pieris management with respect to the Merger. The Aggregate Merger Consideration was determined through negotiation between Pieris and Palvella, and the decision for Pieris to enter into the Merger was solely that of the Pieris board of directors.
Summary of Material Financial Analyses
The following is a summary of the material financial analyses performed by Stifel in arriving at the Stifel Opinion. These summaries of financial analyses alone do not constitute a complete description of the financial analyses Stifel employed in reaching its conclusions. None of the analyses performed by Stifel were assigned a greater significance by Stifel than any other, nor does the order of analyses described represent the relative importance or weight given to those analyses by Stifel. Some of the summaries of the financial analyses performed by Stifel include information presented in tabular format. In order to understand the financial analyses performed by Stifel more fully, you should read the tables together with the text of each summary. The tables alone do not constitute a complete description of Stifel’s financial analyses, including the methodologies and assumptions underlying the analyses, and if viewed in isolation could create a misleading or incomplete view of the financial analyses performed by Stifel. The summary data set forth below does not represent and should not be viewed by anyone as constituting conclusions reached by Stifel with respect to any of the analyses performed by it in connection with the Stifel Opinion. Rather, Stifel made its determination as to the fairness, from a financial point of view, to Pieris of the Aggregate Merger Consideration to be paid by Pieris to the holders of Palvella capital stock in the Merger on the basis of its experience and professional judgment after considering the results of all of the analyses performed. Accordingly, the data presented and the corresponding imputed ranges of values for Palvella and corresponding imputed ranges of ownership percentages of the combined company for Pieris should be considered as a whole and in the context of the full narrative description of all of the financial analyses set forth in the following pages, including the assumptions underlying these analyses.
Except as otherwise noted, the information utilized by Stifel in its analyses, to the extent that it was based on market data, was based on market data as it existed on or before July 19, 2024 and is not necessarily indicative of current market conditions. The analyses described below do not purport to be indicative of actual future results, or to reflect the prices at which any securities may trade in the public markets, which may vary depending upon various factors, including changes in interest rates, dividend rates, market conditions, economic conditions and other factors that influence the price of securities.
For purposes of the financial analyses described below, Stifel utilized an assumed Public Company Allocation Percentage for purposes of the Merger Agreement of 18.1034%, based on the Merger Partner Valuation provided for in the Merger Agreement of $95,000,000 and the unadjusted Public Company Valuation provided for in the Merger Agreement (reflecting the estimated closing net cash position of Pieris provided by Pieris) of $21,000,000. In addition to the financial analyses described below, Stifel reviewed with the Pieris board of directors for informational purposes, among other things, the implied ownership percentage of the combined company on a fully diluted basis (using the treasury stock method based on the closing price per share of Pieris common stock on July 19, 2024), also giving effect to an assumed concurrent financing of 4,370,270 additional shares of Pieris common stock issued and sold for $60,000,777 in cash and 1,376,556 additional shares of Pieris common stock issued upon conversion of the Palvella convertible notes, of 10.9770% attributable to Public Company Outstanding Shares (as defined in the Merger Agreement).
Selected Publicly-Traded Companies Analysis
Stifel reviewed publicly available financial and stock market information of six selected publicly-traded biotechnology companies that Stifel deemed to be relevant to Palvella based on their business profiles because each of their respective most advanced value-generating asset has a dermatology or rare focus and finished Phase 2 trial, is Phase 3 ready, or currently is in Phase 3 (but without having yet received final Phase 3 data). Oncology companies and platform companies were excluded from the selected publicly-traded companies given that these groups generally have a market performance that differs from that of biotechnology companies like Palvella, which is focused on therapeutic areas other than oncology and is a single product company. The group of selected publicly-traded companies reviewed were as follows (shown in descending order of market capitalization):
Company Name
Viridian Therapeutics, Inc.
Pharvaris N.V.
Pliant Therapeutics, Inc.
Astria Therapeutics, Inc.
Fulcrum Therapeutics, Inc.
Inozyme Pharma, Inc.
For each of the selected publicly-traded companies, Stifel calculated the enterprise value, which Stifel defined as fully-diluted equity value using the treasury stock method, plus debt, preferred stock and minority interests, less cash and cash equivalents. Financial data for the selected publicly-traded companies was based on publicly available data obtained from SEC filings and other data sources and closing stock prices on July 19, 2024. Stifel reviewed the high, mean, median and low implied enterprise values of the selected publicly-traded companies.
The results of this selected publicly-traded companies analysis are summarized below:
| Selected Publicly-Traded Companies Analysis | ||||
|
High |
Mean |
Median |
Low |
|
|
Implied Enterprise Value ($mm) |
$642 |
$387 |
$317 |
$204 |
Using a selected range of implied enterprise values reflecting the median and mean implied enterprise values of the selected publicly-traded companies, this analysis yielded a range of implied enterprise values for Palvella of approximately $317 million to $387 million from which Stifel calculated a range of implied equity values for Palvella using cash and debt data for Palvella as of June 30, 2024 provided by the management of Palvella. This analysis indicated an implied equity value reference range for Palvella of approximately $322 million to $391 million.
Based on the implied equity value reference range for Palvella indicated in the above selected publicly-traded companies analysis and the estimated closing net cash position of Pieris provided by Pieris, Stifel calculated a range of implied ownership percentages of the combined company for Pieris using (i) the estimated closing net cash position of Pieris as a percentage of the sum of the estimated closing net cash position of Pieris plus the low value of the implied equity value reference range for Palvella, and (ii) the estimated closing net cash position of Pieris as a percentage of the sum of the estimated closing net cash position of Pieris plus the high value of the implied equity value reference range for Palvella. The resulting implied ownership percentage reference range of the combined company for Pieris was 2.7% to 3.3%, as compared to the assumed Public Company Allocation Percentage for purposes of the Merger Agreement of 18.1034%.
No company utilized in this selected publicly-traded companies analysis is identical to Palvella. In performing this selected publicly-traded companies analysis, Stifel made judgments and assumptions with regard to industry performance, general business, economic, market and financial conditions and other matters, such as the impact of competition, industry growth and the absence of any adverse material change in the financial condition and prospects of Palvella or the selected publicly-traded companies or the industry or in the financial markets in general.
Selected Precedent IPO Analysis
Stifel reviewed publicly available information concerning the terms of six selected biotechnology IPOs since 2021 that Stifel considered to be relevant to Palvella based on the business profiles of the respective issuers because each of their respective lead assets was considered the main value driver and finished Phase 2 trial, was Phase 3 ready, or was in Phase 3 at the time of the relevant IPO (but without having yet received final Phase 3 data). IPOs of oncology companies and platform companies were excluded from the selected IPOs given that these groups generally have a market performance that differs from that of biotechnology companies like Palvella, which is focused on therapeutic areas other than oncology and is a single product company. The group of selected IPOs reviewed were as follows:
|
IPO Date |
|
Issuer Name |
|
6/27/2024 10/20/2023 4/28/2022 4/8/2021 2/5/2021 2/3/2021 |
Alumis, Inc. Abivax SA Belite Bio, Inc VectivBio Holding AG Angion Biomedica Corp. Landos Biopharma |
For each issuer of the selected IPOs, Stifel calculated, among other things, the implied pre-IPO fully-diluted equity value using the treasury stock method based on the IPO price. Stifel reviewed the high, mean, median and low implied pre-IPO fully-diluted equity value of the issuers of the selected IPOs.
The results of this selected precedent IPO analysis are summarized below:
| Selected Precedent IPO Analysis | ||||
|
High |
Mean |
Median |
Low |
|
|
Implied Pre-IPO Fully-diluted Equity Value ($mm) |
$676 |
$450 |
$484 |
$111 |
Stifel then used a selected range of implied pre-IPO fully-diluted equity values reflecting the mean and median implied pre-IPO fully-diluted equity values of the issuers of the selected IPOs to derive a range of implied equity values for Palvella. This analysis indicated an implied equity value reference range for Palvella of approximately $450 million to $484 million.
Based on the implied equity value reference range for Palvella indicated in the above selected precedent IPO analysis and the estimated closing net cash position of Pieris provided by Pieris, Stifel calculated a range of implied ownership percentages of the combined company for Pieris using (i) the estimated closing net cash position of Pieris as a percentage of the sum of the estimated closing net cash position of Pieris plus the low value of the implied equity value reference range for Palvella, and (ii) the estimated closing net cash position of Pieris as a percentage of the sum of the estimated closing net cash position of Pieris plus the high value of the implied equity value reference range for Palvella. The resulting implied ownership percentage reference range of the combined company for Pieris was 2.2% to 2.4%, as compared to the assumed Public Company Allocation Percentage for purposes of the Merger Agreement of 18.1034%.
No issuer in the selected IPOs is identical to Palvella. In performing this selected precedent IPOs analysis, Stifel made judgments and assumptions with regard to industry performance, general business, economic, market and financial conditions, and other matters, such as the impact of competition, industry growth and the absence of any adverse material change in the financial condition and prospects of Palvella or the issuers of the selected IPOs or the industry or in the financial markets in general.
Discounted Cash Flow Analysis
Stifel used financial forecasts of Palvella, as prepared by the management of Palvella and reflecting the probabilities of technical success determined by the management of Palvella, as directed for Stifel’s use by Pieris management, to perform a discounted cash flow analysis, which analyzes a company’s future cash flow projections by discounting them to arrive at the net present value of these cash flows.
Stifel calculated a range of estimated terminal values of the projected free cash flows of Palvella by applying a range of perpetuity growth rates of negative 100% to negative 90% to probability-adjusted projected calendar year 2040 free cash flow. Stifel then discounted the probability-adjusted projected free cash flows of Palvella for the second half of calendar year 2024 through calendar year 2040 and estimated terminal value range to present values using discount rates ranging from 12% to 14% (based on an analysis of weighted average cost of capital, using the capital asset pricing model, considering Palvella’s company-specific circumstances and Stifel’s judgment). This analysis yielded a range of implied enterprise values for Palvella of $411.8 million to $503.4 million from which Stifel calculated a range of implied equity values for Palvella using cash and debt data for Palvella as of June 30, 2024 provided by the management of Palvella. This analysis indicated an implied equity value reference range for Palvella of approximately $416 million to $508 million.
Based on the implied equity value reference range for Palvella indicated in the above discounted cash flow analysis and the estimated closing net cash position of Pieris provided by Pieris, Stifel calculated a range of implied ownership percentages of the combined company for Pieris using (i) the estimated closing net cash position of Pieris as a percentage of the sum of the estimated closing net cash position of Pieris plus the low value of the implied equity value reference range for Palvella, and (ii) the estimated closing net cash position of Pieris as a percentage of the sum of the estimated closing net cash position of Pieris plus the high value of the implied equity value reference range for Palvella. The resulting implied ownership percentage reference range of the combined company for Pieris was 2.1% to 2.6%, as compared to the assumed Public Company Allocation Percentage for purposes of the Merger Agreement of 18.1034%.
Miscellaneous
The preparation of a fairness opinion is a complex process and is not necessarily susceptible to a partial analysis or summary description. In arriving at the Stifel Opinion, Stifel considered the results of all of its analyses as a whole and did not attribute any particular weight to any analysis or factor considered by it. Stifel believes that the summary provided and the analyses described above must be considered as a whole and that selecting portions of these analyses, without considering all of them, would create an incomplete view of the process underlying Stifel’s analyses and the Stifel Opinion; therefore, the ranges of valuations and relative valuations resulting from any particular analysis described above should not be taken to be Stifel’s view of the actual valuation of the company or its relative valuation.
Pieris paid Stifel a fee, which is referred to in this proxy statement/prospectus as the opinion fee, of $500,000 for providing the Stifel Opinion to the Pieris board of directors (not contingent upon the consummation of the Merger). Stifel will receive an additional fee of approximately $1,500,000 contingent upon the consummation of the Merger. In addition, Pieris agreed to reimburse Stifel for certain expenses in connection with its engagement, subject to certain limitations, and to indemnify Stifel for certain liabilities arising out of its engagement. There are no material relationships that existed during the two years prior to the date of the Stifel Opinion or that as of such date were mutually understood to be contemplated in which any compensation was received or is intended to be received as a result of the relationship between Stifel and Pieris or Palvella. Stifel may seek to provide investment banking services to Pieris or Palvella or their affiliates in the future, for which Stifel would seek customary compensation. In the ordinary course of its business, Stifel, its affiliates and their respective clients may transact in the securities of Pieris and may at any time hold a long or short position in such securities.
Certain Unaudited Financial Projections of Palvella
Neither Pieris nor Palvella, as a matter of course, makes public long-term Projections or internal projections as to future performance, revenues, earnings or other financial or operating results due to, among other reasons, the uncertainty of the underlying assumptions and estimates. However, in connection with the evaluation of the Merger, certain unaudited internal financial Projections relating to the business, operations, earnings, cash flow, assets, liabilities and prospects of Palvella, or the “Palvella Projections”, were prepared by management of Palvella.
The Palvella Projections were provided to and considered by the Pieris board of directors in connection with its evaluation of the Merger and to Stifel for use in its financial analyses and for purposes of the Stifel Opinion (as summarized under the section entitled “—Opinion of Pieris’ Financial Advisor”). The Palvella Projections (on a probability-adjusted basis to reflect the probabilities of technical success determined by Palvella’s management) were the only financial projections relied upon by Stifel in rendering the Stifel Opinion.
In considering the reasonableness of Palvella Projections, and understanding the inherent uncertainty to short- and long-range forecasting, Pieris’ management performed a robust assessment that included multiple rounds of review related to key inputs and the accuracy of data that were based on currently available estimates and judgments of the management of Palvella. The possibility that Palvella's product candidate may not successfully complete clinical trials in microcystic lymphatic malformations was reflected in the probability of technical success included in the financial projections. These probabilities were determined by Palvella’s management and reviewed by Pieris’ management and advisors and, in turn, reviewed and considered by the Pieris Board.
The inclusion of the Palvella Projections should not be regarded as an indication that any of Pieris, Palvella, their respective affiliates, officers, directors, advisors, other representatives, or any other recipient of the Palvella Projections considered, or now considers, such Palvella Projection to be necessarily predictive of actual future performance or events, or that they should be construed as financial guidance, and Pieris stockholders and Palvella stockholders are cautioned not to place undue reliance on the Palvella Projections. Accordingly, the Palvella Projections are not included to influence any person’s views on the Merger and are summarized in this proxy statement/prospectus solely to provide access to information that was provided to the Pieris board of directors and to Stifel in connection with the Merger.
The Palvella Projections were prepared solely for internal use and are subjective in many respects. Material assumptions that are underlying the Palvella Projections for QTORIN rapamycin for microcystic LM include:
|
● |
the Palvella revenue projections begin in 2027 which is the current estimated year of approval for the microcystic LM indication in the US. Palvella did not assume any ex-US revenues as Palvella is not currently conducting any trial outside the US. Palvella also provided revenue projections for cutaneous venous malformations, or CVM, starting in 2029 in the US only. However, Pieris fully disregarded the CVM indication in connection with the evaluation of the Merger given the earlier stage of development of the CVM indication, that Palvella had not run a clinical trial for QTORIN rapamycin in the CVM indication, and that Palvella's management's assumptions were not as fully developed for the CVM indication as compared to the microcystic LM indication; |
|
● |
the revenue model for QTORIN rapamycin for the treatment of microcystic LM revenue is US only. The revenue projections assumed reaching peak penetration within 5 years of launch. After year five, the model assumes mid-single digit growth which ultimately declines to low single digit growth by the end of the forecast period; |
|
● |
the projections did not factor in the possibility of FDA approval of competitive products; |
| ● |
Palvella arrived at the probability of regulatory approval for QTORIN rapamycin for the treatment of microcystic LM based on a research report jointly compiled by three organizations, BIO, QLS Advisors, and Informa UK Ltd, titled “Clinical Development Success Rates and Contributing Factors 2011–2020” and was published in February of 2021. This research report included the therapeutic area of dermatology into a therapeutic area group titled “Others” and provides success rates for each phase of development. The report finds that 60.0% of Phase 3 trials that are categorized in the "Others" category successfully transition to the NDA/BLA phase where then 88.4% receive approval. Given the probabilities of approval in the “Others” category represent an average for the class of therapeutic areas, Palvella adjusted the ultimate probability of regulatory approval upwards for QTORIN rapamycin for the treatment of microcystic LM based on the phase 2 clinical study results, safety profile, and Breakthrough Therapy Designation suggesting greater likelihood of approval compared to the average. Specifically, based on this report Palvella assumed the probability of regulatory approval, which includes a successful Phase 3 trial followed by NDA approval, to be 65.0% which is above the "Others" category average. Palvella believes this upward adjustment to the probability of success is reasonable based on multiple factors. First, the February 2021 report cited provided an analysis of probability of success comparing 'Rare Disease' and 'All Disease'. Thus, this suggests QTORIN rapamycin for the treatment of microcystic LM, a rare disease, to have an incrementally higher probability of success as compared to all disease. Here it was reported 60.4% of rare disease trials successfully completed Phase 3 and 93.6% gained regulatory approval, as compared with 57.8% and 90.6%, respectively, for 'All Disease'. Second, it was reported non-NME drugs see 67.5% probability of Phase 3 success as compared to 50.6% for NME drugs. Given that rapamycin has already been approved in other formulations, this supports the rationale for the higher probability of success rate. In addition, a report published in November 2023 in the European Journal of Health Economics titled "Special FDA designations for drug development: orphan, fast track, accelerated approval, priority review, and breakthrough therapy" suggests clinical studies with special FDA designation, including Breakthrough Designation, experience higher clinical trial success rates. Despite all of this supporting evidence, drug development is inherently risky and this historical data represents averages and may not be indicative of future results; |
|
● |
clinical and regulatory timelines based on the current stage of Palvella’ s program; |
|
● |
Breakthrough Therapy Designation, Fast Track Designation, and Orphan Drug Designation from the FDA for QTORIN rapamycin for the treatment of microcystic LM; |
|
● |
net sales commence in the same year as the regulatory approval is obtained; |
|
● |
net sales and operating income assumptions were based on industry data, industry research and management analysis; and |
|
● |
(i) estimates of microcystic LM U.S. prevalence: >30,000 diagnosed patients; (ii) estimates of market growth rates were based on patient population growth rates; (iii) estimates of pricing for QTORIN rapamycin for the treatment of microcystic LM were derived from the pricing of similar orphan therapies; (iv) price increases, if any, are consistent with industry standards and historical targeted inflation rates; and (v) market acceptance and patient compliance rates are based on currently approved products. |
While presented with numeric specificity, the Palvella Projections reflect numerous estimates and assumptions made at the time the Palvella Projections were prepared that are inherently uncertain and many of which are beyond the control of Palvella’s management. Modeling and projecting the future development and commercialization of product candidates by an emerging biotechnology company is a highly speculative endeavor. Further, given that the Palvella Projections cover multiple years, by their nature, they become subject to greater uncertainty with each successive year beyond their preparation. The Palvella Projections beyond year five are based on the assumptions set forth below and among other things, ordinary and customary inflationary increases. Product and operating costs beyond year 5 reflect straight line growth assumptions. The revenue model for QTORIN rapamycin for the treatment of microcystic LM revenue is US only, patient-based model with growth rates in patients-on-therapy after year five that trend from the mid-single digits to the low single digits by the end of the forecast period. The Palvella Projections are subject to various risks, including, among others, the ability of Palvella to successfully develop and commercialize its product candidates, the effect of future regulatory or legislative actions on Palvella and the industry in which it operates, the potential impact of the announcement or completion of the Merger on relationships with customers, providers, vendors, competitors, management and other employees, changes in the general economic environment, or social or political conditions, that could affect Palvella’s business, potential liability resulting from pending or future litigation, and the uncertainties, costs and risks involved in Palvella’s operations. As a result, there can be no assurance that any of the Palvella Projections accurately reflect future trends or accurately estimate the future market for Palvella’s product or product candidates. There also can be no assurance that Palvella will obtain the regulatory approvals necessary for the commercialization of its products or product candidates, or that Palvella’s competitors will not commercialize products that are safer, more effective, or more successfully marketed and sold than any product that Palvella may market or commercialize.
None of Pieris or Palvella or any of their respective affiliates, officers, directors, advisors or other representatives has made, makes or is authorized in the future to make any representation to any Pieris stockholder or Palvella stockholder or other person regarding Palvella’s ultimate performance compared to the information contained in the Palvella Projections or that the Palvella Projections will be achieved. The inclusion of the Palvella Projections herein should not be deemed an admission or representation by Pieris, Palvella, their respective affiliates, officers, directors, advisors or other representatives or any other person that it is viewed as material information, particularly in light of the inherent risks and uncertainties associated with such Projections. The summary of the Palvella Projections included below is not being included in this proxy statement/prospectus in order to influence the decision of any Pieris stockholder or Palvella stockholder or to induce any Palvella stockholder to vote in favor of any of the proposals at the Pieris special meeting or to take any other action relating to the Merger.
The Palvella Projections reflect both assumptions as to certain business decisions that are subject to change and, in many respects, personal judgment, and thus are susceptible to multiple interpretations and, in the ordinary course, would be expected to undergo periodic revisions based on actual experience and business developments. None of Pieris, Palvella or their respective affiliates, officers, directors, advisors or other representatives can give assurance that the Palvella Projections and the underlying estimates and assumptions will be realized. The Palvella Projections constitute “forward-looking statements” and actual results may differ materially and adversely from those set forth below.
Neither Pieris nor Palvella, as a matter of course, make public projections as to future sales, earnings or other financial results. As such, the Palvella Projections were not prepared with a view toward public disclosure. The Palvella Projections do not take into account any circumstances or events occurring after the date they were prepared and neither Pieris nor Palvella can give assurance that, had the Palvella Projections been prepared either as of the date of the Merger Agreement or as of the date of this proxy statement/prospectus, similar estimates and assumptions would be used. The Palvella Projections do not take into account all of the possible financial and other effects of the Merger on Palvella, the effect on Palvella of any business or strategic decision or action that has been or will be taken as a result of the Merger Agreement having been executed, or the effect of any business or strategic decisions or actions that would likely have been taken if the Merger Agreement had not been executed, but which were instead altered, accelerated, postponed or not taken in anticipation of the Merger. Further, the Palvella Projections do not take into account the effect of any possible failure of the Merger to occur.
The Palvella Projections were not prepared with a view toward compliance with GAAP, published guidelines of the SEC or the guidelines established by the American Institute of Certified Public Accountants, or the “AICPA” for preparation or presentation of prospective financial information. Ernst & Young LLP has not audited, reviewed, examined, compiled or applied agreed- upon procedures with respect to the Palvella Projections and, accordingly, Ernst & Young LLP does not express an opinion or any other form of assurance on such information or its achievability, and assumes no responsibility for, and disclaims any association with, the prospective financial information with respect thereto. The report of Ernst & Young LLP which is included in this proxy statement/prospectus relates to historical financial information of Palvella, and such report does not extend to the Palvella Projections and should not be read to do so.
The Palvella Projections include financial measures, including net sales, gross profit and EBIT. These projections are based on a variety of sources including primary and secondary market research, scientific literature, clinical study data, and the experience of management and consultants. These financial measures should not be considered in isolation from, or as a substitute for, financial information presented as non-GAAP, and these financial measures may not be comparable to one another or to similarly titled measures used by other companies. We are not presenting a reconciliation of the financial measures included in the Palvella Projections to the relevant GAAP financial measures in this proxy statement/prospectus and we undertake no obligation to update or otherwise revise or reconcile any of the Palvella Projections to reflect circumstances existing after the date the Palvella Projections were generated or to reflect the occurrence of future events, except as otherwise required by law.
In light of the foregoing, and considering that the Pieris special meeting will be held a substantial amount of time after the Palvella Projections were prepared, as well as the uncertainties inherent in any forecasted information, Pieris Stockholders and Palvella Stockholders are cautioned not to place undue reliance on such information, and each of Pieris and Palvella caution you that the Palvella Projections should be evaluated, if at all, in conjunction with the historical financial statements and other information regarding Palvella contained elsewhere in this proxy statement/prospectus. You are urged to read the sections entitled “Cautionary Statement Concerning Forward-Looking Statements” and “Risk Factors” of this proxy statement/prospectus, for additional information regarding the risks inherent in forward-looking information such as the Palvella Projections.
| ($ in mm) |
2024E
|
2025E
|
2026E
|
2027E
|
2028E
|
2029E
|
2030E
|
2031E
|
2032E
|
2033E
|
2034E
|
2035E
|
2036E
|
2037E
|
2038E
|
| Total Net Sales | - | - | - |
$3.0
|
$38.0
|
$75.2
|
$145.3
|
$234.7
|
$305.2
|
$357.6
|
$406.1
|
$452.2
|
$495.5
|
$536.1
|
$574.0
|
| Gross Profit | - | - | - |
$2.7
|
$34.2
|
$67.1
|
$128.6
|
$206.8
|
$268.1
|
$313.3
|
$354.7
|
$393.9
|
$430.2
|
$463.9
|
$494.9
|
| Gross Margin (%) | - | - | - |
90%
|
90%
|
89%
|
89%
|
88%
|
88%
|
88%
|
87%
|
87%
|
87%
|
87%
|
86%
|
| EBIT | ($10.1) | ($14.0) | ($14.7) | ($27.5) | ($0.3) | $33.8 | $92.0 | $166.5 | $223.8 | $264.5 | $301.1 | $334.9 | $367.7 | $398.6 | $426.7 |
Note: The Palvella Projections through 2038 shown in the table above include projections for QTORIN rapamycin for microcystic LM only and are probability-adjusted to reflect the probabilities of technical success determined by Palvella’s management. Palvella's management believes projections for 2039 and 2040 are substantially more uncertain than the Palvella Projections through 2038 and therefore were not included. Palvella owns issued patents in the United States and pending applications in the United States directed to anhydrous gel formulations of rapamycin and methods of using the same to treat certain skin disorders, including microcystic LM that naturally expire in 2038. As such, the shown Palvella Projections are through the end of calendar year 2038 to align with the expected period of patent protection.
Interests of Pieris Directors and Executive Officers in the Merger
In considering the recommendation of the Pieris board of directors with respect to the Merger, holders of shares of Pieris common stock should be aware that Pieris’ executive officers and directors may have interests in the Merger that may be different from, or in addition to, those of Pieris stockholders generally. These interests may create potential conflicts of interest. The Pieris board of directors was aware of these potential conflicts of interest and considered them, among other matters, in reaching its decision to approve the Merger and to recommend that Pieris stockholders vote in favor of the proposals.
Severance Benefits.
In connection with Pieris’ initial hiring of Mr. Stephen S. Yoder, Chief Executive Officer and President, Pieris entered into an employment agreement with him dated December 17, 2014, or the “Yoder Employment Agreement”. For more information on the Yoder Employment Agreement, please see the section titled “Pieris Executive and Director Compensation—Employment and Severance Arrangements” elsewhere in this proxy statement/prospectus.
In the event that Mr. Yoder’s employment is terminated by Pieris without cause or by Mr. Yoder with good reason during a change in control period, as defined in the Yoder Employment Agreement, Mr. Yoder will be entitled, subject to his execution and nonrevocation of a release of claims in Pieris’ favor and his continued compliance with certain restrictive covenants, to the extent applicable, to (i) receive twelve months of his then-current annual base salary, payable as a lump sum on the first regular payroll date following the effective date of his release of claims, (ii) his target bonus for the year, payable as a lump sum, and (iii) reimbursement of Consolidated Omnibus Budget Reconciliation Act, or “COBRA”, premiums for health benefit coverage for a period of up to twelve months commencing on the first day of the first full calendar month following the effective date of his release of claims. In addition, under the terms of the Yoder Employment Agreement or the applicable award agreements, Mr. Yoder will be entitled to full acceleration of vesting and lapsing of any forfeiture restrictions on all his outstanding equity awards. Under the Yoder Employment Agreement, if payments and benefits payable to Mr. Yoder in connection with a change of control would constitute a “parachute payment” within Section 280G of the Code or are subject to Section 4999 of the Code, then such payments and benefits will either be paid in full or be reduced so that the Section 4999 excise tax does not apply, whichever results in the better after-tax result for Mr. Yoder.
In connection with Pieris’ promotion of Mr. Thomas Bures to Chief Financial Officer, Pieris entered into an employment agreement, or the “Bures Employment Agreement”, with him dated October 7, 2021. For more information on the Bures Employment Agreement, please see the section titled “Pieris Executive and Director Compensation—Employment and Severance Arrangements” elsewhere in this proxy statement/prospectus.
In the event that Mr. Bures’ employment is terminated by Pieris without cause or by Mr. Bures with good reason during a change in control period, as defined in the Bures Employment Agreement, Mr. Bures will be entitled, subject to his execution and nonrevocation of a release of claims in Pieris’ favor and his continued compliance with certain restrictive covenants, to the extent applicable, to (i) receive twelve months of his then-current annual base salary, payable in twelve equal installments on the first regular payroll date following the effective date of his release of claims, (ii) his target bonus for the year, payable as a lump sum, and (iii) reimbursement of COBRA premiums for health benefit coverage for a period of up to twelve months commencing on the first day of the first full calendar month following the effective date of his release of claims. In addition, under the terms of the Bures Employment Agreement or the applicable award agreements, Mr. Bures will be entitled to full acceleration of vesting and lapsing of any forfeiture restrictions on all his outstanding equity awards. Under the Bures Employment Agreement, if payments and benefits payable to Mr. Bures in connection with a change of control would constitute a “parachute payment” within Section 280G of the Code or are subject to Section 4999 of the Code, then such payments and benefits will either be paid in full or be reduced so that the Section 4999 excise tax does not apply, whichever results in the better after-tax result for Mr. Bures.
The below table sets forth the payments and benefits payable to each of Pieris’ executive officers in connection with certain expected actions of the Pieris board of directors and certain terminations following a change of control.
| Payments | ||||
|
Employee |
Severance (1) |
COBRA (2) |
Acceleration of Stock Options (3) |
Total |
|
Stephen S. Yoder |
$876,893 |
$29,544 |
$0 |
$906,437 |
|
Thomas Bures |
$560,217 |
$38,208 |
$0 |
$598,425 |
|
(1) |
The benefits disclosed herein are payable upon termination of the applicable executive’s employment by Pieris without cause or upon such executive’s resignation with good reason, in either case beginning with the signing of an agreement, which, if consummated, would result in a change in control, and ending twelve months following the effective date of a change of control. The signing of the Merger Agreement constitutes a change of control for purposes of such payments. |
|
(2) |
COBRA payments are estimates based on the employees’ current election of healthcare benefits. |
|
(3) |
The exercise price for all unvested stock options exceeds the current trading value per share, and so it is determined that each share subject to an unvested stock option to purchase common stock that would be accelerated in the circumstances described above has zero value. |
Following the closing of the Merger, Pieris expects to incur approximately $1,580,118 in cash severance obligations, which includes approximately $1,437,110 payable to the executive officers above and $143,000 payable to Pieris’ other employees.
Continued Service.
Mr. Christopher Kiritsy, one of Pieris’ existing directors, is expected to continue as a director of the combined company, and following the closing of the Merger will be eligible to be compensated as a non-employee director of the combined company pursuant to the combined company’s non-employee director compensation policy that will be put in place following the Effective Time. Mr. Kiritsy will not receive any change of control payments, transaction completion bonuses or other additional compensation if the Merger is completed.
See section titled “Management Following the Merger—Executive Officers and Directors of the Combined Company Following the Merger” beginning on page 289.
Ownership Interests.
As of October 28, 2024, Pieris’ directors and executive officers beneficially owned, in the aggregate, approximately 0.07% of the shares of Pieris common stock, which for purposes of this subsection excludes any shares of common stock issuable upon exercise of stock options to purchase shares of Pieris common stock held by such individual. Certain of Pieris’ officers, directors and affiliated stockholders have also entered into a support agreement in connection with the Merger. For a more detailed discussion of the support agreements, please see the section titled “Agreements Related to the Merger—Support Agreements” beginning on page 179 of this proxy statement/prospectus.
Treatment of Stock Options.
As of October 28, 2024, there are no unvested stock options to purchase Pieris common stock for Pieris’ non-employee directors.
As of October 28, 2024, Mr. Yoder owned unvested stock options to purchase 8,714 shares of Pieris common stock and Mr. Bures owned unvested stock options to purchase 3,195 shares of Pieris common stock. The treatment of stock options held by Pieris’ executive officers is described above under “—Severance Benefits.”
Indemnification of Officers and Directors.
Pieris has entered into indemnification agreements with each of Pieris’ current directors and executive officers. These agreements require Pieris to indemnify these individuals to the fullest extent permitted under Nevada law against liabilities that may arise by reason of their service to Pieris, and to advance expenses incurred as a result of any proceeding against them as to which they could be indemnified. In addition, pursuant to the terms of the Merger Agreement, Pieris has agreed to purchase a six-year prepaid “D&O tail policy” for the non-cancellable extension of the directors’ and officers’ liability coverage of Pieris’ existing directors’ and officers’ insurance policies for a claims reporting or discovery period of at least six years from and after the Effective Time with respect to any claim related to any period of time at or prior to the Effective Time, with terms, conditions, retentions and limits of liability to be mutually agreed by Pieris and Palvella prior to the closing of the Merger (which approval will not be unreasonably withheld, conditioned or delayed), but that are no less favorable than the coverage provided under Pieris’ existing policies as of the date of the Merger Agreement with respect to coverage of any actual or alleged error, misstatement, misleading statement, act, omission, neglect, breach of duty or any matter claimed against a director or officer of Pieris by reason of him or her serving in such capacity that existed or occurred at or prior to the Effective Time (including in connection with the Merger Agreement or the Merger).
Interests of Palvella Directors and Executive Officers in the Merger
In considering the recommendation of the Palvella board of directors with respect to approving the Merger, stockholders should be aware that Palvella’s directors and executive officers may have interests in the Merger that are different from, or in addition to, the interests of Palvella stockholders generally. These interests may present them with actual or potential conflicts of interest, and these interests, to the extent material, are described below.
The Palvella board of directors was aware of these potential conflicts of interest and considered them, among other matters, in reaching its decision to approve the Merger Agreement and the Merger, and to recommend that the Palvella stockholders approve the Merger Agreement and the Merger as contemplated by this proxy statement/prospectus.
Ownership Interests
As of October 28, 2024, Palvella’s current directors and executive officers beneficially owned, in the aggregate, approximately 28.53% of the shares of Palvella capital stock, which for purposes of this subsection excludes any Palvella shares issuable upon exercise or settlement of Palvella stock options held by such individual. Each of Palvella’s officers, directors and affiliated stockholders have also entered into a support agreement in connection with the Merger. For a more detailed discussion of the support agreements, please see the section titled “Agreements Related to the Merger—Support Agreements” beginning on page 179 of this proxy statement/prospectus.
Treatment of Palvella Options
In connection with the Merger, each stock option granted under the Palvella Stock Plan that is outstanding immediately prior to the Effective Time, will be assumed by Pieris and will become an option to acquire, on the same terms and conditions as were applicable to such Palvella stock option immediately prior to the Effective Time, a number of shares of Pieris common stock equal to the number of shares of Palvella common stock subject to the unexercised portion of the Palvella stock option immediately prior to the Effective Time, multiplied by the exchange ratio (rounded down to the nearest whole share number), with an exercise price per share for the options equal to the exercise price per share of such Palvella stock option immediately prior to the Effective Time divided by the exchange ratio (rounded up to the nearest whole cent). Such assumed options will continue to be governed by the terms and conditions of the Palvella Therapeutics, Inc. 2024 Equity Incentive Plan.
The table below sets forth information regarding the Palvella stock options held as of October 28, 2024 by each of Palvella’s current executive officers and directors.
|
Name |
Number |
Weighted |
Number of |
Weighted |
||||||||
|
Executive Officers |
713,017 | $2.64 | 632,552 | $2.62 | ||||||||
|
Wesley H. Kaupinen |
81,097 | $2.60 | 251,386 | $2.43 | ||||||||
|
Matthew Korenberg |
- | - | - | - | ||||||||
|
Kathleen Goin |
327,358 | $2.73 | 246,565 | $2.81 | ||||||||
|
Jeffrey Martini, Ph.D. |
158,920 | $2.83 | 102,546 | $2.67 | ||||||||
| Kathleen A. McGowan | 145,642 | $2.24 | 32,055 | $2.45 |
|
Name |
Number |
Weighted |
Number |
Weighted |
||||||||
|
Non-Employee Directors |
51,390 | $2.24 | 60,186 | $2.33 | ||||||||
|
George M. Jenkins |
29,445 | $2.24 | 30,093 | $2.33 | ||||||||
|
Scott Morenstein |
- | - | - | - | ||||||||
|
Aditya Asokan, Ph.D. |
- | - | - | - | ||||||||
|
Todd C. Davis |
21,945 | $2.25 | 30,093 | $2.33 | ||||||||
|
Tadd Wessel |
- | - | - | - |
Management Following the Merger
As described elsewhere in this proxy statement/prospectus, including in the section captioned “Management Following the Merger,” one of Pieris’ existing directors, who is expected to be Christopher Kiritsy, will continue as a director of the combined company after the Effective Time, and, following the closing of the Merger, will be eligible to be compensated as a non-employee director of Pieris pursuant to the Pieris non-employee director compensation policy that is expected to remain in place following the Effective Time.
Limitations of Liability and Indemnification
For a discussion of the indemnification and insurance provisions related to the Palvella directors and officers under the Merger Agreement, please see the section titled “The Merger Agreement—Indemnification and Insurance for Directors and Officers” beginning on page 171 of this proxy statement/prospectus.
Limitations of Liability and Indemnification
As described elsewhere in this proxy statement/prospectus, including in the section captioned “Management Following the Merger,” Palvella’s directors and executive officers are expected to become the directors and executive officers of the combined company upon the closing of the Merger.
Subject to the terms and conditions of the Merger Agreement, and in accordance with Delaware law, at the completion of the Merger, Merger Sub, a wholly-owned subsidiary of Pieris formed by Pieris in connection with the Merger, will merge with and into Palvella, with Palvella surviving as a wholly-owned subsidiary of Pieris.
At the Effective Time, upon the terms and subject to the conditions set forth in the Merger Agreement, each outstanding share of Palvella capital stock (excluding dissenting shares) will be automatically converted solely into the right to receive a number of shares of Pieris common stock equal to the exchange ratio described in more detail below.
No fractional shares of Pieris common stock will be issued in connection with the Merger, and no certificates or scrip for any such fractional shares will be issued and such fractional share interests shall not entitle the owner thereof to vote or to any other rights of a stockholder of Pieris. Any fractional shares of Pieris common stock resulting from the conversion of Palvella capital stock into the right to receive a number of shares of Pieris common stock equal to the exchange ratio (after aggregating all fractional shares of Pieris common stock issuable to such holder) will be rounded down to the nearest whole share of Pieris common stock, with cash being paid in lieu of such fractional shares of Pieris common stock eliminated by such rounding.
Based on the estimates set forth above, and certain other assumptions, including, but not limited to, a valuation for Pieris equal to $21 million, provided, that (a) if Pieris’ net cash as of the closing is greater than $11 million, then Pieris’ valuation will be adjusted upwards on a dollar-for-dollar basis by the difference of: (i) Pieris’ net cash, minus (ii) $11 million, and (b) if Pieris’ net cash is less than $11 million, then Pieris’ valuation will be adjusted downwards on a dollar-for-dollar basis by the difference of: (i) $11 million, minus (ii) Pieris’ net cash, as further described in the Merger Agreement, without giving effect to the PIPE Financing, Pieris securityholders as of immediately prior to the Merger would own approximately 18% of the common stock of the combined company, and Palvella securityholders as of immediately prior to the Merger would own approximately 82% of the common stock of the combined company, in each case, on a fully-diluted basis, calculated using the treasury stock method. For more information on the PIPE Financing, please see the section titled “Agreements Related to the Merger—Securities Purchase Agreement” beginning on page 180 in this proxy statement/prospectus.
The exchange ratio formula is the quotient obtained by dividing the number of Palvella merger shares (defined below) by the number of Palvella outstanding shares (defined below), in which:
|
● |
“Pieris allocation percentage” means the quotient determined by dividing (i) the Pieris valuation by (ii) the aggregate valuation; |
|
● |
“Pieris closing price” means the volume weighted average closing trading price of a share of Pieris common stock on Nasdaq for the five consecutive trading days ending five trading days immediately prior to the date upon which the Effective Time occurs; |
|
● |
“Pieris outstanding shares” means the total number of shares of Pieris common stock that are issued and outstanding immediately prior to the Effective Time, expressed on a fully-diluted and as-converted to Pieris common stock basis, calculated using the treasury stock method and assuming, without duplication, (i) the acceleration and exercise of all Pieris stock options and Pieris warrants outstanding as of immediately prior to the Effective Time with an exercise price less than the Pieris closing price, and (ii) the conversion of all Pieris preferred stock outstanding as of immediately prior to the Effective Time (on an as-converted to Pieris common stock basis). For the avoidance of doubt, (A) no Pieris stock options outstanding as of immediately prior to the Effective Time with an exercise price equal to or greater than the Pieris closing price shall be included in the total number of shares of Pieris common stock outstanding for purposes of determining the Pieris outstanding shares, and (B) other than with respect to Pieris common stock underlying outstanding Pieris stock options and Pieris warrants with an exercise price less than the Pieris closing price, shares of Pieris common stock reserved for issuance under the Pieris stock plans as of immediately prior to the Effective Time shall not be included in the total number of shares of Pieris common stock outstanding for purposes of determining the Pieris outstanding shares; |
|
● |
“Pieris valuation” means $21,000,000; provided, that (i) if the Pieris final net cash is greater than $11,000,000, then the Pieris Valuation will be adjusted upwards on a dollar-for-dollar basis by the difference of: (A) the Pieris final net cash, minus (B) $11,000,000, and (ii) if the Pieris final net cash is less than $11,000,000, then the Pieris Valuation will be adjusted downwards on a dollar-for-dollar basis by the difference of: (A) $11,000,000, minus (B) the Pieris final net cash; |
|
● |
“aggregate valuation” means the sum of the (i) Palvella valuation, plus (ii) the Pieris valuation; |
|
● |
“Palvella allocation percentage” means the quotient determined by dividing (i) the Palvella valuation by (ii) the aggregate valuation; |
|
● |
“Palvella merger shares” means the product determined by multiplying (i) the post-closing Pieris shares by (ii) the Palvella allocation percentage; |
|
● |
“Palvella outstanding shares” means the total number of shares of Palvella capital stock outstanding immediately prior to the Effective Time expressed on a fully-diluted and as-converted to Palvella common stock basis calculated using the treasury stock method and assuming, without duplication, (i) the issuance of all shares of Palvella common stock that would be issued assuming the acceleration and exercise of all Palvella stock options outstanding as of immediately prior to the Effective Time, and (ii) the conversion of all Palvella preferred stock outstanding as of immediately prior to the Effective Time (on an as-converted to Palvella common stock basis). Notwithstanding the foregoing, any Palvella stock options granted on or after the date of the Merger Agreement would be included in the total number of Palvella outstanding shares on a fully-diluted and as-converted to Palvella common stock basis without applying the treasury stock method. For the avoidance of doubt, other than with respect to Palvella common stock underlying outstanding Palvella stock options, shares of Palvella common stock reserved for issuance under the Palvella Stock Plan as of immediately prior to the Effective Time shall not be included in the total number of shares of Palvella common stock outstanding for purposes of determining the Palvella outstanding shares; |
|
● |
“Palvella valuation” means $95 million; and |
|
● |
“Post-closing Pieris shares” mean the quotient determined by dividing (i) the Pieris outstanding shares by (ii) the Pieris allocation percentage. |
Calculation of Pieris’ Final Net Cash
Pursuant to the terms of the Merger Agreement, Pieris’ “final net cash” means, as of the cash determination time (which is as of the close of business on the last business day prior to the anticipated closing date of the Merger) the sum (without duplication) of the following:
|
● |
Pieris’ and its subsidiaries’ unrestricted cash, cash equivalents, and marketable securities; |
|
● |
accounts receivable that would be required to be set forth in a balance sheet prepared in accordance with GAAP and deposits (to the extent refundable or applicable to Palvella, Pieris or any subsidiary of Pieris following the Effective Time); |
|
● |
expenses paid, or liabilities incurred, prior to closing of the Merger, that are approved in writing to be covered and reimbursed by the Pieris D&O tail policy in excess of the deductible and within overall policy limits; |
|
● |
any amounts owed to Pieris or any of its subsidiaries in respect of a Legacy Asset Disposition (as defined in the Merger Agreement), which amounts have not been received as of the closing date of the Merger (in each case, net of any expenses, fees, or taxes payable by Pieris or its subsidiaries in respect of such Legacy Asset Disposition), if the proceeds of such Legacy Asset Disposition would be reflected as a bona fide account receivable in accordance with GAAP; |
|
● |
the amount of tax refund or credit (including, for the avoidance of doubt, the German research and development tax credit) (i) received by Pieris or any of its affiliates or (ii) awarded to Pieris or any of its affiliates, whether or not such amount is pending, due and owning, or unpaid as of the closing of the Merger, if such tax refund would be reflected as a bona fide account receivable in accordance with GAAP and approved by Palvella in its reasonable discretion; |
|
● |
all prepaid expenses and deposits of Pieris set forth in the Merger Agreement usable by Palvella, Pieris or any subsidiary of Pieris following the Effective Time, or any other prepaid expenses or deposits of Pieris or any of its affiliates mutually agreed upon by Pieris and Palvella; |
|
● |
50% of the amount of any costs or expenses, including attorney’s fees, settlement amounts or judgments, incurred or paid in connection with any securityholder litigation (including all amounts paid or payable up to the retention amount of any insurance policy that covers or may cover such costs or expenses or amounts not covered by any such insurance policy); |
|
● |
50% of any fees or expenses incurred by Pieris prior to the Effective Time in connection with filing a listing application with Nasdaq; and |
|
● |
50% of any fees or expenses incurred by Pieris prior to the Effective Time in connection with Nasdaq, proxy solicitation, exchange agent services, and any and all SEC matters, including, without limitation, the printing, filing and mailing of this proxy statement/prospectus (including any related preliminary materials) and the Registration Statement on Form S-4 and any amendments or supplements thereto. |
Minus the sum (without duplication) of the following:
|
● |
Pieris’ and its subsidiaries’ accrued and unpaid accounts payable and other accrued and unpaid expenses (other than transaction expenses), including legal settlements that are not covered by a director and officer insurance policy and the cost of the Pieris D&O tail policy, without duplication, any accrued but unpaid taxes related to or attributable to all taxable periods, or portions of taxable periods, ending on or before the closing date of the Merger, including, for the avoidance of doubt, any tax liabilities relating to the disposition of any Legacy Assets (as defined in the Merger Agreement) after taking into account any available NOL carryforwards or other tax attributes of Pieris and any foreign tax credits which foreign tax credits are directly related to the disposition of the Legacy Assets (if any) and any employer portion employment taxes incurred in connection with any compensatory payments made to employees and any and all other bona fide current and long-term liabilities payable in cash that would be required to be set forth in a balance sheet prepared in accordance with GAAP; |
|
● |
Pieris’ or its subsidiaries’ unpaid transaction expenses; |
|
● |
any change of control, bonus, deferred compensation, severance (voluntary or otherwise) (including a reasonable estimate of payment or reimbursement for continued coverage under any employee benefit plan), retention or similar payments (whether “single trigger” or “double trigger”) that become due and payable by Pieris or any of its subsidiaries pursuant to contracts entered into at or prior to the Effective Time as a result of or in connection with the Merger or any Legacy Asset Disposition and remain unpaid as of the Effective Time; |
|
● |
the amount of any notice payments, fines or other payments to be made by Pieris in order to terminate any existing agreement to which Pieris is a party and to wind down any current and future clinical trial obligations, in each case, that remain unpaid as of the Effective Time; and |
|
● |
50% of the amount of costs or expenses, including attorney’s fees or settlement amounts or judgments, incurred or paid in connection with any securityholder litigation (including all amounts paid or payable up to the retention amount of any insurance policy that covers or may cover such costs or expenses or amounts not covered by any such insurance policy) that remain unpaid as of the Effective Time. |
In connection with his anticipated termination of employment following the effective time of the Merger, certain officers of Pieris, including Stephen S. Yoder, Pieris’ Chief Executive Officer, and Thomas Bures, Pieris’ Chief Financial Officer, would be entitled to receive certain severance payments and benefits under the terms of their respective employment agreements with Pieris, as described under “Employment and Severance Arrangements” beginning on page 191 and “Severance Benefits” beginning on page 148 in this proxy statement/prospectus. The amount of such cash payments will reduce Pieris’ final net cash. Pieris has already assumed such reductions in its estimated cash at closing of $11 million.
Not less than ten business days prior to the anticipated closing date of the Merger, Pieris will deliver to Palvella a draft net cash schedule setting forth, in reasonable detail, Pieris’ good faith estimated calculation of its net cash and the exchange ratio as of the anticipated closing date of the Merger, together with the relevant work papers and back-up materials used or useful in preparing the net cash schedule. Pieris will consider in good faith any comments provided by Palvella within four business days of the delivery of the draft net cash schedule.
Not less than four (but no more than six) business days prior to the anticipated closing date of the Merger, Pieris will deliver to Palvella a proposed final net cash schedule, prepared and certified by Pieris’ Chief Financial Officer (or if there is no Chief Financial Officer, the Chief Executive Officer), together with the relevant work papers and back-up materials used or useful in preparing the net cash schedule, which such proposed final net cash schedule will be subject to the agreement and consent of Palvella.
On or before the third calendar day after Pieris’ delivery of such proposed final net cash schedule, Palvella will have the right to dispute any part of the net cash schedule by delivering a written notice to that effect to Pieris (referred to herein as a dispute notice). Any dispute notice will identify in reasonable detail each item in dispute and Palvella’s proposed revisions to Pieris’ net cash schedule. On the fourth calendar day following the delivery of the proposed final net cash schedule, if Palvella does not dispute the proposed final net cash schedule, it will become final and binding on all parties.
If Palvella disputes the net cash schedule, the parties shall attempt in good faith to resolve the disputed items and negotiate an agreed-upon determination of net cash. If the parties are unable to negotiate an agreed-upon determination of net cash or any component thereof within two calendar days of the anticipated closing date of the Merger, any remaining disagreements will be referred to an independent auditor of recognized national standing jointly selected by Pieris and Palvella which is not serving as auditor of either Pieris or Palvella. The determination of the amount of net cash made by such accounting firm shall be final and binding on Pieris and Palvella.
Pieris’ net cash balance is subject to numerous factors, some of which are outside of Pieris’ control. The actual amount of net cash will depend significantly on the timing of the closing of the Merger. In addition, the closing of the Merger could be delayed if Pieris and Palvella are not able to agree upon the amount of Pieris’ net cash as of the cash determination.
Procedures for Exchanging Palvella Stock Certificates
At the Effective Time, Pieris will deposit with Computershare Trust Company, N.A. or another bank or trust company designated by Pieris and reasonably acceptable to Palvella, as the exchange agent, (i) certificates representing the shares of Pieris common stock issuable pursuant to the terms of the Merger Agreement in exchange for shares of Palvella capital stock, (ii) cash payable in lieu of fractional shares of Pieris common stock otherwise issuable pursuant to the terms of the Merger Agreement, and (iii) any dividends or distributions to which pre-closing holders of Palvella common stock may be entitled to under the terms of the Merger Agreement.
As soon as reasonably practicable after the Effective Time, the exchange agent will mail to each record holder of Palvella capital stock (i) a letter of transmittal and (ii) instructions for surrendering the record holder’s stock certificates, if applicable, in exchange for the merger consideration. Upon delivery to the exchange agent of a duly executed letter of transmittal in accordance with the exchange agent’s instructions, the surrender of the record holder’s stock certificates, if applicable, and delivery to the exchange agent of such other documents as may be reasonably required by the exchange agent or Pieris, the record holder of such Palvella capital stock will be entitled to receive in exchange therefor a stock certificate or book-entry shares representing the number of whole shares of Pieris common stock issuable to such holder pursuant to the Merger Agreement. The surrendered certificates representing shares of Palvella common stock or Palvella preferred stock will be canceled.
After the Effective Time, each certificate representing Palvella common stock or Palvella preferred stock that has not been surrendered will represent only the right to receive shares of Pieris common stock issuable pursuant to the Merger Agreement to which the holder of any such certificate is entitled.
The Merger Agreement requires the parties to consummate the Merger no later than two business days following the satisfaction or (to the extent permitted by law) wavier of the conditions to the consummation of the Merger contained in the Merger Agreement, including the adoption of the Merger Agreement by the Palvella stockholders and the approval by the Pieris stockholders of the issuance of Pieris common stock and the other transactions proposed under the Merger Agreement, other than those conditions that by their nature are to be satisfied at the closing of the Merger, unless another date or time is agreed to in writing by Pieris and Palvella. The Merger will become effective upon the filing of a certificate of merger with the Secretary of State of the State of Delaware or at such later time as is agreed by Pieris and Palvella and specified in the certificate of merger. Neither Pieris nor Palvella can predict the exact timing of the consummation of the Merger.
In the United States, Pieris must comply with applicable federal and state securities laws and the rules and regulations of Nasdaq in connection with the issuance of shares of Pieris common stock to Palvella stockholders in connection with the transactions contemplated by the Merger Agreement and the filing of this proxy statement/prospectus with the SEC.
Material U.S. Federal Income Tax Consequences of the Merger
The following discussion is a summary of the material U.S. federal income tax consequences of the Merger to Palvella U.S. holders (as defined below) that exchange Palvella capital stock for Pieris common stock pursuant to the Merger, but does not purport to be a complete analysis of all potential tax consequences that may be relevant to Palvella U.S. holders. The effects of other U.S. federal tax laws, such as estate and gift tax laws, and any applicable state, local or non-U.S. tax laws are not discussed. This discussion is based on the Code, Treasury Regulations promulgated thereunder, judicial decisions, and published rulings and administrative pronouncements of the IRS, in each case in effect as of the date hereof. These authorities may change or be subject to differing interpretations. Any such change or differing interpretation may be applied retroactively in a manner that could adversely affect a Palvella U.S. holder. Palvella has not sought and does not intend to seek a ruling from the IRS regarding the matters discussed below and, even though an opinion of counsel has been sought and obtained by Palvella, such opinion is not binding upon the IRS or a court. Consequently, there can be no assurance the IRS or a court will not take a position contrary to that discussed below regarding the tax consequences of the Merger.
This discussion is limited to Palvella U.S. holders that hold Palvella capital stock as a “capital asset” within the meaning of Section 1221 of the Code (generally, property held for investment). This discussion does not address all U.S. federal income tax consequences that may be relevant to a Palvella U.S. holder’s particular circumstances, including the impact of the alternative minimum tax, the Medicare contribution tax on net investment income or the rules related to “qualified small business stock” within the meaning of Section 1202 of the Code, or as “Section 1244 stock” for purposes of Section 1244 of the Code. In addition, it does not address consequences relevant to Palvella U.S. holders subject to special rules, including, without limitation:
|
● |
U.S. expatriates and former citizens or long-term residents of the U.S.; |
|
● |
U.S. holders whose functional currency is not the U.S. dollar; |
|
● |
persons holding Palvella capital stock as part of a hedge, straddle or other risk reduction strategy or as part of a conversion transaction or other integrated investment; |
|
● |
banks, insurance companies and other financial institutions; |
|
● |
real estate investment trusts or regulated investment companies; |
|
● |
brokers, dealers or traders in securities; |
|
● |
persons subject to special tax accounting rules as a result of any item of gross income with respect to Palvella capital stock being taken into account in an “applicable financial statement” (as defined in the Code); |
|
● |
persons who acquired their shares of Palvella capital stock in a transaction subject to the gain rollover provisions of Section 1045 of the Code; |
|
● |
persons who hold “Section 305 stock”; |
|
● |
S corporations, partnerships or other entities or arrangements classified as partnerships for U.S. federal income tax purposes (and investors therein); |
|
● |
tax-exempt organizations or governmental organizations; |
|
● |
persons deemed to sell Palvella capital stock under the constructive sale provisions of the Code; |
|
● |
persons who hold or received Palvella capital stock pursuant to the exercise of any employee stock option or otherwise as compensation; and |
|
● |
tax-qualified retirement plans. |
If an entity (or arrangement) classified as a partnership for U.S. federal income tax purposes holds Palvella common stock, the tax treatment of a partner in the partnership will depend on the status of the partner, the activities of the partnership and certain determinations made at the partner level. Accordingly, partnerships holding Palvella common stock and the partners in such partnerships should consult their tax advisors regarding the U.S. federal income tax consequences to them.
In addition, the following discussion does not address: (a) the tax consequences of transactions effectuated before, after or at the same time as the Merger, whether or not they are in connection with the Merger, including, without limitation, transactions in which shares of Palvella capital stock are acquired or disposed of other than in exchange for shares of Pieris common stock in the Merger (such as through the exercise of dissenters’ rights), (b) the tax consequences to holders of convertible notes or options or warrants of Palvella, or (c) the tax consequences of the ownership of shares of Pieris common stock following the Merger.
IT IS RECOMMENDED THAT ALL HOLDERS CONSULT THEIR TAX ADVISORS WITH RESPECT TO THE APPLICATION OF THE U.S. FEDERAL INCOME TAX LAWS TO THEIR PARTICULAR SITUATIONS AS WELL AS ANY TAX CONSEQUENCES OF THE MERGER ARISING UNDER THE U.S. FEDERAL ESTATE OR GIFT TAX LAWS OR UNDER THE LAWS OF ANY STATE, LOCAL OR NON-U.S. TAXING JURISDICTION OR UNDER ANY APPLICABLE INCOME TAX TREATY.
For purposes of this discussion, a “Palvella U.S. holder” is a beneficial owner of Palvella capital stock that, for U.S. federal income tax purposes, is or is treated as:
|
● |
an individual who is a citizen or resident of the U.S.; |
|
● |
a corporation created or organized in or under the laws of the U.S., any state thereof, or the District of Columbia; |
|
● |
an estate, the income of which is subject to U.S. federal income tax regardless of its source; or |
|
● |
a trust that (i) is subject to the primary supervision of a U.S. court and the control of one or more “United States persons” (within the meaning of Section 7701(a)(30) of the Code) over all of its substantial decisions or (ii) has a valid election in effect to be treated as a United States person for U.S. federal income tax purposes. |
Tax Characterization of the Merger
In the opinion of Mintz and Troutman the Merger will qualify as a “reorganization” within the meaning of Section 368(a) of the Code, and the material U.S. federal income tax consequences of the Merger to Palvella U.S. holders are as described below under the heading “—Tax Treatment of Palvella U.S. Holders in the Merger.” These opinions are based on facts and representations contained in representation letters provided to Mintz and Troutman by Pieris and Palvella and certain assumptions, including that the merger is completed in the manner set forth in the Merger Agreement and the Registration Statement on Form S-4 of which this proxy statement/prospectus forms a part. The accuracy of such facts, representations and assumptions, could affect the conclusions set forth in such opinions and, as previously noted above, such opinions will not be binding on the IRS or the courts. Consequently, there can be no assurance the IRS or a court will not take a position contrary to the intended tax treatment of the Merger. Palvella stockholders are encouraged to consult their own tax advisors concerning the intended characterization of the Merger as a “reorganization” under Section 368(a) of the Code.
If the Merger does not qualify as a “reorganization” within the meaning of Section 368(a) of the Code (including if the IRS successfully challenges the qualification of the Merger as such), then each Palvella U.S. holder would recognize gain or loss on the exchange of Palvella capital stock for Pieris common stock in the Merger equal to the difference between (x) the fair market value of the shares of Pieris common stock received in exchange for the Palvella capital stock plus any cash received in lieu of a fractional share and (y) such Palvella U.S. holder’s adjusted tax basis in the shares of Palvella capital stock surrendered.
The remainder of this discussion assumes that the Merger will be treated as a “reorganization” within the meaning of Section 368(a) of the Code in accordance with the opinions referred to above.
Tax Treatment of Palvella U.S. Holders that Participate in the Merger. The material U.S. federal income tax consequences to a Palvella U.S. holder as a result of the Merger will be as follows: (i) except as described below with respect to the receipt of cash in lieu of a fractional share of Pieris common stock, a Palvella U.S. holder will not recognize gain or loss upon the exchange of the holder’s Palvella capital stock for Pieris common stock, (ii) a Palvella U.S. holder will obtain an aggregate adjusted tax basis in the Pieris common stock the holder receives in the Merger equal to the holder’s adjusted tax basis in the Palvella capital stock exchanged therefor, reduced by the basis allocable to any fractional share of Pieris common stock for which cash is received, and (iii) the holding period of the shares of Pieris common stock received by a Palvella U.S. holder in the Merger will include the holding period of the shares of Palvella capital stock surrendered in exchange therefor. Treasury Regulations provide detailed rules for allocating the tax basis and holding period of the shares of Palvella capital stock surrendered to the shares of Pieris common stock received. Palvella U.S. holders of shares of Palvella capital stock acquired on different dates and at different prices should consult their tax advisors regarding the allocation of the tax basis and holding period of such shares.
Tax Treatment of Cash Received in Lieu of a Fractional Share of Pieris Common Stock. A Palvella U.S. holder that receives cash in lieu of a fractional share of Pieris common stock will be treated as having received such fractional share and then as having received such cash in redemption of the fractional share. A Palvella U.S. holder will recognize gain or loss equal to the difference between the amount of cash received in lieu of the fractional share of Pieris common stock and the portion of the U.S. holder’s aggregate adjusted tax basis in the shares of Palvella capital stock allocable to the fractional share. Such gain or loss will generally be capital gain or loss and will be long-term capital gain or loss if the Palvella U.S. holder’s holding period for the Palvella capital stock surrendered in the Merger exceeds one year at the Effective Time. Long-term capital gains of certain non-corporate holders of Palvella capital stock, including individuals, are taxed at preferential rates. The deductibility of capital losses is subject to limitations.
Reporting Requirements. Each Palvella U.S. holder who receives shares of Pieris common stock in the Merger is required to retain permanent records pertaining to the Merger and make such records available to any authorized IRS officers and employees. Such records should specifically include information regarding the amount, basis, and fair market value of the Palvella capital stock exchanged and the amount of Pieris common stock and cash received in exchange therefor. Palvella U.S. holders who owned immediately before the Merger at least 1% (by vote or value) of the total outstanding stock of Palvella are required to attach a statement to their tax returns for the year in which the Merger is consummated that contains the information listed in Treasury Regulations Section 1.368-3(b). Such statement must include the Palvella U.S. holder’s tax basis in such holder’s Palvella capital stock surrendered in the Merger, the fair market value of such stock, the date of the Merger and the name and employer identification number of each of Palvella and Pieris.
Palvella U.S. holders are urged to consult with their tax advisors to comply with these rules.
Backup Withholding and Information Reporting. A Palvella U.S. holder may, under certain circumstances, be subject to information reporting and backup withholding on any payments of cash in lieu of fractional shares, unless such holder properly establishes an exemption or provides its correct taxpayer identification number and otherwise complies with the applicable requirements of the backup withholding rules. Backup withholding is not an additional tax. Any amounts withheld under the backup withholding rules may be refunded or credited against a payee’s U.S. federal income tax liability, if any, so long as such payee furnishes the required information to the IRS in a timely manner.
The foregoing summary is not intended to be, and should not be construed to be, legal, business or tax advice to any particular Palvella U.S. holder. This summary does not take into account your particular circumstances and does not address consequences that may be particular to you. Therefore, you should consult your tax advisor regarding the particular consequences of the Merger to you.
Anticipated Accounting Treatment
The Merger is expected to be accounted for as a reverse recapitalization in accordance with GAAP. Under this method of accounting, Palvella will be deemed to be the accounting acquirer for financial reporting purposes. This determination was primarily due to Pieris being determined to be a shell company at the time of the Merger, immediately following the dividend of the CVR and following the discontinuation of historical research and development efforts, because it will not meet the GAAP definition of a business, will not have more than nominal assets, and will not have more than nominal operations. Further, it is expected, immediately following the Merger: (i) Palvella stockholders will own a substantial majority of the voting rights; (ii) Palvella will designate a majority (four of five) of the initial members of the board of directors of the combined company; (iii) Palvella’s executive management team will become the management team of the combined company; and (iv) the combined company will be named Palvella Therapeutics, Inc. and be headquartered in Wayne, Pennsylvania. Accordingly, for accounting purposes, the Merger will be treated as the equivalent of Palvella issuing stock to acquire the net assets of Pieris. As a result of the Merger, the net assets of Pieris will be recorded at their acquisition-date fair value in the financial statements of Palvella and the reported operating results prior to the Merger will be those of Palvella. See the section titled “Unaudited Pro Forma Condensed Combined Financial Information” elsewhere in this proxy statement/prospectus for additional information.
Shares of Pieris common stock are currently listed on The Nasdaq Capital Market under the symbol “PIRS.” Pieris has agreed to cause the shares of Pieris common stock being issued in the Merger to be approved for listing on Nasdaq at or prior to the Effective Time.
Pieris has filed a listing application for the combined company with Nasdaq. The Nasdaq Capital Market objective listing criteria are currently satisfied. After completion of the Merger, the combined company will be renamed “Palvella Therapeutics, Inc.” and, assuming approval of the application for continued listing, the common stock of the combined company will trade on The Nasdaq Capital Market under the symbol “PVLA.” However, Nasdaq’s determination may not be known at the time stockholders are asked to vote on the Merger.
In addition, under the Merger Agreement, each of Pieris’ and Palvella’s obligation to complete the Merger is subject to the satisfaction or waiver by each of the parties, at or prior to the Merger, of various conditions, including that the shares of Pieris common stock to be issued in the Merger have been approved for listing on Nasdaq, subject to official notice of issuance, as of the closing of the Merger.
Appraisal Rights and Dissenters’ Rights
Holders of Pieris common stock are not entitled to dissenter’s or appraisal rights under Nevada law in connection with the Merger.
Under the DGCL, Pieris stockholders are not entitled to appraisal rights in connection with the Merger.
If the Merger is consummated, Palvella stockholders and beneficial owners who (1) do not vote in favor of or deliver a written consent adopting the Merger Agreement, (2) have not waived their right to appraisal, (3) continuously hold their shares from the date of making a demand for appraisal through the effective date of the Merger and (4) have otherwise complied with the applicable procedures and requirements set forth in Section 262 of the DGCL, which is hereinafter referred to as Section 262, are entitled to exercise appraisal rights in connection with the Merger under Section 262.
The discussion below is not a complete summary regarding Palvella stockholders’ appraisal rights under Delaware law and is qualified in its entirety by reference the text of Section 262. In the event of any inconsistency between the information contained in this summary, and the actual text of Section 262 of the DGCL, the actual text of Section 262 of the DGCL controls. All references in Section 262 and in this summary to a “stockholder” are to the record holder of shares of Palvella capital stock unless otherwise expressly noted herein, and all such references to a “beneficial owner” mean a person who is the beneficial owner of shares of Palvella capital stock held either in voting trust or by a nominee on behalf of such person unless otherwise expressly noted herein. Stockholders and beneficial owners intending to exercise appraisal rights, or to preserve their right to do so, should carefully review Section 262. Failure to follow precisely any of the statutory procedures set forth in Section 262 will result in a termination or waiver of these rights. This summary does not constitute legal or other advice, nor does it constitute a recommendation that Palvella stockholders or beneficial owners exercise their appraisal rights under Delaware law.
Under Section 262, where a merger is adopted by stockholders by written consent in lieu of a meeting of stockholders pursuant to Section 228 of the DGCL, either the constituent corporation before the effective date of such merger or the surviving corporation, within ten days after the effective date of such merger, must notify each stockholder of the constituent corporation entitled to appraisal rights of the approval of such merger, and that appraisal rights are available and shall include in such notice either a copy of Section 262 of the DGCL or information directing the stockholders to a publicly available electronic resource at which Section 262 of the DGCL may be accessed without subscription or cost. Such notice may, and, if given on or after the effective date of the merger, shall, also notify such stockholders of the effective date of the Merger.
If the Merger is completed, following stockholder approval of the Merger, and no later than 10 days after the effective date of the Merger, Palvella will notify its stockholders entitled thereto that the Merger has been approved and that appraisal rights are available to any stockholder who is entitled to exercise appraisal rights, including by not having delivered a written consent to the adoption of the Merger Agreement. Such notice may, and, if given on or after the effective date of the Merger, shall, also notify such stockholders of the effective date of the Merger. If such notice did not notify stockholders of the effective date of the Merger, then Palvella will send a second notice notifying each of the stockholders of Palvella that are entitled to appraisal rights of the effective date of the Merger either before the effective date of the Merger or on or within 10 days after such effective date; provided, however, that if such second notice is sent more than 20 days following the sending of the first notice, such second notice need only be sent to each stockholder who is entitled to appraisal rights and who has demanded appraisal of such holder’s shares in accordance with Section 262(d)(2) of the DGCL and any beneficial owner who has demanded appraisal under Section 262(d)(3) of the DGCL. Holders of record or beneficial owners of shares of Palvella capital stock who desire to exercise their appraisal rights must deliver a written demand for appraisal to Palvella within 20 days after the date of giving a notice of appraisal rights, and that the shares subject to such appraisal demand must not have been voted, including by written consent, in favor of the Merger and adopting the Merger Agreement. A demand for appraisal must reasonably inform Palvella of the identity of the stockholder or the beneficial owner and that such stockholder or beneficial owner intends thereby to demand appraisal of the shares of Palvella capital stock held by such stockholder or beneficial owner. In addition, in the case of a demand for appraisal made by a beneficial owner, the demand must (1) reasonably identify the holder of record of the shares for which the demand is made, (2) provide documentary evidence of such beneficial owner’s beneficial ownership and a statement that such documentary evidence is a true and correct copy of what it purports to be and (3) provide an address at which such beneficial owner consents to receive notices given by Palvella and to be set forth on the verified list of persons who have demanded appraisal for their shares pursuant to Section 262(f) of the DGCL. Failure to deliver a written consent adopting the Merger Agreement will not in and of itself constitute a written demand for appraisal satisfying the requirements of Section 262. All demands for appraisal should be addressed to Palvella Therapeutics, Inc., c/o Palvella Therapeutics, Inc., 125 Strafford Avenue, Suite 360, Wayne, Pennsylvania 19087, Attention: Secretary, and should be executed by, or on behalf of, the record holder of shares of Palvella capital stock. ALL DEMANDS MUST BE RECEIVED BY PALVELLA WITHIN 20 DAYS AFTER THE DATE PALVELLA SENDS A NOTICE TO ITS STOCKHOLDERS ENTITLED THERETO NOTIFYING THEM THAT THE MERGER HAS BEEN APPROVED, AND THE AVAILABILITY OF APPRAISAL RIGHTS IN CONNECTION WITH THE MERGER.
The failure to deliver a written demand for appraisal within the time period specified above, will result in the loss of appraisal rights and such person will be entitled to receive the merger consideration for his, her or its shares of Palvella capital stock as provided for in the Merger Agreement.
To be effective, a demand for appraisal by a holder of shares of Palvella capital stock or beneficial owner must be made by, or in the name of, the registered stockholder or beneficial owner. In addition, the stockholder or beneficial owner must continuously hold or own, as applicable, the shares from the date of making the demand through the Effective Time.
At any time within 60 days after the Effective Time, any person who has demanded and who is entitled to appraisal rights, but has neither commenced an appraisal proceeding nor joined an appraisal proceeding as a named party, has the right to withdraw such person’s demand and accept the merger consideration by delivering a written withdrawal to Palvella. No appraisal proceeding in the Delaware Court of Chancery, or the “Delaware Court”, will be dismissed as to any person without the approval of the Delaware Court, with such approval conditioned upon such terms as the Delaware Court deems just, including without limitation, a reservation of jurisdiction for any application to the Delaware Court made pursuant to Section 262(j) for expenses, including, without limitation, reasonable attorneys’ fees and the fees and expenses of experts, to be charged pro rata against the value of all shares entitled to appraisal, provided, however, that this provision will not affect the right of any Palvella stockholder or beneficial owner who has not commenced an appraisal proceeding or joined that proceeding as a named party to withdraw such stockholder’s or beneficial owner’s demand for appraisal and to accept the merger consideration within 60 days after the Effective Time.
If, following a demand for appraisal, a person has withdrawn such person’s demand for appraisal in accordance with Section 262, such person will have the right to receive the merger consideration for such person’s shares of Palvella capital stock.
Within 120 days after the effective date of the Merger, any person who has delivered a demand for appraisal in accordance with Section 262 will, upon written request to the surviving corporation, be entitled to receive a written statement setting forth the aggregate number of shares not voted in favor of the Merger and with respect to which demands for appraisal rights have been received and the aggregate number of stockholders or beneficial owners holding or owning these shares, provided that, where a beneficial owner makes such demand, the record holder of such shares shall not be considered a separate stockholder holding such shares for the purpose of such aggregate number. This written statement will be given to the requesting person within 10 days after such person’s written request is received by the surviving corporation or within 10 days after expiration of the period for delivery of demands for appraisal, whichever is later. Within 120 days after the effective date of the Merger, either the surviving corporation or any person who has properly and timely demanded appraisal and otherwise complied with Section 262 may file a petition in the Delaware Court demanding a determination of the fair value of the shares held by all such stockholders. Upon the filing of the petition by a person other than the surviving corporation, service of a copy of the petition must be made upon the surviving corporation. The surviving corporation has no obligation to file a petition in the Delaware Court, and Palvella, which is expected to be the surviving corporation, has no present intent to file a petition in the Delaware Court. Accordingly, persons who desire to have their shares of Palvella capital stock appraised should initiate any petitions necessary for the perfection of their appraisal rights within the time periods and in the manner prescribed in Section 262. If, within 120 days after the Effective Time, no petition has been filed as required by Section 262, all rights to appraisal will cease and any person that previously demanded appraisal will become entitled only to the merger consideration under the Merger Agreement.
If a petition for appraisal is duly filed by a person other than the surviving corporation and a copy of the petition is delivered to the surviving corporation, the surviving corporation will then be obligated, within 20 days after receiving service of a copy of the petition, to provide the Delaware Register in Chancery with a duly verified list containing the names and addresses of all persons who have demanded an appraisal of their shares and with whom agreements as to the value of their shares have not been reached by the surviving corporation. The Delaware Register in Chancery, if so ordered by the Delaware Court, shall give notice of the time and place fixed for the hearing of such petition by registered or certified mail to the surviving corporation and to all such person set forth on such verified list.
The Delaware Court is empowered to conduct a hearing upon the petition, and to determine those persons who have complied with Section 262 and who have become entitled to the appraisal rights provided thereby. The Delaware Court may require the persons who have demanded an appraisal for their shares and who hold stock represented by certificates to submit their certificates of stock to the Register in Chancery for notation thereon of the pendency of the appraisal proceedings; and if any person fails to comply with that direction, the Delaware Court may dismiss the proceedings as to that person. Upon application by the surviving corporation or any person entitled to participate in the appraisal proceedings, the Delaware Court may, in its discretion, proceed to trial upon the appraisal prior to the final determination of the persons entitled to appraisal. Any person whose name appears on the verified list may participate fully in all proceedings until it is finally determined that such person is not entitled to appraisal rights under Section 262.
The appraisal proceeding shall be conducted in accordance with the rules of the Delaware Court, including any rules specifically governing appraisal proceedings. Through such proceedings, the Delaware Court will appraise the “fair value” of the shares taking into account all relevant factors, exclusive of any element of value arising from the accomplishment or expectation of the Merger, together with interest, if any, to be paid upon the amount determined to be the fair value. Unless the Delaware Court in its discretion determines otherwise for good cause shown, and except as provided in subsection (h) of Section 262, interest from the effective date of the Merger through the date of payment of the judgment will be compounded quarterly and will accrue at 5% over the Federal Reserve discount rate (including any surcharge) as established from time to time during the period between the effective date of the Merger and the date of payment of the judgment. However, the surviving corporation has the right, at any time prior to the Delaware Court’s entry of judgment in the proceedings, to make a voluntary cash payment to each stockholder of record and beneficial owner entitled to appraisal. If the surviving corporation makes a voluntary cash payment pursuant to subsection (h) of Section 262, interest will accrue thereafter only on the sum of (1) the difference, if any, between the amount paid by the surviving corporation in such voluntary cash payment and the fair value of the shares as determined by the Delaware Court and (2) interest accrued on the amount of the voluntary cash payment before such payment was made, unless such interest was paid at the time the voluntary cash payment is made. When the value is determined, the Delaware Court will direct the payment of the fair value, with interest thereon accrued during the pendency of the proceeding to the persons entitled to receive the same.
In determining fair value, the Delaware Court is required to take into account all relevant factors. In Weinberger v. UOP, Inc., the Delaware Supreme Court discussed the factors that could be considered in determining fair value in an appraisal proceeding, stating that “proof of value by any techniques or methods which are generally considered acceptable in the financial community and otherwise admissible in court” should be considered, and that “[f]air price obviously requires consideration of all relevant factors involving the value of a company.” The Delaware Supreme Court has stated that in making this determination of fair value, the court must consider market value, asset value, dividends, earnings prospects, the nature of the enterprise and any other facts which were known or could be ascertained as of the date of the Merger which throw any light on future prospects of the merged corporation.
Section 262 provides that fair value is to be “exclusive of any element of value arising from the accomplishment or expectation of the merger.” In Cede & Co. v. Technicolor, Inc., the Delaware Supreme Court stated that this exclusion is a “narrow exclusion [that] does not encompass known elements of value,” but which rather applies only to the speculative elements of value arising from such accomplishment or expectation. In Weinberger, the Delaware Supreme Court construed Section 262 to mean that “elements of future value, including the nature of the enterprise, which are known or susceptible of proof as of the date of the merger and not the product of speculation, may be considered.”
Persons considering exercising appraisal rights should be aware that the fair value of such person’s shares as determined under Section 262 could be more than, the same as, or less than the value that such holder is entitled to receive under the terms of the Merger Agreement.
Moreover, neither Pieris nor Palvella anticipates offering more than the merger consideration to any Palvella stockholder or beneficial owner exercising appraisal rights and reserves the right to make a voluntary cash payment pursuant to subsection (h) of Section 262 and to assert, in any appraisal proceeding, that, for purposes of Section 262, the “fair value” of a share of Palvella capital stock is less than the merger consideration. No representation is made as to the outcome of the appraisal of fair value as determined by the Delaware Court.
Costs of the appraisal proceeding may be determined by the Delaware Court and taxed upon the parties as the Delaware Court deems equitable in the circumstances. Upon the application of a person whose name appears on the verified list who participated in the proceeding and incurred expenses in connection therewith, the Delaware Court may order all or a portion of the expenses, including, without limitation, reasonable attorneys’ fees and the fees and expenses of experts, to be charged pro rata against the value of all shares entitled to appraisal that were not dismissed or subject to an award pursuant to a reservation of jurisdiction pursuant to subsection (k) of Section 262. In the absence of such a determination or assessment, each party bears its own expenses. Any person who has demanded appraisal rights will not, after the Effective Time, be entitled to vote shares subject to that demand for any purpose or to receive payments of dividends or any other distribution with respect to those shares, other than with respect to payment to stockholders of record of Palvella as of a record date prior to the Effective Time.
To the extent there are any inconsistencies between the foregoing summary, on the one hand, and Section 262, on the other hand, Section 262 will govern.
Failure to follow the steps required by Section 262 for perfecting appraisal rights will result in the loss of appraisal rights. In view of the complexity of Section 262, persons who may wish to exercise appraisal rights, or preserve their right to do so, should consult their legal advisors.
The following is a summary of the material terms of the Merger Agreement. A copy of the Merger Agreement is attached to this proxy statement/prospectus as Annex A and is incorporated by reference into this proxy statement/prospectus. The Merger Agreement has been attached to this proxy statement/prospectus to provide you with information regarding its terms. It is not intended to provide any other factual information about Pieris, Palvella or Merger Sub. The following description does not purport to be complete and is qualified in its entirety by reference to the Merger Agreement. You should refer to the full text of the Merger Agreement for details of the Merger and the terms and conditions of the Merger Agreement.
The Merger Agreement contains representations and warranties that Pieris and Merger Sub, on the one hand, and Palvella, on the other hand, have made to one another as of specific dates. These representations and warranties have been made for the benefit of the other parties to the Merger Agreement and may be intended not as statements of fact but rather as a way of allocating the risk to one of the parties if those statements prove to be incorrect. In addition, the assertions embodied in the representations and warranties are qualified by information in confidential disclosure schedules exchanged by the parties in connection with signing the Merger Agreement. While Pieris and Palvella do not believe that these disclosure schedules contain information required to be publicly disclosed under the applicable securities laws, other than information that has already been so disclosed, the disclosure schedules do contain information that modifies, qualifies and creates exceptions to the representations and warranties set forth in the attached Merger Agreement. Accordingly, you should not rely on the representations and warranties as current characterizations of factual information about Pieris or Palvella, because they were made as of specific dates, may be intended merely as a risk allocation mechanism between Pieris, Merger Sub and Palvella and are modified by the disclosure schedules.
Subject to the terms and conditions of the Merger Agreement, and in accordance with Delaware law, at the completion of the Merger, Merger Sub, a wholly-owned subsidiary of Pieris formed by Pieris in connection with the Merger, will merge with and into Palvella, with Palvella surviving the Merger as a wholly owned subsidiary of Pieris.
Completion and Effectiveness of the Merger
The Merger will be completed as promptly as practicable after all of the conditions to completion of the Merger are satisfied or waived, including the approval by the stockholders of Pieris and Palvella. Pieris and Palvella are working to complete the Merger as quickly as practicable and expect that the Merger will be completed soon after the Pieris special meeting scheduled to be held on Wednesday, December 11, 2024 at 9:00 a.m. Eastern Time. However, Pieris and Palvella cannot predict the completion of the Merger or the exact timing of the completion of the Merger because it is subject to various conditions.
In connection with the Merger, each stock option granted under the Palvella Stock Plan that is outstanding immediately prior to the Effective Time, will be assumed by Pieris and will become an option to acquire, on the same terms and conditions as were applicable to such Palvella stock option immediately prior to the Effective Time, a number of shares of Pieris common stock equal to the number of shares of Palvella common stock subject to the unexercised portion of the Palvella stock option immediately prior to the Effective Time, multiplied by the exchange ratio (rounded down to the nearest whole share number), with an exercise price per share for the options equal to the exercise price per share of such Palvella stock option immediately prior to the Effective Time divided by the exchange ratio (rounded up to the nearest whole cent). Such assumed options will continue to be governed by the terms and conditions of the Palvella Stock Plan, including the adjustment and change in control provisions contained therein.
Treatment of Pieris Common Stock and Pieris Options
Each share of Pieris common stock and option to purchase Pieris common stock that is issued and outstanding at the Effective Time will remain issued and outstanding and such shares and options will be unaffected by the Merger.
Directors and Officers of Pieris Following the Merger
Pursuant to the Merger Agreement, each of the directors and officers of Pieris who will not continue as directors or officers of the combined company following the consummation of the Merger will resign effective as of the Effective Time. Effective as of the Effective Time, the combined company board of directors will consist of a total of five directors, one of whom will be designated by Pieris, namely Christopher Kiritsy, and four of whom will be designated by Palvella, namely Wesley H. Kaupinen, Todd C. Davis, George M. Jenkins and Tadd S. Wessel. In addition, upon the Effective Time, Wesley H. Kaupinen will serve as President and Chief Executive Officer, Matthew Korenberg will serve as Chief Financial Officer, Kathleen Goin will serve as Chief Operating Officer, Jeffrey Martini, Ph.D., will serve as Chief Scientific Officer, and Kathleen A. McGowan will serve as Vice President, Finance and Operations of the combined company.
Representations and Warranties
The Merger Agreement contains customary representations and warranties of Pieris, Merger Sub and Palvella for a transaction of this type relating to, among other things:
|
● |
corporate organization, standing and power, and similar corporate matters; |
|
● |
capitalization; |
|
● |
subsidiaries; |
|
● |
authority to enter into the Merger Agreement and the related agreements and the absence of certain conflicts; |
|
● |
financial statements and, with respect to Pieris, documents filed with the SEC and the accuracy of information contained in those documents; |
|
● |
liabilities; |
|
● |
material changes or events; |
|
● |
tax matters; |
|
● |
real property and leaseholds; |
|
● |
intellectual property; |
|
● |
contracts; |
|
● |
litigation; |
|
● |
environmental matters; |
|
● |
employee benefit plans; |
|
● |
compliance with laws; |
|
● |
permits and regulatory matters; |
|
● |
employee matters; |
|
● |
insurance; |
|
● |
brokers, fees and expenses; |
|
● |
certain transactions or relationships with affiliates; |
|
● |
internal controls and procedures; |
|
● |
books and records; |
|
● |
privacy and data protection; |
|
● |
with respect to Pieris, the opinion of Pieris’ financial advisor; |
|
● |
with respect to Pieris, matters related to the valid issuance in the Merger of Pieris common stock; |
|
● |
with respect to Pieris, matters related to NRS 78.411 through 78.444; |
|
● |
with respect to Pieris, the operations of Merger Sub; |
|
● |
with respect to Pieris, compliance with the Merger Agreement; and |
|
● |
with respect to Palvella, ownership of Pieris common stock. |
The representations and warranties are, in many respects, qualified by materiality and knowledge, and will not survive the Merger, but their accuracy forms the basis of one of the conditions to the obligations of Pieris, Merger Sub and Palvella to complete the Merger.
Covenants; Conduct of Business Pending the Merger
Pieris has agreed that, except as permitted by the Merger Agreement or unless Palvella has provided written consent, or to the extent necessary to comply with any applicable law or as required in connection with the PIPE Financing, during the period commencing on the date of the Merger Agreement and continuing until the earlier to occur of the Effective Time and the termination of the Merger Agreement, Pieris and its subsidiaries will use commercially reasonably efforts to, act and carry on its business in the ordinary course of business. Pieris has also agreed that, except as permitted by the Merger Agreement or unless Palvella has provided written consent, or to the extent necessary to comply with any applicable law or as required in connection with the PIPE Financing, it will not, and will not cause or permit any of its subsidiaries to, during the period commencing on the date of the Merger Agreement and continuing until the earlier to occur of the Effective Time and the termination of the Merger Agreement:
|
● |
(i) subject to certain exceptions, declare, set aside, or pay any dividends on, or make any other distributions (whether in cash, securities or other property) in respect of, any of its capital stock; (ii) split, combine or reclassify any of its capital stock or issue or authorize the issuance of any other securities in respect of, in lieu of or in substitution for shares of its capital stock or any of its other securities; or (iii) subject to certain exceptions, purchase, redeem or otherwise acquire any shares of its capital stock or any other of its securities or any rights, warrants or options to acquire any such shares or other securities; |
|
● |
subject to certain exceptions, issue, deliver, sell, grant, pledge or otherwise dispose of or encumber any shares of its capital stock, any other voting securities or any securities convertible into or exchangeable for, or any rights, warrants or options to acquire, any such shares, voting securities or convertible or exchangeable securities; |
|
● |
except as required or appropriate to give effect to anything in contemplation of the closing of the Merger, amend its amended and restated articles of incorporation, amended and restated bylaws or other comparable charter or organizational documents or effect or be a party to any merger, consolidation, share exchange, business combination, liquidation, dissolution, reorganization, statutory conversion, recapitalization, reclassification of shares, stock split or reverse stock split or form any new subsidiary or acquire any equity interest or other interest in any other person; |
|
● |
acquire (i) by merging or consolidating with, or by purchasing all or a substantial portion of the assets or any stock of, or by any other manner, any business or any corporation, partnership, joint venture, limited liability company, association or other business organization or division thereof or (ii) any assets that are material, individually or in the aggregate, to Pieris and its subsidiaries, taken as a whole; |
|
● |
sell, lease, license, pledge, or otherwise dispose of or encumber any properties or assets material to Pieris or any of its subsidiaries; |
|
● |
enter into any material transaction; |
|
● |
license any material intellectual property rights to or from any third party; |
|
● |
(i) incur or suffer to exist any indebtedness for borrowed money or guarantee any such indebtedness of another person, (ii) issue, sell or amend any debt securities or warrants or other rights to acquire any debt securities of Pieris or any of its subsidiaries, guarantee any debt securities of another person, enter into any “keep well” or other agreement to maintain any financial statement condition of another person or enter into any arrangement having the economic effect of any of the foregoing, (iii) make any loans, advances (other than routine advances to employees of Pieris in the ordinary course of business) or capital contributions to, or investment in, any other person, other than Pieris or any of its direct or indirect wholly owned subsidiaries or (iv) enter into any hedging agreement or other financial agreement or arrangement designed to protect Pieris or its subsidiaries against fluctuations in commodities prices or exchange rates; |
|
● |
forgive any loans to any person, including its employees, officers, directors or affiliates; |
|
● |
enter into any agreement to purchase or sell any interest in real property, grant any security interest in any real property, enter into any lease, sublease, license or other occupancy agreement with respect to any real property or alter, amend, modify any agreement that terminated any Pieris lease; |
|
● |
make any (i) capital expenditures or other expenditures with respect to property, plant or equipment or (ii) other material expenditures in excess of $50,000 in the aggregate (other than any expenditures in the ordinary course of business); |
|
● |
make any changes in accounting methods, principles or practices, except insofar as may have been required by the SEC or a change in GAAP or, except as so required, change any assumption underlying, or method of calculating, any bad debt, contingency or other reserve; |
|
● |
subject to certain exceptions, (i) modify or amend in any material respect, or terminate, any material contract or agreement to which Pieris or any of its subsidiaries is party, or (ii) knowingly waive, release or assign any material rights or claims (including any write-off or other compromise of any accounts receivable of Pieris or any of its subsidiaries); |
|
● |
(i) enter into any contract or agreement, including those relating to the rendering of services or the distribution, sale or marketing by third parties of the products, of, or products licensed by, Pieris or any of its subsidiaries or (ii) license any material intellectual property rights to or from any third party; |
|
● |
except as required to comply with any Pieris employee plan, (i) take any action with respect to, adopt, enter into, terminate (other than termination for cause) or amend any Pieris employee plan (or any other employee benefit or compensation plan, program, policy, agreement or arrangement that would have constituted a Pieris employee plan had it been in effect on the date of the Merger Agreement) or any collective bargaining agreement, (ii) increase the compensation (including any compensation opportunities) or fringe benefits of, or pay any bonus or grant any bonus opportunity to, any director, officer, employee or consultant, (iii) amend or accelerate the payment, right to payment or vesting of any compensation or benefits, including any outstanding equity or equity-based incentive awards, (iv) pay any benefit not provided for as of the date of the Merger Agreement under any benefit plan under any Pieris employee plan, (v) grant any awards under any Pieris employee plan (or under any other employee benefit or compensation plan, program, policy, agreement or arrangement that would have constituted a Pieris employee plan had it been in effect on the date of the Merger Agreement), or (vi) take any action other than in the ordinary course of business to fund or in any other way secure the payment of compensation or benefits under any Pieris employee plan (or under any other employee benefit or compensation plan, program, policy, agreement or arrangement that would have constituted a Pieris employee plan had it been in effect on the date of the Merger Agreement); |
|
● |
make, change or revoke any tax election (other than elections made in the ordinary course of business), change an annual accounting period, enter into any closing agreement, waive or extend any statute of limitations with respect to taxes (other than any automatic extension granted in the ordinary course of business and consistent with past custom and practice of Pieris), settle or compromise any material tax liability, claim or assessment, knowingly surrender any right to claim a refund of material taxes, or amend any income or other material tax return; |
|
● |
commence any offering of shares of Pieris common stock, including pursuant to any employee stock purchase plan; |
|
● |
subject to certain exceptions, initiate, threaten, compromise or settle any litigation or arbitration proceeding (other than any litigation to enforce its rights under the Merger Agreement); |
|
● |
fail to use commercially reasonable efforts to maintain insurance levels substantially comparable to levels existing as of the date of the Merger Agreement; |
|
● |
open or close any facility or office; |
|
● |
delay or fail to pay accounts payable and other obligations when due; or |
|
● |
authorize any of, or commit or agree, in writing or otherwise, to take any of, the foregoing actions or any action that would reasonably be expected to, individually or in the aggregate, make any representation or warranty of Pieris in the Merger Agreement untrue or incorrect in any material respect, or would materially impair, delay or prevent the satisfaction of any conditions to obligations of the parties to effect the Merger. |
Palvella has agreed that, except as permitted by the Merger Agreement or as consented to in writing by Pieris, or to the extent necessary to comply with any applicable law or as required in connection with the PIPE Financing, during the period commencing on the date of the Merger Agreement and continuing until the earlier to occur of the Effective Time and the termination of the Merger Agreement, Palvella will use commercially reasonable efforts to, act and carry on its business in the ordinary course of business. Palvella has also agreed that, except as permitted by the Merger Agreement or unless Pieris has provided written consent, or to the extent necessary to comply with any applicable law or as required in connection with the PIPE Financing, it will not, during the period commencing on the date of the Merger Agreement and continuing until the earlier to occur of the Effective Time and the termination of the Merger Agreement:
|
● |
(i) declare, set aside or pay any dividends on, or make any other distributions (whether in cash, securities or other property) in respect of, any of its capital stock; (ii) split, combine or reclassify any of its capital stock or issue or authorize the issuance of any other securities in respect of, in lieu of or in substitution for shares of its capital stock or any of its other securities, other than the issuance of shares upon exercise or conversion of any Palvella preferred stock, Palvella stock options, or other convertible securities of Palvella; or (iii) subject to certain exceptions, purchase, redeem or otherwise acquire any shares of its capital stock or any other of its securities or any rights, warrants or options to acquire any such shares or other securities; |
|
● |
subject to certain exceptions, issue, deliver, sell, grant, pledge or otherwise dispose of or encumber any shares of its capital stock, any other voting securities or any securities convertible into or exchangeable for, or any rights, warrants or options to acquire, any such shares, voting securities or convertible or exchangeable securities; |
|
● |
except as required to give effect to anything in contemplation of the closing of the Merger, amend its certificate of incorporation, bylaws or other comparable charter or organizational documents or effect or be a party to any merger, consolidation, share exchange, business combination, liquidation, dissolution, reorganization, statutory conversion, recapitalization, reclassification of shares, stock split or reverse stock split or form any new subsidiary or acquire any equity interest or other interest in any other person; |
|
● |
acquire, by merging or consolidating with, or by purchasing all or a substantial portion of the assets or any stock of, or by any other manner, any business or any corporation, partnership, joint venture, limited liability company, association or other business organization or division thereof; |
|
● |
except in the ordinary course of business, sell, lease, license, pledge, or otherwise dispose of or encumber any material properties or assets material to Palvella; |
|
● |
enter into any material transaction other than in the ordinary course of business; |
|
● |
license any material intellectual property to or from any third party; |
|
● |
initiate, threaten, compromise or settle any litigation or arbitration proceeding (other than any litigation to enforce its rights under the Merger Agreement) other than waivers, settlements or agreements which (i) do not exceed, in the aggregate, $200,000 more than the amounts covered by insurance policies (ii) do not impose any material restrictions on the operations or businesses of Palvella (including following the Merger and/or the surviving corporation), taken as a whole, (C) do not provide for equitable relief on, or the admission of wrongdoing by Palvella, and (D) do not relate to claims, litigations, investigations or proceedings brought by a governmental entity; |
|
● |
subject to certain exceptions, (i) incur or suffer to exist any indebtedness for borrowed money or guarantee any such indebtedness of another person in excess of $200,000, (ii) issue, sell, or amend any debt securities or warrants or other rights to acquire any debt securities of Palvella, guarantee any debt securities of another person, enter into any “keep well” or other agreement to maintain any financial statement condition of another person, or enter into any arrangement having the economic effect of any of the foregoing, or (iii) make any loans, advances (other than routine advances to employees of Palvella in the ordinary course of business) or capital contributions to, or investment in, any other person; |
|
● |
make, change or revoke any tax election (other than elections made in the ordinary course of business), change an annual accounting period, enter into any closing agreement, waive or extend any statute of limitations with respect to taxes (other than any automatic extension granted in the ordinary course of business and consistent with past custom and practice of Palvella), settle or compromise any material tax liability, claim or assessment, knowingly surrender any right to claim a refund of material taxes, or amend any income or other material tax return; or |
|
● |
authorize any of, or commit or agree, in writing or otherwise, to take any of, the foregoing actions or any action that would reasonably be expected to, individually or in the aggregate, (i) make any representation or warranty of Palvella in the Merger Agreement untrue or incorrect, or (ii) impair, delay or prevent the satisfaction of any conditions to obligations of the parties to effect the Merger. |
Prior to the Effective Time, Pieris will declare a dividend to each holder of record of shares of its common stock and shares of its preferred stock entitled to such a dividend in accordance with its terms the right to receive one CVR for each outstanding share of Pieris common stock held by such stockholder as of such date, each representing the contractual right to receive payments upon the receipt of payments by Pieris or any of its affiliates under certain strategic partner agreements, including existing collaboration agreements pursuant to which Pieris may be entitled to milestones and royalties in the future and other out-licensing agreements for certain of Pieris’ legacy assets, which Pieris may enter into prior to Closing, and upon the receipt of certain research and development tax credits in favor of Pieris or any of its affiliates, in each case as set forth in, and subject to and in accordance with the terms and conditions of, the CVR Agreement in the form attached to the Merger Agreement, discussed in greater detail under the section titled “Agreements Related to the Merger—CVR Agreement” beginning on page 182 in this proxy statement/prospectus. The record date for such dividend will be the close of business on the last business day prior to the day on which the Effective Time occurs and the payment date for which shall be three business days after the Effective Time; provided that the payment of such dividend may be conditioned upon the occurrence of the Effective Time. In connection with such dividend, Pieris will cause the CVR Agreement to be duly authorized, executed and delivered by Pieris and a rights agent selected by Pieris with Palvella’s prior approval (such approval not to be unreasonably withheld, delayed or conditioned).
Each of Pieris and Palvella have agreed that, except as described below, Pieris and Palvella and any of their respective subsidiaries will not, and each party will cause their respective directors, officers, employees and consultants not to, and will instruct their respective attorneys and financial advisors not to, directly or indirectly:
|
● |
solicit, seek or initiate or knowingly take any action to facilitate or encourage any offers, inquiries or the making of any proposal or offer that constitutes, or would reasonably be expected to lead to, any Acquisition Proposal; |
|
● |
enter into, continue or otherwise participate or engage in any discussions or negotiations regarding any Acquisition Proposal, or furnish to any person any non-public information or afford any person other than Pieris or Palvella, as applicable, access to such party’s property, books or records (except pursuant to a request by a governmental entity) in connection with any offers, inquiries or the making of any proposal or offer that constitutes, or could reasonably be expected to lead to, any Acquisition Proposal; |
|
● |
take any action to make the provisions of any “fair price,” “business combination” or “control share acquisition” statute or other similar statute or regulation inapplicable to any transactions contemplated by an Acquisition Proposal; or |
|
● |
publicly propose to do any of the foregoing. |
An “Acquisition Proposal” means, with respect to Pieris or Palvella, (a) any inquiry, proposal or offer for a merger, consolidation, dissolution, sale of substantial assets, recapitalization, share exchange, tender offer or other business combination involving such party and its subsidiaries, other than mergers, consolidations, recapitalizations, share exchanges or other business combinations involving solely such party and/or one or more subsidiaries of such party, (b) any proposal for the issuance by such party of 15% or more of its equity securities, or (c) any proposal or offer to acquire in any manner, directly or indirectly, 15% or more of the equity securities or consolidated total assets of such party and its subsidiaries, in each case other than the transactions contemplated by the Merger Agreement; provided, however, that no inquiry, proposal, or offer received pursuant to the terms of or in connection with the PIPE Financing or any Legacy Asset Disposition (as defined in the Merger Agreement) shall be an Acquisition Proposal.
Notwithstanding the restrictions described above or anything to the contrary set forth in the Merger Agreement, subject to compliance with the terms of the Merger Agreement, before the earliest to occur of: the Effective Time, the applicable party obtaining the approval of its respective stockholders required to consummate the Merger, or the termination of the Merger Agreement, each party, and their respective representatives may (A) furnish non-public information with respect to Pieris and its subsidiaries or Palvella, as the case may be, to any third party (and the representatives of such third party), or (B) engage in discussions or negotiations (including solicitation of revised Acquisition Proposal) with any third party (and the representatives of such third party) regarding any bona fide, unsolicited written Acquisition Proposal which such party’s board of directors determines in good faith, after consultation with such party’s financial advisors and outside legal counsel, constitute a Superior Proposal (and is not withdrawn), provided:
|
● |
that such party receives from the third party an executed confidentiality agreement containing terms not less restrictive than those contained in the confidentiality agreement between Pieris and Palvella; |
|
● |
that such party has not materially breached the non-solicitation provisions of the Merger Agreement; and |
|
● |
that such party’s board of directors has determined in good faith, after consultation with outside legal counsel, that the failure to take such actions would reasonably be expected to be inconsistent with the fiduciary duties of such board of directors under applicable law. |
A “Superior Proposal” means, with respect to Pieris or Palvella, any bona fide, unsolicited written Acquisition Proposal (replacing all references in such definition to 15% with 50%), (a) on terms which the board of directors of such party determines in its good faith judgment to be more favorable to the holders of such party’s capital stock from a financial point of view than the transactions contemplated by the Merger Agreement, after consultation with its financial and outside legal advisors, taking into account all the terms and conditions of such proposal and the Merger Agreement (including any termination or break-up fees and conditions to consummation, as well as any written, binding offer by the other party hereto to amend the terms of the Merger Agreement, which offer is not revocable for at least four Business Days) that the board of directors of such party determines to be relevant, and (b) which board of directors of such party has determined to be reasonably capable of being completed on the terms proposed, taking into account all financial, regulatory, legal and other aspects of such proposal that board of directors of such party determines to be relevant (including the likelihood and timing of consummation as compared to the transactions contemplated by the Merger Agreement).
The Merger Agreement also provides that each party will as promptly as reasonably practicable advise the other of the status and terms of, and keep the other party reasonably informed with respect to, any Acquisition Proposal or any material change or proposed material change to that Acquisition Proposal. Such party in receipt of an Acquisition Proposal must provide the other party with written notice of the first decision by its board of directors to consider any Acquisition Proposal, to enter into discussions or negotiations concerning any Acquisition Proposal or to provide non-public information with respect to such to any person.
Under the Merger Agreement, subject to certain exceptions described below, Pieris agreed that its board of directors (and any committee thereof) may not take any of the following actions, each of which are referred to in this proxy statement/prospectus as a Pieris board of directors recommendation change:
|
● |
fail to include its recommendation to Pieris stockholders in connection with the solicitation of their approval of the share issuance and the amendment to Pieris’ amended and restated articles of incorporation to increase the number of authorized shares of Pieris common stock and ratification of the Pieris board of directors’ approval of the amendment to Pieris’ amended and restated articles of incorporation to change the name of Pieris to “Palvella Therapeutics, Inc.” at the Pieris special meeting in this proxy statement/prospectus or shall have withdrawn or modified such recommendation in a manner adverse to Palvella; |
|
● |
withhold, withdraw or modify, or publicly propose to withhold, withdraw or modify, the approval or recommendation of the Pieris board of directors with respect to the share issuance or the amendment to Pieris’ amended and restated articles of incorporation to increase the number of authorized shares of Pieris common stock and the amendment to Pieris’ amended and restated articles of incorporation to change the name of Pieris to “Palvella Therapeutics, Inc.”; |
|
● |
after the receipt by Pieris of an Acquisition Proposal and Palvella’s subsequent request in writing that the Pieris board of directors reconfirm its recommendation to Pieris stockholders to solicit their approval of the required Pieris voting proposals at the Pieris special meeting, fail to reconfirm its recommendation within ten business days after its receipt of Palvella’s request; |
|
● |
fail to recommend against acceptance of a tender offer within ten business days after commencement; or |
|
● |
propose publicly to approve, endorse, adopt or recommend, or has approved, endorsed, adopted or recommended any Acquisition Proposal. |
However, notwithstanding the foregoing, at any time prior to the approval of the proposals to be considered at the Pieris special meeting by the necessary vote of Pieris stockholders, with respect to a Superior Proposal, the Pieris board of directors may make a Pieris board of directors recommendation change if:
|
● |
the Pieris board of directors determines in good faith, after consultation with outside legal counsel, that the failure to make a Pieris board of directors recommendation change would reasonably be expected to be inconsistent with its fiduciary duties under applicable law; |
|
● |
Pieris has provided at least four business days’ prior written notice to Palvella that it intends to effect a Pieris board of directors recommendation change and written copies of any relevant proposed transactions agreements with any party making a potential Superior Proposal, including the identity of the person making such Superior Proposal; |
|
● |
Pieris has complied in all material respects with the non-solicitation provisions of the Merger Agreement in connection with any potential Superior Proposal; |
|
● |
Pieris has, and has caused its financial advisors and outside legal counsel to, during the applicable four business days period, negotiate with Palvella in good faith to make such adjustments to the terms and conditions of the Merger Agreement so that such Acquisition Proposal cases to constitute a Superior Proposal (to the extent Palvella desires to negotiate); and |
|
● |
if after Palvella has delivered to Pieris a written, binding and irrevocable offer to alter the terms or conditions of the Merger Agreement during the applicable four business day notice period, the Pieris board of directors has determined in good faith, after consultation with outside legal counsel and after considering the terms of such offer by Palvella, that the failure to effect a Pieris board of directors recommendation change would reasonably be expected to be inconsistent with its fiduciary duties under applicable law. |
Under the Merger Agreement, subject to certain exceptions described below, Palvella agreed that its board of directors may not take any of the following actions, each of which are referred to in this proxy statement/prospectus as a Palvella board of directors recommendation change:
|
● |
withhold, withdraw or modify (or publicly propose to withhold, withdraw or modify) the approval or recommendation of the Palvella board of directors with respect to the Merger; |
|
● |
fail to recommend against acceptance of a tender offer within ten business days after commencement; or |
|
● |
propose publicly to approve, endorse, adopt or recommend, or has approved, endorsed, adopted, or recommended any Acquisition Proposal. |
However, notwithstanding the foregoing or anything to the contrary set forth in the Merger Agreement, at any time prior to the approval of the Merger by the necessary vote of Palvella stockholders, with respect to a Superior Proposal, the Palvella board of directors may make a Palvella board of directors recommendation change if, but only if:
|
● |
the Palvella board of directors determines in good faith, after consultation with outside legal counsel, that the failure to make a Palvella board of directors recommendation change would reasonably be expected to be inconsistent with its fiduciary duties under applicable law; |
|
● |
Palvella has provided at least four business days’ prior written notice to Pieris that it intends to effect a Palvella board of directors recommendation change and written copies of any relevant proposed transactions agreements with any party making a potential Superior Proposal, including the identity of the person making such Superior Proposal; |
|
● |
Palvella has complied in all material respects with the non-solicitation provisions of the Merger Agreement in connection with any potential Superior Proposal; |
|
● |
Palvella has, and has caused its financial advisors and outside legal counsel to, during the applicable four business days period, negotiate with Pieris in good faith to make such adjustments to the terms and conditions of the Merger Agreement so that such Acquisition Proposal cases to constitute a Superior Proposal (to the extent Pieris desires to negotiate); and |
|
● |
if after Pieris has delivered to Palvella a written, binding and irrevocable offer to alter the terms or conditions of the Merger Agreement during the applicable four business day notice period, the Palvella board of directors has determined in good faith, after consultation with outside legal counsel and after considering the terms of such offer by Pieris, that the failure to effect a Palvella board of directors recommendation change would reasonably be expected to be inconsistent with its fiduciary duties under applicable law. |
Meeting of the Pieris Stockholders and Written Consent of the Palvella Stockholders
Pieris is obligated under the Merger Agreement to take all action necessary under applicable law, its amended and restated articles of incorporation and amended and restated bylaws, and Nasdaq rules to duly call, give notice of, convene and hold a meeting of the holders of Pieris common stock for the purpose of considering and voting to approve the proposals. The Pieris special meeting will be held as promptly as practicable after the Registration Statement on Form S-4 is declared effective under the Securities Act, and in any event no later than 35 days after the effective date of the Registration Statement on Form S-4.
Promptly after the Registration Statement on Form S-4 has been declared effective under the Securities Act, and in any event no later than two business days thereafter, Palvella will solicit and obtain the consent of Palvella stockholders for purposes of (i) evidencing the adoption of the Merger Agreement and the approval of the Merger and the other transactions contemplated therein, (ii) acknowledging that the approval given thereby is irrevocable and that such stockholder is aware of its rights to demand appraisal for its shares pursuant to Section 262 of the DGCL, a copy of which will be attached thereto or incorporated by reference therein, and that such stockholder has received and read a copy of Section 262 of the DGCL and (iii) acknowledging that by its approval of the Merger it is not entitled to appraisal rights with respect to its shares in connection with the Merger and thereby waives any rights to receive payment of the fair value of the Palvella capital stock under Section 262 of the DGCL.
Indemnification and Insurance for Directors and Officers
Under the Merger Agreement, from the Effective Time through the sixth anniversary of the date on which the Effective Time occurs, Pieris and the surviving corporation in the Merger have agreed to indemnify and hold harmless each person who was at the time of the execution of the Merger Agreement, or has been at any time prior to the date of the Merger Agreement, or who becomes prior to the Effective Time, a director or officer of Pieris or Palvella or any of their respective subsidiaries, against all claims, losses, liabilities, damages, judgments, fines and reasonable fees, costs and expenses, including attorneys’ fees and disbursements, incurred in connection with any claim, action, suit, proceeding or investigation, whether civil, criminal, administrative or investigative, arising out of or pertaining to the fact that the indemnified person is or was an officer, director, employee or agent of Pieris or of Palvella or any of their respective subsidiaries, whether asserted or claimed prior to, at or after the Effective Time, to the extent permitted under the applicable certificate or articles of incorporation and bylaws.
From the Effective Time through the sixth anniversary of the date on which the Effective Time occurs, the certificate of incorporation and bylaws of the combined company will contain provisions at least as favorable as the provisions relating to indemnification, advancement of expenses and elimination of liability for monetary damages as those set forth in the certificate or articles of incorporation and bylaws of Pieris and Palvella immediately prior to the Effective Time.
Each of Pieris and Palvella will secure and purchase a six year “tail policy” on their respective existing directors’ and officers’ liability insurance policies.
Each of Pieris and Palvella has agreed to use reasonable best efforts to:
|
● |
take, or cause to be taken, all actions, and do, or cause to be done, and to assist and cooperate with the other parties in doing, all things necessary, proper or advisable to consummate and make effective the transactions contemplated by the Merger Agreement as promptly as practicable; |
|
● |
as promptly as practicable, obtain from any governmental entity or any other third party any consents, licenses, permits, waivers, approvals, authorizations, or orders required to be obtained or made by Pieris or Palvella or any of their subsidiaries in connection with the authorization, execution and delivery of the Merger Agreement and the consummation of the transactions contemplated by the Merger Agreement; |
|
● |
as promptly as practicable, make all necessary filings, and thereafter make any other required submissions, with respect to the Merger Agreement and the Merger required under (a) the Securities Act and the Exchange Act, and any other applicable federal or state securities laws and (b) any other applicable laws; and |
|
● |
execute or deliver any additional instruments necessary to consummate the transactions contemplated by, and to fully carry out the purposes of, the Merger Agreement. |
Pursuant to the Merger Agreement, Pieris and Palvella have further agreed that:
|
● |
Pieris will use its commercially reasonable efforts, and shall take all reasonably necessary actions, to continue the listing of Pieris common stock on Nasdaq during the term of the Merger Agreement (through and until the Effective Time) and to cause the shares of Pieris common stock being issued in the Merger to be approved for listing, subject to official notice of issuance, on Nasdaq at or prior to the Effective Time; and |
|
● |
Palvella will cooperate with Pieris with respect to the listing application for the Pieris common stock and promptly furnish to Pieris all information concerning Palvella and its officers, directors and equityholders and such other matters that may be required or reasonably requested in connection with the Nasdaq listing. |
Conditions to the Completion of the Merger
The following contains a description of the material conditions to the completion of the Merger. Each party’s obligation to complete the Merger is subject to the satisfaction or waiver by each of the parties (to the extent permitted by law), at or prior to the closing, of various conditions, which include the following:
|
● |
the adoption of the Merger Agreement having been approved by means of written consents by the requisite vote of the stockholders of Palvella under applicable law and Palvella’s certificate of incorporation. The share issuance and the amendment to Pieris’ amended and restated articles of incorporation to increase the number of authorized shares of Pieris common stock and the Pieris board of directors’ approval of the amendment to Pieris’ amended and restated articles of incorporation to change the name of Pieris to “Palvella Therapeutics, Inc.” having been approved and ratified, respectively, at a meeting of Pieris stockholders, at which a quorum is present, by the requisite vote of the stockholders of Pieris under applicable law and stock market regulations; |
|
● |
the Registration Statement on Form S-4, of which this proxy statement/prospectus is a part having become effective under the Securities Act and no stop order suspending the effectiveness of the Registration Statement on Form S-4 shall have been issued and no proceeding for that purpose, and no similar proceeding with respect to this proxy statement/prospectus, shall have been initiated or threatened in writing by the SEC or its staff; |
|
● |
no governmental entity of competent jurisdiction having enacted, issued, promulgated, enforced or entered any order, executive order, stay, decree, judgment or injunction (preliminary or permanent) or statute, rule or regulation which is in effect and which has the effect of making the Merger illegal or otherwise prohibiting consummation of the Merger; |
|
● |
the approval of the listing of the additional shares of Pieris common stock on Nasdaq having been obtained and the shares of Pieris common stock to be issued in the Merger pursuant to the Merger Agreement having been approved for listing, subject to official notice of issuance, on Nasdaq; |
|
● |
the PIPE Financing having been consummated or being consummated concurrently with the closing of the Merger or immediately following the closing of the Merger in accordance with the terms of the Purchase Agreement; and |
|
● |
Pieris’ final net cash having been finally determined in accordance with the Merger Agreement. |
Notwithstanding the foregoing, certain closing conditions may not be waived due to applicable law or otherwise. The following closing conditions may not be waived: receipt of the requisite stockholder approvals; the effectiveness of the Registration Statement on Form S-4 of which this proxy statement/prospectus forms a part; and the absence of any order or injunction that has the effect of prohibiting the consummation of the Merger.
In addition, the obligation of Pieris and Merger Sub to complete the Merger is further subject to the satisfaction or waiver of the following conditions, any of which may be waived exclusively by Pieris and Merger Sub:
|
● |
the representations and warranties of Palvella regarding certain matters related to corporate organization and power, and similar corporate matters, capitalization, authority to enter into the Merger Agreement and the related agreements and lack of certain conflicts, and material changes or events must be true and correct on the date of the Merger Agreement and on the closing date of the Merger with the same force and effect as if made on the date on which the Merger is to be completed or, if such representations and warranties address matters as of a particular date, then as of that particular date, except where the failure to be true and correct (giving effect to any references therein to materiality qualifications), individually or in the aggregate is not material to Palvella; |
|
● |
the remaining representations and warranties of Palvella in the Merger Agreement must be true and correct on the date of the Merger Agreement and on the closing date of the Merger with the same force and effect as if made on the date on which the Merger is to be completed or, if such representations and warranties address matters as of a particular date, then as of that particular date, except in each case, or in the aggregate, where the failure to be so true and correct would not reasonably be expected to have a material adverse effect on Palvella (without giving effect to any references therein to materiality qualifications); |
|
● |
Palvella must have performed in all material respects all obligations required to be performed by it under the Merger Agreement on or prior to the closing date of the Merger; |
|
● |
no material adverse effect on Palvella having occurred since the date of the Merger Agreement and be continuing; |
|
● |
Pieris must have received an officers’ certificate duly executed by Palvella’s chief executive officer to the effect that certain closing conditions have been satisfied; |
|
● |
certain specified Palvella contracts must have been terminated; and |
|
● |
Palvella must have obtained certain specified consents and approvals of third parties, and any other consents or approvals of third parties (other than a governmental entity) the failure of which to obtain, individually or in the aggregate, is reasonably likely to have a material adverse effect on Palvella. |
In addition, the obligation of Palvella to complete the Merger is further subject to the satisfaction or waiver of the following conditions, any of which may be waived exclusively by Palvella:
|
● |
the representations and warranties regarding certain matters related to corporate organization and power, and similar corporate matters, capitalization, authority to enter into the Merger Agreement and the related agreements and lack of certain conflicts, and material changes or events must be true and correct on the date of the Merger Agreement and on the closing date of the Merger with the same force and effect as if made on the date on which the Merger is to be completed or, if such representations and warranties address matters as of a particular date, then as of that particular date, except where the failure to be true and correct (giving effect to any references therein to materiality or material adverse effect qualifications), individually or in the aggregate is not material to Pieris; |
|
● |
the remaining representations and warranties of Pieris and Merger Sub in the Merger Agreement must be true and correct on the date of the Merger Agreement and on the closing date of the Merger with the same force and effect as if made on the date on which the Merger is to be completed or, if such representations and warranties address matters as of a particular date, then as of that particular date, except in each case, or in the aggregate, where the failure to be so true and correct would not reasonably be expected to have a material adverse effect on Pieris (without giving effect to any references therein to materiality or material adverse effect qualifications); |
|
● |
Pieris and Merger Sub each must have performed in all material respects all obligations required to be performed by it under the Merger Agreement on or prior to the closing date of the Merger; |
|
● |
no material adverse effect on Pieris shall have occurred since the date of the Merger Agreement and be continuing; |
|
● |
Palvella must have received copies of the resignations, effective as of the Effective Time, of each director and officer (for such officers, limited to the offices held by such officers and not to such officer’s employment) of Pieris and its subsidiaries, other than the resignation from the individual designated a director to the Pieris board of directors by Pieris pursuant to the Merger Agreement; |
|
● |
Pieris must have obtained certain specified consents and approvals of third parties, and any other consents or approvals of third parties (other than a governmental entity) the failure of which to obtain, individually or in the aggregate, is reasonably likely to have a material adverse effect on Pieris; and |
|
● |
Palvella must have received an officers’ certificate duly executed by Pieris’ chief executive officer to the effect that certain closing conditions have been satisfied. |
With respect to Palvella, a “material adverse effect” for purposes of the Merger Agreement means any change, effect, event, circumstance or development, or an “Effect”, that, individually or in the aggregate with all other Effects that have occurred through the date of determination, has had, or is reasonably likely to have, a material adverse effect on the business, assets and liabilities, financial condition or results of operations of Palvella, taken as a whole; provided, however, that no Effect, to the extent resulting from or arising out of any of the following, will be deemed to be a material adverse effect with respect to Palvella or be taken into account for purposes of determining whether a material adverse effect with respect to Palvella has occurred or is reasonably likely to occur:
|
● |
changes after the date of the Merger Agreement in prevailing economic or market conditions in the United States or any other jurisdiction (except to the extent those changes have a disproportionate effect on Palvella relative to the other participants in the industry or industries in which Palvella operates); |
|
● |
changes or events after the date of the Merger Agreement affecting the industry or industries in which Palvella operates generally (except to the extent those changes or events have a disproportionate effect on Palvella relative to the other participants in the industry or industries in which Palvella operates); |
|
● |
changes after the date of the Merger Agreement in GAAP or requirements of the interpretation thereof (except to the extent those changes have a disproportionate effect on Palvella relative to the other participants in the industry or industries in which Palvella operates); |
|
● |
changes after the date of the Merger Agreement in laws, rules or regulations of general applicability or interpretations thereof by any governmental entity (except to the extent those changes have a disproportionate effect on Palvella relative to the other participants in the industry or industries in which Palvella operates); |
|
● |
any natural disaster, epidemic, pandemic or other disease outbreak (including the COVID-19 pandemic) or any outbreak of major hostilities or any act of terrorism (except to the extent those changes or events have a disproportionate effect on Palvella relative to the other participants in the industry or industries in which Palvella operates); |
|
● |
the execution, delivery, announcement or performance of the obligations under the Merger Agreement or the announcement, pendency or anticipated consummation of the transactions contemplated by the Merger Agreement; |
|
● |
any failure by Palvella to meet any internal guidance, budgets, plans or forecasts of its revenues, earnings or other financial performance or results of operations (but not the underlying cause of such changes or failures, unless such changes or failures would otherwise be excepted from the definition of material adverse effect); or |
|
● |
any equity holder or derivative litigation arising from or relating to the Merger Agreement or the transactions contemplated by the Merger Agreement. |
With respect to Pieris, a “material adverse effect” for purposes of the Merger Agreement means any Effect that, individually or in the aggregate with all other Effects that have occurred through the date of determination, has had, or is reasonably likely to have, a material adverse effect on the business, assets and liabilities, financial condition or results of operations of Pieris and its subsidiaries, taken as a whole; provided, however, that no Effect, to the extent resulting from or arising out of any of the following, will be deemed to be a material adverse effect with respect to Pieris or be taken into account for purposes of determining whether a material adverse effect with respect to Pieris has occurred or is reasonably likely to occur:
|
● |
changes after the date of the Merger Agreement in prevailing economic or market conditions in the United States or any other jurisdiction in which such entity has substantial business operations (except to the extent those changes have a disproportionate effect on Pieris and its subsidiaries relative to the other participants in their industries); |
|
● |
changes or events after the date of the Merger Agreement affecting the industry or industries in which Pieris and its subsidiaries operate generally (except to the extent those changes or events have a disproportionate effect on Pieris and its subsidiaries relative to the other participants in the industry or industries in which Pieris and its subsidiaries operate); |
|
● |
changes after the date of the Merger Agreement in GAAP or requirements or the interpretation thereof (except to the extent those changes have a disproportionate effect on Pieris and its subsidiaries relative to the other participants in the industry or industries in which Pieris and its subsidiaries operate); |
|
● |
changes after the date of the Merger Agreement in laws, rules or regulations of general applicability or interpretations thereof by any governmental entity (except to the extent those changes have a disproportionate effect on Pieris and its subsidiaries relative to the other participants in the industry or industries in which Pieris and its subsidiaries operate); |
|
● |
any natural disaster, epidemic, pandemic or other disease outbreak (including the COVID-19 pandemic) or any outbreak of major hostilities or any act of terrorism (except to the extent those changes or events have a disproportionate effect on Pieris and its subsidiaries relative to the other participants in the industry or industries in which Pieris and its subsidiaries operate); |
|
● |
a change in the public trading price of Pieris common stock or the implications thereof (it being understood that any Effect causing or giving rise to any such change shall be taken into account for purposes of determining whether a material adverse effect has occurred or is reasonably likely to occur, unless such changes or failures would otherwise be expected from the definition of material adverse effect); |
|
● |
the execution, delivery, announcement or performance of the obligations under the Merger Agreement or the announcement, pendency or anticipated consummation of the transactions contemplated by the Merger Agreement (it being understood that the underlying cause of such changes or failures shall be taken into account for purposes of determining whether a material adverse effect has occurred or is reasonably likely to occur, unless such changes or failures would otherwise be excepted from the definition of material adverse effect); |
|
● |
any failure by Pieris or any of its subsidiaries to meet any public estimates or expectations of its revenue, earnings or other financial performance or results of operations for any period; |
|
● |
any failure by Pieris or any of its subsidiaries to meet any internal guidance, budgets, plans or forecasts of its revenues, earnings or other financial performance or results of operations (it being understood that the underlying cause of such changes or failures shall be taken into account for purposes of determining whether a material adverse effect has occurred or is reasonably likely to occur, unless such changes or failures would otherwise be excepted from the definition of material adverse effect); |
|
● |
any changes in or affecting research and development, preclinical studies, clinical trials or other drug development activities (including the failure to obtain positive results from clinical trials, the occurrence of adverse events or serious adverse events in any clinical trial, development activities or favorable responses from any applicable governmental entity) conducted by or on behalf of Pieris or any of its subsidiaries or licensees in respect of its products or product candidates; |
|
● |
any rejection or non-acceptance by a governmental entity of a registration or filing by Pieris or any of its subsidiaries relating to any intellectual property rights of Pieris or any of its subsidiaries; |
|
● |
regulatory approval of, or regulatory action or announcement with respect to, any products, or product candidates, of a third party that are similar to, or expected to compete against, any of Pieris’ or any of its subsidiaries’ product candidates, including product candidates licensed out to the third parties; |
|
● |
any stockholder or derivative litigation arising from or relating to the Merger Agreement or the transactions contemplated by the Merger Agreement; |
|
● |
any Legacy Asset Disposition; or |
|
● |
other specified matters. |
Termination and Termination Fees
Termination of the Merger Agreement
The Merger Agreement may be terminated at any time before the Effective Time, whether before or (subject to the terms of the Merger Agreement) after the required stockholder approvals to complete the Merger have been obtained, as set forth below:
|
(a) |
by mutual written consent of Pieris and Palvella; |
|
(b) |
by either Pieris or Palvella, if the Merger has not been consummated by March 31, 2025; provided, that the right to terminate the Merger Agreement on or after such date will not be available to any party whose failure to fulfill any obligation under the Merger Agreement has been a principal cause of the failure of the Merger to occur on or before March 31, 2025; provided, that, if as of such date all conditions to the closing of the Merger have been satisfied or waived, such date shall automatically be extended until the date that is two business days following the final determination of Pieris’ final net cash, in accordance with the terms of the Merger Agreement; |
|
(c) |
by either Pieris or Palvella, if a governmental entity of competent jurisdiction has issued a nonappealable final order, decree or ruling or taken any other nonappealable final action that permanently restrains, enjoins or otherwise prohibits the Merger; provided, that this right to terminate the Merger Agreement will not be available to any party if the issuance of any such order, decree, ruling or other action is principally attributable to the failure of such party, or any affiliate of such party, to perform in any material respect any covenant in the Merger Agreement required to be performed by such party, or any affiliate of such party, at or prior to the Effective Time; |
|
(d) |
by either Pieris or Palvella, if the Pieris special meeting (including, if applicable, following any adjournment or postponement thereof as permitted or required under the Merger Agreement) shall have been held and completed and the stockholders of Pieris have taken a final vote on the share issuance, the amendment to Pieris’ amended and restated articles of incorporation to increase the number of authorized shares of Pieris common stock and the Pieris board of directors’ approval of the amendment to Pieris’ amended and restated articles of incorporation to change the name of Pieris to “Palvella Therapeutics, Inc.”, and such proposals have not been approved or ratified, as applicable, by the Pieris stockholders; provided, however, that Pieris may not terminate the Merger Agreement pursuant to this provision if Pieris’ failure to fulfill any obligation under the Merger Agreement has been a principal cause of the failure to obtain the required approval or ratification of the Pieris stockholders; |
|
(e) |
by Pieris, at any time prior to the approval by Palvella stockholders of the adoption of the Merger Agreement, if any of the following circumstances shall occur: |
|
● |
The Palvella board of directors has effected a Palvella board of directors recommendation change; or |
|
● |
Palvella has materially breached certain of its non-solicitation obligations under the Merger Agreement or the obligation to deliver the required stockholder approval under the Merger Agreement; |
|
(f) |
by Palvella, at any time prior to the approval by Pieris stockholders of the share issuance and the amendment to Pieris’ amended and restated articles of incorporation to increase the number of authorized shares of Pieris common stock and the ratification by the Pieris stockholders of approval by the Pieris board of directors of the amendment to Pieris’ amended and restated articles of incorporation to change the name of Pieris to “Palvella Therapeutics, Inc.”, if any of the following circumstances shall occur: |
|
● |
The Pieris board of directors has effected a Pieris board of directors recommendation change; or |
|
● |
Pieris has materially breached certain of its non-solicitation obligations under the Merger Agreement or the obligation to deliver the required stockholder approval under the Merger Agreement; |
|
(g) |
by Pieris, if Palvella has breached or failed to perform any of its representations, warranties, covenants or agreements contained in the Merger Agreement such that certain conditions to the closing of the Merger would not be satisfied; provided that Pieris is not then in material breach of any representation, warranty, or covenant under the Merger Agreement; provided, further, if such breach or failure to perform is curable by Palvella, then the Merger Agreement will not terminate pursuant to this paragraph as a result of such particular breach or failure until the expiration of a 30-day period after delivery of written notice of such breach or failure from Pieris to Palvella (it being understood that the Merger Agreement will not terminate as a result of such particular breach or failure if such breach or failure by Palvella is cured prior to such termination becoming effective); |
|
(h) |
by Palvella, if Pieris has breached or failed to perform any of its representations, warranties, covenants or agreements contained in the Merger Agreement such that certain conditions to the closing of the Merger would not be satisfied; provided that Palvella is not then in material breach of any representation, warranty, or covenant under the Merger Agreement; provided, further, if such breach or failure is curable, then the Merger Agreement will not terminate pursuant to this paragraph as a result of such particular breach or failure until the expiration of a 30-day period after delivery of written notice of such breach or inaccuracy from Palvella to Pieris (it being understood that the Merger Agreement will not terminate as a result of such particular breach or failure if such breach by Pieris is cured prior to such termination becoming effective); |
|
(i) |
by Pieris, if the written consent of Palvella stockholders necessary to adopt the Merger Agreement and approve the Merger and related matters has not been obtained on or prior to 5:00 p.m., New York City time, on the date that is two business days after the Registration Statement on Form S-4, of which this proxy statement/prospectus is a part, is declared effective; |
|
(j) |
by Palvella if, at any time prior to the receipt of the approval of the Palvella stockholders of the adoption of the Merger Agreement, each of the following occur: (A) Palvella having received a Superior Proposal; (B) Palvella having complied in all material respects with its obligations under Section 6.1 of the Merger Agreement with respect to such Superior Proposal; (C) the Palvella board of directors approves, and Palvella substantially concurrently with the termination of the Merger Agreement enters into, a definitive agreement with respect to such Superior Proposal; and (D) prior to or substantially concurrently with such termination, Palvella pays to Pieris $2 million; or |
|
(k) |
by Pieris if, at any time prior to the receipt of the approval of the Pieris stockholders of the share issuance and the other Pieris stockholder proposals contemplated by the Merger Agreement, each of the following occur: (A) Pieris shall have received a Superior Proposal; (B) Pieris shall have complied in all material respects with its obligations under Section 6.1 of the Merger Agreement with respect to such Superior Proposal; (C) the Pieris board of directors approves, and Pieris substantially concurrently with the termination of the Merger Agreement enters into, a definitive agreement with respect to such Superior Proposal; and (D) prior to or substantially concurrently with such termination, Pieris pays to Palvella $1 million. |
The party desiring to terminate the Merger Agreement will give the other party written notice of such termination.
Termination Fees Payable by Pieris
Pieris must pay Palvella a termination fee of $1 million if the Merger Agreement is terminated by (i) Pieris or Palvella pursuant to clause (b), (d) or (h) above so long as (A) prior to the termination of the Merger Agreement, any person makes an Acquisition Proposal or amends an Acquisition Proposal made prior to the date of the Merger Agreement with respect to Pieris; and (B) within 12 months after such termination Pieris enters into a definitive agreement to consummate, or consummates, any Acquisition Proposal (replacing all references in such definition to 15% with 50%), regardless of whether made before or after the termination of the Merger Agreement, (ii) Palvella pursuant to clause (f) above, or (iii) Pieris pursuant to clause (k) above.
Termination Fees Payable by Palvella
Palvella must pay Pieris a termination fee of $2 million if the Merger Agreement is terminated by (i) Pieris or Palvella pursuant to clause (b), (g) or (i) above so long as (A) prior to the termination of the Merger Agreement, any person makes an Acquisition Proposal or amends an Acquisition Proposal made prior to the date of the Merger Agreement with respect to Palvella; and (B) within 12 months after such termination Palvella enters into a definitive agreement to consummate, or consummates, any Acquisition Proposal (replacing all references in such definition to 15% with 50%), regardless of whether made before or after the termination of the Merger Agreement, (ii) Pieris pursuant to clause (e) above, or (iii) Palvella pursuant to clause (j) above.
The Merger Agreement may be amended with the approval of the respective boards of directors of Palvella and Pieris at any time, except that after the Merger Agreement has been adopted and approved by the Palvella stockholders or Pieris stockholders, no amendment to the Merger Agreement may be made without the further approval by the Palvella stockholders or Pieris stockholders, as the case may be, if such further approval is required by law.
The Merger Agreement provides that all fees and expenses incurred in connection with the Merger Agreement and the transactions contemplated thereby shall be paid by the party incurring such expenses, except as described above in the section titled “The Merger Agreement—Termination and Termination Fees” beginning on page 176 of this proxy statement/prospectus, and except that it shall not exceed $500,000 and Palvella and Pieris will share equally (i) all fees and expenses of the exchange agent, (ii) all fees and expenses, other than attorneys’ and accountants’ fees, incurred with respect to the printing, filing and mailing of this proxy statement/prospectus and the Registration Statement on Form S-4 of which this proxy statement/prospectus is a part, and any amendments or supplements thereto, and (iii) all fees and expense incurred in connection with the engagement of any proxy soliciting firms.
AGREEMENTS RELATED TO THE MERGER
In order to induce Pieris to enter into the Merger Agreement, certain Palvella stockholders are parties to support agreements with Pieris pursuant to which, among other things, each such stockholder has agreed, solely in his, her or its capacity as a Palvella stockholder, to vote all of his, her or its shares of Palvella capital stock in favor of the adoption of the Merger Agreement. These Palvella stockholders also agreed to vote against any competing Acquisition Proposal with respect to Palvella, subject to certain exceptions.
These Palvella stockholders have also granted Pieris an irrevocable proxy to vote their respective shares of Palvella capital stock in accordance with the support agreements. The Palvella stockholders may vote their shares of Palvella capital stock on all other matters not referred to in such proxy.
As of October 28, 2024, the Palvella stockholders that are party to a support agreement with Pieris owned an aggregate of 13,160,619 shares of Palvella capital stock, representing approximately 64.3% of the outstanding shares of Palvella voting capital stock on an as converted to common stock basis. These stockholders include (i) all executive officers and directors of Palvella and their affiliates which own shares of Palvella capital stock and (ii) certain stockholders owning 5% or more of the outstanding shares of Palvella capital stock. Following the effectiveness of the Registration Statement on Form S-4 of which this proxy statement/prospectus is a part and pursuant to the Merger Agreement, Palvella stockholders holding a sufficient number of shares of Palvella capital stock to adopt the Merger Agreement and approve the Merger and related transactions will be asked to execute written consents providing for such adoption and approval. The Palvella stockholders that entered into the support agreements are Wesley H. Kaupinen, George M. Jenkins, Todd C. Davis, RoyaltyRx Capital, LLC, Tadd S. Wessel, Aditya Asokan, Ph.D., Scott Morenstein, Kathleen A. McGowan, Jeffrey Martini, Ph.D., Kathleen Goin, Samsara BioCapital L.P., CDK Associates, L.L.C., Third Street Holdings LLC, Biotechnology Value Fund, L.P, Biotechnology Value Trading Fund OS, L.P., Biotechnology Value Fund II, L.P., Petrichor Opportunities Fund I LP, Petrichor Opportunities Fund I Intermediate LP, Adams Street Growth Equity Fund VII LP, Adams Street 2018 Direct Venture/Growth Fund LP, Adams Street 2019 Direct Growth Equity Fund LP, and Adams Street 2020 Direct Growth Equity Fund LP. Petrichor Opportunities Fund I LP and Petrichor Opportunities Fund I Intermediate LP are affiliates of Tadd S. Wessel, a member of the Palvella board of directors.
Under these support agreements, subject to certain exceptions, such stockholders have also agreed not to sell or transfer their shares of Palvella capital stock held by them, or any voting rights with respect thereto, until the earlier of the termination of the Merger Agreement, the Effective Time, or the date a Pieris board of directors recommendation change or a Palvella board of directors recommendation change is made, subject to certain exceptions. To the extent that any such sale or transfer is permitted pursuant to the exceptions included in the support agreement, each person to which any shares of Palvella capital stock are so sold or transferred must sign a substantially similar support agreement with respect to the transferred securities.
In addition, in order to induce Palvella to enter into the Merger Agreement, certain Pieris stockholders have entered into support agreements with Palvella pursuant to which, among other things, each such stockholder has agreed, solely in his, her or its capacity as a Pieris stockholder, to vote all of his, her or its shares of Pieris common stock in favor of Proposals 1, 2 and 4. These Pieris stockholders also agreed to vote against any competing Acquisition Proposal with respect to Pieris.
These Pieris stockholders have also granted Palvella an irrevocable proxy to vote their respective shares of Pieris common stock in accordance with the support agreements. Pieris stockholders may vote their shares of Pieris common stock on all other matters not referred to in such proxy.
As of October 28, 2024, the Pieris stockholders that are party to a support agreement owned an aggregate of 258,496 shares of Pieris common stock representing approximately 19.6% of the outstanding shares of Pieris common stock. These stockholders include certain executive officers and directors of Pieris and certain other Pieris stockholders holding 5% or more of the outstanding shares of Pieris common stock, such as certain of those beneficial owners mentioned in the table in the section titled “Principal Stockholders of Pieris” beginning on page 328. The Pieris stockholders that entered into the support agreements are: Biotechnology Value Fund, L.P., Biotechnology Value Fund II, L.P., Biotechnology Value Trading Fund OS LP. Investment 10 LLC, MSI BVF SPV, LLC, Stephen S. Yoder, Thomas Bures, Shane Olwill, James Geraghty, Matthew L. Sherman, M.D., Ann Barbier, M.D., Ph.D., Peter Kiener, D. Phil, Christopher Kiritsy, Michael Richman, and Maya R. Said, Sc.D.
Under these support agreements, subject to certain exceptions, such stockholders have also agreed not to sell or transfer their shares of Pieris common stock and Pieris preferred stock held by them until the earlier of the termination of the Merger Agreement, the Effective Time, and the date a Pieris board of directors recommendation change or a Palvella board of directors recommendation change is made, subject to certain exceptions. To the extent that any such sale or transfer is permitted pursuant to the exceptions included in the support agreements, each person to which any shares of Pieris common stock or securities convertible into shares of Pieris common stock are so sold or transferred must sign a substantially similar support agreement with respect to the transferred securities.
The foregoing description of the support agreements does not purport to be complete and is qualified in its entirety by the full text of the forms of support agreements, which are attached hereto as Annex E and Annex F.
Certain of Palvella’s and Pieris’ executive officers, directors and stockholders have entered into lock-up agreements, pursuant to which such parties have agreed not to, except in limited circumstances, offer, pledge, sell, contract to sell, sell any option to purchase, purchase any option or contract to sell, grant any option, right or warrant to purchase, or otherwise transfer or dispose of, any shares of Pieris common stock, until 180 days after the Effective Time.
The Palvella stockholders who have executed lock-up agreements as of October 28, 2024, owned in the aggregate, approximately 63.3% of the shares of Palvella’s outstanding capital stock. The Pieris stockholders who have executed lock-up agreements as of October 28, 2024, owned in the aggregate, approximately 19.6% of the shares of Pieris’ outstanding common stock.
The foregoing description of the lock-up agreements does not purport to be complete and is qualified in its entirety by the full text of the form of lock-up agreement, which is attached hereto as Annex G.
On July 23, 2024, the PIPE Investors entered into the Purchase Agreement with Pieris, pursuant to which such PIPE Investors have agreed to subscribe for and purchase (either for cash or in exchange for the termination and cancellation of outstanding convertible notes issued by Palvella), and Pieris has agreed to issue and sell to the PIPE Investors, an aggregate of approximately 3,154,241 of shares of Pieris common stock at a price per share equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, and/or in lieu of Pieris common stock to certain purchasers who so choose, Pre-Funded Warrants to purchase up to 2,592,585 shares of the combined company common stock at a purchase price per Pre-Funded Warrant equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, minus $0.001. The gross proceeds from the PIPE Financing are expected to be approximately $78.9 million, consisting of approximately $60.0 million in cash and the conversion of approximately $18.9 million of principal and interest payable under the outstanding convertible notes issued by Palvella, before paying estimated expenses. The closing of the PIPE Financing is conditioned upon the closing of the Merger as well as certain other conditions. The Merger is conditioned upon the PIPE Financing concurrently with or immediately following the closing of the Merger as well as certain other conditions. The Purchase Agreement contains customary representations and warranties of Pieris. The Purchase Agreement also contains customary representations and warranties of the investors party thereto.
Each investor’s obligation to purchase shares of Pieris common stock from Pieris pursuant to the Purchase Agreement are subject to the satisfaction or waiver of certain conditions, including, but not limited to the ones listed below. These conditions can be waived: (i) by a PIPE Investor solely as to itself or (ii) by PIPE Investors who collectively have subscribed to purchase at least a majority in interest of the aggregate shares to be issued in the PIPE Financing, or the “Purchaser Majority”:
|
(a) |
the representations and warranties of Pieris in the Purchase Agreement must be true and correct in all material respects on the date of the Purchase Agreement and on the closing date of the PIPE Financing (other than representations and warranties specifically made as of a particular date, in which case such representations and warranties shall be true and correct as of such date), except where individually or in the aggregate, where the failure to be so true and correct has not had or would not reasonably be expected to have a material adverse effect (without regard to any materiality or material adverse effect qualifications contained therein); |
|
(b) |
Pieris shall have performed, satisfied and complied in all material respects with all covenants, agreements, obligations required by the Purchase Agreement to be performed, satisfied or complied with by it on or prior to the closing of the PIPE Financing; |
|
(c) |
no statute, rule, regulation, order, executive order, decree, judgment, writ, order, ruling or injunction shall have been enacted, entered, promulgated, issued or endorsed by any court of competent jurisdiction or any governmental authority that enjoins, prevents or prohibits the consummation of any of the transactions contemplated by the Purchase Agreement; |
|
(d) |
the satisfaction or waiver of each of the conditions precedent to the consummation of the Merger set forth in the Merger Agreement (other than those conditions which, by their nature, are to be satisfied at the closing of the Merger pursuant to the Merger Agreement) and the approval of the share issuance and the amendment to Pieris’ amended and restated articles of incorporation to increase the number of authorized shares of Pieris common stock, and the ratification of the Pieris board of directors’ approval of an amendment to Pieris’ amended and restated articles of incorporation to change the name of Pieris to “Palvella Therapeutics, Inc.” shall have occurred; |
|
(e) |
Pieris shall have obtained in a timely fashion any and all consents, permits, approvals, registrations and waivers necessary for consummation of the purchase and sale of the Pieris common stock, all of which shall be and remain so long as necessary in full force and effect; |
|
(f) |
since the date of the Purchase Agreement, no event or series of events shall have occurred that has had or would reasonably be expected to have a material adverse effect; |
|
(g) |
the Pieris common stock shall not have been suspended by the SEC or the Nasdaq from trading on the Nasdaq nor shall suspension by the SEC or the Nasdaq have been threatened, either (i) in writing by the SEC or the Nasdaq or (ii) by falling below the minimum listing maintenance requirements of the Nasdaq. No stop order shall have been imposed by Nasdaq, the SEC or any other governmental authority or regulatory body with respect to public trading in the Pieris common stock. Pieris shall have filed the Notification Form: Listing of Additional Shares for the listing of the Pieris common stock and shares underlying the Pre-Funded Warrants with Nasdaq, and Nasdaq shall have raised no objection to such notice and the transactions contemplated hereby; |
|
(h) |
Pieris shall have delivered the deliverables required under the Purchase Agreement; |
|
(i) |
Pieris shall have delivered to the placement agent, on behalf of each PIPE Investor, a certificate, dated as of the closing date of the PIPE Financing and signed by its chief executive officer and its chief financial officer, certifying to the fulfillment of certain conditions specified in the Purchase Agreement; and |
|
(j) |
the Purchase Agreement shall not have been terminated as to such PIPE Investor. |
Pieris’ obligation to sell shares of Pieris common stock to each PIPE Investor pursuant to the Purchase Agreement are subject to the satisfaction or waiver of certain conditions by Pieris, including:
|
(a) |
all representations and warranties of the PIPE Investors contained in the Purchase Agreement shall be true and correct in all material respects at and as of the date of the Purchase Agreement and on the date of the closing of the PIPE Financing (other than representations and warranties specifically made as of a particular date, in which case such representations and warranties shall be true and correct as of such date), except for those representations and warranties which are qualified as to materiality, in which case such representations and warranties shall be true and correct in all respects; |
|
(b) |
each PIPE Investor shall have performed, satisfied and complied in all material respects with all covenants, agreements, obligations and conditions required by the Purchase Agreement to be performed, satisfied or complied with by it at or prior to the closing of the PIPE Financing; |
|
(c) |
no statute, rule, regulation, order, executive order, decree, judgment, writ, order, ruling or injunction shall have been enacted, entered, promulgated, issued or endorsed by any court of competent jurisdiction or any governmental authority that enjoins, prevents or prohibits the consummation of any of the transactions contemplated by the Purchase Agreement; |
|
(d) |
each PIPE Purchaser shall have delivered the deliverables required under the Purchase Agreement; |
|
(e) |
the satisfaction or waiver of each of the conditions precedent to the consummation of the Merger set forth in the Merger Agreement (other than those conditions which, by their nature, are to be satisfied at the closing of the Merger pursuant to the Merger Agreement) and the approval of the share issuance and the amendment to Pieris’ amended and restated articles of incorporation to increase the number of authorized shares of Pieris common stock and change the name of Pieris to “Palvella Therapeutics, Inc.” shall have occurred; and |
|
(f) |
the Purchase Agreement shall not have been terminated. |
Amendment and Waiver of the Securities Purchase Agreement
The Purchase Agreement may be amended, modified or waived only with the written consent of Pieris and the Purchaser Majority, provided that certain closing conditions shall require the consent of all investors as described above. The Purchase Agreement terminates upon the earlier to occur of (i) the termination of the Merger Agreement without the Merger being consummated, (ii) by a PIPE Investor (with respect to itself) if any of the closing conditions under the Purchase Agreement shall have become incapable of fulfillment, and shall not have been waived by such PIPE Investor, or (iii) 5:00 P.M., New York time, on April 30, 2025.
Overview
At or prior to the Effective Time, Pieris will enter into the CVR Agreement with a rights agent and a CVR holder representative, pursuant to which each holder of record of shares of Pieris common stock and preferred stock entitled to receive a dividend in accordance with the terms of such preferred stock will receive the right to one CVR for each outstanding share of Pieris common stock held by such stockholder, or share of common stock underlying such preferred stock, held by such stockholder, on such date. Furthermore, to the extent a holder of a Pieris warrant outstanding as of the date of the CVR Agreement exercises such Pieris warrant after the date of the CVR Agreement, a number of CVRs equal to the number of shares of Pieris common stock issued to such holder in connection with such exercise shall be issued to such holder, subject to and in accordance with the terms and conditions of such Pieris Warrant, as applicable.
Each CVR will represent the contractual right to receive cash payments (net of certain expenses and taxes) upon the receipt of payments by Pieris or any of its affiliates under certain strategic partner agreements, including existing collaboration agreements pursuant to which Pieris may be entitled to milestones and royalties in the future and other out-licensing agreements for certain of Pieris’ legacy assets, and upon the receipt of certain research and development tax credits in favor of Pieris or any of its affiliates, in each case as set forth in, and subject to and in accordance with the terms and conditions of, the CVR Agreement. Pursuant to the terms of the CVR Agreement, holders of CVRs would be entitled to cash payments (net of certain expenses and taxes) upon the receipt by Pieris or any of its affiliates of certain payments under any license, collaboration or similar agreement involving any legacy assets of Pieris entered into prior to the effective date of the CVR Agreement. Pieris retains the ability under the Merger Agreement to seek to enter into such agreements related to its legacy assets and intellectual property, including PRS-400 and PRS-220, though currently does not have any such agreements in place and does not have any plans to divest any material assets. Pieris continues to explore potential transactions for PRS-400 and PRS-220. Management has determined that there is no value associated with the CVRs as the likelihood of any payments received in connection with Pieris’ legacy assets is remote. Unless earlier terminated by the representative of the CVR holders and the combined company, the CVR Agreement will terminate on the date on which no strategic partner agreement is in effect and no payments are payable to Pieris or any of its affiliates under any Strategic Partner Agreement.
The contingent payments under the CVR Agreement, if they become payable, will become payable to the rights agent for subsequent distribution to the holders of the CVRs. In the event that no such proceeds are received, holders of the CVRs will not receive any payment pursuant to the CVR Agreement. There can be no assurance that holders of CVRs will receive any amounts with respect thereto. The right to the contingent payments contemplated by the CVR Agreement is a contractual right only and will not be transferable, except in the limited circumstances specified in the CVR Agreement. The CVRs will not be evidenced by a certificate or any other instrument and will not be registered with the SEC. The CVRs will not have any voting or dividend rights and will not represent any equity or ownership interest in Pieris or any of its affiliates. No interest will accrue on any amounts payable in respect of the CVRs.
The foregoing description of the CVR Agreement does not purport to be complete and is qualified in its entirety by the full text of the form of CVR Agreement, which is included in Annex D to this proxy statement/prospectus.
Material U.S. Federal Income Tax Consequences of the Receipt of CVRs
The following discussion is a summary of the material U.S. federal income tax consequences of the receipt of CVRs to Pieris stockholders who receive CVRs with respect to Pieris common stock, but this discussion does not purport to be a complete analysis of all potential tax consequences that may be relevant to a Pieris stockholder. The effects of other U.S. federal tax laws, such as estate and gift tax laws, and any applicable state, local or non-U.S. tax laws are not discussed. This discussion is based on the Code, Treasury Regulations promulgated thereunder, judicial decisions, and published rulings and administrative pronouncements of the IRS, in each case in effect as of the date hereof. These authorities may change or be subject to differing interpretations. Any such change or differing interpretation may be applied retroactively in a manner that could adversely affect a Pieris stockholder. Pieris has not sought and does not intend to seek any opinions of counsel or rulings from the IRS regarding the matters discussed below. There can be no assurance the IRS or a court will not take a position contrary to that discussed below regarding the tax consequences of the receipt of CVRs.
This discussion is limited to Pieris stockholders that hold Pieris common stock as a “capital asset” within the meaning of Section 1221 of the Code (generally, property held for investment). This discussion does not address all U.S. federal income tax consequences that may be relevant to a Pieris stockholder’s particular circumstances, including the impact of the alternative minimum tax or the Medicare contribution tax on net investment income or the rules related to “qualified small business stock” within the meaning of Section 1202 of the Code, or as “Section 1244 Stock” for purposes of Section 1244 of the Code. In addition, it does not address consequences relevant to Pieris stockholders subject to special rules, including, without limitation:
|
● |
U.S. expatriates and former citizens or long-term residents of the U.S.; |
|
● |
Pieris U.S. holders whose functional currency is not the U.S. dollar; |
|
● |
persons holding Pieris common stock as part of a hedge, straddle or other risk reduction strategy or as part of a conversion transaction or other integrated investment; |
|
● |
banks, insurance companies, and other financial institutions; |
|
● |
real estate investment trusts or regulated investment companies; |
|
● |
brokers, dealers or traders in securities; |
|
● |
“Controlled foreign corporations,” “passive foreign investment companies,” and corporations that accumulate earnings to avoid U.S. federal income tax; |
|
● |
S corporations, partnerships or other entities or arrangements classified as partnerships for U.S. federal income tax purposes (and investors therein); |
|
● |
tax-exempt organizations or governmental organizations; |
|
● |
persons subject to special tax accounting rules as a result of any item of gross income with respect to Pieris common stock being taken into account in an “applicable financial statement” (as defined in the Code); |
|
● |
persons who acquired their shares of Pieris common stock in a transaction subject to the gain rollover provisions of Section 1045 of the Code; |
|
● |
persons who hold “Section 305 stock”; |
|
● |
persons deemed to sell Pieris common stock under the constructive sale provisions of the Code; |
|
● |
persons who hold or received Pieris common stock pursuant to the exercise of any employee stock option or otherwise as compensation; and |
|
● |
tax-qualified retirement plans. |
If an entity (or arrangement) classified as a partnership for U.S. federal income tax purposes holds Pieris common stock, the tax treatment of a partner in the partnership will depend on the status of the partner, the activities of the partnership and certain determinations made at the partner level. Accordingly, partnerships holding Pieris common stock and the partners in such partnerships should consult their tax advisors regarding the U.S. federal income tax consequences to them.
IT IS RECOMMENDED THAT PIERIS STOCKHOLDERS CONSULT THEIR TAX ADVISORS WITH RESPECT TO THE APPLICATION OF THE U.S. FEDERAL INCOME TAX LAWS TO THEIR PARTICULAR SITUATIONS AS WELL AS ANY TAX CONSEQUENCES OF THE RECEIPT OF CVRs ARISING UNDER THE U.S. FEDERAL ESTATE OR GIFT TAX LAWS OR UNDER THE LAWS OF ANY STATE, LOCAL OR NON-U.S. TAXING JURISDICTION OR UNDER ANY APPLICABLE INCOME TAX TREATY.
For purposes of this discussion, a “Pieris U.S. holder” is a beneficial owner of Pieris common stock that, for U.S. federal income tax purposes, is or is treated as:
|
● |
an individual who is a citizen or resident of the U.S.; |
|
● |
a corporation created or organized in or under the laws of the U.S., any state thereof, or the District of Columbia; |
|
● |
an estate, the income of which is subject to U.S. federal income tax regardless of its source; or |
|
● |
a trust that (i) is subject to the primary supervision of a U.S. court and the control of one or more “United States persons” (within the meaning of Section 7701(a)(30) of the Code) over all of its substantial decisions or (ii) has a valid election in effect to be treated as a United States person for U.S. federal income tax purposes. |
For purposes of this discussion, a “Pieris non-U.S. holder” means a beneficial owner of Pieris common stock that is neither a Pieris U.S. holder nor a partnership (or other entity (or arrangement) classified as a partnership) for U.S. federal income tax purposes.
Receipt of CVRs by Pieris U.S. Holders. There is substantial uncertainty as to the tax treatment of the CVRs. Specifically, there is no authority directly addressing whether CVRs with characteristics similar to the CVRs should be treated as a distribution of property with respect to Pieris common stock, a distribution of equity, a “debt instrument” or an “open transaction” for U.S. federal income tax purposes, and such determinations are inherently factual in nature. As a result, it is not possible to express a definitive conclusion as to the U.S. federal income tax treatment of receipt of the CVRs or receipt of any payment pursuant to the CVRs. Based on the specific characteristics of the CVRs, and unless otherwise required by a change in law after the date of the CVR Agreement, Pieris intends to take the position that the fair market value of the CVRs cannot be reasonably ascertained on the date of the issuance of the CVRs, and accordingly, the issuance of the CVRs constitutes an “open transaction.” Absent a change in law requiring otherwise, Pieris will not report the issuance of the CVRs as a current distribution with respect to its stock and will instead report each future cash payment (if any) on the CVRs as a distribution by Pieris in respect of its stock for U.S. federal income tax purposes, with each such payment being reported as a dividend to the extent of Pieris’ current or accumulated earning and profits in the year in which such payment is made.
If Pieris’ intended reporting position is correct, a Pieris U.S. holder would not generally recognize income in respect of the CVRs on the date of issuance and would take no tax basis in the CVRs. Any future cash payments would constitute a dividend to the extent of Pieris’ current or accumulated earnings and profits (as determined for U.S. federal income tax purposes) in the taxable year of such payment, then as a non-taxable return of capital to the extent of such holder’s basis in its Pieris common stock, and finally as capital gain from the sale or exchange of Pieris common stock. Dividends received by individual Pieris U.S. holders are eligible for reduced rates of taxation applicable to long-term capital gains, provided certain holding period requirements are met.
Treatment of CVRs under the “Closed Transaction.” The IRS could instead assert that the issuance of the CVRs should be treated as a “closed transaction.” Under “closed transaction” treatment, a Pieris U.S. holder would be treated as receiving a distribution equal to the fair market value of the CVRs issued to such Pieris U.S. holder on the date of issuance. The amount of this distribution generally would be treated first as a taxable dividend to the extent of the Pieris U.S. holder’s pro rata share of Pieris’ current or accumulated earnings and profits (as determined for U.S. federal income tax purposes), then as a non-taxable return of capital to the extent of the Pieris U.S. holder’s basis in its Pieris common stock, and finally as capital gain from the sale or exchange of Pieris common stock. A Pieris U.S. holder’s tax basis in the CVRs received would equal the fair market value of the CVRs on the date of issuance and the holding period of the CVRs received would begin on the day following the date of issuance. Although not free from doubt, a future cash payment under a CVR would likely be treated as a non-taxable return of a Pieris U.S. holder’s adjusted tax basis in the CVR to the extent thereof, although the timing of the recovery of a Pieris U.S. holder’s tax basis is unclear. A payment in excess of such amount may be treated as a payment with respect to a sale of a capital asset, ordinary income or dividends. Additionally, it is possible that a portion of future cash payments would constitute imputed interest and taxed as such. A Pieris U.S. holder might recognize loss, which might be a capital loss and could be a long-term capital loss, upon the expiration of the CVR to the extent cash payments ultimately received pursuant to such CVR were less than the Pieris U.S. holder’s adjusted tax basis in the CVRs, but whether and when such a loss would be recognized is unclear. The deductibility of capital losses is subject to limitations. As discussed above, Pieris will not report the issuance of the CVRs as a “closed transaction” for U.S. federal income tax purposes.
Treatment as Debt Instrument or Equity. It is also possible, although Pieris believes unlikely, that the issuance of the CVRs could be treated as one or more “debt instruments” or as a distribution of equity. Pieris U.S. holders are urged to consult their tax advisors with respect to the proper characterization of the CVRs and the tax consequences thereof (including any future cash payments under the CVRs).
Receipt of CVRs by Pieris Non-U.S. Holders. As discussed above under “Receipt of CVRs by Pieris U.S. Holders,” Pieris intends to take the position that any future cash payments on the CVRs are distributions with respect to Pieris common stock and that such distributions constitute dividends to the extent payable out of Pieris’ current or accumulated earnings and profits (as determined under U.S. federal income tax principles) in the taxable year of such future cash payment. Assuming such position is correct, amounts not treated as dividends for U.S. federal income tax purposes may constitute a return of capital and first be applied against and reduce a Pieris non-U.S. holder’s adjusted tax basis in its common stock, but not below zero, and any excess may be treated as capital gain with respect to such Pieris non-U.S. holder’s Pieris common stock. However, the tax treatment of CVRs is subject to uncertainty, and, accordingly, Pieris non-U.S. holder are urged to consult their tax advisors with respect to the proper characterization of the CVRs and the tax consequences thereof (including any future cash payments made under the CVRs).
In light of Pieris’ intended reporting position, it is expected that Pieris non-U.S. holders would generally be subject to U.S. federal withholding tax at a rate of 30% on any future cash payments that are treated as dividends on the CVRs. If a Pieris non-U.S. holder is eligible for a lower treaty rate, withholding will be at such lower treaty rate only if such Pieris non-U.S. holder provides a valid IRS Form W-8BEN or Form W-8BEN-E (or applicable successor form) certifying such Pieris non-U.S. holder’s qualification for the reduced rate. If a Pieris non-U.S. holder holds the stock through a financial institution or other intermediary, the Pieris non-U.S. holder will be required to provide appropriate documentation to the intermediary, which then will be required to provide certification to the applicable withholding agent, either directly or through other intermediaries. Pieris non-U.S. holders who do not timely provide the applicable withholding agent with the required certification, but who qualify for a reduced treaty rate, may obtain a refund of any excess amounts withheld by timely filing an appropriate claim for refund with the IRS.
Subject to the discussions below regarding FATCA (as defined below) and backup withholding, if the issuance of the CVRs is effectively connected with a Pieris non-U.S. holder’s conduct of a trade or business within the United States (and, if required by an applicable income tax treaty, the Pieris non-U.S. holder maintains a permanent establishment in the United States to which the distribution of the CVRs is attributable), the Pieris non-U.S. holder will be exempt from U.S. federal withholding tax and future payments made pursuant to the CVRs generally will be subject to U.S. federal income tax on a net income basis in the same manner as if such Pieris non-U.S. holder were a U.S. holder. To claim the exemption, the Pieris non-U.S. holder must furnish to the applicable withholding agent a valid IRS Form W-8ECI (or applicable successor form), certifying that the payment is effectively connected with the Pieris non-U.S. holder’s conduct of a trade or business within the United States. A Pieris non-U.S. holder that is a corporation also may be subject to a branch profits tax at a rate of 30% (or such lower rate specified by an applicable income tax treaty) on all or a portion of its effectively connected earnings and profits for the taxable year.
Future payments, if any, to a Pieris non-U.S. holder with respect to a CVR may also be subject to withholding at a 30% rate unless the holder establishes a reduced treaty rate or that such income is exempt from withholding because it is effectively connected with the holder’s conduct of a trade or business within the United States or otherwise.
Under the provisions of the Code commonly referred to as the Foreign Account Tax Compliance Act, or FATCA, future payments made pursuant to the CVRs, if any, to a Pieris non-U.S. holder with respect to the CVRs may be subject to withholding at a rate of 30% if the Pieris non-U.S. holder fails to satisfy prescribed certification requirements. In general, no such withholding will be required with respect to a Pieris non-U.S. holder that timely provides certifications that establish an exemption from FATCA withholding on a valid IRS Form W-8. If withholding under FATCA is required, Pieris non-U.S. holders not otherwise subject to withholding (or that otherwise would be entitled to a reduced rate of withholding) may be required to seek a refund or credit from the IRS.
Any withholding required by Pieris or other applicable withholding agents may be satisfied by Pieris or such agent by withholding a portion of the issued CVRs, from future payments, if any, on the CVRs, or from other property of the Pieris non-U.S. holder held in an account with the applicable withholding agent.
To the extent that future payments made pursuant to the CVRs is treated as capital gain from the sale or exchange of Pieris common stock, such gain generally will not be subject to U.S. federal income tax unless (i) such gain is effectively connected with the conduct by a Pieris non-U.S. holder of a trade or business in the United States (and, if an income tax treaty applies, the gain is generally attributable to a U.S. permanent establishment maintained by such Pieris non-U.S. holder), (ii) in the case of gain realized by a Pieris non-U.S. holder that is an individual, such Pieris non-U.S. holder is present in the United States for 183 days or more in the taxable year of the sale and certain other conditions are met or (iii) Pieris is or has been a United States real property holding corporation, or USRPHC, for U.S. federal income tax purposes and, if the shares are “regularly traded on an established securities market,” such Pieris non-U.S. holder owned, directly or indirectly, at any time during the five-year period ending on the date of the distribution, more than 5% of the shares of Pieris common stock and such Pieris non-U.S. holder is not eligible for any treaty exemption. Pieris believes it is not, and has not been, a USRPHC for U.S. federal income tax purposes. In addition, although not free from doubt, Pieris believes that Pieris common stock currently should be considered to be regularly traded.
A Pieris non-U.S. holder should consult its tax advisor regarding its entitlement to benefits and the various rules under applicable tax treaties.
If the IRS were to successfully assert that the issuance of the CVRs should be treated as a “closed transaction,” each Pieris non-U.S. holder would be treated as receiving a distribution in an amount equal to the fair market value of the CVRs issued to such Pieris non-U.S. holder on the date of the issuance. This distribution would be treated first as a taxable dividend to the extent of the Pieris non-U.S. holder’s pro rata share of Pieris’ current or accumulated earnings and profits for the year of issuance (as determined for U.S. federal income tax purposes), then as a non-taxable return of capital to the extent of the Pieris non-U.S. holder’s basis in its Pieris common stock, and finally as capital gain from the sale or exchange of Pieris common stock with respect to any remaining amount. Principles described in this section “Receipt of CVRs by Pieris Non-U.S. Holders,” relating to withholding would apply. In addition, any future cash payments under the CVRs may also be subject to withholding to the extent that such payments exceed the fair market value of the CVRs on the date of issuance.
Due to the legal and factual uncertainty regarding the tax treatment of the CVRs (and any future cash payments under the CVRs), Pieris non-U.S. holders are urged to consult their tax advisors concerning the recognition of gain, income and/or loss or withholding that may apply in connection with the CVRs. Pieris non-U.S. holders should consult their tax advisors regarding the applicability of information reporting and backup withholding and/or withholding under the Foreign Account Tax Compliance Act with respect to the CVRs and any future cash payments under the CVRs, particularly in light of the uncertainty under U.S. federal income tax law relating to the tax treatment of the CVRs.
Information Reporting and Backup Withholding. In general, any payments made pursuant to the CVRs (if any) to Pieris U.S. holders will be reported to the IRS unless the holder is an exempt recipient. Backup withholding, currently at a rate of 24%, may apply to payments made pursuant to the CVRs (if any) unless the Pieris U.S. holder (1) is an exempt recipient or (2) provides a certificate (generally on an IRS Form W-9) containing the Pieris U.S. holder’s name, address, correct federal taxpayer identification number and statement that the Pieris U.S. holder is a U.S. person and is not subject to backup withholding.
A Pieris non-U.S. holder will not be subject to backup withholding with respect to the issuance of the CVRs, provided the Pieris non-U.S. holder certifies its non-U.S. status, such as by providing a valid IRS Form W-8BEN, Form W-8BEN-E, or Form W-8ECI, or otherwise establishes an exemption. However, information returns will be filed with the IRS in connection with payments made pursuant to the CVRs (if any), regardless of whether any tax was actually withheld. Copies of these information returns may also be made available under the provisions of a specific treaty or agreement to the tax authorities of the country in which the Pieris non-U.S. holder resides or is established.
Backup withholding is not an additional tax. Any amounts withheld under the backup withholding rules may be allowed as a refund or credit against a holder’s U.S. federal income tax liability, provided the required information is timely furnished to the IRS.
PLEASE CONSULT YOUR TAX ADVISOR WITH RESPECT TO THE PROPER CHARACTERIZATION OF THE RECEIPT OF THE CVRs.
PIERIS EXECUTIVE AND DIRECTOR COMPENSATION
Compensation Overview
This section describes the material elements of compensation awarded to, earned by or paid to Pieris’ named executive officers and directors. Pieris qualifies as a “smaller reporting company” under the rules promulgated by the SEC, and it has elected to comply with the disclosure requirements applicable to smaller reporting companies. Accordingly, this executive compensation summary is not intended to meet the “Compensation Discussion and Analysis” disclosure required of larger reporting companies.
Compensation Philosophy
The objectives of Pieris’ rewards programs are:
|
● |
to attract and retain talented individuals with an entrepreneurial mindset; |
|
● |
to motivate these individuals towards high performance in pursuing Pieris’ corporate strategies and to achieve its business goals; |
|
● |
to recognize individual excellence while fostering a shared commitment of success; |
|
● |
to align the interests of patients, employees, and stockholders; |
|
● |
to be financially prudent, yet competitive; and |
|
● |
to further foster Pieris’ core values of passion, integrity, excellence, responsibility, innovation and the spirit of collaboration. |
When establishing target pay levels, Pieris seeks to target the median of the market and allow for discretion in hiring best-in-class talent, while providing future compensation opportunities that are commensurate with the market and reflect the individual’s role and experience. It is expected that some individuals will be positioned above or below market median based on particular facts and circumstances. Incentive pay outcomes (e.g., actual earned bonus) are not guaranteed, and actual pay will be reflective of success towards Pieris’ stated company and individual goals and objectives, as well as stockholder value creation.
Role of the Compensation and Management Development Committee and Compensation Consultant
Pieris’ Compensation and Management Development Committee is responsible for the determination of the compensation of Pieris’ Chief Executive Officer and conducts its decision-making process with respect to that topic without the Chief Executive Officer, or any other executive officers, present. Pieris’ Compensation and Management Development Committee has been delegated the authority to administer its 2014 Employee, Director and Consultant Equity Incentive Plan, or 2014 Plan, its 2016 Employee, Director and Consultant Equity Incentive Plan, or 2016 Plan, its 2018 Employee, Director and Consultant Equity Incentive Plan, or 2018 Plan, its 2018 Employee Stock Purchase Plan, or 2018 ESPP, its 2019 Employee, Director and Consultant Equity Incentive Plan, or 2019 Plan, its 2020 Employee, Director and Consultant Equity Incentive Plan, as amended, or 2020 Plan, and its 2023 Employee Stock Purchase Plan, or 2023 ESPP.
The Compensation and Management Development Committee has the authority to directly retain the services of independent consultants and other experts to assist in fulfilling its responsibilities. The Compensation and Management Development Committee has engaged the services of Pearl Meyer & Partners LLC, or Pearl Meyer, a national executive compensation consulting firm, to review and provide recommendations concerning all of the components of Pieris’ executive compensation program. Pearl Meyer performs services solely on behalf of the Compensation and Management Development Committee and has no relationship with Pieris or management except as it may relate to performing such services. Pearl Meyer assists the Compensation and Management Development Committee in defining Pieris’ peer companies for executive compensation and practices, and in benchmarking Pieris’ executive compensation program against the peer group. Pearl Meyer also assists the Compensation and Management Development Committee in benchmarking its director compensation program and practices against those of its peers. The Compensation and Management Development Committee has assessed the independence of Pearl Meyer pursuant to SEC rules and the corporate governance rules of Nasdaq and concluded that no conflict of interest exists that would prevent Pearl Meyer from independently representing the Compensation and Management Development Committee.
The Compensation and Management Development Committee reviews the performance of each Named Executive Officer in light of the above factors, as described in more detail in “Narrative Disclosure to Summary Compensation Table” below, and determines whether the Named Executive Officer should receive any increase in base salary, annual bonus award or discretionary equity award based on such evaluation.
Say-on-Pay Vote on Executive Compensation
At the 2023 annual meeting of stockholders of Pieris, 85.9% of the votes cast voted in favor of Pieris’ executive compensation.
Summary Compensation Table
The following table summarizes the compensation earned in each of Pieris’ fiscal years that ended December 31, 2023 and 2022 by its Named Executive Officers, which consisted of its principal executive officer and its two next most highly compensated executive officers who earned more than $100,000 during the fiscal year ended December 31, 2023 and were serving as executive officers as of such date. Pieris refers to the executive officers listed below as the Named Executive Officers.
|
Name and Principal Position |
Year |
Salary ($) |
Bonus ($) (1) |
Option Awards ($)(2) |
Non-equity Incentive Plan Compensation ($)(3) |
All Other Compensation ($) |
Total ($) |
||||||||||||
|
Stephen S. Yoder |
2023 |
584,595 | 146,149 | 614,818 | - | 13,390 | (4) | 1,358,952 | |||||||||||
|
Chief Executive Officer |
2022 |
564,826 | - | 1,288,128 | 197,689 | 12,200 | (4) | 2,062,843 | |||||||||||
|
Thomas Bures |
2023 |
400,155 | 80,031 | 241,536 | - | 7,315 | (4) | 729,037 | |||||||||||
|
Senior Vice President and Chief Financial Officer |
2022 |
381,100 | - | 444,183 | 106,078 | 12,200 | (4) | 943,561 | |||||||||||
|
Shane Olwill |
2023 |
328,614 | 127,702 | 241,536 | - | 7,593 | (5) | 705,445 | |||||||||||
|
Senior Vice President and Chief Development Officer |
|
(1) |
Represents discretionary bonus paid for 2023 performance, which is described in more detail in the "Bonus Payments" section below. |
|
(2) |
These amounts represent the aggregate grant date fair value for the option awards granted during the fiscal years presented, determined in accordance with the Financial Accounting Standards Board, or FASB, ASC Topic 718. A discussion of the assumptions used in determining grant date fair value may be found in Note 11 to Pieris’ Financial Statements included in Pieris’ Annual Report on Form 10-K for the fiscal year ended December 31, 2023. |
|
(3) |
These amounts represent payments made for 2022 performance under Pieris’ 2022 performance-based incentive bonus plan. |
|
(4) |
Represents matching contributions Pieris made under its 401(k) Plan. |
|
(5) |
Represents $568 for matching contributions Pieris made under applicable pension scheme, $5,404 in commuter allowance benefits and $1,621 in German inflation adjustment premiums paid. Dr. Olwill will depart Pieris effective October 31, 2024. |
Narrative to Summary Compensation Table
The Compensation and Management Development Committee has adopted the following processes and procedures for the consideration and determination of executive and director compensation: In establishing compensation amounts for executives, the Compensation and Management Development Committee seeks to provide compensation that is competitive in light of current market conditions and industry practices. Accordingly, the Compensation and Management Development Committee will generally review market data, which is comprised of proxy-disclosed data from peer companies and information from nationally recognized published surveys for the biopharmaceutical industry, adjusted for size. The market data helps the Compensation and Management Development Committee gain perspective on the compensation levels and practices at the peer companies and to assess the relative competitiveness of the compensation paid to Pieris’ executives. The market data thus guides the Compensation and Management Development Committee in its efforts to set executive compensation levels and program targets at competitive levels for comparable roles in the marketplace. The Compensation and Management Development Committee then considers other factors, such as the importance of each executive officer’s role to Pieris, individual expertise, experience, performance, retention concerns and relevant compensation trends in the marketplace, in making its final compensation determinations. The Compensation and Management Development Committee works with its consultant, Pearl Meyer, in these activities.
Elements of Compensation
In addition to each officer’s base salary, Pieris’ executive officer compensation program consists of a cash incentive bonus plan and discretionary stock option awards in addition to customary benefits. The amounts of compensation awarded for each element of Pieris’ compensation program (i.e., base salary, bonuses and stock options) are reviewed in connection with Pieris’ performance.
Base Salary
Annual base salaries compensate Pieris’ executive officers for fulfilling the requirements of their respective positions and provide them with a level of cash income predictability and stability with respect to a portion of their total compensation. Pieris believes that the level of an executive officer’s base salary should reflect the executive’s performance, experience and breadth of responsibilities, Pieris’ understanding of salaries for similar positions within its industry and any other factors relevant to that particular job.
Base salaries are typically negotiated at the outset of an executive’s employment in connection with an offer of employment and employment agreement. Salary levels are considered annually as part of Pieris’ performance review process, but also in cases including promotion or other changes in the job responsibilities of an executive officer. Merit and company-wide general increases are also taken into consideration when determining increases to base salary.
Bonus Payments
Performance Goals. In general, Pieris’ Compensation and Management Development Committee approves all bonus payments to Pieris’ executive officers based upon the attainment of performance targets established by the Compensation and Management Development Committee and related to scientific, operational, and financial metrics with respect to Pieris or any of its subsidiaries, or the Performance Goals, which may include, among others, business development and financing milestones; developmental, clinical or regulatory milestones; strategic transactions; clinical trial milestones and results, any of which may be measured in absolute terms or compared to any incremental increase or measured in terms of growth. Further, any Performance Goals may be used to measure the performance of Pieris as a whole or as a business unit or other segment of Pieris. Bonuses paid are typically based upon objectively determinable bonus formulas that tie such bonuses to one or more Performance Goals. The bonus formulas are adopted by the Compensation and Management Development Committee and communicated to each executive officer. No bonuses are paid unless and until the Compensation and Management Development Committee makes a determination with respect to the attainment of the performance objectives. Notwithstanding the foregoing, the Compensation and Management Development Committee may adjust bonuses payable based on achievement of individual performance goals or pay bonuses (including, without limitation, discretionary bonuses) to executives based upon such other terms and conditions as the Compensation and Management Development Committee may in its discretion determine. The bonuses payable may be paid in equity or cash.
2023 Performance Goals and Performance Goal Achievement. In early 2023, Pieris’ Compensation and Management Development Committee reviewed and approved Pieris’ 2023 Performance Goals, or the Original 2023 Performance Goals. The Original 2023 Performance Goals were divided into several categories, that the Compensation and Management Development Committee determined advanced Pieris’ overall corporate strategy including: (i) continued clinical advancement of elarekibep in collaboration with AstraZeneca; (ii) continued advancement of PRS-220 in the Phase 1 clinical trial and preparation for a Phase 2 clinical trial; (iii) continued advancement of PRS-400 for an investigational new drug application, or “NDA”, filing; (iv) advancement of the Phase 1 study of PRS-344 and announcement of topline results; (v) achievement of certain internal milestones for its partnered preclinical programs with Pfizer (formerly Seagen); and (vi) corporate development and financing, which involved closing a business development transaction for PRS-220 or PRS-400. In light of AstraZeneca's decision to discontinue the elarekibep clinical trials, as well as Pieris’ decision to explore strategic transactions in July 2023, the Compensation and Management Development Committee reviewed and approved restated 2023 Performance Goals, or the Amended 2023 Performance Goals, in August 2023, which were approved with respect to employees other than Pieris’ executive officers. The Amended 2023 Performance Goals were divided into several categories that the Compensation and Management Development Committee determined advanced Pieris’ overall corporate strategy: (a) internal project completions and decisions, including PRS-220 Phase 1 clinical trial completion, determination of the future strategy for elarekibep, and handover of Pieris’ partnered assets to its partners (i.e., PRS-344 to Servier and the two preclinical programs to Pfizer, formerly Seagen) (33.3%); (b) operational winddowns which involved winding down Pieris’ operational activities at Pieris’ Hallbergmoos site in Germany and reducing various liabilities and costs, including Pieris’ lease liability (33.3%); and (c) pursuing and assessing potential strategic transactions, which included exploring partnering or funding opportunities for PRS-220, PRS-400, cinrebafusp alfa, and Pieris’ platform generally, and assessing potential reverse merger opportunities (33.3%).
Subsequently, the Compensation and Management Development Committee evaluated Pieris’ performance against the Amended 2023 Performance Goals in early 2024. Performance under each category is evaluated on a 0-100% scale. Pieris’ evaluation system also permits consideration of areas of overachievement when Pieris exceeds its stated goals and when the Compensation and Management Development Committee sets examples of potential areas for overachievement. The Compensation and Management Development Committee considers any overachievement when assessing Pieris’ level of goal achievement. In early 2024, the Compensation and Management Development Committee considered each of the Amended 2023 Performance Goals in detail, including any potential overachievement, and determined that Pieris had achieved 100% of the Amended 2023 Performance Goals on an overall basis. The Compensation and Management Development Committee also assessed the Original 2023 Performance Goals and determined that 0% had been achieved due to the discontinuation of the elarekibep clinical trials and the winddown or discontinuation of Pieris’ other respiratory programs.
Cash Bonus Compensation. Taking into account the achievement levels of the Original 2023 Performance Goals and the Amended 2023 Performance Goals as well as the efforts of Pieris’ executives, its Compensation and Management Development Committee approved discretionary cash bonus payments to (i) Mr. Yoder in the amount of $146,149; (ii) Mr. Bures in the amount of $80,031 and (iii) Dr. Olwill in the amount of $127,702.
Stock-Based Awards
Historically, Pieris has generally granted stock options to its employees, including its Named Executive Officers, in connection with their initial employment with Pieris. Pieris has also historically granted stock options on an annual basis as part of annual performance reviews of its employees.
Pieris’ equity award program is the primary vehicle for offering long-term incentives to its executives. Pieris does not have any equity ownership guidelines for its executives, which is consistent with other pre-commercial biotechnology companies that use stock options as the long-term incentive vehicle. Further, Pieris believes that equity grants provide its executives with a strong link to its long-term performance, create an ownership culture and help to align the interests of its executives and its stockholders. In addition, the vesting feature of Pieris’ equity awards is designed to contribute to executive retention by providing an incentive for its executives to remain in its employment during the vesting period. Pieris may also make additional discretionary grants, typically in connection with the promotion of an employee, to reward an employee, for retention purposes or in other circumstances as the Compensation and Management Development Committee deems appropriate.
Employee Benefits
From time to time, Pieris has provided its Named Executive Officers with employee benefits that its board of directors believes are reasonable. Pieris’ Named Executive Officers are eligible to participate in the same broad-based employee benefit plans that are offered to Pieris’ other employees, such as health insurance, disability insurance, life insurance and a 401(k) plan.
These benefits are provided as part of the basic conditions of employment for all of Pieris’ employees, and therefore providing them to Pieris’ Named Executive Officers does not represent a significant incremental cost to Pieris. Pieris does not view employee benefits as a significant element of its comprehensive compensation structure, but does believe they can be useful in attracting, motivating and retaining the executive talent for which Pieris competes. Pieris believes that these additional benefits may assist its Named Executive Officers in performing their duties and provide time efficiencies for its Named Executive Officers in appropriate circumstances, and Pieris may consider providing additional employee benefits in the future. All future practices regarding employee benefits will be approved and subject to periodic review by the Compensation and Management Development Committee.
Clawback Policy
Pieris has adopted an incentive compensation recoupment policy that is applicable to its executive officers, and such other of its senior executives as may be determined by the Compensation and Management Development Committee. If Pieris determines that it must restate its financial results as reported in a periodic or other report filed with the SEC to correct an accounting error due to material noncompliance with any financial reporting requirement under the U.S. securities laws, Pieris will seek to recover or require forfeiture, at the direction of the Compensation and Management Development Committee, after it has reviewed the facts and circumstances that led to the requirement of the restatement and the costs and benefits of seeking recovery, any excess incentive based compensation, received by an officer covered by the policy during the three completed fiscal years immediately preceding the date on which Pieris is required to prepare the accounting restatement. Furthermore, Pieris will seek to recoup incentive compensation that is used in such a way that violates Pieris’ “Insider Trading Policy,” as made available on its website at www.pieris.com, for example, by engaging in transactions involving hedging devices or Pieris securities that are used to secure a margin or other loan.
Employment and Severance Arrangements
Pieris considers it essential to the best interests of its stockholders to foster the continuous employment of its key management personnel. In this regard, Pieris recognizes that the possibility of a change in control may exist and that the uncertainty and questions that it may raise among management could result in the departure or distraction of management personnel to the detriment of Pieris and its stockholders. In order to reinforce and encourage the continued attention and dedication of certain key members of management, Pieris has entered into written employment agreements with certain of its Named Executive Officers that, while at-will, contain certain change in control and severance provisions.
Stephen S. Yoder, Chief Executive Officer
Mr. Yoder serves as Pieris’ President and Chief Executive Officer pursuant to the Yoder Employment Agreement, which provides for a continuous term and may be terminated by either party at any time, provided that if Mr. Yoder resigns, he shall provide Pieris with at least 90 days’ prior written notice. Mr. Yoder is eligible to receive an annual bonus of up to 50% of his annual base salary based upon achievement of individual and corporate performance objectives as determined by the Compensation and Management Development Committee in its sole discretion. Mr. Yoder’s annual base salary for 2024 is $584,595 (as set by the Compensation and Management Development Committee in 2023).
Pursuant to his employment agreement, Mr. Yoder is prohibited during the term of the agreement, subject to certain exceptions, from (i) accepting any other employment or consultancy, (ii) serving on the board of directors or similar body of any other entity, unless approved by the Chairman of the Pieris board of directors, and (iii) acquiring, assuming or participating in, directly or indirectly, any financial position, investment or interest known by Mr. Yoder to be adverse or antagonistic to Pieris, Pieris’ business or prospects, financial or otherwise, or in any competing business.
Mr. Yoder’s employment agreement also contains (i) customary confidentiality obligations which are not limited by the term of the agreement, (ii) certain non-compete provisions extending during the term of the agreement and one year thereafter, and (iii) certain non-solicitation provisions during the term of the agreement and for one year thereafter. Mr. Yoder also agreed to assign certain intellectual property rights to Pieris.
Thomas Bures, Senior Vice President and Chief Financial Officer
Thomas Bures serves as Pieris’ Senior Vice President and Chief Financial Officer and is employed at will pursuant to the Bures Employment Agreement. Mr. Bures is eligible to receive an annual bonus of up to 40% of his annual base salary based upon achievement of individual and corporate performance objectives as determined by the Compensation and Management Development Committee in its sole discretion. Mr. Bures' annual base salary for 2024 is $400,155 (as set by the Compensation and Management Development Committee in 2023).
Pursuant to his employment agreement, Mr. Bures is prohibited during the term of the agreement, subject to certain exceptions, from (i) accepting any other employment or consultancy, (ii) serving on the board of directors or similar body of any other entity, and (iii) acquiring, assuming or participating in, directly or indirectly, any financial position, investment or interest known by Mr. Bures to be adverse or antagonistic to Pieris, its business or prospects, financial or otherwise, or in any competing business.
Mr. Bures also signed agreements that include (i) customary confidentiality obligations which are not limited by the term of the agreement, (ii) certain non-compete provisions extending during the term of the agreement and one year thereafter, and (iii) certain non-solicitation provisions during the term of the agreement and for one year thereafter. Mr. Bures also agreed to assign certain intellectual property rights to Pieris.
Shane Olwill, Senior Vice President, Chief Development Officer
Shane Olwill serves as Pieris’ Senior Vice President and Chief Development Officer, and is employed pursuant to an employment contract effective June 15, 2011, and with a continuous term, and may be terminated by either party provided that a statutory notice period is observed in accordance with German law. Dr. Olwill is currently eligible to receive a bonus of up to 40% of his annual base salary based upon achievement of individual and corporate performance objectives as determined by the Compensation and Management Development Committee in its sole discretion. Dr. Olwill’s annual base salary for 2024 is €304,501 (as set by the Compensation and Management Development Committee in 2023), which is approximately $328,614 using an assumed 2023 exchange rate of 1.0808 U.S. dollars per euro.
Pursuant to his employment agreement, Dr. Olwill is prohibited during the term of the agreement, subject to certain exceptions, from (i) accepting any other employment or consultancy, (ii) engaging in any paid external activities that would impair Dr. Olwill from performing his duties or unpaid side-line activities that would be competitive with or capable of being competitive with Pieris’ business, and (iii) acquiring, assuming or participating in, directly or indirectly, any financial position, investment or interest known by Dr. Olwill to be adverse or antagonistic to Pieris, its business or prospects, financial or otherwise, or in any competing business.
Dr. Olwill’s employment agreement also includes customary confidentiality obligations which are not limited by the term of the agreement. Dr. Olwill also agreed to assign certain intellectual property rights to Pieris.
Pieris and Dr. Olwill mutually agreed that in connection with the signing of the Merger Agreement, Dr. Olwill would step down effective October 31, 2024. Pieris and Dr. Olwill entered into a separation agreement, or the “Separation Agreement”, dated as of July 23, 2024, which provides that Dr. Olwill is entitled to a lump sum payment of €417,800, and is released from the obligation to work between August 1, 2024 and October 31, 2024 during which he will continue to receive his fixed salary. Dr. Olwill will depart Pieris effective October 31, 2024.
Potential Payments upon Termination or Change in Control
Stephen S. Yoder, Chief Executive Officer and President
Mr. Yoder’s employment agreement provides that if Mr. Yoder’s employment is terminated (i) by Pieris without cause or (ii) by him for good reason, then Pieris must pay Mr. Yoder (a) a lump-sum payment equal to 12 months of his base salary, (b) a pro rata portion of the bonus for the year in which the termination occurs, based on year-to-date performance as determined by the Pieris board of directors, or a committee thereof, in its sole discretion, (c) an amount equal to his health insurance premium, paid directly or as a reimbursement to Mr. Yoder, for up to a maximum of 12 months and (d) all unvested equity awards then held by Mr. Yoder will immediately vest in full and become exercisable. The severance and acceleration of any unvested options is expressly conditioned on Mr. Yoder executing and delivering to Pieris a release of claims.
Mr. Yoder’s employment agreement also provides that if within 12 months following a change of control, Mr. Yoder’s employment is terminated (i) by Pieris without cause or (ii) by him for good reason, and Mr. Yoder executes and delivers to Pieris a release of claims, then he will receive (a) a lump-sum payment equal to 12 months of his base salary at the time of his termination, (b) his target bonus amount for the year in which the termination occurs, (c) an amount equal to 12 months of his health insurance premium, paid directly or as a reimbursement to Mr. Yoder, and (d) all outstanding unvested equity awards will immediately vest in full and become exercisable following termination and any forfeiture restrictions will immediately lapse.
Thomas Bures, Senior Vice President and Chief Financial Officer
Thomas Bures’ employment agreement provides that if Mr. Bures’ employment is terminated (i) by Pieris without cause or (ii) by him for good reason, then Pieris must pay Mr. Bures a lump sum payment equal to 9 months or 75% of his base salary. The severance and acceleration of any unvested options is expressly conditioned on Mr. Bures executing and delivering to Pieris a release of claims.
Mr. Bures’ employment agreement also provides that if, following a change of control, Mr. Bures’ employment is terminated (i) by Pieris without cause or (ii) by him for good reason, and Mr. Bures executes and delivers to Pieris a release of claims, then he will receive (a) a lump-sum payment equal to 12 months of his base salary at the time of his termination, (b) his target bonus amount for the year in which the termination occurs, (c) an amount equal to 12 months of his health insurance premium, paid directly or as a reimbursement to Mr. Bures, and (d) all outstanding unvested equity awards will immediately vest in full and become exercisable following termination and any forfeiture restrictions will immediately lapse.
Shane Olwill, Senior Vice President and Chief Development Officer
Pursuant to a retention agreement provided to Shane Olwill in February 2024, if Dr. Olwill’s employment is terminated (i) by Pieris without cause or (ii) by him between July 1, 2024 and July 15, 2024, and Dr. Olwill executes and delivers to Pieris a release of claims, then he will receive a lump sum payment of €445,300. Pieris and Dr. Olwill mutually agreed that in connection with the signing of the Merger Agreement, Dr. Olwill would step down effective October 31, 2024. Pieris and Dr. Olwill entered into the Separation Agreement , dated as of July 23, 2024, which provides that Dr. Olwill is entitled to a lump sum payment of €417,800, and is released from the obligation to work between August 1, 2024 and October 31, 2024 during which he will continue to receive his fixed salary. Dr. Olwill will depart Pieris effective October 31, 2024.
Anti-Hedging and Pledging Policies
As part of Pieris’ insider trading policy, Pieris prohibits employees and directors from engaging in transactions that are designed to, or have the effect of, hedging or offsetting any decrease in the market value of Pieris’ shares owned by such employees or directors. In particular, Pieris’ insider trading policy prohibits the following transactions:
|
● |
trading in Pieris’ securities on a short-term basis. Any shares of Pieris common stock purchased in the open market must be held for a minimum of six months and ideally longer. |
|
● |
short sales of Pieris’ securities. |
|
● |
use of Pieris’ securities to secure a margin or other loan. |
|
● |
transactions in straddles, collars or other similar risk reduction or hedging devices. |
|
● |
transactions in publicly-traded options relating to Pieris’ securities (i.e., options that are not granted by Pieris). |
Outstanding Equity Awards at Fiscal Year-End
The table below summarizes the aggregate stock and option awards held by Pieris’ Named Executive Officers as of December 31, 2023, on a reverse split adjusted basis.
|
Name and Principal Position |
Number of Securities Underlying Unexercised Options (#) Exercisable |
Number of Securities Underlying Unexercised Options (#) Unexercisable |
Equity Incentive Plan Awards: Number of Securities Underlying Unexercised Unearned Options (#) Unexercisable |
Option Exercise Price ($) |
Option Expiration Date |
||
|
Stephen S. Yoder |
16,000 |
(1) |
- |
(1) |
- |
160.00 |
12/17/2024 |
|
Chief Executive Officer |
6,150 |
(1) |
- |
(1) |
- |
121.60 |
2/12/2026 |
|
5,430 |
(1) |
- |
(1) |
- |
159.20 |
2/23/2027 |
|
|
4,063 |
(1) |
- |
(1) |
- |
684.80 |
2/20/2028 |
|
|
4,750 |
(1) |
- |
(1) |
- |
247.20 |
2/26/2029 |
|
|
4,922 |
(2) |
328 |
(2) |
- |
252.00 |
2/27/2030 |
|
|
4,555 |
(3) |
2,070 |
(3) |
- |
200.00 |
3/4/2031 |
|
|
3,423 |
(4) |
4,401 |
(4) |
- |
242.40 |
2/22/2032 |
|
|
- |
(5) |
7,926 |
(5) |
- |
111.20 |
2/24/2033 |
|
|
Thomas Bures |
1,563 |
(1) |
- |
(1) |
- |
451.20 |
12/18/2027 |
|
Senior Vice President and Chief Financial Officer |
891 |
(1) |
- |
(1) |
- |
247.20 |
2/26/2029 |
|
938 |
(2) |
62 |
(2) |
- |
252.00 |
2/27/2030 |
|
|
1,289 |
(3) |
586 |
(3) |
- |
200.00 |
3/4/2031 |
|
|
1,181 |
(4) |
1,517 |
(4) |
- |
242.40 |
2/22/2032 |
|
|
- |
(5) |
3,114 |
(5) |
- |
111.20 |
2/24/2033 |
|
|
Shane Olwill |
1,813 |
(1) |
- |
(1) |
- |
160.00 |
12/17/2024 |
|
Senior Vice President and Chief Development Officer |
750 |
(1) |
- |
(1) |
- |
121.60 |
2/12/2026 |
|
704 |
(1) |
- |
(1) |
- |
159.20 |
2/23/2027 |
|
|
1,125 |
(1) |
- |
(1) |
- |
684.80 |
2/20/2028 |
|
|
1,025 |
(1) |
- |
(1) |
- |
247.20 |
2/26/2029 |
|
|
1,055 |
(2) |
70 |
(2) |
- |
252.00 |
2/27/2030 |
|
|
1,289 |
(3) |
586 |
(3) |
- |
200.00 |
3/4/2031 |
|
|
1,181 |
(4) |
1,517 |
(4) |
- |
242.40 |
2/22/2032 |
|
|
- |
(5) |
3,114 |
(5) |
- |
111.20 |
2/24/2033 |
|
(1) |
The option award is fully vested. |
|
(2) |
The option award has a grant date of February 27, 2020 and vests pursuant to the following schedule: 25% of the option vested on the one-year anniversary of the grant date and the remaining 75% of the option shall vest as to 6.25% of the option shares at the end of each successive three-month period thereafter. |
|
(3) |
The option award has a grant date of March 4, 2021 and vests pursuant to the following schedule: 25% of the option vested on the one-year anniversary of the grant date and the remaining 75% of the option shall vest as to 6.25% of the option shares at the end of each successive three-month period thereafter. |
|
(4) |
The option award has a grant date of February 22, 2022 and vests pursuant to the following schedule: 25% of the option vested on the one-year anniversary of the grant date and the remaining 75% of the option shall vest as to 6.25% of the option shares at the end of each successive three-month period thereafter. |
|
(5) |
The option award has a grant date of February 24, 2023 and vests pursuant to the following schedule: 25% of the option vests on the one-year anniversary of the grant date and the remaining 75% of the option shall vest as to 6.25% of the option shares at the end of each successive three-month period thereafter. |
Director Compensation
Pieris’ director compensation program is administered by the Pieris board of directors with the assistance of the Compensation and Management Development Committee. The Compensation and Management Development Committee conducts a periodic review of director compensation and makes recommendations to the Pieris board of directors with respect thereto. The Compensation and Management Development Committee has periodically engaged the services of Pearl Meyer to review and provide recommendations concerning Pieris’ non-employee director compensation policy.
During 2023, the Pieris board of directors was compensated pursuant to its Amended and Restated Non-Employee Director Compensation Policy, or the Director Compensation Policy, which provides for cash and equity-based compensation for service on the Pieris board of directors and its committees. Under the Director Compensation Policy, Pieris’ non-employee directors receive the following cash compensation for their service on the Pieris board of directors and its committees:
|
Board of Directors or Committee of Board of Directors |
Annual Retainer Amount for Director (non-Chairperson) |
Annual Retainer Amount for Chairperson |
|
Board of Directors |
$35,000 |
$65,000 |
|
Audit Committee |
$7,500 |
$15,000 |
|
S&T Committee(1) |
$5,000 |
$10,000 |
|
Compensation and Management Development Committee |
$5,000 |
$10,000 |
|
Nominating and Corporate Governance Committee(1) |
$4,000 |
$8,000 |
(1) On July 23, 2024, the Pieris board of directors determined to dissolve the S&T Committee and the Nominating and Corporate Governance Committees effective as of September 30, 2024.
During 2023, the Pieris board of directors was also compensated with stock option grants, on a reverse split adjusted bases, under the Director Compensation Policy as shown in the table below:
|
Board of Directors |
Annual Stock Option Grants |
Initial Stock Option Grant for Newly Appointed or Elected Directors |
|
Director |
500 shares of common stock |
1,000 shares of common stock |
|
Chairperson |
Additional 63 shares of common stock |
|
The annual stock options vest as to 100% one (1) year after the date of grant of such option, and the initial stock options vest initial stock options vest as to 33% one (1) year after the date of grant of such option, with the remaining 67% vesting in eight (8) equal quarterly installments at the end of each full fiscal quarter following the initial vesting date. The annual stock options granted to the non-employee directors and the Chairperson under the Director Compensation Policy will be granted on the date of the meeting of the Pieris board of directors coincident with or immediately following Pieris’ annual meeting of stockholders and will vest on the date of the next regular annual stockholders meeting. All stock options granted under the Director Compensation Policy have an exercise price equal to the fair market value of Pieris common stock on the grant date and terminate 10 years after the grant date.
In addition, in 2023 Pieris announced its intention to explore engaging in one or more strategic transactions, including mergers, reverse mergers, acquisitions, other business combinations or sales of assets, or other strategic transactions. The Pieris board of directors appointed a Transaction Committee of the Pieris board of directors to help facilitate the Pieris board of directors’ consideration of such potential strategic transactions, which consists of Pieris’ directors James Geraghty, Christopher Kiritsy, and Peter Kiener, D.Phil. Each member of the Transaction Committee is compensated for their service on the committee with an annual fee of $10,000 paid quarterly in arrears following the end of each fiscal quarter, provided that the amount of such payment will be prorated for any portion of such quarter that such director was not serving as a member of the Transaction Committee. On July 23, 2024, the Pieris board of directors determined to dissolve the Transaction Committee effective as of September 30, 2024.
The table below summarizes all compensation earned by each of Pieris’ non-employee directors for services performed during its fiscal year ended December 31, 2023, on a reverse split adjusted basis. Mr. Yoder is not in the table below because he receives no separate compensation for his services as a director of Pieris, and all of the compensation earned by Mr. Yoder during Pieris’ fiscal year ended December 31, 2023 as an executive officer of Pieris is reflected in the Summary Compensation Table above.
|
Name |
Fees Earned or Paid in Cash ($) |
Option Awards ($) (8) |
Total ($) |
||||||
|
James Geraghty (1) |
$84,984 | $9,103 | $94,087 | ||||||
|
Michael Richman (2) |
$44,000 | $8,092 | $52,092 | ||||||
|
Christopher Kiritsy (3) |
$64,484 | $8,092 | $72,576 | ||||||
|
Ann Barbier, M.D., Ph.D. (4) |
$44,000 | $8,092 | $52,092 | ||||||
|
Peter Kiener, D.Phil. (5) |
$56,984 | $8,092 | $65,076 | ||||||
|
Matthew L. Sherman, M.D. (6) |
$45,000 | $8,092 | $53,092 | ||||||
|
Maya R. Said, Sc.D. (7) |
$44,000 | $8,092 | $52,092 |
|
(1) |
As of December 31, 2023, Mr. Geraghty held option awards for 4,035 shares at exercise prices ranging from $20.80 to $617.60. |
|
(2) |
As of December 31, 2023, Mr. Richman held option awards for 5,487 shares at exercise prices ranging from $20.80 to $617.60. |
|
(3) |
As of December 31, 2023, Mr. Kiritsy held option awards for 3,312 shares at exercise prices ranging from $20.80 to $617.60. |
|
(4) |
As of December 31, 2023, Dr. Barbier held option awards for 2,875 shares at exercise prices ranging from $20.80 to $514.40. |
|
(5) |
As of December 31, 2023, Dr. Kiener held option awards for 2,875 shares at exercise prices ranging from $20.80 to $356.80. |
|
(6) |
As of December 31, 2023, Dr. Sherman held option awards for 2,875 shares at exercise prices ranging from $20.80 to $356.80. |
|
(7) |
As of December 31, 2023, Dr. Said held option awards for 2,625 shares at exercise prices ranging from $20.80 to $440.00. |
|
(8) |
These amounts represent the aggregate grant date fair value of option awards granted to each director in Pieris’ fiscal year ended December 31, 2023 computed in accordance with FASB ASC Topic 718. A discussion of the assumptions used in determining grant date fair value may be found in Note 11 to Pieris’ Financial Statements included in Pieris’ Annual Report on Form 10-K for the fiscal year ended December 31, 2023. |
Delinquent Section 16(a) Reports
Section 16(a) of the Exchange Act requires the Pieris officers and directors, and persons who own more than 10% of a registered class of Pieris’ equity securities, to file reports of ownership and changes in ownership with the SEC. Officers, directors and greater-than 10% stockholders are required by SEC regulation to furnish us with copies of all Section 16(a) forms they file.
Based solely on Pieris’ review of the copies of forms that it has received, and written representations from certain reporting persons that they were not required to file Form 5s relating to Pieris’ stock, Pieris believes that in in 2023 and to date, other than James Geraghty, who had one inadvertent late filing on Form 4, all filing requirements pursuant to Section 16(a) of the Exchange Act relating to Pieris’ officers, directors and principal shareholders were satisfied.
Pay versus Performance
As required by SEC rules, Pieris is providing the following information about the relationship between executive compensation actually paid and certain financial performance of Pieris. The disclosure included in this section is prescribed by SEC rules and does not necessarily align with how Pieris or its Compensation and Management Development Committee view the link between Pieris’ performance and the pay of its principal executive officer, or PEO, and its NEOs.
|
Year |
Summary Compensation Table Total for Stephen S. Yoder, Principal Executive Officer ($) |
Compensation Actually Paid to PEO ($)(1) |
Average Summary Compensation Table Total for Non-PEO NEOs ($)(2) |
Average Compensation Actually Paid to Non-PEO NEOs ($)(3) |
Value of Initial Fixed $100 Investment Based on Total Shareholder Return ($)(4) |
Net Loss (in thousands) ($)(5) |
|
2023 |
1,358,952 |
553,945 |
717,241 |
448,400 |
7.28 |
(24,543) |
|
2022 |
2,062,843 |
(526,880) |
954,079 |
190,343 |
41.60 |
(33,277) |
|
2021 |
1,692,917 |
3,005,107 |
1,061,609 |
1,350,793 |
151.20 |
(45,738) |
(1) The dollars amounts reported in this column represent the amount of "compensation actually paid" to Mr. Yoder. The amounts are computed in accordance with SEC rules by deducting and adding the following amounts from the "Total" column of the Summary Compensation Table, or SCT, on page 188 (pursuant to SEC rules, fair value at each measurement date is computed in a manner consistent with the fair value methodology used to account for share-based payments in our financial statements under GAAP):
|
Year |
Reported Summary Compensation Table Total for PEO ($) |
Reported Value of Equity Awards (a) ($) |
Equity Award Adjustments (b) ($) |
Compensation Actually Paid to PEO ($) |
|
2023 |
1,358,952 |
(614,818) |
(190,189) |
553,945 |
|
2022 |
2,062,843 |
(1,288,128) |
(1,301,595) |
(526,880) |
|
2021 |
1,692,917 |
(858,754) |
2,170,944 |
3,005,107 |
(a) The grant date fair value of equity awards represents the total of the amounts reported in the "Option Awards" column in the SCT for the applicable year.
(b) The amounts deducted or added in calculating the equity award adjustments are as follows:
|
Year |
Year End Fair Value of Outstanding and Unvested Equity Awards Granted in the Year ($) |
Year over Year Change in Fair Value of Outstanding and Unvested Equity Awards Granted in Prior Years ($) |
Fair Value as of Vesting Date of Equity Awards Granted and Vested in the Year ($) |
Year over Year Change in Fair Value of Equity Awards Granted in Prior Years that Vested in the Year ($) |
Fair Value at the End of the Prior Year of Equity Awards that Failed to Meeting Vesting Conditions in the Year ($) |
Value of Dividends or other Earnings Paid on Stock or Option Awards not Otherwise Reflected in Fair Value or Total Compensation ($) |
Total Equity Award Adjustments ($) |
||||||||||||||
|
2023 |
57,995 | (237,467 | ) | — | (10,716 | ) | — | — | (190,189 | ) | |||||||||||
|
2022 |
312,186 | (1,011,551 | ) | — | (602,230 | ) | — | — | (1,301,595 | ) | |||||||||||
|
2021 |
1,469,796 | 436,967 | — | 264,181 | — | — | 2,170,944 |
(2) The dollar amounts reported this column represent the average of the amounts reported for Pieris’ NEOs as a group (excluding Mr. Yoder, who has served as Pieris’ CEO since 2010) in the “Total” column of the SCT in each applicable year. The names of each of the NEOs (excluding Mr. Yoder) included for purposes of calculating the average amounts in each applicable year are as follows: (i) for 2023, Shane Olwill and Thomas Bures, (ii) for 2022, Ahmed Mousa and Thomas Bures, and (iii) for 2021, Hitto Kaufmann and Tim Demuth.
(3) The dollar amounts reported in this column represent the average amount of "compensation actually paid" to the NEOs as a group (excluding Mr. Yoder), as computed in accordance with SEC rules by deducting and adding the following amounts from the "Total" column of the SCT, using the same methodology described above in Note 1. The amounts added and deducted are as follows:
|
Year |
Average Reported Summary Compensation Table Total for Non-PEO NEOs ($) |
Average Reported Value of Equity Awards (a) ($) |
Average Equity Award Adjustments (b) ($) |
Average Compensation Actually Paid to Non- PEO NEOs ($) |
||||||||
|
2023 |
717,241 | (241,536 | ) | (27,305 | ) | 448,400 | ||||||
|
2022 |
954,079 | (444,183 | ) | (319,553 | ) | 190,343 | ||||||
|
2021 |
1,061,609 | (603,882 | ) | 893,066 | 1,350,793 |
(a) The grant date fair value of equity awards represents the total of the amounts reported in the "Option Awards" column in the SCT for the applicable year.
(b) The amounts added or deducted in calculating the average equity award adjustments are as follows:
|
Year |
Average Year End Fair Value of Outstanding and Unvested Equity Awards Granted in the Year ($) |
Year over Year Average Change in Fair Value of Outstanding and Unvested Equity Awards Granted in Prior Years ($) |
Average Fair Value as of Vesting Date of Equity Awards Granted and Vested in the Year ($) |
Year over Year Average Change in Fair Value of Equity Awards Granted in Prior Years that Vested in the Year ($) |
Average Fair Value at the End of the Prior Year of Equity Awards that Failed to Meeting Vesting Conditions in the Year ($) |
Average Value of Dividends or other Earnings Paid on Stock or Option Awards not Otherwise Reflected in Fair Value or Total Compensation ($) |
Total Equity Award Adjustments ($) |
||||||||||||||
|
2023 |
22,789 | (48,705 | ) | — | (1,389 | ) | — | — | (27,305 | ) | |||||||||||
|
2022 |
107,651 | (271,860 | ) | — | (155,343 | ) | — | — | (319,553 | ) | |||||||||||
|
2021 |
767,328 | 84,218 | — | 41,520 | — | — | 893,066 |
(4) Cumulative TSR measures the cumulative value of $100 invested on the last trading day before the earliest fiscal year in the table, or December 31, 2020, including the reinvestment of dividends, through and including the end of the applicable fiscal year for which TSR is calculated, or December 31, 2021, December 31, 2022, and December 31, 2023. Pieris did not pay dividends in the periods presented.
(5) The dollar amounts reported represent the amount of net income (loss) reflected in the Pieris ' audited financial statements for the applicable year.
Analysis of the Information Presented in the Pay Versus Performance Table
Pieris generally seeks to incentivize long-term performance, and therefore does not specifically align its performance measures with "compensation actually paid" (as computed in accordance with the SEC rules) for a particular year. In accordance with the SEC rules, Pieris is providing the following descriptions of the relationships between information presented in the Pay versus. Performance table above.
Relationship between Compensation and Cumulative Total Shareholder Return
The following chart sets forth the relationship between compensation actually paid to Pieris’ PEO, the average compensation actually paid to Pieris’ non-PEO NEOs, and Pieris’ TSR over the period covering fiscal years 2021, 2022, and 2023. A large component of Pieris’ executive compensation is equity based to align compensation with performance but also includes other appropriate incentives such as cash bonuses that are designed to incentivize Pieris’ executives to achieve annual corporate goals. Pieris believes the equity-based compensation strongly aligns Pieris’ PEO's and non-PEOs' interests with those of stockholders to maximize long-term value and encourages long-term employment.

Relationship between Compensation and Net Income (Loss)
The following chart sets forth the relationship between compensation actually paid to Pieris’ PEO, the average compensation actually paid to Pieris’ non-PEO NEOs, and Pieris’ net loss over the period covering fiscal years 2021, 2022, and 2023. Because Pieris is not a commercial-stage company, Pieris did not have any revenue during the periods presented, other than milestones and grant income in the reported years. Consequently, Pieris has not historically looked to net income as a performance measure for its executive compensation program. In 2021, 2022, and 2023 Pieris’ net loss decreased, and the compensation actually paid for both its PEO and its non-PEO NEOs decreased between 2021 and 2022, and increased in 2023.
All information provided under the heading "Pay versus. Performance" and "Analysis of the Information Presented in the Pay Versus Performance Table" will not be deemed to be incorporated by reference in any filing of Pieris’ under the Securities Act of 1933, as amended, whether made before or after the date hereof and irrespective of any general incorporation language in any such filing.
PALVELLA EXECUTIVE AND DIRECTOR COMPENSATION
Executive Compensation
This section discusses the material components of the executive compensation program for Palvella’s executive officers who are named in the “2023 Summary Compensation Table” below. As a “smaller reporting company”, Palvella is not required to include a Compensation Discussion and Analysis and Palvella has elected to comply with the scaled disclosure requirements applicable to smaller reporting companies. Palvella’s named executive officers or the Palvella NEOs, for the year ended December 31, 2023, which consists of Palvella’s principal executive officer, and its other two most highly compensated executive officers, are:
|
• |
Wesley H. Kaupinen, President and Chief Executive Officer; |
|
• |
Kathleen Goin, Chief Operating Officer; and |
|
• |
Jeffrey Martini, Ph.D., Chief Scientific Officer |
2023 Summary Compensation Table
The following table sets forth information concerning the compensation of the Palvella NEOs for the year ended December 31, 2023.
|
Name and Principal Position |
Year |
Salary |
Bonus |
Option Awards |
Non-Equity Incentive Plan Compensation |
All Other Compensation |
Total |
||||||||||||
|
Wesley H. Kaupinen |
|||||||||||||||||||
|
President and Chief Executive Officer |
2023 |
$375,462 | $- | $209,858 | - | $643 | $585,963 | ||||||||||||
|
2022 |
$350,000 | $140,000 | - | - | $643 | $490,643 | |||||||||||||
|
Kathleen Goin |
|||||||||||||||||||
|
Chief Operating Officer |
2023 |
$354,975 | $49,056 | $941,776 | - | - | $1,345,807 | ||||||||||||
|
2022 |
$300,000 | $90,000 | - | - | - | $390,000 | |||||||||||||
|
Jeffrey Martini, PhD |
|||||||||||||||||||
|
Chief Scientific Officer |
2023 |
$293,588 | $39,852 | $281,539 | - | - | $614,979 | ||||||||||||
|
2022 |
$275,000 | $82,500 | - | - | - | $357,500 |
___________________
|
(1) |
Amounts shown are cash incentive payments earned in respect of 2023 or 2022, as the case may be, performance and paid in 2024 or 2023, as the case may be. |
|
(2) |
Amounts shown in this column represent the aggregate grant date fair value of the stock options awarded to the Palvella NEO in fiscal year 2023. For a discussion of the assumptions and methodologies used to calculate the amounts referred to above, please see the discussion of option awards contained in Note 7, 2019 Equity Inventive Plan, to Palvella’s financial statements included elsewhere in this proxy statement/prospectus. The amounts reported in this column reflect the accounting cost for these stock options and do not correspond to the actual economic value that may be received by the NEO upon exercise of the stock options. |
|
(3) |
Amounts shows are the cost of a term life insurance policy for Mr. Kaupinen. |
Narrative Disclosure to the Summary Compensation Table 2023 Elements of compensation
The compensation of Palvella’s NEOs generally consists of base salary, annual cash bonus opportunities, and other benefits, as described below.
2023 Base Salaries
The base salary payable to Palvella NEO is intended to provide a fixed component of compensation reflecting the executive’s skill set, experience, role, responsibilities, and contributions. For 2023, the Palvella’s annual base salary rates were: $375,462 for Mr. Kaupinen, $354,975 for Ms. Goin and $293,588 for Dr. Martini. These reflect temporary reductions in salary for a portion of 2023.
Annual Cash Bonus Opportunities
Each of Palvella NEOs’ performance-based cash bonus opportunity is expressed as a percentage of base salary that can be achieved at a target level by meeting predetermined corporate and individual performance objectives. Palvella’s compensation committee annually sets each executive’s target bonus for the year. The 2023 annual bonus opportunity for Mr. Kaupinen, Ms. Goin and Dr. Martini were targeted at 40%, 30% and 30% of their respective base salaries.
For 2023, all Palvella NEOs were eligible to earn their annual bonuses pursuant to the achievement of the following corporate performance goals: based on corporate development and management goals, including leadership training and retention, corporate development, QTORIN platform expansion and CMC milestones; and achievement of certain goals related to QTORIN rapamycin for the treatment of microcystic LM.
Following a review of the performance goals attained in 2023, Palvella’s compensation committee recommended, and Palvella’s board of directors approved, 2023 annual bonus payments to Ms. Goin and Dr. Martini in amounts equal to $49,056 and $39,852, respectively. Mr. Kaupinen recommended a deferral of any consideration of a 2023 annual cash bonus for his compensation.
Equity Compensation
Palvella grants stock options and restricted stock to its employees from time to time. Typically, equity awards granted upon an individual’s commencement of employment vest 25% upon the first anniversary of the individual’s start date, and monthly over 36 months thereafter, subject to continued employment. Subsequent grants generally vest monthly over 48 months, contingent on continued employment. In 2023, Palvella granted option awards to Palvella’s NEOs, Mr. Kaupinen of 69,260, Ms. Goin of 310,817 and Dr. Martini of 92,917.
Other Elements of Compensation
As described in the “All Other Compensation” column in the “— 2023 Summary Compensation Table” above, Palvella covers the cost of a term life insurance policy for Mr. Kaupinen.
Palvella does not maintain any retirement plans or nonqualified deferred compensation plans.
Named Executive Officer Arrangements
Wesley H. Kaupinen
Palvella entered into an employment agreement with Mr. Kaupinen, dated May 20, 2020, or the “Kaupinen Employment Agreement”, providing for his position as President and Chief Executive Officer and an annual base salary of $350,000. Under the Kaupinen Employment Agreement, Mr. Kaupinen is eligible to receive an annual bonus. In 2021, Palvella’s compensation committee set Mr. Kaupinen’s annual bonus target at up to 40% of Mr. Kaupinen’s annual base salary, based on the achievement of corporate performance objectives established by Palvella’s compensation committee. Under the Kaupinen Employment Agreement, Mr. Kaupinen is eligible to participate in Palvella’s employee benefit plans, subject to the eligibility requirements of those plans.
The Kaupinen Employment Agreement provides for an indefinite term and is terminable (i) at will by Palvella or by Mr. Kaupinen, provided that 30 days’ advance notice must be provided by the terminating party in the event of a termination of employment without “cause” by Palvella or Mr. Kaupinen’s resignation without “good reason”; (ii) on the date that Mr. Kaupinen provides Palvella with written notice that he is terminating his employment for good reason (subject to any applicable cure period); and (iii) on the date of his death or on the date of his disability, as reasonably determined by Palvella.
Under the Kaupinen Employment Mr. Kaupinen is entitled to certain benefits upon termination of employment as described below in the section entitled “—Termination Payments.”
Kathleen Goin
Ms. Goin is party to an offer letter dated August 19, 2019, or the Goin Offer Letter, that provides for at-will employment and provides for an initial base salary of $300,000 for full-time work commencing in 2020. Under the Goin Offer Letter, Ms. Goin is eligible to receive an annual cash incentive award opportunity under Palvella’s bonus plan. In addition, in connection with her commencement of employment, Ms. Goin received two option awards to purchase, in the aggregate, 83,291 shares of Palvella common stock, with each option award vesting in accordance with Palvella’s standard vesting schedule for new hire grants. Ms. Goin is eligible to participate in Palvella’s employee benefits plans that are generally made available by Palvella to its employees, subject to the eligibility requirements of those plans.
Jeffrey Martini, PhD
Dr. Martini is party to an offer letter dated July 27, 2020, or the “Martini Offer Letter”, that provides for at-will employment and provides for an initial base salary of $275,000. Under the Martini Offer Letter, Dr. Martini is eligible to receive an annual cash incentive award opportunity under Palvella’s bonus plan targeted at 30% of base salary. Dr. Martini was also eligible for a one-time payment of $43,925 related to the achievement of his 2020 Palvella Goals and Objectives. In addition, in connection with his commencement of employment, Dr. Martini received an option award to purchase 104,283 shares of Palvella common stock, which vests in accordance with Palvella’s standard vesting schedule for new hire grants. Dr. Martini is eligible to participate in Palvella’s employee benefits plans that are generally made available by Palvella to its employees, subject to the eligibility requirements of those plans.
Confidentiality, Non-Competition, Non-Solicitation and Inventions Agreements
Mr. Kaupinen, Ms. Goin and Dr. Martini each entered into a Confidentiality, Assignment of Inventions, and Restrictive Covenant Agreement, or the Restrictive Covenant Agreements, with Palvella that includes customary prohibitions against competition with Palvella and solicitation of Palvella’s customers and employees, both during employment and for the 12-month following any cessation of employment. The Restrictive Covenant Agreements also include standard provisions relating to Palvella’s intellectual property rights and prohibiting the executive from disclosing confidential information.
Payment of any severance benefits under each Palvella NEO’s agreement is conditioned on continued compliance with such Palvella NEO’s Restrictive Covenant Agreement.
Termination Payments
Regardless of the manner in which an NEO’s employment or service with Palvella terminates, the Palvella NEO is entitled to receive accrued and unpaid base salary. In addition, Mr. Kaupinen and Ms. Goin are contractually entitled to severance upon certain termination events, as described below.
Wesley H. Kaupinen
Pursuant to the Kaupinen Employment Agreement, upon termination of employment by Palvella without “cause” or by Mr. Kaupinen for “good reason”, Palvella will provide Mr. Kaupinen with 12 months of salary continuation. Payment of Mr. Kaupinen’s severance is conditioned on (i) Mr. Kaupinen’s execution of a general release of claims in favor of Palvella and its affiliates; (ii) Mr. Kaupinen’s continued compliance with the provisions of his Restrictive Covenant Agreement; and (iii) Palvella being financially solvent at the time any such severance payment becomes due, and that the payment of any such severance amounts would not cause Palvella to become insolvent.
Under the Kaupinen Employment Agreement, “cause” generally means any of the following: (i) indictment, commission of, or other entry of a plea of guilty or no contest to, (A) a felony or (B) any crime (other than a felony) that causes Palvella or its affiliates public disgrace or disrepute, or adversely affects Palvella or its affiliates’ operations or financial performance or the relationship Palvella has with its affiliates, customers and suppliers; (ii) commission of an act of gross negligence, willful misconduct, fraud, embezzlement, theft or material dishonesty with respect to Palvella or any of its affiliates; (iii) a breach of Mr. Kaupinen’s fiduciary duties to Palvella or any of its affiliates; (iv) alcohol abuse or use of controlled substances (other than prescription drugs taken in accordance with a physician’s prescription); (v) material breach of any agreement with Palvella or any of its affiliates, including the Kaupinen Employment Agreement and his Restrictive Covenant Agreement; (vi) a material breach of any of Palvella’s policies regarding employment practices; or (vii) refusal to perform or repeated failure to perform, the lawful directives of the Palvella board of directors, if not cured within 15 days following his receipt of Palvella’s written notice.
Under the Kaupinen Employment Agreement, “good reason” generally means Mr. Kaupinen’s resignation for any of the following reasons, provided he provides notice to Palvella within 90 days of the initial occurrence of the event, Palvella fails to cure the issue within 30 days, and he resigns within 30 days of the end of the cure period: (i) a material reduction in his title, duties, authority or responsibilities, provided that such reduction would not be deemed to have occurred if, following a change of control, (A) Palvella remains a separate entity, and he remains the most senior executive directly responsible for Palvella’s operations, or (B) if Palvella does not remain a separate entity, Palvella is the most senior executive directly responsible for the operations of the acquiring entity; (ii) a material breach of the Kaupinen Employment Agreement by Palvella; (iii) a material reduction in his base salary paid by Palvella to which he has not provided written consent, other than a decrease in which Palvella contemporaneously decrease the salaries of all of its senior executives; or (iv) a change of more than 50 miles in the geographic location at which he performs his services.
Under the Kaupinen Employment Agreement, if payments and benefits payable to Mr. Kaupinen in connection with a change in control constitute “excess parachute payments” under Section 280G of the Code, then such payments and benefits will be reduced to the minimum extent necessary so that no portion thereof will fail to be tax-deductible by Palvella or its affiliates under Section 280G of the Code.
Kathleen Goin
Palvella entered into a severance agreement with Ms. Goin, dated May 22, 2020, or the Goin Severance Agreement. The Goin Severance Agreement provides that upon termination of employment by Palvella without “cause,” Palvella will provide Ms. Goin with three months of salary continuation. Payment of Ms. Goin’s severance is conditioned on (i) Ms. Goin’s execution of a general release of claims in favor of Palvella and its affiliates; (ii) Ms. Goin’s continued compliance with the provisions of her Restrictive Covenant Agreement; and (iii) Palvella being financially solvent at the time any such severance payment becomes due, and that the payment of any such severance amounts would not cause Palvella to become insolvent.
Under the Goin Severance Agreement, “cause” generally has the same meaning as such term has in the Kaupinen Employment Agreement.
Palvella’s compensation committee will review its compensatory arrangements with Palvella NEOs prior to the closing of the Merger to determine whether any adjustments are appropriate.
Outstanding Equity Awards at Fiscal Year-End
The following table summarizes the number of shares of common stock underlying outstanding equity incentive plan awards for each Palvella NEO as of December 31, 2023.
|
Option Awards |
Stock Awards |
|||||||
|
Name |
Grant Date |
Number of Securities Underlying Unexercised Options (#) Exercisable |
Number of Securities Underlying Unexercised Options (#) Unexercisable |
Option Exercise Price |
Option Expiration Date |
Number of Shares of Stock That Have Not Vested |
Market Value of Shares of Stock That Have Not Vested |
|
|
— |
— |
— |
— |
— |
— |
— |
— |
|
|
Wesley H. Kaupinen |
2/23/2023 |
27,415 (1) |
41,845 |
$3.03 |
2/22/2033 |
— |
— |
|
|
Wesley H. Kaupinen |
5/28/2024 |
32,903 (2) |
230,320 |
$2.33 |
5/27/2034 |
— |
— |
|
|
Kathleen Goin |
10/30/2019 |
38,314 (3) |
- |
$2.21 |
10/29/2029 |
— |
— |
|
|
Kathleen Goin |
10/30/2019 |
45,248 (3) |
- |
$2.21 |
10/29/2029 |
— |
— |
|
|
Kathleen Goin |
10/15/2020 |
81,555 (4) |
1,736 |
$2.81 |
10/14/2030 |
— |
— |
|
|
Kathleen Goin |
2/23/2023 |
109,968 (1) |
167,846 |
$3.03 |
2/22/2033 |
— |
— |
|
|
Kathleen Goin |
2/23/2023 |
13,063 (1) |
19,940 |
$3.03 |
2/22/2033 |
— |
— |
|
|
Kathleen Goin |
5/28/2024 |
12,032 (2) |
84,221 |
$2.33 |
5/27/2034 |
— |
— |
|
|
Jeffrey Martini, Ph.D |
10/12/2020 |
104,283 (5) |
- |
$2.81 |
10/11/2030 |
— |
— |
|
|
Jeffrey Martini, Ph.D |
2/23/2023 |
23,715 (1) |
36,199 |
$3.03 |
2/22/2033 |
— |
— |
|
|
Jeffrey Martini, Ph.D |
2/23/2023 |
13,063 (1) |
19,940 |
$3.03 |
2/22/2033 |
— |
— |
|
|
Jeffrey Martini, Ph.D |
5/28/2024 |
8,033(2) |
56,233 |
$2.33 |
5/27/2034 |
— |
— |
|
_______________________
|
(1) |
This option was granted on February 23, 2023 and vests in 48 substantially equal monthly installments. Vesting of the award requires continued employment through the applicable vesting dates. |
|
(2) |
This option was granted on May 28, 2024 and vests in 48 substantially equal monthly installments. Vesting of the award requires continued employment through the applicable vesting dates. |
|
(3) |
This option was granted on October 30, 2019, and vests as follows: 25% of the option shares vested on October 30, 2020, and the remaining 75% of the option shares vest in 36 substantially equal monthly installments thereafter. Vesting of the award requires continued employment through the applicable vesting dates. |
|
(4) |
This option was granted on October 15, 2020, and vests in 48 substantially equal monthly installments. Vesting of the award requires continued employment through the applicable vesting dates. |
|
(5) |
This option was granted on October 12, 2020, and vests as follows: 25% of the option shares vested on October 12, 2021, and the remaining 75% of the option shares vest in 36 substantially equal monthly installments thereafter. Vesting of the award requires continued employment through the applicable vesting dates. |
Equity Compensation Plans
2019 Equity Incentive Plan
Palvella maintains the 2019 Plan. The purposes of the 2019 Plan are to enable Palvella to recruit and retain highly qualified employees, directors, consultants and other service providers and to provide them with an incentive for productivity and the opportunity to share in Palvella’s growth and value. Palvella may provide these incentives through the grant of stock options, SARs, restricted stock and RSUs.
As noted above, Palvella expects to terminate the 2019 Plan and will cease granting awards thereunder upon the effective date of the 2024 equity plan described under “Proposal 3 – Approval of the 2024 Equity Incentive Plan.” Any outstanding awards will continue to be subject to the terms of the 2019 Plan and the applicable award agreements, until such awards are exercised or settled, or until they terminate or expire by their terms.
The material terms of the 2019 Plan are summarized below. These summaries are qualified in their entirety by reference to the actual text of the 2019 Plan, which is filed as an exhibit to the Registration Statement on Form S-4 of which this proxy statement/prospectus forms a part.
Eligibility and Administration. Palvella’s employees, directors, consultants, and other service providers are eligible to receive awards as participants under the 2019 Plan. The Palvella board of directors administers the 2019 Plan, and it may appoint a committee to perform some or all of its administrative functions thereunder. Directors who are eligible for awards or have received awards may vote on any matters affecting the administration of the 2019 Plan or the grant of awards, but no director may act upon the grant of an award to himself or herself. The Palvella board of directors has full authority to make all determinations and interpretations under the 2019 Plan, and the authority to determine award recipients, the numbers and types of awards to be granted, and the provisions of each award, including the period of their exercisability and the vesting schedule applicable to an award, among other powers under the 2019 Plan.
Shares Subject to the Plan. The maximum number of shares that may be issued in respect of awards under the 2019 Plan is 2,861,768 shares, which consist of authorized but unissued shares of Palvella’s non-voting common stock. All shares subject to the 2019 Plan are eligible to be granted pursuant to incentive stock option, or ISOs, awards, which are intended to qualify for tax treatment as set forth under Section 422 of the Code. As of October 28, 2024, 78,959 options to purchase shares of Palvella’s non-voting common stock had been exercised and options to purchase 2,149,138 shares of Palvella’s non-voting common stock remain outstanding, with a weighted average exercise price of $2.62 per share, and 633,671 shares remain available for issuance.
Share Recycling. If an award under the 2019 Plan expires, terminates, is canceled, forfeited or is settled for cash, any shares subject to such award may, to the extent of such expiration, termination, cancellation, forfeiture or cash settlement, be used again for new grants under the 2019 Plan. In addition, the delivery of any share withheld in settlement of a tax withholding obligation associated with an award, or in satisfaction of the exercise price payable upon exercise of an option will cause that share to become available for grant again under the 2019 Plan.
Adjustments. In the event of any recapitalization, reclassification, reorganization, merger, consolidation stock split or combination, stock dividend or other similar event or transaction affecting the shares, Palvella’s board of directors will make equitable substitutions or adjustments to (i) the aggregate number, class and/or issuer of the securities that may be issued under the 2019 Plan, (ii) to the number, class and/or issuer of securities subject to outstanding awards, and (iii) to the exercise price of outstanding options or SARs.
Options. The 2019 Plan provides for the grant of both incentive stock options and non-qualified stock options to purchase shares of Palvella common stock at a stated exercise price. The exercise price of stock options granted under the 2019 Plan and the term applicable to each option will be determined by Palvella’s board of directors at the time of grant, however the ISO rules impose certain limitations on the exercise price and maximum term of such grants.
Restricted stock. The 2019 Plan also allows for the grant or sale of restricted stock. The price, if any, of shares of restricted stock will be determined by the Palvella board of directors. During the vesting period, a participant will have the right to receive any dividends with respect to restricted stock, provided that the plan administrator may specify that any such dividends are subject to the same vesting schedule as the shares to which they relate.
Stock appreciation rights and restricted stock units. In addition, the 2019 Plan allows for the grant of SARs and RSUs, with terms as determined by the Palvella board of directors in accordance with the 2019 Plan. However, as noted above, Palvella has not granted any SARs or RSUs under the 2019 Plan.
Change of Control. If Palvella experiences a “change of control” (including certain dissolution, liquidation, asset sale or merger transactions), the Palvella board of directors will determine how to treat outstanding awards under its 2019 Plan. This may include, without limitation: (i) the acceleration of the vesting conditions on outstanding awards, (ii) the cancellation and/or forfeiture of outstanding awards, unless exercised prior to the change in control or assumed, substituted or continued buy the surviving entity, or (iii) the cashout or redemption of outstanding awards. The Palvella board of directors need not treat all outstanding awards in an identical manner.
Transferability. Except as otherwise determined by the Palvella board of directors with respect to a particular award, awards under the 2019 Plan are generally not transferable prior to vesting other than by will or by the laws or descent and distribution.
Plan Amendment and Termination. The Palvella board of directors may amend, alter or discontinue the 2019 Plan at any time, provided that no amendment, alteration or discontinuation will be made which would adversely change the terms of an outstanding award, without that participant’s consent. The 2019 Plan will continue in effect until it is terminated by the Palvella board of directors.
Director Compensation
Palvella has omitted the director compensation table because Palvella did not pay any cash compensation or grant any equity or non-equity awards to its non-employee directors during 2023. The aggregate number of option awards outstanding as of December 31, 2023 for each non-employee director who was serving as of such date was as follows: Mr. Jenkins, 22,500 and Mr. Davis 15,000 and Dr. Asokan and Messrs. Morenstein and Wessel, 0 each.
Prior to closing of the Merger, Palvella did not have a formal policy to provide cash or equity compensation to its non-employee directors for their service on the Palvella board of directors or committees of the Palvella board of directors. Following the completion of the Merger, the combined company’s board of directors intends to adopt an annual non-employee director compensation program.
MATTERS BEING SUBMITTED TO A VOTE OF PIERIS STOCKHOLDERS
APPROVAL OF THE ISSUANCE OF SHARES OF PIERIS COMMON STOCK PURSUANT TO THE TERMS OF THE MERGER AGREEMENT AND THE PURCHASE AGREEMENT FOR PURPOSES OF NASDAQ LISTING RULES 5635(A), (B) AND (D)
General
At the Pieris special meeting, Pieris stockholders will be asked to approve (A) the issuance of shares of Pieris common stock pursuant to the terms of (i) the Merger Agreement (as it may be amended from time to time) and (ii) the Purchase Agreement (as it may be amended from time to time) and (B) the change of control of Pieris pursuant to the Merger, in accordance with Nasdaq Listing Rules 5635(a), (b) and (d).
Immediately after the Merger, but without giving effect to the PIPE Financing (as defined below), Pieris securityholders as of immediately prior to the Merger are expected to own approximately 18% of the outstanding shares of capital stock of the combined company and former Palvella securityholders, are expected to own approximately 82% of the outstanding shares of capital stock of the combined company, in each case, on a fully-diluted basis, calculated using the treasury stock method, subject to certain assumptions, including, but not limited to, a valuation for Pieris equal to $21 million, provided, that (a) if Pieris’ net cash as of the closing is greater than $11 million, then Pieris’ valuation will be adjusted upwards on a dollar-for-dollar basis by the difference of: (i) Pieris’ net cash, minus (ii) $11 million, and (b) if Pieris’ net cash is less than $11 million, then Pieris’ valuation will be adjusted downwards on a dollar-for-dollar basis by the difference of: (i) $11 million, minus (ii) Pieris’ net cash, as further described in the Merger Agreement.
The PIPE Investors, have agreed to subscribe for and purchase (in cash or by cancellation of convertible notes) an aggregate of approximately 3,154,241 of shares of Pieris common stock, at a price per share equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, and/or in lieu of Pieris common stock to certain purchasers who so choose, Pre-Funded Warrants to purchase up to 2,592,585 shares of the combined company common stock, at a purchase price per Pre-Funded Warrant equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, minus $0.001, referred to as the “PIPE Financing”, which is expected to close immediately following the closing of the Merger. The gross proceeds from the PIPE Financing are expected to be approximately $78.9 million, consisting of approximately $60.0 million in cash and the conversion of approximately $18.9 million of principal and interest payable under the outstanding convertible notes issued by Palvella, before paying estimated expenses. The closing of the PIPE Financing is conditioned upon the closing of the Merger as well as certain other conditions.
Additionally, immediately following the PIPE Financing, the Pieris securityholders as of immediately prior to the Merger are expected to hold approximately 10.47% of the shares of combined company capital stock, the Palvella securityholders as of immediately prior to the Merger are expected to hold approximately 50.18% of the shares of combined company capital stock, and the PIPE Investors are expected to hold approximately 39.35% of the shares of combined company capital stock, in each case, on a fully diluted basis, subject to certain assumptions.
The terms of reasons for and other aspects of the Merger Agreement, the Merger and the issuance of Pieris common stock in the Merger are described in detail in the other sections in this proxy statement/prospectus. A copy of the Merger Agreement is attached as Annex A to this proxy statement/prospectus.
Stockholder Approval Requirement for Purposes of Nasdaq Listing Rules 5635(a), (b) and (d)
Pursuant to Nasdaq Listing Rule 5635(a), stockholder approval is required prior to the issuance of common stock or other securities convertible into or exercisable for common stock, in connection with the acquisition of the stock or assets of another company, if such securities are not issued in a public offering and (i) the common stock has, or will have upon issuance, voting power equal to or in excess of 20% of the voting power outstanding before the issuance of such securities, or (ii) the number of shares of common stock to be issued is or will be equal to or in excess of 20% of the number of shares of common stock outstanding before the issuance of such securities. Because Pieris expects to issue approximately 6,650,532 shares of Pieris common stock to the stockholders of Palvella in accordance with the terms and subject to the conditions of the Merger Agreement, and approximately 3,154,241 of shares of Pieris common stock to the PIPE investors in accordance with the terms and subject to the conditions of the Purchase Agreement which number exceeds 20% of both the voting power and the number of shares of Pieris common stock outstanding before such issuance, Pieris is seeking the approval of its stockholders for the issuance of shares of Pieris common stock pursuant to the Merger Agreement pursuant to Nasdaq Listing Rule 5635(a).
Additionally, pursuant to Nasdaq Listing Rule 5635(b), stockholder approval is required prior to the issuance of common stock that will result in a change of control of a listed company. Because Pieris expects that the consummation of the Merger, including the issuance of shares of Pieris common stock to the stockholders of Palvella pursuant to the Merger Agreement and pursuant to the Purchase Agreement, will constitute a change of control for purposes of Nasdaq Listing Rule 5635(b), Pieris is seeking the approval of its stockholders for the issuance of shares of Pieris common stock pursuant to both the Merger Agreement and the Purchase Agreement pursuant to Nasdaq Listing Rule 5635(b).
Finally, pursuant to Nasdaq Listing Rule 5635(d), stockholder approval is required for a transaction other than a public offering involving the sale, issuance or potential issuance by an issuer of common stock (or securities convertible into or exercisable for common stock) at a price that is less than the lower of (i) the closing price immediately preceding the signing of the binding agreement or (ii) the average closing price of the common stock for the five trading days immediately preceding the signing of the binding agreement, if the number of shares of common stock (or securities convertible into or exercisable for common stock) to be issued equals to 20% or more of the common stock, or 20% or more of the voting power, outstanding before the issuance. The closing price of Pieris’ common stock on July 22, 2024, which immediately preceded the signing of the Merger Agreement and the Purchase Agreement, was $7.48 per share, and the average closing price of Pieris’ common stock for the five trading days immediately preceding the signing of the Merger Agreement and the Purchase Agreement was $7.468 per share. Because the price at which Pieris will issue the shares of Pieris common stock to the stockholders of Palvella in accordance with the terms and subject to the conditions of the Merger Agreement may be deemed to be lower than $7.468 per share, Pieris is seeking the approval of its stockholders for the issuance of shares of Pieris common stock pursuant to the Merger Agreement pursuant to Nasdaq Listing Rule 5635(d). Additionally, because the price at which Pieris will issue the shares of Pieris common stock to PIPE Investors in accordance with the terms and subject to the conditions of the Purchase Agreement may be deemed to be lower than $7.468 per share, Pieris is seeking the approval of its stockholders for the issuance of shares of Pieris common stock pursuant to the Purchase Agreement pursuant to Nasdaq Listing Rule 5635(d).
Required Vote
The affirmative vote of the holders of a majority of the votes cast on the proposal by the holders of shares of Pieris common stock present or represented at the meeting on such matter is required to approve this Proposal No. 1. Abstentions and broker non-votes, if any, are not considered votes cast and therefore will have no effect on the outcome of Proposal No. 1.
Pursuant to support agreements, each of Pieris’ directors and officers and certain other stockholders have agreed to vote in favor of this Proposal No. 1. As of the date of this proxy statement/prospectus, such stockholders own approximately 19.6% of the outstanding shares of Pieris common stock.
THE PIERIS BOARD OF DIRECTORS RECOMMENDS A VOTE “FOR” THIS PROPOSAL NO. 1.
Unless otherwise instructed, it is the intention of the persons named in the accompanying proxy card to vote shares represented by properly executed proxy cards “FOR” the approval of this Proposal No. 1.
APPROVAL OF AN AMENDMENT TO THE AMENDED AND RESTATED ARTICLES OF INCORPORATION OF PIERIS TO INCREASE THE NUMBER OF AUTHORIZED SHARES OF PIERIS COMMON STOCK
General
Pieris is seeking stockholder approval for a proposal to approve an amendment to its amended and restated articles of incorporation to increase the number of authorized shares of Pieris common stock from 3,750,000 shares to 200,000,000 shares, which is referred to as Proposal No. 2 or the Authorized Shares Increase Proposal.
Pieris’ amended and restated articles of incorporation currently authorizes 3,750,000 shares of common stock, par value $0.001 per share, and 10,000,000 shares of preferred stock, par value $0.001 per share, of which 1,320,240 shares of common stock and 15,618 shares of preferred stock were outstanding as of October 28, 2024, the record date for the Pieris special meeting. The proposed amendment to Pieris’ amended and restated articles of incorporation would not increase or otherwise affect its authorized preferred stock. Pieris common stock is all of a single class, with equal voting, distribution, liquidation and other rights. The additional Pieris common stock to be authorized by adoption of the amendment would have rights identical to Pieris’ currently outstanding common stock.
A copy of the amendment to Pieris’ amended and restated articles of incorporation is attached as Annex I to this proxy statement/prospectus. If Pieris stockholders approve this proposal, subject to the discretion of the Pieris board of directors, Pieris intends to file the amendment to its amended and restated articles of incorporation with the Nevada Secretary of State prior to the Effective Time.
Purpose
As described in greater detail in Proposal No. 1, Pieris will be required to issue shares of its common stock to Palvella stockholders pursuant to the terms of the Merger Agreement and to certain PIPE investors pursuant to the terms of the Purchase Agreement. In addition, if Proposal No. 3 is approved, Pieris will reserve additional shares of its common stock for future issuance under the 2024 Equity Incentive Plan.
The Pieris board of directors believes that as a result of the foregoing, the number of authorized shares of common stock that would be authorized and unissued and not reserved for issuance will not be an adequate number of shares to assure that there will be sufficient shares available for future issuance under the 2024 Equity Incentive Plan. In addition, there will not be sufficient shares available for issuance in connection with possible future acquisitions, equity and equity-based financings, possible future awards under employee benefit plans, and other corporate purposes. Therefore, the Pieris board of directors has determined that it is in the best interests of Pieris and its stockholders to amend its amended and restated articles of incorporation as described herein.
Except for (i) the issuance of shares pursuant to the terms of the Merger Agreement and the Purchase Agreement, which is the subject of Proposal No. 1 and which is described elsewhere in this proxy statement/prospectus and (ii) the issuance of shares that may result from grants under the 2024 Equity Incentive Plan, which is the subject of Proposal No. 3, Pieris does not currently have any plans, proposals or arrangement to issue any of its authorized but unissued shares of common stock.
Possible Effects of the Amendment
If the amendment to Pieris’ amended and restated articles of incorporation is approved, the additional authorized shares would be available for issuance at the discretion of the Pieris board of directors and without further stockholder approval, except as may be required by law or the rules of The Nasdaq Capital Market on which Pieris common stock is listed. The additional shares of authorized common stock would have the same rights and privileges as the shares of Pieris common stock currently issued and outstanding. Holders of Pieris common stock have no preemptive rights.
The issuance of additional shares of common stock may, among other things, have a dilutive effect on earnings per share and on stockholders’ equity and voting rights. Furthermore, future sales of substantial amounts of Pieris common stock, or the perception that these sales might occur, could adversely affect the prevailing market price of Pieris common stock or limit Pieris’ ability to raise additional capital. Stockholders should recognize that, as a result of this proposal, they will own a smaller percentage of shares relative to the total authorized shares of the company than they presently own.
Required Vote
The affirmative vote of the holders of a majority in voting power of the outstanding shares of Pieris common stock and preferred stock, voting as a single class, entitled to vote thereon is required to approve this Authorized Shares Increase Proposal. The Series F Preferred Stock requires the holder thereof, as such, to (i) vote together with the holders of Pieris common stock on the Authorized Shares Increase Proposal and (ii) cast a portion of the 25,000,000 votes of the Series F Preferred Stock, in person or by proxy, FOR the Authorized Shares Increase Proposal in a manner that is proportionate to the percentage of shares of Pieris common stock that are voted FOR the Authorized Shares Increase Proposal as compared with the total shares of Pieris common stock that are voted FOR and AGAINST the Authorized Shares Increase Proposal, excluding any shares of common stock that are not voted “FOR” or “AGAINST” such proposal for any reason, including, without limitation, any abstentions or broker non-votes. For the avoidance of doubt, the Series F Preferred Stock votes on a proportionate basis to the shares voted FOR and AGAINST the Authorized Shares Increase Proposal and does not reflect any shares of common stock that are not voted “FOR” or “AGAINST” such proposal for any reason, including, without limitation, any abstentions or broker non-votes. As an example, if 50.5% of the shares of Pieris common stock are voted FOR the Authorized Shares Increase Proposal, 50.5% of the votes cast by the holder of the Series F Preferred Stock will be cast as votes FOR the Authorized Shares Increase Proposal. Abstentions and broker non-votes, if any, will have the same effect as a vote AGAINST Proposal No. 2, but will not be considered as votes cast AGAINST for the purposes of determining the proportion of Series F Preferred stock votes that will be cast FOR and AGAINST.
No dividends shall be paid on the Series F Preferred Stock, including the CVRs being issued in connection with the closing of the Merger. The Series F Preferred Stock may be redeemed at any time at the option of the Pieris board of directors (in its sole discretion) for $0.01, payable in cash and only out of funds legally available therefor, and will be automatically so redeemed upon approval of the Authorized Shares Increase Proposal.
The holder of the Series F Preferred Stock may not, directly or indirectly, transfer (by sale, assignment, encumbrance, hypothecation, pledge, conveyance in trust, gift, bequest, devise or descent, by operation of law or by any other transfer or disposition of any kind, including to any receivers, creditors, trustees in bankruptcy or other insolvency proceeding) such share to any other person or entity without the prior written consent of the Pieris board of directors (acting in its sole discretion).
Pursuant to support agreements, each of Pieris’ directors and officers and certain other stockholders have agreed to vote in favor of this Proposal No. 2. As of the date of this proxy statement/prospectus, such stockholders own approximately 19.6% of the outstanding shares of Pieris common stock.
THE PIERIS BOARD OF DIRECTORS RECOMMENDS A VOTE “FOR” THIS PROPOSAL NO. 2.
Unless otherwise instructed, it is the intention of the persons named in the accompanying proxy card to vote shares represented by properly executed proxy cards “FOR” the approval of this Proposal No. 2.
APPROVAL OF THE 2024 EQUITY INCENTIVE PLAN
General
At its meeting held on September 12, 2024, the Pieris board of directors adopted a resolution approving the Palvella Therapeutics, Inc. 2024 Equity Incentive Plan, referred to as the “2024 Equity Incentive Plan” or the “Plan”, and directing that the Plan be submitted to the stockholders for their approval at the Pieris special meeting. If approved by Pieris stockholders, the 2024 Equity Incentive Plan will become effective as of the date approved by stockholders, and no further awards will be made under the Pieris Pharmaceuticals, Inc. (i) 2014 Employee, Director and Consultant Equity Incentive Plan, the 2016 Employee, Director and Consultant Equity Incentive Plan, the 2018 Employee, Director and Consultant Equity Incentive Plan, the 2019 Employee, Director and Consultant Equity Incentive Plan, and the 2020 Employee, Director and Consultant Equity Incentive Plan, as amended, or the “Prior Plan”.
If approved, Pieris intends to use the 2024 Equity Incentive Plan to enhance the profitability and value of Pieris for the benefit of its stockholders by enabling Pieris to offer employees, directors, consultants and other service providers of Pieris Company and its affiliates, stock and stock-based incentive awards, to create a means to raise the level of stock ownership by employees, directors, consultants and service providers in order to attract, retain and reward such individuals and strengthen the mutuality of interests between such individuals and Pieris stockholders. The Pieris board of directors believes that an equity incentive plan is an integral part of Pieris’ approach to long-term incentive compensation, focused on stockholder return, and Pieris’ continuing efforts to align stockholder and management interests. Pieris believes that growth in stockholder value depends on, among other things, Pieris’ continued ability to attract and retain non-employee directors, employees, consultants and service providers, in a competitive workplace market, with the experience and capacity to perform at the highest levels.
A copy of the 2024 Equity Incentive Plan is attached as Annex H to this proxy statement/prospectus.
Summary of Plan Provisions
The principal provisions of the 2024 Equity Incentive Plan are summarized below. This summary is qualified in its entirety by reference to the 2024 Equity Incentive Plan document, a copy of which is attached as Annex H and has been filed with the SEC with this proxy statement/prospectus. To the extent the description below differs from the text of the 2024 Equity Incentive Plan, the text of the 2024 Equity Incentive Plan will control.
Administration
The 2024 Equity Incentive Plan vests broad powers in a committee to administer and interpret the Plan. The Pieris board of directors will designate the Pieris Compensation Committee to administer the 2024 Equity Incentive Plan. Except when limited by the terms of the 2024 Equity Incentive Plan, the Pieris Compensation Committee has the authority to, among other things: select the persons to be granted awards; determine the type, size and term of awards; establish performance objectives and conditions for earning awards; and determine whether such performance objectives and conditions have been met. Subject to the requirements of applicable law and Pieris’ governing documents, the Pieris Compensation Committee may delegate its authority, including its authority to grant awards, under the 2024 Equity Incentive Plan to one or more individuals or another committee. The Pieris board of directors may at any time exercise the rights and duties of the Pieris Compensation Committee under the 2024 Equity Incentive Plan; accordingly, references herein to the Pieris Compensation Committee will also include the Pieris board of directors.
The Pieris board of directors may amend, alter or discontinue the 2024 Equity Incentive Plan and the Pieris Compensation Committee may amend any outstanding award at any time; provided, however, that no such amendment or termination may adversely affect awards then outstanding without the holder’s permission. In addition, any amendments seeking to increase the total number of shares reserved for issuance under the 2024 Equity Incentive Plan or modifying the classes of participants eligible to receive awards under the Plan will require ratification by the Pieris stockholders in accordance with applicable law.
Eligibility
Pieris’ employees, directors, consultants, and other service providers, or those of Pieris’ affiliates, are eligible to participate in the 2024 Equity Incentive Plan and may be selected by the Pieris Compensation Committee to receive an award. However, in accordance with applicable tax rules, only Pieris’ employees (and the employees of Pieris’ parent or subsidiary corporations) are eligible to be granted incentive stock options.
Vesting
The Pieris Compensation Committee determines the vesting conditions for awards. A time-based condition requires that the participant be employed or otherwise in the service of Pieris and its affiliates for a certain amount of time in order for the award to vest. A performance-based condition requires that certain performance criteria be achieved in order for the award to vest. Awards may also vest in connection with a participant’s termination of employment or in connection with a Change in Control (as described and defined below).
Shares of Stock Available for Issuance
Subject to certain adjustments, the maximum number of shares of Pieris common stock (referred to in this proposal as “common stock” or “shares”) that may be issued under the 2024 Equity Incentive Plan is the sum of: (i) 3,340,639 shares, minus (ii) the number of shares subject to Prior Plan awards granted between September 12, 2024 and the Effective Date, plus (iii) up to 115,294 additional shares underlying awards outstanding under the Prior Plan that expire, terminate are canceled or forfeited without issuance to the holder thereof of the full number of shares to which the award related, or the “Share Pool”.
If any award granted under the 2024 Equity Incentive Plan or Prior Plan expires, terminates, is canceled or is forfeited, the shares underlying the award will be available for new grants under the 2024 Equity Incentive Plan. Any shares that are withheld for the payment of taxes or in satisfaction of the exercise price an award, will again become available for grant under the 2024 Equity Incentive Plan.
Any shares issued in respect of awards granted in substitution for equity-based awards of an entity acquired by the Company or a subsidiary, or with which the Company or a subsidiary combine, will not reduce the Share Pool.
The maximum aggregate number of shares under the 2024 Equity Incentive Plan that may be issued in respect of incentive stock options is 10,367,799.
In the event of any merger, consolidation, reorganization, recapitalization, stock split, reverse stock split, split up, spin-off, combination of shares, exchange of shares, stock dividend, dividend in kind, or other like change in capital structure (other than ordinary cash dividends) to stockholders of the Company, or other similar corporate event or transaction that affects Pieris common stock, the Pieris Compensation Committee shall make appropriate adjustments in the number and kind of shares authorized by the 2024 Equity Incentive Plan and covered under outstanding awards as it determines appropriate and equitable.
Types of Awards
The 2024 Equity Incentive Plan provides for the grant of the following equity-based and cash-based incentive awards to participants: (i) stock options, (ii) stock appreciation rights, (iii) restricted stock, (iv) restricted stock units, or “RSUs”, and (v) other cash or stock-based awards.
Stock Options. A stock option entitles the holder to purchase from Pieris a stated number of its shares at a specified price for a limited period of time. The Pieris Compensation Committee will specify the number of shares of common stock subject to each option and the exercise price for such option, provided that, in case of an ISO, the exercise price may not be less than the fair market value of a share of common stock on the date the option is granted. However, for an ISO granted to a 10% stockholder, the exercise price shall not be less than 110% of the fair market value of common stock on the date the option is granted.
Generally, options may be exercised in whole or in part through a cash payment. The Pieris Compensation Committee, however, may in its discretion permit payment of the exercise price by other methods. For example, unless the Pieris Compensation Committee decides otherwise, the option holder may pay the exercise price of an option through the surrender of previously acquired shares or may “net settle” an option (which involves the cancellation of a portion of the option to cover the cost of exercising the balance of the option).
All options shall be exercisable in accordance with the terms of the applicable award agreement. The maximum term of an option shall be determined by the Pieris Compensation Committee on the date of grant. In the case of ISOs, the aggregate fair market value (determined as of the date of grant) of common stock with respect to which such ISOs become exercisable for the first time during any calendar year cannot exceed $100,000. ISOs granted in excess of this limitation will be treated as non-qualified stock options.
Stock Appreciation Rights. A stock appreciation right represents the right to receive, upon exercise, any appreciation in a share of common stock over a particular time period. The base price of a stock appreciation right shall not be less than the fair market value of the underlying Pieris common stock on the date the stock appreciation right is granted. The maximum term of a stock appreciation right shall be determined by the Pieris Compensation Committee on the date of grant but shall not exceed 10 years. Stock appreciation right payouts may be made in cash, shares of common stock, or a combination of both, at the Pieris Compensation Committee’s discretion.
Unless otherwise provided in an award agreement or determined by the Pieris Compensation Committee, if a participant’s service with Pieris (or its affiliates) terminates due to death or disability, the participant’s unexercised options and stock appreciation rights may be exercised, to the extent they were exercisable at the time of the participant’s death or disability (or on such accelerated basis as the Pieris Compensation Committee may determine at or after grant), for a period of twelve months from the termination date or until the expiration of the original award term, whichever period is shorter. If a participant’s service with Pieris (or its affiliates) is terminated for cause (as defined in the 2024 Equity Incentive Plan), (i) all unexercised options and stock appreciation rights (whether vested or unvested) shall terminate and be forfeited on the termination date, and (ii) any option or stock appreciation right exercise then in progress will be cancelled.
Unless otherwise provided in an award agreement or determined by the Pieris Compensation Committee, if a participant’s service terminates for any other reason, the participant’s unexercised options and stock appreciation rights may be exercised, to the extent they were exercisable at the time of the participant’s termination (or on such accelerated basis as the Pieris Compensation Committee may determine at or after grant), for a period of ninety days from the termination date or until the expiration of the original option or stock appreciation right term, whichever period is shorter. Unless otherwise provided by the Pieris Compensation Committee, any options and stock appreciation rights that are not exercisable at the time of the termination of service shall terminate and be forfeited on the termination date.
Restricted Stock. A restricted stock award is a grant of shares of common stock that are subject to forfeiture and transfer restrictions during a specified period. The Pieris Compensation Committee will determine the price, if any, to be paid by the participant for each share of restricted stock. If the specified vesting conditions are not attained, the underlying Pieris common stock will be forfeited to Pieris. Conversely, if and when the vesting conditions are satisfied, the restrictions imposed will lapse. During the restriction period, a participant will have the right to vote the shares underlying the restricted stock and receive dividends with respect to restricted stock. However, unless otherwise determined by the Pieris Compensation Committee, any such dividends will be subject to the same vesting conditions as the restricted stock to which they relate. The Pieris Compensation Committee may also require that the dividends be reinvested in additional restricted shares. The Pieris Compensation Committee may issue a certificate or certificates representing the number of shares subject to an award of restricted stock or placed shares in a restricted stock account with the transfer agent during the restriction period. As a condition to any award of restricted stock, a participant may be required to deliver to Pieris a share power, endorsed in blank, relating to the shares of common stock covered an award. Unless otherwise provided in an award agreement or determined by the Pieris Compensation Committee, upon termination of service a participant will forfeit all restricted stock that then remains subject to forfeiture.
Restricted Stock Units. An RSU represents a right to receive, on the achievement of specified vesting conditions, an amount equal to the fair market value of one share of Pieris common stock. An RSU may be settled in shares of Pieris common stock, cash or a combination of both, at the discretion of the Pieris Compensation Committee. Unless otherwise provided in an award agreement or determined by the Pieris Compensation Committee, upon a termination of service, a participant will forfeit all of the participant’s RSUs that then remain subject to forfeiture.
Cash and Other Stock-Based Awards. Cash and other stock-based awards (including awards to receive unrestricted shares of Pieris common stock) may be granted to participants. The Pieris Compensation Committee will determine the terms and conditions of each such award, including, as applicable, the term, any exercise or purchase price, vesting conditions and other terms and conditions.
Change in Control
In the event of a change in control (as defined in the 2024 Equity Incentive Plan), the Pieris Compensation Committee may, on a participant-by-participant basis: (i) cause any or all outstanding awards to become vested and immediately exercisable (as applicable), in whole or in part; (ii) cause any outstanding option or stock appreciation right to become exercisable for a reasonable period in advance of the change in control and, to the extent not exercised prior to that change in control, cancel that option or stock appreciation right upon closing of the change in control; (iii) cancel any unvested award or unvested portion thereof, with or without consideration; (iv) cancel any Award in exchange for a substitute award; (v) redeem any restricted stock or restricted stock unit for cash and/or other substitute consideration with value equal to the fair market value of an unrestricted share on the date of the change in control; (vi) cancel any outstanding option or stock appreciation right with respect to all common stock for which the award remains unexercised in exchange for a cash payment equal to the excess (if any) of the fair market value of the common stock subject to the option or stock appreciation right over the exercise price of the option or stock appreciation right; (vii) take such other action as the Pieris Compensation Committee determines to be appropriate under the circumstances; and/or (viii) in the case of any award subject to Section 409A of the Code, the Pieris Compensation Committee shall only be permitted to use discretion to the extent that such discretion would be consistent with the intended treatment of such award under Section 409A of the Code.
Repricing Prohibited
Neither the Pieris board of directors nor the Pieris Compensation Committee may, without obtaining prior approval of Pieris stockholders: (i) implement any cancellation/re-grant program pursuant to which outstanding options or stock appreciation rights under the 2024 Equity Incentive Plan are cancelled and new options or stock appreciation rights are granted in replacement with a lower exercise per share; (ii) cancel outstanding options or stock appreciation rights under the 2024 Equity Incentive Plan with an exercise price per share in excess of the then current fair market value per share for consideration payable in Pieris equity securities; or (iii) otherwise directly reduce the exercise price in effect for outstanding options or stock appreciation rights under the 2024 Equity Incentive Plan.
Clawback
Awards under the 2024 Equity Incentive Plan (and any shares subject to the awards) will be subject to rescission, cancellation or recoupment, in whole or in part, or other similar action in accordance with the terms of any company clawback or similar policy or any applicable law related to such actions, as may be in effect from time-to-time.
Non-Employee Director Compensation Limits
Under the 2024 Equity Incentive Plan, the aggregate amount of equity and cash compensation payable to a non-employee director with respect to a fiscal year, whether under the 2024 Equity Incentive Plan or otherwise, for services as a non-employee director, shall not exceed $750,000, provided that such amount shall be $1,000,000 for the fiscal year in which the applicable non-employee director is initially elected or appointed to the Pieris board of directors. Such non-employee director limit shall not apply to (i) compensation earned by a non-employee director solely in his or her capacity as chairperson of the Pieris board of directors or lead independent director, (ii) compensation earned with respect to services a non-employee director provides in a capacity other than as a non-employee director, such as an advisor or consultant, and (iii) compensation awarded by the Pieris board to a non-employee director in extraordinary circumstances, in each case provided that the non-employee director receiving such additional compensation does not participate in the decision to award such compensation.
Miscellaneous
Generally, awards granted under the 2024 Equity Incentive Plan may not be transferred, except by will or intestate succession. However, the Pieris Compensation Committee may in its discretion authorize the gratuitous transfer of awards (other than incentive stock options) to family members of the grantee, partnerships owned by such family members, trusts for the benefit of such family members or other similar estate planning vehicles. Awards under the 2024 Equity Incentive Plan will be subject to withholding for applicable taxes, to the extent required by law, and the Pieris Compensation Committee may authorize the withholding of shares subject to an award to satisfy required tax withholding. Awards under the 2024 Equity Incentive Plan are intended to be exempt from or comply with the requirements of Section 409A of the Code and will be interpreted accordingly. Unless the 2024 Equity Incentive Plan is extended with the approval of Pieris stockholders, the 2024 Equity Incentive Plan will expire on September 12, 2034 (ten years after the Pieris board of directors adopted the 2024 Equity Incentive Plan).
Federal Tax Consequences
The federal income tax consequences of the issuance, exercise and/or settlement of awards under the 2024 Equity Incentive Plan are described below. The following information is only a summary and does not address all aspects of taxation that may be relevant to a particular participant in light of his or her personal circumstances. Participants should consult with their own tax advisors with respect to the tax consequences inherent in the ownership and exercise of the awards and the ownership and disposition of any underlying securities. The summary does not address the effects of other federal taxes (including possible “golden parachute” excise taxes) or taxes imposed under state, local or foreign tax laws. Tax laws are subject to change. Generally, all amounts taxable as ordinary income to participants under the 2024 Equity Incentive Plan in respect of awards are expected to be deductible by the Company as compensation at the same time the participant recognizes the ordinary income, subject to the limitations of Section 162(m) of the Code. Under Section 162(m), the Company cannot deduct compensation paid to certain covered employees in excess of $1 million per year.
Nonqualified Stock Options
A participant recognizes no taxable income when a non-qualified stock option is granted. Upon exercise of a non-qualified stock option, a participant will recognize ordinary income equal to the excess of the fair market value of the shares received over the exercise price of the non-qualified stock option. A participant’s tax basis in shares of common stock received upon exercise of a non-qualified stock option will generally be equal to the fair market value of those shares on the exercise date, and the participant’s holding period for such shares will begin at that time. Upon sale of shares of common stock received upon exercise of a non-qualified stock option, the participant will realize short-term or long-term capital gain or loss, depending on the period the shares are held. The amount of such gain or loss will be equal to the difference between the amount realized in connection with the sale of the shares and the participant’s tax basis in such shares.
Incentive Stock Options
A participant recognizes no taxable income when an incentive stock option is granted or exercised. So long as the participant meets the applicable holding period requirements for shares received upon exercise of an incentive stock option (two years from the date of grant and one year from the date of exercise), gain or loss realized by a participant upon sale of the shares received upon exercise will be long-term capital gain or loss, and the Company will not be entitled to a deduction. If, however, the participant disposes of the shares before meeting the applicable holding period requirements, or a “disqualifying disposition”, the participant will then recognize ordinary income. The amount of ordinary income recognized by the participant is limited to the lesser of the gain on such sale and the difference between the fair market value of the shares of common stock on the date of exercise and the option exercise price. Any gain realized in excess of this amount will be treated as short- or long-term capital gain (depending on how long the shares are held). If the option price exceeds the amount realized upon such a disposition, the difference will be short- or long-term capital loss (depending on how long the shares are held). Notwithstanding the above, individuals subject to Alternative Minimum Tax may recognize ordinary income upon exercise of an incentive stock option.
Stock Appreciation Rights
A participant recognizes no taxable income when a stock appreciate right is granted or vests as long as the grant price is at least equal to the fair market value of Pieris common stock on the date of grant and the stock appreciation right has no additional deferral feature. Upon the exercise of a stock appreciate right, a participant will recognize ordinary income equal to the excess of the fair market value of the shares of common stock underlying the stock appreciate right over the grant price of the stock appreciate right. A participant’s tax basis in shares of common stock received upon exercise of a stock appreciate right will generally be equal to the fair market value of those shares on the exercise date, and the participant’s holding period for such shares will begin at that time. Upon sale of shares of common stock received upon exercise of a stock appreciate right, the participant will realize short-term or long-term capital gain or loss, depending on the period the shares are held. The amount of such gain or loss will be equal to the difference between the amount realized in connection with the sale of the shares and the participant’s tax basis in such shares.
Restricted Stock
If a participant receives shares of restricted stock under the 2024 Equity Incentive Plan and does not make the election described in the next paragraph, the participant will recognize no taxable income upon the receipt of the shares. When the forfeiture conditions with respect to the restricted stock lapse, the participant will recognize ordinary income equal to the fair market value of the shares at that time, less any amount paid for the shares. A participant’s tax basis in shares of restricted stock will generally be equal to the income recognized when the forfeiture conditions lapse, and the participant’s holding period for the shares will begin at that time. Upon sale of the shares, the participant will realize short- or long-term gain or loss, depending on how long the shares are held after the forfeiture conditions lapse. Such gain or loss will be equal to the difference between the amount realized upon the sale of the shares and the participant’s tax basis in the shares.
Participants receiving shares of restricted stock may make an election under Section 83(b) of the Code. By making a Section 83(b) election, the participant elects to recognize compensation income when the shares are received rather than at the time the forfeiture conditions lapse. The amount of such compensation income will be equal to the fair market value of the shares upon receipt (valued without regard to the forfeiture conditions and transfer restrictions applicable to the shares), less any amount paid for the shares. By making a Section 83(b) election, the participant will recognize no additional compensation income when the forfeiture conditions lapse. The participant’s tax basis in shares with respect to which a Section 83(b) election is made will generally be equal to the income recognized at grant, and the participant’s holding period for such shares will begin at that time. Upon sale of the shares, the participant will realize short- or long-term capital gain or loss, depending on the period the shares were held. However, if the shares are forfeited, the participant will not be entitled to claim a deduction with respect to any income tax paid upon making the Section 83(b) election. To make a Section 83(b) election, a participant must file an appropriate form of election with the IRS and with his or her employer, each within 30 days after the shares of restricted stock are issued.
Restricted Stock Units
When shares of common stock or cash with respect to RSU awards are delivered to the participant, the value of the shares or cash is then taxable to the participant as ordinary income.
Other Stock-Based Awards
The taxation of other stock-based awards will depend upon the design of such awards.
New Plan Benefits
The benefits that will be awarded or paid under the 2024 Equity Incentive Plan are currently not determinable. The awards granted under the incentive plan will depend on the Pieris board of directors or the Pieris Compensation Committee’s actions and the fair market value of shares at various future dates and the Pieris board of directors or the Pieris Compensation Committee has not determined future awards or who might receive them. As a result, it is not possible to determine the benefits that executive officers and other employees and non-employee directors and consultants will receive if the 2024 Equity Incentive Plan is approved by the stockholders.
On September 12, 2024, the closing market price per share of Pieris’ common stock was $17.03, as reported by the Nasdaq Stock Market.
Required Vote
The affirmative vote of the holders of a majority of the votes cast on the proposal by the holders of all of the shares of Pieris common stock present or represented at the meeting is required to approve this Proposal No. 3. Abstentions and broker non-votes, if any, are not considered votes cast and therefore will have no effect on the outcome of Proposal No. 3.
Pursuant to support agreements, each of Pieris’ directors and officers and certain other stockholders have agreed to vote in favor of this Proposal No. 3. As of the date of this proxy statement/prospectus, such stockholders own approximately 19.6% of the outstanding shares of Pieris common stock.
THE PIERIS BOARD OF DIRECTORS RECOMMENDS A VOTE “FOR” THIS PROPOSAL NO. 3.
Unless otherwise instructed, it is the intention of the persons named in the accompanying proxy card to vote shares represented by properly executed proxy cards “FOR” the approval of this Proposal No. 3.
RATIFICATION OF THE PIERIS BOARD OF DIRECTORS’ APPROVAL OF AN AMENDMENT TO THE AMENDED AND RESTATED ARTICLES OF INCORPORATION OF PIERIS TO CHANGE THE NAME OF THE CORPORATION FROM “PIERIS PHARMACEUTICALS, INC.” TO “PALVELLA THERAPEUTICS, INC.”
Overview
On August 6, 2024, the Pieris board of directors approved and deemed advisable an amendment to Pieris’ amended and restated articles of incorporation, which, if given effect, would change the name of the corporation from “Pieris Pharmaceuticals, Inc.” to “Palvella Therapeutics, Inc.” The change in the name of the corporation would become effective upon the filing of a certificate of amendment with the Nevada Secretary of State. This Proposal No. 4 is conditioned upon and subject to the approval of Proposal Nos. 1 and 2 and the consummation of the Merger. Pursuant to Section 78.390(8) of the NRS, no stockholder approval is required in connection with the amendment to Pieris’ amended and restated articles of incorporation to change the corporation’s name as described above. However, the Pieris board of directors believes it is important to receive the views of its stockholders in connection with the proposed amendment, and seeks ratification of the Pieris board of directors’ approval of the amendment in this Proposal No. 4.
In the judgment of the Pieris board of directors, the change of the name of the corporation is desirable and necessary to appropriately reflect the fact that the business of Palvella will become the principal operating business of the combined company upon the consummation of the Merger.
The complete text of the form of certificate of amendment to the amended and restated articles of incorporation reflecting the foregoing proposed amendment is set forth as Annex J to this proxy statement/prospectus, and stockholders are urged to review the full text of the certificate of amendment together with the foregoing information, which is qualified in its entirety by reference to the full text of the certificate of change.
Required Vote
The affirmative vote of the holders of a majority of the votes cast on the proposal by the holders of shares of Pieris common stock present or represented at the meeting is required to approve this Proposal No. 4. Abstentions and broker non-votes, if any, are not considered votes cast and therefore will have no effect on the outcome of Proposal No. 4.
Pursuant to support agreements, each of Pieris’ directors and officers and certain other stockholders have agreed to vote in favor of this Proposal No. 4. As of the date of this proxy statement/prospectus, such stockholders own approximately 19.6% of the outstanding shares of Pieris common stock.
THE PIERIS BOARD OF DIRECTORS RECOMMENDS A VOTE “FOR” THIS PROPOSAL NO. 4.
Unless otherwise instructed, it is the intention of the persons named in the accompanying proxy card to vote shares represented by properly executed proxy cards “FOR” the approval of this Proposal No. 4.
APPROVAL OF POSSIBLE ADJOURNMENT OF THE PIERIS SPECIAL MEETING
If Pieris fails to receive a sufficient number of votes to approve Proposal Nos. 1 and 2, Pieris may propose to adjourn the Pieris special meeting, for a period of not more than 30 days, for the purpose of soliciting additional proxies to approve Proposal Nos. 1 and 2. Pieris currently does not intend to propose adjournment at the Pieris special meeting if there are sufficient votes to approve Proposal Nos. 1 and 2. Additionally, pursuant to Article 1, Section 3 of Pieris’ amended and restated bylaws, any meeting of stockholders may be adjourned from time to time to reconvene at any other time and to any other place at which a meeting of stockholders may be held under Pieris’ amended and restated bylaws.
Required Vote
The affirmative vote of the holders of a majority of the votes cast on the proposal by the holders of shares of Pieris common stock present or represented at the meeting is required to approve this Proposal No. 5. Abstentions and broker non-votes, if any, are not considered votes cast and therefore will have no effect on the outcome of Proposal No. 5.
THE PIERIS BOARD OF DIRECTORS RECOMMENDS A VOTE “FOR” THIS PROPOSAL NO. 5.
Unless otherwise instructed, it is the intention of the persons named in the accompanying proxy card to vote shares represented by properly executed proxy cards “FOR” the approval of this Proposal No. 5.
Pieris is a biotechnology company that historically discovered and developed Anticalin® protein-based drugs to target validated disease pathways in unique and transformative ways. Proprietary to Pieris, Anticalin proteins are a novel class of therapeutics validated in the clinic and through partnerships with leading pharmaceutical companies, including Pfizer (formerly Seagen) and Boston Pharmaceuticals in IO. Pieris’ clinical pipeline consists of IO bispecifics in partnership with collaborators, including SGN-BB228 (also referred to as PRS-346) targeting CD228 and 4-1BB, and BOS-342 (also referred to as PRS-342) targeting GPC3 and 4-1BB.
On June 21, 2023, Pieris announced that AstraZeneca, one of its former partners, had communicated to Pieris its decision to discontinue and cease dosing in the Phase 2a clinical study of elarekibep based on lung findings from a non-clinical 13-week GLP toxicology study in non-human primates with dry powder inhaler-formulated elarekibep, which impacted Pieris’ inhaled respiratory franchise.
In July 2023, Pieris decided to explore one or more strategic transactions, including reverse mergers, company sale, merger, divestiture of assets, or other strategic transactions, as well as the potential for new or expanded partnerships to advance its proprietary therapeutic programs including, cinrebafusp alfa, PRS-220, and PRS-400. Pieris also announced that it would scale back research, development and clinical projects, including stopping future investments in PRS-220 Phase 2a readiness activities and research and development activities for PRS-400, and opting out of co-development of PRS-344/S095012 in the United States. These decisions were primarily related to recent events that impacted Pieris’ inhaled respiratory franchise, including AstraZeneca's discontinuation of enrollment of the Phase 2a study for elarekibep. In connection with this announcement, the Pieris board of directors approved a reduction in Pieris’ workforce by approximately 70%.
On March 27, 2024, Pieris announced its decision to implement measures that were expected to extend its cash runway into at least 2027, while maximizing its ability to collect potential milestones from its clinical pipeline of partnered drug candidates. These measures included discontinuing all of its research and development efforts and conducting additional reductions in its workforce.
Additionally, on June 28, 2024, Servier provided Pieris with a written notice of termination of the Collaboration Agreement. The Non-Exclusive License Agreement with Servier terminates upon termination of the Collaboration Agreement. The Collaboration Agreement and Non-Exclusive License Agreement will terminate effective December 27, 2024 and Servier will discontinue and cease dosing in the Phase 1 clinical study of S095012. Servier's decision to terminate the agreements was based on a potential safety concern in the S095012 Phase 1 clinical study.
Exploration of Strategic Options
Following an extensive process of evaluating strategic alternatives, including identifying and reviewing potential assets for an acquisition, a potential independent path forward to develop cinrebafusp alfa, a potential acquiror of Pieris, and other reverse merger candidates, Pieris, Merger Sub, and Palvella entered into the Merger Agreement on July 23, 2024. Pursuant to the Merger Agreement and subject to the satisfaction or waiver of the conditions set forth in the Merger Agreement, among other things, Merger Sub will merge with and into Palvella, with Palvella continuing as a wholly owned subsidiary of Pieris and the surviving corporation of the Merger. If the Merger is completed, the business of Palvella will continue as the business of the combined company and the combined company will focus on developing Palvella’s product candidates, and it is anticipated that the combined company will not continue to develop any of Pieris’ legacy product candidates. The Merger Agreement and the transaction contemplated thereby are described in detail elsewhere in this proxy statement/prospectus.
Pieris expects to devote significant time and resources to the complete the Merger. However, there can be no assurances that such activities will result in the completion of the Merger. If the Merger is not completed, Pieris will reconsider its strategic alternatives. Pieris considers one of the following courses of action to be the most likely alternatives if the Merger is not completed:
|
● |
operate its business. As announced on March 27, 2024, Pieris’ current strategy is to maximize its ability to capture the potential milestones from partnered IO assets with Pfizer (formerly Seagen) and Boston Pharmaceuticals by operating a lean business model and reducing operating expenses. If for some reason, the Merger does not close, the Pieris board of directors may conclude it is in the best interest to continue to operate the business as announced, while maintaining its ability to consider other potential strategic options; |
|
● |
dissolve and liquidate its assets. If, for any reason, the Merger does not close, the Pieris board of directors may conclude that it is in the best interest of stockholders to dissolve the company and liquidate its assets. In that event, Pieris would be required to pay all of its debts and contractual obligations, and to set aside certain reserves for potential future claims. There would be no assurances as to the amount or timing of available cash remaining to distribute to stockholders after paying Pieris’ obligations and setting aside funds for reserves; or |
|
● |
pursue another strategic transaction. Pieris may resume the process of evaluating a potential strategic transaction to attempt another strategic transaction like the Merger. |
The order of these alternatives does not indicate any preference towards any particular alternative.
Historical Business and Programs
Prior to the June 2023 discontinuation of the elarekibep clinical trials and the subsequent winddown of Pieris’ proprietary clinical and preclinical programs, Pieris’ business focused on discovering and developing Anticalin-based drugs to target validated disease pathways in unique and transformative ways. Pieris’ pipeline was focused on two pillars: respiratory diseases and IO. Elarekibep (formerly PRS-060/AZD1402) was the lead respiratory Anticalin-based drug candidate that was partnered with AstraZeneca. Pieris’ pipeline also formerly included two other proprietary respiratory programs, PRS-220, a clinical-stage Anticalin protein targeting connective tissue growth factor, or CTGF, and PRS-400, a pre-clinical Anticalin protein targeting Jagged-1 for the treatment of muco-obstructive lung diseases.
Cinrebafusp alfa, formerly Pieris’ lead fully proprietary clinical-stage IO asset, was a Mabcalin® (antibody-Anticalin) bispecific protein comprising a HER2-targeting antibody genetically linked to 4-1BB-targeting Anticalin proteins that was being developed for HER2-expressing cancers. Pieris’ IO pipeline currently includes its partnered IO assets: SGN-BB228 (formerly, PRS-346), a clinical-stage CD228/4-1BB bispecific antibody-Anticalin compound that is currently being advanced in the clinic by Pfizer, and BOS-342 (formerly, PRS-342), a clinical-stage GPC3/4-1BB bispecific Mabcalin compound that Pieris exclusively licensed to Boston Pharmaceuticals and is being advanced in the clinic by Boston Pharmaceuticals, as discussed in more detail below.
SGN-BB228 (PRS-346)
SGN-BB228, a CD228 x 4-1BB bispecific antibody-Anticalin compound, is one of the assets being developed in connection with the Pfizer collaboration. Pieris conducted the initial research and development work after which it handed the program to Pfizer, which is responsible for further advancement and funding of the asset. SGN-BB228 is currently in Phase 1 studies in patients with advanced melanoma and other solid tumors and is one of three programs in the Pfizer collaboration. During the third quarter of 2021, Pieris initiated the second program, and during the fourth quarter of 2022, Pieris initiated the third program within the collaboration with Pfizer. The second and third programs were handed over to Pfizer at the end of 2023, and Pfizer is responsible for continuing development of these two programs. Pieris retains a co-promotion option for one program in the Pfizer collaboration in the United States.
BOS-342 (PRS-342)
BOS-342 is a GPC3 x 4-1BB bispecific Mabcalin compound that Pieris exclusively licensed to Boston Pharmaceuticals. In August 2023, the first patient was dosed in a Boston Pharmaceuticals sponsored Phase 1/2 study of BOS-342 in hepatocellular carcinoma. BOS-342 continues to progress in the clinic.
Historical Product Candidates.
Elarekibep
Elarekibep is former a drug candidate that was being developed to treat asthma and antagonizes IL-4Rα immunoreceptor, thereby inhibiting the downstream action of IL-4 and IL-13, two cytokines known to be key mediators in the inflammatory cascade that drive the pathogenesis of asthma and other inflammatory diseases. Pieris sponsored the Phase 1 single ascending dose/multiple ascending dose studies for elarekibep, after which AstraZeneca took responsibility for further clinical development of elarekibep. As previously disclosed, in June 2023, AstraZeneca informed Pieris of its decision to discontinue and cease dosing in the Phase 2 clinical studies of elarekibep. This decision was based on lung findings from the non-clinical 13-week non-human primates GLP toxicology study, which did not support long-term use and progression to later-stage development. The 13-week non-human primates study included three active dose cohorts. AstraZeneca concluded that there were no clinical observations across any of the doses but that there were respiratory tract pathology findings. These findings included inflammation-mediated lung tissue damage, which did not appear to be dose related. AstraZeneca’s decision was made independent of any data from the Phase 2a study. Following this decision and after review, Pieris decided to not continue further development of elarekibep.
PRS-220
PRS-220 was an orally inhaled Anticalin protein targeting CTGF that was being developed as a local treatment for idiopathic pulmonary fibrosis, or IPF, and other forms of fibrotic lung diseases. CTGF, a matricellular protein, has been demonstrated to be a driver of fibrotic tissue remodeling and the protein has been found over-expressed in lung tissue from patients suffering from IPF. Pieris conducted a Phase 1 study of PRS-220 in healthy volunteers in Australia, which was completed in August 2023. The study was a randomized, two-part, blinded, placebo-controlled study, designed to assess the safety, tolerability, pharmacokinetics, and immunogenicity of single and multiple ascending doses of PRS-220 when administered by oral inhalation to healthy subjects. The clinical study report was finalized at the end of December 2023, and data from the single and multiple ascending doses of PRS-220, when administered by oral inhalation to healthy subjects, demonstrated that PRS-220 was safe and generally well-tolerated by subjects in this study at all administered doses. With the completion of the Phase 1 clinical studies and in connection with its announcement in July 2023, Pieris discontinued further development of the program for strategic and scientific reasons. After completion of the Phase 1 studies, Pieris wound down the program. As of the date of this proxy statement/prospectus, Pieris has not partnered this program and continues to explore potential transactions.
PRS-400
PRS-400 was a fully proprietary Anticalin protein targeting Jagged-1 and was being developed as a local treatment for muco-obstructive lung diseases. Jagged-1 is one of five cell surface ligands interacting with Notch receptors. It has been demonstrated that Jagged-1/Notch signaling drives secretory cell trans-differentiation in the airways and that blocking Jagged-1/Notch signaling reduces secretory cell number, mucin expression and mucus plugging in vivo. In August 2022, Pieris presented preclinical data at the European Respiratory Society International Congress 2022 indicating that candidate molecules inhibit Jagged-1-induced Notch 2 signaling in a dose-dependent manner and also demonstrate that PRS-400 reduces mucin expression ex vivo. Additionally, PRS-400 was found in vivo to reduce mucin gene expression and goblet cells in mice with IL-13-induced airway inflammation. These findings suggested that PRS-400 represents a promising opportunity to address muco-obstructive respiratory diseases locally with an attractive therapeutic index. In connection with Pieris’ July 2023 announcement, Pieris decided to discontinue additional investments into PRS-400 and explore potential partnerships. As of the date of this proxy statement/prospectus, Pieris has not partnered this program and continues to explore potential transactions.
Cinrebafusp alfa
Cinrebafusp is a bispecific Mabcalin compound comprising a HER2-targeting antibody genetically linked to 4-1BB-targeting Anticalin proteins. Cinrebafusp alfa is designed to drive tumor localized T-cell activation through tumor-targeted drug clustering mediated by HER2 expressed on tumor cells. This program was the first 4-1BB bispecific T-cell co-stimulatory agonist to enter clinical development. In July 2022, Pieris received Fast Track Designation from the FDA for cinrebafusp alfa. In August 2022, Pieris announced its decision to cease further enrollment in the two-arm, multicenter, open-label Phase 2 study of cinrebafusp alfa as part of a strategic pipeline prioritization to focus its resources on its respiratory assets. Cinrebafusp alfa has demonstrated clinical benefit in Phase 1 studies, including single agent activity in a monotherapy setting, and in the Phase 2 study in HER2-expressing gastric cancer. In April 2023, clinical data showing an unconfirmed 100% objective response rate and promising emerging durability profile were presented at the American Association of Cancer Research annual meeting. This data provided encouraging evidence of clinical activity for this program. In July 2023, Pieris announced that it was focusing on exploring new or expanded partnerships to advance cinrebafusp alfa. As of the date of this proxy statement/prospectus, Pieris has not partnered this program.
S095012 (PRS-344)
S095012 is a bispecific Mabcalin compound comprising a PD-L1-targeting antibody genetically linked to 4-1BB-targeting Anticalin proteins that was being developed by Servier on a worldwide basis in connection with the Servier collaboration agreement. The first-in-human Phase 1/2 multicenter open-label dose escalation study was designed to determine the safety and preliminary activity of S095012 in patients with advanced and/or metastatic solid tumors. In July 2023, Pieris notified Servier that it was opting out of co-development and commercialization of S095012 in the United States. Servier retained exclusive, even as to Pieris, worldwide rights to the program including the right to advance development and potential commercialization in the United States. On June 28, 2024, Servier communicated its intention to discontinue development of S095012 due to potential safety concerns and to terminate the Servier collaboration agreement effective December 27, 2024.
Pieris is not currently conducting sales and marketing efforts with respect to any of its previous programs.
The process of researching and developing drugs for human use is lengthy, unpredictable and subject to many risks. Historically, Pieris has incurred substantial expenses as it continued to develop its clinical and preclinical drug candidates and programs. Pieris is not currently conducting research and development of any of its previous programs.
Pieris’ commercial success may depend in part on its ability to obtain and maintain exclusivity of its proprietary Anticalin-based technologies through intellectual property protection for its drug candidates, libraries of different protein scaffolds and consensus sequences, the fundamental Anticalin platform technology, including novel therapeutic and diagnostic discoveries, as well as other proprietary know-how and trade secrets, and to operate without infringing on the intellectual property rights of others.
Pieris seeks to protect its exclusive position of Anticalin technologies by, among other means, prosecuting its own international, U.S. and foreign patent applications related to its proprietary technology, inventions and improvements that are important to the development and implementation of its business. Pieris has established intellectual property protection in relation to its Anticalin technologies in key global markets, including in North America, Europe and Asia. Pieris also relies on trade secrets for confidential know-how, which it generally seeks to protect through contractual (for example, confidentiality) agreements with employees and third parties.
Pieris has protected the goodwill of itself and its drug candidates, created through innovation and development, by putting in place trademark registrations of the Pieris and Anticalin marks, as well as several defensive registrations.
Pieris historically has, and may continue to, file and prosecute patent applications and maintain granted patents directed to its key drug candidates in an effort to establish intellectual property positions relating to new compositions of matter for these drug candidates, as well as novel medical applications of these compounds in the treatment, prevention or diagnosis of various indications.
Pieris owns, or is the exclusive licensee of, a patent portfolio consisting of several issued U.S. patents, and their respective counterparts in a number of foreign jurisdictions, including pending patent applications under the Patent Cooperation Treaty, pending U.S. patent applications and corresponding pending patent applications in a number of foreign jurisdictions, as well as pending provisional patent applications, as described in further detail below.
In applicable jurisdictions, such as the United States, and to the extent feasible, Pieris seeks patent term extensions for certain of its issued patents. If Pieris obtains marketing approval for its drug candidates in the United States or certain jurisdictions outside of the United States, it may be eligible for regulatory protection, such as 4 years of data exclusivity and 12 years of market exclusivity for new biological entities in the United States and as mentioned below, up to five years of patent term extension potentially available in the United States, 8 to 11 years of data and marketing exclusivity potentially available for new drugs in the European Union, up to five and a half years of patent extension in Europe (supplemental protection certificate) and eight years of exclusivity, similar to data exclusivity in the United States, potentially available in Japan under its re-examination system. There can be no assurance that Pieris will qualify for any such regulatory exclusivity or that any such exclusivity will prevent competitors from seeking approval solely on the basis of their own studies. See “Government Regulation” below.
Pieris holds issued patents and pending patent applications in the United States and other foreign jurisdictions, which patents or patent applications are related to libraries of different scaffolds and consensus sequences, such as human NGAL and human tear lipocalin, and are expiring or expected to expire between 2024 and 2043, subject to any patent term adjustments and terminal disclaimers in the United States. Pieris also owns a number of patents and patent applications at various stages of prosecution directed towards compositions of matter and in some cases, formulations or methods of use, of its clinical drug candidates. Where possible, Pieris will pursue patent term adjustments in the United States and any applicable foreign jurisdictions.
As a result of its research and licensing agreement, or the TUM License, with TUM, Pieris holds a worldwide exclusive license to multiple issued patents and pending patent applications. These patents and patent applications relate to Anticalin proteins derived from hNGAL lipocalin muteins and/or a library of an hNGAL scaffold of a certain consensus sequence, which patent is expected to expire in 2029, subject to any patent term adjustments or terminal disclaimers in the United States.
Pieris holds a number of issued patents and pending patent applications in the United States and foreign jurisdictions directed to newly-discovered or improved scaffold libraries of lipocalin muteins, compounds derived therefrom (i.e., specific drug candidates) or the uses of such compounds to treat, prevent and mitigate certain diseases and conditions whose pathological development involve the targets of interest as well as to diagnose, prognose and select treatments for the diseases and conditions. It expects that these patents and any patents that may issue from pending applications would likely expire between 2029 and 2043 without taking into account possible patent term adjustments or other extensions. However, any and all of these pending patent applications may not result in issued patents, and not all issued patents may be maintained in force for their entire term. The following is an overview of Pieris’ patent portfolio:
|
Patent Family No. |
Total No. of Patents (Patent Applications) |
Ownership Status |
Type of Patent |
Expiration Date (without patent term adjustments or extensions) |
Jurisdiction |
Technology |
|
PCT/US2023/074629 |
(1) |
Joint |
Utility |
N/A (Expected Sep. 20, 2043) |
PCT |
CD137/CD228 bispecifics |
|
PCT/EP2023/057627 |
(1) |
Owned |
Utility |
N/A (Estimated Mar. 24, 2043) |
PCT |
Production process for Mabcalin proteins |
|
PCT/EP2021/075629 |
(3) |
Owned |
Utility |
N/A (Estimated Sep. 17, 2041) |
US, JP, EU |
Biomarker methods and uses |
|
PCT/EP2020/054821 |
2 (14) |
Owned |
Utility |
Feb. 25, 2040 |
AU, BR, CA, CN, EP, IL, IN, JP, KR, MX, RU, SG, US, VN, ZA |
CD137/GPC3 Bispecific |
|
PCT/EP2016/061071 |
11 (6) |
Owned |
Utility |
May 18, 2036 |
AU, BR, CA, CN, EP, IN, JP, KR, MX, RU, SG, US, ZA |
CD137/GPC3 Bispecific |
|
PCT/EP2010/069028 |
19 (2) |
Owned |
Utility |
Dec. 7, 2030 |
AU, BR, CA, CN, EP, IN, JP, KR, RU, SG, US |
hNGAL protein library |
|
PCT/EP2009/057925 |
8 |
Licensed |
Utility |
June 24, 2029 |
AU, CA, EP, JP, KR, US |
hNGAL protein library |
|
US 7,585,940 |
1 |
Owned |
Utility |
Feb. 4, 2026 (includes PTA) |
US |
hNGAL protein library |
In addition to issued patents, Pieris holds trademarks in the United States for the Pieris and Anticalin marks. Similarly, it holds their respective counterparts, as registered trademarks, in a number of foreign jurisdictions.
Pieris also has relied on unpatented trade secrets and know-how and continuing technological innovation to develop and maintain its competitive advantage. Pieris strives to protect its proprietary information, in part, by using CDAs and/or invention assignment agreements with its collaborators, scientific advisors, employees and consultants. The CDAs are designed to protect its proprietary information and, in the case of agreements requiring invention assignment, to grant it ownership of technologies that are developed through a relationship with a third party. While Pieris considers trade secrets and know-how to be a critical component of its intellectual property, trade secrets and know-how can be difficult to protect. In particular, with respect to its technology platform, Pieris anticipates that these trade secrets and know-how will, over the course of time, be disseminated within the industry through independent development, the publication of journal articles describing the methodology and the movement of personnel skilled in the technology from academic to industry positions and vice versa. As a result, those proprietary trade secrets and know-how may lose their value to Pieris over a period of time, and Pieris may lose any competitive advantage afforded by them, as they become public knowledge.
Strategic Partnerships.
Pfizer
On February 8, 2018, Pieris entered into a license and collaboration agreement, or the Pfizer Collaboration Agreement (formerly the Seagen Collaboration Agreement), and a non-exclusive Anticalin platform technology license agreement, or the Pfizer Platform License (formerly the Seagen Platform License), and together with the Pfizer Collaboration Agreement, the Pfizer Agreements (formerly the Seagen Agreements), with Pfizer (formerly Seagen), pursuant to which they agreed to develop multiple targeted bispecific IO treatments for solid tumors and blood cancers. The programs under the Pfizer Agreements include SGN-BB228 and two pre-clinical programs. Pfizer is solely responsible for further advancement and funding of the SGN-BB228 asset. Under the Pfizer Agreements, Pfizer retains an exclusive license to develop, manufacture, and commercialize collaboration products, and also received a non-exclusive license to certain platform technology intellectual property.
Under the terms of the Pfizer Agreements, the companies pursued multiple Anticalin-antibody fusion proteins during the research phase. The Pfizer Agreements provided Pfizer a base option to select up to three programs for further development. Prior to the initiation of a pivotal trial, Pieris may opt into global co-development and U.S. commercialization of one of the three programs and share in global costs and profits on an equal basis. Pfizer will solely develop, fund and commercialize the other two programs.
Furthermore, under the terms of the Pfizer Agreements, Pfizer paid Pieris a $30 million upfront fee and will pay tiered royalties on net sales between low to high single digits, and up to the low double-digits if Pieris were to exercise its global co-development and U.S. commercialization option. Additionally, as of June 30, 2024, Pieris could receive the following potential milestone payments: up to $759 million in research, development, regulatory and commercial milestones, and up to $450 million in sales milestones.
The term of each of the Pfizer Agreements ends upon the expiration of all of Pfizer’s payment obligations under such Pfizer Agreement. The royalty term The Pfizer Collaboration Agreement may be terminated by Pfizer on a product-by-product basis for convenience beginning 12 months after its effective date upon 90 days’ notice or, for any program where a pivotal study has been initiated, upon 180 days’ notice. Any program may be terminated at Pfizer’s option. If any program is terminated by Pfizer after a pre-defined pre-clinical stage, Pieris will have full rights to continue such program. If any program is terminated by Pfizer prior to such pre-defined pre-clinical stage, Pieris will have the right to continue to develop such program but will be obligated to offer a co-development option to Pfizer for such program. The Pfizer Collaboration Agreement may also be terminated by Pfizer or Pieris for an uncured material breach by the other party upon 90 days’ notice, subject to extension for an additional 90 days if the material breach relates to diligence obligations and subject, in all cases, to dispute resolution procedures. The Pfizer Collaboration Agreement may also be terminated due to the other party’s insolvency and may in certain instances, including for reasons of safety, be terminated on a product-by-product basis. Each party may also terminate the Pfizer Agreements if the other party challenges the validity of any patents licensed under the Pfizer Agreements, subject to certain exceptions. The Pfizer Platform License will terminate upon termination of the Pfizer Collaboration Agreement, whether in its entirety or on a product-by-product basis.
The royalty term is based on a country-by-country basis and product-by-product basis, and for the Platform Agreement it starts at the first commercial sale of such product and ending with respect to such product in such country on the later of (a) ten (10) years thereafter in such country of sale; (b) last to expire regulatory exclusivity relating to such product in such country of sale; or (c) expiration of the last to expire valid claim of certain patent rights covering the import, use, sale or offer for sale product in such country of sale. The expected natural expiration of the current last-to-expire patent under the Platform Agreement is in 2036, not accounting for any potential extensions or adjustments granted by governmental authorities.
On March 24, 2021, Pieris announced that Pfizer made a strategic equity investment in Pieris, and that the companies had entered into a Second Pfizer Amendment (formerly Second Seagen Amendment), in which their existing IO collaboration agreement has been amended relating to joint development and commercial rights for one program in the alliance. Under the Second Pfizer Amendment, Pieris’ option to co-develop and co-commercialize one of three programs in the collaboration was converted to a co-promotion option in the United States, with Pfizer solely responsible for the development and overall commercialization of that program. Pieris will also be entitled to increased royalties from that program in the event that it chooses to exercise the co-promotion option.
In September 2023, Pfizer and Pieris entered into an amendment of the Second Pfizer Amendment that provides Pfizer with collaboration product licenses and no changes to the amounts achievable under the collaboration agreement. The effect of the September 2023 amendment was to transfer responsibility for substantially all activities previously performed by Pieris, including research and development, to Pfizer. Subsequently, in December 2023, the transfer of the two pre-clinical programs was fully approved by the combined joint steering committee.
Boston Pharmaceuticals
On April 24, 2021, Pieris and Boston Pharmaceuticals entered into an exclusive product license agreement, or the BP Agreement, to develop BOS-342, a 4-1BB/GPC3 preclinical IO Mabcalin bispecific protein. Under the terms of the BP Agreement, Boston Pharmaceuticals exclusively licensed worldwide rights to BOS-342.
The term of the BP Agreement ends upon the expiration of all of Boston Pharmaceuticals’ payment obligations thereunder. The BP Agreement may be terminated by Boston Pharmaceuticals in its entirety for convenience beginning nine months after its effective date upon 60 days’ notice or, for any program under the BP Agreement which has received marketing approval, upon 120 days’ notice. If any program is terminated by Boston Pharmaceuticals, Pieris will have full rights to continue such program. The BP Agreement may also be terminated by Boston Pharmaceuticals or Pieris for an uncured material breach by the other party upon 180 days’ notice (60 days in the case of non-payment of undisputed amounts due and payable), subject to extension for an additional 180 days in certain cases and subject, in all cases, to dispute resolution procedures. The Agreement may also be terminated due to the other party’s insolvency. Pieris may also terminate the BP Agreement if Boston Pharmaceuticals challenges the validity of any patents licensed under the BP Agreement, subject to certain exceptions.
Pieris does not have any obligations to assist in the research and development efforts of Boston Pharmaceuticals under the BP Agreement. However, Pieris had an obligation to fund up to $4.0 million in costs, including out-of-pocket costs incurred by Boston Pharmaceuticals, in connection with the manufacture of products under the BP Agreement. The arrangement with Boston Pharmaceuticals provides for the transfer of the following: (i) exclusive license of BOS-342, (ii) non-exclusive Pieris platform license, (iii) initial know-how, (iv) product cell line license, and (v) materials (as each such term is defined under the BP Agreement).
In August 2023, the first patient was dosed in a Boston Pharmaceuticals sponsored phase 1/2 study of BOS-342 in hepatocellular carcinoma (HCC), for which Pieris received a $2.5 million milestone payment. As of June 30, 2024, Pieris could receive the following potential milestone payments from Boston Pharmaceuticals: up to $85 million in research, development, regulatory and commercial milestones, and up to $265 million in sales milestones, as well as tiered royalties from mid-single digits to low-double digits on potential sales of BOS-342.
Servier
On January 4, 2017, Pieris entered into the Servier Collaboration Agreement and a non-exclusive Anticalin platform license agreement with Servier, or the Servier Platform License, collectively referred to as the Servier Agreements. Pursuant to the terms of the Servier Agreements, Pieris, along with Servier, initially pursued five bispecific therapeutic programs, one of which was the S095012 program.
In July 2023, Pieris notified Servier that it was opting out of co-development and commercialization of S095012 in the United States. Servier retained exclusive, even as to Pieris, worldwide rights to the program including the right to advance development and potential commercialization in the United States. As a result of Pieris’ election to opt out, Pieris was entitled to increased royalty rates and potential royalties and milestones, if any, for S095012. On June 28, 2024, Servier notified Pieris that it was discontinuing the Phase 1/2 clinical study of S095012 due to potential safety concerns and that it was terminating the Servier Agreements effective December 27, 2024. As of the date of this proxy statement/prospectus, Servier has terminated all of the five bispecific therapeutic programs under the collaboration.
Pieris may continue to explore other partnerships or arrangements to license out its legacy assets and intellectual property, including PRS-400 and PRS-220. Pieris continues to explore potential transactions for PRS-400 and PRS-220. Management has determined that there is no value associated with the CVRs as the likelihood of any payments received in connection with Pieris’ legacy assets is remote.
Government Regulation and Product Approvals
Government authorities in the United States at the federal, state and local level, and in other countries and jurisdictions, such as the European Union, extensively regulate, among other things, the research, development, testing, manufacture, pricing, quality control, approval, packaging, storage, recordkeeping, labeling, advertising, promotion, distribution, marketing, post-approval monitoring and reporting, and import and export of biopharmaceutical products. The processes for obtaining marketing approvals in the United States and in foreign countries and jurisdictions, along with compliance with applicable statutes and regulations and other regulatory authorities, require the expenditure of substantial time and financial resources. See the section titled “Palvella’s Business – Government Regulation” in this proxy statement/prospectus for further discussion.
Palvella is a clinical-stage biopharmaceutical company whose vision is to become the leading rare disease biopharmaceutical company focused on developing and, if approved, commercializing novel therapies to treat patients suffering from serious, rare genetic skin diseases for which there are no FDA-approved therapies. Palvella intends to leverage its versatile QTORIN platform to treat these patients. QTORIN is designed to generate new therapies that penetrate the deep layers of the skin to locally treat a broad spectrum of rare genetic skin diseases. Palvella’s lead product candidate, QTORIN rapamycin, is in clinical development for two of these diseases: microcystic LM, and cutaneous venous malformations. QTORIN rapamycin contains the active pharmaceutical ingredient rapamycin, also known as sirolimus, which is an inhibitor of mTOR, a kinase that plays a key role in cell growth and proliferation.
Palvella currently has one ongoing clinical trial and one clinical trial planned to start in the fourth quarter of 2024. Palvella’s ongoing trial, SELVA, is a Phase 3 Baseline-Controlled Study Evaluating the Safety and Efficacy of QTORIN rapamycin in the Treatment of Microcystic LM. Palvella previously announced topline Phase 2 clinical trial results from the multi-center, open-label study of 12 subjects receiving QTORIN™ rapamycin once-daily for 12-weeks. The Phase 2 clinical trial featured multiple pre-specified efficacy assessments, including clinician and patient global impression assessments as well as assessments of individual clinical manifestations that are important disease burdens for individuals living with microcystic LMs. All participants in the Phase 2 clinical trial demonstrated improvements on the Clinician Global Impression of Change scale, with all participants in the study rated as either "Much Improved" (n=7, 58%) or "Very Much Improved" (n=5, 42%) after 12-weeks of treatment compared to the pre-treatment baseline period. Palvella expects to report top-line data for the Phase 3 study in approximately 40 participants with microcystic LM in the first quarter of 2026.
Microcystic LM is a serious, chronically debilitating, and lifelong disease of the lymphatic system characterized by lymphorrhea and acute cellulitis. It is estimated that there are more than 30,000 diagnosed patients in the United States with microcystic LM. The specific pathophysiology of microcystic LM is primarily the result of somatic activating mutations in PIK3CA which result in increased activation of the PI3K/mTOR pathway and subsequent lymphatic hyperplasia. Because microcystic LM has a well-understood pathophysiology and a well-defined disease course, Palvella believes an appropriate clinical study for this rare disease is a baseline-controlled Phase 3 study using clinician assessments.
Palvella has received Breakthrough Therapy Designation, Fast Track Designation, and Orphan Drug Designation from the FDA for QTORIN rapamycin for the treatment of microcystic LM. Palvella has also received Fast Track Designation from the FDA for the treatment of venous malformations.
There are no FDA-approved therapies currently indicated for either microcystic LM or cutaneous venous malformations. If approved for the treatment of microcystic LM or cutaneous venous malformations, Palvella believes QTORIN rapamycin has the potential to become the standard of care for these diseases.
Palvella also has a planned study for cutaneous venous malformations, a Phase 2 Baseline-Controlled Study Evaluating the Safety and Efficacy of QTORIN rapamycin for the Treatment of Cutaneous Venous Malformations expected to start in the fourth quarter of 2024. Cutaneous venous malformations are a serious disease with a high unmet need characterized by dysregulated growth of malformed veins impacting the skin, causing functional impairment and deformity. It is estimated that there are more than 75,000 diagnosed patients in the United States with cutaneous venous malformations. Palvella is conducting a Phase 2 baseline-controlled clinical trial in approximately 15 participants in this patient population and expect to report top-line data in the fourth quarter of 2025.
Palvella also has additional preclinical research programs based on Palvella’s QTORIN platform for the treatment of serious, rare genetic skin diseases for which Palvella believes there are significant unmet needs. As Palvella plans to expand its pipeline into new rare skin diseases, it plans to generate new product candidates with its QTORIN platform. Despite Palvella’s intentions with respect to its QTORIN platform, Palvella’s business carries substantial risks. The QTORIN platform is novel and has only generated one program to date, QTORIN rapamycin, and clinical evidence to support this candidate is preliminary and limited at this time.
Palvella has multiple patents and patent applications directed to anhydrous gel formulations of rapamycin, including QTORIN rapamycin, and the use of such anhydrous gel formulations for the treatment certain skin disorders, including microcystic LM and venous malformations. Palvella’s issued U.S. patents with claims directed to certain anhydrous gel formulations containing rapamycin and methods of treatment expire in 2038.
Palvella has assembled a management team with extensive experience in building and operating innovative biopharmaceutical companies across a variety of therapeutic areas and technologies. Palvella’s management team is led by Wesley H. Kaupinen, Palvella’s founder, President and Chief Executive Officer, who previously served as Senior Vice President of Corporate Development and Commercialization at Insmed, Inc. and has 20 years of broad leadership and management experience in the life sciences industry. Palvella’s executive team is composed of individuals with deep experience in rare disease drug development including Kathleen Goin, Palvella’s Chief Operating Officer, who previously served in leadership roles at Trevena, Inc and Endo Pharmaceuticals, Inc and Jeffrey Martini Ph.D., Palvella’s Chief Scientific Officer, who previously served in leadership roles at Marinus Pharmaceuticals, Teva Pharmaceuticals, and Cephalon, Inc. Members of Palvella’s management team have previously led various discovery, development, manufacturing and commercialization programs within the rare disease and dermatology fields, including ARIKAYCE, Differin, Diflucan, VALCHLOR, ZTALMY, and Vectical. Further, Palvella is supported by a leading group of investors including, among others, Adams Street Partners, BVF Partners L.P., CAM Capital, Petrichor, and Samsara BioCapital.
Palvella has assembled a medical and scientific advisory board, or MSAB, comprised of leading scientists and academic clinicians who have backgrounds in various disciplines, including genetics, rare skin diseases, and novel therapeutic discovery and development. Palvella partners with its MSAB for idea generation into high unmet need diseases in need of novel therapeutic development, scientific and clinical input on clinical study design including endpoints, and generation of new product candidate ideas.
Palvella’s Vision and Approach
Palvella’s vision, supported by its mission of serving patients, is to become the leading rare disease biopharmaceutical company focused on developing and, if approved, commercializing novel therapies for serious, rare genetic skin diseases, for which there are no FDA-approved therapies. Palvella envisions a future treatment paradigm in which individuals suffering from serious, rare genetic skin diseases, and the physicians treating those diseases, have significantly improved treatment options which address the underlying causes of those diseases. The core components of Palvella’s approach include the following:
|
● |
supported by its pipeline, build upon its experience in serious, rare genetic skin diseases through indication expansion and the generation of new product candidates. Palvella believes serious, rare genetic skin diseases represent a substantial opportunity to develop and, if approved, commercialize first-in-disease therapies. More than 98% of the reported 597 rare skin diseases do not have a therapy approved by the FDA. Palvella’s goal is to select diseases caused by genetic mutations with a well-understood etiology, pathophysiology and a strong rationale for a specific pathway intervention, many of which have a debilitating, lifelong impact on individual lives. In addition to exploring other selected diseases for which QTORIN rapamycin could provide a viable therapy, Palvella is investigating several new product candidates for additional rare genetic skin diseases that it selects; |
|
● |
maximize the potential of the QTORIN platform across a wide range of molecules. To date, Palvella has developed QTORIN rapamycin which Palvella believes has broad clinical potential in several serious, rare genetic skin diseases without FDA-approved therapies. Palvella intends to further leverage its scalable QTORIN platform to generate additional product candidates that target the known causes of serious, rare genetic skin diseases for which there are no FDA approved therapies; and |
|
● |
forge meaningful patient and physician collaborations. A key element of Palvella’s approach is to take a rigorous, systematic approach to understanding the disease of the patient populations that Palvella is addressing. A foundational pillar of this approach is to forge and maintain meaningful collaborations with physicians and disease advocacy organizations. Through this engagement, valuable learnings inform the selection and development of efficacy endpoints and what constitutes clinical meaningfulness and acceptable risk-benefit. Palvella believes these learnings significantly inform its product development approach, which may contribute to the regulatory acceptability of its product candidates. |
To achieve Palvella’s vision, the key elements of its strategy include:
|
● |
successfully develop and, if approved, commercialize QTORIN rapamycin for the treatment of microcystic LM, cutaneous venous malformations and other rare genetic skin diseases. QTORIN rapamycin is in clinical development for microcystic LM and cutaneous venous malformations. There are no FDA-approved therapies for these specific indications, and, if approved, Palvella believes QTORIN rapamycin has the potential to become the standard of care. Palvella expects to report top-line data from its Phase 3 SELVA trial in microcystic LM in the first quarter of 2026. Assuming favorable results from SELVA, Palvella plans to request FDA agreement to begin a rolling submission of an NDA in the second half of 2026. Palvella plans to initiate its Phase 2 trial in patients with cutaneous venous malformations in the fourth quarter of 2024 and expects to report top-line data in the fourth quarter of 2025. Given there is a growing body of real-world evidence that rapamycin has the potential to treat a broad number of cutaneous diseases, Palvella plans to evaluate QTORIN rapamycin in other cutaneous indications; |
|
● |
build an independent commercial organization to commercialize, if approved, Palvella’s genetic skin diseases therapies in the United States. If approved for the treatment of microcystic LM and/or cutaneous venous malformations in the United States, Palvella intends to independently commercialize QTORIN rapamycin by building out a focused specialty sales force that will target vascular anomaly centers and dermatologists who cover the majority of patients being treated. Palvella expects this will provide it with a significant competitive advantage in addition to providing it future operational leverage. Outside of the United States, Palvella may consider building its own commercial infrastructure or out license where appropriate, elect to utilize strategic collaborators, distributors or other partners to assist in the commercialization of its products candidates, if approved; |
|
● |
evaluate the potential of the QTORIN platform to treat additional rare genetic skin diseases. As noted above, more than 98% of the reported 597 rare skin diseases do not have a therapy approved by the FDA. Palvella has identified several rare genetic skin diseases where there are no FDA approved therapies, and the cause of pathophysiology or genetic mutation is known. The QTORIN platform provides the opportunity to target delivery of active pharmaceutical ingredients (APIs) with a very diverse chemical structure and size to the dermis and epidermis with limited systemic absorption. The incorporation and evaluation of alternative APIs in the QTORIN platform would expand the possible range of indications for which novel, life-changing therapies may be created. This provides the opportunity for expansion of novel QTORIN products to address the needs of hundreds of thousands of patients with genetic skin diseases who have no FDA approved therapies for their disease; and |
|
● |
continue to establish barriers to entry through intellectual property and regulatory exclusivities. Palvella has significant intellectual property rights in its current development programs, including issued patents in the U.S. directed to QTORIN rapamycin and methods of using such anhydrous gel formulations of Exrapamycin. Palvella owns issued patents in the US, as well as Australia, China, Israel and Japan and pending applications in the US, Europe and Japan directed to anhydrous gel formulations of rapamycin and methods of using the same to treat certain skin disorders, including microcystic LM and venous malformations that naturally expire in 2038. Palvella also owns issued US patents and a pending US application that encompass anhydrous gel formulations of mTOR inhibitors, including rapamycin, and methods of using the same to treat skin disorders including microcystic LM and venous malformations that naturally expire as early as 2038. Palvella also owns pending applications in the US and other major markets directed to the use of QTORIN rapamycin for the treatment of microcystic LM that, if issued, would expire in 2042. Any of Palvella’s product candidates that receive regulatory approval may also potentially be protected by regulatory exclusivity, such as through the exclusive marketing period provided from Orphan Drug Designation and/or drugs approved based on new clinical investigations (other than bioavailability studies) that are conducted by the sponsor that are essential to approval. Palvella expects to continue to expand its intellectual property portfolio as it continues to develop its product candidates. |
Palvella’s research team developed and designed QTORIN by testing over 80 combinations of excipients. QTORIN is a patented and versatile platform designed to generate potential new therapies that penetrate the deep layers of the skin to locally treat a broad spectrum of rare skin diseases. Identification and development of novel QTORIN products begins with the Palvella team identifying serious, rare genetic skin diseases with no FDA-approved therapies which clinically have a localized presentation and therefore could be suitable for targeted, topical drug intervention. Once target diseases are selected and key biological pathways that can be causative drivers of that specific disease have been identified, a rigorous formulation development process is undertaken with product development objectives of achieving (i) high payloads of the active pharmaceutical ingredient in the anhydrous gel, and (ii) penetration and distribution of pharmacologically active quantities of the active ingredient to the site of pathophysiology, including the dermis, while achieving minimal to no systemic absorption of the active ingredient.
The QTORIN platform is composed of an anhydrous gel comprising excipients, or inactive substances, that serve as the vehicle or medium for a drug or other active substance, selected in what Palvella believes is an optimized ratio in order to achieve therapeutic levels of drug delivered to the site of origin of the disease, often within the deepest regions of the skin. Palvella’s QTORIN product candidates have been developed to accommodate the cargo at high concentrations in order to drive sufficient drug to its target deep in the epidermis and dermis. Inclusion of agents like penetration enhancers have been avoided in order to minimize systemic absorption. The final formulation of the drug product is designed to be less than 100% of the maximum solubility to avoid physical instability due to factors such as temperature change.
Palvella believes its QTORIN platform provides the following advantages:
|
● |
reproducible platform across multiple molecules. In a preclinical study conducted by Palvella, the QTORIN platform has demonstrated compatibility with more than 15 high potential pharmacologic agents. As a result of such compatibility, Palvella believes it will be able to generate new product candidates and reproduce the formulations results from QTORIN rapamycin while minimizing the challenges and timelines typically associated with formulation development activities; |
|
● |
versatility across a range of indications. We believe QTORIN’s ability to accommodate a wide range of therapeutic cargoes enables versatility in the targets for molecular intervention, thereby potentially being able to develop novel QTORIN therapies across a diverse set of serious, rare genetic skin diseases; |
|
● |
tailored penetration and distribution of molecules to the site of where the disease originates. In order to engage the target, a product candidate must deliver therapeutic concentrations of drug substances to the site of the pathophysiology, which is often rendered challenging due to certain agents, such as rapamycin, possessing high molecular weights or structures that prevent skin penetration. By optimizing the individual QTORIN excipient ratio for each therapeutic molecule, Palvella’s platform is designed to deliver therapeutic agents to the specific site of disease origin; |
|
● |
delivery of therapeutic agents designed to minimize systemic exposure. Well-accepted mechanisms of action of rapamycin or other therapeutic agents represent potential therapies for rare genetic skin diseases. However, the adverse event profile of those agents through systemic exposure poses significant barriers to patient adoption. As observed in all completed clinical trials with QTORIN rapamycin to date, Palvella’s QTORIN product candidates are designed for targeted, localized delivery of therapeutic agents to pathogenic tissue of interest while minimizing systemic absorption and thereby reduce the risk of unwanted adverse events associated with systemic therapy; |
|
● |
enhanced stability at ambient temperatures. Palvella has data to support long-term stability of QTORIN rapamycin at room temperature, which Palvella believes is an important feature for patient acceptability, particularly for a chronic dosing regimen; and |
|
● |
scalable QTORIN manufacturing. Palvella intends to scale up QTORIN manufacturing in the future. Under cGMP conditions, Palvella believes it has overcome many of the challenges associated with manufacturing QTORIN rapamycin, including solubility, stability, and scalability. Based on Palvella’s work to date, Palvella believes that it can successfully scale up QTORIN rapamycin and future QTORIN product candidates to meet its future development and commercial needs. |
Despite Palvella’s intentions with respect to its QTORIN platform, therapeutic development with a novel platform carries substantial risks. The QTORIN platform is novel and has only generated one program to date, QTORIN rapamycin, and clinical evidence to support this candidate is preliminary and limited at this time. In addition, as a novel platform, the QTORIN platform may never result in a product candidate that receives regulatory approval.
The Role of mTOR in Cutaneous Disorders
The PI3K/mTOR family of kinases play vital roles in cellular function by regulating proliferation, growth and survival. Dysregulation of the PI3K/mTOR pathway is associated with several cutaneous disorders, including serious, rare genetic skin diseases. Often these pathological diseases are characterized by hyperproliferation and tumorigenesis, defective keratinocyte differentiation, senescence-like growth arrest and resistance to apoptosis.
____________________________
ANG: angiopoietin, VEGF: vascular endothelial growth factor, VEGFR: vascular endothelial growth factor receptor, PIK3CA: phosphoinositide 3-kinase, mTOR
Over the past two decades, several studies have been published on the use of oral rapamycin in cutaneous diseases, including genetic skin diseases, due to rapamycin’s well-documented anti-proliferative, anti-angiogenic, and immunosuppressive properties.
Rapamycin Challenges and Palvella’s Novel Product Candidate, QTORIN Rapamycin
Rapamycin Has Demonstrated Activity in Rare Genetic Skin Diseases
A systematic review by Swarbrick and colleagues found over 200 publications demonstrating the broad potential of rapamycin in cutaneous diseases. This publication built upon an early publication by Teng and colleagues in May 2015 which highlighted the substantial promise of mTOR inhibitors, including rapamycin, in a number of difficult to treat dermatologic diseases while advocating for targeted, topical approaches suited to improve tolerability. Despite the preliminary evidence of clinical benefit in many cutaneous diseases, rapamycin’s use in cutaneous diseases, including rare genetic skin diseases, remains limited, primarily due to the undesirable toxicity profile of oral rapamycin, including immunosuppression, for cutaneous diseases and the limited biodistribution of oral rapamycin to the dermis.
Barriers to Oral Rapamycin’s Use in Cutaneous Diseases
Rapamycin is FDA approved as an oral product for the prevention of organ transplant rejection and for the treatment of lymphangioleiomyomatosis. It has been well-established that inhibition of mTOR by rapamycin has the potential to have broad application in dermatology, but there are several challenges which have limited its use:
|
● |
systemic exposure to oral rapamycin is associated with severe and unwanted toxicities. In addition to its immunosuppressive nature, the most common (≥30%) adverse reactions observed in clinical studies for organ rejection prophylaxis include: peripheral edema, hypertriglyceridemia, hypertension, hypercholesterolemia, increased creatinine and constipation, along with several other intolerable adverse reactions. Additionally, because rapamycin suppresses immune function, chronic systemic use may cause serious side effects such as thrombocytopenia and hyperlipidemia, nephrotoxicity and altered insulin sensitivity; |
|
● |
oral rapamycin has low biodistribution to the skin which limits the clinical activity of the systemic mode of administration against genetic skin diseases; |
|
● |
rapamycin is a challenging molecule to formulate and deliver topically as its high molecular weight, poor solubility and chemical instability restrict penetration into the deeper layers of the skin, including the dermis, where many manifestations of rare genetic skin diseases originate. Rapamycin has a molecular weight of 914 daltons, almost two-fold higher than the generally accepted rule that the molecular weight of a compound should be under 500 daltons to penetrate the skin; |
|
● |
the listed drug for the Section 505(b)(2) NDA intends to reference is oral RAPAMUNE (Reference sponsor: Pfizer). Palvella intends to rely on the FDA’s conclusions of safety from its review of the reference sponsor’s studies, along with Palvella generated data, to support approval. These include nonclinical mammalian systemic toxicity, gene toxicology, carcinogenicity, and safety pharmacology studies. Clinical studies include systemic safety, PK, and PD studies. |
Palvella’s Novel Product Candidate: QTORIN rapamycin
Palvella has developed QTORIN rapamycin, a novel, 3.9% anhydrous topical gel formulation containing rapamycin, for the treatment of microcystic LM and cutaneous venous malformations. If approved, Palvella believes QTORIN rapamycin has the potential to become the standard of care in each of these diseases.
Palvella believes it has optimized QTORIN rapamycin to deliver therapeutically active levels of rapamycin to the deep layers of the skin, including the dermis, with minimal systemic absorption below immunosuppressive levels. Palvella estimates, based on preclinical studies, that QTORIN rapamycin will deliver concentrations of rapamycin — approximately 1000-fold higher than oral rapamycin — to the cutaneous tissue with minimal systemic absorption. During the discovery and development of QTORIN rapamycin, 25 excipients were evaluated in more than 80 different combinations. QTORIN rapamycin was designed to utilize a combination of excipients that Palvella believes maximized solubility while maintaining chemical stability. QTORIN rapamycin has completed formulation optimization and in vitro penetration assays and has demonstrated low systemic absorption in Palvella’s human clinical trials to date.
QTORIN Rapamycin for the Treatment of Microcystic LM
|
Objective |
● Palvella is developing QTORIN rapamycin for the treatment of microcystic LM ● There are no FDA-approved therapies; Palvella is developing the first targeted therapy for microcystic LM |
|
Palvella’s Targeted Approach |
● Utilizing QTORIN to confer site-directed delivery of rapamycin to the dermis where microcystic LM originates |
|
Program Status; Upcoming Milestones |
● Palvella completed its Phase 2 clinical trial in the 4th quarter of 2022 ● Palvella initiated its Phase 3 clinical trial in the 3rd quarter of 2024. ● Palvella expects to report top-line data in first quarter of 2026 |
|
Disease Burden |
● Serious, rare and chronic genetic disease characterized by lymphorrhea and acute cellulitis ● Usually present at birth; progresses throughout life ● Localized masses of malformed lymphatic vessels protrude through the skin barrier |
|
Genetic Basis and Molecular Pathways |
● Somatic gain of function mutation primarily in PIK3CA leads to hyperactivated PI3K/mTOR signaling |
|
Scientific Rationale |
● mTOR is hyperactivated in microcystic LM ● Rapamycin directly inhibits overactivated mTOR activity and decreases lymphangiogenesis |
|
Market Dynamics |
● Estimated prevalence: > 30,000 diagnosed patients in US ● Based on >30,000 diagnosed patients in the U.S., Palvella believes the estimated TAM opportunity on an annualized basis is greater than $1 billion |
|
Intellectual Property; Regulatory Designations* |
● Palvella holds U.S. patents and applications in the U.S. and major foreign markets with claims directed to anhydrous gel formulations of rapamycin and methods of use for treating microcystic LM, expiring in 2038 and, for certain applications, if issued, as late as 2042 ● FDA Fast Track Designation ● FDA Orphan Drug Designation ● FDA Breakthrough Therapy Designation ● European Medicines Agency Orphan Drug Designation |
_________________________
* Fast Track or Orphan Drug Designation may not result in a faster development process, review or approval as compared to conventional FDA approval procedures. Please see “Special FDA Expedited Review and Approval Programs” herein for more information.
Disease Overview
Microcystic LM is a serious, rare genetic disease of the lymphatic system characterized by lymphorrhea, which is the persistent discharge of internal lymph fluid from disrupted lymphatic vessels, and acute cellulitis, or a bacterial infection of the skin underlying tissues (Figure 1). Microcystic LM primarily arise from somatic activating mutations in PIK3CA resulting in hyperactivation of the PI3K/mTOR signaling pathway. Microcystic LM is one of three morphologic types of LMs based on the size of the individual cysts (as opposed to the overall size of the LM): macrocystic (>2cm), microcystic (<2cm) and combined. Microcystic LM present at birth and is the result of congenital abnormalities of the lymphatic system thought to originate during the embryologic development of lymphatic vessels. Microcystic LM leads to malformed lymphatic vasculature, persistent infiltration of lymph fluid into soft tissues, and locally invasive masses with pathologic sequelae.
Due to the chronic lymphorrhea, cellulitis and other symptoms, microcystic LM is associated with a high degree of morbidity and has a significant impact on daily life. Microcystic LM can be located on any region of the body but is most commonly found in high areas of lymphatic vessels, including the trunk, head and neck. The malformations connect to the epidermis in the form of vesicles, papules, and plaques which can leak at the surface. Infections of malformations can occur and may lead to cellulitis of surrounding tissues or severe, life-threatening infections. The natural history of microcystic LM is progressive, with symptoms generally worsening during life, including increases in the number size of cysts that lead to complications, and morbidity.
Microcystic LMs arise due to post zygotic mutations during early embryonic development, are usually present at birth, and are persistent and progressive throughout life. Patients are usually diagnosed at a young age by pediatric dermatologists or pediatric hematologists and are managed by multi-disciplinary teams. Due to the genetic nature of the disease, microcystic LM is programmed to be on the skin and do not spontaneously regress. In a 2017 review of 153 patients over a 34-year period to determine if LM sub-types had spontaneous regression, spontaneous regression was observed in 0% of patients with microcystic LM (n=28;TABLE 1).
FIGURE 1. Example of Microcystic LM
Despite the high rate of morbidity and life-threatening cellulitis associated with microcystic LM, there are currently no FDA-approved medications for this disease. Currently available treatment options include surgery, sclerotherapy with bleomycin or other sclerotic agents, laser, and cryotherapy, which are invasive, can induce further inflammation and result in high recurrence rates. Surgical resection remains challenging and ineffective due to the infiltrative, diffuse nature of microcystic LM. In addition, due to underlying associated somatic mutation, it is difficult to achieve accurate and clear surgical margins, resulting in high recurrence rates post resection. The high unmet need and drawbacks associated with surgical approaches have spurred the search for treatment alternatives that target the underlying pathological mechanisms of this disorder.
Microcystic LM Does Not Have Spontaneous Regression
Due to the genetic nature of the disease, microcystic LMs are persistent and progressive throughout life, without spontaneous regression. A review article, which followed subjects over a 34-year observation period, found no spontaneous regression throughout that time among 28 participants with microcystic LM (Table 1).
TABLE 1: Clinical Characteristics of Spontaneous Regression of the LM Patients

Because microcystic LM does not have spontaneous regression, a baseline-controlled study, in which subjects’ status on therapy is compared with the status before therapy, can be suitable for this disease because improvement does not reflect the natural history of the disease in the absence of treatment and can therefore be attributed to be a direct therapeutic effect.
Discovery of mTOR as key driver of microcystic LMP
Important insights gained over the last decade have implicated increased activation of the PI3K/mTOR signaling pathway in microcystic LM. Enhanced mTOR signaling has been observed to increase the expression of the vascular endothelial growth factor, or VEGF, a key promoter of angiogenesis and lymphangiogenesis. This leads, in turn, to uncontrolled, disorganized, and malformed lymphatic development.
Hyperactivation of the PI3K/mTOR pathway results in lymphatic endothelial cell proliferation and migration, defective mural cell coverage and aberrant lymphatic vascular network formation. This ultimately results in the anatomic malformations in lymphatic channels seen in this disease.
Rapamycin inhibits mTOR, which is a downstream element of the over-activated PI3K/mTOR pathway (Figure 2). Rapamycin demonstrated in preclinical studies an ability to decrease mTOR signaling, thereby reducing endothelial cell proliferation and subsequently the formation of malformed lymphatic vessels. Additionally, rapamycin reduces lymph fluid formation in the affected tissue, helping to minimize clinical symptoms associated with microcystic LM.
FIGURE 2. PI3K/mTOR Pathway Is Overactivated in Microcystic LM and Point of Rapamycin Pharmacologic Inhibition
A large and growing evidence base exists demonstrating rapamycin’s activity in treating microcystic LM: since 2011, a total of 16 studies evaluating the off-label use of rapamycin in microcystic LM have been published. In a 2021 article by Kalwani et al, the authors stated “Sirolimus [rapamycin], a strong inhibitor of mTOR, has shown tremendous promise in the treatment of LM.” Systematic reviews of rapamycin for the treatment of microcystic LMs have demonstrated that rapamycin can significantly improve the prognosis.
Oral rapamycin is sometimes used in clinical practice in leading academic vascular anomalies clinics where microcystic LM patients are often treated. Importantly, off-label use of oral rapamycin is associated with an adverse event profile that requires frequent patient monitoring and limits its use for a chronic disease such as microcystic LM (Figure 3). Particularly for pediatric and adolescent patients, these toxicities limit the use of oral rapamycin. In addition, oral rapamycin is associated with a narrow therapeutic window due to the adverse event profile described above and the poor biodistribution of oral rapamycin to the dermis, which is where microcystic LMs originate.
FIGURE 3. Adverse Events Observed with Oral Rapamycin Treatment In the Study of Prophylaxis of Organ Rejection Following Renal Transplantation
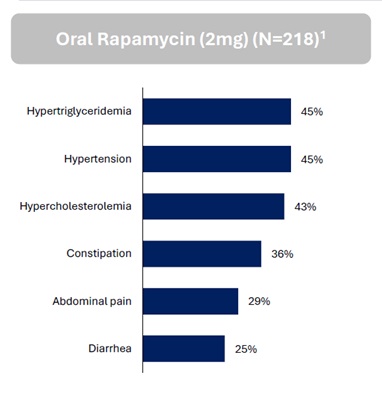
As a result, there remains a significant unmet need for a targeted rapamycin therapy for microcystic LM that limits systemic absorption and the adverse effects, or AEs, associated with systemic therapy.
Advancing QTORIN Rapamycin in Microcystic LM
Palvella is evaluating QTORIN rapamycin for the treatment of microcystic LM. QTORIN rapamycin has the potential to be the first therapy and standard of care in the U.S. for microcystic lymphatic malformations, if approved.
Based on preclinical studies, Palvella believes that QTORIN rapamycin will deliver concentrations of rapamycin approximately 1000-fold higher than systemic rapamycin to the cutaneous tissue with minimal systemic absorption. Palvella therefore believes that QTORIN rapamycin has the potential to harness the potential therapeutic benefits of rapamycin while minimizing the well-known side effects of oral rapamycin.
Palvella completed an open-label Phase 2 trial to evaluate QTORIN rapamycin in patients with microcystic LM in the 4th quarter of 2022. Results of that trial are detailed below. Based on those results and discussions with the FDA at a Type C Meeting in 2023 and a Type B Breakthrough Therapy Meeting in 2024 regarding the proposed patient population, dosing, and endpoint selection for its next clinical trial, Palvella has initiated a Phase 3 trial, SELVA (PALV-09), to evaluate QTORIN rapamycin in patients with microcystic LM in the 3rd quarter of 2024. Palvella expects to report top-line data from this trial in the first quarter of 2026.
QTORIN rapamycin has been granted FDA Fast Track Designation, Orphan Drug Designation, and Breakthrough Therapy Designation for the treatment of microcystic LM.
Clinical Development Overview
Palvella completed an open-label, Phase 2 trial (PALV-06) with QTORIN rapamycin in patients with microcystic LM and based on the results of that trial, Breakthrough Therapy Designation was granted to QTORIN rapamycin for the treatment of microcystic LM. A subsequent Breakthrough Therapy Designation meeting with the FDA was held and the study was initiated in the third quarter of 2024.
PALV-06 Overview and Efficacy Results
The Phase 2 PALV-06 trial was a multi-center, open-label study of subjects receiving QTORIN™ rapamycin once-daily for 12-weeks. The Phase 2 clinical trial featured multiple pre-specified efficacy assessments, including clinician and patient global impression assessments as well as assessments of individual clinical manifestations that are important disease burdens for individuals living with microcystic LMs. As is common in Phase 2 studies, efficacy was evaluated as secondary endpoints without multiplicity adjustment or formal statistical analysis. The PALV-06 trial enrolled a total of 12 participants, all of whom completed 12-weeks of QD QTORIN rapamycin treatment as well as all study related activities.
A baseline-controlled study is a clinical study in which the patient's condition during treatment is compared with their condition before treatment. In such studies, participants serve as their own control. In a placebo-controlled study, patients are randomized prior to treatment to receive either study drug or matching placebo and to determine how the efficacy of the treatment compares to placebo. Baseline-controlled studies are appropriate when the effects are dramatic, occur rapidly following treatment, and are unlikely to have occurred spontaneously (e.g., general anesthesia, cardioversion, measurable tumor shrinkage).
FIGURE 4: PALV-06 Improvement in Clinician- and Patient- Reported Impression of Change
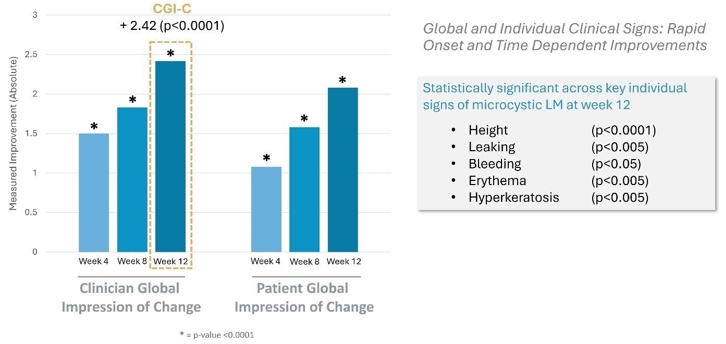
Efficacy data from the Phase 2 open-label study demonstrated nominally statistically significant and clinically meaningful improvements for microcystic LM participants treated with QTORIN rapamycin on several of the efficacy endpoints studied. The data demonstrated improvements as compared to pre-treatment (baseline) across several clinically relevant and important endpoints, including many of the static and impression of change global instruments (Table 2). Statistically significant improvements in the clinician global impression of severity (CGI-S), clinician global impression of change (CGI-C), and patient global impression of change (PGI-C) were supported by visual improvements of target lesions captured in photographs. Clinical meaningfulness was confirmed by participant interviews.
TABLE 2: PALV-06 Study Efficacy Results on Clinician- and Patient-Reported Impression of Change Instruments As Compared to Pre-Treatment (Baseline)
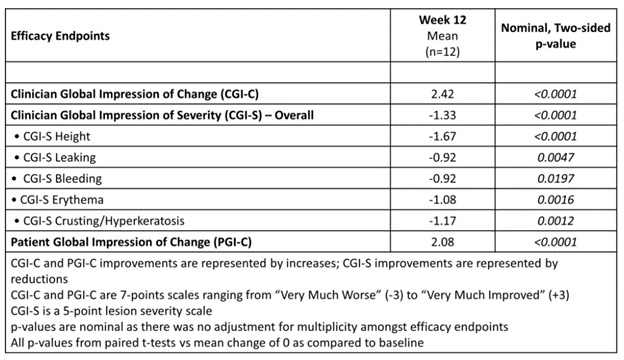
In addition to meaningful improvements in clinician- and patient-reported outcomes, visible improvement in lesions was observed following treatment with QTORIN rapamycin.
FIGURE 5: Visible Improvement in Microcystic LM Lesions During QTORIN Rapamycin Treatment in PALV-06
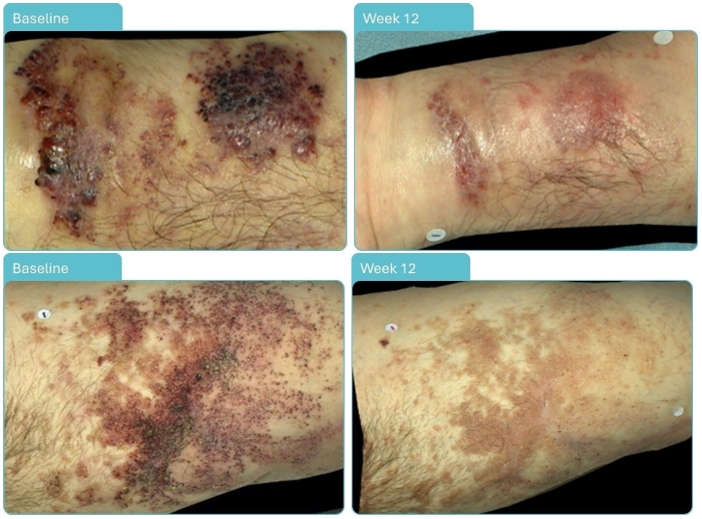
PALV-06 Phase 2 Pharmacokinetic and Safety/Tolerability Results
Systemic concentrations of rapamycin following administration of QTORIN rapamycin in the PALV-06 trial were <2 ng/mL for all participants at all time points tested with an average of 120-98 pg/mL across all patients and time points tested. Safety data obtained in the PALV-06 trial was similar to that observed in larger clinical studies of QTORIN rapamycin, including clinical trials in Pachyonychia Congenita (PALV-02, -03, -05) and Gorlin Syndrome (PALV-04). QTORIN rapamycin was generally well tolerated with all treatment related adverse events either mild or moderate. No study participants discontinued or withdrew from the study. No SAEs, clinically significant lab abnormalities or vital sign abnormalities were reported. The most common TRAEs occurring in >2 participants were application site pain (n=3, 25.0%), application site pruritus (n=3, 25.0%), and nausea (n=2, 16.7%).
TABLE 3: PALV-06 Treatment Related Adverse Events in Microcystic LM Participants
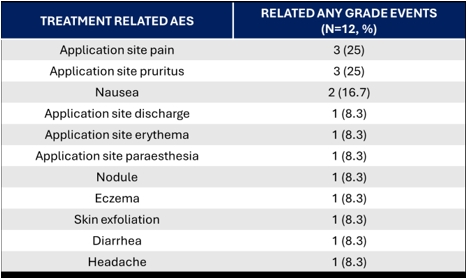
Phase 3 trial – PALV-09 (SELVA) and Anticipated pre-NDA Meeting
Palvella designed its Phase 3 trial (Figure 6) based on results from the Phase 2 trial and consideration of comments from the FDA during End of Phase 2 and Breakthrough Therapy Designation Meetings. Discussions with the FDA focused on several aspects of the proposed clinical trial design, including the patient population, dosing, and endpoint selection. The FDA commented on each of these areas and advised where further clarification was requested.
Subsequent to the Breakthrough Therapy Designation Meeting and incorporation of certain FDA feedback into the Phase 3 trial design, Palvella was notified in September 2024 that it had received an FDA Orphan Drug Clinical Trials Grant for up to $2.6 million to support its Phase 3 trial of QTORIN rapamycin for the treatment of microcystic LM. Since the program's inception, the FDA has awarded approximately 700 Orphan Products Clinical Trial Grants to fund clinical trials of products evaluating the efficacy and/or safety in support of a new indication or change in labeling to address unmet needs for patients with rare diseases or conditions. Grant applications are peer reviewed and evaluated for scientific and technical merit by a panel of experts in the subject field of the specific application. Consultation with the relevant FDA review division may also occur during this phase of the review to determine whether the proposed study will provide acceptable data that could contribute to product approval. A score is assigned to each application based on the scientific/technical review criteria including:
| ● | rationale | |
| ● | study design | |
| ● | inclusion of patient input | |
| ● | investigator(s) | |
| ● | infrastructure | |
| ● | financial resources | |
| ● | ability to advance the current field. |
The review panel may advise the Orphan Products Grant program staff about the appropriateness of the proposal to the goals of the grant program. Since inception, the FDA Orphan Products Grants Program has funded clinical trials that have facilitated the approval of more than 85 products. Palvella’s receipt of the grant does not guarantee FDA approval of QTORIN rapamycin for the treatment of microcystic LM or any other indication.
The Phase 3 trial to evaluate QTORIN rapamycin in patients with microcystic LM includes up to 40 participants who will administer QTORIN rapamycin QD for 24+ weeks. The primary and key secondary endpoints are a 7-point change mLM-IGA, a dynamic assessment that uses a comparative rating scale, and a blinded evaluation using the microcystic LM multi-component static scale, respectively. Clinician-reported change in severity from the start of treatment as measured by the mLM-IGA scale is supported by Phase 2 trial results as exit interviews conducted with the clinicians who were part of the trial. More specifically, these data support that clinicians can accurately rate change in microcystic LM disease severity across each level of disease activity. The endpoints have been designed to capture clinical changes in key aspects of a patient’s disease, as reported by the clinicians and patients.
| ● | the mLM-IGA is an endpoint that was specifically designed for this rare disease population with extensive endpoint development incorporating both physician and patient views; and | |
| ● | the use of a global instrument was the strong and consistent preference of clinician investigators due to it being a multi-sign/symptom disease. |
The FDA has recommended that primary efficacy in the treatment of microcystic LM be evaluated on a static multicomponent assessment scale but recommended that Palvella provide a rationale for selecting the comparative rating scale should Palvella proceed with a comparative rating scale. While static scales were explored, these scores were shown to be less sensitive. Furthermore, the mLM-IGA is different from the traditional comparative rating scales in that investigators must score individual clinical signs before filling out the mLM-IGA and the mLM-IGA leverages baseline photographs to provide more objective scoring.
The mLM-IGA also leverages the well-accepted 7-point dynamic change scale that has been used in FDA labeling across many diseases/therapeutic areas.
Palvella believes that a baseline-controlled study is an appropriate trial in patients with microcystic LM because there is evidence the effects of QTORIN rapamycin in this setting are dramatic and occur rapidly following treatment, and effects are unlikely to have occurred spontaneously. The Phase 2 study was a baseline-controlled study, and provided evidence that the treatment effect with QTORIN rapamycin was dramatic and occurred rapidly as evidenced by nominally significant results at the first timepoint measured, 4 weeks. These effects, as well as any results we may see from the ongoing Phase 3 study, are unlikely to have occurred spontaneously. Microcystic LM has a well-understood pathophysiology and a well-defined disease course such that the natural history of the disease shows that patients with microcystic LM do not have spontaneous regression. Therefore, any improvement can more confidently be attributed to study drug rather than natural fluctuations or spontaneous improvement of the disease. This aligns with the FDA's Draft Guidance on Expedited Programs for Serious Conditions – Drugs and Biologics (May 2014), which states “Single-arm trials may be an important option in rare diseases with well-understood pathophysiology and a well-defined disease course.” However, the FDA may not agree microcystic LM is an appropriate setting for a baseline-controlled Phase 3 study and has commented that a placebo-controlled trial or additional trials assessing different clinical endpoints may be required to assess the efficacy of QTORIN rapamycin for the treatment of microcystic LM.
FIGURE 6. Microcystic LM Baseline-Controlled Phase 3 Trial Design

Assuming positive results from the Phase 3 study, we plan to submit a Section 505(b)(2) NDA for QTORIN rapamycin for the treatment of microcystic LM. Palvella’s NDA strategy is to provide the clinical evidence generated from the Phase 3 study to demonstrate the safety and efficacy of a treatment in the microcystic LM patient population and combine it with confirmatory evidence from the Phase 2 data, real-world evidence and natural history of the disease. In addition to the Phase 3 safety and efficacy data, Palvella plans to use the Phase 3 trial to bridge QTORIN rapamycin to the oral listed drug based on a cross-study comparison between the PK of QTORIN rapamycin from the Phase 3 trial and the published PK of RAPAMUNE and will rely on the listed drug, RAPAMUNE, for additional components of the NDA. If the Phase 3 trial yields a statistically significant result, Palvella plans to review the nonclinical and clinical data with the FDA at a pre-NDA meeting, including to determine whether additional clinical safety or efficacy trials or additional bridging studies may be required to pursue a Section 505(b)(2) NDA pathway for QTORIN rapamycin.
To support Palvella’s 505(b)(2) NDA, Palvella plans to bridge QTORIN rapamycin and RAPAMUNE based on a cross-study comparison between pharmacokinetic data from QTORIN rapamycin and the prescribing information for RAPAMUNE, the FDA recommends that bridging to support an NDA for the treatment of microcystic LM be done in a relative bioavailability study comparing the pharmacokinetics of a topical product applied under maximal use conditions and the approved oral drug. The planned cross study analysis will allow for comparison of systemic pharmacokinetic parameters, key criteria for assessing the applicability of safety findings from the listed drug, which are a result of systemic exposure from the oral formulation. Palvella believes the proposed clinical pharmacology plan will address the requirements for bridging to support reliance on the FDA’s previous findings of safety for RAPAMUNE tablets to support a 505(b)(2) NDA submission by establishing relative bioavailability to known pharmacokinetic parameters of RAPAMUNE as well as pharmacokinetics under maximal use conditions. Population pharmacokinetic analyses, including covariate analyses, will be conducted as data allows. No additional studies are planned, as a bridging approach is planned to enable labeling guidance for specific populations and drug-drug interactions.
Potential Market Opportunity and Market Research
Palvella believes that QTORIN rapamycin, if approved, has commercial potential for microcystic LM in the U.S. The treatment regimen in microcystic LM, Palvella believes, would be chronic dosing due to the genetic nature of the condition. As discussed below, based on a published real-world occurrence study of U.S. physicians, Palvella estimates that there are over 30,000 diagnosed microcystic LM patients in the United States. Furthermore, the introduction of a new treatment may lead to improved awareness of the disease, better and sooner diagnosis, and more patients actively seeking therapy.
As part of better understanding the market opportunity in microcystic LM, Palvella commissioned a primary market research study in May 2024 that surveyed 52 dermatologists and hematologists (Figure 7). Furthermore, as part of Palvella’s market research, a target product profile, or Product X, was presented based on the Phase 2 results.
FIGURE 7. Market Research Report (May 2024) On Product X
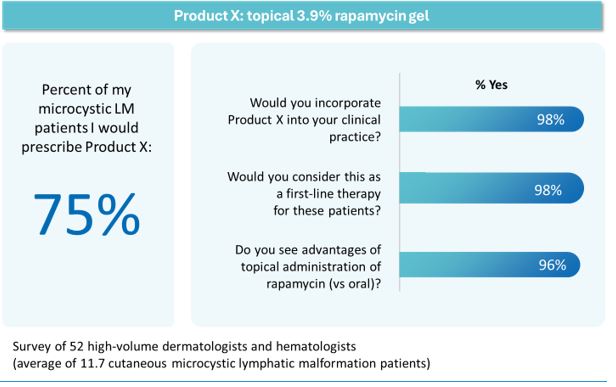
Palvella believes that this preliminary market research underscores both the unmet need and the significant market opportunity for QTORIN rapamycin for the treatment of microcystic LM.
QTORIN Rapamycin for the Treatment of Cutaneous Venous Malformations
|
Objective |
● |
There are currently no FDA-approved therapies indicated for the treatment of cutaneous venous malformations; Palvella is developing the first targeted therapy |
|
Program Status and Upcoming Milestones |
● |
Palvella plans to initiate its Phase 2 clinical trial in cutaneous venous malformation patients in the 4th quarter of 2024 |
| ● | Palvella expects to report top-line data in the fourth quarter of 2025 | |
|
Genetic Basis and Molecular Pathways |
● |
Somatic mutations in TEK or PIK3CA lead to aberrant PI3K/mTOR signaling |
|
Disease Burden |
● |
Cutaneous venous malformations are a serious, rare condition characterized by the overgrowth of veins that protrude through the skin and is characterized by deformities, functional impairment and hemorrhaging |
| ● | Usually present early in life; progresses throughout life | |
| ● | Localized masses of malformed veins protrude through the skin barrier | |
|
Scientific Rationale for Cutaneous Venous Malformations |
● |
TIE2 and PI3K overactivation converge on mTOR |
| ● | Rapamycin directly inhibits overactivated mTOR activity and decreases endothelial proliferation and venous formation |
|
Palvella’s Targeted Approach |
● |
QTORIN is designed to confer site-directed delivery of rapamycin to the epidermis and dermis |
|
Market Dynamics |
● |
Estimated prevalence: >75,000 in the United States |
| ● | Palvella believes the estimated TAM opportunity on an annualized basis is greater than $1 billion | |
|
Intellectual Property; Regulatory Designations |
● |
Palvella holds U.S. patents and applications in the U.S. and major foreign markets with claims directed to anhydrous gel formulations of rapamycin and methods of use for treating cutaneous venous malformations, expiring in 2038 |
Disease Overview
Cutaneous venous malformations are congenital vascular anomalies characterized by dysregulated growth of veins within the skin. They present as dilated, tortuous vessels that manifest as bluish or purplish patches or nodules on the skin. These malformations result from developmental errors in venous morphogenesis during embryogenesis, leading to abnormal connections between veins and capillaries. These anomalies are typically present at birth and can expand or become more prominent with age. They vary in size and distribution, ranging from small, localized lesions to more extensive areas of affected skin. Cutaneous venous malformations cause functional impairment, significantly impact quality of life and are associated with severe long-term complications.
Management of cutaneous venous malformations depends on factors such as symptomatology, location, and patient preferences. Treatment options, which are limited and insufficient, include conservative approaches such as observation and compression therapy, as well as interventional techniques like sclerotherapy (injection of sclerosing agents to induce vessel shrinkage), laser therapy, and surgical excision for larger or symptomatic malformations. Procedures to remove venous malformations are often not curative, with high rate of recurrence/regrowth. Complications from serial attempts to remove venous malformations including scarring, swelling, and nerve deficits are also compounded when multiple procedures are required. There are no FDA approved treatments for cutaneous venous malformations and there is an urgent need for an approved pharmacologic treatment for these patients.
Cutaneous venous malformations is a chronic disease that worsens over time with no spontaneous regression. The invasiveness and limited effectiveness of current treatments, coupled with the lack of approved pharmacotherapy options, demonstrate the urgent need for an FDA approved therapy for cutaneous venous malformations. A targeted topical therapy that directly addresses disease pathology is of interest to this patient population, as it could abolish the need for systemic treatments that have wider toxicity or invasive procedural interventions.
FIGURE 8. Patients with Cutaneous Venous Malformations
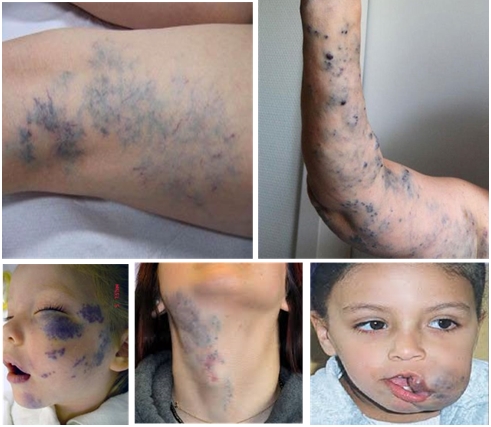
Discovery of mTOR as key driver of Venous Malformations
Cutaneous venous malformations are primarily caused by somatic mutations in either TEK or PIK3CA leading to overactivated PI3K/mTOR signaling. TEK encodes for the endothelial cell-specific receptor tyrosine kinase (TIE2) which in turn activates phosphatidylinositol-3-kinase (PI3K) with mutations in this gene accounting for approximately 70% of cutaneous venous malformation cases. Mutations in the PIK3CA gene, which encodes the p110α catalytic subunit of PI3K, have also been identified in cutaneous venous malformations accounting for approximately 30% of cases that do not have TEK mutations. The PI3K/mTOR pathway plays a crucial role in regulating cell growth, proliferation, and survival. Mutations in TEK or PIK3CA lead to increased activation of this pathway, promoting abnormal endothelial cell proliferation and result in the formation of cutaneous venous malformations. Rapamycin, an mTOR inhibitor, dampens PI3K/mTOR signaling, thus garnering attention as a potential therapeutic option for cutaneous venous malformations.
Palvella’s Solution: QTORIN Rapamycin
Palvella is developing QTORIN rapamycin for the treatment of cutaneous venous malformations. Rapamycin inhibits mTOR, which is a downstream element of the PI3K/mTOR pathway. In doing so, rapamycin is thought to diminish PI3K/mTOR overactivation, thereby reducing endothelial cell proliferation and subsequently the formation of malformed vessels. Several published case studies and clinical trials have demonstrated efficacy of the off-label use of oral rapamycin for the treatment of venous malformations.
Clinical Development Overview
Palvella plans to initiate its Phase 2 clinical trial, a 12-week, multicenter, baseline-controlled Phase 2 clinical trial of QTORIN rapamycin in up to 20 patients with cutaneous venous malformations in the 4th quarter of 2024. Palvella plans to enroll patients aged 6 years or older, who will receive treatment for 12 weeks. Assuming favorable results, Palvella plans to meet with the FDA for an end of Phase 2 meeting to discuss a Phase 3 clinical study.
FIGURE 9. PALV-10 Trial Design
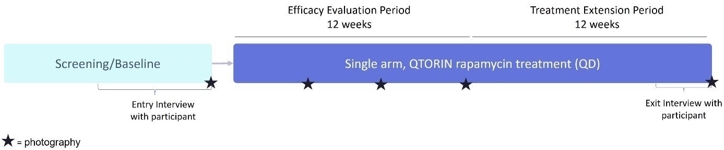
Potential Market Opportunity
Palvella believes that QTORIN rapamycin, if approved, has significant commercial potential in cutaneous venous malformations in the U.S. and other markets. The treatment regimen in cutaneous venous malformations, Palvella believes, would be chronic dosing due to the genetic nature of the condition. Palvella estimates, based on published epidemiologic work, that there are >75,000 patients living with cutaneous venous malformations in the United States. Based on this estimated U.S. prevalence, Palvella believes the TAM opportunity on an annualized basis for QTORIN rapamycin in cutaneous venous malformations is greater than $1B. Furthermore, the introduction of a new therapy may lead to improved awareness of these diseases, better and sooner diagnosis, and more patients actively seeking therapy.
Additional mTOR Driven Diseases
Palvella has identified several other rare genetic skin diseases that are driven by mTOR and available clinical data suggests that inhibition of mTOR may be a good therapeutic target in these populations. These diseases include but are not limited to refractory vascular tumors, capillary malformations, and cutaneous sarcoidosis. Palvella is currently evaluating several of these opportunities for clinical development.
Palvella intends to build commercial infrastructure in the United States to support the commercialization of its product candidates, if approved. Palvella plans to implement a staggered approach to building its commercial team aligned with the progress of its clinical development and advancement towards registration. This approach allows it to grow the organization while appropriately supporting the necessary market development and launch objectives.
The initial focus of Palvella’s commercial sales effort will be on the subset of multidisciplinary care teams and medical dermatologists at vascular anomaly centers, many of whom Palvella has established relationships through its clinical development initiatives. Palvella plans to engage these physicians by building an experienced rare disease and dermatology-oriented sales force which will be supported by patient consumer and health care provider marketing programs tailored to the indications and communities its products treat. The potential to bring forth new differentiated treatments in rare genetic skin diseases for which no treatments currently exist will help position Palvella to engage this population of physicians. Over time, Palvella hopes to generate operational leverage from its field organization as it expands to potential future rare genetic skin diseases indications.
Palvella expects that the patients who are prescribed its product will be serviced by a highly customized support system of programs and resources to support both their access to and appropriate use of Palvella’s therapies. This will include distribution through specialty pharmacy partners, reimbursement and product administration support through a patient services team trained specifically on the needs of people with rare genetic skin diseases and access programs aimed at providing copay assistance. In certain instances, it may include access to free product through a patient assistance program for eligible individuals. These programs and resources will be built specifically with feedback from the individuals with these diseases and their caregivers in mind. The patient services team will act with the highest levels of integrity and also be highly focused on ensuring that all individuals and physicians who interact with Palvella’s programs, distribution partners and company have a high level of satisfaction.
To support access to and reimbursement for its therapies, Palvella expects to deploy an experienced patient access team to collaboratively engage with payors, provide education regarding the diseases it treats and provide education regarding its products’ value propositions. Value propositions based on clinical data will be key to supporting Palvella’s product pricing strategies. Palvella plans on engaging with payors leading up to the potential product launch and continuing to support ongoing access creation throughout the life cycle of the product candidate, if approved. As Palvella seeks to develop and receive regulatory approval for the treatment of new indications for existing product candidates or develop and commercialize new products, once approved, Palvella’s patient access team will seek to position itself to provide market access and education.
Palvella expects its commercial organization to be complemented by a medical affairs team tasked with appropriately educating clinical decision makers on the scientific data on the company’s products in development, and those that are approved, if applicable. Medical affairs will do this through support of appropriate medical education initiatives, supporting the publication of relevant data at scientific meetings, executing a publication strategy to disseminate new scientific details on Palvella’s products, and responding to all incoming requests for medical information. Palvella also plans to identify where appropriate the opportunity to support investigator-initiated trials that may expand the scientific body of evidence for Palvella’s products, and potentially to provide grants to researchers in areas of company interest.
Palvella anticipates that it will be required to invest significant amounts of financial and management resources to develop the appropriate infrastructure to prepare for commercialization. Palvella intends to scale certain investments so that they align with achievement of regulatory hurdles, but significant expenditures may be required prior to the receipt of any regulatory approval of its product candidate.
Outside of the United States, Palvella may consider building its own commercial infrastructure, or out licensing , where appropriate, and may elect in the future to utilize strategic collaborators, distributors, or other partners with making its products available to patients.
While Palvella has personnel with substantial manufacturing experience, it does not own or operate manufacturing facilities for the production of clinical quantities of its product candidates and Palvella currently has no plans to build its own clinical or commercial-scale manufacturing capabilities. Palvella relies on contract manufacturing organizations, or CMOs, to manufacture and supply Palvella’s materials to be used for the development and commercialization of its current and any future product candidate and expect such reliance to continue for the foreseeable future. Palvella also relies, and expects to continue to rely, on third parties to package, label, store and distribute its current and any future investigational product candidate, as well as its commercial products if marketing approval is obtained. Palvella believes that this strategy allows it to maintain a more efficient infrastructure by eliminating the need for it to invest in its own manufacturing facilities, equipment and personnel while also enabling it to focus its expertise and resources on the development of Palvella’s current and any future product candidate.
Palvella has agreements with Altasciences and PMRS, both cGMP facilities, for the manufacture of its clinical supply of its product candidate for clinical trials and for the manufacture of a commercial supply of its QTORIN rapamycin, if approved. Palvella has agreements with Medpharm UK for the manufacture of its clinical supply of its product candidate for clinical trials. Palvella obtains supplies of drug substance for its product candidate on a purchase order basis from three sources.
As Palvella advances QTORIN rapamycin through development, it will add backup suppliers for drug product manufacture and packaging to protect against any potential supply disruptions.
Additionally, Palvella has a supply agreement with Nemera, for the supply of pumps it intends to use to package QTORIN rapamycin, if its product candidate is approved. Nemera is a sole source supplier with respect to these pumps, and Palvella is required under the supply agreement to purchase from Nemera. Palvella believes that the Nemera metered dose pump used to deliver its QTORIN rapamycin products will not require separate FDA approval (or approval as a combination product), based upon a preliminary determination from the FDA that the pump is exempt from such requirements. However, if the FDA ultimately disagrees, Palvella’s product candidate, if approved, may be regulated as a combination products requiring clearance or review of the delivery device by the FDA.
The use of CMOs and reliance on collaboration partners is cost-efficient and has eliminated the need for Palvella’s direct investment in manufacturing facilities and additional staff early in development. Palvella believes available CMOs are capable of providing sufficient quantities of its product candidate, if approved, to meet anticipated full-scale commercial demands. However, there are a limited number of manufacturers capable of producing Palvella’s product candidates, particularly its current product candidates which incorporate rapamycin.
Significant uncertainty exists as to the coverage and reimbursement status of any product that receives regulatory approval. In the United States, sales of any product candidates for which regulatory approval for commercial sale is obtained will depend in part on the availability of coverage and adequate reimbursement from third-party payors. Third-party payors include government authorities and health programs in the United States such as Medicare and Medicaid, managed care providers, private health insurers and other organizations. These third-party payors are increasingly reducing reimbursements for medical products and services. The process for determining whether a payor will provide coverage for a drug product may be separate from the process for setting the reimbursement rate that the payor will pay for the drug product. Third-party payors may limit coverage to specific drug products on an approved list, or formulary, which might not include all of FDA-approved drugs for a particular indication. Additionally, the containment of healthcare costs has become a priority of federal and state governments, and the prices of drugs have been a focus in this effort.
The U.S. government, state legislatures and foreign governments have shown significant interest in implementing cost-containment programs, including price controls, restrictions on reimbursement and requirements for substitution of generic products. Adoption of price controls and cost-containment measures, and adoption of more restrictive policies in jurisdictions with existing controls and measures, could further limit Palvella’s net revenue and results.
A payor’s decision to provide coverage for a drug product does not imply that an adequate reimbursement rate will be approved. Further, coverage and reimbursement for drug products can differ significantly from payor to payor. As a result, the coverage determination process is often a time-consuming and costly process that will require it to provide scientific and clinical support for the use of any Palvella product which receives marketing authorization to each payor separately, with no assurance that coverage and adequate reimbursement will be applied consistently or obtained in the first instance.
Third-party payors are increasingly challenging the price and examining the medical necessity and cost-effectiveness of medical products and services, in addition to their safety and efficacy. New metrics frequently are used as the basis for reimbursement rates, such as average sales price, average manufacturer price and actual acquisition cost. In order to obtain coverage and reimbursement for any product that might be approved for sale, it may be necessary to conduct expensive pharmacoeconomic studies in order to demonstrate the medical necessity and cost-effectiveness of the products, in addition to the costs required to obtain regulatory approvals. If third-party payors do not consider a product to be cost-effective compared to other available therapies, they may not cover the product after approval as a benefit under their plans or, if they do, the level of payment may not be sufficient to allow a company to sell its products at a profit.
The marketability of any of its current or any future product candidates for which Palvella receives regulatory approval for commercial sale may suffer if the government and third-party payors fail to provide adequate coverage and reimbursement. In addition, emphasis on managed care in the United States has increased and Palvella expects will continue to increase the pressure on pharmaceutical pricing. Coverage policies and third-party reimbursement rates may change at any time. Even if favorable coverage and reimbursement status is attained for one or more products for which Palvella or its collaborators receive regulatory approval, less favorable coverage policies and reimbursement rates may be implemented in the future.
In the European Union, pricing and reimbursement schemes vary widely from country to country. Some countries provide that products may be marketed only after a reimbursement price has been agreed. Some countries may require the completion of additional studies that compare the cost-effectiveness of a particular product candidate to currently available therapies. European Union member states may approve a specific price for a product, or it may instead adopt a system of direct or indirect controls on the profitability of the company placing the product on the market. Other member states allow companies to fix their own prices for products but monitor and control company profits. The downward pressure on health care costs has become intense. As a result, increasingly high barriers are being erected to the entry of new products. In addition, in some countries, cross-border imports from low-priced markets exert competitive pressure that may reduce pricing within a country. Any country that has price controls or reimbursement limitations may not allow favorable reimbursement and pricing arrangements.
Palvella’s success depends in part on its ability to obtain and maintain proprietary protection for product candidates and any of its future product candidates, core technologies, novel discoveries, product development technologies and know-how; to operate without infringing on the proprietary rights of others; and to prevent others from infringing its proprietary rights. Palvella’s policy is to seek to protect its proprietary position by, among other methods, filing patents and patent applications in the United States and select foreign countries related to Palvella’s proprietary technology, inventions and improvements that are important to the development and implementation of its business. Palvella also seeks to avoid infringing proprietary rights of others. For this reason, Palvella routinely monitors and evaluates third-party patents and publications, and, if necessary, takes appropriate action based on that evaluation. In addition, Palvella relies on trademarks, trade secrets, know-how, continuing technological innovation and potential in-licensing opportunities to develop and maintain its proprietary position.
As of July 10, 2024, Palvella owns or has an exclusive license to issued U.S. and foreign patents and pending U.S. and foreign patent applications and U.S. provisional applications relating to QTORIN rapamycin and uses thereof. Of these QTORIN rapamycin patents and patent applications:
Palvella owns issued patents in the US, as well as Australia, China, Israel and Japan and pending applications in the US, Europe and Japan directed to anhydrous gel formulations of rapamycin and methods of using the same to treat certain skin disorders, including microcystic LM and venous malformations that naturally expire in 2038. Palvella also owns issued US patents and a pending US application that encompass anhydrous gel formulations of mTOR inhibitors, including rapamycin, and methods of using the same to treat skin disorders including microcystic LM and venous malformations that naturally expire as early as 2038. Palvella owns pending applications in the US, Europe and Japan that are directed to the use of QTORIN rapamycin for the treatment of microcystic LM, which if issued, would naturally expire in September 2042. A summary of these patent families is presented in the following table.
|
Owned / Licensed |
# Patents and Countries |
# Applications and Countries |
Natural Expiry Date |
Type of Patent |
|
|
Anhydrous gel formulations of rapamycin and methods of use |
Owned by Palvella |
9 patents in U.S., Australia, China, Israel, and Japan |
4 pending applications in U.S., Canada, Europe and Japan |
January 2038 |
Utility |
|
Anhydrous gel formulations of mTOR inhibitors and methods of use |
Owned by Palvella |
2 U.S. Patents |
1 pending U.S. application |
As early as January 2038 |
Utility |
|
Use of QTORIN rapamycin for treating microcystic LM |
Owned by Palvella |
N/A |
3; U.S., Europe and Japan |
September 2042 |
Utility |
Patent term is based on the filing or grant date of the patent, as well as the governing law of the country in which the patent is obtained. The protection provided by a patent varies from country to country, and is dependent on the type of patent granted, the scope of the patent claims, and the legal remedies available in a given country.
Obtaining patent protection is not the only method that Palvella employs to protect its propriety rights. Palvella also utilizes other forms of intellectual property protection, including trademark, and trade secrets, when those other forms are better suited to protect a particular aspect of its intellectual property. Palvella’s belief is that its propriety rights are strengthened by its comprehensive approach to intellectual property protection.
Maintaining the confidential nature of Palvella’s non-publicly disclosed products and technologies is of paramount importance. For this reason, Palvella’s employees, contractors, consultants, and advisors are required to enter into nondisclosure and invention assignment agreements when their employment or engagement commences. Those individuals also enter into agreements that prohibit the communication or implementation of any third-party proprietary rights during the course of their employment with Palvella. Palvella also requires any third-party that may receive its confidential information or materials to enter into CDAs prior to receipt of that information or material. See “Risk Factors — Risks Related to Intellectual Property” for more information.
Ligand Development Funding Agreement
In December 2018, Palvella entered into the Ligand Agreement, or the “Original Ligand Agreement”, with Ligand, whereby Ligand agreed to make a one-time payment of $10.0 million to fund the development of QTORIN rapamycin. As partial consideration for the one-time payment, Palvella granted Ligand the right to receive up to $8.0 million in milestone payments upon the achievement of certain corporate, financing and regulatory milestones by Palvella related to QTORIN rapamycin for the treatment of any and all indications. In addition, Palvella agreed to pay to Ligand tiered royalties from 5.0% to 9.8% based on any aggregate annual worldwide net product sales of any products based on QTORIN rapamycin. On a licensed product-by-licensed product and country-by-country basis, the royalty period is from the date of first commercial sale of such licensed product in a country until the latest of (i) the expiration of the last valid claim within the licensed patent rights covering such licensed product in the country in which such licensed product is made, used or sold, (ii) the expiration of the regulatory exclusivity term conferred by the applicable regulatory authority in such country with respect to such licensed product, and (iii) the fifteenth anniversary of the first commercial sale of such licensed product in such country. In certain circumstances, Palvella has the right to reduce the royalty rates under the Original Ligand Agreement by making payments, or “Royalty Buy Down Payments”. Specifically, once Palvella has made royalty payments to Ligand equal to certain specified amounts in the mid eight figures, Palvella has the option to make Royalty Buy Down Payments at any time during the remainder of the term of the Original Ligand Agreement to reduce its certain royalty tier percentages on annual worldwide net sales of any products by one or two percentage points. Such Royalty Buy Down Payments range in size from the low seven figures to the low eight figures.
Ligand may terminate the agreement for any or no reason upon a 90-day notice to Palvella. Ligand may also terminate the agreement for cause in connection with a material breach that Palvella does not cure within a certain period of time.
The total amount of potential future milestone payments remaining under the arrangement were $5.0 million as of December 31, 2023 and 2022. The potential future milestone payments represent derivative liabilities with a fair value of $1.0 million and $1.5 million as of December 31, 2023 and 2022, respectively, which are classified as derivative liabilities – royalty agreement on the balance sheets.
Palvella’s obligation under the Original Ligand Agreement was determined to be a debt instrument based on the likelihood of repaying the amounts provided to fund the development of QTORIN rapamycin and that Palvella has significant continuing involvement in the generation of the cash flows potentially due to Ligand. This obligation is reflected as royalty agreement liability which is classified as a long-term liability on the accompanying balance sheets. Interest expense with respect to the royalty agreement liability is determined using the effective interest method based upon risk-adjusted cash flow estimates of Palvella’s potential future royalty payments under the Ligand Agreement, yielding an effective interest rate of 38.9% and 30.3% as of December 31, 2023 and 2022, respectively. The effective interest rate is estimated at each balance sheet date based on the rate that would enable the liability to be repaid in full over the anticipated life of the arrangement. Changes in these estimates will impact the amount of interest expense recognized through the accompanying statements of operations. During the second quarter of 2023, Palvella received data from certain of its clinical trials that reduced the projected net product sales related to QTORIN rapamycin and the corresponding probabilities of successful commercialization, resulting in a significant reduction in the expected future royalty payments and a corresponding reduction in the royalty agreement liability as of December 31, 2023. Palvella incurred non-cash interest income (expense) of $6.3 million and ($10.4) million for the years ended December 31, 2023 and 2022, respectively, all of which is a component of the royalty agreement liability on the accompanying balance sheets.
In November 2023, the Original Ligand Agreement was amended, or the “Amended Ligand Agreement” and together with the Original Ligand Agreement, the “Ligand Agreements”, whereby Ligand paid Palvella an additional $5.0 million in return for an increase in the future tiered royalties to 8.0% to 9.8% of any aggregate annual worldwide net product sales of any products based on QTORIN rapamycin The Royalty Buy Down Payments, and the associated rate modifications, in the Original Ligand Agreement were eliminated as part of the Amended Ligand Agreement. The Amended Ligand Agreement also replaced the termination provision so that the agreement may be terminated by the earlier of a mutual written agreement of the parties or when the royalties contemplated by the agreement are paid to Ligand. Palvella evaluated the accounting for the Amended Ligand Agreement under ASC 470, Debt, and concluded that the present value of the cash flows under the Amended Ligand Agreement differed by more than 10% from the present value of the cash flows under the Original Ligand Agreement. As such, the Original Ligand Agreement was extinguished, and the Amended Ligand Agreement was recorded at the estimated fair value of the royalty agreement liability on the date of the amendment. This resulted in a one-time, non-cash gain on extinguishment of approximately $23.1 million being recorded in the accompanying statement of operations.
The Ligand Agreements require Palvella to make certain estimates and assumptions about the timing and probability of FDA approval and commercialization, and the amount of future net sales for any product containing QTORIN rapamycin. The estimated future net sales are based on subjective assumptions that include the estimated size of the addressable patient population and the anticipated pricing of Palvella’s products. These estimates and assumptions are subject to significant variability and are thus subject to significant uncertainty. Therefore, these estimates and assumptions are likely to change as Palvella develops and commercializes products containing QTORIN rapamycin that may result in future adjustments to the royalty agreement liability, the derivative liabilities, and the accretion of interest expense.
The pharmaceutical and biotechnology industries are characterized by rapidly advancing technologies, intense competition, and a strong emphasis on proprietary products. Potential competitors with product candidates in development for rare genetic skin diseases include Kaken Pharmaceutical Co., Ltd., Nobelpharma Co., Ltd., Novartis Pharmaceuticals, Relay Therapeutics, Inc. and Vaderis Therapeutics AG. While Palvella believes that its technology, expertise, scientific knowledge and intellectual property provide it with competitive advantages, it faces and will continue to face competition from these companies and other sources, including major pharmaceutical, specialty pharmaceutical and biotechnology companies, academic institutions, governmental agencies and public and private research organizations that conduct research, seek patent protection, and establish collaborative arrangements for research, development, manufacturing, and commercialization. Any product candidates that Palvella successfully develops and, if approved, commercializes may compete with existing therapies or procedures and new therapies that may become available in the future. Moreover, Palvella’s industry is characterized by the existence of large numbers of patents and frequent allegations of patent infringement.
There are no approved pharmacotherapies currently available for the treatment of microcystic LMs or cutaneous venous malformations. The current treatment options for microcystic LMs include a high-risk surgical procedure and off-label use of sclerosants, including doxycycline, bleomycin, ethanol and sodium tetradecyl sulfate. The current treatment options for cutaneous venous malformations include conservative approaches such as observation and compression therapy, as well as interventional techniques like sclerotherapy, laser therapy and surgical excision for larger or symptomatic malformations. There are a number of drug development companies and academic researchers exploring oral and topical formulations of various agents for the treatment of LMs and VMs including macrolides, phosphodiesterase inhibitors, P13K inhibitors, AKT inhibitors, and mTOR inhibitors. A majority of these are in early development.
The key competitive factors affecting the success of Palvella’s current or future product candidates, if approved, are likely to be their efficacy, safety, convenience and price, the level of competition and the availability of coverage and adequate reimbursement from third-party payors. If any of Palvella’s product candidates are approved and successfully commercialized, it is likely that it will face increased competition as a result of other companies pursuing development of products to address similar diseases.
Many of the companies against which Palvella is competing or against which it may compete in the future have significantly greater financial resources and expertise in research and development, manufacturing, preclinical testing, conducting clinical trials, obtaining regulatory approvals and marketing approved products than it does. These competitors also compete with Palvella in recruiting and retaining qualified scientific and management personnel and establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies complementary to, or necessary for, its programs. Mergers and acquisitions in the pharmaceutical and biotechnology industries may result in even more resources being concentrated among a smaller number of Palvella’s competitors. Smaller and other early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies.
Palvella may pursue the in-license or acquisition of rights to complementary technologies and product candidates on an opportunistic basis. The acquisition and licensing of technologies and product candidates is a competitive area, and a number of more established companies also have similar strategies to in-license or acquire technologies and product candidates that Palvella may consider attractive. These established companies may have a competitive advantage over Palvella due to their size, cash resources and greater development and commercialization capabilities. In addition, companies that perceive it to be a competitor may be unwilling to assign or license rights to Palvella. Palvella also may be unable to in-license or acquire the relevant technology or product candidate on terms that would allow it to make an appropriate return on its investment.
Palvella’s commercial opportunity could be reduced or eliminated if its competitors develop and commercialize products that are safer, more effective, have fewer or less severe side effects, are more convenient or are less expensive than any product candidates that Palvella may develop, and, if approved, successfully commercialize. Palvella’s competitors also may obtain FDA or other regulatory approval for their products more rapidly than Palvella may obtain approval for its own, which could result in Palvella’s competitors establishing a strong market position before Palvella is able to enter the market. In addition, Palvella’s ability to compete may be affected in many cases by insurers or other third-party payors seeking to encourage the use of generic products. Because of Palvella’s primary focus on rare diseases, if its product candidates achieve marketing approval, Palvella expects to seek premium pricing.
In addition, some of the market demand for topical rapamycin may be satisfied by compounding pharmacies. Although such pharmacies will be unable to compound any drug that is essentially a copy of QTORIN rapamycin, if approved, a compounded product would not be considered a copy of QTORIN rapamycin if there were a difference between Palvella’s product and the compounded product that was made for an individual patient and which the prescribing practitioner determines produces a significant difference for that patient. Physicians may determine that such differences exist for some or all of their patients and may choose to prescribe compounded rapamycin provided rapamycin appears on a list established by the FDA of bulk drug substances for which there is a clinical need or satisfies other limited conditions. In the event compounders are authorized to compound rapamycin products following approval of QTORIN rapamycin, if approved, Palvella could be subject to significant competition from those formulations.
Government authorities in the United States, at the federal, state, and local level, and in other countries and jurisdictions, extensively regulate, among other things, the research, development, testing, manufacture, quality control, approval, packaging, storage, recordkeeping, labeling, advertising, promotion, distribution, marketing, post-approval monitoring and reporting, and import and export of pharmaceutical products. The processes for obtaining regulatory approvals in the United States and in foreign countries and jurisdictions, along with subsequent compliance with applicable statutes and regulations and other regulatory authorities, require the expenditure of substantial time and financial resources.
FDA Approval Process
In the United States, pharmaceutical products are subject to extensive regulation by the FDA under the Food, Drug, and Cosmetic Act, or FDCA, and other federal and state statutes and regulations. The failure to comply with applicable U.S. requirements may subject a company to a variety of administrative or judicial sanctions, such as a clinical hold, the FDA’s refusal to approve pending NDAs, warning or untitled letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, civil penalties, and criminal prosecution.
Pharmaceutical product development for a new product or certain changes to an approved product in the U.S. typically involves preclinical laboratory and animal tests, the submission to the FDA of an Investigational New Drug Application, or “IND”, which must become effective before clinical testing may commence, and adequate and well-controlled clinical trials to establish the safety and effectiveness of the drug for each indication for which FDA approval is sought. Satisfaction of FDA pre-market approval requirements typically takes many years and the actual time required may vary substantially based upon the type, complexity and novelty of the product or disease.
Preclinical tests include laboratory evaluation of product chemistry, formulation, and toxicity, as well as animal trials to assess the characteristics and potential safety and efficacy of the product. The conduct of the preclinical tests must comply with federal regulations and requirements, including good laboratory practices. The results of preclinical testing are submitted to the FDA as part of an IND along with other information, including information about product CMC, and a proposed clinical trial protocol. Long-term preclinical tests, such as animal tests of reproductive toxicity and carcinogenicity, may continue after the IND is submitted. A 30-day waiting period after the submission of each IND is required prior to the commencement of clinical testing in humans.
If the FDA has neither commented on nor questioned the IND within this 30-day period, the clinical trial proposed in the IND may begin. Clinical trials involve the administration of the investigational new drug to healthy volunteers or patients under the supervision of a qualified investigator. Clinical trials must be conducted: (i) in compliance with federal regulations; (ii) in compliance with GCP, an international standard meant to protect the rights and health of patients and to define the roles of clinical trial sponsors, administrators, and monitors; as well as (iii) under protocols detailing the objectives of the trial, the parameters to be used in monitoring safety and the effectiveness criteria to be evaluated. Each protocol involving testing on U.S. patients and subsequent protocol amendments must be submitted to the FDA as part of the IND.
The FDA may order the temporary, or permanent, discontinuation of a clinical trial at any time, or impose other sanctions, if it believes that the clinical trial either is not being conducted in accordance with FDA requirements or presents an unacceptable risk to the clinical trial patients. The imposition of a clinical hold may be full or partial. The study protocol and informed consent information for patients in clinical trials must also be submitted to an IRB for approval. An IRB may also require the clinical trial at the site to be halted, either temporarily or permanently, for failure to comply with the IRB’s requirements, or may impose other conditions.
Clinical trials to support NDAs for marketing approval are typically conducted in three sequential phases, but the phases may overlap. In Phase 1, the initial introduction of the drug into healthy human patients, the drug is tested to assess metabolism, pharmacokinetics, pharmacological actions, side effects associated with increasing doses, and, if possible, early evidence of effectiveness. Phase 2 usually involves trials in a limited patient population to determine the effectiveness of the drug for a particular indication, determine optional dose and regimen, and to identify common adverse effects and safety risks. If a drug demonstrates evidence of effectiveness and an acceptable safety profile in Phase 2 evaluations, Phase 3 trials are undertaken to obtain additional information about clinical efficacy and safety in a larger number of patients, typically at geographically dispersed clinical trial sites, to permit the FDA to evaluate the overall benefit-risk relationship of the drug and to provide adequate information for the labeling of the drug. In most cases, the FDA requires two adequate and well-controlled Phase 3 clinical trials to demonstrate the efficacy and safety of the drug. A single Phase 3 or Phase 2 trial may be sufficient in rare instances, including when either (i) the trial is a large, multicenter trial demonstrating internal consistency and a statistically very persuasive finding of a clinically meaningful effect on mortality, irreversible morbidity or prevention of a disease with a potentially serious outcome and confirmation of the result in a second trial would be practically or ethically impossible or (ii) the trial is supported by other confirmatory evidence. Approval on the basis of a single trial may be subject to a requirement for additional post-approval studies.
The manufacturer of an investigational drug in a Phase 2 or Phase 3 clinical trial for a serious or life- threatening disease is required to disclose, such as by posting on its website, its policy on evaluating and responding to requests for expanded access to such investigational drug.
After completion of the required clinical testing, an NDA is prepared and submitted to the FDA. FDA approval of the NDA is required before marketing of the product may begin in the U.S. The NDA must include the results of all preclinical, clinical and other testing and a compilation of data relating to the product’s pharmacology, chemistry, manufacture and controls. The cost of preparing and submitting an NDA is substantial. Furthermore, under the Prescription Drug User Fee Act, or PDUFA, the submission of most NDAs is additionally subject to a substantial application user fee, and the applicant under an approved NDA is also subject to an annual program fee for each prescription product. These fees are typically increased annually. An NDA for a drug that has been designated as an orphan drug is not subject to an application fee, unless the NDA includes an indication for other than a rare disease or condition.
The FDA has 60 days from its receipt of an NDA to determine whether the application will be filed based on the agency’s threshold determination that it is sufficiently complete to permit substantive review. Once the submission is filed, the FDA begins an in-depth review. Under PDUFA, the FDA has agreed to certain performance goals in the review of NDAs to encourage timeliness. NDAs for most standard review drug products are reviewed within 12 months from submission of NDAs for new molecular entities, or NMEs, and ten months from submission of NDAs for non-NMEs. Priority review can be applied to drugs that the FDA determines offer major advances in treatment or provide a treatment where no adequate therapy exists. NDAs for most priority review drug products are reviewed within eight months from submission of NDAs for NMEs and six months from submission of NDAs for non-NMEs. The review process for both standard and priority review may be extended by the FDA for three additional months to consider certain late-submitted information or information intended to clarify information already provided in the NDA submission.
The FDA may also refer applications for novel drug products, or drug products that present difficult questions of safety or efficacy, to an outside advisory committee — typically a panel that includes clinicians, statisticians and other experts — for review, evaluation and a recommendation as to whether the application should be approved. The FDA is not bound by the recommendation of an advisory committee, but it generally follows such recommendations.
Before approving an NDA, the FDA will typically inspect one or more clinical sites to assure compliance with GCP. Additionally, the FDA will inspect the facility or the facilities at which the drug is manufactured. The FDA will not approve the product unless compliance with cGMPs is satisfactory and the NDA contains data that provide substantial evidence that the drug is safe and effective in the claimed indication.
After the FDA evaluates the NDA and completes clinical and manufacturing site inspections, it issues either an approval letter or a complete response letter. A complete response letter generally outlines the deficiencies in the NDA and may require substantial additional testing, or information, in order for the FDA to reconsider the application. The applicant may either resubmit the NDA, addressing all of the deficiencies identified in the letter, or withdraw the application. If, or when, those deficiencies have been addressed to the FDA’s satisfaction in a resubmission of the NDA, the FDA will issue an approval letter. The FDA has committed to reviewing such resubmissions in two or six months depending on the type of information included. An approval letter authorizes commercial marketing of the drug with specific prescribing information for specific indications. As a condition of NDA approval, the FDA may require a REMS to help ensure that the benefits of the drug outweigh the potential risks. REMS can include medication guides, communication plans for healthcare professionals, and elements to assure safe use, or ETASU. ETASU can include, but are not limited to, special training or certification for prescribing or dispensing, dispensing only under certain circumstances, special monitoring, and the use of patient registries. The requirement for a REMS can materially affect the potential market and profitability of the drug. Moreover, the FDA may require substantial post-approval testing and surveillance to monitor the drug’s safety or efficacy. Once granted, product approvals may be withdrawn if compliance with regulatory standards is not maintained, or problems are identified following initial marketing.
Once granted, product approvals may be withdrawn if compliance with regulatory standards is not maintained, or problems are identified following initial marketing. Changes to some of the conditions established in an approved application, including changes in indications, labeling, or manufacturing processes or facilities, require submission and the FDA approval of a new NDA or NDA supplement before the change can be implemented. An NDA supplement for a new indication typically requires clinical data similar to that in the original application, and the FDA uses the same procedures and actions in reviewing NDA supplements as it does in reviewing NDAs.
Special FDA Expedited Review and Approval Programs
Fast Track Designation
The FDA is required to facilitate the development, and expedite the review, of drugs that are intended for the treatment of a serious or life-threatening disease or condition for which there is no effective treatment, and which demonstrate the potential to address unmet medical needs for the disease. The FDA has various programs, including Fast Track Designation, priority review, accelerated approval, and Breakthrough Therapy Designation, the purpose of which is to provide important new drugs or biologics to patients earlier than under standard FDA review procedures.
Under the Fast Track program, the sponsor of a new drug candidate may request that the FDA designate the drug candidate for a specific indication as a Fast Track drug concurrent with, or after, the filing of the IND for the drug candidate. FDA must determine if the drug candidate qualifies for Fast Track Designation within 60 days of receipt of the sponsor’s request. Fast Track designation may not result in a faster development process, review or approval compared to conventional FDA procedures. The FDA may withdraw Fast Track designation if it believes that the designation is no longer supported by data from Palvella’s clinical development program.
If a development product is granted Fast Track Designation, the sponsor may engage in more frequent interactions with the FDA, and the FDA may review sections of the NDA before the application is complete. This rolling review is available if the applicant provides, and the FDA approves, a schedule for the submission of the remaining information and the applicant pays applicable user fees. However, the FDA’s time period goal for reviewing an application does not begin until the last section of the NDA is submitted.
The FDA may give a priority review designation to drugs that offer major advances in treatment or provide a treatment where no adequate therapy exists. A priority review means that the goal for the FDA to review an application is six months, rather than the standard review of ten months under current PDUFA guidelines. Under the new PDUFA agreement, these six- and ten-month review periods are measured from the “filing” date rather than the receipt date for NDAs or biologics license application, or BLAs, for new molecular entities, which typically adds approximately two months to the timeline for review and decision from the date of submission. Most products that are eligible for Fast Track Designation are also likely to be considered appropriate to receive a priority review.
In addition, products tested for their safety and effectiveness in treating serious or life-threatening illnesses and that provide meaningful therapeutic benefit over existing treatments may be eligible for accelerated approval and may be approved on the basis of adequate and well-controlled clinical trials establishing that the drug product has an effect on a surrogate endpoint that is reasonably likely to predict clinical benefit, or on a clinical endpoint that can be measured earlier than irreversible morbidity or mortality, or IMM, that is reasonably likely to predict an effect on irreversible morbidity or mortality or other clinical benefit, taking into account the severity, rarity or prevalence of the disease and the availability or lack of alternative treatments. As a condition of approval, the FDA may require a sponsor of a drug or biologic receiving accelerated approval to perform post-marketing studies to verify and describe the predicted effect on IMM or other clinical endpoint, and the drug or biologic may be subject to accelerated withdrawal procedures.
Orphan Drugs
Under the Orphan Drug Act, the FDA may grant Orphan Drug Designation to drugs intended to treat a rare disease or condition. This generally means a disease or condition that affects fewer than 200,000 individuals in the U.S. Orphan Drug Designation must be requested before submitting an NDA. After the FDA grants Orphan Drug Designation, the generic identity of the drug and its potential orphan use are disclosed publicly by the FDA. Orphan Drug Designation does not convey any advantage in, or shorten the duration of, the regulatory review and approval process. The first NDA applicant to receive FDA approval for a particular active ingredient to treat a particular disease with the FDA Orphan Drug Designation is entitled to a seven-year exclusive marketing period in the U.S. for that product, for that orphan indication. During the seven-year exclusivity period, the FDA may not approve any other applications to market the same drug for the same orphan disease, except in limited circumstances, such as a showing of clinical superiority to the product with orphan drug exclusivity. A product can be considered clinically superior if it is safer, more effective or makes a major contribution to patient care. Orphan drug exclusivity does not prevent the FDA from approving a different drug for the same disease or condition, or the same drug for a different disease or condition. Among the other benefits of Orphan Drug Designation are tax credits for certain research and an exemption from the NDA application user fee.
Breakthrough Therapy Designation
The Breakthrough Therapy Designation is a program by the FDA that aims to speed up the development and review of drugs and biologics for serious or life-threatening conditions. The designation is based on preliminary clinical evidence that the drug may provide a substantial improvement over current therapies.
To qualify for Breakthrough Therapy Designation, the drug must meet the following criteria:
|
● |
demonstrate a clear advantage over available therapies; |
|
● |
have an effect on irreversible morbidity or mortality, or “IMM”; |
|
● |
have an effect on symptoms that represent serious consequences of the disease; and |
|
● |
have a significantly improved safety profile compared to available therapies. |
Disclosure of Clinical Trial Information
Sponsors of clinical trials of FDA regulated products, including drugs, are required to register and disclose certain clinical trial information in the ClinicalTrials.gov database. Information related to the product, patient population, phase of investigation, study sites and investigators, and other aspects of the clinical trial is then made public as part of the registration. Sponsors are also obligated to disclose the results of their clinical trials after completion. Disclosure of the results of these trials can be delayed in certain circumstances for up to two years after the date of completion of the trial. Competitors may use this publicly available information to gain knowledge regarding the progress of development programs as well as clinical trial design.
Pediatric Information
Under the Pediatric Research Equity Act, or PREA, NDAs or supplements to NDAs must contain data to assess the safety and effectiveness of the drug for the claimed indications in all relevant pediatric subpopulations and to support dosing and administration for each pediatric subpopulation for which the drug is safe and effective. The FDA may grant full or partial waivers, or deferrals, for submission of data. With certain exceptions, PREA does not apply to any drug for an indication for which orphan designation has been granted.
The Best Pharmaceuticals for Children Act, or BPCA, provides NDA holders a six-month extension of any exclusivity — patent or nonpatent — for a drug if certain conditions are met. Conditions for exclusivity include the FDA’s determination that information relating to the use of a new drug in the pediatric population may produce health benefits in that population, the FDA making a written request for pediatric studies, and the applicant agreeing to perform, and reporting on, the requested studies within the statutory timeframe. Applications under the BPCA are treated as priority applications, with all of the benefits that designation confers.
Post-Approval Requirements
Once an NDA is approved, a product will be subject to certain post-approval requirements, including, among other things, record-keeping requirements, providing the FDA with updated safety information, product sampling and distribution requirements, and promotion and advertising requirements. For instance, the FDA closely regulates the post-approval marketing and promotion of drugs, including standards and regulations for direct-to-consumer advertising, off-label promotion, industry-sponsored scientific and educational activities and promotional activities involving the internet. Drugs may be marketed or promoted only for the approved indications and in accordance with the provisions of the approved labeling.
Adverse event reporting and submission of periodic reports are required following FDA approval of an NDA. The FDA also may require post-marketing testing, known as Phase 4 testing, REMS, and surveillance to monitor the effects of an approved product, or the FDA may place conditions on an approval that could restrict the distribution or use of the product. In addition, quality control, drug manufacture, packaging and labeling procedures must continue to conform to cGMPs after approval. Drug manufacturers and certain of their subcontractors are required to register their establishments with the FDA and certain state agencies. Registration with the FDA subjects entities to periodic unannounced inspections by the FDA, during which the agency inspects manufacturing facilities to assess compliance with cGMPs. Accordingly, manufacturers must continue to expend time, money, and effort in the areas of production and quality-control to maintain compliance with cGMPs. Regulatory authorities may withdraw product approvals, request product recalls, or take other administrative or judicial enforcement actions if a company fails to comply with regulatory standards, if it encounters problems following initial marketing, or if previously unrecognized problems are subsequently discovered.
The Hatch-Waxman Amendments
Orange Book Listing
NDA applicants are required to list with the FDA each patent whose claims cover the applicant’s drug or approved method of using the drug. Upon approval of a drug, each of the patents listed in the application for the drug is then published in the FDA’s Approved Drug Products with Therapeutic Equivalence Evaluations, commonly known as the Orange Book. Drugs listed in the Orange Book can, in turn, be referenced by potential generic competitors in support of approval of an ANDA. An ANDA provides for marketing of a drug product that has the same active ingredients, strengths, and routes of administration in the same strengths and dosage form as the listed drug and has been shown through bioequivalence testing to be therapeutically equivalent to the listed drug. Other than the requirement for bioequivalence testing, ANDA applicants are not required to conduct, or submit results of, preclinical or clinical tests to prove the safety or effectiveness of their drug product. Drugs approved in this way are commonly referred to as “generic equivalents” to the listed drug and can often be substituted by pharmacists under prescriptions written for the original listed drug pursuant to each state’s laws on drug substitution.
The ANDA applicant is required to certify to the FDA concerning any patents listed for the approved product in the FDA’s Orange Book. Specifically, the applicant must certify to each patent in one of the following ways: (i) the required patent information has not been filed; (ii) the listed patent has expired; (iii) the listed patent has not expired but will expire on a particular date and approval is sought after patent expiration; or (iv) the listed patent is invalid, unenforceable or will not be infringed by the new product. The ANDA applicant may also elect to submit a section viii statement certifying that its proposed ANDA label does not contain (or carve out) any language regarding the patented method-of-use rather than certify to a listed method-of-use patent. If the applicant does not challenge the listed patents, the ANDA application will not be approved until all the listed patents claiming the referenced product have expired. A certification that the new product will not infringe the already approved product’s listed patents, or that such patents are invalid or unenforceable is called a Paragraph IV certification. If the ANDA applicant has provided a Paragraph IV certification to the FDA, the applicant must also send notice of the Paragraph IV certification to the NDA and patent holders once the ANDA has been received for filing by the FDA. The NDA and patent holders may then initiate a patent infringement lawsuit in response to the notice of the Paragraph IV certification. The filing of a patent infringement lawsuit within 45 days of the receipt of a Paragraph IV certification automatically prevents the FDA from approving the ANDA until the earlier of 30 months, expiration of the patent, settlement of the lawsuit, or a decision in the infringement case that is favorable to the ANDA applicant.
The ANDA application also will not be approved until any applicable non-patent exclusivity listed in the Orange Book for the referenced product has expired.
Exclusivity
Upon NDA approval of a drug that contains an active moiety that has been previously approved by the FDA in any other NDA, when the application contains reports of a new clinical investigations (other than bioavailability studies) that were essential to approval, that drug product receives three years of exclusivity. During this three-year period of exclusivity, the FDA may not approve any Section 505(b)(2) NDA or ANDA seeking approval of a version of that drug that includes the same conditions of approval.
Section 505(b)(2) NDAs
A special type of NDA, commonly referred to as a Section 505(b)(2) NDA, enables the applicant in certain circumstances to rely, in part, on the FDA’s prior findings in approving a similar product or published literature in support of its application. A Section 505(b)(2) NDA may provide an alternate path to FDA approval for a new or improved formulation, a new route of administration, or a new use of a previously approved product.
Section 505(b)(2) permits the submission of an NDA where at least some of the information required for approval comes from studies not conducted by, or for, the applicant and for which the applicant has not obtained a right of reference. If the Section 505(b)(2) applicant can establish that reliance on the FDA’s prior findings of safety and/or effectiveness is scientifically appropriate, it may eliminate the need to conduct certain preclinical or clinical studies of the new product. The FDA may also require companies to perform additional studies or measurements to support the change from the approved product. The FDA may then approve the new product candidate for all, or some, of the indications for which the referenced product has been approved, as well as for any new indication sought by the Section 505(b)(2) applicant. If Palvella chooses to rely on the Section 505(b)(2) NDA process to seek approval for its product candidates in various indications, there can be no assurance that the FDA will agree with its use of that pathway. See “Risk Factors—Risks Related to the Development and Regulatory Approval of Palvella’s Product Candidates— Palvella’s development and commercialization strategy for its product candidates depends, in part, on published scientific literature and the FDA’s prior findings regarding the safety and efficacy of rapamycin. If Palvella is not able to pursue this strategy, it may be delayed in receiving regulatory authority approval.”
To the extent that the Section 505(b)(2) applicant is relying on the FDA’s prior findings of safety or effectiveness for an already approved product, the applicant is required to certify to the FDA concerning any patents listed for the approved product in the Orange Book to the same extent that an ANDA applicant would. Thus, approval of a Section 505(b)(2) NDA can be stalled until all the listed patents claiming the referenced product have expired, until any non-patent exclusivity, such as exclusivity for obtaining approval of a new chemical entity, listed in the Orange Book for the referenced product has expired, and, in the case of a Paragraph IV certification and subsequent patent infringement suit, until the earlier of 30 months, settlement of the lawsuit or a decision in the infringement case that is favorable to the Section 505(b)(2) applicant.
Combination Products
A combination product is a product comprised of two or more regulated components, e.g., drug and medical device, that are physically combined and produced as a single entity, packaged together in a single package, or packaged separately but intended to be labeled for use together.
The FDA is divided into various branches, or Centers, by product type. Different Centers typically review drug, biologic, or device applications. In order to review an application for a combination product, the FDA must decide which Center should be responsible for the review. FDA regulations require that the FDA determine the combination product’s primary mode of action, or PMOA, which is the single mode of a combination product that provides the most important therapeutic action of the combination product. The Center that regulates that portion of the product that generates the PMOA becomes the lead evaluator. If there are two independent modes of action, neither of which is subordinate to the other, the FDA makes a determination as to which Center to assign the product based on consistency with other combination products raising similar types of safety and effectiveness questions or to the Center with the most expertise in evaluating the most significant safety and effectiveness questions raised by the combination product. When evaluating an application, a lead Center may consult other Centers but still retain complete reviewing authority, or it may collaborate with another Center, by which the Center assigns review of a specific section of the application to another Center, delegating its review authority for that section. Typically, the FDA requires a single marketing application submitted to the Center selected to be the lead evaluator, although the agency has the discretion to require separate applications to more than one Center.
Medical Device Products
A medical device is an instrument, apparatus, implement, machine, contrivance, implant, in vitro reagent, or other similar or related article, including any component part, or accessory which is: (i) recognized in the official National Formulary, or the U.S. Pharmacopoeia, or any supplement to them; (ii) intended for use in the diagnosis of disease or other conditions, or in the cure, mitigation, treatment, or prevention of disease, in man or other animals; or (iii) intended to affect the structure or any function of the body of man or other animals, and which does not achieve any of its primary intended purposes through chemical action within or on the body of man or other animals and which is not dependent upon being metabolized for the achievement of any of its primary intended purposes.
The FDCA classifies medical devices into one of three categories based on the risks associated with the device and the level of control necessary to provide reasonable assurance of safety and effectiveness. Class I devices are deemed to be low risk and are subject to the fewest regulatory controls. Class II devices are subject to the FDA’s general controls, and any other special controls as deemed necessary by the FDA to provide reasonable assurance of the safety and effectiveness of the devices. Class III devices are generally the highest risk devices and are subject to the highest level of regulatory control to provide reasonable assurance of the device’s safety and effectiveness. Class III devices must typically be approved by the FDA before they are marketed.
Most medical devices can be legally sold within the United States only if the FDA has: (i) approved a premarket approval application, or PMA, prior to marketing, generally applicable to Class III devices; or (ii) cleared the device in response to a premarket notification, or 510(k) submission, generally applicable to Class I and II devices. However, most Class I and some Class II devices can be marketed without prior FDA authorization. If a device falls into a generic category of Class I or Class II devices that the FDA has exempted by regulation, a premarket notification is not required before marketing the device in the United States. Some 510(k)-exempt devices are also exempt from Quality System Regulation, or QSR, requirements.
After a device is placed on the market, numerous regulatory requirements apply. These include: the QSR, labeling regulations, the FDA’s general prohibition against promoting products for unapproved or “off-label” uses, the Medical Device Reporting regulation (which requires that manufacturers report to the FDA if their device may have caused or contributed to a death or serious injury or malfunctioned in a way that would likely cause or contribute to a death or serious injury if it were to recur), and the Reports of Corrections and Removals regulation (which requires manufacturers to report recalls and field actions to the FDA if initiated to reduce a risk to health posed by the device or to remedy a violation of the FDCA).
The FDA enforces these requirements by inspection and market surveillance. If the FDA finds a violation, it can institute a wide variety of enforcement actions, ranging from a public warning letter to more severe sanctions such as: fines, injunctions, and civil penalties; recall or seizure of products; operating restrictions, partial suspension or total shutdown of production; refusing requests for 510(k) clearance or PMA approval of new products; withdrawing 510(k) clearance or PMA approvals already granted; and criminal prosecution.
U.S. Anti-kickback, False Claims and Other Healthcare Fraud and Abuse Laws
In the United States, there are federal and state anti-kickback laws that prohibit offering, the payment, solicitation, or receipt of kickbacks, bribes or other remuneration intended to induce the purchase or recommendation of healthcare products and services. Violations of these laws can lead to civil and criminal penalties, including exclusion from participation in federal healthcare programs. These laws apply to manufacturers of products, such as Palvella, with respect to Palvella’s financial relationship with hospitals, physicians and other potential purchasers or acquirers of Palvella’s products. The U.S. government has published regulations that identify “safe harbors” or exemptions for certain practices from enforcement actions under the federal anti-kickback statute, and Palvella will seek to comply with the safe harbors where possible. To qualify for a safe harbor, the activity must fit squarely within the safe harbor. Arrangements that do not meet a safe harbor are not necessarily illegal but must be evaluated on a case-by-case basis. A person or entity may be found to violate the anti-kickback statute even absent actual knowledge of this statute or specific intent to violate it. In addition, the government may assert that a claim that includes items or services resulting from a violation of the federal anti-kickback statute constitutes a false or fraudulent claim for purposes of the federal FCA.
The civil FCA prohibits, among other things, any person or entity from knowingly presenting, or causing to be presented, a false or fraudulent claim for payment to, or approval by, the federal government, knowingly making, using, or causing to be made or used a false record or statement material to a false or fraudulent claim to the federal government, or avoiding, decreasing, or concealing an obligation to pay money to the federal government. A claim includes “any request or demand” for money or property presented to the U.S. government. The civil FCA has been used to assert liability on the basis of kickbacks and other improper referrals, improper use of Medicare provider or supplier numbers when detailing a provider of services, improper promotion of off-label uses not covered by a device’s clearance or approval, and allegations as to misrepresentations with respect to products, contract requirements, and services rendered. In addition, private payors have been filing follow-on lawsuits alleging fraudulent misrepresentation, although establishing liability and damages in these cases is more difficult than under the FCA. Intent to deceive is not required to establish liability under the civil FCA. Civil FCA actions may be brought by the government or may be brought by private individuals on behalf of the government, called “qui tam” actions. If the government decides to intervene in a qui tam action and prevails in the lawsuit, the individual will share in the proceeds from any fines or settlement funds. If the government declines to intervene, the individual may pursue the case alone. The civil FCA provides for treble damages and a civil penalty for each false claim, such as an invoice or pharmacy claim for reimbursement, which can aggregate into millions of dollars. For these reasons, FCA lawsuits against biopharmaceutical and device companies have increased significantly in volume and breadth, leading to several substantial civil and criminal settlements, as much as $3.0 billion, regarding certain sales practices and promoting off label uses. Civil FCA liability may further be imposed for known Medicare or Medicaid overpayments that are not refunded within 60 days of discovering the overpayment, even if the overpayment was not caused by a false or fraudulent act. In addition, conviction or civil judgment for violating the FCA may result in exclusion from federal health care programs, and suspension and debarment from government contracts, and refusal of orders under existing government contracts.
The government may further prosecute conduct constituting a false claim under the criminal FCA. The criminal FCA prohibits the making or presenting of a claim to the government knowing such claim to be false, fictitious, or fraudulent and, unlike the civil FCA, requires proof of intent to submit a false claim.
The civil monetary penalties statute is another statute under which medical device companies may potentially be subject to enforcement. Among other things, the civil monetary penalties statue imposes fines against any person who offers to provide remuneration to any individual eligible for benefits under Medicare or Medicaid that the offeror knows or should know is likely to influence the individual to order or receive from a particular provider or supplier of any item or service reimbursable under those programs.
The federal HIPAA also created federal criminal statutes that prohibit, among other actions, knowingly and willfully executing, or attempting to execute, a scheme to defraud or to obtain, by means of false or fraudulent pretenses, representations or promises, any of the money or property owned by, or under the custody or control of, a healthcare benefit program, regardless of whether the payor is public or private, in connection with the delivery or payment for health care benefits, knowingly and willfully embezzling or stealing from a health care benefit program, willfully obstructing a criminal investigation of a health care offense and knowingly and willfully falsifying, concealing, or covering up by any trick or device a material fact or making any materially false statements in connection with the delivery of, or payment for, healthcare benefits, items, or services relating to healthcare matters. Additionally, the Affordable Care Act amended the intent requirement of certain of these criminal statutes under HIPAA so that a person or entity no longer needs to have actual knowledge of the statute, or the specific intent to violate it, to have committed a violation.
The Sunshine Act requires annual reporting, by applicable device and drug manufacturers, of covered products, payments, and other transfers of value to certain health care providers, and ownership and investment interests held by physicians and their immediate family members.
Further, Palvella may be subject to data privacy and security regulation by both the federal government and the states in which Palvella conducts its business. HIPAA, as amended by the HITECH and its respective implementing regulations imposes certain requirements on covered entities relating to the privacy, security, and transmission of certain individually identifiable health information, known as protected health information. Among other things, HITECH, through its implementing regulations, makes HIPAA’s security standards and certain privacy standards directly applicable to business associates, defined as a person or organization, other than a member of a covered entity’s workforce, that creates, receives, maintains, or transmits protected health information on behalf of a covered entity for a function or activity regulated by HIPAA. HITECH also strengthened the civil and criminal penalties that may be imposed against covered entities, business associates, and individuals, and gave state attorneys general new authority to file civil actions for damages or injunctions in federal courts to enforce the federal HIPAA laws and seek attorneys’ fees and costs associated with pursuing federal civil actions. In addition, other federal and state laws may govern the privacy and security of health and other information in certain circumstances, many of which differ from each other in significant ways and may not be preempted by HIPAA, thus complicating compliance efforts.
Many states have also adopted laws similar to each of the above federal laws, which may be broader in scope and apply to items or services reimbursed by any third-party payor, including commercial insurers. Certain states also require implementation of commercial compliance programs and compliance with the medical device industry’s voluntary compliance guidelines and the applicable compliance guidance promulgated by the federal government, or otherwise restrict payments or the provision of other items of value that may be made to healthcare providers and other potential referral sources; impose restrictions on marketing practices; or require companies to track and report information related to payments, and other items of value to physicians and other healthcare providers.
If Palvella’s operations are found to be in violation of any of the laws or regulations described above or any other applicable laws, Palvella may be subject to penalties or other enforcement actions, including criminal and significant civil monetary penalties, damages, fines, disgorgement, imprisonment, exclusion from participation in government healthcare programs, corporate integrity agreements, suspension and debarment from government contracts, and refusal of orders under existing government contracts, reputational harm, diminished profits and future earnings, and the curtailment or restructuring of Palvella’s operations, any of which could adversely affect Palvella’s ability to operate its business and its results of operations. Enforcement actions can be brought by federal or state governments, or as “qui tam” actions brought by individual whistleblowers in the name of the government under the civil FCA if the violations are alleged to have caused the government to pay a false or fraudulent claim.
To the extent that any of Palvella’s products are sold in a foreign country, Palvella may be subject to similar foreign laws and regulations, which may include, for instance, applicable post-marketing requirements, including safety surveillance, anti-fraud and abuse laws, and implementation of corporate compliance programs and reporting of payments or transfers of value to healthcare professionals.
Foreign Corrupt Practices Act
The FCPA, generally prohibits offering, promising, giving, or authorizing others to give anything of value, either directly or indirectly, to a non-U.S. government official in order to influence official action, or otherwise obtain or retain business. The FCPA also requires public companies to make and keep books and records that accurately and fairly reflect the transactions of the corporation and to devise and maintain an adequate system of internal accounting controls. Palvella’s industry is heavily regulated and therefore involves significant interaction with public officials, including officials of non-U.S. governments. Additionally, in many other countries, the health care providers who prescribe pharmaceuticals and medical devices are employed by their government, and the purchasers are government entities; therefore, Palvella’s dealings with these prescribers and purchasers are subject to regulation under the FCPA. The SEC and DOJ have increased their FCPA enforcement activities with respect to pharmaceutical and medical device companies. Violations could result in fines, criminal sanctions against Palvella, its officers, or its employees, the closing down of Palvella’s facilities, requirements to obtain export licenses, cessation of business activities in sanctioned countries, implementation of compliance programs, and prohibitions on the conduct of Palvella’s business. Enforcement actions may be brought by the DOJ and SEC, and legislation has expanded the SEC’s power to seek disgorgement in all FCPA cases filed in federal court and extended the statute of limitations in SEC enforcement actions in intent-based claims such as those under the FCPA from five years to ten years.
Coverage and Reimbursement
Palvella’s ability to successfully commercialize any approved product candidates will depend in part on the extent to which governmental payor programs at the federal and state levels, including Medicare and Medicaid, private health insurers and other third-party payors provide coverage for and establish adequate reimbursement levels. However, decisions regarding the extent of coverage and amount of reimbursement to be provided are made on a payor-by-payor basis. Government health administration authorities, private health insurers and other organizations generally decide which drugs they will pay for and establish reimbursement levels for healthcare. In particular, in the United States, private health insurers and other third-party payors often provide reimbursement for products and services based on the level at which the government provides reimbursement through the Medicare or Medicaid programs for such treatments. In the United States, the European Union and other potentially significant markets for Palvella’s product candidates, government authorities and third-party payors are increasingly attempting to limit or regulate the price of medical products and services, particularly for new and innovative products and therapies, which often has resulted in average selling prices lower than they would otherwise be. Further, the increased emphasis on managed healthcare in the United States and on country and regional pricing and reimbursement controls in the European Union will put additional pressure on product pricing, reimbursement and usage, which may adversely affect Palvella’s future product sales and results of operations. These pressures can arise from rules and practices of managed care groups, judicial decisions and governmental laws and regulations related to Medicare, Medicaid and healthcare reform, pharmaceutical coverage and reimbursement policies and pricing in general.
Third-party payors are increasingly imposing additional requirements and restrictions on coverage and limiting reimbursement levels for medical products. For example, in the United States, federal and state governments reimburse covered prescription drugs at varying rates generally below average wholesale price. These restrictions and limitations influence the purchase of healthcare services and products. Third party payors may limit coverage to specific drug products on an approved list, or formulary, which might not include all of the FDA approved drug products for a particular indication. Third-party payors are increasingly challenging the price and examining the medical necessity and cost effectiveness of medical products and services, in addition to their safety and efficacy. Palvella may need to conduct expensive pharmacoeconomic studies in order to demonstrate the medical necessity and cost effectiveness of Palvella’s products, in addition to the costs required to obtain FDA approvals. Palvella’s product candidates may not be considered medically necessary or cost effective. A payor’s decision to provide coverage for a drug product does not imply that an adequate reimbursement rate will be approved. Adequate third-party reimbursement may not be available to enable Palvella to maintain price levels sufficient to realize an appropriate return on Palvella’s investment in drug development.
The IRA contains substantial drug pricing reforms, including the establishment of a drug price negotiation program within the HHS that would require manufacturers to charge a negotiated “maximum fair price” for certain selected drugs or pay an excise tax for noncompliance, the establishment of rebate payment requirements on manufacturers of certain drugs payable under Medicare Parts B and D to penalize price increases that outpace inflation, and requires manufacturers to provide discounts on Part D drugs. Substantial penalties can be assessed for noncompliance with the drug pricing provisions. Although the IRA exempts orphan drugs that treat only one rare disease from the drug pricing negotiation provisions, Palvella does not know if additional drug pricing reforms could eliminate this exemption and therefore affect the prices Palvella can charge and reimbursement Palvella receives for its product candidates, if approved, thereby reducing Palvella’s profitability. The effect of IRA on Palvella’s business and the pharmaceutical industry in general is not yet known.
Future efforts to reform healthcare or reduce costs under government insurance programs may result in lower reimbursement for Palvella’s products and product candidates or exclusion of Palvella’s products and product candidates from coverage. The cost containment measures that healthcare payors and providers are instituting and any healthcare reform could significantly reduce Palvella’s revenue from the sale of any approved product candidates. Palvella cannot provide any assurances that Palvella will be able to obtain and maintain third-party coverage or adequate reimbursement for Palvella’s product candidates in whole or in part.
Healthcare Reform
The United States and many foreign jurisdictions have enacted or proposed legislative and regulatory changes affecting the healthcare system that could restrict or regulate post-approval activities relating to Palvella’s product candidates, if approved, and affect Palvella’s ability to successfully commercialize its product candidates, if approved, including implementing cost-containment programs to limit the growth of government-paid healthcare costs, including price controls, restrictions on reimbursement and requirements for substitution of generic products for branded prescription drugs.
There have been significant ongoing judicial, administrative, executive and legislative efforts by the federal government, state governments, regulators and third-party payors to control or manage the increased costs of health care and, more generally, to reform the U.S. healthcare system. The pharmaceutical industry has been a particular focus of these efforts and has been significantly affected by major legislative initiatives. For example, in March 2010, the Affordable Care Act was enacted, which was intended to broaden access to health insurance, reduce or constrain the growth of healthcare spending, enhance remedies against fraud and abuse, add new transparency requirements for the healthcare and health insurance industries, impose new taxes and fees on the health industry and impose additional health policy reforms.
Several healthcare reform proposals culminated in the enactment of the IRA in August 2022, which will eliminate, beginning in 2025, the coverage gap under Medicare Part D by significantly lowering the enrollee maximum out-of-pocket cost and requiring manufacturers to subsidize, through a newly established manufacturer discount program, 10% of Part D enrollees’ prescription costs for brand drugs below the out-of-pocket maximum, and 20% once the out-of-pocket maximum has been reached. Among other things, the IRA also requires HHS to negotiate the selling price of a statutorily specified number of drugs and biologics each year that CMS reimburses under Medicare Part B and Part D. Only high-expenditure single-source drugs that have been approved for at least 7 years (11 years for biologics) are eligible to be selected by CMS for negotiation, with the negotiated price taking effect two years after the selection year. Negotiations for Medicare Part D products began in 2024 with the negotiated price taking effect in 2026, and negotiations for Medicare Part B products begin in 2026 with the negotiated price taking effect in 2028. In August 2023, HHS announced the ten Medicare Part D drugs and biologics that it selected for negotiations and has since announced the negotiated maximum fair prices for these drugs. This price cap, which cannot exceed a statutory ceiling price, will come into effect on January 1, 2026, and will represent a significant discount from average prices to wholesalers and direct purchasers. The IRA also imposes rebates on Medicare Part B and Part D drugs whose prices have increased at a rate greater than the rate of inflation. The IRA permits the Secretary of HHS to implement many of these provisions through guidance, as opposed to regulation, for the initial years. Manufacturers that fail to comply with the IRA may be subject to various penalties, including civil monetary penalties. These provisions may be subject to legal challenges. For example, the provisions related to the negotiation of selling prices of high-expenditure single-source drugs and biologics have been challenged in multiple lawsuits brought by pharmaceutical manufacturers. The outcome of these lawsuits is uncertain. Thus, while it is unclear how the IRA will be implemented, it will likely have a significant impact on the pharmaceutical industry and the pricing of prescription drug products.
Palvella expects that the Affordable Care Act, the IRA, as well as other healthcare reform measures that have and may be adopted in the future, may result in more rigorous coverage criteria and in additional downward pressure on the price that Palvella receives for its product candidates, if approved, and could seriously harm Palvella’s future revenues. Any reduction in reimbursement from Medicare, Medicaid, or other government programs may result in a similar reduction in payments from private payers. The implementation of cost containment measures or other healthcare reforms may prevent Palvella from being able to generate revenue, attain profitability or successfully commercialize Palvella’s product candidates, if approved.
Foreign Regulatory Requirements
Palvella may be subject to widely varying foreign regulations, which may be quite different from those of the FDA, governing clinical trials, manufacturing, product registration and approval, pharmaceutical sales and data protection. Whether or not FDA approval has been obtained, Palvella must obtain a separate approval for a product by the comparable regulatory authorities of foreign countries prior to the commencement of product marketing in these countries. In certain countries, regulatory authorities also establish pricing and reimbursement criteria. The approval process varies from country to country, and the time may be longer or shorter than that required for FDA approval. Regulatory approval in one country does not ensure regulatory approval in another, but a failure or delay in obtaining regulatory approval in one country may negatively impact the regulatory process in others.
Palvella’s principal executive office is located in Wayne, Pennsylvania, where it leases 3,379 square feet of space that it uses for its administrative, research and development and other activities. Palvella believes that its existing facilities are adequate for its near-term needs, but expects to need additional space as it grows. Palvella believes that suitable additional or alternative space would be available as required in the future on commercially reasonable terms.
Employees and Human Capital Resources
As of October 28, 2024, Palvella had nine full-time employees, of which one has a Ph.D. None of Palvella’s employees are represented by a labor union or covered by collective bargaining agreements, and Palvella believes its relationship with its employees is good.
Palvella’s human capital resources objectives include, as applicable, identifying, recruiting, retaining, incentivizing, and integrating its existing and additional employees. The principal purposes of Palvella’s equity incentive plans are to attract, retain and motivate selected employees and directors through the granting of stock-based compensation awards and cash-based performance bonus awards.
From time to time, Palvella may become involved in legal proceedings or be subject to claims arising in the ordinary course of its business. Palvella is not currently subject to any material legal proceedings.
PIERIS MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The following discussion and analysis provides information which Pieris’ management believes is relevant to an assessment and understanding of Pieris’ results of operations and financial condition. This discussion and analysis should be read together with the section of this proxy statement/prospectus entitled Pieris’ audited and unaudited financial statements and related notes that are included elsewhere in this proxy statement/prospectus. In addition to historical information, this discussion and analysis contains forward-looking statements that involve risks, uncertainties and assumptions. Pieris’ actual results may differ materially from those anticipated in these forward-looking statements as a result of certain factors, including but not limited to those set forth under the caption “Risk Factors” set forth in this proxy statement/prospectus, Pieris’ Annual Report on Form 10-K for the fiscal year ended December 31, 2023, and those included in Pieris’ Quarterly Reports on Form 10-Q for the quarters ended March 31, 2024 and June 30, 2024.
Business Overview
Pieris is a biotechnology company that historically discovered and developed Anticalin® protein-based drugs to target validated disease pathways in unique and transformative ways. Proprietary to Pieris, Anticalin proteins are a novel class of therapeutics validated in the clinic and through partnerships with leading pharmaceutical companies, including Pfizer (formerly Seagen) and Boston Pharmaceuticals in IO. Pieris’ clinical pipeline consists of IO bispecifics in partnership with collaborators, including SGN-BB228 (also referred to as PRS-346) targeting CD228 and 4-1BB and BOS-342 (also referred to as PRS-342) targeting GPC3 and 4-1BB.
On March 27, 2024, Pieris announced an update on its review of strategic alternatives, and its decision to implement measures that are expected to extend its cash runway into at least 2027, while maximizing its ability to collect potential milestones from its clinical pipeline of partnered drug candidates, potentially obtain value for cinrebafusp alfa and other proprietary platform capabilities, and consider other strategic opportunities. As part of this strategy, Pieris discontinued all of its research and development efforts, reduced its workforce, which affected additional employees, including the executive leadership. Pieris remains eligible to receive potential contingent milestone and royalty payments from its partnered 4-1BB bispecific Mabcalin protein franchise from Pfizer and Boston Pharmaceuticals. These include aggregated milestones of approximately $15.0 million in connection with dosing a first patient in the Phase 2 trials for SGN-BB228 and BOS-342, and aggregated milestones of approximately $40.0 million in connection with dosing a first patient in the pivotal clinical trials for SGN-BB228 and BOS-342. This follows from Pieris’ July 2023 announcement where Pieris stated its intention to explore one or more strategic transactions with the assistance of Pieris’ financial advisor, Stifel, and announced a reduction in its workforce by approximately 70% due to its decision to opt out of and terminate programs, thus reducing its operating footprint and expenses.
Discovery and Development Programs
Pieris currently has several IO drug candidates partnered with major biopharmaceutical companies, which are at varying stages of development:
|
● |
Pieris’ IO partnered portfolio includes the following drug candidates that are multi-specific Anticalin-based fusion protein drug candidates designed to engage immunomodulatory targets, in partnership with Pfizer (formerly Seagen) and Boston Pharmaceutics; |
|
● |
in the Pfizer collaboration, SGN-BB228 (also referenced as PRS-346), a CD228 x 4-1BB bispecific antibody-Anticalin compound, was previously handed over to Pfizer, which is responsible for further advancement and funding of the asset. In January 2023, the first patient was dosed in a Pfizer-sponsored Phase 1 study of SGN-BB228, upon which Pieris achieved a $5.0 million milestone. Pfizer presented preclinical data for this program at the Society for Immunotherapy of Cancer 37th Annual Meeting in November 2022 and at the American Association for Cancer Research (AACR) Annual Meeting in April 2023. Pfizer presented the study design of the Phase 1 study of SGN-BB228 at the American Society of Clinical Oncology (ASCO) Annual Meeting in June 2023. The program is one of three programs in the Pfizer alliance, and Pieris believes the previous achievement of a key development milestone for SGN-BB228 validates its approach in IO bispecifics, complementing the encouraging clinical data seen with cinrebafusp alfa. Pieris transferred the second and third programs to Pfizer at the end of 2023, and retains a co-promotion option for one program in the Pfizer collaboration in the United States; and |
|
● |
BOS-342 (also referenced as PRS-342) is a GPC3 x 4-1BB bispecific Mabcalin compound that Pieris has exclusively licensed to Boston Pharmaceuticals. In August 2023, the first patient was dosed in a Boston Pharmaceuticals sponsored Phase 1/2 study of BOS-342 in hepatocellular carcinoma (HCC), for which Pieris received a $2.5 million milestone payment and is entitled to receive up to approximately $350 million in potential development, regulatory and sales-based milestone payments, and tiered royalties on potential sales of BOS-342. |
Pieris’ former drug candidates include:
|
● |
Cinrebafusp alfa is designed to drive tumor localized T cell activation through tumor-targeted drug clustering mediated by HER2 expressed on tumor cells. This program was the first 4-1BB bispecific T cell co-stimulatory agonist to enter clinical development. In July 2022, Pieris received Fast Track Designation from FDA for cinrebafusp alfa. In August 2022, Pieris announced the decision to cease further enrollment in the two-arm, multicenter, open-label Phase 2 study of cinrebafusp alfa as part of a strategic pipeline prioritization to focus Pieris’ resources. Cinrebafusp alfa has demonstrated clinical benefit in Phase 1 studies, including single agent activity in a monotherapy setting, and in the Phase 2 study in HER2-expressing gastric cancer, giving Pieris confidence in its broader 4-1BB franchise. In April 2023, clinical data showing an unconfirmed 100% objective response rate and promising emerging durability profile were presented at the American Association of Cancer Research annual meeting. This data provided encouraging evidence of clinical activity for this program; |
|
● |
S095012 (also referenced as PRS-344) is a bispecific Mabcalin compound comprising a PD-L1-targeting antibody genetically linked to 4-1BB-targeting Anticalin proteins being developed by Servier on a worldwide basis. The first-in-human Phase 1/2 multicenter open-label dose escalation study was designed to determine the safety and preliminary activity of S095012 in patients with advanced and/or metastatic solid tumors. In July 2023, Pieris notified Servier that it was opting out of co-development and commercialization of S095012 in the United States. Servier retained exclusive, even as to Pieris, worldwide rights to the program including the right to advance development and potential commercialization in the United States. On June 28, 2024, Servier provided Pieris with a written notice of termination of the License and Collaboration Agreement between Servier, Pieris, and Pieris Pharmaceuticals GmbH, dated January 4, 2017, as subsequently amended, or the Collaboration Agreement. Pursuant to Section 7.1 of the Non-Exclusive Anticalin® Platform Technology License Agreement, between Servier, Pieris, and Pieris Pharmaceuticals GmbH, dated January 4, 2017, or the Non-Exclusive License Agreement, the Non-Exclusive License Agreement terminates upon termination of the Collaboration Agreement. The Collaboration Agreement and Non-Exclusive License Agreement will terminate effective December 27, 2024, or 180 days from the date on which Servier notified Pieris of its intent to terminate each agreement; |
|
● |
Elarekibep, a former respiratory program that was partnered with AstraZeneca for the treatment of asthma, was a drug candidate that antagonizes IL-4Rα, thereby inhibiting the downstream action of IL-4 and IL-13, two cytokines known to be key mediators in the inflammatory cascade that drive the pathogenesis of asthma and other inflammatory diseases; |
|
○ |
in June 2023, AstraZeneca communicated to Pieris its decision to discontinue and cease dosing in the Phase 2a clinical studies of elarekibep. This decision was based on lung findings from a non-clinical 13-week GLP toxicology study with dry powder inhaler-formulated elarekibep, which did not support long-term use and progression to later-stage development. The 13-week non-human primates study included three active dose cohorts. AstraZeneca concluded that there were no clinical observations across any of the doses but that there were respiratory tract pathology findings. These findings included inflammation-mediated lung tissue damage, which did not appear to be dose related. AstraZeneca’s decision was made independent of any data from the Phase 2a study; |
|
○ |
in July 2023, AstraZeneca notified Pieris of its intention to terminate the AstraZeneca Collaboration Agreement and the AstraZeneca Platform License, which terminations became effective October 15, 2023. AstraZeneca’s decision to terminate these agreements was based on the non-clinical safety findings in a 13-week toxicology study of elarekibep in non-human primates previously disclosed by Pieris. Based upon Pieris’ review, Pieris determined to discontinue the program for scientific reasons; |
|
● |
PRS-220, an orally inhaled Anticalin protein targeting connective tissue growth factor, or CTGF, was being developed as a local treatment for idiopathic pulmonary fibrosis, or IPF, and other forms of fibrotic lung diseases. CTGF, a matricellular protein, has been demonstrated to be a driver of fibrotic tissue remodeling and the protein has been found over-expressed in lung tissue from patients suffering from IPF; |
|
○ |
in 2021, Pieris received a €14.2 million grant from the Bavarian Ministry of Economic Affairs, Regional Development and Energy supporting research and development of the PRS-220 program. Pieris conducted a Phase 1 study of PRS-220 in healthy volunteers in Australia, which Pieris completed in August 2023. The study was a randomized, two-part, blinded, placebo-controlled study, designed to assess the safety, tolerability, pharmacokinetics, and immunogenicity of single and multiple ascending doses of PRS-220 when administered by oral inhalation to healthy subjects. The clinical study report was finalized at the end of December 2023. Data from the single and multiple ascending doses of PRS-220, when administered by oral inhalation to healthy subjects, demonstrated that PRS-220 was safe and generally well tolerated by subjects in this study at all administered doses. With the completion of the Phase 1 clinical studies, Pieris decided to discontinue further development of the program for strategic and scientific reasons; and |
|
● |
in May 2021, Pieris also entered into a multi-program research collaboration and license agreement with Genentech, a member of the Roche Group, to discover, develop and commercialize locally delivered respiratory and ophthalmology therapies. In April and May 2023, the ophthalmology and respiratory programs were jointly discontinued, respectively. Genentech also had options to select two additional programs with the payment of a fee, which expired in May 2024. With the expiration of these options and no programs active or ongoing, the Genentech Agreement also expired. |
Since inception, Pieris has devoted nearly all of its efforts and resources to its research and development activities and has incurred significant net losses. For the three months ended June 30, 2024 and 2023, Pieris reported net loss of $3.6 million and net income of $4.0 million, respectively. As of June 30, 2024, Pieris had an accumulated deficit of $323.4 million. Pieris expects to continue incurring substantial losses as it devotes time and resources into exploring strategic transactions. Pieris’ operating expenses have historically been comprised of research and development expenses and general and administrative expenses.
Pieris has not generated any revenues from product sales to date and it does not expect to generate revenues from product sales for the foreseeable future. Pieris’ revenues for the three and six months ended June 30, 2024 were from license and collaboration agreements with its partners.
A significant portion of Pieris’ operations are conducted in countries other than the United States. Since Pieris conducts its business in U.S. dollars, its main exposure, if any, results from changes in the exchange rates between the euro and the U.S. dollar. At each period end, Pieris remeasures assets and liabilities to the functional currency of that entity (for example, U.S. dollar payables recorded by Pieris’ subsidiary, Pieris Pharmaceuticals GmbH). Remeasurement gains and losses are recorded in the statement of operations line item “Other income (expense), net”. All assets and liabilities denominated in euros are translated into U.S. dollars at the exchange rate on the balance sheet date. Revenues and expenses are translated at the weighted average rate during the period. Equity transactions are translated using historical exchange rates. All adjustments resulting from translating foreign currency financial statements into U.S. dollars are included in accumulated other comprehensive loss.
Merger Agreement
On July 23, 2024, Pieris entered into the Merger Agreement with Palvella. Please see the sections titled “The Merger” and “The Merger Agreement” beginning on pages 122 and 163, respectively, of this proxy statement/prospectus for more information.
Key Financial Terms and Metrics
The following discussion summarizes the key factors Pieris management believes are necessary for an understanding of Pieris’ consolidated financial statements.
Revenues
Pieris has not generated any revenues from product sales to date and it does not expect to generate revenues from product sales for the foreseeable future. Pieris’ revenues for the last two years have been from the license and collaboration agreements with AstraZeneca, Servier, Pfizer, Genentech and Boston Pharmaceuticals.
The revenues from AstraZeneca, Servier, Pfizer, Genentech and Boston Pharmaceuticals have been comprised primarily of upfront payments, research and development services and milestone payments. For additional information about Pieris’ revenue recognition policy, see “Note 2— Summary of Significant Accounting Policies.”
Research and Development Expenses
The process of researching and developing drugs for human use is lengthy, unpredictable and subject to many risks. Historically, Pieris has incurred substantial expenses as it continued to develop its clinical and preclinical drug candidates and programs. Also included in research and development costs in 2023 were severance costs associated with the workforce reduction announced in July 2023. In the third quarter of 2023, Pieris had stopped or taken actions to wind down research and development costs related to all proprietary programs.
On March 27, 2024, Pieris announced that Pieris would be discontinuing all of Pieris’ research and development activities. Pieris has no further spending obligations related to its partnered IO programs. Pieris expects research and development costs to be significantly lower than historical amounts.
General and Administrative Expenses
General and administrative expenses consist primarily of salaries, employee benefits, equity compensation and other personnel-related costs associated with executive, administrative and other support staff. Other significant general and administrative expenses include the costs associated with professional fees for accounting, auditing, insurance costs, consulting and legal services along with facility and maintenance costs attributable to general and administrative functions. Included in general and administrative costs in 2023 were severance costs associated with the workforce reduction announced in July 2023. On March 27, 2024, Pieris announced a reduction in workforce that would impact additional employees and the executive leadership team and was implemented in the third quarter of 2024. Pieris expects general and administrative costs to be significantly lower than historical amounts given the leaner organization and elimination of research and development spending going forward.
Results of Operations
Comparison of the three and six months ended June 30, 2024 and 2023
The following table sets forth Pieris’ revenues and operating expenses (in thousands):
|
Three Months Ended June 30, |
Six Months Ended June 30, |
|||||||||||||||
|
2024 |
2023 |
2024 |
2023 |
|||||||||||||
|
Revenues |
$ | - | $ | 20,055 | $ | 53 | $ | 21,991 | ||||||||
|
Research and development expenses |
751 | 14,328 | 1,969 | 27,752 | ||||||||||||
|
General and administrative expenses |
3,426 | 3,664 | 7,564 | 7,687 | ||||||||||||
|
Total operating expenses |
4,177 | 17,992 | 9,533 | 35,439 | ||||||||||||
|
Other (expense) income |
||||||||||||||||
|
Interest income |
201 | 490 | 441 | 847 | ||||||||||||
|
Grant income |
- | 1,584 | - | 3,612 | ||||||||||||
|
Other (expense) income |
386 | (161 | ) | 557 | (218 | ) | ||||||||||
|
Net income (loss) |
$ | (3,590 | ) | $ | 3,976 | $ | (8,482 | ) | $ | (9,207 | ) | |||||
Revenues
The following table provides a comparison of revenue for the three months ended June 30, 2024 and 2023 (in thousands):
|
Three Months Ended June 30, |
||||||||||||
|
2024 |
2023 |
Increase/(Decrease) |
||||||||||
|
Customer revenue |
$ | - | $ | 20,086 | $ | (20,086 | ) | |||||
|
Collaboration revenue |
- | (31 | ) | 31 | ||||||||
|
Total Revenue |
$ | - | $ | 20,055 | (20,055 | ) | ||||||
|
● |
the $20.1 million decrease in customer revenue in the three months ended June 30, 2024 compared to the three months ended June 30, 2023 is entirely due to no revenue being recognized in the current period as all obligations related to customer revenue have previously been satisfied; and |
|
● |
the $31 thousand increase in collaboration revenues in the three months ended June 30, 2024 compared to the three months ended June 30, 2023 is due to no revenue pass through cost or collaboration reimbursement being recognized in the current period as all obligations related to the collaboration agreement have previously been satisfied. |
The following table provides a comparison of revenue for the six months ended June 30, 2024 and 2023 (in thousands):
|
Six Months Ended June 30, |
||||||||||||
|
2024 |
2023 |
Increase/(Decrease) |
||||||||||
|
Customer revenue |
$ | 6 | $ | 22,096 | $ | (22,090 | ) | |||||
|
Collaboration revenue |
47 | (105 | ) | 152 | ||||||||
|
Total Revenue |
$ | 53 | $ | 21,991 | (21,938 | ) | ||||||
|
• |
the $22.1 million decrease in customer revenue in the six months ended June 30, 2024 compared to the six months ended June 30, 2023 reflects only the final, minimal amounts of reimbursement revenue being recognized in the current period as all obligations related to customer revenue have previously been satisfied; and |
|
• |
the $0.2 million increase in collaboration revenues in the six months ended June 30, 2024 compared to the six months ended June 30, 2023 reflects final reimbursement revenue recorded in the current period as compared to changes in the estimated progress for S095012 under the Servier collaboration that led to higher revenue offsets in the prior period. |
Research and Development Expenses
The following table provides a comparison of the research and development expenses for the three months ended June 30, 2024 and 2023 (in thousands):
|
Three Months Ended June 30, |
||||||||||||
|
2024 |
2023 |
Increase/(Decrease) |
||||||||||
|
Respiratory |
$ | (116 | ) | $ | 6,743 | $ | (6,859 | ) | ||||
|
IO |
43 | 2,076 | (2,033 | ) | ||||||||
|
Other research and development activities |
824 | 5,509 | (4,685 | ) | ||||||||
|
Total |
$ | 751 | $ | 14,328 | (13,577 | ) | ||||||
|
● |
the $6.9 million decrease in Pieris’ respiratory programs for the three months ended June 30, 2024 compared to the three months ended June 30, 2023 is due primarily to lower overall costs for PRS-220 and lower pre-clinical costs for PRS-400, as these programs were stopped or wound down in connection with Pieris’ strategic update announced in July 2023. The credit balance for respiratory program expense in the current period reflects a final cash payment collected from a single vendor; |
|
● |
the $2.0 million decrease in Pieris’ IO programs for the three months ended June 30, 2024 compared to the three months ended June 30, 2023 is due primarily to decreases in manufacturing and clinical costs for both cinrebafusp alfa and S095012, as such programs have been discontinued or handed over to partners; and |
|
● |
the $4.7 million decrease in other research and development activities expenses for the three months ended June 30, 2024 compared to the three months ended June 30, 2023 is driven by lower overall personnel costs due to lower headcount as a result of restructuring actions announced in July 2023, no depreciation in 2024 as a result of Pieris’ asset sale, and lower overall lab supply costs due to the lab facility wind down. |
The following table provides a comparison of the research and development expenses for the six months ended June 30, 2024 and 2023 (in thousands):
|
Six Months Ended June 30, |
||||||||||||
|
2024 |
2023 |
Increase/(Decrease) |
||||||||||
|
Respiratory |
$ | (83 | ) | $ | 10,888 | $ | (10,971 | ) | ||||
|
Immuno-oncology |
530 | 4,585 | (4,055 | ) | ||||||||
|
Other R&D activities |
1,522 | 12,279 | (10,757 | ) | ||||||||
|
Total |
$ | 1,969 | $ | 27,752 | (25,783 | ) | ||||||
|
• |
the $11.0 million decrease in Pieris’ respiratory programs for the six months ended June 30, 2024 compared to the six months ended June 30, 2023 is due primarily to lower overall costs for PRS-220 and lower pre-clinical costs PRS-400, as these programs were stopped or wound down in connection with Pieris’ strategic update announced in July 2023. The credit balance for respiratory program expense in the current period reflects a final cash payment collected from a single vendor; |
|
• |
the $4.1 million decrease in Pieris’ IO programs for the six months ended June 30, 2024 compared to the six months ended June 30, 2023 is due primarily to decreases in manufacturing and clinical costs for both cinrebafusp alfa and S095012, as such programs have been discontinued or handed over to partners; and |
|
• |
The $10.8 million decrease in other research and development activities expenses for the six months ended June 30, 2024 compared to the six months ended June 30, 2023 is driven by lower overall personnel costs due to lower headcount as a result of restructuring actions announced in July 2023, no depreciation in 2024 as a result of Pieris’ asset sale, and lower overall lab supply costs due to the lab facility wind down. |
General and Administrative Expenses
General and administrative expenses were $3.4 million for the three months ended June 30, 2024 and $3.7 million for the three months ended June 30, 2023. The period-over-period decrease was driven by lower overall personnel costs due to lower headcount as a result of restructuring actions announced in July 2023 and March 2024, no depreciation in 2024 as a result of Pieris’ asset sale process, lower professional services spending given the winddown of programs and activities, and lower rent and facility costs. These benefits were partially offset by higher legal spending related to ongoing strategic transaction costs and the impact of lower allocated facility and IT costs to research and development departments given the winddown of research and development programs and activities.
General and administrative expenses were $7.6 million for the six months ended June 30, 2024 and $7.7 million for the six months ended June 30, 2023. The period-over-period decrease was driven by lower overall personnel costs due to lower headcount as a result of restructuring actions announced in July 2023 and March 2024 and no depreciation in 2024 as a result of Pieris’ asset sale process and lower professional services spending given the winddown of programs and activities. These benefits were partially offset by higher legal spending related to ongoing strategic transaction costs and the impact of lower allocated facility and IT costs to research and development departments given the winddown of research and development programs and activities.
Other Income (Expense)
Pieris’ other income was $0.6 million for the three months ended June 30, 2024 and $1.9 million for the three months ended June 30, 2023. The period-over-period decrease was primarily due to lower grant income offset slightly by unrealized gains in the current period due to an overall strengthening U.S. dollar and a net gain on the sales of all property and equipment compared to the estimated fair value of the assets held for sale.
Pieris’ other income was $1.0 million for the six months ended June 30, 2024 and $4.2 million for the six months ended June 30, 2023. The period-over-period decrease was primarily due to lower grant income offset slightly by unrealized gains in the current period due to an overall strengthening U.S. dollar and a net gain on the sales of all property and equipment compared to the estimated fair value of the assets held for sale.
Comparison of Years Ended December 31, 2023 and December 31, 2022
The following table sets forth Pieris’ revenues and operating expenses for the fiscal years ended December 31, 2023 and 2022 (in thousands):
|
Years ended December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
Revenues |
$ | 42,810 | $ | 25,902 | ||||
|
Research and development expenses |
41,801 | 52,982 | ||||||
|
General and administrative expenses |
16,853 | 16,394 | ||||||
|
Asset impairment |
13,912 | — | ||||||
|
Total operating expenses |
72,566 | 69,376 | ||||||
|
Interest income |
1,851 | 721 | ||||||
|
Grant income |
3,612 | 8,173 | ||||||
|
Other (expense) income , net |
(250 | ) | 1,303 | |||||
|
Loss before income taxes |
(24,543 | ) | (33,277 | ) | ||||
|
Benefit for income tax |
— | — | ||||||
|
Net loss |
$ | (24,543 | ) | $ | (33,277 | ) | ||
Revenues
The following table provides a comparison of revenues for the years ended December 31, 2023 and 2022 (in thousands):
|
Year Ended December 31, |
||||||||||||
|
2023 |
2022 |
Increase/(Decrease) |
||||||||||
|
Customer revenue |
$ | 38,711 | $ | 25,469 | $ | 13,242 | ||||||
|
Collaboration revenue |
4,099 | 433 | 3,666 | |||||||||
|
Total Revenue |
$ | 42,810 | $ | 25,902 | 16,908 | |||||||
|
• |
The $13.2 million increase in customer revenue for the year ended December 31, 2023 compared to the year ended December 31, 2022 is driven by the following: |
|
◦ |
Revenue for the year ended December 31, 2023 is primarily due to event-driven revenue recognized for the discontinuation of programs or termination of agreements with both the Genentech ($12.5 million) and AstraZeneca ($7.4 million), revenue recognized due to the Pfizer collaboration amendment driving acceleration of program handover ($10.1 million) and the milestone achieved for the Phase 1 first patient dose under the Boston Pharmaceuticals collaboration ($2.5 million) in the year ended December 31, 2023. These increases were partially offset by event-driven revenue recognized in the prior year for the discontinuation of two early-stage programs under the AstraZeneca collaboration ($9.2 million), completion of the performance obligation related to the material right for S095025 (PRS-352) ($4.9 million), milestone revenue recorded under the Pfizer collaboration ($5.0 million) and completion of the performance obligation related to the expiration of the target swap for the second program under the Pfizer collaboration ($1.5 million). |
|
• |
Collaboration revenue increased by $3.7 million in the year ended December 31, 2023 compared to the year ended December 31, 2022. The increase is primarily due to event-driven revenue recognized upon the opt-out co-development for S095012 (PRS-344) ($4.6 million), offset partially by higher Servier efforts and expenses for S095012 (PRS-344) that are reductions of our portion of revenue for activities managed by us under the Servier collaboration. |
Research and Development Expenses
The following table provides a comparison of the research and development expenses for the years ended December 31, 2023 and 2022 (in thousands):
|
Year Ended December 31, |
||||||||||||
|
2023 |
2022 |
Increase/(Decrease) |
||||||||||
|
Immuno-oncology |
$ | 6,982 | $ | 13,743 | $ | (6,761 | ) | |||||
|
Respiratory |
11,511 | 12,845 | (1,334 | ) | ||||||||
|
Other R&D activities |
23,308 | 26,394 | (3,086 | ) | ||||||||
|
Total
|
$ | 41,801 | $ | 52,982 | (11,181 | ) | ||||||
|
• |
The $6.8 million decrease in our immuno-oncology program spending period-over-period is due primarily to a decrease in clinical and manufacturing costs for both cinrebafusp alfa and S095012 (PRS-344), as well as lower professional services and consulting fees for S095012 (PRS-344). |
|
• |
The $1.3 million decrease for Pieris’ respiratory program spending period-over-period is due generally to PRS-220 and PRS-400, and the changing stage of development for these programs year-over-year. PRS-220 incurred lower preclinical and manufacturing costs in 2023, which was partially offset by higher clinical and consulting costs as the program moved from pre-clinical stage in 2022 to clinical stage in 2023. PRS-400 incurred higher manufacturing and pre-clinical costs in 2023 as compared to 2022 as Pieris was planning for IND-enabling activities prior to stopping this program in the middle of the year after its strategic announcement in July 2023. |
|
• |
The $3.1 million decrease in other research and development activities expenses is driven by lower overall personnel costs due to lower headcount, facility, software and travel expenses, all of which was partially offset higher severance costs after the mid-year strategic announcement. |
General and Administrative Expenses
General and administrative expenses were $16.9 million for the year ended December 31, 2023 as compared to $16.4 million for the year ended December 31, 2022. The period-over-period increase was driven primarily by severance expense along with higher audit and tax costs recorded in the current period, offset partially by lower salary and related costs (prior to severance costs), lower facilities, software, legal, travel and insurance costs.
Asset Impairment
During the third quarter of 2023, as part of Pieris’ strategic process for maximizing the value of assets, Pieris committed to a plan to prepare and sell all property and equipment. As a result of this decision, Pieris incurred impairment expenses totaling $13.9 million, of which $1.8 million was related to impairment of our right-of-use asset under the Hallbergmoos Lease for which Pieris and the landlord terminated the lease in December 2023.
Other income (expense), net
Pieris’ other income was $5.2 million for the year ended December 31, 2023 as compared to a other expense of $10.2 million for the year ended December 31, 2022. The decrease year over year was primarily due to lower grant income and unrealized losses. Lower grant income was a result of reaching the threshold for reimbursable costs in 2023. Lower unrealized losses in the current period were due to an overall weakening U.S. dollar on a year to date basis as compared to the prior comparable period. This was partially offset by higher interest income on investment in the current period as a result of higher interest rates.
Liquidity and Capital Resources
On March 27, 2024, Pieris announced the implementation of a new strategy along with relevant cost-saving measures that are expected to extend its cash runway into at least 2027, while maximizing its ability to capture the potential milestones from its partnered 4-1BB bispecific Mabcalin protein IO assets. Pieris may be entitled to aggregate milestones of up to approximately $15.0 million upon first patient dosed in the Phase 2 trials for SGN-BB228 and BOS-342, which are currently in Phase 1 clinical development, and up to approximately $40.0 million upon first patient dosed in pivotal clinical trials for SGN-BB228 and BOS-342.
Through June 30, 2024, Pieris has funded its operations primarily through private and public sales of equity, payments received under its license and collaboration agreements (including research and development services costs, and upfront and milestone payments), government grants and loans.
As of June 30, 2024, Pieris had a total of $19.7 million in cash, cash equivalents and investments. Pieris has incurred losses in every period since inception, with the exception of the three months ended June 30, 2023, and have a total accumulated deficit of $323.4 million as of June 30, 2024. Net losses and negative cash flows have had, and will continue to have, an adverse effect on its stockholders’ equity and working capital. Pieris expects to continue to incur operating losses for at least the next several years.
Pieris has historically devoted substantially all of its financial resources and efforts to research and development and general and administrative expenses to support such research and development.
Pieris expects cash necessary to fund operations will continue to decrease significantly in the near term as it has taken measures to preserve cash, including implementing significant workforce reductions and terminating all research and development activities.
The following table provides a summary of operating, investing and financing cash flows (in thousands):
|
Six Months Ended June 30, |
||||||||
|
2024 |
2023 |
|||||||
|
Net cash used in operating activities |
$ | (8,476 | ) | $ | (24,427 | ) | ||
|
Net cash provided by investing activities |
11,176 | 10,537 | ||||||
|
Net cash provided by financing activities |
- | 19,781 | ||||||
Net cash used in operating activities for the six months ended June 30, 2024 was $8.5 million compared to net cash used in operating activities of $24.4 million for the six months ended June 30, 2023. The decrease in cash used in operations in the current period is predominantly attributable to the $21.5 million impact of a reduction in deferred revenue in the six months ended June 30, 2023 as compared to the current period in which there was no cash used in operations related to deferred revenue.
Net cash provided by investing activities for the six months ended June 30, 2024 was $11.2 million, as compared to net cash provided by investing activities of $10.5 million for the same period in 2023. The change in net cash used is predominantly attributable to the impact of net investments changes and the timing of maturities in the current period, as well as proceeds on the sale of all assets, as compared to the prior period.
There was no net cash provided by financing activities for the six months ended June 30, 2024, as compared to net cash provided by financing activities of $19.8 million for the same period in 2023. The change in net cash used is predominantly attributable to the $19.7 million impact of the issuance of stock resulting from the ATM sales activity performed during the six months ended June 30, 2023.
The following table provides a summary of operating, investing, and financing cash flows for the years ended December 31, 2023 and 2022 respectively (in thousands):
|
Year Ended December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
Net cash used in operating activities |
$ | (53,819 | ) | $ | (59,932 | ) | ||
|
Net cash provided by (used in) investing activities |
12,002 | (21,236 | ) | |||||
|
Net cash provided by financing activities |
19,795 | 7,214 | ||||||
Net cash used in operating activities for the year ended December 31, 2023 and 2022 was $53.8 million and $59.9 million, respectively. Cash used in operations in the year ended December 31, 2023 is impacted by lower deferred revenue, primarily driven by higher revenue recognized across all of Pieris’ collaborations, lower lease liability due to lease termination payment, lower accounts payable, and lower accrued expenses. These changes are offset partially by lower accounts receivable and lower overall prepaid and other assets. This compares to the impact of lower deferred revenue, primarily driven by higher revenue recognized for AstraZeneca, Servier and Pfizer out of the deferred balance, lower accounts payable and accrued expenses and higher accounts receivable and prepaid expenses in the prior period.
The change in net cash provided by investing activities for the year ended December 31, 2023 compared to net cash used in investing activities in 2022 is solely attributable to the impact of net investments changes (more maturities in the current period versus more purchases of investments in the comparable prior year period).
Financing activities for the year ended December 31, 2023 and 2022 provided cash of $19.8 million and $7.2 million, respectively. The increase in net cash provided by financing activities is due to an increase in sales under the ATM Program.
Effective at 5:00 p.m. Eastern Time on April 22, 2024, Pieris effected a 1-for-80 reverse stock split of its common stock. All references to shares of common stock outstanding, average number of shares outstanding and per share amounts in this proxy statement/prospectus have been restated to reflect the reverse stock split on a retroactive basis.
In August 2021, Pieris established the ATM Program under a sales agreement with Jefferies LLC, pursuant to which Pieris may offer and sell shares of its common stock, from time to time, up to an aggregate amount of gross sales proceeds of $50.0 million. In November 2022, the sales agreement was amended to provide for an increase in the aggregate offering amount, such that under the ATM Program, as amended, Pieris may offer and sell shares of its common stock, from time to time, up to an aggregate amount of gross sales proceeds of $75.0 million. The ATM Program, as amended, is offered under a shelf registration statement on Form S-3 that was filed with and declared effective by the SEC in August 2021. For the year ended December 31, 2023, Pieris sold 0.3 million shares for gross proceeds of $20.3 million under the ATM Program at an average stock price of $67.07 per share. For the year ended December 31, 2022, Pieris sold 25,000 shares for gross proceeds of $7.2 million under the predecessor ATM program at an average stock price of $276.51.
On July 23, 2024, Pieris entered into the Purchase Agreement with the PIPE Investors, including BVF, an existing stockholder of Pieris, pursuant to which, among other things, the PIPE Investors have agreed to subscribe for and purchase (either for cash or in exchange for the termination and cancellation of outstanding convertible notes issued by Palvella), and Pieris agreed to issue and sell to the PIPE Investors, an aggregate of approximately 3,154,241 of shares of Pieris common stock at a price per share equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, and/or in lieu of Pieris common stock to certain purchasers who so choose, Pre-Funded Warrants to purchase up to 2,592,585 shares of Pieris common stock at a purchase price per Pre-Funded Warrant equal to the Purchase Price minus $0.001, subject to adjustment as set forth in the Purchase Agreement. The Purchase Agreement contains customary representations and warranties of Pieris, on the one hand, and the PIPE Investors, on the other hand, and customary conditions to closing, including the consummation of the Merger. The gross proceeds from the PIPE Financing are expected to be approximately $78.9 million, consisting of approximately $60.0 million in cash and the conversion of approximately $18.9 million of principal and interest payable under the outstanding convertible notes issued by Palvella, before paying estimated expenses. The closing of the PIPE Financing is expected to occur in connection with and immediately following the consummation of the Merger.
The Pre-Funded Warrants do not expire, and each Pre-Funded Warrant will be exercisable at any time after the date of issuance of such Pre-Funded Warrant, subject to a beneficial ownership limitation. A holder of a Pre-Funded Warrant may not exercise such Pre-Funded Warrant if the holder, together with its affiliates, would beneficially own more than 4.99% or 9.99% of the number of shares of Pieris common stock outstanding immediately after giving effect to such exercise, provided, however, that a holder may increase or decrease the beneficial ownership limitation by giving 61 days’ notice to us, but not to any percentage in excess of 19.99%.
At or prior to the closing of the PIPE Financing, we will enter into the Registration Rights Agreement with the PIPE Investors pursuant to which the PIPE Investors will be entitled to certain resale registration rights with respect to shares of Pieris common stock issued to the PIPE Investors and any shares of Pieris common stock issued upon exercise of the Pre-Funded Warrants. Pursuant to the Registration Rights Agreement, Pieris will be required to prepare and file a resale registration statement with the SEC within 30 days following the closing of the PIPE Financing. Pieris shall use its commercially reasonable efforts to cause this registration statement to be declared effective by the SEC within 90 days following the closing of the PIPE Financing (or within 120 days following the PIPE Financing if the SEC reviews such resale registration statement).
Pieris is currently subject to the SEC general instructions of Form S-3 known as the “baby shelf rules.” Under these instructions, the amount of funds Pieris can raise through primary public offerings of securities in any 12-month period using its registration statement on Form S-3 is limited to one-third of the aggregate market value of the shares of its common stock held by non-affiliates. Therefore, Pieris will be limited in the amount of proceeds it is able to raise by selling shares of its common stock using its Form S-3, including under the ATM Program, until such time as its public float exceeds $75 million.
Pieris has historically devoted substantially all of its financial resources and efforts to research and development and general and administrative expenses to support such research and development. Pieris expects cash necessary to fund operations will continue to decrease significantly as it has decided to discontinue all research and development activities and implement a further workforce reduction that will affect additional employees and the executive leadership team.
Pieris believes that its currently available funds will be sufficient to fund its remaining limited operations through at least the next 12 months from the issuance of this proxy statement/prospectus. As part of its March 27, 2024 strategic update, as discussed above, Pieris decided to implement measures to reduce discretionary expenditures and other fixed or variable personnel costs as it discontinued all remaining research and development obligations.
If Pieris seeks to raise additional capital to fulfill its operating and capital requirements through public or private equity financings, utilization of its current ATM Program, strategic collaborations, licensing arrangements, government grants and/or the achievement of milestones under its collaborative agreements, there is no assurance that Pieris will be successful in obtaining sufficient funding on terms acceptable to Pieris to fund continuing operations, if at all, and the terms of any future financing may adversely affect the holdings or the rights of its stockholders.
Off-Balance Sheet Arrangements
Pieris does not have any off-balance sheet arrangements, as defined under applicable SEC rules.
Critical Accounting Policies and Estimates
Refer to Part II, Item 7, “Critical Accounting Policies and Estimates” of Pieris’ Annual Report on Form 10-K for the fiscal year ended December 31, 2023 for a discussion of its critical accounting policies and estimates.
This discussion and analysis of Pieris’ financial condition and results of operations is based on its financial statements, which Pieris has prepared in accordance with U.S. GAAP. Pieris believes that several accounting policies are important to understanding its historical and future performance. Pieris refers to these policies as critical because these specific areas generally require Pieris to make judgments and estimates about matters that are uncertain at the time Pieris makes the estimate, and different estimates-which also would have been reasonable-could have been used. On an ongoing basis, Pieris evaluates its estimates and judgments, including those described in greater detail elsewhere in this proxy statement/prospectus. Pieris bases its estimates on historical experience and other market-specific or other relevant assumptions that Pieris believes to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions.
Pieris believes that its most critical accounting policies are those relating to revenue recognition, contingencies, research and development expense and income taxes, and there have not been significant changes to its accounting policies discussed in the Annual Report on Form 10-K for the fiscal year ended on December 31, 2023.
Recently Issued Accounting Pronouncements
Pieris reviews new accounting standards to determine the expected financial impact, if any, that the adoption of each standard will have. For the recently issued accounting standards that Pieris believes may have an impact on its consolidated financial statements, see “Note 2—Summary of Significant Accounting Policies” in its condensed consolidated financial statements.
Smaller Reporting Company Status
Currently, Pieris qualifies as a smaller reporting company.
As a smaller reporting company, Pieris is eligible for and has taken advantage of certain exemptions from various reporting requirements that are not available to public reporting companies that do not qualify for this classification, including, but not limited to:
|
● |
an opportunity for reduced disclosure obligations regarding executive compensation in Pieris’ periodic and annual reports, including exemption from the requirements to provide a compensation discussion and analysis describing compensation practices and procedures; |
|
● |
an opportunity for reduced financial statement disclosure in registration statements and in annual reports on Form 10-K, which only requires two years of audited financial statements rather than the three years of audited financial statements that are required for other public companies; and |
|
● |
an opportunity to utilize the non-accelerated filer time-line requirements. |
For as long as Pieris continues to be a smaller reporting company, Pieris expects that it will take advantage of both the reduced internal control audit requirements and the disclosure obligations available to it as a result of this classification.
PALVELLA MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The following discussion and analysis provides information which Palvella’s management believes is relevant to an assessment and understanding of Palvella’s results of operations and financial condition. This discussion and analysis should be read together with the section of this proxy statement/prospectus entitled Palvella’s audited and unaudited financial statements and related notes that are included elsewhere in this proxy statement/prospectus. This discussion and analysis should also be read together with the section of this proxy statement/ prospectus entitled “Palvella’s Business” and the unaudited pro forma condensed combined financial information as of and for the three and six months ended June 30, 2024 and for the year ended December 31, 2023 included in the section of this proxy statement/ /prospectus entitled “Unaudited Pro Forma Financial Information.” In addition to historical financial analysis, this discussion and analysis contains forward-looking statements based upon current expectations that involve risks, uncertainties and assumptions, as described under the heading “Cautionary Statement Concerning Forward-Looking Statements and Market and Industry Information.” Actual results and timing of selected events may differ materially from those anticipated in these forward-looking statements as a result of various factors, including those set forth under “Risk Factors—Risks Relating to Palvella’s” or elsewhere in this proxy statement/prospectus.
Overview
Palvella is a clinical-stage biopharmaceutical company whose vision is to become the leading rare disease biopharmaceutical company focused on developing and, if approved, commercializing novel therapies to treat patients suffering from serious, rare genetic skin diseases for which there are no FDA-approved therapies. Palvella intends to leverage its versatile QTORIN platform to treat these patients. QTORIN is designed to generate new therapies that penetrate the deep layers of the skin to locally treat a broad spectrum of rare genetic skin diseases. Palvella’s lead product candidate, QTORIN rapamycin, is in clinical development for two of these diseases: microcystic LM, and cutaneous venous malformations. QTORIN rapamycin contains the active pharmaceutical ingredient rapamycin, also known as sirolimus, which is an inhibitor of mTOR, a kinase that plays a key role in cell growth and proliferation.
Palvella currently has one ongoing clinical trial and one clinical trial planned to start in the fourth quarter of 2024. Palvella’s ongoing trial, SELVA, is a Phase 3 Baseline-Controlled Study Evaluating the Safety and Efficacy of QTORIN rapamycin in the Treatment of Microcystic LM. Palvella previously announced topline Phase 2 clinical trial results from the multi-center, open-label study of 12 subjects receiving QTORIN™ rapamycin once-daily for 12-weeks. The Phase 2 clinical trial featured multiple pre-specified efficacy assessments, including clinician and patient global impression assessments as well as assessments of individual clinical manifestations that are important disease burdens for individuals living with microcystic LMs. All participants in the Phase 2 clinical trial demonstrated improvements on the Clinician Global Impression of Change scale, with all participants in the study rated as either "Much Improved" (n=7, 58%) or "Very Much Improved" (n=5, 42%) after 12-weeks of treatment compared to the pre-treatment baseline period. Palvella expects to report top-line data for the Phase 3 study in approximately 40 participants with microcystic LM in the first quarter of 2026.
The FDA defines a baseline-controlled study as a clinical study in which the patient's condition during treatment is compared with their condition before treatment. In such studies, participants serve as their own control. In a placebo-controlled study, patients are randomized prior to treatment to receive either study drug or matching placebo and to determine how the efficacy of the treatment compares to placebo.
Microcystic LM is a serious, chronically debilitating, and lifelong disease of the lymphatic system characterized by lymphorrhea and acute cellulitis. It is estimated that there are more than 30,000 diagnosed patients in the United States with microcystic LM. The specific pathophysiology of microcystic LM is primarily the result of somatic activating mutations in PIK3CA which result in increased activation of the PI3K/mTOR pathway and subsequent lymphatic hyperplasia. Because microcystic LM has a well-understood pathophysiology and has a well-defined disease course, Palvella believes the optimal clinical study for this rare disease is a baseline-controlled Phase 3 study that incorporates both live clinician assessments and review by a blinded committee.
Palvella has received Breakthrough Therapy Designation, Fast Track Designation, and Orphan Drug Designation from the FDA for QTORIN rapamycin for the treatment of microcystic LM. Palvella has also received Fast Track Designation from the FDA for the treatment of venous malformations.
There are no FDA-approved therapies currently indicated for either microcystic LM or cutaneous venous malformations. If approved for the treatment of microcystic LM or cutaneous venous malformations, Palvella believes QTORIN rapamycin has the potential to become the standard of care for these diseases.
Palvella also has a planned study for cutaneous venous malformations, a Phase 2 Baseline-Controlled Study Evaluating the Safety and Efficacy of QTORIN rapamycin for the Treatment of Cutaneous Venous Malformations expected to start in the fourth quarter of 2024. Cutaneous venous malformations are a serious disease with a high unmet need characterized by dysregulated growth of malformed veins impacting the skin, causing functional impairment and deformity. It is estimated that there are more than 75,000 diagnosed patients in the United States with cutaneous venous malformations. Palvella is conducting a Phase 2 baseline-controlled clinical trial in approximately 15 participants in this patient population and expects to report top-line data in the fourth quarter of 2025.
Palvella also has additional preclinical research programs based on Palvella’s QTORIN platform for the treatment of serious, rare genetic skin diseases for which Palvella believes there are significant unmet needs. As Palvella plans to expand its pipeline into new rare skin diseases, it plans to generate new product candidates with its QTORIN platform.
Palvella has multiple patents and patent applications directed to anhydrous gel formulations of rapamycin, including QTORIN rapamycin, and the use of such anhydrous gel formulations for the treatment certain skin disorders, including microcystic LM and venous malformations. Palvella’s issued U.S. patents with claims directed to certain anhydrous gel formulations containing rapamycin and methods of treatment expire as in 2038.
Background
Palvella was formed under the laws of the State of Delaware on September 11, 2015 as Palvella Therapeutics LLC, a limited liability company. On May 30, 2018, Palvella converted into a Delaware corporation and changed its name to Palvella Therapeutics, Inc. Since Palvella’s inception, it has devoted substantially all of its time to identifying, researching and conducting preclinical and clinical activities for its product candidates, acquiring and developing its platform technology, organizing and staffing its company, business planning, raising capital and establishing its intellectual property portfolio.
Since Palvella’s inception in 2015, Palvella has incurred significant operating losses, and Palvella has never generated any revenue. Palvella’s ability to generate revenue from product sales sufficient to achieve profitability will depend heavily on the successful development and commercialization of QTORIN rapamycin and other future product candidates. Further, if Palvella enters into license or collaboration agreements for any of its product candidates or intellectual property, Palvella may generate revenue in the future from payments as a result of such license or collaboration agreements; however, there can be no assurance that Palvella will be able to enter into any license or collaboration agreements. Palvella’s operating loss was $2.9 million and $4.4 million and $4.7 million and $8.9 million for the three and six months ended June 30, 2024 and 2023, respectively, and $11.9 million and $18.0 million for the years ended December 31, 2023 and 2022, respectively. Since inception, Palvella’s operations have been financed primarily by aggregate net proceeds of $76.5 million from the issuance of convertible preferred stock and convertible notes and $15.0 million from the Ligand Agreements, with Ligand which is discussed further below. As of December 31, 2023, Palvella had an accumulated deficit of $76.3 million and cash and cash equivalents of $7.4 million. As of June 30, 2024, Palvella had an accumulated deficit of $83.0 million and cash equivalents of $14.5 million.
Palvella expects to continue to incur significant operating losses for the foreseeable future and expects to incur increased expenses as Palvella continues to advance its product candidates through clinical trials and regulatory submissions. Palvella may also incur expenses in connection with the in-licensing or acquisition of additional product candidates. Furthermore, upon the closing of the Merger (see the section below entitled “Proposed Merger”), Palvella expects to incur additional costs associated with operating as a public company, including significant legal, accounting, investor relations and other expenses that Palvella did not incur as a private company. If Palvella receives regulatory approval for QTORIN rapamycin for treatment of Microcystic LM, venous malformations or any future product candidates, Palvella expects to incur significant commercialization expenses related to product manufacturing, marketing, sales and distribution. Palvella’s losses may fluctuate significantly from quarter-to-quarter and year-to-year, depending on the timing of its clinical trials and Palvella’s expenditures on other research and development activities.
Ligand Development Funding and Royalty Agreement
In December 2018, Palvella entered into the Original Ligand Agreement, whereby Ligand made a one-time payment of $10.0 million to fund the development of QTORIN rapamycin. In November 2023, pursuant to the Amended Ligand Agreement Ligand made an additional one-time payment of $5.0 million to fund the development of QTORIN rapamycin. Under the Amended Ligand Agreement, Ligand is entitled to receive up to $8.0 million in milestone payments upon the achievement of certain milestones by Palvella related to QTORIN rapamycin for the treatment of any and all indications, of which $5.0 million of potential future milestone payments remain under the arrangement. In addition, Palvella agreed to pay to Ligand tiered royalties from 8.0% to 9.8% based on any aggregate annual worldwide net product sales of any products based on QTORIN rapamycin. The Amended Ligand Agreement includes an option for Ligand to purchase additional product revenue participation rights from Palvella over a certain period of time (as set forth in the Amended Ligand Agreement). The option allows Ligand, for each product developed on the QTORIN platform that completes the first human clinical trial in the United States, the opportunity to make an upfront payment (as set forth in the Amended Ligand Agreement) to Palvella in return for a royalty rate (as set forth in the Amended Ligand Agreement). Palvella’s obligation to make future milestone payments under the Amended Ligand Agreement was determined to be a derivative liability and Palvella’s obligation to make future royalty payments was determined to be a debt instrument. Please see “—Critical Accounting Policies and Significant Judgments and Estimates—Ligand Agreement” and “Business—Ligand Development Funding Agreement.”
Recent Developments
Proposed Merger
On July 23, 2024, Palvella entered into the Merger Agreement with Pieris. Please see the sections titled “The Merger” and “The Merger Agreement” beginning on pages 122 and 163, respectively, of this proxy statement/prospectus for more information.
PIPE Financing
On July 23, 2024, Pieris entered into a securities purchase agreement (the “Purchase Agreement”) with certain investors, including BVF Partners, L.P., an existing stockholder of Pieris (the “PIPE Investors”). Please see the sections titled “The Merger” and “The Merger Agreement” beginning on pages 122 and 163, respectively, of this proxy statement/prospectus for more information.
Impact of Global Economic Events
Uncertainty in the global economy presents significant risks to Palvella’s business. Palvella is subject to continuing risks and uncertainties in connection with the current macroeconomic environment, including increases in inflation and geopolitical factors, including the ongoing conflict between Russia and Ukraine and the responses thereto, and supply chain disruptions. While Palvella’s management is closely monitoring the impact of the current macroeconomic conditions on all aspects of Palvella’s business, including the impacts on its participants in its Phase 3 clinical trials, employees, suppliers, vendors and business partners, the ultimate extent of the impact on Palvella’s business remains highly uncertain and will depend on future developments and factors that continue to evolve. Most of these developments and factors are outside Palvella’s control and could exist for an extended period of time. Management will continue to evaluate the nature and extent of the potential impacts to Palvella’s business, results of operations, liquidity and capital resources. For additional information, see the section entitled “Risk Factors—Risks Related to Palvella.”
Components of Operating Results
Operating Expenses
Palvella’s operating expenses since inception have consisted primarily of research and development expenses and general and administrative costs.
Research and Development Expenses
Palvella’s research and development expenses consist primarily of costs incurred for the development of its product candidates, which include:
|
● |
costs related to production of preclinical and clinical materials, including CMC fees paid to CMOs; |
|
● |
personnel costs, including salaries, related benefits and stock-based compensation expense for employees engaged in research and development functions; |
|
● |
vendor expenses related to the execution of preclinical studies and clinical trials; |
|
● |
expenses incurred under agreements with consultants that conduct research and development activities on Palvella’s behalf; |
|
● |
costs related to compliance with regulatory requirements; and |
|
● |
allocated overhead, including rent, equipment and information technology costs. |
Palvella expenses all research and development expenses in the periods in which they are incurred. Costs for certain research and development activities are recognized based on an evaluation of the progress to completion of specific tasks using information and data provided to Palvella by its vendors and other service providers. This process involves reviewing open contracts, communicating with Palvella’s personnel to identify services that have been performed on Palvella’s behalf and estimating the level of service performed and the associated cost incurred for the service when Palvella has not yet been invoiced or otherwise notified of actual costs. Any nonrefundable advance payments that Palvella makes for goods or services to be received in the future for use in research and development activities are recorded as prepaid expenses. Such amounts are expensed as the related goods are delivered or the related services are performed, or until it is no longer expected that the goods will be delivered or the services rendered.
Palvella’s indirect research and development expenses are not currently tracked on a program-by-program basis. Palvella uses its personnel and infrastructure resources across multiple research and development programs to identify and develop product candidates.
Research and development activities account for a significant portion of Palvella’s operating expenses. Palvella expects its research and development expenses to increase substantially for the foreseeable future as Palvella continues to invest in research and development activities related to developing its product candidates, including investments in advancing its programs and conducting clinical trials. In particular, Palvella expects to incur substantial research and development expenses to continue late-stage clinical development and pursue regulatory approvals of QTORIN rapamycin for the treatment of microcystic LM, venous malformations and the development of Palvella’s preclinical programs. Product candidates in later stages of clinical development generally incur higher development costs than those in earlier stages, primarily due to the increased size and duration of later-stage clinical trials. As a result, Palvella expects its research and development expenses to increase as its product candidates advance into later stages of clinical development.
Because of the numerous risks and uncertainties associated with product development and the current stage of development of Palvella’s product candidates and programs, Palvella cannot reasonably estimate or know the nature, timing and estimated costs necessary to complete the remainder of the development of Palvella’s product candidates or programs. The duration, costs and timing of preclinical studies and clinical trials and development of Palvella’s product candidates will depend on a variety of factors, including:
|
● |
timely completion of Palvella’s preclinical studies and clinical trials, which may be significantly slower or cost more than it currently anticipate and may depend substantially upon the performance of certain third-party contractors; |
|
● |
delays in validating, or inability to validate, any endpoints utilized in a clinical trial; |
|
● |
the prevalence, duration and severity of potential side effects or other safety issues experienced with Palvella’s product candidates, if any, or experienced by competitors who are developing topical rapamycin products or who are targeting the same indications in the rare genetic skin diseases space; |
|
● |
the ability of CMOs upon which Palvella relies to manufacture clinical supplies of its product candidates or any future product candidates to remain in good standing with relevant regulatory authorities and to develop, validate and maintain commercially viable manufacturing processes that are compliant with cGMP; |
|
● |
Palvella’s ability to retain patients who have enrolled in a clinical study but may be prone to withdraw due to the rigors of the clinical trial, lack of efficacy, side effects, personal issues or loss of interest; |
|
● |
Palvella’s ability to establish and enforce intellectual property rights in and to its current product candidates or any future product candidates; and |
|
● |
minimizing and managing any delay or disruption to Palvella’s ongoing or planned clinical trials. |
A change in the outcome of any of these factors with respect to the development of any of Palvella’s product candidates would significantly change the costs and timing associated with the development of that product candidate.
Palvella may never succeed in achieving regulatory approval for any of its product candidates. Palvella’s preclinical studies and clinical trials may be unsuccessful. Palvella may elect to discontinue, suspend or modify clinical trials of some product candidates or focus on others. A change in the outcome of any of these factors could mean a significant change in the costs and timing associated with the development of Palvella’s current and future preclinical and clinical product candidates. For example, if the FDA or another regulatory authority were to require Palvella to conduct additional clinical trials beyond those that Palvella currently anticipates will be required for the completion of any of Palvella’s product candidates’ clinical development, or if Palvella experiences significant delays in execution of or enrollment in any of its preclinical studies or clinical trials, Palvella could be required to expend significant additional financial resources and time on the completion of preclinical and clinical development for such product candidates.
General and Administrative Expenses
Palvella’s general and administrative expenses consist primarily of the following costs:
|
● |
personnel costs, including salaries, related benefits, travel and stock-based compensation expense for personnel in executive, finance and administrative functions; and |
|
● |
professional fees for legal, intellectual property, information technology, financial, human resources, consulting, audit and accounting services not otherwise included in research and development expenses. |
Palvella anticipates that its general and administrative expenses will increase substantially in the future as Palvella increases its headcount to support its organizational growth. Following the completion of the Merger, Palvella also anticipates that it will incur increased accounting, audit, legal, regulatory, compliance and director and officer insurance costs as well as investor and public relations expenses associated with Palvella’s operations as a public company. In addition, if Palvella obtains regulatory approval for a product candidate and does not enter into a third-party commercialization collaboration, Palvella expects to incur significant expenses related to building a sales and marketing organization to support product sales, marketing and distribution activities.
Other (Expense) Income
Palvella’s other (expense) income for the years ended December 31, 2023 and 2022 primarily consists of non-cash interest expense with respect to the royalty agreement liability, and fair value adjustments on the derivative liability components of the Ligand Agreements. Palvella’s other (expense) income is subject to variability due to changes in the fair value of the derivative liabilities as well as the potential variability of the royalty agreement liability, both of which are based on significant estimates regarding the timing and success of future development and commercialization activities. During the second quarter of 2023, Palvella received data from certain of its clinical trials that reduced the projected net product sales related to QTORIN rapamycin and the corresponding probabilities of successful commercialization, resulting in a significant reduction in the expected future royalty payments and a corresponding reduction in the royalty agreement liability. In November 2023, the Ligand Agreement was extinguished and the Amended Ligand Agreement was recorded at the estimated fair value of the royalty agreement liability on the date of the amendment. This resulted in a non-cash gain on extinguishment being recorded in other (expense) income related to the difference between the carrying value of the liability and its estimated fair value on the date of amendment.
Income Taxes
Since May 2018, Palvella has not recorded any income tax benefits for NOLs. Palvella believes, based upon the weight of available evidence, that it is more likely than not that all of Palvella’s NOLs and tax credits will not be realized. Accordingly, Palvella has established a valuation allowance against such deferred tax assets for all periods since inception.
Palvella assesses its income tax positions and records tax benefits based upon management’s evaluation of the facts, circumstances, and information available at the reporting date. For those tax positions where it is more likely than not that a tax benefit will be sustained, Palvella records the amount of tax benefit with a greater than 50% likelihood of being realized upon ultimate settlement with a taxing authority having full knowledge of all relevant information. For those income tax positions for which it is not more likely than not that a tax benefit will be sustained, no tax benefit is recognized in the financial statements.
The Company had no provision for income taxes for the year ended December 31, 2023. The Company recorded a benefit for income taxes of $1.0 million for the year ended December 31, 2022 which consisted of approximately $0.1 million of current federal tax benefit and $0.9 million of current state tax benefit. The 2022 tax benefit is attributed to the reversal of the company's uncertain tax position due to the lapse of the 2018 Pennsylvania statute of limitations concerning the timing of the payment received under the Original Ligand Agreement.
As of December 31, 2023, Palvella had federal and state NOL carryforwards in the amount of $36.7 million and $37.6 million, respectively, which may be available to offset future taxable income. The state NOL carryforwards begin expiring at various dates through 2038, unless previously utilized. All federal NOL carryforwards were generated subsequent to January 1, 2018 and therefore are able to be carried forward indefinitely. As of December 31, 2023, Palvella has orphan drug credits of $0.2 million to reduce future federal taxes through 2039.
Results of Operations
Comparison of Three Months Ended June 30, 2024 and 2023
The following sets forth Palvella’s results of operations:
|
Three Months Ended June 30, |
Change |
|||||||||||||||
|
2024 |
2023 |
$ |
% |
|||||||||||||
|
Operating expenses: |
||||||||||||||||
|
Research and development |
$ | 1,442 | $ | 3,382 | $ | (1,940 | ) | (57 | )% | |||||||
|
General and administrative |
1,466 | 978 | 488 | 49 | % | |||||||||||
|
Total operating expenses |
2,908 | 4,360 | (1,452 | ) | (33 | )% | ||||||||||
|
Operating loss |
(2,908 | ) | (4,360 | ) | 1,452 | (33 | )% | |||||||||
|
Other (expense) income: |
||||||||||||||||
|
Interest expense - royalty agreement |
(942 | ) | 11,284 | (12,226 | ) | (108 | )% | |||||||||
|
Interest expense – convertible notes payable |
(29 | ) | - | (29 | ) | (100 | )% | |||||||||
|
Fair value adjustments on derivative liabilities |
(271 | ) | 678 | (949 | ) | (140 | )% | |||||||||
|
Other income, net |
(22 | ) | 94 | (116 | ) | (123 | )% | |||||||||
|
Net loss |
$ | (4,172 | ) | $ | 7,696 | $ | (11,832 | ) | (154 | )% | ||||||
Research and Development Expenses
The table below summarizes Palvella’s research and development expenses incurred by development program:
|
Three Months Ended June 30, |
Change |
|||||||||||||||
|
(in thousands) |
2024 |
2023 |
$ |
% |
||||||||||||
|
QTORIN rapamycin for PC and GS |
$ | - | $ | 1,550 | $ | (1,550 | ) | (100 | )% | |||||||
|
QTORIN rapamycin for microcystic LM |
422 | 69 | 353 | 512 | % | |||||||||||
|
QTORIN rapamycin CMC costs |
40 | 214 | (174 | ) | (81 | )% | ||||||||||
|
Non-program specific and unallocated research and development expenses: |
||||||||||||||||
|
Salaries and stock-based compensation |
704 | 853 | (149 | ) | (17 | )% | ||||||||||
|
Consultants |
185 | 617 | (432 | ) | (70 | )% | ||||||||||
|
Other |
91 | 79 | 12 | 15 | % | |||||||||||
|
Total research and development expenses |
$ | 1,442 | $ | 3,382 | $ | (1, 940 | ) | (57 | )% | |||||||
For the three months ended June 30, 2024, research and development expenses were $1.4 million, compared to $3.4 million for the three months ended June 30, 2023. The decrease in research and development expenses during the three months ended June 30, 2024 was primarily due to lower clinical trial costs of $1.5 million and consulting costs of $0.4 million due to the decision to curtail program spending in July 2023 for programs for pachyonychia congenita, or “PC”, and gorlin syndrome, or “GS”, as a result of the clinical trial data readouts for these programs. Partially offsetting the decrease was the start-up costs for the microcystic LM study commencing in Q3 2024.
General and Administrative Expenses
For the three months ended June 30, 2024, general and administrative expenses were $1.5 million, compared to $1 million for the three months ended June 30, 2023. The increase in general and administrative expenses during the three months ended June 30, 2024 was primarily due to an increase in professional services and legal costs as a result of the activity associated with the Merger Agreement.
Other (Expense) Income
Other (expense) income for the three months ended June 30, 2024 and 2023 was ($1.3) million and $12.0 million, respectively. The decrease of $13.3 million was attributable to a decrease in non-cash interest (expense) income of $12.2 million related to the decrease in carrying amounts of the royalty agreement liability primarily as a result of changes in net sales assumptions for QTORIN rapamycin and the removal of projected royalty payments for PC and GS in the second quarter of 2023.
Comparison of Six Months Ended June 30, 2024 and 2023
The following sets forth Palvella’s results of operations:
|
Six Months Ended June 30, |
Change |
|||||||||||||||
|
2024 |
2023 |
$ |
% |
|||||||||||||
|
Operating expenses: |
||||||||||||||||
|
Research and development |
$ | 2,426 | $ | 6,998 | $ | (4,572 | ) | (65 | )% | |||||||
|
General and administrative |
2,241 | 1,902 | 339 | 18 | % | |||||||||||
|
Total operating expenses |
4,667 | 8,900 | (4,206 | ) | (48 | )% | ||||||||||
|
Operating loss |
(4,667 | ) | (8,900 | ) | (4,233 | ) | (48 | )% | ||||||||
|
Other income (expense): |
||||||||||||||||
|
Interest income/ (expense) - royalty agreement |
(1,747 | ) | 8,705 | (10,452 | ) | (120 | )% | |||||||||
|
Interest expense – convertible notes payable |
(29 | ) | - | (29 | ) | (100 | )% | |||||||||
|
Fair value adjustments on derivative liabilities income/ (expense) |
(329 | ) | 593 | (922 | ) | (155 | )% | |||||||||
|
Other income, net |
64 | 586 | (522 | ) | (89 | )% | ||||||||||
|
Net income (loss) |
$ | (6,708 | ) | $ | 984 | $ | (7,692 | ) | (782 | )% | ||||||
Research and Development Expenses
The table below summarizes Palvella’s research and development expenses incurred by development program:
|
Six Months Ended June 30, |
Change |
|||||||||||||||
|
(in thousands) |
2024 |
2023 |
$ |
% |
||||||||||||
|
QTORIN rapamycin for PC and GS |
$ | - | $ | 3,241 | $ | (3,241 | ) | (100 | )% | |||||||
|
QTORIN rapamycin for microcystic LM |
425 | 150 | 275 | 183 | % | |||||||||||
|
QTORIN rapamycin CMC costs |
153 | 572 | (419 | ) | (73 | )% | ||||||||||
|
Non-program specific and unallocated research and development expenses: |
||||||||||||||||
|
Salaries and stock-based compensation |
1,351 | 1,697 | (346 | ) | (20 | )% | ||||||||||
|
Consultants |
351 | 1,048 | (697 | ) | (67 | )% | ||||||||||
|
Other |
146 | 290 | (144 | ) | (49 | )% | ||||||||||
|
Total research and development expenses |
$ | 2,426 | $ | 6,998 | $ | (4,572 | ) | (65 | )% | |||||||
Research and development expenses for the six months ended June 30, 2024 were $2.4 million compared to $7.0 million for the six months ended June 30, 2023. The decrease in research and development expenses during the six months ended June 30, 2024 was due to decreased expenses incurred from the PC and GS programs as a result of the 2023 second quarter readouts of the PC and GS clinical trials as well as decreased consulting and CMC costs across all programs.
General and Administrative Expenses
For the six months ended June 30, 2024, general and administrative expenses were $2.2 million compared to $1.9 million for the six months ended June 30, 2023. The increase in general and administrative expenses during the six months ended June 30, 2024 was primarily due to the activity associated with the Merger Agreement.
Other Income (Expense)
Other income (expense) during the six months ended June 30, 2024 was ($2.0) million of expense as compared to $9.9 million of income during the six months ended June 30, 2023. During the second quarter of 2023, the Company received data from certain of its clinical trials that reduced the projected net product sales related to QTORIN rapamycin and the corresponding probabilities of successful commercialization, resulting in a significant reduction in the expected future royalty payments and a corresponding reduction in the royalty agreement liability. The Company incurred non-cash interest income (expense) related to the royalty agreement of ($1.7) million and $8.7 million for the six months ended June 30, 2024 and 2023.
Comparison of Fiscal Years Ended December 31, 2023 and 2022
The following sets forth Palvella’s results of operations:
|
Year Ended December 31, |
Change |
|||||||||||||||
|
2023 |
2022 |
$ |
% |
|||||||||||||
|
Operating expenses: |
||||||||||||||||
|
Research and development |
$ | 8,793 | $ | 13,884 | $ | (5,091 | ) | (37 | )% | |||||||
|
General and administrative |
3,076 | 4,156 | (1,080 | ) | (26 | )% | ||||||||||
|
Total operating expenses |
11,869 | 18,040 | (6,171 | ) | (34 | )% | ||||||||||
|
Operating loss |
(11,869 | ) | (18,040 | ) | 6,171 | 34 | % | |||||||||
|
Other income (expense): |
||||||||||||||||
|
Interest income/ (expense) - royalty agreement |
6,265 | (10,364 | ) | 16,629 | 160 | % | ||||||||||
|
Fair value adjustments on derivative liabilities income/ (expense) |
485 | (300 | ) | 785 | 262 | % | ||||||||||
|
Gain on extinguishment - royalty agreement |
23,098 | - | 23,098 | 100 | % | |||||||||||
|
Other income, net |
712 | 126 | 586 | 465 | % | |||||||||||
|
Income/ (loss) before income taxes |
18,691 | (28,578 | ) | 47,269 | 165 | % | ||||||||||
|
Income tax - (expense) benefit |
- | 1,026 | (1,026 | ) | (100 | )% | ||||||||||
|
Net income (loss) |
$ | 18,691 | $ | (27,552 | ) | $ | 46,243 | (168 | )% | |||||||
Research and Development Expenses
The table below summarizes Palvella’s research and development expenses incurred by development program:
|
Year Ended December 31, |
Change |
|||||||||||||||
|
(in thousands) |
2023 |
2022 |
$ |
% |
||||||||||||
|
QTORIN rapamycin for PC and GS |
$ | 3,682 | $ | 5,684 | $ | (2,002 | ) | (35 | )% | |||||||
|
QTORIN rapamycin for microcystic LM |
164 | 961 | (797 | ) | (83 | )% | ||||||||||
|
QTORIN rapamycin CMC costs |
878 | 2,561 | (1,683 | ) | (66 | )% | ||||||||||
|
Non-program specific and unallocated research and development expenses: |
||||||||||||||||
|
Salaries and stock-based compensation |
2,383 | 2,795 | (412 | ) | (15 | )% | ||||||||||
|
Consultants |
1,229 | 1,222 | 7 | 1 | % | |||||||||||
|
Other |
457 | 661 | (204 | ) | (31 | )% | ||||||||||
|
Total research and development expenses |
$ | 8,793 | $ | 13,884 | $ | (5,091 | ) | (37 | )% | |||||||
Research and development expenses for the year ended December 31, 2023 were $8.8 million compared to $13.9 million for the year ended December 31, 2022. The decrease in research and development expenses during the year ended December 31, 2023 was due to decreased expenses incurred from the PC and GS programs as a result of the 2023 readouts of the PC and GS clinical trials as well as decreased CMC costs across all programs.
General and Administrative Expenses
For the year ended December 31, 2023, general and administrative expenses were $3.1 million compared to $4.2 million for the year ended December 31, 2022. The decrease in general and administrative expenses during the year ended December 31, 2023 was primarily due to decreases in personnel-related costs as a result of salary reductions in the second half of 2023 following the readouts of the PC and GS clinical trials.
Other Income (Expense)
Other income (expense) during the year ended December 31, 2023 was $30.6 million of income as compared to ($10.5) million of expense during the year ended December 31, 2022. During the second quarter of 2023, the Company received data from certain of its clinical trials that reduced the projected net product sales related to QTORIN rapamycin and the corresponding probabilities of successful commercialization, resulting in a significant reduction in the expected future royalty payments and a corresponding reduction in the royalty agreement liability. The Company incurred non-cash interest income (expense) related to the royalty agreement of $6.3 million and ($10.4) million for the years ended December 31, 2023 and 2022. In addition, the Company recorded a $23.1 million gain on extinguishment of the royalty agreement liability in connection with the Amended Ligand Agreement in November 2023.
Liquidity and Capital Resources
Sources of Liquidity
Since inception, Palvella’s operations have been financed primarily by aggregate net proceeds of $76.5 million from the issuance of convertible preferred stock and convertible notes, and $15.0 million received under the Ligand Agreements. Palvella will continue to be dependent upon equity and debt financings, collaborations or other sources of third-party capital at least until Palvella is able to generate positive cash flows from product sales, if ever.
Palvella incurred net losses of $4.1 million for the three months ended June 30, 2024. Palvella recorded net income of $7.7 million for the three months ended June 30, 2023. As of June 30, 2024, Palvella had an accumulated deficit of $83.0 million and cash and cash equivalents of $14.5 million. Palvella’s primary use of cash is to fund operating expenses, which consist primarily of research and development expenditures, and, to a lesser extent, general and administrative expenditures. Cash used to fund operating expenses is impacted by the timing of when Palvella pays these expenses, as reflected in the change in accounts payable and accrued expenses.
Going Concern
Palvella’s financial statements included elsewhere in the proxy statement/prospectus have been prepared on a basis which assumes Palvella is a going concern. As discussed in Note 1 to those financial statements, Palvella has incurred losses from operations and negative cash flows from operations, and does not expect to generate revenues or operating cash flows for the foreseeable future, and had stated that substantial doubt exists about Palvella’s ability to continue as a going concern for one year period following the date of this filing. Palvella’s ability to continue as a going concern may be viewed unfavorably by current and prospective investors, as well as by analysts and creditors. This may in turn make it more difficult for Palvella to raise the additional financing necessary to continue to operate Palvella’s business and Palvella may be forced to significantly alter their business strategy, substantially curtail their current operations, or cease operations altogether. If unanticipated difficulties or circumstances arise, and depending on the ultimate outcome of the Merger, Palvella may require additional capital sooner to support its operations. If Palvella is unable to complete the Merger or raise additional capital when necessary, it may be forced to further decelerate or curtail its operations until such time as additional capital becomes available, which could have a material adverse effect on it and its financial statements. There can be no assurance that management’s plans will be successful. There is no assurance that Palvella will complete the Merger and/or additional financing will be available when needed or that it will be able to obtain such financing on reasonable terms.
Pre-Close Financing and PIPE Financing
In June 2024, in contemplation of the Merger, Palvella entered into a convertible note purchase agreement under which Palvella raised $12.4 million of gross proceeds from the issuance of convertible promissory notes that are convertible into Pieris common stock based on certain conditions and events, including the consummation of the Merger Agreement as discussed above.
In connection with the Merger Agreement, certain third parties have entered into the PIPE financing as disclosed above. The PIPE financing is contingent on and will occur concurrent to the closing of the Merger, subject to customary closing conditions.
Future Funding Requirements
Palvella has not generated product revenue or achieved profitability since its inception and expects to continue to incur net losses for the foreseeable future. As of June 30, 2024, Palvella had approximately $14.5 million in cash and cash equivalents. Based on its current business plans, Palvella does not believe that its existing cash and cash equivalents will be sufficient to fund its planned operations for the one year period following the date of this filing. Moreover, Palvella expects its losses to increase as it continues to advance Palvella’s product candidates through clinical trials and regulatory submissions. Palvella may also incur expenses in connection with the in-licensing or acquisition of additional product candidates. Furthermore, upon the closing of the Merger, Palvella expects to incur additional costs associated with operating as a public company, including significant legal, accounting, investor relations and other expenses that Palvella did not incur as a private company. Palvella’s primary uses of capital have been, and Palvella expects will continue to be, compensation and related expenses, third-party clinical research, manufacturing and development services, license payments or milestone obligations that may arise, manufacturing costs, legal and other regulatory expenses and general overhead costs.
Based upon Palvella’s operating plan, Palvella believes that the anticipated net proceeds from the PIPE Financing, together with Palvella’s and Pieris’ projected available cash and cash equivalents upon the closing of the Merger, will be sufficient to fund Palvella operating expenses into the second half of 2027. However, there is no certainty that the PIPE or Merger will close and actual cash balances at closing could differ from these projected cash amounts based on actual spending through the date of closing. To continue to finance Palvella’s operations beyond that point, Palvella may need to raise additional capital, the success of which cannot be assured. Palvella has based this estimate on assumptions that may prove to be wrong, and Palvella could exhaust its available capital resources sooner than Palvella currently expects. If Palvella receives regulatory approval for QTORIN rapamycin for the treatment of microcystic LM, or any of Palvella’s future product candidates, Palvella expects to incur significant commercialization expenses related to manufacturing, sales, marketing, and distribution, or from any out licensing of the product. Palvella is also responsible for up to $5.0 million in milestone payments to Ligand under the Amended Ligand Agreement upon the achievement of certain regulatory milestones by Palvella related to QTORIN rapamycin, which may be triggered prior to the commercialization of any of Palvella’s product candidates and ability to generate revenue. Please see “—Critical Accounting Policies and Significant Judgments and Estimates—Ligand Agreement” and “Palvella’s Business-Ligand Development Funding Agreement”.
Palvella will continue to require additional capital to advance its current product candidates through clinical development, to develop, acquire or in-license other potential product candidates and to fund Palvella’s operations for the foreseeable future. Palvella may finance its cash needs through public or private equity or debt offerings or other third party sources such as strategic collaborations. However, Palvella may be unable to raise additional funds or enter into such other arrangements when needed or on terms that are acceptable to Palvella, or at all. To the extent that Palvella raises additional capital by issuing equity securities, Palvella’s existing stockholders may experience substantial dilution, and the terms of these securities may include liquidation or other preferences detrimental to the rights of Palvella common stockholders. Any agreements for future debt or preferred equity financings, if available, may involve covenants limiting or restricting Palvella’s ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends. If Palvella raises additional funds through collaborations, strategic alliances or marketing, distribution or licensing arrangements with third parties, Palvella may be required to relinquish valuable rights to its technologies, future revenue streams, research programs or product candidates, or grant licenses on terms that may not be favorable to Palvella. Palvella may seek additional capital due to favorable market conditions or strategic considerations even if Palvella believes it has sufficient funds for its current or future operating plans.
Palvella’s future funding requirements depend on many factors, including, but not limited to:
|
● |
Palvella’s ability to complete the Merger and the PIPE Financing; |
|
● |
timing and outcome of regulatory review for QTORIN rapamycin for the treatment of microcystic LM, or Palvella’s other product candidates; |
|
● |
the cost of commercialization and manufacturing activities with respect to QTORIN rapamycin and Palvella’s ability to successfully commercialize this product candidate, if approved; |
|
● |
the scope, progress, results and costs of researching and developing QTORIN rapamycin, or any future product candidates, and conducting preclinical studies and clinical trials; |
|
● |
the number and scope of clinical programs Palvella decides to pursue; |
|
● |
the cost of manufacturing Palvella’s product candidates and any products Palvella commercializes, including costs associated with developing Palvella’s supply chain; |
|
● |
the cost of commercialization activities if any of Palvella’s product candidates are approved for sale, including marketing, sales and distribution costs; |
|
● |
Palvella’s ability to establish and maintain strategic collaborations, licensing or other arrangements and the financial terms of any such agreements that Palvella may enter into; |
|
● |
the timing and sales of any future approved products, if any; |
|
● |
the potential size of the markets for Palvella’s approved products, if any; |
|
● |
the timing and amount of milestone or royalty payments due under the Ligand Agreements or under similar arrangements with any future collaboration or licensing partners; |
|
● |
the expenses needed to attract and retain skilled personnel; |
|
● |
Palvella’s need to implement additional internal systems and infrastructure, including financial and reporting systems, and other costs associated with being a public company; and |
|
● |
the costs involved in preparing, filing, prosecuting, maintaining, defending and enforcing Palvella’s intellectual property portfolio |
Further, Palvella’s development and commercialization operating plans may change, and Palvella may need additional funds to meet operational needs and capital requirements for clinical trials and other research and development activities and commercialization of QTORIN rapamycin, if approved. Because of the numerous risks and uncertainties associated with the development and commercialization of Palvella’s product candidates, Palvella may be unable to estimate the amounts of increased capital outlays and operating expenditures associated with its current and anticipated product development programs.
Cash Flows
The following table summarizes Palvella’s cash flows for the six months ended June 30, 2024 and 2023:
| Six Months Ended June 30, | ||||||||
| (in thousands) | 2024 | 2023 | ||||||
| Net cash (used in) provided by: | ||||||||
|
Operating activities |
$ | (2,705 | ) | (8,158 | ) | |||
|
Financing activities |
9,982 | - | ||||||
|
Net increase (decrease) in cash and cash equivalents |
$ | 7,187 | (8,158 | ) | ||||
Net cash used in operating activities
Net cash used in operating activities for the six months ended June 30, 2024 and June 30, 2023 consisted of net income for the period adjusted for non-cash items and changes in components of operating assets and liabilities. For the six months ended June 30, 2024, a net loss of ($6.7) million was adjusted for non-cash items of $4.0 million, including non-cash interest expense of $1.7 million, change in fair value of derivative liabilities-royalty agreement of $0.3 million, stock-based compensation expense of $0.4 million, and a net increase of $1.4 million due to changes in operating assets and liabilities. For the six months ended June 30, 2023, net income of $1.0 million was adjusted for non-cash items of $9.1 million, including non-cash interest income of $8.7 million, change in fair value of derivative liabilities-royalty agreement of $0.6 million, stock-based compensation expense of $0.3 million, and a net decrease of $0.1 million due to changes in operating assets and liabilities.
Net cash provided by financing activities
For the six months ended June 30, 2024, net cash provided by financing activities were $9.9 million, entirely attributable to proceeds from issuance of convertible notes payable of $10.0 million less issuance costs of $0.1 million.
The following table summarizes Palvella’s cash flows for the years ended December 31, 2023 and 2022:
|
Year Ended December 31, |
||||||||
|
(in thousands) |
2023 |
2022 |
||||||
|
Net cash (used in) provided by: |
||||||||
|
Operating activities |
$ | (13,703 | ) | $ | (14,840 | ) | ||
|
Financing activities |
5,000 | 9,566 | ||||||
|
Net decrease in cash and cash equivalents |
$ | (8,703 | ) | $ | (5,274 | ) | ||
Net cash used in operating activities
Net cash used in operating activities for the years ended December 31, 2023 and December 31, 2022 consisted of net income for the period adjusted for non-cash items and changes in components of operating assets and liabilities. For the year ended December 31, 2023, a net income of $18.7 million was adjusted for non-cash items of $32.3 million, including gain on extinguishment of royalty agreement of ($23.1) million, non-cash interest income of ($6.3) million, change in fair value of derivative liabilities-royalty agreement of ($0.5) million, stock-based compensation expense of $0.6 million, and a net decrease of $3.1 million due to changes in operating assets and liabilities. For the year ended December 31, 2022, a net loss of $27.6 million was adjusted for non-cash items of $12.6 million, including non-cash interest expense of $10.4 million, change in fair value of derivative liabilities-royalty agreement of $0.3 million, stock-based compensation expense of $0.4 million, and a net increase of $1.6 million due to changes in operating assets and liabilities.
Net cash provided by financing activities
For the years ended December 31, 2023 and December 31, 2022, net cash provided by financing activities consisted of $5.0 million and $9.6 million, respectively, primarily attributable to proceeds from the Amended Ligand Agreement and issuance of Series D preferred stock, respectively.
Contractual Obligations and Commitments
Leases
Palvella leases office space in Wayne, Pennsylvania. Palvella’s future lease payments for these facilities is $0.1 million for the remaining term, which shall automatically renew in October 2024.
Ligand Agreement
In December 2018, Palvella entered into the Original Ligand Agreement with Ligand, whereby Ligand agreed to make a one-time payment of $10.0 million to fund the development of QTORIN rapamycin. As partial consideration for the one-time payment, Palvella granted Ligand the right to receive up to $8.0 million in milestone payments upon the achievement of certain corporate, financing and regulatory milestones by Palvella related to QTORIN rapamycin for the treatment of any and all indications. The total amount of potential future milestone payments remaining under the arrangement were $5.0 million as of December 31, 2023. In addition, Palvella agreed to pay to Ligand tiered royalties from 5.0% to 9.8% based on any aggregate annual worldwide net product sales of any products based on QTORIN rapamycin. On a licensed product-by-licensed product and country-by-country basis, the royalty period is from the date of first commercial sale of such licensed product in a country until the latest of (i) the expiration of the last valid claim within the licensed patent rights covering such licensed product in the country in which such licensed product is made, used or sold, (ii) the expiration of the regulatory exclusivity term conferred by the applicable regulatory authority in such country with respect to such licensed product, and (iii) the fifteenth anniversary of the first commercial sale of such licensed product in such country.
In November 2023, Palvella and Ligand entered into the Amended Ligand Agreement, whereby Ligand paid Palvella an additional $5.0 million in return for an increase in the future tiered royalties to 8.0% to 9.8% of any aggregate annual worldwide net product sales of any products based on QTORIN rapamycin.
Other
Further, Palvella enters into contracts in the normal course of business with service providers for clinical trials, preclinical research studies and testing, manufacturing, and other services and products for operating purposes. Palvella’s payment obligations under these contracts generally provide for termination upon notice and, therefore, Palvella believes that its non-cancelable obligations under these agreements are not material and Palvella cannot reasonably estimate the timing of any such payments or if and when they will occur.
Palvella may also enter into additional research, manufacturing, supplier and other agreements in the future, which may require up-front payments or long-term commitments of cash.
Critical Accounting Policies and Significant Judgments and Estimates
Management’s discussion and analysis of Palvella’s financial condition and results of operations is based on the audited financial statements included elsewhere in this proxy statement/prospectus, which have been prepared in accordance with GAAP in the United States. The preparation of these financial statements requires Palvella to make estimates and assumptions that affect the reported amounts of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the financial statements, as well as the reported expenses incurred during the reporting periods. Palvella’s estimates are based on Palvella’s historical experience and on various other factors that Palvella believes are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. Palvella’s actual results may differ from these estimates under different assumptions or conditions.
While Palvella’s significant accounting policies are more fully described in the notes to the audited financial statements included elsewhere in this proxy statement/prospectus, Palvella believes that the accounting policies discussed below are critical to understanding Palvella’s historical and future performance, as these policies relate to the more significant areas involving management’s judgments and estimates.
Research and Development Expenses and Accruals
Palvella estimates costs of research and development activities conducted by service providers, which include, the conduct of sponsored research, preclinical studies and contract manufacturing activities. Palvella records the estimated costs of research and development activities based upon the estimated amount of services provided but not yet invoiced and include these costs in accrued expenses and other current liabilities or prepaid expenses and other current assets on the balance sheets and within research and development expense on the statements of operations.
Palvella estimates these costs based on factors such as estimates of the work completed and budget provided and in accordance with agreements established with Palvella’s collaboration partners and third-party service providers. Palvella makes significant judgments and estimates in determining the accrued liabilities and prepaid expense balances in each reporting period. As actual costs become known, Palvella adjusts accrued liabilities or prepaid expenses. While Palvella’s actual results could differ from their estimates, Palvella has not experienced any material differences between accrued costs and actual costs incurred since Palvella’s inception.
Palvella’s expenses related to clinical trials are based on estimates of patient enrollment and related expenses at clinical trial investigator sites as well as estimates for the services received and efforts expended pursuant to contracts with multiple research institutions that may be used to conduct and manage clinical trials on Palvella’s behalf. Palvella generally accrues expenses related to clinical trials based on contracted amounts applied to the level of patient enrollment and activity. If timelines or contracts are modified based upon changes in the clinical trial protocol or scope of work to be performed, Palvella modifies estimates of accrued expenses accordingly on a prospective basis
Ligand Agreement
Under the terms of the Ligand Agreements, Palvella received $15.0 million to fund the development of QTORIN rapamycin, in exchange for Ligand’s right to receive future payments based on the development and commercialization of products covered under the Ligand Agreements. Ligand is entitled to receive up to an additional $5.0 million of milestone payments upon the achievement of certain regulatory milestones by Palvella related to QTORIN rapamycin for the treatment of any and all indications. Palvella’s obligation to make milestone payments under the Ligand Agreements was determined to be a derivative liability, and Palvella’s obligation to make future royalty payments was determined to be a debt instrument.
The accounting for liabilities under the Ligand Agreements requires Palvella to make certain estimates and assumptions about the timing and probability of FDA approval and commercialization, and the amount of future net sales for any product containing QTORIN rapamycin. The estimated future net sales are based on subjective assumptions that include the estimated size of the addressable patient population and the anticipated pricing of the Company’s products. These assumptions are subject to significant variability, and are thus subject to significant uncertainty.
Royalty payments will be recorded as debt service payments on the royalty agreement liability. Interest expense is determined using the effective interest method based upon risk adjusted cash flow estimates of Palvella’s expected future royalty payments, yielding an effective interest rate of 38.9% and 30.3% as of December 31, 2023 and 2022, respectively. The effective interest rate is estimated at each balance sheet date based on the rate that would enable the liability to be repaid in full over the anticipated life of the arrangement. This effective interest rate will likely be subject to variability as Palvella continues the development and commercialization of Palvella’s products. The derivative liabilities — royalty agreement is classified as long term on Palvella’s balance sheet according to the estimated timing of the occurrence of potential payments.
The fair value of the derivative liabilities — royalty agreement with respect to the potential milestone payments is determined based upon the estimated probabilities and timing of the achievement of milestones, discounted to present value using Palvella’s estimated weighted average cost of capital. The assumptions used to determine the fair value of the derivative liabilities — royalty agreement at December 31, 2023 and 2022 were (a) weighted cost of capital of 25%; and (b) 50% probability of achieving regulatory approval of a product by the FDA with a term of 3.5 and 1.75 years, respectively. Gains and losses arising from changes in fair value of the derivative liabilities — royalty agreement are recognized within Palvella’s statements of operations as fair value adjustments on the derivative liabilities — royalty agreement and in the balance sheet as a non-current liability for each financial reporting period.
Palvella’s estimates and assumptions with respect to the royalty agreement liability and derivative liabilities — royalty agreement are likely to change as Palvella develops and commercializes QTORIN rapamycin, if approved. Any such adjustments that may become necessary will impact the recorded value of the royalty agreement liability and the derivative liabilities — royalty agreement, the accretion of interest expense on the royalty agreement liability and the fair value adjustments on derivative liabilities — royalty agreement.
Stock-Based Compensation
Palvella accounts for stock-based compensation in accordance with Accounting Standards Codification, Compensation-Stock Compensation, or ASC 718. In accordance with ASC 718, compensation cost is measured at estimated fair value and is included as compensation expense over the vesting period during which service is provided in exchange for the award.
Palvella uses the Black-Scholes option pricing model, or “Black-Scholes”, to determine the fair value of Palvella’s stock options. Black-Scholes utilizes various assumptions, including the fair value per share of the underlying common stock issuable upon exercise of the options, the expected life of the options, the expected stock price volatility from peer companies and the expected risk-free interest rate. These assumptions reflect Palvella’s best estimates, but they involve inherent uncertainties based on market conditions generally outside Palvella’s control.
The fair value of each stock option grant is estimated on the date of grant using the Black-Scholes option pricing model. Black-Scholes requires inputs based on certain subjective assumptions, including (i) the expected stock price volatility, (ii) the expected term of the award, (iii) the risk-free interest rate and (iv) expected dividends. Due to the lack of a public market for Palvella’s common stock and lack of company-specific historical and implied volatility data, Palvella has based its computation of expected volatility on the historical volatility of a representative group of public companies with similar characteristics to Palvella, including stage of product development and life science industry focus. The historical volatility is calculated based on a period of time commensurate with the expected term assumption. Palvella used the simplified method to calculate the expected term for options granted whereby the expected term equals the arithmetic average of the vesting term and the original contractual term of the options due to its lack of sufficient historical data. The risk-free interest rate is based on U.S. Treasury securities with a maturity date commensurate with the expected term of the associated award. The expected dividend yield is assumed to be zero as Palvella has never paid dividends and has no current plans to pay any dividends on its common stock.
As a result, if other assumptions had been used, stock-based compensation cost could have been materially impacted. Furthermore, if Palvella uses different assumptions for future grants, stock-based compensation cost could be materially impacted in future periods.
Palvella will continue to use judgement in evaluating the assumptions utilized for its stock-based compensation expense calculations on a prospective basis. In addition to the assumptions used in the Black-Scholes, Palvella’s policy is to account for forfeitures as they occur in accordance with ASC 718. Palvella reverses compensation expense cost previously recognized, in the period the award is forfeited, for an award that is forfeited before completion of the requisite service period.
Determination of Fair Value of Common Stock on Grant Date
As Palvella common stock has not been publicly traded, Palvella periodically estimated the fair value of the Palvella common stock considering, among other things, contemporaneous valuations of its common stock prepared by an unrelated third-party valuation firm in accordance with the guidance provided by the AICPA”, Valuation of Privately-Held-Company Equity Securities Issued as Compensation. Palvella’s stock valuations were prepared using either a hybrid method, where the equity value in one or more of the scenarios is calculated using an option-pricing method, or a probability-weighted expected return method, or “PWERM”, where the fair value of common stock is estimated based upon an analysis of future values for Palvella, assuming various outcomes. Under the PWERM, the common stock value is based on the probability-weighted present value of expected future investment returns considering each of the possible outcomes available as well as the rights of each class of stock. The future value of the common stock under each outcome is discounted back to the valuation date at an appropriate risk-adjusted discount rate and probability weighted to arrive at an indication of value for the common stock. In addition to considering the results of these third-party valuations, Palvella considered various objective and subjective factors to determine the price of its common stock as of each grant date, which may be as of a date later than the most recent third-party valuation date.
Recently Adopted Accounting Pronouncements
Refer to Note 2, “Summary of Significant Accounting Policies,” in the notes to Palvella’s audited financial statements for the periods ended June 30, 2024 and December 31, 2023 appearing elsewhere in this proxy statement/prospectus for a discussion of recent accounting pronouncements.
Off-Balance Sheet Arrangements
During the periods presented, Palvella did not have, nor does it currently have, any off-balance sheet arrangements as defined in the rules and regulations of the SEC.
Quantitative and Qualitative Disclosures about Market Risk
Palvella is a smaller reporting company as defined by Item 10 of Regulation S-K and is not required to provide the information otherwise required under this item.
MANAGEMENT FOLLOWING THE MERGER
Executive Officers and Directors of the Combined Company Following the Merger
The combined company’s board of directors will initially be fixed at five members, consisting of one current Pieris board member, namely Christopher Kiritsy, and four of the six current Palvella board members, namely Wesley H. Kaupinen, Todd C. Davis, George M. Jenkins and Tadd S. Wessel.
The following table lists the names and ages, as of October 28, 2024, and positions of the individuals who are expected to serve as executive officers and directors of the combined company upon completion of the Merger.
|
Name |
Age |
Position |
|
Executive Officers |
||
|
Wesley H. Kaupinen |
47 |
Chief Executive Officer, Founder, President and Director |
|
Matthew Korenberg |
49 |
Chief Financial Officer |
|
Kathleen Goin |
54 |
Chief Operating Officer |
|
Jeffrey Martini, Ph.D. |
46 |
Chief Scientific Officer |
| Kathleen A. McGowan | 63 | Vice President, Finance and Operations |
|
Non-employee Directors |
||
|
George M. Jenkins |
72 |
Chairman of the Board |
|
Todd C. Davis |
63 |
Director |
|
Christopher Kiritsy |
59 |
Director |
|
Tadd S. Wessel |
48 |
Director |
___________________
|
(1) |
Member of the Audit Committee. |
|
(2) |
Member of the Compensation Committee. |
|
(3) |
Member of the Nominating and Corporate Governance Committee. |
Wesley H. Kaupinen, Palvella’s founder, has served as Palvella’s Chief Executive Officer and as a member of the Palvella board of directors since December 2015 and was further appointed President in August 2016. Prior to founding Palvella, Mr. Kaupinen served as Senior Vice President, Corporate Development and Commercialization at Insmed, Inc., a publicly traded commercial stage biopharmaceutical company focused on developing novel therapies to treat serious rare diseases, from 2013 to August 2015. Previously, Mr. Kaupinen was a Principal at Quaker Partners, an investment firm focused on public market and venture capital investments in innovative life sciences companies, and an associate in the healthcare group at Apax Partners, a global private equity firm. Mr. Kaupinen is a member of the board of directors of Primrose Bio, a private equity-backed company focused on developing and licensing its manufacturing technologies for nucleic acids and proteins used in therapeutics and vaccines. Mr. Kaupinen previously served on the board of directors of Biocoat (acquired by GTCR), Intact Vascular (acquired by Philips), and TELA Bio (NASDAQ: TELA). Earlier in his career, Mr. Kaupinen also held commercial and general management positions at Synthes (now a part of Johnson & Johnson) and Johnson & Johnson Cordis Cardiology. Mr. Kaupinen earned an M.B.A. from The Wharton School of the University of Pennsylvania and a B.A. in Economics from the University of Virginia.
Mr. Kaupinen is qualified to serve on the combined company’s board of directors because of his knowledge of the combined company’s business, as well as his extensive leadership experience and successful record of commercial operations and product pipeline development.
Matthew Korenberg has served as Palvella’s Chief Financial Officer since October 2024. Prior to joining Palvella, Mr. Korenberg served as President and Chief Operating Officer of Ligand Pharmaceuticals Inc. from November 2022 to October 2024 and as Chief Financial Officer from August 2015 to October 2022. Before his tenure at Ligand Pharmaceuticals Inc., Mr. Korenberg was the founder, Chief Executive Officer, and a director of NeuroCircuit Therapeutics, a company focused on developing drugs to treat genetic disorders of the brain with an initial focus on Down syndrome, from September 2013. Earlier in his career, Mr. Korenberg was a Managing Director and member of the healthcare investment banking team at Goldman Sachs from July 1999 through August 2013, where he advised and financed companies in the biotechnology and pharmaceutical sectors across New York, London, and San Francisco. Prior to Goldman Sachs, Mr. Korenberg was a healthcare investment banker at Dillon, Read & Co. Inc., working with healthcare and industrial companies.
Mr. Korenberg is also a member of the board of directors and serves on the audit and compensation committees of Qualigen Therapeutics, Inc., a publicly traded company developing oncology-focused therapeutics, since May 2020. Additionally, he serves on the board of Primrose Bio, Inc., a private biotech company, since November 2023. Mr. Korenberg holds a B.B.A. in Finance and Accounting from the University of Michigan.
Kathleen Goin has served as Palvella’s Vice President, Development Operations since October 2019. From February 2017 to October 2019, Ms. Goin served as Vice President of Clinical Operations at Clinical Works, a MyClin company, a specialty consulting firm focused on helping companies start their clinical programs and execute trials. Prior to that, Ms. Goin served as Vice President, Clinical Operations of Trevena, Inc., a publicly held biotechnology company, from November 2013 to February 2017. Ms. Goin holds a Master of Science in Occupational Therapy from Misericordia University and a B.S. in Political Science from Rosemont College.
Jeffrey Martini, Ph.D., has served as Palvella’s Chief Scientific Officer since October 2024. Dr. Martini was previously Palvella’s Senior Vice President, Research and Development and Scientific Affairs from August 2020 to October 2024. Prior to joining Palvella, Dr. Martini served in various capacities, including as Executive Director, Business Development and Corporate Strategy and as Executive Director, Program Management, at Marinus Pharmaceuticals, Inc. from July 2018 to August 2020. He also served as Senior Director, Project Champion at Teva Pharmaceuticals Industries Limited from July 2013 to July 2018. Dr. Martini holds a Ph.D. in Molecular Pharmacology and Structural Biology from Jefferson University and a B.S. in Life Sciences from Pennsylvania State University.
Kathleen A. McGowan has served as Palvella’s Vice President, Finance & Operations since September 2020 and previously served as Palvella’s Chief Financial Officer from September 2018 to August 2020. Prior to joining Palvella, Ms. McGowan served as consultant for Danforth Advisors LLC, a consulting firm, from February 2018 to October 2018, and as Chief Financial Officer of VaxInnate Corporation, a privately held company developing vaccines for influenza, zika and dengue fever, from January 2013 to August 2018. Prior to VaxInnate, she led financial operations at Tobira Therapeutics (acquired by Allergan plc), a clinical-stage company developing and commercializing therapies for nonalcoholic steatohepatitis (NASH) and other liver diseases from 2008 to 2012. Earlier in her career, Ms. McGowan held senior management financial roles at Discovery Laboratories, Inc. (now Windtree Therapeutics, Inc.), Claneil Enterprises, Liposome Company (acquired by Elan Corp.), and Johnson & Johnson from 2004 to 2008, 2000 to 2004, 1996 to 2000 and 1983 to 1995, respectively. She holds an M.B.A. in Finance from Fairleigh Dickinson University and a B.S. in Finance and Accounting from Drexel University.
George M. Jenkins has served as a member of the Palvella board of directors since March 2017 and as its Chair since January 2018. From 1987 until 2005, Mr. Jenkins was a general partner of Apax Partners, a global private equity firm where he served as chief operating officer. Mr. Jenkins currently serves as a board member of several private companies, including Conventus Orthopaedics, Inc. He has previously served on the board of various public and private companies, including SkinMedica (acquired by Allergan plc), Colorescience, Sunglass Hut and Spyder Active Sports. Mr. Jenkins holds an M.B.A. from Pace University and a B.A. in Economics from Lafayette College, where he currently serves as a Trustee Emeritus.
We believe that Mr. Jenkins is qualified to serve on the combined company’s board of directors because of his extensive experience in healthcare investment management as well as his executive leadership and directorship experience.
Todd C. Davis has served as a member of the Palvella board of directors since April 2017. Mr. Davis has served as the Executive Chairman of Benuvia Holdings, Inc. since November 2019. He is the founder and managing partner of RoyaltyRx Capital, LLC, a special opportunities investment firm founded in September 2018. From 2006 until January 2018, Mr. Davis was a Co-founder and Managing Partner of Cowen/HealthCare Royalty Partners, a global healthcare investment firm. Previously, Mr. Davis was a Partner at Paul Capital Partners, where he co-managed that firm’s royalty investments as a member of the Royalty Management Committee from 2004 to 2006. He also served as a Partner responsible for biopharmaceutical growth equity investments at Apax Partners from 2001 to 2004. Mr. Davis began his business career in various sales and product management roles at Abbott Laboratories where he held several commercial roles of increasing responsibility during the period from 1990 to 1995. He subsequently held general management, business development, and licensing roles at Elan Pharmaceuticals from 1997 to 2001. He currently serves as Chief Executive Officer of Ligand and currently serves on the boards of Ligand, a publicly held biopharmaceutical company, BioDelivery Sciences International, Inc., a publicly held specialty pharmaceutical company, Vaxart, Inc., a publicly held biotechnology company, and Virocell Biologics, where he has served since 2007, 2018, 2019, and 2021, respectively. Mr. Davis holds an M.B.A. from Harvard University and a B.S. from the U.S. Naval Academy.
Mr. Davis is qualified to serve on the combined company’s board of directors because of his extensive experience in the healthcare industry and healthcare investment management as well as his experience as a director of a publicly held biopharmaceutical company.
Christopher Kiritsy has served as a member of the Pieris board of directors September 2016. Mr. Kiritsy is founder and managing member of Precision Kapital, LLC, a private investment and advisory firm. Prior to forming Precision Kapital, Mr. Kiritsy co-founded Arisaph Pharmaceuticals, Inc., or Arisaph, and served as Arisaph’s President and Chief Executive Officer from 2005 through March 2018. At Arisaph, Mr. Kiritsy oversaw the development of a broad preclinical and clinical pipeline, taking several cardiometabolic products into clinical development. Additionally, Mr. Kiritsy employed a unique, shareholder friendly financing strategy, raising nearly two thirds of all capital nondilutively through royalty monetization and grant funding. Prior to Arisaph, Mr. Kiritsy served as Executive Vice President, Corporate Development and Chief Financial Officer of Kos Pharmaceuticals, Inc., or Kos, responsible for finance, corporate communications, strategic planning, and business development functions. During his decade long tenure, Mr. Kiritsy raised approximately $500 million in public equity capital, including Kos’ IPO, and spearheaded 10 major corporate development transactions, including product acquisitions, in/out licensing and co-promotion arrangements. Mr. Kiritsy played central role in building Kos from a start-up into publicly traded, profitable, 1,000 person fully-integrated company, where Kos internally developed and commercialized the blockbuster Niaspan® franchise. Kos was acquired by Abbott Laboratories for $4 billion in 2006. Mr. Kiritsy previously served on the board of directors of HTG Molecular Diagnostics, Inc. In addition, Mr. Kiritsy previously served as a board member and audit committee chair of Melinta Pharmaceuticals, Inc., as a board member of Arisaph and as chairman of the board of Avaxia Biologics, Inc. Mr. Kiritsy received his A.B. in Biology from Bowdoin College and his M.B.A. at night from Boston University School of Business. Mr. Kiritsy is a seasoned entrepreneur, possessing 30 years of unique business and technical experience, and a track record of building successful fully integrated biopharma businesses.
Mr. Kiritsy is qualified to serve on the combined company’s board of directors based on his considerable experience in the pharmaceutical industry and his expertise in finance and corporate development.
Tadd S. Wessel has served as a member of the Palvella board of directors since January 2023. Mr. Wessel is the founder and Managing Partner of Petrichor, a private investment firm focused on the healthcare sector. He is also a founder and Managing Partner of Scion Life Sciences, an affiliate of Petrichor. Tadd has more than 25 years of experience, primarily focused on investing and building companies in the life sciences sectors. Previously, he was a Partner at OrbiMed Advisors where he led the build-out of the structured investment business. Prior to OrbiMed, Tadd was a Vice President at Fortress Investment Group focused on healthcare investments. Tadd began his career in the life sciences investment banking groups at Citigroup and Robertson Stephens. Tadd has served on more than 30 boards most recently including Aurion Biotech and ITM Isotope Technologies Munich SE. He also serves on the Advisory Board of the AIM at Melanoma Foundation, whose mission is dedicated to finding more effective treatments and, ultimately, the cure for melanoma. He also serves on the Board of the International Centers for Precision Oncology (ICPO) whose mission is to scale access of molecularly targeted precision oncology diagnostics and therapeutics for the benefit of cancer patients globally. Tadd holds an AB in biology from Princeton University.
Mr. Wessel is qualified to serve on the combined company’s Board of Directors based on his extensive experience in the healthcare and finance industries.
Each executive officer will serve at the discretion of the combined company’s board of directors. There are no family relationships among any of the combine company’s directors or executive officers.
Board of Directors of the Combined Company Following the Merger
The Pieris board of directors currently consists of eight directors divided into three staggered classes, with one class to be elected at each annual meeting to serve for a three-year term. Following the completion of the Merger, the Pieris board will consist of five members. The staggered structure of the Pieris board of directors will remain in place for the combined company following the completion of the Merger. It is anticipated that the incoming directors will be appointed to applicable vacant director seats of the combined company board of directors. There are no family relationships among any of the proposed combined company directors and officers.
Committees of the Board of Directors
The Pieris board of directors currently has the following standing committees: audit committee and compensation and management development committee. Following the completion of the Merger, the combined company will have the following standing committees: audit committee, compensation committee and nominating and corporate governance committee.
Audit Committee
Following the completion of the Merger, the members of the combined company’s audit committee are expected to be Messrs. , , and , each of whom qualifies as an independent director for audit committee purposes, as defined under the rules of the SEC and the applicable Nasdaq listing rules and has sufficient knowledge in financial and auditing matters to serve on the combined company’s audit committee. Mr. is expected to chair the audit committee. Mr. is an “audit committee financial expert” as defined in Item 407(d)(5)(ii) of Regulation S-K promulgated under the Securities Act. This designation does not impose any duties, obligations or liabilities that are greater than those generally imposed on members of the combined company’s audit committee and the combined company’s board of directors.
The combined company’s audit committee will be directly responsible for, among other things:
|
● |
selecting a firm to serve as the independent registered public accounting firm to audit the combined company’s consolidated financial statements and overseeing the retention, compensation, evaluation and, when appropriate, termination of such independent registered public accounting firm; |
|
● |
approving or, as permitted, pre-approving all audit and non-audit services to be performed by the independent registered public accounting firm; |
|
● |
ensuring the independence of the independent registered public accounting firm; |
|
● |
discussing the scope and results of the audit with the independent registered public accounting firm and reviewing, with management and that firm, the combined company’s interim and year-end operating results and related disclosures as well as critical accounting policies and practices used by the combined company; |
|
● |
monitoring and reviewing legal, regulatory, and administrative compliance to the extent affecting the combined company’s financial results; |
|
● |
establishing procedures for employees to anonymously submit concerns about questionable accounting or audit matters; |
|
● |
considering the adequacy of the combined company’s internal controls and internal audit function; |
|
● |
determining and reviewing risk assessment guidelines and policies, including cybersecurity risks, financial risk exposure, and internal controls regarding information security; |
|
● |
preparing and approving the audit committee report required to be included in the combined company’s annual proxy statement; |
|
● |
reviewing material related party transactions or those that require disclosure; and |
|
● |
reviewing quarterly and year-end earnings releases. |
Compensation Committee
Following the completion of the Merger, the members of the combined company’s compensation committee are expected to be Messrs. , and , each of whom qualifies as an independent director, as defined under applicable Nasdaq listing rules and also meets the additional, heightened independence criteria applicable to members of the compensation committee. Mr. is expected to chair the compensation committee.
The combined company’s compensation committee will be responsible for, among other things:
|
● |
reviewing and making recommendations to the combined company’s board of directors as to the combined company’s general compensation philosophy and overseeing the development and implementation of an executive compensation program and policies related to such program; |
|
● |
annually reviewing and recommending to the board of directors the corporate performance goals and objectives relevant to the compensation of the combined company’s Chief Executive Officer, and annually reviewing the performance of the combined company’s Chief Executive Officer and recommending to the combined company’s board of directors the compensation level for the combined company’s Chief Executive Officer; |
|
● |
annually reviewing and recommending to the board of directors the corporate performance goals and objectives relevant to the compensation of the combined company’s other executive officers, and annually reviewing the performance of the combined company’s other executive officers and recommending to the combined company’s board of directors the compensation level for the combined company’s other executive officers; |
|
● |
reviewing and recommending to the combined company’s board of directors the compensation of the combined company’s directors; |
|
● |
overseeing the administration of the combined company’s stock and equity incentive plans; |
|
● |
reviewing and approving, or making recommendations to the combined company’s board of directors with respect to, incentive compensation and equity plans; |
|
● |
reviewing and approving the retention or termination of any consulting firm or outside advisor to assist in the evaluation of compensation matters; and |
|
● |
evaluating and assessing potential and current compensation advisors in accordance with the independence standards identified in the applicable Nasdaq rules. |
Nominating and Corporate Governance Committee
Following the completion of the Merger, the members of the combined company’s nominating and corporate governance committee are expected to be Messrs. , and , each of whom qualifies as an independent director, as defined under applicable Nasdaq listing rules. Mr. is expected to chair the nominating and corporate governance committee.
The combined company’s nominating and corporate governance committee will be responsible for, among other things:
|
● |
developing criteria for the selection of new directors and committee membership, including policies regarding the desired knowledge, experience, skills, independence, diversity, and other characteristics of board and committee members; |
|
● |
identifying, reviewing and evaluating candidates for membership on the combined company’s board of directors, including candidates submitted by the combined company’s stockholders, and making recommendations to the combined company’s board of directors regarding nominees to fill vacancies or new positions on the combined company’s board of directors and the slate of nominees to stand for election by the combined company’s stockholders at each annual meeting of stockholders; |
|
● |
considering proposals submitted by the combined company’s stockholders and establishing any policies, requirements, criteria and procedures to facilitate stockholder communications with the combined company’s board of directors; |
|
● |
annually reviewing and recommending to the combined company’s board of directors determinations with respect to the independence of continuing and prospective directors within the meaning prescribed by the SEC and Nasdaq; |
|
● |
annually reviewing and recommending to the combined company’s board of directors (i) the assignment of directors to serve on each of the combined company’s board of directors committees, (ii) the chair of each committee and (iii) the chair of the combined company’s board of directors or lead independent director, as appropriate, and recommending additional committee members to fill vacancies or as otherwise needed; |
|
● |
reviewing all resignations tended by directors and recommending to the combined company’s board of directors the action, if any, to be taken with respect to such resignation; |
|
● |
developing, recommending and overseeing the implementation of the combined company’s corporate governance guidelines and a code of business conduct and ethics; |
|
● |
overseeing compliance with and reviewing proposed waivers of the corporate governance guidelines or the code of business conduct and ethics for directors, executive officers and other senior financial officers, and reporting on such compliance to the combined company’s board of directors; |
|
● |
overseeing the process of evaluating the performance of the combined company’s board of directors and its committees; and |
|
● |
assisting the combined company’s board of directors on corporate governance matters. |
Background and Experience of Directors; Board Diversity
When considering whether directors and nominees have the experience, qualifications, attributes or skills, taken as a whole, to enable the combined company’s board of directors to satisfy its oversight responsibilities effectively in light of the combined company’s business and structure, the combined company’s board of directors focused primarily on each person’s background and experience as reflected in the information discussed in each of the directors’ individual biographies set forth above. Palvella believes that the combined company’s directors provide an appropriate mix of experience and skills relevant to the size and nature of the combined company’s business.
Although the combined company’s board of directors has not adopted a specific policy regarding diversity in identifying director nominees, both the combined company’s nominating and corporate governance committee and the combined company’s board of directors seek the talents and backgrounds that would be most helpful to the combined company in selecting director nominees. In particular, the combined company’s nominating and corporate governance committee, when recommending director candidates to the combined company’s board of directors for nomination, may consider whether a director candidate, if elected, assists in achieving a mix of board of directors members that represents a diversity of background and experience.
Code of Business Conduct and Ethics
Following the completion of the Merger, the combined company will adopt a written code of business conduct and ethics that will apply to all of the combined company’s directors, officers and employees, including the combined company’s principal executive officer, principal financial officer, principal accounting officer or controller, or persons performing similar functions. The code of business conduct and ethics will cover fundamental ethics and compliance-related principles and practices such as accurate accounting records and financial reporting, avoiding conflicts of interest, the protection and use of the combined company’s property and information, reporting of illegal or unethical behavior, competition and fair dealing and compliance with legal and regulatory requirements. Following the completion of the Merger, a current copy of the combined company’s code of business conduct and ethics will be posted on the investor relations section of the combined company’s website. If the combined company makes any substantive amendments to, or grant any waivers from, the code of business conduct and ethics for any officer or director, the combined company will disclose the nature of such amendment or waiver on the combined company website or in a Current Report on Form 8-K.
Compensation Committee Interlocks and Insider Participation
In connection with the closing of the Merger, the combined company’s board of directors is expected to select members of the compensation committee. Each member of the compensation committee is expected to be a “non-employee” director within the meaning of Rule 16b-3 of the rules promulgated under the Exchange Act and independent within the meaning of the independent director guidelines of Nasdaq. None of the proposed combined company’s executive officers serves as a member of the board of directors or compensation committee of any entity that has one or more executive officers who is proposed to serve on the combined company’s board of directors or compensation committee following the completion of the Merger.
Non-Employee Director Compensation
Prior to the Merger, Palvella did not have a formal policy to provide any cash or equity compensation to its non-employee directors for their service on its board of directors or committees of its board of directors. In connection with closing of the Merger, it is expected that the board of directors of the combined company will adopt a non-employee director compensation policy designed to enable the combined company to attract and retain, on a long-term basis, highly qualified non-employee directors and align its directors’ interests with those of its stockholders. Employee directors will not receive additional compensation for their services as directors. It is expected that each director who is not an employee will be paid cash and equity compensation for serving on the board of directors of the combined company, the amount and terms of which have not yet been determined. The combined company will also reimburse its non-employee directors for reasonable travel and out-of-pocket expenses incurred in connection with attending the board of director and committee meetings.
CERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONS OF THE COMBINED COMPANY
Pieris Transactions
Pieris’ Audit Committee Charter requires the Audit Committee to review, consider, and approve in advance all future transactions, in which Pieris is a participant, that involve amounts that equal or exceed $120,000 and in which any Related Person has or will have a direct or indirect material interest in such transaction. Related Persons include any of Pieris’ directors, executive officers, holder of 5% or more of any class of Pieris capital stock, or any member of the immediate family of, or entities affiliated with, any of them, or any other related persons, as defined in Item 404 of Regulation S-K. In approving or rejecting any such proposal, Pieris’ Audit Committee is to consider all available information deemed relevant by the Audit Committee, including, but not limited to, the extent of the related person’s interest in the transaction, and whether the transaction is on terms no less favorable to Pieris than terms Pieris could have generally obtained from an unaffiliated third party under the same or similar circumstances.
Related Person Transactions
On July 23, 2024, BVF, which holds more than 5% of Pieris’ voting securities, along with other PIPE Investors, entered into the Purchase Agreement with Pieris, pursuant to which such PIPE Investors have agreed to subscribe for and purchase (either for cash or in exchange for the termination and cancellation of outstanding convertible notes issued by Palvella), and Pieris has agreed to issue and sell to the PIPE Investors, an aggregate of approximately 3,154,241 of shares of Pieris common stock at a price per share equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, and/or in lieu of Pieris common stock to certain purchasers who so choose, Pre-Funded Warrants to purchase up to 2,592,585 shares of the combined company common stock at a purchase price per Pre-Funded Warrant equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, minus $0.001. Each of BVF, Samsara Biocapital, L.P., Averill Master Fund, Ltd. and Integrated Finance Group have agreed to purchase shares pursuant to the Purchase Agreement and, together with each of their respective affiliates, are expected to be beneficial owners of 5% or more than the outstanding shares of Pieris following the PIPE Financing.
Other than the foregoing, since July 1, 2022, there has not been, nor is there currently proposed, any transaction to which Pieris is or was a party in which the amount involved exceeds the lesser of $120,000 and 1% of the average of Pieris’ total assets at year-end for the last two completed fiscal years, and in which any of Pieris’ directors, executive officers, holders of more than 5% of any class of Pieris’ voting securities or any of their respective affiliates or immediate family members, had, or will have, a direct or indirect material interest.
Indemnification Agreements with Directors and Executive Officers
Pieris has entered into indemnification agreements with each of its directors and executive officers. Each of those indemnification agreements is in the form approved by the Pieris board of directors. Those indemnification agreements require that, under the circumstances and to the extent provided for therein, Pieris indemnifies such persons to the fullest extent permitted by applicable law against certain expenses and other amounts incurred by any such person as a result of such person being made a party to certain actions, suits, and proceedings by reason of the fact that such person is or was a director, officer, employee, or agent of Pieris, any entity that was a predecessor corporation of Pieris, or any of Pieris’ affiliates. The rights of each person who is a party to such an indemnification agreement are, in addition to any other rights such person may have under applicable Nevada law, Pieris’ amended and restated articles of incorporation, Pieris’ amended and restated bylaws, any other agreement, a vote of Pieris stockholders, a resolution adopted by the Pieris board of directors, or otherwise.
Director Independence
The Pieris board of directors undertook a review of the composition of the board and independence of each of Pieris’ current and former directors. Based upon information requested from and provided by each director concerning his or her background, employment and affiliations, including family relationships, the Pieris board of directors has determined that each of James Geraghty, Michael Richman, Peter Kiener, D.Phil., Christopher Kiritsy, Ann Barbier, M.D., Ph.D., Matthew L. Sherman, M.D., and Maya R. Said, Sc.D. qualify as “independent” as that term is defined by Nasdaq Listing Rule 5605(a)(2). Stephen S. Yoder does not qualify as “independent” under applicable Nasdaq Listing Rules applicable to the board of directors generally or to separately designated board committees because he currently serves as Pieris’ Chief Executive Officer. In making such determinations, the Pieris board of directors considered the relationships that each of its non-employee directors has with Pieris, and all other facts and circumstances deemed relevant in determining independence, including the beneficial ownership of Pieris capital stock by each non-employee director.
Palvella Transactions
The following includes a summary of transactions since January 1, 2021, to which Palvella has been a party in which the amount involved exceeded or will exceed the lesser of (i) $120,000 and (ii) 1% of the average of Palvella’s total assets at year-end for the prior two fiscal years, and in which any of Palvella’s directors, executive officers or, to Palvella’s knowledge, beneficial owners of more than 5% of Palvella capital stock or any member of the immediate family of any of the foregoing persons had or will have a direct or indirect material interest, other than equity and other compensation, termination, change in control and other arrangements, which are described under “Palvella Executive Compensation.” Palvella also describes below certain other transactions with its directors, executive officers and holder of more than 5% of Palvella capital stock.
Series D Preferred Stock Financing
In December 2022, Palvella sold an aggregate of 1,835,227 shares of Palvella’s Series D preferred stock at a purchase price of $5.2879 per share for aggregate gross cash proceeds of approximately $9.7 million. At the Effective Time, each share of Palvella’s Series D preferred stock will convert into the right to receive a number of shares of Pieris common stock (subject to the payment of cash in lieu of fractional shares) calculated in accordance with the exchange ratio set forth in the Merger Agreement.
The following table summarizes the Series D preferred stock purchased by certain members of the Palvella board of directors or their affiliates and holders of more than 5% of Palvella’s outstanding capital stock. The terms of these purchases were the same for all purchasers of Palvella’s Series D preferred stock. Please refer to the section entitled “Palvella Principal Stockholders” for more details regarding the shares held by certain of these purchasers.
|
Name of Stockholder |
Shares of Series D Preferred Stock |
Total Purchase Price |
||||
|
Entities affiliates with Petrichor Healthcare Capital Management (1) |
945,555 | $5,000,000 | ||||
|
Samsara BioCapital L.P.(2) |
189,111 | $1,000,000 | ||||
|
Entities affiliates with BVF Partners (3) |
94,555 | $499,998 | ||||
|
George M. Jenkins(4) |
28,367 | $150,002 | ||||
|
Wesley H. Kaupinen(5) |
17,034 | $90,074 | ||||
|
Kathleen A. McGowan (6) |
1,127 | $5,959 |
___________________
|
(1) |
Consists of (i) 288,300 shares of Series D Preferred Stock held by Petrichor Opportunities Fund I Intermediate LP and (ii) 657,255 shares of Series D Preferred Stock held by Petrichor Opportunities Fund I LP. Tadd S. Wessel, a member of the Palvella board of directors, is a managing partner of Petrichor Healthcare Capital Management, the investment manager of Petrichor Opportunities Fund I Intermediate LP and Petrichor Opportunities Fund I LP. |
|
(2) |
Aditya Asokan, Ph.D. is currently a member of the Palvella board of directors, and is affiliated with Samsara BioCapital GP, LLC. |
|
(3) |
Consists of (i) 526,075 shares of Series D Preferred Stock held by Biotechnology Value Fund II, L.P., (ii) 50,810 shares of Series D Preferred Stock held by Biotechnology Value Fund L.P., and (iii) 4,471 shares of Series D Preferred Stock held by Biotechnology Value Trading Fund OS, L.P. BVF Partners was a holder of more than 5% of our outstanding capital stock at the time of the consummation of the Series D preferred stock financing. |
|
(4) |
George Jenkins is a member of the Palvella board of directors. |
|
(5) |
Wesley H. Kaupinen is Palvella’s President and Chief Executive Officer and a member of the Palvella board of directors. |
|
(6) |
Kathleen A. McGowan is Palvella’s Vice President, Finance and Operations. |
Convertible Note Financing
Between June and July 2024, Palvella issued and sold convertible notes in the aggregate principal amount of $12,433,000. Simple interest accrues on the outstanding principal amount of the convertible notes at an annual rate of SOFR plus 2.0% per annum. Unless earlier converted, the maturity date is the earliest to occur of (i) the date that Palvella receives approval of an NDA by the FDA of the QTORIN rapamycin in the United States, or (ii) the date that is July 3, 2027. Upon the closing of the PIPE Financing, the entire outstanding principal amount and unpaid accrued interest on the convertible notes will automatically convert into the common stock of Pieris at the same price per share of the Pieris common stock issued in a PIPE Financing.
The following table summarizes the convertible notes purchased by certain members of the Palvella board of directors or their affiliates and holders of more than 5% of Palvella’s outstanding capital stock. The terms of these purchases were the same for all purchasers of Palvella’s convertible notes.
|
Name of Noteholder |
Principal Amount of Convertible Notes |
||
|
Petrichor Opportunities Fund I LP. (1) |
$2,500,000 | ||
|
Ligand Pharmaceuticals Incorporated (2) |
$2,500,000 | ||
|
Todd C. Davis (2) |
$500,000 | ||
|
George M. Jenkins (3) |
$150,000 | ||
|
Wesley H. Kaupinen (4) |
$20,000 |
___________________
|
(1) |
Tadd S. Wessel, a member of the Palvella board of directors, is a managing partner of Petrichor Healthcare Capital Management, the investment manager of Petrichor Opportunities Fund I LP |
|
(2) |
Todd C. Davis is a member of the Palvella board of directors and the Chief Executive Officer of Ligand Pharmaceuticals Incorporated. |
|
(3) |
George M. Jenkins is a member of the Palvella board of directors. |
|
(4) |
Wesley H. Kaupinen is Palvella’s President and Chief Executive Officer and a member of Palvella board of directors. |
Transactions with Ligand Pharmaceuticals, Inc.
In December 2018, Palvella entered into the Original Ligand Agreement with Ligand whereby Ligand made a one-time payment of $10.0 million to fund the development of QTORIN rapamycin. In November 2023, Palvella entered into the Amended Ligand Agreement whereby Ligand made an additional one-time payment of $5.0 million to fund the development of QTORIN rapamycin. Under the Amended Ligand Agreement, Ligand is entitled to receive up to $8.0 million in milestone payments upon the achievement of certain milestones by Palvella related to QTORIN rapamycin for the treatment of any and all indications, of which $5.0 million of potential future milestone payments remain under the arrangement. In addition, Palvella agreed to pay to Ligand tiered royalties from 8.0% to 9.8% based on any aggregate annual worldwide net product sales of any products based on QTORIN rapamycin. The Amended Ligand Agreement includes an option for Ligand to purchase additional product revenue participation rights from Palvella over period of time (as set forth in the Amended Ligand Agreement). The option allows Ligand, for each product developed on the QTORIN platform that completes the first human clinical trial in the United States, the opportunity to make a certain upfront payments to Palvella in return for a specified royalty rate (as set forth in the Amended Ligand Agreement). Palvella’s obligation to make future milestone payments under the Amended Ligand Agreement was determined to be a derivative liability and Palvella’s obligation to make future royalty payments was determined to be a debt instrument. Todd C. Davis is a member of Palvella’s board of directors and is the Chief Executive Officer of Ligand.
Stock Option Grants to Executive Officers and Directors
Palvella has granted options to its executive officers and certain of its directors as more fully described in the section entitled “Palvella Executive Compensation.”
Employment Agreements
Employee Agreements
Palvella has entered into employment agreements, offer letters and/or severance agreements with each of Palvella’s NEOs. See “Palvella Executive Compensation—Palvella Named Executive Officer Employment Agreements” for a further discussion of these arrangements.
Policies and Procedures for Related Party Transactions
The combined company will adopt a written related party transaction policy, to be effective upon the completion of this Merger, setting forth the policies and procedures for the review and approval or ratification of related-party transactions. This policy will cover any transaction, arrangement or relationship or any series of similar transactions, arrangements or relationships, in which the combined company were or are to be a participant and a related party had or will have a direct or indirect material interest, as determined by the audit committee of combined company’s board of directors, including, without limitation, purchases of goods or services by or from the related party or entities in which the related party has a material interest, and indebtedness, guarantees of indebtedness or employment by the combined company of a related party.
All related party transactions described in this section occurred prior to adoption of this policy and as such, these transactions were not subject to the approval and review procedures set forth in the policy. However, these transactions were reviewed and approved by The Palvella board of directors. The Palvella board of directors reviews and approves transactions with directors, officers and holders of 5% or more of Palvella’s voting securities and their affiliates, each a related party. Prior to this Merger, the material facts as to the related party’s relationship or interest in the transaction are disclosed to The Palvella board of directors prior to their consideration of such transaction, and the transaction is not considered approved by The Palvella board of directors unless a majority of the directors who are not interested in the transaction approve the transaction. Further, when Palvella stockholders are entitled to vote on a transaction with a related party, the material facts of the related party’s relationship or interest in the transaction are disclosed to the stockholders, who must approve the transaction in good faith.
UNAUDITED PRO FORMA CONDENSED CONSOLIDATED COMBINED FINANCIAL INFORMATION
The following unaudited pro forma condensed consolidated combined financial information is provided to aid you in your analysis of the financial aspects of the Merger and related transactions and presents the combination of the financial information of Pieris and Palvella adjusted to give effect to the Merger and related transactions, or collectively, the Pro Forma Adjustments. Capitalized terms included but not defined below have the same meaning as defined elsewhere in this proxy statement/prospectus.
On July 23, 2024, Pieris, Merger Sub, and Palvella entered into the Merger Agreement, pursuant to which, and subject to the satisfaction or waiver of the conditions set forth in the Merger Agreement, Merger Sub will merge with and into Palvella, with Palvella continuing as a wholly- owned subsidiary of Pieris. After the completion of the Merger, Pieris will change its corporate name to “Palvella Therapeutics, Inc.” The Merger is intended to qualify for federal income tax purposes as a tax-free reorganization under the provisions of Section 368(a) of the Internal Revenue Code of 1986, as amended. If the Merger is completed, the business of Palvella will continue as the business of the combined company.
Subject to the terms and conditions of the Merger Agreement, at the effective time of the Merger, each outstanding share of Palvella capital stock (including shares of Palvella common stock and Palvella preferred stock) (excluding dissenting shares) will be converted solely into the right to receive a number of shares of Pieris common stock calculated in accordance with the Merger Agreement, equal to the exchange ratio calculated in accordance with the Merger Agreement. Based on Pieris’ and Palvella’s capitalization as of July 23, 2024, the date the Merger Agreement was executed, the exchange ratio is estimated to be approximately 0.315478222 shares of Pieris common stock for each share of Palvella common stock. Immediately after the Merger, but without giving effect to the PIPE Financing, Pieris securityholders as of immediately prior to the Merger are expected to own approximately 18% of the outstanding shares of capital stock of the combined company and Palvella securityholders as of immediately prior to the Merger are expected to own approximately 82% of the outstanding shares of capital stock of the combined company, in each case, on a fully-diluted basis, calculated using the treasury stock method, and subject to certain assumptions, including, but not limited to a valuation for Pieris equal to $21.0 million, provided, that (a) if Pieris’ net cash as of the closing is greater than $11 million, then Pieris’ valuation will be adjusted upwards on a dollar-for-dollar basis by the difference of: (i) Pieris’ net cash, minus (ii) $11 million, and (b) if Pieris’ net cash is less than $11 million, then Pieris’ valuation will be adjusted downwards on a dollar-for-dollar basis by the difference of: (i) $11 million, minus (ii) Pieris’ net cash, as further described in the Merger Agreement.
Because, among other factors, the number of shares of Pieris common stock issuable to Palvella’s securityholders is determined based on Pieris’ net cash balance as of the close of business on the last business day prior to the anticipated closing date of the Merger, Pieris cannot be certain of the exact number of shares that will be issued to (or reserved for issuance to) Palvella’s stockholders. The exchange ratio referenced above is an estimate only and the final exchange ratio will be determined pursuant to a formula described in detail in the Merger Agreement and included elsewhere in this proxy statement/prospectus.
Each stock option granted under the Palvella Stock Plan that is outstanding immediately prior to the effective time of the Merger will be assumed by Pieris and will become an option to acquire, on the same terms and conditions as were applicable to such Palvella stock option immediately prior to the effective time of the Merger, a number of shares of Pieris common stock equal to the number of shares of Palvella’s common stock subject to the unexercised portion of the Palvella stock option immediately prior to the effective time of the Merger, multiplied by the exchange ratio (rounded down to the nearest whole share number) with an exercise price per share for the options equal to the exercise price per share of such Palvella stock option immediately prior to the effective time of the Merger divided by the exchange ratio (rounded up to the nearest whole cent). Such assumed options will continue to be governed by the terms and conditions of the Palvella Stock Plan.
The pre-Merger employment agreements for the two Pieris executives also included severance, bonus and retention payments, the aggregate of which will be treated as pre-combination compensation expense of Pieris and is included in the liabilities assumed by Palvella upon closing of the Merger. In addition, certain non-executive Pieris employees entered into separation agreements prior to Merger negotiations with Palvella, pursuant to which they are entitled to severance, bonus, and retention payments. These payments will be treated as pre-combination compensation expense of Pieris and will also be included in the liabilities assumed by Palvella upon closing of the Merger.
Contingent Value Rights Agreement
At or prior to the effective time of the Merger, Pieris will enter into a CVR Agreement with a rights agent (the “Rights Agent”) and a CVR holder representative, pursuant to which each holder of record of shares of Pieris’ common stock and preferred stock entitled to receive a dividend in accordance with the terms of such preferred stock will receive the right to one contingent value right (each, a “CVR”) for each outstanding share of Pieris’ common stock held by such stockholder, or share of common stock underlying such preferred stock, held by such stockholder, on such date. Furthermore, to the extent a holder of a Pieris warrant outstanding as of the date of the CVR Agreement exercises such Pieris warrant after the date of the CVR Agreement, a number of CVRs equal to the number of shares of Pieris common stock issued to such holder in connection with such exercise shall be issued to such holder, subject to and in accordance with the terms and conditions of such Pieris Warrant, as applicable.
Each CVR will represent the contractual right to receive cash payments (net of certain expenses and taxes) upon the receipt of payments by Pieris or any of its affiliates under certain strategic partner agreements, including existing collaboration agreements pursuant to which Pieris may be entitled to milestones and royalties in the future and other out-licensing agreements for certain of Pieris’ legacy assets, and upon the receipt of certain research and development tax credits in favor of Pieris or any of its affiliates, in each case as set forth in, and subject to and in accordance with the terms and conditions of, the CVR Agreement. Pursuant to the terms of the CVR Agreement, holders of CVRs would be entitled to cash payments (net of certain expenses and taxes) upon the receipt by Pieris or any of its affiliates of certain payments under any license, collaboration or similar agreement involving any legacy assets of Pieris entered into prior to the effective date of the CVR Agreement. Pieris retains the ability under the Merger Agreement to seek to enter into such agreements related to its legacy assets and intellectual property, including PRS-400 and PRS-220, though currently does not have any such agreements in place and does not have any plans to divest any material assets. Pieris continues to explore potential transactions for PRS-400 and PRS-220. Management has determined that there is no value associated with the CVRs as the likelihood of any payments received in connection with Pieris’ legacy assets is remote. Unless earlier terminated by the representative of the CVR holders and the combined company, the CVR Agreement will terminate on the date on which no strategic partner agreement is in effect and no payments are payable to Pieris or any of its affiliates under any Strategic Partner Agreement.
The contingent payments in respect of the CVRs, if they become payable, will become payable to the Rights Agent for subsequent distribution to the holders of the CVRs. In the event that no such proceeds are received, holders of the CVRs will not receive any payment pursuant to the CVR Agreement. There can be no assurance that holders of CVRs will receive any amounts with respect thereto. The right to the contingent payments contemplated by the CVR Agreement is a contractual right only and will not be transferable, except in the limited circumstances specified in the CVR Agreement. The CVRs will not be evidenced by a certificate or any other instrument and will not be registered with the SEC. The CVRs will not have any voting or dividend rights and will not represent any equity or ownership interest in Pieris or any of its affiliates. No interest will accrue on any amounts payable in respect of the CVRs.
Management concluded that these CVRs represent financial instruments that are to be accounted for under the fair value option election in ASC 825, Financial Instruments. Under the fair value option election, the CVRs are initially measured at the aggregate estimated fair value of the CVRs and will be subsequently remeasured at estimated fair value on a recurring basis at each reporting period date. The estimated fair value of the CVR liability was determined using the discounted cash flow method to estimate future cash flows associated with Pieris’ legacy assets, including the expected milestone and royalty payments associated with Pieries’ legacy assets. Changes in fair value of the CVR liability will be presented in the consolidated statements of operations and comprehensive income (loss). The liability value is based on significant inputs not observable in the market such as estimated cash flows, estimated probabilities of success, and risk-adjustment discount rates, which represent a Level 3 measurement within the fair value hierarchy. Management has concluded that there is no value associated with the CVRs as the likelihood of any payments received in connection with Pieris’ legacy assets is remote.
Palvella Private Financing and PIPE Financing
On June 6, 2024, Palvella initiated a sequence of convertible notes with certain investors via the Convertible Note Purchase Agreement, or the Note Purchase Agreement. Under the Note Purchase Agreement, the investors have committed to extend credit to Palvella, providing up to a total of $20 million, or the Authorized Principal Amount, via convertible promissory notes. Through the issuance date of this filing, the Company has received $12.4 million of gross proceeds in exchange for convertible promissory notes issued. The convertible note bears an annual interest of 2.0% plus SOFR and shall be due and payable upon the earlier to occur of June 2027 or certain events defined in the Note Purchase Agreement. Under certain circumstances, the convertible note is convertible at the option of requisite holders into the Company’s equity securities at defined conversion prices. The terms of the convertible note specify that upon the consummation of the Merger, all outstanding principal and any unpaid accrued interest on the notes shall be automatically converted into common stock.
On July 23, 2024, concurrently with the execution and delivery of the Merger Agreement, the PIPE Investors entered into the Purchase Agreement with Pieris, pursuant to which such PIPE Investors have agreed to subscribe for and purchase (either for cash or in exchange for the termination and cancellation of outstanding convertible notes issued by Palvella), and Pieris has agreed to issue and sell to the PIPE Investors, an aggregate of up to 3,154,241 of shares of Pieris common stock at a price per share equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, and/or in lieu of Pieris common stock to certain purchasers who so choose, Pre-Funded Warrants to purchase up to 2,592,585 shares of the combined company common stock at a purchase price per Pre-Funded Warrant equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, minus $0.001. The gross proceeds from the PIPE Financing are expected to be approximately $78.9 million, consisting of approximately $60.0 million in cash and the conversion of approximately $18.9 million of principal and interest payable under the outstanding convertible notes issued by Palvella, before paying estimated expenses.
The Merger is expected to be accounted for as a reverse recapitalization in accordance with GAAP. Under this method of accounting, Pieris, which is the legal acquirer, is treated as the “acquired” company for financial reporting purposes and Palvella is treated as the accounting acquirer. This determination was primarily due to Pieris being determined to be a shell company at the time of the Merger, immediately following the dividend of the CVR and following the discontinuation of historical research and development efforts, because it will not meet the GAAP definition of a business, will not have more than nominal assets, and will not have more than nominal operations. Further, it is expected that, immediately following the Merger, Palvella’s stockholders will have a majority of the voting power of the combined company, Palvella will control four of five seats on the board of directors of the combined company, and Palvella’s senior management will comprise all of the senior management of the combined company. Accordingly, for accounting purposes, the Merger will be treated as the equivalent of a capital transaction in which Palvella is issuing stock for the net assets of Pieris. The net assets of Pieris will be stated at historical cost, with no goodwill or other intangible assets recorded.
The following unaudited pro forma condensed consolidated combined balance sheet as of June 30, 2024 combines the historical condensed consolidated balance sheet of Pieris as of June 30, 2024 with the historical balance sheet of Palvella as of June 30, 2024 giving further effect to the Pro Forma Adjustments, as if they had been consummated as of June 30, 2024.
The following unaudited pro forma condensed consolidated combined statements of operations for the year ended December 31, 2023 combine the historical condensed consolidated statement of operations of Pieris for the year ended December 31, 2023 and the historical statements of operations of Palvella for the year ended December 31, 2023, giving effect to the Pro Forma Adjustments as if they had been consummated on January 1, 2023, the beginning of the earliest period presented.
The following unaudited pro forma condensed consolidated combined statements of operations for the six months ended June 30, 2024 combine the historical condensed consolidated statement of operations of Pieris for the six months ended June 30, 2024 and the historical statements of operations of Palvella for the six months ended June 30, 2024, giving effect to the Pro Forma Adjustments as if they had been consummated on January 1, 2023, the beginning of the earliest period presented.
The unaudited pro forma condensed consolidated combined financial statements have been derived from and should be read in connection with:
|
● |
the accompanying notes to the unaudited pro forma condensed consolidated combined financial statements; |
|
● |
the historical unaudited condensed consolidated financial statements of Pieris as of and for the six months ended June 30, 2024 and the related notes included elsewhere in this proxy statement/prospectus; |
|
● |
the historical unaudited condensed financial statements of Palvella as of and for the six months ended June 30, 2024 and the related notes included elsewhere in this proxy statement/prospectus; |
|
● |
the historical audited consolidated financial statements of Pieris as of and for the year ended December 31, 2023 and the related notes included elsewhere in this proxy statement/prospectus; |
|
● |
the historical audited financial statements of Palvella as of and for the year ended December 31, 2023 and the related notes included elsewhere in this proxy statement/prospectus; |
|
● |
the sections entitled “Pieris Management’s Discussion and Analysis of Financial Condition and Results of Operations,” “Palvella Management’s Discussion and Analysis of Financial Condition and Results of Operations,” and other financial information relating to Pieris and Palvella included elsewhere in this proxy statement/prospectus. |
The unaudited pro forma condensed consolidated combined financial information is based on the assumptions and adjustments that are described in the accompanying notes. The accounting for the Merger requires the financial calculation of Pieris’ net cash. Accordingly, the pro forma adjustments are preliminary, subject to further revision as additional information becomes available and additional analyses are performed, and have been made solely for the purpose of providing unaudited pro forma condensed consolidated combined financial information. Differences between these preliminary estimates and the final accounting, expected to be completed after the closing of the Merger, will occur and these differences could have a material impact on the accompanying unaudited pro forma condensed consolidated combined financial information and the combined company’s future results of operations and financial position.
The unaudited pro forma condensed combined consolidated financial information does not give effect to the potential impact of current financial conditions, regulatory matters, operating efficiencies or other savings or expenses that may be associated with the integration of the two companies. The unaudited pro forma condensed consolidated combined financial information is not necessarily indicative of the financial position or results of operations in the future periods or the result that actually would have been realized had Pieris and Palvella been a combined organization during the specified periods. The actual results reported in periods following the Merger may differ significantly from those reflected in the unaudited condensed consolidated combined pro forma financial information presented herein for a number of reasons, including, but not limited to, differences in the assumptions used to prepare this unaudited pro forma condensed consolidated combined financial information.
UNAUDITED PRO FORMA CONDENSED CONSOLIDATED COMBINED BALANCE SHEET AS OF JUNE 30, 2024
(in thousands)
| Historical | ||||||||||||||||||||||
| Palvella | Pieris |
Private Financing Adjustments |
Note 4 |
Transaction Accounting Adjustments |
Note 4 | Pro Forma Combined Total | ||||||||||||||||
|
Asset |
||||||||||||||||||||||
|
Current assets: |
||||||||||||||||||||||
|
Cash and cash equivalents |
$ | 14,537 | $ | 19,731 | $ | 2,399 |
(a) |
$ | 60,001 |
(b) (f) |
$ | 96,668 | ||||||||||
|
Accounts receivable |
- | 1,485 | - | - | 1,485 | |||||||||||||||||
|
Receivable from public grants |
- | 3,049 | - | - | 3,049 | |||||||||||||||||
|
Other receivables |
- | 137 | - | - | 137 | |||||||||||||||||
|
Assets held for sale, property and equipment |
- | - | - | - | - | |||||||||||||||||
|
Deferred transaction costs |
500 | - | - | (500 | ) |
(f) |
- | |||||||||||||||
|
Prepaid expenses and other current assets |
307 | 649 | - | - | 956 | |||||||||||||||||
|
Total Current Assets |
15,344 | 25,051 | 2,399 | 59,501 | 102,295 | |||||||||||||||||
|
Intellectual property |
- | - | - | - | - | |||||||||||||||||
|
Total assets |
$ | 15,344 | $ | 25,051 | $ | 2,399 | $ | 59,501 | $ | 102,295 | ||||||||||||
|
Liabilities, convertible preferred stock and stockholders’ equity (deficit) |
||||||||||||||||||||||
|
Current liabilities: |
||||||||||||||||||||||
|
Accounts Payable |
$ | 1,446 | $ | 969 | $ | - | $ | - | $ | 2,415 | ||||||||||||
|
Accrued expenses and other current liabilities |
2,949 | 5,377 | - | 49 |
(c) (f) |
8,375 | ||||||||||||||||
|
Total Current Liabilities |
4,395 | 6,346 | - | 49 | 10,790 | |||||||||||||||||
|
Royalty agreement liability |
9,802 | - | - | - | 9,802 | |||||||||||||||||
|
Derivative liabilities – royalty agreement |
1,343 | - | - | - | 1,343 | |||||||||||||||||
|
Convertible promissory notes |
10,029 | - | 2,399 |
(a) |
(12,428 | ) |
(d) |
- | ||||||||||||||
|
Total liabilities |
25,569 | 6,346 | 2,399 | (12,379 | ) | 21,935 | ||||||||||||||||
|
Palvella convertible preferred stock |
70,603 | - | - | (70,603 | ) |
(e) |
- | |||||||||||||||
|
Pieris convertible preferred stock |
- | - | - | - | - | |||||||||||||||||
|
Stockholders’ equity (deficit): |
||||||||||||||||||||||
|
Palvella common stock, $0.00001 par value |
- | - | - | - | - | |||||||||||||||||
|
Pieris common stock |
- | 1 | - | 13 |
(b) (d) (e) |
14 | ||||||||||||||||
|
Additional paid-in capital |
2,181 | 342,586 | - | (180,611 | ) |
(b) (d) (e) (f) (g) (h) |
164,156 | |||||||||||||||
|
Accumulated other comprehensive (loss) income |
- | (436 | ) | - | 436 |
(g) |
- | |||||||||||||||
|
Accumulated deficit |
(83,009 | ) | (323,446 | ) | - | 322,646 |
(c) (f) (h) |
(83,809 | ) | |||||||||||||
|
Total stockholders’ equity (deficit) |
(10,255 | ) | 18,705 | - | 71,880 | 80,360 | ||||||||||||||||
|
Total liabilities, convertible preferred stock and stockholders’ equity |
$ | 15,344 | $ | 25,051 | $ | 2,399 | $ | 59,501 | $ | 102,295 | ||||||||||||
See accompanying notes to the unaudited pro forma condensed combined financial statements
UNAUDITED PRO FORMA CONDENSED CONSOLIDATED COMBINED STATEMENT OF OPERATIONS
FOR THE SIX MONTHS ENDED JUNE 30, 2024
(in thousands, except share and per share data)
| Historical | ||||||||||||||||||||||
| Palvella | Pieris | Private Financing Adjustments | Note 4 |
Transaction |
Note 4 | Pro Forma Combined Total | ||||||||||||||||
|
Revenue: |
||||||||||||||||||||||
|
Customer revenue |
$ | - | $ | 6 | $ | - | $ | - | $ | 6 | ||||||||||||
|
Collaboration revenue |
- | 47 | - | - | 47 | |||||||||||||||||
|
Total revenue |
- | 53 | - | - | 53 | |||||||||||||||||
|
Operating expenses: |
||||||||||||||||||||||
|
Research and development |
2,426 | 1,969 | - | - | 4,395 | |||||||||||||||||
|
General and administrative |
2,241 | 7,564 | - | - | 9,805 | |||||||||||||||||
|
Asset impairment |
- | - | - | - | - | |||||||||||||||||
|
Total operating expenses |
4,667 | 9,533 | - | - | 14,200 | |||||||||||||||||
|
Operating loss |
(4,667 | ) | (9,480 | ) | - | - | (14,147 | ) | ||||||||||||||
|
Other income (expense), net: |
||||||||||||||||||||||
|
Interest income (expense) |
(1,747 | ) | 441 | - | - | (1,306 | ) | |||||||||||||||
|
Interest income (expense) – convertible notes payable |
(29 | ) | - | - | 29 |
(i) |
- | |||||||||||||||
|
Fair value adjustments on derivative liabilities |
(329 | ) | - | - | - | (329 | ) | |||||||||||||||
|
Other income (loss) |
64 | 557 | - | - | 621 | |||||||||||||||||
|
Total other income (expense), net |
(2,041 | ) | 998 | - | 29 | (1,014 | ) | |||||||||||||||
|
Net Income (loss) |
(6,708 | ) | (8,482 | ) | - | 29 | (15,161 | ) | ||||||||||||||
|
Net profit (loss) per share |
||||||||||||||||||||||
|
Basic |
$ | (1.24 | ) | $ | (6.69 | ) | $ | (1.09 | ) | |||||||||||||
|
Diluted |
$ | (1.24 | ) | $ | (6.69 | ) | $ | (1.09 | ) | |||||||||||||
|
Weighted average number of common shares outstanding |
||||||||||||||||||||||
|
Basic |
5,720,009 | 1,268,000 |
(j) |
13,874,627 | ||||||||||||||||||
|
Diluted |
5,720,009 | 1,268,000 |
(j) |
13,874,627 | ||||||||||||||||||
See accompanying notes to the unaudited pro forma condensed combined financial statements
UNAUDITED PRO FORMA CONDENSED CONSOLIDATED COMBINED STATEMENT OF OPERATIONS
FOR THE YEAR ENDED DECEMBER 31, 2023
(in thousands, except share and per share data)
| Historical | ||||||||||||||||||||||
| Palvella | Pieris | Private Financing Adjustments | Note 4 | Transaction Accounting Adjustments | Note 4 | Pro Forma Combined Total | ||||||||||||||||
|
Revenue: |
||||||||||||||||||||||
|
Customer revenue |
$ | - | $ | 38,711 | $ | - | $ | - | $ | 38,711 | ||||||||||||
|
Collaboration revenue |
- | 4,099 | - | - | 4,099 | |||||||||||||||||
|
Total revenue |
- | 42,810 | - | - | 42,810 | |||||||||||||||||
|
Operating expenses: |
||||||||||||||||||||||
|
Research and development |
8,793 | 41,801 | - | - | 50,594 | |||||||||||||||||
|
General and administrative |
3,076 | 16,853 | - | 800 |
(f) |
20,729 | ||||||||||||||||
|
Asset impairment |
- | 13,912 | - | - | 13,912 | |||||||||||||||||
|
Total operating expenses |
11,869 | 72,566 | - | 800 | 85,235 | |||||||||||||||||
|
Operating loss |
(11,869 | ) | (29,756 | ) | - | - | (42,425 | ) | ||||||||||||||
|
Other income (expense), net: |
||||||||||||||||||||||
|
Interest income (expense) |
6,265 | 1,851 | - | - | 8,116 | |||||||||||||||||
|
Grant income |
- | 3,612 | - | - | 3,612 | |||||||||||||||||
|
Gain on extinguishment – royalty agreement |
23,098 | - | - | 23,098 | ||||||||||||||||||
|
Fair value adjustments on derivative liabilities |
485 | - | - | - | 485 | |||||||||||||||||
|
Other income (loss) |
712 | (250 | ) | - | - | 462 | ||||||||||||||||
|
Total other income (expense), net |
30,560 | 5,213 | - | - | 35,773 | |||||||||||||||||
|
Net Income (loss) |
18,691 | (24,543 | ) | - | 800 | (6,652 | ) | |||||||||||||||
|
Net profit (loss) per share |
||||||||||||||||||||||
|
Basic |
$ | 0.68 | $ | (21.80 | ) | $ | (0.48 | ) | ||||||||||||||
|
Diluted |
$ | 0.67 | $ | (21.80 | ) | $ | (0.48 | ) | ||||||||||||||
|
Weighted average number of common shares outstanding |
||||||||||||||||||||||
|
Basic |
5,720,009 | 1,125,795 |
(j) |
13,815,974 | ||||||||||||||||||
|
Diluted |
5,796,956 | 1,125,795 |
(j) |
13,815,974 | ||||||||||||||||||
See accompanying notes to the unaudited pro forma condensed combined financial statements
NOTES TO THE UNAUDITED PRO FORMA CONDENSED CONSOLIDATED COMBINED FINANCIAL INFORMATION
1. Description of the Transactions
The Merger
On July 23, 2024, Pieris, Merger Sub, and Palvella entered into the Merger Agreement, pursuant to which, and subject to the satisfaction or waiver of the conditions set forth in the Merger Agreement, Merger Sub will merge with and into Palvella, with Palvella continuing as a wholly- owned subsidiary of Pieris. After the completion of the Merger, Pieris will change its corporate name to “Palvella Therapeutics, Inc.” The Merger is intended to qualify for federal income tax purposes as a tax-free reorganization under the provisions of Section 368(a) of the Internal Revenue Code of 1986, as amended. If the Merger is completed, the business of Palvella will continue as the business of the combined company.
Subject to the terms and conditions of the Merger Agreement, at the effective time of the Merger, each outstanding share of Palvella capital stock (including shares of Palvella common stock and Palvella preferred stock) (excluding dissenting shares) will be converted solely into the right to receive a number of shares of Pieris common stock calculated in accordance with the Merger Agreement, equal to the exchange ratio calculated in accordance with the Merger Agreement. Based on Pieris’ and Palvella’s capitalization as of July 23, 2024, the date the Merger Agreement was executed, the exchange ratio is estimated to be approximately 0.315478222 shares of Pieris common stock for each share of Palvella common stock. Immediately after the Merger, but without giving effect to the PIPE Financing, Pieris securityholders as of immediately prior to the Merger are expected to own approximately 18% of the outstanding shares of capital stock of the combined company and Palvella securityholders as of immediately prior to the Merger are expected to own approximately 82% of the outstanding shares of capital stock of the combined company, in each case, on a fully-diluted basis, calculated using the treasury stock method, and subject to certain assumptions, including, but not limited toa valuation for Pieris equal to $21.0 million, provided, that (a) if Pieris’ net cash as of the closing is greater than $11 million, then Pieris’ valuation will be adjusted upwards on a dollar-for-dollar basis by the difference of: (i) Pieris’ net cash, minus (ii) $11 million, and (b) if Pieris’ net cash is less than $11 million, then Pieris’ valuation will be adjusted downwards on a dollar-for-dollar basis by the difference of: (i) $11 million, minus (ii) Pieris’ net cash, as further described in the Merger Agreement.
Because, among other factors, the number of shares of Pieris common stock issuable to Palvella’s securityholders is determined based on Pieris’ net cash balance as of the close of business on the last business day prior to the anticipated closing date of the Merger, Pieris cannot be certain of the exact number of shares that will be issued to (or reserved for issuance to) Palvella’s stockholders. The exchange ratio referenced above is an estimate only and the final exchange ratio will be determined pursuant to a formula described in detail in the Merger Agreement and included elsewhere in this proxy statement/prospectus.
Each stock option granted under the Palvella Stock Plan that is outstanding immediately prior to the effective time of the Merger will be assumed by Pieris and will become an option to acquire, on the same terms and conditions as were applicable to such Palvella stock option immediately prior to the effective time of the Merger, a number of shares of Pieris common stock equal to the number of shares of Palvella’s common stock subject to the unexercised portion of the Palvella stock option immediately prior to the effective time of the Merger, multiplied by the exchange ratio (rounded down to the nearest whole share number) with an exercise price per share for the options equal to the exercise price per share of such Palvella stock option immediately prior to the effective time of the Merger divided by the exchange ratio (rounded up to the nearest whole cent). Such assumed options will continue to be governed by the terms and conditions of the Palvella Stock Plan.
The pre-Merger employment agreements for the two Pieris executives also included severance, bonus and retention payments, the aggregate of which will be treated as pre-combination compensation expense of Pieris and is included in the liabilities assumed by Palvella upon closing of the Merger. In addition, certain non-executive Pieris employees entered into separation agreements prior to Merger negotiations with Palvella, pursuant to which they are entitled to severance, bonus, and retention payments. These payments will be treated as pre-combination compensation expense of Pieris and will also be included in the liabilities assumed by Palvella upon closing of the Merger.
Contingent Value Rights Agreement
At or prior to the effective time of the Merger, Pieris will enter into a CVR Agreement with a rights agent (the “Rights Agent”) and a CVR holder representative, pursuant to which Pieris stockholders as of the close of business on the last business day prior to the day on which the Effective Time occurs will receive a dividend of one contingent value right (“CVR”) for each outstanding share of Pieris common stock held by such stockholder, or each share of common stock underlying Pieris preferred stock outstanding and entitled to receive such dividend in accordance with the terms of such Pieris preferred stock held by such stockholder, as applicable, on such date. Furthermore, to the extent a holder of a Pieris warrant outstanding as of the date of the CVR Agreement exercises such Pieris warrant after the date of the CVR Agreement, a number of CVRs equal to the number of shares of Pieris common stock issued to such holder in connection with such exercise shall be issued to such holder, subject to and in accordance with the terms and conditions of such Pieris Warrant, as applicable.
Each CVR will represent the contractual right to receive cash payments (net of certain expenses and taxes) upon the receipt by Pieris or any of its affiliates of certain payments under certain strategic partner agreements, including existing collaboration agreements pursuant to which Pieris may be entitled to milestones and royalties in the future and other out-licensing agreements for certain of Pieris’ legacy assets, and upon the receipt of certain research and development tax credits in favor of Pieris or any of its affiliates, in each case as set forth in, and subject to and in accordance with the terms and conditions of, the CVR Agreement. Pursuant to the terms of the CVR Agreement, holders of CVRs would be entitled to cash payments (net of certain expenses and taxes) upon the receipt by Pieris or any of its affiliates of certain payments under any license, collaboration or similar agreement involving any legacy assets of Pieris entered into prior to the effective date of the CVR Agreement. Pieris retains the ability under the Merger Agreement to seek to enter into such agreements related to its legacy assets and intellectual property, including PRS-400 and PRS-220, though currently does not have any such agreements in place and does not have any plans to divest any material assets. Pieris continues to explore potential transactions for PRS-400 and PRS-220. Management has determined that there is no value associated with the CVRs as the likelihood of any payments received in connection with Pieris’ legacy assets is remote. Unless earlier terminated by the representative of the CVR holders and the combined company, the CVR Agreement will terminate on the date on which no strategic partner agreement is in effect and no payments are payable to Pieris or any of its affiliates under any Strategic Partner Agreement.
The contingent payments in respect of the CVRs, if they become payable, will become payable to a rights agent for subsequent distribution to the holders of the CVRs. In the event that no such proceeds are received, holders of the CVRs will not receive any payment pursuant to the CVR Agreement. There can be no assurance that holders of CVRs will receive any amounts with respect thereto. The right to the contingent payments contemplated by the CVR Agreement is a contractual right only and will not be transferable, except in the limited circumstances specified in the CVR Agreement. The CVRs will not be evidenced by a certificate or any other instrument and will not be registered with the SEC. The CVRs will not have any voting or dividend rights and will not represent any equity or ownership interest in Pieris or any of its affiliates. No interest will accrue on any amounts payable in respect of the CVRs.
Management concluded that the CVRs meet the definition of a derivative and will be initially measured at the aggregate estimated fair value of the CVRs and will be subsequently remeasured at estimated fair value on a recurring basis at each reporting period date. The estimated fair value of the CVR derivative was determined using the discounted cash flow method to estimate future cash flows associated with Pieris’ legacy assets, including the expected milestone and royalty payments associated with Pieries’ legacy assets. Changes in fair value of the CVR derivative are presented in the consolidated statements of operations and comprehensive income (loss). The derivative value is based on significant inputs not observable in the market such as estimated cash flows, estimated probabilities of success, and risk-adjustment discount rates, which represent a Level 3 measurement within the fair value hierarchy. Management has concluded that there is no value associated with the CVRs as the likelihood of any payments received in connection with Pieris’ legacy assets is remote.
Palvella Private Financing and PIPE Financing
On June 6, 2024, Palvella initiated a sequence of convertible notes with certain investors via the Convertible Note Purchase Agreement, or the Note Purchase Agreement. Under the Note Purchase Agreement, the investors have committed to extend credit to Palvella, providing up to a total of $20 million, or the Authorized Principal Amount, via convertible promissory notes. Through the issuance date of this filing, the Company has received $12.4 million of gross proceeds in exchange for convertible promissory notes issued. The convertible note bears an annual interest of 2.0% plus SOFR and shall be due and payable upon the earlier to occur of June 2027 or certain events defined in the Note Purchase Agreement. Under certain circumstances, the convertible note is convertible at the option of requisite holders into the Company’s equity securities at defined conversion prices. The terms of the convertible note specify that upon the consummation of the Merger, all outstanding principal and any unpaid accrued interest on the notes shall be automatically converted into common stock.
On July 23, 2024, concurrently with the execution and delivery of the Merger Agreement, the PIPE Investors entered into the Purchase Agreement with Pieris, pursuant to which such PIPE Investors have agreed to subscribe for and purchase (either for cash or in exchange for the termination and cancellation of outstanding convertible notes issued by Palvella), and Pieris has agreed to issue and sell to the PIPE Investors, an aggregate of up to 3,154,241 of shares of Pieris common stock at a price per share equal to the Purchase Price, subject to adjustment as set forth in the Purchase Agreement, and/or in lieu of Pieris common stock to certain purchasers who so choose, Pre-Funded Warrants to purchase up to 2,592,585 shares of Pieris common stock at a purchase price per Pre-Funded Warrant equal to the Purchase Price minus $0.001. The gross proceeds from the PIPE Financing are expected to be approximately $78.9 million, before paying estimated expenses.
The Merger is expected to be accounted for as a reverse recapitalization in accordance with GAAP. Under this method of accounting, Pieris, which is the legal acquirer, is treated as the “acquired” company for financial reporting purposes and Palvella is treated as the accounting acquirer. This determination was primarily due to Pieris being determined to be a shell company at the time of the Merger, immediately following the dividend of the CVR, because it will not meet the GAAP definition of a business, will not have more than nominal assets, and will not have more than nominal operations at the time of the Merger. Further, it is expected that, immediately following the Merger, Palvella’s stockholders will have a majority of the voting power of the combined company, Palvella will control four of five seats on the board of directors of the combined company, and Palvella’s senior management will comprise all of the senior management of the combined company. Accordingly, for accounting purposes, the Merger will be treated as the equivalent of a capital transaction in which Palvella is issuing stock for the net assets of Pieris. The net assets of Pieris will be stated at historical cost, with no goodwill or other intangible assets recorded.
2. Basis of Pro Forma Presentation
The unaudited pro forma condensed consolidated combined financial information was prepared pursuant to the rules and regulations of Article 11 of Regulation S-X. The unaudited pro forma condensed consolidated combined balance sheet as of June 30, 2024 was prepared using the historical balance sheets of Pieris and Palvella as of June 30, 2024, and gives effect to the Merger and the PIPE Financing as if they occurred on June 30, 2024. The unaudited pro forma condensed consolidated combined statement of operations for the six months ended June 30, 2024, and for the year ended December 31, 2023, were prepared using the historical statements of operations of Pieris and Palvella for the six months ended June 30, 2024 and for the year ended December 31, 2023, respectively, and gives effect to the Merger and the PIPE Financing as if they occurred on January 1, 2023.
The Merger is expected to be accounted for as a reverse recapitalization in accordance with GAAP, which is the equivalent of a capital transaction in which Palvella has issued stock for the net assets of Pieris. As the operations of Pieris are in the process of being wound down leading up to the date of the Merger, the net assets of Pieris are expected to be nominal as of the date of the Merger, resulting in Pieris being a public shell company. Upon the Merger, the net assets of Pieris will be stated at historical cost, with no goodwill or other intangible assets recorded. Operations prior to the Merger are of Palvella.
Accounting rules require evaluation of certain assumptions, estimates, or determination of financial statement classifications. During preparation of the unaudited pro forma condensed consolidated combined financial information, management has performed a preliminary analysis and is not aware of any material differences, and accordingly, this unaudited pro forma condensed combined financial information assumes no material differences in accounting policies. Following the Merger and the PIPE Financing, management will conduct a final review of Pieris accounting policies in order to determine if differences in accounting policies require adjustment or reclassification of Pieris results of operations or reclassification of assets or liabilities to conform to Palvella’s accounting policies and classifications. As a result of this review, management may identify differences that, when conformed, could have a material impact on this unaudited pro forma condensed consolidated combined financial information.
Palvella and Pieris may incur significant costs associated with integrating their operations after the Merger is completed. The unaudited pro forma condensed combined financial information does not reflect the costs of any integration activities or benefits that may result from realization of future cost savings from operating efficiencies, which may result from the Merger.
To the extent that there are significant changes to the business following completion of the Merger, the assumptions and estimates set forth in the unaudited pro forma condensed consolidated financial information could change significantly. Accordingly, the pro forma adjustments are subject to further adjustments as additional information becomes available and as additional analyses are conducted following the completion of the Merger. There can be no assurances that these additional analyses will not result in material changes to the estimates of fair value.
|
3. |
Shares of Pieris Common Stock Issued to Palvella’s Stockholders Upon Closing of the Merger |
At the effective time of the Merger, Pieris expects to issue 6,650,532 shares of common stock to the stockholders of Palvella in the Merger, determined as follows:
|
Shares |
||||
|
Palvella shares of common stock outstanding |
5,720,009 | |||
|
Shares of Palvella convertible preferred stock outstanding |
15,360,787 | |||
|
Total Palvella common stock equivalent shares |
21,080,796 | |||
|
Exchange ratio |
0.315478222 | |||
|
Estimated shares of Pieris common stock to be issued to Palvella stockholders upon closing of the Merger |
6,650,532 | |||
|
4. |
Pro Forma Adjustments |
Adjustments included in the column under the heading “Transaction Accounting Adjustments” are primarily based on information contained within the Merger Agreement, the Note Purchase Agreement, and the PIPE. Adjustments included in the column under the heading “Private Financing Adjustments” are primarily based on information contained in the Note Purchase Agreement and the PIPE. Further analysis will be performed upon completion of the Merger to confirm these estimates.
Based on a review of Pieris’ summary of significant accounting policies, the nature and amount of any adjustments to the historical consolidated financial statements of Pieris to conform to the accounting policies of Palvella are not expected to be significant.
Both Palvella and Pieris have a history of generating net operating losses and maintain a full valuation allowance against their net deferred tax assets. As a result, both entities have not reflected an income tax benefit or expense within the historical financial statement periods presented. Management has not identified any changes to the income tax positions due to the Merger that would result in an incremental tax expense or benefit. Accordingly, no tax-related adjustments have been reflected for the pro forma adjustments.
The pro forma adjustments, based on preliminary estimates that may change significantly as additional information is obtained, are as follows:
|
(a) |
To reflect $12.4 million in gross proceeds received pursuant to the Note Purchase Agreement as of the date of this filing, with $10.0 million of those proceeds already accounted for on Palvella’s historical balance sheet as of June 30, 2024. |
|
(b) |
To reflect the $60.0 million received in connection with the PIPE, for which approximately 4,370,270 shares of Pieris common stock are to be issued, and $6.5 million expected to be received in connection with the remaining commitments under the Note Purchase Agreement, offset by $6.5 million in transaction costs incurred. Upon closing of the Merger, any unfunded commitments under the Note Purchase Agreement will be accelerated and received in cash at the time of the merger in the form of shares of Pieris common stock. |
|
(c) |
To reflect Pieris’ estimated compensation expense of $0.5 million related to severance, retention, and bonus payments that were negotiated pre-Merger but had not yet been paid or fully accrued for as of June 30, 2024. As such, the $0.5 million is recorded as an assumed liability within the unaudited combined pro forma balance sheet as of June 30, 2024 and offset to accumulated deficit. As it is considered a preacquisition expense, there is no related adjustment within the unaudited condensed consolidated combined pro forma statements of operations. |
|
(d) |
To reflect the conversion of the $18.9 million issued under the Note Purchase Agreement, which includes $12.4 million (see 4(a) above) and $6.5 million (see 4(b) above) to 1,376,556 shares of Pieries common stock upon closing of the Merger. |
|
(e) |
To reflect the exchange of 15,360,787 shares of Palvella convertible preferred stock into shares of Palvella common stock, which, together with the 5,720,009 shares of outstanding Palvella common stock, were then converted in aggregate into 6,650,532 shares of Pieris common stock based on the exchange ratio. |
|
(f) |
To reflect Palvella’s preliminary estimated transaction costs of $6.5 million in connection with the Merger, such as advisor fees, legal fees, printer fees, and accounting expenses, of which $0.5 million of the transaction costs already accrued and deferred on Palvella’s historical balance sheet as of June 30, 2024. These transaction costs that are directly attributable to the transaction are recorded as an offset to additional paid-in capital. Based on the estimates of management, of the $6.5 million of transaction costs incurred, $5.7 million was offset against additional paid-in capital and $0.8 million was recorded to general and administrative expense in the unaudited condensed consolidated combined pro forma statement of operations. |
|
(g) |
To reflect the reclassification of Pieris accumulated other comprehensive income (loss) into additional paid-in capital. |
|
(h) |
To reflect the reclassification of historical accumulated deficit of Pieris into additional paid-in capital. |
|
(i) |
To reflect the reversal of interest expense accrued on the convertible notes during the period ended June 30, 2024 that will be converted to shares of Pieris common stock upon closing of the Merger. |
|
(j) |
The pro forma combined basic and diluted earnings per share have been adjusted to reflect the pro forma net income (loss) for the six months ended June 30, 2024, and the year ended December 31, 2023. In addition, the number of shares used to calculate the pro forma combined basic and diluted net income (loss) per share has been adjusted to reflect the estimated total number of shares of common stock of the combined company that would be outstanding as of the Merger closing date, including the shares to be issued in the PIPE Financing, as if they have been outstanding for the entirety of the periods presented. For the six months ended June 30, 2024, and the year ended December 31, 2023, the pro forma weighted average shares outstanding has been calculated as follows: |
|
June 30, 2024 |
December 31, 2023 |
|||||||
|
Weighted-average Palvella common shares outstanding – basic and diluted |
5,720,009 | 5,720,009 | ||||||
|
Palvella convertible preferred stock |
15,360,787 | 15,360,787 | ||||||
|
Total |
21,080,796 | 21,080,796 | ||||||
|
Application of exchange ratio |
0.315478 | 0.315478 | ||||||
|
Adjusted Weighted-average Palvella common shares outstanding – basic and diluted |
6,650,532 | 6,650,532 | ||||||
|
$18.9 million of Palvella convertible notes |
1,376,556 | 1,376,556 | ||||||
|
$60.0 million of PIPE Financing |
4,370,270 | 4,370,270 | ||||||
|
Weighted-average Pieris common shares outstanding |
1,268,000 | 1,125,795 | ||||||
|
Pieris convertible preferred stock |
208,331 | 208,331 | ||||||
|
Pieris reverse stock split adjustment |
- | 83,552 | ||||||
|
Pieris planned issuance of common shares |
938 | 938 | ||||||
|
Pro forma combined weighted average number of shares of common stock – basic and diluted |
13,874,627 | 13,815,974 | ||||||
DESCRIPTION OF PIERIS CAPITAL STOCK
The following description of Pieris capital stock and provisions of Pieris’ amended and restated articles of incorporation and amended and restated bylaws are summaries and are qualified in their entirety by reference to such amended and restated articles of incorporation and amended and restated bylaws and applicable provisions of Nevada law, including Chapters 78 and 92A of the NRS. Pieris has filed copies of these documents with the SEC as exhibits to its periodic filings.
Under Nevada law, the issuance of shares of Pieris common stock to Palvella may not exceed the number of authorized shares of Pieris common stock then authorized under Pieris’ amended and restated articles of incorporation. As such, the authorized share increase of Pieris common stock, as described in Proposal No. 2, if approved by the majority of the voting power of all outstanding shares of Pieris capital stock on the record date for the Pieris special meeting, is expected to occur prior to the Merger and prior to issuance of shares of Pieris common stock to Palvella pursuant to the Merger Agreement.
Pieris’ authorized capital stock consists of 3,750,000 shares of Pieris common stock, par value $0.001 per share, and 10,000,000 shares of Pieris preferred stock, par value $0.001 per share, of which authorized Pieris preferred stock 4,963 shares have been designated as Series A Convertible preferred stock, 5,000 shares have been designated as Series B Convertible Preferred Stock, 3,522 shares have been designated as Series C Convertible Preferred Stock, 3,000 shares have been designated as Series D Convertible Preferred Stock, 5,000 shares have been designated as Series E Convertible Preferred Stock, and one share has been designated as Series F Preferred Stock.
Holders of Pieris common stock are entitled to one vote for each share held on all matters submitted to a vote of Pieris stockholders and do not have cumulative voting rights. Holders of Pieris common stock are entitled to receive any dividends as may be declared by the Pieris board of directors, subject to any preferential dividend or other rights of any then outstanding preferred stock.
Voting Rights
For all matters submitted to a vote of stockholders, each holder of Pieris common stock is entitled to one vote for each share registered in the holder’s name on Pieris’ books. At all meetings of the stockholders, except where otherwise provided by applicable law, rules of any stock exchange upon which Pieris’ securities are listed, Pieris’ amended and restated articles of incorporation or Pieris’ amended and restated bylaws, the presence, in person, virtually or by duly authorized proxy, of the holders of a majority of the outstanding shares of Pieris capital stock entitled to vote constitutes a quorum for the transaction of business. Except as otherwise provided by applicable law or by Pieris’ amended and restated articles of incorporation or Pieris’ amended and restated bylaws, all matters, other than the election of directors, proposed at any meeting of the stockholders shall be determined by a majority of the votes cast affirmatively or negatively. Except as otherwise provided by law, Pieris’ amended and restated articles of incorporation, Pieris’ amended and restated bylaws or the terms of any class or series of Pieris preferred stock, directors are elected by a plurality of the votes of the shares of Pieris common stock present virtually or by proxy at the meeting and entitled to vote generally on the election of directors.
Dividends
Subject to limitations under Nevada law, any provision of Pieris’ amended and restated articles of incorporation and any preferences that may be applicable to any then outstanding preferred stock, holders of Pieris common stock are entitled to receive ratably those dividends, if any, as may be declared by the Pieris board of directors, in their sole discretion, out of legally available funds.
Liquidation
Upon Pieris’ liquidation, dissolution or winding up, the holders of Pieris common stock will be entitled to share ratably in the net assets legally available for distribution to stockholders after the payment of all Pieris’ debts and other liabilities of the company, subject to any prior rights of any preferred stock then outstanding.
Other Rights and Restrictions
Holders of Pieris common stock do not have preemptive or subscription rights, and they have no right to convert their Pieris common stock into any other securities. There are no redemption or sinking fund provisions applicable to Pieris common stock. The rights, preferences and privileges of common stockholders are subject to the rights of the stockholders of any series of Pieris preferred stock, including any which Pieris may designate in the future. Pieris’ amended and restated articles of incorporation and amended and restated bylaws do not restrict the ability of a holder of Pieris common stock to transfer the holder’s shares of Pieris common stock.
Listing
Pieris common stock is quoted on The Nasdaq Capital Market under the symbol “PIRS.”
Transfer Agent and Registrar
The transfer agent and registrar for Pieris common stock is Computershare Investor Services, LLC, P.O. Box 43006, Providence, RI 02940-3078, telephone number: 1-877-373-6374.
Under the terms of Pieris’ amended and restated articles of incorporation, the Pieris board of directors is authorized to issue up to 10,000,000 shares of preferred stock in one or more series without stockholder approval by adopting a resolution or resolutions providing for the issuance of the shares thereof and by filing a certificate of designations relating thereto in accordance with the NRS. The Pieris board of directors has the discretion to determine the designations, rights, preferences, privileges and restrictions, including voting powers (full, limited or no voting powers), dividend rights, conversion rights, redemption privileges and liquidation preferences, of each series of preferred stock.
The authorization of the Pieris board of directors to issue preferred stock and determine the rights and preferences of that preferred stock eliminates delays associated with a stockholder vote on specific issuances. The issuance of preferred stock, while providing flexibility in connection with possible acquisitions, future financings and other corporate purposes, could have the effect of making it more difficult for a third party to acquire, or could discourage a third party from seeking to acquire, a majority of Pieris outstanding voting stock.
Voting Rights
The Series F Preferred Stock have no voting rights other than the right to vote on Proposal No. 2. Each share of Series F Preferred Stock outstanding on the record date entitles the holder thereof to 25,000,000 votes on Proposal No. 2, and all shares of Series F Preferred Stock held by such holder must and will be voted, without further action by such holder, in the same proportion as the aggregate shares of Pieris common stock (excluding any shares of Pieris common stock that are not voted) are voted on Proposal No. 2. As an example, if 50.5% of the shares of Pieris common stock that are voted either “FOR” or “AGAINST” the Authorized Shares Increase Proposal are voted FOR the Authorized Shares Increase Proposal, 50.5% of the votes cast by the holder of the Series F Preferred Stock will be cast as votes FOR the Authorized Shares Increase Proposal. Abstentions and broker non-votes, if any, will have the same effect as “AGAINST” votes on the Authorized Shares Increase Proposal, but will not be considered as votes cast “AGAINST” for the purposes of determining the proportion of Series F Preferred stock votes that will be cast “FOR” and “AGAINST”.
Conversion and Redemption
Shares of the Series F Preferred Stock are not convertible into any other security, and are redeemable by Pieris upon the earlier to occur of: (i) the order of the Pieris board of directors in its sole discretion, automatically and effective at such date and time as is determined and specified by the Pieris board of directors in its sole discretion and (ii) automatically and effective immediately after the effectiveness of the increase in the number of authorized shares of Pieris common stock proposed in Proposal No. 2. Shares of the Series F Preferred Stock may not be transferred prior to their redemption without the prior written consent of the Pieris board of directors.
Other Rights and Restrictions
Each holder of Series F Preferred Stock has entered into a written agreement with Pieris to attend the Pieris special meeting, to vote all shares of Series F Preferred Stock with regard to Proposal No. 2 in the same proportion as the aggregate shares of Pieris common stock (excluding any shares of Pieris common stock that are not voted) are voted on Proposal No. 2 and, upon request by Pieris, to grant a designee of Pieris an irrevocable proxy to vote the shares of Series F Preferred Stock in accordance with the foregoing.
Anti-Takeover Effects of Nevada Law and Pieris’ Amended and Restated Articles of Incorporation and Amended and Restated Bylaws.
Nevada law and Pieris’ amended and restated articles of incorporation and amended and restated bylaws contain provisions that could have the effect of delaying, deferring or discouraging another party from acquiring control of Pieris. These provisions, which are summarized below, are expected to discourage coercive takeover practices and inadequate takeover bids. These provisions are also designed to encourage persons seeking to acquire control of Pieris to first negotiate with the Pieris board of directors.
Staggered Board; Removal of Directors. Pieris’ amended and restated articles of incorporation divides the Pieris board of directors into three classes with staggered three-year terms. In addition, a director may only be removed for cause and only by the affirmative vote of the holders of at least 80% of the votes that all of Pieris stockholders would be entitled to cast in an annual election of directors. Any vacancy or newly-created directorship on the Pieris board of directors may only be filled by vote of a majority of Pieris’ directors then in office, even though less than a quorum, or by a sole remaining director, and not by stockholders. The classification of the Pieris board of directors and the limitations on the removal of directors and filling of vacancies could make it more difficult for a third party to acquire, or discourage a third party from seeking to acquire, control of Pieris.
No Stockholder Action by Written Consent; Special Meetings. Pieris’ amended and restated articles of incorporation and amended and restated bylaws provide that any action required or permitted to be taken by Pieris stockholders must be effected at a duly called annual or special meeting of such holders and may not be effected by any consent in writing by such holders. Pieris’ amended and restated articles of incorporation and amended and restated bylaws also provide that, except as otherwise required by law, special meetings of Pieris stockholders can only be called by the Pieris board of directors.
Advance Notice Requirements for Stockholder Proposals. Pieris’ amended and restated bylaws establish an advance notice procedure for stockholder proposals to be brought before an annual meeting of Pieris stockholders, including proposed nominations of persons for election to the Pieris board of directors. Stockholders at a Pieris annual meeting may only consider proposals or nominations specified in the notice of meeting or brought before the meeting by or at the direction of the Pieris board of directors or by a Pieris stockholder of record on the record date for the meeting who is entitled to vote at the meeting and who has delivered timely written notice in proper form to Pieris’ secretary of the stockholder’s intention to bring such business before the meeting. These provisions could have the effect of delaying until the next stockholder meeting stockholder actions that are favored by the holders of a majority of Pieris’ outstanding voting securities.
Amendment of Pieris’ amended and restated articles of incorporation and amended and restated bylaws. The NRS provides generally that the affirmative vote of a majority of the outstanding shares entitled to vote thereon is required to amend a corporation’s articles of incorporation, unless a corporation’s articles of incorporation requires a greater percentage. Pieris’ amended and restated bylaws may be amended or repealed by a majority vote of the Pieris board of directors or by the affirmative vote of the holders of at least 80% of the votes that all of Pieris stockholders would be entitled to cast in any annual election of directors. In addition, the affirmative vote of the holders of at least 80% of the votes that all of Pieris stockholders would be entitled to cast in any annual election of directors is required to amend or repeal or to adopt any provisions inconsistent with any of the provisions of Pieris’ amended and restated articles of incorporation described above under “—Staggered Board; Removal of Directors” and “—No Stockholder Action by Written Consent; Special Meetings,” and under this section “ —Amendment of Pieris’ amended and restated articles of incorporation and amended and restated bylaws.”
Acquisitions, Business Combinations and Change in Control. The NRS contains provisions governing the acquisition of a controlling interest in certain Nevada corporations. Nevada’s “acquisition of controlling interest” statutes (NRS 78.378 through 78.3793, inclusive) contain provisions governing the acquisition of a controlling interest in certain Nevada corporations. These “control share” laws provide generally that any person that acquires a “controlling interest” in certain Nevada corporations may be denied voting rights, unless a majority of the disinterested stockholders of the corporation elect to restore such voting rights. These laws would apply to Pieris as of a particular date if Pieris were to have 200 or more stockholders of record (at least 100 of whom have addresses in Nevada appearing on Pieris’ stock ledger at all times during the 90 days immediately preceding that date) and do business in the State of Nevada directly or through an affiliated corporation, unless Pieris’ amended and restated articles of incorporation or amended and restated bylaws in effect on the tenth day after the acquisition of a controlling interest provide otherwise. These laws provide that a person acquires a “controlling interest” whenever a person acquires shares of a subject corporation that, but for the application of these provisions of the NRS, would enable that person to exercise (1) one-fifth or more, but less than one-third, (2) one-third or more, but less than a majority, or (3) a majority or more of all of the voting power of the corporation in the election of directors. Once an acquirer crosses one of these thresholds, shares which it acquired in the transaction taking it over the threshold and within the 90 days immediately preceding the date when the acquiring person acquired or offered to acquire a controlling interest become “control shares” to which the voting restrictions described above apply. Pieris’ amended and restated articles of incorporation include a provision stating that these laws, or any successor statutes, relating to acquisitions of controlling interests in Pieris, shall not apply to Pieris or to any acquisition of any shares of Pieris capital stock. These laws may have a chilling effect on certain transactions if Pieris’ amended and restated articles of incorporation are amended to eliminate the foregoing provision and these laws otherwise apply according to their terms.
Nevada’s “combinations with interested stockholders” statutes (NRS 78.411 through 78.444, inclusive) provide that specified types of business “combinations” between certain Nevada corporations and any person deemed to be an “interested stockholder” of the corporation are prohibited for two years after such person first becomes an “interested stockholder” unless the corporation’s board of directors approves the combination (or the transaction by which such person becomes an “interested stockholder”) in advance, or unless the combination is approved by the board of directors and 60% of the corporation’s voting power not beneficially owned by the interested stockholder, its affiliates and associates. Furthermore, in the absence of prior approval certain restrictions may apply even after such two-year period. For purposes of these statutes, an “interested stockholder” is any person who is (1) the beneficial owner, directly or indirectly, of 10% or more of the voting power of the outstanding voting shares of the corporation, or (2) an affiliate or associate of the corporation and at any time within the two previous years was the beneficial owner, directly or indirectly, of 10% or more of the voting power of the then-outstanding shares of the corporation. The definition of the term “combination” is sufficiently broad to cover most significant transactions between a corporation and an “interested stockholder”. These laws generally apply to Nevada corporations with 200 or more stockholders of record. However, a Nevada corporation may elect in its articles of incorporation not to be governed by these particular laws, but if such election is not made in the corporation’s original articles of incorporation, the amendment (1) must be approved by the affirmative vote of the holders of stock representing a majority of the outstanding voting power of the corporation not beneficially owned by interested stockholders or their affiliates and associates, and (2) is not effective until 18 months after the vote approving the amendment and does not apply to any combination with a person who first became an interested stockholder on or before the effective date of the amendment. Pieris has not made such an election in its original articles of incorporation, and it has not amended the Pieris amended and restated articles of incorporation to so elect.
Further, NRS 78.139 provides that directors may resist a change or potential change in control of the corporation if the board of directors determines that the change or potential change is opposed to or not in the best interest of the corporation upon consideration of any relevant facts, circumstances, contingencies or constituencies pursuant to NRS 78.138(4).
COMPARISON OF RIGHTS OF HOLDERS OF PIERIS CAPITAL STOCK AND PALVELLA CAPITAL STOCK
If the Merger is completed, Palvella stockholders will receive shares of Pieris common stock, pursuant to the terms of the Merger Agreement. Immediately prior to the closing of the Merger, Pieris’s amended and restated articles of incorporation of Pieris will be amended to increase the number of authorized shares of Pieris common stock, as set forth in the form of certificate of amendment attached as Annex I to this proxy statement/prospectus.
The Pieris amended and restated articles of incorporation differs in several respects from the third amended and restated certificate of incorporation of Palvella. Set forth below is a table summarizing certain material differences in the rights of stockholders of Palvella under Delaware law as compared with the rights of Pieris stockholders under Nevada law, and under the respective charters and bylaws. This chart does not attempt to address each difference, but instead focuses on those differences which we believe are most relevant and material to Palvella stockholders. This chart is qualified in its entirety by reference to the NRS, the Pieris amended and restated articles of incorporation, the Pieris amended and restated bylaws, the DGCL, the Palvella third amended and restated certificate of incorporation and the Palvella bylaws.
|
Provision |
Delaware |
Nevada |
|
Charter Regarding Limitation of Liability |
The Palvella Third Amended and Restated Articles of Incorporation (the “Delaware Charter”) provides that, to the fullest extent permitted by DGCL, any person (an “Indemnified Person”) who was or is made or is threatened to be made a party or is otherwise involved in any action, suit or proceeding, whether civil, criminal, administrative or investigative (a “Proceeding”), by reason of the fact that such person, or a person for whom such person is the legal representative, is or was a director or officer of Palvella or, while a director or officer of Palvella, is or was serving at the request of Palvella as a director, officer, employee or agent of another corporation or of a partnership, joint venture, limited liability company, trust, enterprise or nonprofit entity, including service with respect to employee benefit plans, against all liability and loss suffered and expenses (including attorneys’ fees) reasonably incurred by such Indemnified Person in such Proceeding; provided in the case of a director, such director’s actions or omissions did not constitute fraud, bad faith, a violation of Palvella’s bylaws (the “Delaware Bylaws”) or willful misconduct. |
The Pieris Amended and Restated Articles of Incorporation (the “Nevada Charter”) provides that, to the fullest extent permitted by the NRS, the liability of directors and officers of Pieris shall be eliminated or limited.
Note that, under the NRS, this provision does not exclude exculpation for breaches of duty of loyalty and covers both directors and officers. |
|
Charter Regarding Indemnitee’s Right to Advanced Payment of Expenses |
The Delaware Charter provides that Palvella shall pay the expenses (including attorneys' fees) incurred by any Indemnified Person in defending any Proceeding in advance of its final disposition, provided, however, that, to the extent required by law, such payment of expenses in advance of the final disposition of the Proceeding shall be made only upon receipt of an undertaking by the Indemnified Person to repay all amounts advanced if it should be ultimately determined that the Indemnified Person is not entitled to be indemnified under the Delaware Charter or otherwise. does not specify whether an indemnitee’s right to advanced payment of expenses related to a proceeding is subject to the satisfaction of any standard of conduct nor is it conditioned upon any prior determination that the indemnitee is entitled to indemnification with respect to the related proceeding (or the absence of any prior determination to the contrary). If a claim for indemnification or advancement of expenses is not paid in full within 30 days after a written claim therefor by the Indemnified Person has been received by Palvella, the Indemnified Person may file suit to recover the unpaid amount of such claim and, if successful in whole or in part. shall be entitled to be paid the expense of prosecuting such claim. In any such action Palvella shall have the burden of proving that the Indemnified Person is not entitled to the requested indemnification or advancement of expenses under applicable law. |
The Nevada Charter does not expressly condition an indemnitee’s right to advanced payment of expenses related to a proceeding on the satisfaction of any standard of conduct nor is it conditioned upon any prior determination that the indemnitee is entitled to indemnification with respect to the related proceeding (or the absence of any prior determination to the contrary). |
|
Charter Regarding Forum Adjudication for Disputes |
The Delaware Charter provides that unless Palvella consents in writing to the selection of an alternative forum, the Court of Chancery in the State of Delaware shall be the sole and exclusive forum for any stockholder (including a beneficial owner) to bring (i) any derivative action or proceeding brought on behalf of Palvella, (ii) any action asserting a claim of breach of fiduciary duty owed by any director, officer or other employee of Palvella to Palvella or Palvella’s stockholders, (iii) any action asserting a claim against Palvella, its directors, officers or employees arising pursuant to any provision of the DGCL or Palvella’s third amended and restated certificate of incorporation or bylaws or (iv) any action asserting a claim against Palvella, its directors, officers or employees governed by the internal affairs doctrine, except for, as to each of (i) through (iv) above, any claim as to which the Court of Chancery determines that there is an indispensable party not subject to the jurisdiction of the Court of Chancery (and the indispensable party does not consent to the personal jurisdiction of the Court of Chancery within 10 days following such determination), which is vested in the exclusive jurisdiction of a court or forum other than the Court of Chancery, or for which the Court of Chancery does not have subject matter jurisdiction. |
The Nevada Charter provides that, to the fullest extent permitted by law, and unless the combined company consents to the selection of an alternative forum, the Eighth Judicial District Court of Clark County, Nevada shall be the sole and exclusive forum for any (i) derivative action or proceeding brought in the name or right of the corporation or on its behalf, (ii) action asserting a claim of breach of a fiduciary duty owed by any of its directors, officers, employees or agents to the corporation or any of its stockholders, (iii) any action arising or asserting a claim arising pursuant to any provision of Chapters 78 or 92A of the NRS or any provision of its articles of incorporation or bylaws, (iv) any action to interpret, apply, enforce or determine the validity of its articles of incorporation or bylaws or (v) any action asserting a claim governed by the internal affairs doctrine. Choice-of-forum provisions of the type and scope included in the articles of incorporation of the combined company are expressly permitted by Section 78.046 of the NRS, but application of these choice-of-forum provisions may be limited in some instances by law. Section 27 of the Exchange Act establishes exclusive federal jurisdiction over all suits brought to enforce any duty or liability created by the Exchange Act or the rules and regulations thereunder and therefore the choice-of-forum provision would not apply to actions arising under, or brought to enforce a duty or liability created by, the Exchange Act or any other claim for which the U.S. federal courts have exclusive jurisdiction. However, Section 22 of the Securities Act provides that federal and state courts have concurrent jurisdiction over all suits brought to enforce any duty or liability created by the Securities Act or the rules and regulations thereunder, and the choice-of-forum provision would apply to actions arising under, or brought to enforce a duty or liability created by, the Securities Act. To the extent the text of the choice-of-forum provision in the combined company’s articles of incorporation purports to restrict the courts in which claims arising under the Securities Act may be brought, there remains some uncertainty as to whether a court would enforce such a provision. We note that the choice-of-forum provision will not relieve the combined company of its duties to comply with the federal securities laws and the rules and regulations thereunder, and the combined company’s stockholders will not be deemed to have waived compliance with these laws, rules and regulations. |
|
Classified Board |
The Delaware Charter does not provide for a classified board. The Delaware Charter provides that directors are elected for annual terms. Each director’s term continues until the election and qualification of his or her successor, or his or her earlier death, resignation or removal.
The Delaware Charter provides that the holders of Palvella’s Series D preferred stock, exclusively and as a separate class, shall be entitled to elect two directors of Palvella (the “Series D Directors”), (b) the holders of Palvella’s Series C preferred stock, exclusively and as a separate class, shall be entitled to elect two directors of Palvella (the “Series C Directors”), (c) the holders of the shares of Palvella’s Junior preferred stock, voting together as a single class on an as-converted to Palvella’s voting common stock basis, shall be entitled to elect one director of Palvella (the “Junior Preferred Director”) and (d) the holders Palvella’s shares of voting common stock, exclusively and as a separate class, shall be entitled to elect one director of Palvella (the “Common Directors”).
No such directorship may be filled by Palvella stockholders other than by the stockholders that are entitled to elect such director, voting exclusively and as a separate class. |
The Nevada Charter and the Pieris Amended and Restated Bylaws (the “Nevada Bylaws”) provide that directors shall be divided into three classes, each serving until the third succeeding annual meeting of stockholders following such director’s election. |
|
Amendment to Delaware Charter or the Nevada Charter |
As long as any shares of Palvella preferred stock are outstanding, the written consent or affirmative vote of both (i)(A) Petrichor or (B) the holders of a majority of 70% of the outstanding shares of the Series D preferred stock of Palvella held by stockholders other than Petrichor, and (ii) at least one of (A) CDK Associates L.L.C. or (B) Samsara BioCapital L.P. (the “Requisite Holders”) is needed to amend, alter or repeal any provision of Palvella’s third amended and restated certificate of incorporation or to increase or decrease the authorized number of directors constituting the Palvella board of directors.
As long as at least 36,704 shares of Series D preferred stock are outstanding, the written consent or affirmative vote of the holders of at least 67% of the outstanding shares of Palvella Series D preferred stock, voting as a separate class, is needed to amend, alter, waive or repeal any provision of Palvella’s third amended and restated certificate of incorporation so as to affect the powers, preferences or rights of the Series D preferred stock in a material and adverse manner or to increase or decrease the authorized number of shares of Series D preferred stock.
As long as at least 2,127,498 shares of Series C preferred stock are outstanding, the written consent or affirmative vote of the holders of at least 67% of the outstanding shares of Palvella Series C preferred stock, voting as a separate class, is needed to amend, alter, waive or repeal any provision of Palvella’s third amended and restated certificate of incorporation so as to affect the powers, preferences or rights of the Series C preferred stock in a material and adverse manner or to increase or decrease the authorized number of shares of Series C preferred stock.
As long as at least 383,382 shares of Series B preferred stock are outstanding, the written consent or affirmative vote of the holders of at least 67% of the outstanding shares of Palvella Series B preferred stock, voting as a separate class, is needed to amend, alter, waive or repeal any provision of Palvella’s third amended and restated certificate of incorporation so as to affect the powers, preferences or rights of the Series B preferred stock in a material and adverse manner or to increase or decrease the authorized number of shares of Series B preferred stock.
As long as at least 870,509 shares of Series A-1 preferred stock and Series A-2 preferred stock are outstanding, the written consent or affirmative vote of the holders of at least 67% of the outstanding shares of Palvella Series A-1 preferred stock and Series A-2 preferred stock, voting as a separate class, is needed to amend, alter, waive or repeal any provision of Palvella’s third amended and restated certificate of incorporation so as to affect the powers, preferences or rights of the Series A-1 preferred stock and Series A-2 preferred stock in a material and adverse manner or to increase or decrease the authorized number of shares of Series A-1 preferred stock and Series A-2 preferred stock. |
The NRS provides that, except as otherwise provided by applicable law, an amendment to the Nevada Charter would require approval of the amendment by the Pieris board of directors and by stockholders holding shares representing at least a majority of the voting power, or such greater proportion of the voting power as may be required in the case of a vote by classes or series, or as may be required by the provisions of the Nevada Charter.
The NRS further provides that an amendment to the Nevada Charter that only changes the name of the corporation does not require any stockholder approval.
The Nevada Charter provides that in addition to the affirmative vote of the holders of any class or series of the shares of Pieris capital stock required by law or by the Nevada Charter, the affirmative vote of the holders of at least 80% of the voting power of all of the then outstanding shares of Pieris capital stock entitled to vote generally in the election of directors, voting together as a single class, shall be required to amend, alter or repeal, or adopt any provision inconsistent with any provision of the Nevada Charter other than Article I (name of the corporation), Article II (registered agent and registered office), Article III (purpose of the corporation) and Article IV (authorized capital stock). |
|
Removal of Directors |
The Delaware Charter provides that Series D Directors, the Series C Directors the Junior Preferred Director and the Common Director may be removed without cause only by the affirmative vote or written consent of the holders of the shares of the series of preferred stock or shares of common stock entitled to elect them. |
The Nevada Charter and the Nevada Bylaws provide that subject to the rights of the holders of any series of preferred stock then outstanding, any director, or the entire Pieris board of directors, may be removed from office at any time only for cause and only by the affirmative vote of the holders of at least 80% of the voting power of all of the then outstanding shares of Pieris capital stock entitled to vote at an election of directors, voting together as a single class, which exceeds the lowest permitted voting threshold for removal of a director under the NRS, which is two-thirds of the outstanding voting power of the stockholders. |
|
Vacancies on the Board |
A vacancy in any directorship filled by the holders of any class or series shall be filled only by vote or written consent in lieu of a meeting of the holders of such class or series or by any remaining director or directors elected by the holders of such class or series. |
Subject to the rights of the holders of any series of preferred stock then outstanding, any vacancies in the Pieris board of directors resulting from death, resignation, retirement, disqualification, removal from office or other cause shall, unless otherwise required by law, by the Nevada Charter or by resolution of the Pieris board of directors, be filled only by a majority vote of the directors then in office even though less than a quorum, or by a sole remaining director, and not by the stockholders. |
|
Bylaws Regarding Proxies |
The Delaware Bylaws provide that proxies are valid for three years from the date of creation unless the proxy provides for a longer period. |
Under the NRS, proxies are valid for six months from the date of creation unless the proxy provides for a longer period of up to seven years. |
|
Bylaws Regarding Advance Notice |
The DGCL does not have a statutory advance notice requirement, but a Delaware corporation is permitted to set forth such requirements in its bylaws. The Delaware Bylaws do not provide for advance notice requirement. |
The NRS does not have any statutory advance notice requirements but a Nevada corporation is permitted to set forth such requirements in its bylaws. The Nevada Bylaws provide that the same advance notice of not less than 120 days prior to the first anniversary of Pieris’ preceding year’s annual meeting must be received. |
|
Bylaws Regarding Proxy Access |
The Delaware Bylaws have not adopted a proxy access bylaw. |
The Nevada Bylaws do not have a proxy access bylaw. |
|
Bylaws Regarding Annual Meetings of Stockholders |
The Delaware Bylaws provide that annual meeting of stockholders for the election of directors and for the transaction of such other business as may properly come before the meeting shall be held in person or by means of remote communication, if any, at such place, if any, and at such date and hour as shall be fixed by the Palvella board of directors and designated in the notice or waiver of notice thereof. |
The Nevada Bylaws provide that annual meeting of stockholders for the election of directors and for the transaction of such other business as may properly come before the meeting shall be held at such time and date as shall be designated from time to time by the board. |
|
Bylaws Regarding Quorum |
Except where otherwise provided by Delaware Charter or the Delaware Bylaws, the Delaware Bylaws provide that holders of a majority of the capital stock issued and entitled to vote at a stockholder meeting, present in person or represented by proxy shall constitute a quorum at all meetings of the stockholders for the transaction of business.
Where a separate vote by a class or series or classes or series of Palvella stock is required, a majority of the outstanding shares of such class or series or classes or series, present in person or represented by proxy, shall constitute a quorum entitled to take action with respect to that vote on that matter, except as otherwise provided by law, the Delaware Charter or the Delaware Bylaws. If a quorum fails to attend or be represented at any meeting, the chairperson of the meeting or stockholders entitled to vote at the meeting, present in person or by proxy, at the meeting may adjourn the meeting in accordance with the Delaware Bylaws until a quorum is present or represented. |
The Nevada Bylaws and NRS provide that a majority of the voting power, which includes the voting power that is present in person or by proxy, regardless of whether the proxy has authority to vote on any matter, constitutes a quorum for the transaction of business, except as otherwise provided by Nevada Charter or the rules of any stock exchange upon which the combined company’s securities are listed. |
|
Bylaws Regarding |
The Delaware Bylaws provide that unless otherwise provided by the Palvella board of directors, meetings of stockholders shall be presided over by one of the following in the following order of precedence: (i) the Chairman; (ii) the Chief Executive Officer; (iii) the President; (iv) any other director or officer of Palvella designated by the Palvella board of directors to act as chairman of such meeting and to preside thereat if both the Chairman and the President shall be absent from such meeting; or (v) a stockholder of record who shall be chosen chairman of such meeting by a majority in voting interest of the stockholders present in person or by proxy and entitled to vote. |
The Nevada Bylaws provide that the meetings of stockholders shall be presided over by the Chairman of the board, or the Chief Executive Officer. In the absence or disability of the Chairman or the Chief Executive Officer, the President, or in the absence of any of the foregoing persons, by a chair designated by the board or by a chair chosen by the stockholders shall preside. The Secretary will act as secretary of the meeting, but in the Secretary’s absence, and the absence of any Assistant Secretary, the chair of the meeting may appoint another officer to act as secretary of the meeting. The chairman of any meeting of the stockholders shall determine the order of business and the procedures at the meeting, including such regulation of the manner of voting and the conduct of discussion as he or she deems to be appropriate. The chairman of any meeting of the stockholders shall have the power to adjourn the meeting to another place, if any, date and time. The date and time of the opening and closing of the polls for each matter upon which the stockholders will vote at the meeting shall be announced at the meeting. |
|
Bylaws Regarding Officers |
The Delaware Bylaws provide that officers of Palvella shall be chosen by the board of directors and shall consist of a Chief Executive Officer, President, a Secretary and a Treasurer, the Palvella board of directors, in its discretion may also appoint such other officers, agents and employees as the Palvella board of directors may deem necessary, including a Chief Financial Officer, a Controller, one or more Assistant Secretaries, one or more Assistant Treasurers, and one or more Vice Presidents, each of whom shall hold office for such period, have such authority, and perform such duties as the Palvella board of directors or the President may from time to time determine. The Palvella may delegate to any principal officer the power to appoint and define the authority and duties of, or remove, any such officers, agents, or employees. |
The Nevada Bylaws provide that officers of Pieris shall be chosen by the board and shall include a Chief Executive Officer, President, Secretary, and Treasurer. The board, in its discretion may also appoint such other officers as the board or the Chief Executive Officer may determine, including but not limited to a Chief Financial Officer one or more Vice Presidents, Assistant Treasurers and Assistant Secretaries each of whom shall have such authority and perform such duties as may be prescribed by the board. |
|
Amendment to the Bylaws |
As long as any shares of Palvella preferred stock are outstanding, the written consent or affirmative vote of the Requisite Holders is needed to amend, alter or repeal any provision of Delaware Bylaws. |
The Nevada Charter and Nevada Bylaws provide that the Pieris board of directors is expressly empowered to adopt, amend or repeal the Nevada Bylaws. Any adoption, amendment or repeal of the Nevada Bylaws by the Pieris board of directors shall require the approval of a majority of the total number of authorized directors whether or not there exist any vacancies in previously authorized directorships. The stockholders also have power to adopt, amend or repeal the Nevada Bylaws; provided, that in addition to the affirmative vote of the holders of any class or series of the shares of Pieris capital stock required by law or by the Nevada Charter, the affirmative vote of the holders of at least 80% of the voting power of all of the then outstanding shares of Pieris capital stock entitled to vote generally in the election of directors, voting together as a single class, is required for the stockholders to adopt, amend or repeal any provision of the Nevada Bylaws. |
|
Conversion Rights |
Palvella’s third amended and restated certificate of incorporation provides that holders of Palvella preferred stock have the right to convert such shares into shares of Palvella common stock at any time at a conversion rate in accordance with the terms of Palvella’s third amended and restated certificate of incorporation.
Upon the happening of certain events as described in Palvella’s third amended and restated certificate of incorporation, all outstanding shares of Palvella preferred stock will automatically convert into shares of Palvella common stock at the conversion rate calculated in accordance with Palvella’s third amended and restated certificate of incorporation. |
The certificates of designation for each series of Pieris preferred stock (other than the Series F Preferred Stock, which has no conversion rights) provide that the holders of such series Pieris preferred stock have the right to convert their shares of such series into shares of Pieris common stock in accordance with the terms and conditions set forth in the certificate of designation for such series. |
|
Right of First Refusal |
Pursuant to the Co-Sale Agreement, certain Palvella stockholders party to the agreement, the Key Holders defined therein, wishing to transfer any shares of Palvella’s capital stock must first provide Palvella with the right to purchase such shares. In such an event, if Palvella does not elect to exercise its right of first refusal, certain Palvella stockholders detailed in the Co-Sale Agreement, or the Investors defined therein, have a secondary right of refusal to purchase all or a portion of the shares which are proposed for sale or transfer by the Key Holders. |
Neither the Nevada Charter nor the Nevada Bylaws provide for a right of first refusal relating to Pieris capital stock. |
|
Right of Co-Sale |
Pursuant to the Co-Sale Agreement, each Investor (as defined in the Co-Sale Agreement) has a right of co-sale with respect to the Palvella capital stock proposed to be transferred by any Key Holder which is not earlier purchased by Palvella by exercise of its right of first refusal (as described above) or by any Investor by exercise of their secondary right of first refusal (as described above). |
Neither the Nevada Charter nor the Nevada Bylaws provide for a right of co-sale relating to Pieris capital stock. |
|
Preemptive Rights |
Pursuant to the Investors’ Rights Agreement, if Palvella proposes to offer or sell new equity securities, Palvella shall first offer such securities to the Major Investors (as define din the Investor Rights Agreement). Each of the Major Investors will then have the right to purchase securities in such new offering equal to the proportion of the ownership interest of such Major Investor prior to such offering. |
Pieris stockholders do not have preemptive rights under the Nevada Charter, Nevada Bylaws or pursuant to the NRS. Thus, if additional shares of Pieris common stock are issued, the current holders of Pieris common stock will own a proportionately smaller interest in a larger number of outstanding shares of Pieris common stock to the extent that they do not participate in the additional issuance. |
|
Distributions to Stockholders |
Subject to restrictions by applicable law or those contained in Palvella’s third amended and restated certificate of incorporation, the Palvella board of directors may declare and pay dividends upon the shares of Palvella’s capital stock. Dividends may be paid in cash, in property or in shares of Palvella capital stock, subject to the provisions of Palvella’s third amended and restated certificate of incorporation. Palvella will not declare, pay or set aside dividends on any share of any other class or series of its capital stock (other than dividends on shares of its common stock payable in shares of common stock) in any calendar year unless (in addition to obtaining any consents required by Palvella’s third amended and restated certificate of incorporation) the holders of its preferred stock then outstanding shall first receive, or simultaneously receive, dividends on each outstanding share of Palvella’s preferred stock in accordance with the terms of Palvella’s third amended and restated certificate of incorporation. The Palvella board of directors may fix a record date for the determination of holders of Palvella capital stock entitled to receive payment of a dividend or distribution declared thereon, which record date is not to precede the date upon which the resolution fixing the record date is adopted, and which record date may not be more than 60 days prior to the date fixed for the payment thereof. If no record date is fixed, the record date for determining Palvella stockholders entitled to receive payment of a dividend or distribution will be at the close of business on the day on which Palvella board of directors adopts the resolution declaring such dividend or distribution.
In the event of a voluntary or involuntary liquidation, dissolution or winding up of Palvella, or in the case of any Deemed Liquidation Event (as defined in Palvella’s third amended and restated certificate of incorporation), the holders of outstanding shares of Palvella preferred stock shall be entitled to be paid, on a pari passu basis among each other and before any payment is made to the holders of common stock of Palvella, an amount per share equal to the greater of (i) the applicable Original Issue Price (which is $5.2879 for Series D preferred stock and Series C preferred stock, $and $3.19 for Series B preferred stock, $1.64 for Series A-2 preferred stock and $1.31 for Series A-1 preferred stock), plus any dividends declared but unpaid thereon, or (ii) such amount per share as would have been payable had all shares of such series of Palvella preferred stock been converted into Palvella common stock immediately prior to such event. |
The Nevada Charter provides that dividends may be declared and paid on Pieris common stock from funds legally available therefor, if, as and when determined by the Pieris board of directors in their sole discretion, subject to provisions of law, any provision of the Nevada Charter and subject to the relative rights and preferences of any shares of then outstanding preferred stock.
The NRS provides that no distribution (including dividends on, or redemption or purchases of, shares of capital stock or distributions of indebtedness) may be made if, after giving effect to such distribution, (i) the corporation would not be able to pay its debts as they become due in the usual course of business, or, (ii) except as otherwise specifically permitted by the articles of incorporation, the corporation’s total assets would be less than the sum of its total liabilities plus the amount that would be needed at the time of a dissolution to satisfy the preferential rights of preferred stockholders. The Pieris board of directors may consider financial statements prepared on the basis of accounting practices that are reasonable under the circumstances, a fair valuation, including but not limited to unrealized appreciation and depreciation, and any other method that is reasonable under the circumstances. The payment of dividends following the consummation of the Merger will be within the discretion of the combined company’s board of directors, subject to applicable law. |
|
Registration Rights |
Under the Investors’ Rights Agreement, certain holders of Palvella preferred stock that are party to the Investors’ Rights Agreement, have certain registration rights, including the right to demand that Palvella file a registration statement, so called “demand” registration rights, or request that their shares be covered by a registration statement that Palvella is otherwise filing, so-called “piggyback” registration rights. |
No Pieris stockholder has registration rights. |
COMPARISON OF STOCKHOLDER RIGHTS UNDER DELAWARE AND NEVADA LAW
The rights of Pieris stockholders are currently governed by the NRS, the Nevada Charter and the Nevada Bylaws. Following completion of the Merger, the rights of the combined company’s stockholders will be governed by the DGCL, the Delaware Charter and the Delaware Bylaws.
The statutory corporate laws of Delaware, as governed by the DGCL, are similar in many respects to those of Nevada, as governed by the NRS. However, there are certain differences that may affect your rights as a stockholder, as well as the corporate governance of the combined company. The following are brief summaries of material differences between the current rights of Pieris stockholders and the rights of stockholders of the combined company following consummation of the Merger. The following discussion does not provide a complete description of the differences that may affect your rights as a stockholder. This summary is qualified in its entirety by reference to the NRS and DGCL as well as to the Delaware Charter and Delaware Bylaws and the Nevada Charter and Nevada Bylaws.
Increasing or Decreasing Authorized Capital Stock
Under both Delaware and Nevada law, the stockholders must approve an increase or decrease in the number of authorized shares in accordance with the provisions of the applicable statutes. The NRS also allows the board of directors of a Nevada corporation, unless otherwise provided in the articles of incorporation, to increase or decrease the number of authorized shares of a class or series of the corporation’s shares and correspondingly effect a forward or reverse split of the same class or series of the corporation’s shares (and change the par value thereof) without a vote of the stockholders, as long as the action taken (i) does not adversely change or alter any right or preference of the stockholders and does not include any provision[s] pursuant to which only money will be paid or scrip issued to stockholders who hold 10% or more of the outstanding shares of the affected class and series, and (ii) who would otherwise be entitled to receive fractions of shares in exchange for the cancellation of all of their outstanding shares. Delaware law has no similar provision. In such circumstances, the proposed increase or decrease must be approved by the stockholders holding a majority of the voting power of the affected class or series. The Nevada Charter does not otherwise provide. As long as any shares of Palvella preferred stock are outstanding, the written consent or affirmative vote of the Requisite Holders is needed to amend, alter or repeal any provision of Palvella’s third amended and restated certificate of incorporation or to increase or decrease the authorized number of directors constituting the Palvella board of directors. As long as at least 36,704 shares of Series D preferred stock are outstanding, the written consent or affirmative vote of the holders of at least 67% of the outstanding shares of Palvella Series D preferred stock, voting as a separate class, is needed to increase or decrease the authorized number of shares of Series D preferred stock. As long as at least 2,127,498 shares of Series C preferred stock are outstanding, the written consent or affirmative vote of the holders of at least 67% of the outstanding shares of Palvella Series C preferred stock, voting as a separate class, is needed to increase or decrease the authorized number of shares of Series C preferred stock. As long as at least 383,382 shares of Series B preferred stock are outstanding, the written consent or affirmative vote of the holders of at least 67% of the outstanding shares of Palvella Series C preferred stock, voting as a separate class, is needed to increase or decrease the authorized number of shares of Series B preferred stock. As long as at least 870,509 shares of Series A-1 preferred stock and Series A-2 preferred stock are outstanding, the written consent or affirmative vote of the holders of at least 67% of the outstanding shares of Palvella Series A-1 preferred stock and Series A-2 preferred stock, voting as a separate class, is needed to increase or decrease the authorized number of shares of Series A-1 preferred stock and Series A-2 preferred stock.
Classified Board of Directors
The NRS permits any Nevada corporation to classify its board of directors into any number of classes with staggered terms of office, as long as at least one-fourth of the total number of directors is elected annually. The Nevada Charter and Nevada Bylaws also provide for a classified board of directors, with the election of one class of directors each year for a three-year term. The DGCL also permits any Delaware corporation to classify its board of directors into as many as three classes with staggered terms of office. The Delaware Charter and Delaware Bylaws do not provide for a classified board of directors, and directors are elected for annual terms. Each director’s term continues until the election and qualification of his or her successor, or his or her earlier death, resignation or removal.
Cumulative Voting
Cumulative voting for directors entitles each stockholder to cast a number of votes that is equal to the number of voting shares held by such stockholder multiplied by the number of directors to be elected, and to cast all such votes for one nominee or distribute such votes among up to as many candidates as there are positions to be filled. Cumulative voting may enable a minority stockholder or group of stockholders to elect at least one representative to the board of directors where such stockholders would not be able to elect any directors without cumulative voting.
The NRS permits any Nevada corporation to provide in its articles of incorporation the right to cumulative voting in the election of directors if certain procedures are followed. Although the DGCL does not generally grant stockholders cumulative voting rights, a Delaware corporation may provide in its certificate of incorporation for cumulative voting in the election of directors.
The Nevada Charter does not provide for cumulative voting in the election of directors. Similarly, the Delaware Charter does not provide for cumulative voting.
Vacancies
Under both the DGCL and the NRS, subject to the certificate or articles of incorporation and bylaws, vacancies on the board of directors, including those resulting from any increase in the authorized number of directors, may be filled by the affirmative vote of a majority of the remaining directors then in office, even if less than a quorum. Any director so appointed will hold office for the remainder of the term of the director no longer on the board. The Nevada Charter and Nevada Bylaws provide that, subject to the rights of the holders of any series of preferred stock then outstanding, any vacancies in the Pieris board of directors resulting from death, resignation, retirement, disqualification, removal from office or other cause shall, unless otherwise required by law, by the Nevada Charter or by resolution of the Pieris board of directors, be filled only by a majority vote of the directors then in office even though less than a quorum, or by a sole remaining director, and not by the stockholders. The Delaware Charter provides that Series D Directors of Palvella, the Series C Directors of Palvella, the Junior Preferred Director of Palvella and the Common Director of Palvella may be removed without cause only by the affirmative vote or written consent of the holders of the shares of the series of preferred stock or shares of common stock entitled to elect them.
Removal of Directors
Under the DGCL, the holders of a majority of shares of each class entitled to vote at an election of directors may vote to remove any director or the entire board without cause unless (i) the board is a classified board, in which case directors may be removed only for cause, or (ii) the corporation has cumulative voting, in which case, if less than the entire board is to be removed, no director may be removed without cause if the votes cast against their removal would be sufficient to elect him or her. Currently, the Delaware charter does not provide for a classified board or cumulative voting. The Delaware Charter provides that Series D Directors of Palvella, the Series C Directors of Palvella, the Junior Preferred Director of Palvella and the Common Director of Palvella may be removed without cause only by the affirmative vote or written consent of the holders of the shares of the series of preferred stock or shares of common stock entitled to elect them. The NRS does not make a distinction between the removal for cause or without cause and requires the vote of the holders of at least two-thirds of the shares or class or series of shares of the issued and outstanding stock entitled to vote at an election of directors in order to remove a director or all of the directors. However, the Nevada Charter and the Nevada Bylaws provide that subject to the rights of the holders of any series of preferred stock then outstanding, any director, or the entire Pieris board of directors, may be removed from office at any time only for cause and only by the affirmative vote of the holders of at least 80% of the voting power of all of the then outstanding shares of Pieris capital stock entitled to vote at an election of directors, voting together as a single class.
Limitation on Personal Liability of Directors and Officers
The NRS and the DGCL each, by way of statutory provisions or permitted provisions in corporate charter documents, eliminate or limit the personal liability of directors and officers to the corporation or their stockholders for monetary damages for breach of a director’s fiduciary duty, subject to the differences discussed below.
The DGCL permits corporations to adopt charter provisions exculpating directors from monetary liability to the corporation and its stockholders for breaches of the directors’ duty of care, but the statute precludes liability limitation for breach of the duty of loyalty, acts or omissions not in good faith or involving intentional misconduct and for paying dividends or repurchasing stock out of other than lawfully available funds. With respect to a corporation’s most senior officers—namely, the chief executive officer, president, chief financial officer, chief operating officer, chief legal officer, controller, treasurer and chief accounting officer, as well as any other persons identified as “named executive officers” in a company’s most recent SEC filings or who otherwise consent to jurisdiction under Delaware’s long-arm statute applicable to directors and officers of Delaware corporations—the DGCL authorizes similar limitations of liability, but only in connection with direct claims brought by stockholders, including class actions. The DGCL does not, however, authorize a limitation on liability of officers for breach of fiduciary duty arising out of claims brought by the corporation itself or for derivative claims brought by stockholders in the name of the corporation.
Under the NRS, in order for a director or officer to be individually liable to the corporation or its stockholders or creditors for damages as a result of any act or failure to act, the presumption of the business judgment rule must be rebutted and it must be proven that the director’s or officer’s act or failure to act constituted a breach of their fiduciary duties as a director or officer, and that the breach of those duties involved intentional misconduct, fraud or a knowing violation of law. Unlike the DGCL, however, the limitation on director and officer liability under the NRS does not distinguish the duty of loyalty or transaction from which a director derives an improper personal benefit, but does, pursuant to NRS 78.300, impose limited personal liability on directors for distributions made in violation of NRS 78.288. Further, the NRS permits a corporation to renounce in its articles of incorporation any interest or expectancy to participate in specific or specified classes or categories of business opportunities. Both the DGCL and the NRS permit limitation of liability which applies to both directors and officers, though the NRS also expressly applies this limitation to liabilities owed to creditors of the corporation. Furthermore, under the NRS, it is not necessary for a corporation to adopt provisions in its articles of incorporation limiting personal liability of directors or officers, as this limitation is provided by statute.
The Delaware Charter provides for exculpation of directors to the fullest extent permitted by the DGCL. As described above, the NRS provides broader protection from personal liability for directors and officers than the DGCL. The Nevada Charter provides a limitation to director and officer liability to the fullest extent permitted by Nevada law. The Delaware Charter, in contrast, does not provide for exculpation of directors and officers as described above under Nevada law.
Indemnification
The NRS and the DGCL each have statutory mechanisms that permit corporations to indemnify directors, officers, employees and agents in similar circumstances, subject to the differences discussed below.
In suits that are not brought by or in the right of the corporation, both jurisdictions’ statutory indemnification mechanisms permit a corporation to indemnify current and former directors, officers, employees and agents for attorneys’ fees and other expenses, judgments and amounts paid in settlement that the person actually and reasonably incurred in connection with the action, suit or proceeding. The person seeking indemnity may recover under these statutory provisions as long as they acted in good faith and believed their actions were either in the best interests of or not opposed to the best interests of the corporation. Under the statutory indemnification mechanism provided under the NRS, the person seeking indemnity may also be indemnified if they are not held liable for breach of their fiduciary duties. Similarly, with respect to a criminal proceeding, the person seeking indemnification must not have had any reasonable cause to believe their conduct was unlawful. The articles of incorporation may provide for further indemnification than that described in the statutory mechanism provided under the NRS.
In derivative suits, a corporation in either jurisdiction may indemnify its directors, officers, employees or agents for expenses that the person actually and reasonably incurred. A corporation may not indemnify a person if the person was adjudged to be liable to the corporation unless a court otherwise orders.
Under the statutory indemnification mechanism in either jurisdiction, no corporation may indemnify a party unless it decides that indemnification is proper. Under the DGCL, the corporation through its stockholders, directors or independent legal counsel will determine whether the conduct of the person seeking indemnity conformed to the statutory provisions governing indemnity. Similarly, under the statutory indemnification mechanisms under the NRS, the corporation through its stockholders, directors or independent counsel must determine that the indemnification is proper.
The indemnification pursuant to the statutory mechanisms available under the NRS, as described above, does not exclude any other rights to which a person seeking indemnification or advancement of expenses may be entitled under the articles of incorporation or any bylaw, agreement, vote of stockholders or disinterested directors or otherwise. However, unless otherwise ordered by a court, indemnification may not be made to or on behalf of any director or officer finally adjudged by a court of competent jurisdiction, after exhaustion of any appeals taken therefrom, to be liable for intentional misconduct, fraud or a knowing violation of law, and such misconduct, fraud or violation was material to the cause of action.
Both the Delaware Charter and the Nevada Charter provide for indemnification to the fullest extent permitted by their respective laws, which can help the combined company retain and attract high quality and experienced directors.
Action by Written Consent of Directors
Both the DGCL and NRS provide that, unless the articles or certificate of incorporation or the bylaws provide otherwise, any action required or permitted to be taken at a meeting of the directors or a committee thereof may be taken without a meeting if all members of the board or committee, as the case may be, consent to the action in writing.
Neither the Delaware Charter or Delaware Bylaws, nor the Nevada Charter or Nevada Bylaws, limit the type or nature of a board action taken by written consent.
Actions by Written Consent of Stockholders
Both the DGCL and NRS provide that, unless the articles or certificate of incorporation provide otherwise, any action required or permitted to be taken at a meeting of the stockholders may be taken without a meeting if the holders of outstanding stock, having at least the minimum number of votes that would be necessary to authorize or take the action at a meeting of stockholders, consent to the action in writing. In addition, the DGCL requires the corporation to give prompt notice of the taking of corporate action without a meeting by less than unanimous written consent to those stockholders who did not consent in writing. There is no equivalent notice requirement under the NRS.
The NRS also permits a corporation to prohibit stockholder action by written consent in lieu of a meeting of stockholders by including such prohibition in its articles of incorporation or bylaws.
The Delaware Charter and Bylaws provide that any stockholder may act by written consent if such consent is signed by holder of not less than minimum number of votes that would be necessary to authorize or take the corporate action at a meeting at which all shares entitled to vote thereon were present and voted, so long as action is taken in accordance with the Delaware Charter. The Nevada Charter and the Nevada Bylaws expressly prohibit stockholders from taking any action by written consent, and requires that any action required or permitted to be taken by the stockholders of the combined company may be effected only at a duly called annual or special meeting of stockholders.
Dividends and Distributions
Delaware law is more restrictive than Nevada law with respect to dividend payments. Unless further restricted in the certificate of incorporation, the DGCL permits a corporation to declare and pay dividends out of either (i) surplus, or (ii) if no surplus exists, out of net profits for the fiscal year in which the dividend is declared and/or the preceding fiscal year (provided that the amount of capital of the corporation is not less than the aggregate amount of the capital represented by the issued and outstanding stock of all classes having a preference upon the distribution of assets). The DGCL defines surplus as the excess, at any time, of the net assets of a corporation over its stated capital. In addition, the DGCL provides that a corporation may redeem or repurchase its shares only when the capital of the corporation is not impaired and only if such redemption or repurchase would not cause any impairment of the capital of the corporation.
The NRS provides that no distribution (including dividends on, or redemption or purchases of, shares of capital stock or distributions of indebtedness) may be made if, after giving effect to such distribution, (i) the corporation would not be able to pay its debts as they become due in the usual course of business, or, (ii) except as otherwise specifically permitted by the articles of incorporation, the corporation’s total assets would be less than the sum of its total liabilities plus the amount that would be needed at the time of a dissolution to satisfy the preferential rights of preferred stockholders (this “clause (ii) condition”, the Balance Sheet Test). Directors may consider financial statements prepared on the basis of accounting practices that are reasonable under the circumstances, a fair valuation, including but not limited to unrealized appreciation and depreciation, and any other method that is reasonable under the circumstances. The Nevada Charter does not, however, eliminate the requirement that the combined company comply with the Balance Sheet Test with respect to any distribution. The payment of dividends following the consummation of the Merger will be within the discretion of the combined company’s board of directors, subject to applicable law.
Appraisal or Dissenter’s Rights
In both jurisdictions, dissenting stockholders of a corporation engaged in certain major corporate transactions are entitled to appraisal rights. Appraisal or dissenter’s rights permit a stockholder to receive cash generally equal to the fair value of the stockholder’s shares (as determined by agreement of the parties or by a court) in lieu of the consideration such stockholder would otherwise receive in any such transaction.
Under Section 262 of the DGCL, appraisal rights are generally available for the shares of any class or series of stock of a Delaware corporation in a merger, consolidation or conversion, provided that no appraisal rights are available with respect to shares of any class or series of stock if, at the record date for the meeting held to approve such transaction, such shares of stock, or depositary receipts in respect thereof, are either (i) listed on a national securities exchange or (ii) held of record by more than 2,000 holders, unless the stockholders receive in exchange for their shares anything other than shares of stock of the surviving or resulting corporation (or depositary receipts in respect thereof), or of any other corporation that is listed on a national securities exchange or held by more than 2,000 holders of record, cash in lieu of fractional shares or fractional depositary receipts described above or any combination of the foregoing.
In addition, Section 262 of the DGCL allows beneficial owners of shares to file a petition for appraisal without the need to name a nominee holding such shares on behalf of such owner as a nominal plaintiff and makes it easier than under Nevada law to withdraw from the appraisal process and accept the terms offered in the merger, consolidation or conversion. Under the DGCL, no appraisal rights are available to stockholders of the surviving or resulting corporation if the merger did not require their approval. The Delaware Charter and Delaware Bylaws do not provide for appraisal rights in addition to those provided by the DGCL.
Under the NRS, a stockholder is entitled to dissent from, and obtain payment for, the fair value of the stockholder’s shares in the event of (i) certain acquisitions of a controlling interest in the corporation, (ii) consummation of a plan of merger, if approval by the stockholders is required for the merger, regardless of whether the stockholder is entitled to vote on the merger or if the domestic corporation is a subsidiary and is merged with its parent, or if the domestic corporation is a constituent entity in a merger pursuant to NRS 92A.133, (iii) consummation of a plan of conversion to which the corporation is a party, (iv) consummation of a plan of exchange in which the corporation is a party, (iv) any corporate action taken pursuant to a vote of the stockholders, if the articles of incorporation, bylaws or a resolution of the board of directors provides that voting or nonvoting stockholders are entitled to dissent and obtain payment for their shares, or (v) any corporate action to which the stockholder would be obligated, as a result of the corporate action, to accept money or scrip rather than receive a fraction of a share in exchange for the cancellation of all the stockholder’s outstanding shares, except where the stockholder would not be entitled to receive such payment pursuant to NRS 78.205, 78.2055 or 78.207.
Also under the NRS, holders of covered securities (generally those that are listed on a national securities exchange), any shares traded in an organized market and held by at least 2,000 stockholders of record with a market value of at least $20,000,000, and any shares issued by an open-end management investment company registered under the Investment Company Act of 1940 and which may be redeemed at the option of the holder at net asset value, are generally not entitled to dissenter’s rights. However, this exception is not available if (i) the articles of incorporation of the corporation issuing the shares provide that such exception is not available, (ii) the resolution of the board of directors approving the plan of merger, conversion or exchange expressly provides otherwise or (iii) the holders of the class or series of stock are required by the terms of the corporate action to accept for the shares anything except cash, shares of stock or other securities as described in NRS 92A.390(3) or any combination thereof. The NRS prohibits a dissenting stockholder from voting their shares or receiving certain dividends or distributions after their dissent. As with the Delaware Charter and the Delaware Bylaws, the Nevada Charter and Nevada Bylaws do not provide for dissenter’s rights in addition to those provided by the NRS.
The mechanics and timing procedures vary somewhat between Delaware and Nevada, but both require technical compliance with specific notice and payment protocols.
Special Meetings of the Stockholders
The DGCL permits special meetings of stockholders to be called by the board of directors or by any other person authorized in the certificate of incorporation or bylaws to call a special stockholder meeting. The NRS permits special meetings of stockholders to be called by the entire board of directors, any two directors or the President, unless the articles of incorporation or bylaws provide otherwise.
Under the Delaware Bylaws, a special meeting of stockholders may be called by a majority vote of the board, the Chairman of the Board, the Chief Executive Officer or the President. The Nevada Charter and Nevada Bylaws provide that special meetings of stockholders of the combined company may be called only by the board of directors pursuant to a resolution adopted by a majority of the total number of then authorized directors whether or not there exist any vacancies in previously authorized directorships.
Meetings Pursuant to Petition of Stockholders
The DGCL provides that a director or a stockholder of a corporation may apply to the Court of Chancery of Delaware if the corporation fails to hold an annual meeting for the election of directors or there is no written consent to elect directors in lieu of an annual meeting for a period of 30 days after the date designated for the annual meeting or, if there is no date designated, within 13 months after the last annual meeting.
Under the NRS, stockholders having not less than 15% of the voting power may petition the district court to order a meeting for the election of directors if a corporation fails to call a meeting for that purpose within 18 months after the last meeting at which directors were elected.
Adjournment of Stockholder Special Meetings
Under the DGCL, if a meeting of stockholders is adjourned due to lack of a quorum and the adjournment is for more than 30 days, or if after the adjournment a new record date is fixed for the adjourned meeting, notice of the adjourned meeting must be given to each stockholder of record entitled to vote at the meeting. At the adjourned meeting the corporation may transact any business that might have been transacted at the original meeting.
In contrast, under the NRS, a corporation is not required to give any notice of an adjourned meeting or of the business to be transacted at an adjourned meeting, other than by announcement at the meeting at which the adjournment is taken, unless the board of directors of the corporation fixes a new record date for the adjourned meeting or the meeting date is adjourned to a date more than 60 days later than the date set for the original meeting, in which case a new record date must be fixed and notice given. The Nevada Bylaws reduce this 60-day period to 30 days.
Duration of Proxies
Under the DGCL, a proxy executed by a stockholder will remain valid for a period of three years, unless the proxy provides for a longer period.
Under the NRS, a proxy is effective only for a period of six months, unless it is coupled with an interest or unless otherwise provided in the proxy, which duration may not exceed seven years. The NRS also provides for irrevocable proxies, without limitation on duration, in limited circumstances.
Quorum and Voting
The DGCL provides that the certificate of incorporation and bylaws may establish quorum and voting requirements, but in no event shall a quorum consist of less than one-third of the shares entitled to vote. If the certificate of incorporation and bylaws are silent as to specific quorum and voting requirements: (a) a majority of the shares entitled to vote shall constitute a quorum at a meeting of stockholders; (b) in all matters other than the election of directors, the affirmative vote of the majority of shares present at the meeting and entitled to vote on the subject matter shall be the act of the stockholders; (c) directors shall be elected by a plurality of the votes of the shares present at the meeting and entitled to vote on the election of directors; and (d) where a separate vote by a class or series is required, a majority of the outstanding shares of such class or series shall constitute a quorum entitled to take action with respect to that vote on that matter and, in all matters other than the election of directors, the affirmative vote of the majority of shares of such class or series present at the meeting shall be the act of such class or series or classes or series. A bylaw amendment adopted by stockholders which specifies the votes that shall be necessary for the election of directors shall not be further amended or repealed by the board. The Delaware Bylaws provide that the holders of shares representing a majority of capital stock issued and entitled to vote thereat present in person or represented by proxy shall constitute a quorum at all meetings of the stockholders for the transaction of business. The Delaware Charter provides that the holders of Palvella’s Series D preferred stock, exclusively and as a separate class, shall be entitled to elect two directors of Palvella, (b) the holders of Palvella’s Series C preferred stock, exclusively and as a separate class, shall be entitled to elect two directors of Palvella, (c) the holders of the shares of Palvella’s Junior preferred stock, voting together as a single class on an as-converted to Palvella’s voting common stock basis, shall be entitled to elect one director of Palvella, and (d) the holders Palvella’s shares of voting common stock, exclusively and as a separate class, shall be entitled to elect one director of Palvella.
The NRS provides that, unless the articles of incorporation or bylaws provide otherwise, a majority of the voting power of the corporation, present in person or by proxy at a meeting of stockholders (regardless of whether the proxy has authority to vote on any matter), constitutes a quorum for the transaction of business. Under the NRS, unless the articles of incorporation or bylaws provide for different proportions, action by the stockholders on a matter other than the election of directors is approved if the number of votes cast in favor of the action exceeds the number of votes cast in opposition to the action. Unless provided otherwise in the corporation’s articles of incorporation or bylaws, directors are elected at the annual meeting of stockholders by plurality vote. The Nevada Bylaws provide that unless otherwise required by law or the Nevada Charter or the rules of any stock exchange upon which the combined company’s securities are listed, the holders of a majority of the voting power of the combined company’s capital stock represented in person or by proxy shall constitute a quorum at all meetings of the stockholders for the transaction of business. The Nevada Bylaws provide that except as otherwise provided in the terms of any class or series of preferred stock, (i) all elections of directors of the combined company at any meeting of the stockholders shall be determined by a plurality of the votes cast, and (ii) except as otherwise required by law, the Nevada Charter or the Nevada Bylaws, or the rules of any stock exchange upon which the combined company’s securities are listed, all other matters proposed at any meeting of the stockholders shall be determined by a majority of the votes cast affirmatively or negatively.
Business Opportunities
The NRS permits a Nevada corporation to renounce, in its articles of incorporation or by action of the board of directors, any interest or expectancy to participate in specified business opportunities or specified classes or categories of business opportunities that are presented to the corporation or one or more of its officers, directors or stockholders. The Nevada Charter does not include a renunciation of any interest or expectancy in corporate opportunities.
Similar to the NRS, under Delaware law, the corporate opportunity doctrine holds that a corporate officer or director may not generally and unilaterally take a business opportunity for their own if: (i) the corporation is financially able to exploit the opportunity; (ii) the opportunity is within the corporation’s line of business; (iii) the corporation has an interest or expectancy in the opportunity; and (iv) by taking the opportunity for their own, the corporate fiduciary will thereby be placed in a position inimical to his duties to the corporation. The DGCL permits a Delaware corporation to renounce, in its certificate of incorporation or by action of the board of directors, any interest or expectancy of the corporation in, or being offered an opportunity to participate in, specified business opportunities or specified classes or categories of business opportunities that are presented to the corporation or one or more of its officers, directors or stockholders.
Regulatory Matters
The consummation of the Merger requires the filing of the Certificate of Merger with the Office of the Secretary of State in Delaware.
No Dissenter’s or Appraisal Rights for Pieris Stockholders
Holders of Pieris common stock are not entitled to dissenter’s or appraisal rights under Nevada law in connection with the Merger. Under the DGCL, holders of Palvella common stock are entitled to appraisal rights in connection with the Merger.
No Material Accounting Implications
Effecting the Merger will not have any material accounting implications for the combined company.
PRINCIPAL STOCKHOLDERS OF PIERIS
The following table and accompanying footnotes set forth certain information with respect to the beneficial ownership of Pieris common stock as of October 28, 2024 for:
|
● |
each person, or group of affiliated persons, who is known by Pieris to beneficially own more than 5% of Pieris common stock; |
|
● |
each of Pieris’ named executive officers; |
|
● |
all of Pieris’ directors as of October 28, 2024; and |
|
● |
all of Pieris’ executive officers and directors as a group. |
Beneficial ownership prior to the completion of the Merger is based on 1,320,240 shares of Pieris common stock outstanding as of October 28, 2024.
Beneficial ownership is determined in accordance with the rules and regulations of the SEC and includes voting or investment power with respect to Pieris common stock. Shares of Pieris common stock that an individual has a right to acquire within 60 days after October 28, 2024 are considered outstanding and beneficially owned by the person holding such right for the purpose of calculating the percentage ownership of that person but not for the purpose of calculating the percentage ownership of any other person, except with respect to the percentage ownership of all directors and executive officers. Except as otherwise noted, the persons and entities in this table have sole voting and investing power with respect to all of the shares of Pieris common stock beneficially owned by them, subject to community property laws, where applicable. Except as otherwise set forth below, the address of each beneficial owner is c/o Pieris Pharmaceutics, Inc., 225 Franklin Street, 26th Floor, Boston, Massachusetts 02110.
|
Name of Beneficial Owner |
Shares Beneficially Owned |
Percentage of Shares Beneficially Owned (%) |
||||||
| 5%+ Stockholders: | ||||||||
|
Biotechnology Value Fund, L.P. and affiliates(1) |
257,622 | 19.51 | % | |||||
|
ADAR1 Capital Management, LLC and affiliates(2) |
138,646 | 10.50 | % | |||||
| Dellora Investments LP and affiliates(3) | 108,974 | 8.25 | % | |||||
|
Directors and Named Executive Officers: |
||||||||
|
Stephen S. Yoder(4) |
56,783 | 4.12 | % | |||||
|
Thomas Bures(5) |
8,674 | * | ||||||
|
Shane Olwill(6) |
11,576 | * | ||||||
|
James Geraghty(7) |
4,285 | * | ||||||
|
Ann Barbier, M.D., Ph.D.(8) |
2,875 | * | ||||||
|
Peter Kiener, D.Phil.(9) |
2,875 | * | ||||||
|
Christopher Kiritsy(10) |
3,562 | * | ||||||
|
Michael Richman(11) |
5,487 | * | ||||||
|
Matthew L. Sherman, M.D.(12) |
2,875 | * | ||||||
|
Maya R. Said, Sc.D.(13) |
2,625 | * | ||||||
|
All current executive officers and directors as a group (10 persons)(14) |
101,569 | 7.15 | % | |||||
__________
* Represents beneficial ownership of less than 1%.
|
(1) |
This information is based on a Schedule 13G filed with the SEC on May 15, 2024 and information available to us. The address of the principal business and office of BVF, and certain of its affiliates is 44 Montgomery Street, 40th Floor, San Francisco, California, 94104. BVF and its related entities beneficially own (i) 257,622 shares of common stock, excluding (ii) 85 shares of Series A Convertible Preferred Stock, which is convertible into 1,133 shares of common stock, (iii) 4,026 shares of Series B Convertible Preferred Stock, which is convertible into 53,706 shares of common stock, (iv) 3,506 shares of Series C Convertible Preferred Stock, which is convertible into 46,770 shares of common stock, (v) 3,000 shares of Series D Convertible Preferred Stock, which is convertible into 40,020 shares of common stock, (vi) 5,000 shares of Series E Convertible Preferred Stock, which is convertible into 66,700 shares of common stock and (vii) warrants exercisable for 46,999 shares of common stock. The Series A, Series B, Series C, Series D and Series E Preferred Stock may not be converted and the warrants may not be exercised if, after such conversion or exercise, BVF and its affiliates would beneficially own more than 9.99% of the number of shares of common stock then issued and outstanding. As a result of the limitation in the previous sentence, for purposes of the table above, no shares of common stock are included from the preferred stock and warrants. BVF I GP LLC, or BVF GP, is the general partner of BVF; BVF II GP LLC, or BVFII GP, is the general partner of Biotechnology Value Fund II, L.P., or BVF II; BVF Inc., or BVF Inc., is the general partner of Biotechnology Value Trading Fund OS LP, or Trading Fund OS. BVF GP Holdings LLC, or BVF GPH, is the sole member of each of BVF GP and BVF II GP. BVF Partners L.P., or Partners, is the investment manager of BVF, BVF2, Trading Fund OS and certain managed accounts and the sole member of Partners OS. BVF Inc. is the general partner of Partners and the managing member of BVF GPH, and Mark N. Lampert is the sole director and officer of BVF Inc. BVF GP disclaims beneficial ownership of the shares of common stock beneficially owned by BVF. BVF2 GP disclaims beneficial ownership of the shares of common stock beneficially owned by BVF2. Partners OS disclaims beneficial ownership of the shares of common stock beneficially owned by Trading Fund OS. BVF GPH disclaims beneficial ownership of the shares of common stock beneficially owned by BVF and BVF2. Each of Partners, BVF Inc. and Mr. Lampert disclaims beneficial ownership of the shares of common stock beneficially owned by BVF, BVF 2, Trading Fund OS and certain Partners managed accounts. |
|
(2) |
This information is based on a Schedule 13G filed with the SEC on August 1, 2024 and information available to us. The address of the principal business and office of ADAR1 Capital Management, LLC, or ADAR1 Capital Management, and certain of its affiliates is 3503 Wild Cherry Drive, Building 9, Austin, Texas 78738. ADAR1 and its related entities beneficially own 138,646 shares of common stock. ADAR1 Capital Management acts as investment manager to, and manages investment and trading accounts of ADAR1 Partners, LP. ADAR1, pursuant to an investment sub-advisory agreement, acts as the subadvisor for Spearhead Insurance Solutions IDF, LLC – Series ADAR1, or Spearhead, and manages and controls the trading accounts for Spearhead. ADAR1 Capital Management GP, LLC, or ADAR1 General Partner acts as the general partner of ADAR1 Partners, LP, or ADAR1 Partners, and Mr. Daniel Schneeberger is the sole member of ADAR1 Capital Management and ADAR1 General Partner. As such, each of ADAR1 Capital Management, ADAR1 General Partner and Mr. Schneeberger may be deemed the indirect beneficial owner of securities held by ADAR1 Partners and Spearhead. |
|
(3) |
This information is based on a Schedule 13G filed with the SEC on August 7, 2024. The address of the principal business and office of Dellora Investments LP, or Dellora, and certain of its affiliates is 283 Greenwich Avenue, 3rd floor, Greenwich, Connecticut 06830. Dellora, which serves as investment adviser to private funds, including but not limited to Dellora Investments Master Fund, LP, or collectively, the "Dellora Funds", may be deemed to be the beneficial owner of all shares of Pieris common stock held by the Dellora Funds. Kevin Pyun, as Principal of Dellora, with the power to exercise investment and voting discretion, may be deemed to be the beneficial owner of all shares of common stock held by the Dellora Funds. Pursuant to Rule 13d-4 under the Exchange Act, Dellora and Mr. Pyun expressly disclaim beneficial ownership over any of the securities held by the Dellora Funds. |
|
(4) |
Includes 75 shares of our common stock and 56,708 shares issuable upon the exercise of options to purchase common stock, which are exercisable within 60 days of October 28, 2024. |
|
(5) |
Includes 241 shares of our common stock and 8,433 shares issuable upon the exercise of options to purchase common stock, which are exercisable within 60 days of October 28, 2024. |
|
(6) |
Includes 58 shares of common stock and 11,518 shares issuable upon the exercise of options to purchase common stock, which are exercisable within 60 days of October 28, 2024. Dr. Olwill will depart Pieris effective October 31, 2024. |
|
(7) |
Includes 250 shares of common stock and 4,035 shares issuable upon the exercise of options to purchase common stock, which are exercisable within 60 days of October 28, 2024. On August 7, 2024, Mr. Geraghty purchased one (1) share of Series F Preferred Stock from Pieris which entitles the holder to the voting power described in this proxy statement/prospectus on Form S-4, as amended. |
|
(8) |
Includes 2,875 shares issuable upon the exercise of options to purchase common stock, which are exercisable within 60 days of October 28, 2024. |
|
(9) |
Includes 2,875 shares issuable upon the exercise of options to purchase common stock, which are exercisable within 60 days of October 28, 2024. |
|
(10) |
Includes 250 shares of common stock and 3,312 shares issuable upon the exercise of options to purchase common stock, which are exercisable within 60 days of October 28, 2024. |
|
(11) |
Includes 5,487 shares issuable upon the exercise of options to purchase common stock, which are exercisable within 60 days of October 28, 2024. |
|
(12) |
Includes 2,875 shares issuable upon the exercise of options to purchase common stock, which are exercisable within 60 days of October 28, 2024. |
|
(13) |
Includes 2,625 shares issuable upon the exercise of options to purchase common stock, which are exercisable within 60 days of October 28, 2024. |
|
(14) |
See notes 4 through 13 above. |
PRINCIPAL STOCKHOLDERS OF PALVELLA
The following table sets forth the beneficial ownership of Palvella common stock as of October 28, 2024 by:
|
● |
each person or group of affiliated persons, who is known by Palvella to beneficially own more than 5% of its common stock; |
|
● |
each of Palvella’s named executive officers; |
|
● |
each of Palvella’s directors; and |
|
● |
all of Palvella’s executive officers and directors as a group. |
Palvella has determined beneficial ownership in accordance with the rules of the SEC, and thus it represents sole or shared voting or investment power with respect to Palvella’s securities. Unless otherwise indicated below, to Palvella’s knowledge, the persons and entities named in the table have sole voting and sole investment power with respect to all shares that they beneficially owned, subject to community property laws where applicable. The information does not necessarily indicate beneficial ownership for any other purpose, including for purposes of Sections 13(d) and 13(g) of the Exchange Act.
Palvella has based its calculation of the percentage of beneficial ownership prior to the completion of the Merger on 21,080,796 shares of Palvella common stock outstanding as of October 28, 2024, after giving effect to the conversion of all outstanding shares of its preferred stock and non-voting common stock. Palvella has deemed shares of its common stock subject to stock options that are currently exercisable or exercisable within 60 days of October 28, 2024 to be outstanding and to be beneficially owned by the person holding the stock option for the purpose of computing the percentage ownership of that person. Palvella did not deem these shares outstanding, however, for the purpose of computing the percentage ownership of any other person.
Unless otherwise indicated, the address of each beneficial owner listed in the table below is c/o Palvella Therapeutics, Inc., 125 Strafford Avenue, Suite 360, Wayne, Pennsylvania 19087.
|
Name of Beneficial Owner |
Number of Shares Beneficially Owned |
Percentage of Shares Owned (%) |
||||||
|
5% stockholders: |
||||||||
|
Samsara BioCapital(1) |
2,080,221 | 9.87 | % | |||||
|
Entities Affiliated with Caxton Alternative Management(2) |
1,323,777 | 6.28 | % | |||||
|
Entities affiliated with BVF Partners(3) |
1,418,332 | 6.73 | % | |||||
|
Entities affiliated with Adams Street Partners (4) |
1,323,777 | 6.28 | % | |||||
|
Directors and Named Executive Officers: |
||||||||
|
Wesley H. Kaupinen(5) |
5,269,852 | 24.90 | % | |||||
|
Kathleen Goin(6) |
327,358 | 1.53 | % | |||||
|
Jeffrey Martini, Ph.D (7) |
158,920 | * | ||||||
|
George M. Jenkins(8) |
600,329 | 2.84 | % | |||||
|
Todd C. Davis(9) |
266,930 | 1.27 | % | |||||
|
Tadd S. Wessel |
— | — | ||||||
|
Aditya Asokan, Ph.D. |
— | — | ||||||
|
Scott Morenstein |
— | — | ||||||
|
All current executive officers and directors as a group (10 individuals) |
6,778,212 | 31.03 | % | |||||
* Less than 1%.
|
(1) |
Consists of 2,080,221 shares of common stock issuable upon conversion of 1,891,110 shares of Series C preferred stock and 187,111 shares of Series D preferred stock held of record by Samsara BioCapital, L.P., or Samsara LP. Samsara BioCapital GP, LLC, or Samsara GP, is the sole general partner of Samsara LP and may be deemed to beneficially own the shares held of record by Samsara LP. Srinivas Akkaraju is a managing member of Samsara GP and may be deemed to beneficially own the shares held of record by Samsara LP. The mailing address of Samsara LP is 628 Middlefield Road, Palo Alto, California 94301. |
|
(2) |
Consists of (i) 1,244,350 shares of common stock issuable upon conversion of 1,244,350 shares of Series C preferred stock held of record by CDK Associates, L.L.C., or CDK, and (ii) 79,427 shares of common stock issuable upon the conversion of 79,427 shares of common stock issuable upon conversion of 79,427 shares of Series C preferred stock held of record by Third Street Holdings LLC, or TSH, and together with CDK, the CAM Entities. The 1,323,777 shares held of record in the aggregate by the CAM Entities may be deemed to be beneficially owned by (i) Caxton Corporation, the manager of CDK and (ii) Bruce Kovner, the chairman and sole shareholder of Caxton Corporation. Peter D’Angelo, as managing member of TSH, may be deemed to beneficially own the 79,427 shares of common stock issuable upon conversion of the Series C preferred stock held by TSH. The mailing address of the CAM Entities is c/o Caxton Alternative Management LP, 731 Alexander Road, Bldg 2, Suite 500, Princeton, New Jersey 08540. |
|
(3) |
Consists of (i) 757,178 shares of common stock issuable upon conversion of 757,178 shares of Series C preferred stock held of record by Biotechnology Value Fund, L.P., or BVF, (ii) 565,349 shares of common stock issuable upon conversion of 565,349 shares of Series C preferred stock held of record by Biotechnology Value Fund II, L.P., or BVF II, and (iii) 95,805 shares of common stock issuable upon conversion of 95,805 shares of Series C and Series D preferred stock held of record by Biotechnology Value Trading Fund OS, L.P., or Trading Fund OS, and together with BVF and BVF II, the BVF Entities. BVF I GP LLC, or BVF GP, as the general partner of BVF, may be deemed to beneficially own the 757,178 shares held of record by BVF. BVF II GP LLC, or BVF II GP, as the general partner of BVF II, may be deemed to beneficially own the 565,349 shares held of record by BVF II. BVF Partners OS Ltd., or Partner OS, as the general partner of Trading Fund OS, may be deemed to beneficially own the 95,805 shares held of record by Trading Fund OS. BVF GP Holdings LLC, or BVF GPH, as the sole member of each of BVF GP and BVF II GP, may be deemed to beneficially own the 1,232,443 shares held of record in the aggregate by BVF and BVF II. BVF Partners L.P., or Partners, as the investment manager of the BVF Entities, and the sole member of Partners OS, may be deemed to beneficially own the 1,418,332 shares held of record in the aggregate by the BVF Entities. BVF Inc., as the general partner of Partners, may be deemed to beneficially own the 1,418,332 shares beneficially owned by Partners. Mark Lampert, as a director and officer of BVF Inc., may be deemed to beneficially own the 1,418,332 shares beneficially owned by BVF Inc. The mailing address of each of the BVF Entities is 44 Montgomery Street, 40th Floor, San Francisco, California 94104. |
|
(4) |
Consists of (i) 447,755 shares of common stock issuable upon conversion of 447,755 shares of Series C preferred stock held of record by Adams Street 2018 Direct Venture/Growth Fund LP, or AS 2018, (ii) 251,530 shares of common stock issuable upon conversion of 251,530 shares of Series C preferred stock held of record by Adams Street 2019 Direct Growth Equity Fund LP, or AS 2019, (iii) 173,470 shares of common stock issuable upon conversion of 173,470 shares of Series C preferred stock held of record by Adams Street 2020 Direct Growth Equity Fund LP, or AS 2020, and (iv) 451,022 shares of common stock issuable upon conversion of 451,022 shares of Series C preferred stock held of record by Adams Street Growth Equity Fund VII LP, or AS GEF VII, and together with AS 2018, AS 2019 and AS 2020, the Adams Street Entities. Adams Street Partners, LLC is the managing member of the general partner of the general partner of the Adam Street Entities, and may be deemed to beneficially own the shares held by the Adams Street Entities. Thomas S. Bremner, Jeffrey T. Diehl, Elisha P. Gould III, Robin P. Murray and Fred Wang, each of whom is a partner of Adams Street Partners, LLC (or a subsidiary thereof), may be deemed to have shared voting and investment power over the shares held of record by the Adams Street Entities, or the Shares. Adams Street Partners, LLC and Thomas S. Bremner, Jeffrey T. Diehl, Elisha P. Gould III, Robin P. Murray and Fred Wang disclaim beneficial ownership of the Shares except to the extent of their pecuniary interest therein. The mailing address of each of the Adams Street Entities is One North Wacker Drive, Suite 2700, Chicago, Illinois 60606. |
|
(5) |
Consists of (i) 2,525,000 shares of common stock held of record by Wesley H. Kaupinen 2019 Irrevocable Trust dated February 28, 2019 for the benefit of Wesley H. Kaupinen, (ii) 2,525,000 shares of common stock held of record by Christine L. Kaupinen 2019 Irrevocable Trust dated February 28, 2019 for the benefit of Wesley H. Kaupinen’s spouse, (iii) 138,755 shares of common stock issuable upon conversion of 138,755 shares of Series A-1 preferred stock, Series A-2 preferred stock, Series B preferred stock, Series C preferred stock and Series D preferred stock held of record by Wesley H. Kaupinen, and (iv) 81,097 shares of common stock subject to options that are exercisable within 60 days of October 28, 2024. |
|
(6) |
Consists of 327,358 shares of common stock subject to options that are exercisable within 60 days of October 28, 2024. |
|
(7) |
Consists of 158,920 shares of common stock subject to options that are exercisable within 60 days of October 28, 2024. |
|
(8) |
Consists of (i) 66,980 shares of common stock, (ii) and 503,904 shares of common stock issuable upon conversion of 503,904 shares of Series A-1 preferred stock, Series A-2 preferred stock, Series B preferred stock, Series C preferred stock and Series D preferred stock held of record by George M. Jenkins, and (ii) 29,445 shares of common stock subject to options that are exercisable within 60 days of October 28, 2024. |
|
(9) |
Consists of (i) 66,980 shares of common stock and 88,859 shares of common stock issuable upon conversion of 88,859 shares of Series A-1 preferred stock held of record by Todd C. Davis, (ii) 89,146 shares of common stock issuable upon conversion of 89,146 shares of Series A-2 preferred stock, Series B preferred stock and Series C preferred stock held by RoyaltyRx Capital LLC, and (iii) 21,945 shares of common stock held by Todd C. Davis subject to options exercisable within 60 days of October 28, 2024. |
PRINCIPAL STOCKHOLDERS OF THE COMBINED COMPANY
The following table sets forth certain information regarding beneficial ownership of the combined company common stock immediately after consummation of the Merger, assuming the consummation of the Merger, including the PIPE Financing, occurred on October 28, 2024 for:
|
● |
each person or group of affiliated persons expected by Pieris and Palvella to become the beneficial owner of more than 5% of the combined company common stock; |
|
● |
each person expected to be a named executive officer of the combined company; |
|
● |
each person expected to be a director of the combined company; and |
|
● |
all of the combined company’s expected directors and executive officers as a group. |
Beneficial ownership in accordance with the rules of the SEC, and thus it represents sole or shared voting or investment power with respect to Palvella’s securities. Unless otherwise indicated below, to Palvella’s knowledge, the persons and entities named in the table have sole voting and sole investment power with respect to all shares that they beneficially owned, subject to community property laws where applicable. The information does not necessarily indicate beneficial ownership for any other purpose, including for purposes of Sections 13(d) and 13(g) of the Exchange Act.
The percentage of beneficial ownership is calculated based on 13,929,867 shares of common stock expected to be outstanding upon consummation of the Merger and the closing of the PIPE Financing. The number of shares beneficially owned includes shares of common stock that each person has the right to acquire within 60 days of October 28, 2024, including upon the exercise of stock options. These stock options shall be deemed to be outstanding for the purpose of computing the percentage of outstanding shares of the combined company common stock expected to be owned by such person but shall not be deemed to be outstanding for the purpose of computing the percentage of outstanding shares of the combined organization’s common stock expected to be owned by any other person.
Immediately after the Merger, but without giving effect to the PIPE Financing, Pieris securityholders as of immediately prior to the merger are expected to own approximately 18% of the outstanding shares of the combined company and former Palvella securityholders are expected to own approximately 82% of the outstanding shares of the combined company on a fully-diluted basis, calculated using the treasury stock method, subject to certain assumptions, including, but not limited to, a valuation for Pieris equal to $21 million, provided, that (a) if Pieris’ net cash as of the closing is greater than $11 million, then Pieris’ valuation will be adjusted upwards on a dollar-for-dollar basis by the difference of: (i) Pieris’ net cash, minus (ii) $11 million, and (b) if Pieris’ net cash is less than $11 million, then Pieris’ valuation will be adjusted downwards on a dollar-for-dollar basis by the difference of: (i) $11 million, minus (ii) Pieris’ net cash, as further described in the Merger Agreement.
The table below assumes that, based on Pieris’ and Palvella’s capitalization as of October 28, 2024, the exchange ratio is estimated to be equal to approximately 0.315478222 shares of Pieris common stock for each share of Palvella capital stock. The final exchange ratio is subject to adjustment prior to closing of the Merger based upon Pieris’ net cash as of the date immediately preceding the closing of the Merger.
Unless otherwise indicated, the address of each beneficial owner listed in the table below is c/o Palvella Therapeutics, Inc., 125 Strafford Avenue, Suite 360, Wayne, Pennsylvania 19087.
|
Name of Beneficial Owner |
Number of Shares Beneficially Owned |
Percentage of Shares Owned (%) |
||||||
|
5% stockholders: |
||||||||
|
Samsara BioCapital(1) |
692,680 | 6.23 | % | |||||
|
Averill Master Fund, Ltd (2) |
728,337 | 6.55 | % | |||||
|
Biotechnology Value Fund, L.P. and affiliates (3) |
1,157,075 | 9.99 | % | |||||
|
Integrated Finance Group(4) |
728,337 | 6.55 | % | |||||
|
Directors and Named Executive Officers: |
||||||||
|
Wesley H. Kaupinen(5) |
1,663,979 | 14.92 | % | |||||
|
Kathleen Goin(6) |
103,274 | * | ||||||
|
Jeffrey Martini, Ph.D (7) |
50,136 | * | ||||||
|
George M. Jenkins(8) |
200,316 | 1.80 | % | |||||
|
Todd C. Davis(9) |
120,627 | 1.08 | % | |||||
|
Tadd S. Wessel |
— | * | ||||||
|
Christopher Kiritsy(10) |
3,562 | * | ||||||
|
All current executive officers and directors as a group (9 individuals) |
2,190,737 | 19.27 | % | |||||
* Less than 1%.
|
(1) |
Consists of 692,680 shares common stock held of record by Samsara BioCapital, L.P., or Samsara LP. Samsara BioCapital GP, LLC, or Samsara GP, is the sole general partner of Samsara LP and may be deemed to beneficially own the shares held of record by Samsara LP. Srinivas Akkaraju is a managing member of Samsara GP and may be deemed to beneficially own the shares held of record by Samsara LP. The mailing address of Samsara LP is 628 Middlefield Road, Palo Alto, California 94301. |
|
(2) |
Consists of 728,337 shares common stock held of record by Averill Master Fund, Ltd., or Averill. Aaron Cowen is the manager of Averill and may be deemed to beneficially own the shares of record held by Averill. The mailing address of Averill is c/o Maples Corporate Services Limited, P.O. Box 309, Ugland House, Grand Cayman KY1-1104, Cayman Islands. |
|
(3) |
Biotechnology Value Fund, L.P., or BVF, and its related entities beneficially own 1,157,075 shares of common stock consisting of (i) 376,465 shares of common stock held of record by BVF, (ii) 285,619 shares of common stock held of record by Biotechnology Value Fund II, L.P., or BVF II, (iii) 39,536 shares of common held of record by Biotechnology Value Trading Fund OS, L.P., or Trading Fund OS (iv) 888 shares of common held of record by Investment 10 LLC, or Investment 10, (v) 2,566 shares of common held of record by MSI BVF SPV, LLV, or MSI, (vi) 236,817 pre-funded warrants which are exercisable for shares of common stock to be held of record by BVF, (vii) 188,014 pre-funded warrants which are exercisable for shares of common stock to be held of record by BVF II, (viii) 18,883 pre-funded warrants which are exercisable for shares of common stock to be held of record by Trading Fund OS, and (ix) 8,287 pre-funded warrants which are exercisable for shares of common stock to be held of record by MSI, and excluding (i) 335,621 pre-funded warrants which are exercisable for shares of common stock to be held of record by BVF, (ii) 266,458 pre-funded warrants which are exercisable for shares of common stock to be held of record by BVF II, (iii) 26,761 pre-funded warrants which are exercisable for shares of common stock to be held of record by Trading Fund OS, (iv) 11,745 pre-funded warrants which are exercisable for shares of common stock to be held of record by MSI (v) warrants exercisable for 3,522,000 shares of common stock, (vi) 85 shares of Series A Convertible Preferred Stock held of record by BVF and its related entities, which is convertible into 1,133 shares of common stock, (vii) 4,026 shares of Series B Convertible Preferred Stock held of record by BVF and its related entities, which is convertible into 53,706 shares of common stock, (viii) 3,506 shares of Series C Convertible Preferred Stock held of record by BVF and its related entities, which is convertible into 46,770 shares of common stock, (ix) 3,000 shares of Series D Convertible Preferred Stock held of record by BVF and its related entities, which is convertible into 40,020 shares of common stock, and (x) 5,000 shares of Series E Convertible Preferred Stock held of record by BVF and its related entities, which is convertible into 66,700 shares of common stock,. The pre-funded warrants and warrants may not be exercised if, after such exercise, BVF and its affiliates would beneficially own more than 9.99% of the number of shares of common stock then issued and outstanding. As a result of the limitation in the previous sentence, for purposes of the table above, a portion of the shares of common stock issuable upon the exercise of the pre-funded warrants are included and no shares of common stock are included from the warrants. BVF I GP LLC, or BVF GP, as the general partner of BVF, may be deemed to beneficially own the 613,281 shares held of record by BVF. BVF II GP LLC, or BVF II GP, as the general partner of BVF II, may be deemed to beneficially own the 473,633 shares held of record by BVF II. BVF Partners OS Ltd., Partner OS, as the general partner of Trading Fund OS, may be deemed to beneficially own the 58,419 shares held of record by Trading Fund OS. BVF GP Holdings LLC, or BVF GPH, as the sole member of each of BVF GP and BVF II GP, may be deemed to beneficially own the 1,086,915 shares held of record in the aggregate by BVF and BVF II. BVF Partners L.P., or Partners, as the investment manager of BVF and its related entities, and the sole member of Partners OS, may be deemed to beneficially own the 1,157,075 shares held of record in the aggregate by the BVF and its related entities. BVF Inc., as the general partner of Partners, may be deemed to beneficially own the 1,157,075 shares beneficially owned by Partners. Mark Lampert, as a director and officer of BVF Inc., may be deemed to beneficially own the 1,157,075 shares beneficially owned by BVF Inc. The mailing address of each of the BVF is 44 Montgomery Street, 40th Floor, San Francisco, California 94104. |
|
(4) |
Consists of 728,337shares common stock held of record by Integrated Finance Group, or IFG. Mario Patone and William Oldham are the owners of IFG and may each be deemed to beneficially own the shares of record held by IFG. The mailing address of IFG is 1055 Westlake Dr., Ste 200, Berwyn, PA 19312. |
|
(5) |
Consists of (i) 796,582 shares of common stock held of record by Wesley H. Kaupinen 2019 Irrevocable Trust dated February 28, 2019 for the benefit of Wesley H. Kaupinen, (ii) 796,582 shares of common stock held of record by Christine L. Kaupinen 2019 Irrevocable Trust dated February 28, 2019 for the benefit of Wesley H. Kaupinen’s spouse, (iii) 45,231 held of record by Wesley H. Kaupinen, and (iv) 25,584 shares of common stock subject to options and restricted stock units that are exercisable within 60 days of October 28, 2024. |
|
(6) |
Consists of 103,274 shares of common stock subject to options that are exercisable within 60 days of October 28, 2024. |
|
(7) |
Consists of 50,136 shares of common stock subject to options that are exercisable within 60 days of October 28, 2024. |
|
(8) |
Consists of (i) 191,026 shares of common stock, (ii) 9,289 shares of common stock subject to options and restricted stock units that are exercisable within 60 days of October 28, 2024. |
|
(9) |
Consists of (i) 28,124 shares of common stock held by RoyaltyRx Capital LLC, and (ii) 85,581 shares of common stock held by Todd C. Davis and (iii) 6,923 shares of common stock held by Todd C. Davis subject to options exercisable within 60 days of October 28, 2024. |
|
(10) |
Consists of (i) 250 shares of common stock, and (ii) 3,312 shares of common stock subject to options exercisable within 60 days of October 28, 2024. |
Brownstein Hyatt Farber Schreck, LLP, Las Vegas, Nevada, will pass upon the validity of the Pieris common stock offered by this proxy statement/prospectus.
Pieris
The consolidated financial statements of Pieris Pharmaceuticals, Inc. at December 31, 2023 and 2022, and for each of the years then ended, included in the Proxy Statement of Pieris Pharmaceuticals, Inc., which is referred to and made a part of this Prospectus and Registration Statement have been audited by Ernst & Young LLP, independent registered public accounting firm, as set forth in their report thereon appearing elsewhere herein, and are included in reliance upon such report given on the authority of such firm as experts in accounting and auditing.
Palvella
The financial statements of Palvella Therapeutics, Inc. at December 31, 2023 and 2022, and for each of the years then ended, included in the Proxy Statement of Pieris Pharmaceuticals, Inc., which is referred to and made a part of this Prospectus and Registration Statement have been audited by Ernst & Young LLP, independent registered public accounting firm, as set forth in their report thereon (which contains an explanatory paragraph describing conditions that raise substantial doubt about the Palvella Therapeutics, Inc.'s ability to continue as a going concern as described in Note 1 to the financial statements) appearing elsewhere herein, and are included in reliance upon such report given on the authority of such firm as experts in accounting and auditing.
WHERE YOU CAN FIND MORE INFORMATION
Pieris is subject to the informational requirements of the Exchange Act and in accordance therewith, files annual, quarterly and current reports, proxy statements and other information with the SEC electronically, and the SEC maintains a website that contains Pieris’ filings as well as reports, proxy and information statements, and other information issuers file electronically with the SEC at www.sec.gov.
Pieris also makes available free of charge on or through its website at www.pieris.com its Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and amendments to those reports filed or furnished pursuant to Section 13(a) or 15(d) of the Exchange Act as soon as reasonably practicable after Pieris electronically files such material with or otherwise furnishes it to the SEC. The website addresses for the SEC and Pieris are inactive textual references and information on those websites is not part of this proxy statement/prospectus.
Pieris has filed with the SEC a Registration Statement on Form S-4, of which this proxy statement/prospectus is a part, under the Securities Act to register the shares of Pieris common stock to be issued to Palvella stockholders in the Merger. This proxy statement/prospectus is a part of that Registration Statement on Form S-4 and constitutes a prospectus of Pieris, as well as a proxy statement of Pieris for the Pieris special meeting, and it will also serve as an information statement for the stockholders of Palvella. The Registration Statement on Form S-4, including the attached annexes, exhibits and schedules, contains additional relevant information about Pieris and Pieris common stock. This proxy statement/prospectus does not contain all of the information set forth in the Registration Statement on Form S-4 because certain parts of the Registration Statement on Form S-4 are omitted in accordance with the rules and regulations of the SEC.
The SEC allows Pieris to “incorporate by reference” information into this proxy statement/prospectus. This means that Pieris can disclose important information to you by referring you to another document filed separately with the SEC. The information incorporated by reference is considered to be a part of this proxy statement/prospectus, and later information that Pieris files with the SEC will automatically update and supersede the information included in this proxy statement/prospectus. This document incorporates by reference the documents that are listed below that Pieris has previously filed with the SEC, except to the extent that any information contained in such filings is deemed “furnished” in connection with SEC rules.
|
● |
Pieris’ Annual Report on Form 10-K for the fiscal year ended December 31, 2023, filed with the SEC on March 29, 2024, as amended on April 29, 2024. |
|
● |
Pieris’ Quarterly Reports on Form 10-Q for the quarters ended March 31, 2024 filed with the SEC on May 15, 2024 and June 30, 2024 filed with the SEC on August 14, 2024. |
|
● |
Pieris’ Current Reports on Form 8-K, filed with the SEC on March 27, 2024, April 18, 2024, May 9, 2024, July 3, 2024, July 24, 2024, August 8, 2024, and October 3, 2024. |
Notwithstanding the statements in the preceding paragraphs, no document, report or exhibit (or portion of any of the foregoing) or any other information that Pieris has “furnished” to but not “filed” with the SEC pursuant to the Exchange Act shall be incorporated by reference in this proxy statement/prospectus.
In addition, Pieris incorporates by reference any documents that it may subsequently file with the SEC pursuant to Sections 13(a), 13(c), 14 or 15(d) of the Exchange Act after the date of this proxy statement/prospectus and prior to the date of the Pieris special meeting, other than the portions of such documents not deemed to be filed. Any statement contained in this proxy statement/prospectus or in a document incorporated or deemed to be incorporated by reference in this proxy statement/prospectus is deemed to be modified or superseded to the extent that a statement contained herein or in any subsequently filed document that also is, or is deemed to be, incorporated by reference herein modified or superseded such statement. Any statement so modified or superseded will not be deemed, except as so modified or superseded, to constitute a part of this proxy statement/prospectus. Pieris has supplied all the information contained in this proxy statement/prospectus relating to Pieris, and Palvella has supplied all information contained in this proxy statement/prospectus relating to Palvella.
If you would like to request documents from Pieris or Palvella, please send a request in writing or by telephone to either Pieris or Palvella at the following addresses:
|
Pieris Pharmaceuticals, Inc. |
Palvella Therapeutics, Inc. |
|
|
225 Franklin Street, 26th Floor |
125 Strafford Avenue, Suite 360 |
|
|
Boston, Massachusetts 02110 (857) 246-8998 |
Wayne, Pennsylvania 19087 (484) 253-1461 |
If you are a Pieris stockholder and would like additional copies of this proxy statement/prospectus without charge you may (1) visit www.ProxyVote.com, (2) call 1-800-579-1639 or (3) send an email to sendmaterial@proxyvote.com.
If you are a Pieris stockholder and have questions about the Merger, including the procedures for voting your shares, you should contact Pieris’ proxy solicitor, Alliance Advisors, at the following address and telephone number:
Alliance Advisors LLC
200 Broadacres Drive, 3rd Floor
Bloomfield, New Jersey 07003
833-782-7190 (toll free)
PIRS@allianceadvisors.com (Email)
Pieris Pharmaceuticals, Inc.
Palvella Therapeutics, Inc.
PIERIS PHARMACEUTICALS, INC.
CONDENSED CONSOLIDATED BALANCE SHEETS
(unaudited, in thousands)
|
June 30, |
December 31, |
|||||||
|
2024 |
2023 |
|||||||
|
Assets |
||||||||
|
Current assets: |
||||||||
|
Cash and cash equivalents |
$ | 19,731 | $ | 17,396 | ||||
|
Short term investments |
— | 8,970 | ||||||
|
Accounts receivable |
1,485 | 572 | ||||||
|
Receivable from public grants |
3,049 | 3,141 | ||||||
|
Other receivables |
137 | 2,326 | ||||||
|
Assets held for sale, property and equipment |
— | 2,188 | ||||||
|
Prepaid expenses and other current assets |
649 | 4,087 | ||||||
|
Total current assets |
$ | 25,051 | $ | 38,680 | ||||
|
Liabilities and stockholders’ equity |
||||||||
|
Current liabilities: |
||||||||
|
Accounts payable |
$ | 969 | $ | 3,372 | ||||
|
Accrued expenses and other current liabilities |
5,377 | 8,550 | ||||||
|
Total current liabilities |
6,346 | 11,922 | ||||||
|
Stockholders’ equity: |
||||||||
|
Preferred stock |
— | — | ||||||
|
Common stock |
1 | 1 | ||||||
|
Additional paid-in capital |
342,586 | 341,693 | ||||||
|
Accumulated other comprehensive loss |
(436 | ) | 28 | |||||
|
Accumulated deficit |
(323,446 | ) | (314,964 | ) | ||||
|
Total stockholders’ equity |
18,705 | 26,758 | ||||||
|
Total liabilities and stockholders’ equity |
$ | 25,051 | $ | 38,680 | ||||
The accompanying notes are an integral part of these condensed consolidated financial statements.
CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS
(unaudited)
(in thousands, except per share data)
|
Three Months Ended June 30, |
Six Months Ended June 30, |
|||||||||||||||
|
2024 |
2023 |
2024 |
2023 |
|||||||||||||
|
Revenue |
||||||||||||||||
|
Customer revenue |
$ | — | $ | 20,086 | $ | 6 | $ | 22,096 | ||||||||
|
Collaboration revenue |
— | (31 | ) | 47 | (105 | ) | ||||||||||
|
Total revenue |
— | 20,055 | 53 | 21,991 | ||||||||||||
|
Operating expenses |
||||||||||||||||
|
Research and development |
751 | 14,328 | 1,969 | 27,752 | ||||||||||||
|
General and administrative |
3,426 | 3,664 | 7,564 | 7,687 | ||||||||||||
|
Total operating expenses |
4,177 | 17,992 | 9,533 | 35,439 | ||||||||||||
|
Loss from operations |
(4,177 | ) | 2,063 | (9,480 | ) | (13,448 | ) | |||||||||
|
Other income (expense) |
||||||||||||||||
|
Interest income |
201 | 490 | 441 | 847 | ||||||||||||
|
Grant income |
— | 1,584 | — | 3,612 | ||||||||||||
|
Other income (loss) |
386 | (161 | ) | 557 | (218 | ) | ||||||||||
|
Net income (loss) |
$ | (3,590 | ) | $ | 3,976 | $ | (8,482 | ) | $ | (9,207 | ) | |||||
|
Other comprehensive income loss: |
||||||||||||||||
|
Foreign currency translation |
(90 | ) | 287 | (463 | ) | 45 | ||||||||||
|
Unrealized gain (loss) on available-for-sale securities |
— | 2 | (1 | ) | 72 | |||||||||||
|
Comprehensive loss |
$ | (3,680 | ) | $ | 4,265 | $ | (8,946 | ) | $ | (9,090 | ) | |||||
|
Net income (loss) per share |
||||||||||||||||
|
Basic |
$ | (2.76 | ) | $ | 3.63 | $ | (6.69 | ) | $ | (9.08 | ) | |||||
|
Diluted |
$ | (2.76 | ) | $ | 3.62 | $ | (6.69 | ) | $ | (9.08 | ) | |||||
|
Weighted average number of common shares outstanding |
||||||||||||||||
|
Basic |
1,299 | 1,095 | 1,268 | 1,014 | ||||||||||||
|
Diluted |
1,299 | 1,098 | 1,268 | 1,014 | ||||||||||||
The accompanying notes are an integral part of these condensed consolidated financial statements.
CONDENSED CONSOLIDATED STATEMENTS OF CHANGES IN STOCKHOLDERS’ EQUITY
(unaudited, in thousands)
For the Three Months Ended June 30, 2023 and 2024
|
Accumulated |
||||||||||||||||||||||||||||||||
|
Preferred shares |
Common shares |
Additional |
other |
Total |
||||||||||||||||||||||||||||
|
No. of |
Share |
No. of |
Share |
paid-in |
comprehensive |
Accumulated |
Stockholders’ |
|||||||||||||||||||||||||
|
shares |
capital |
shares |
capital |
capital |
income (loss) |
deficit |
equity |
|||||||||||||||||||||||||
|
Balance as of March 31, 2023 |
16 | $ | — | 931 | $ | 1 | $ | 319,487 | $ | (426 | ) | $ | (303,604 | ) | $ | 15,458 | ||||||||||||||||
|
Net loss |
— | — | — | — | — | — | 3,976 | 3,976 | ||||||||||||||||||||||||
|
Stock based compensation expense |
— | — | — | — | 1,048 | — | — | 1,048 | ||||||||||||||||||||||||
|
Foreign currency translation adjustment |
— | — | — | — | — | 287 | — | 287 | ||||||||||||||||||||||||
|
Unrealized gain on investments |
— | — | — | — | — | 2 | — | 2 | ||||||||||||||||||||||||
|
Issuance of common stock resulting from purchase of employee stock purchase plan shares |
— | — | 1 | — | 52 | — | — | 52 | ||||||||||||||||||||||||
|
Issuance of common stock pursuant to ATM offering program, net of $0.7 million in offering costs |
— | — | 303 | — | 19,675 | — | — | 19,675 | ||||||||||||||||||||||||
|
Balance at June 30, 2023 |
16 | $ | — | 1,236 | $ | 1 | $ | 340,262 | $ | (137 | ) | $ | (299,628 | ) | $ | 40,498 | ||||||||||||||||
|
Balance as of March 31, 2024 |
16 | $ | — | 1,237 | $ | 1 | $ | 342,165 | $ | (346 | ) | $ | (319,856 | ) | $ | 21,964 | ||||||||||||||||
|
Net loss |
— | — | — | — | — | — | (3,590 | ) | (3,590 | ) | ||||||||||||||||||||||
|
Stock based compensation expense |
— | — | — | — | 421 | — | — | 421 | ||||||||||||||||||||||||
|
Foreign currency translation adjustment |
— | — | — | — | — | (90 | ) | — | (90 | ) | ||||||||||||||||||||||
|
Round-Up shares from the 1-for-80 reverse split effective April 23, 2024 |
— | — | 83 | — | — | — | — | — | ||||||||||||||||||||||||
|
Balance at June 30, 2024 |
16 | $ | — | 1,320 | $ | 1 | $ | 342,586 | $ | (436 | ) | $ | (323,446 | ) | $ | 18,705 | ||||||||||||||||
PIERIS PHARMACEUTICALS, INC.
CONDENSED CONSOLIDATED STATEMENTS OF CHANGES IN STOCKHOLDERS’ EQUITY
(unaudited, in thousands)
For the Six Months Ended June 30, 2023 and 2024
|
Accumulated |
||||||||||||||||||||||||||||||||
|
Preferred shares |
Common shares |
Additional |
other |
Total |
||||||||||||||||||||||||||||
|
No. of |
Share |
No. of |
Share |
paid-in |
comprehensive |
Accumulated |
Stockholders’ |
|||||||||||||||||||||||||
|
shares |
capital |
shares |
capital |
capital |
income (loss) |
deficit |
equity |
|||||||||||||||||||||||||
|
Balance at December 31, 2022 |
16 | $ | — | 931 | $ | 1 | $ | 318,603 | $ | (254 | ) | $ | (290,421 | ) | $ | 27,929 | ||||||||||||||||
|
Net loss |
— | — | — | — | — | — | (9,207 | ) | (9,207 | ) | ||||||||||||||||||||||
|
Stock based compensation expense |
— | — | — | — | 1,932 | — | — | 1,932 | ||||||||||||||||||||||||
|
Foreign currency translation adjustment |
— | — | — | — | — | 45 | — | 45 | ||||||||||||||||||||||||
|
Unrealized loss on investments |
— | — | — | — | — | 72 | — | 72 | ||||||||||||||||||||||||
|
Issuance of common stock resulting from purchase of employee stock purchase plan shares |
— | — | 1 | — | 52 | — | — | 52 | ||||||||||||||||||||||||
|
Issuance of common stock pursuant to ATM offering program, net of $0.3 million in offering costs |
— | — | 303 | — | 19,675 | — | — | 19,675 | ||||||||||||||||||||||||
|
Balance at June 30, 2023 |
16 | $ | — | 1,236 | $ | 1 | $ | 340,262 | $ | (137 | ) | $ | (299,628 | ) | $ | 40,498 | ||||||||||||||||
|
Balance at December 31, 2023 |
16 | $ | — | 1,237 | $ | 1 | $ | 341,693 | $ | 28 | $ | (314,964 | ) | $ | 26,758 | |||||||||||||||||
|
Net loss |
— | — | — | — | — | — | (8,482 | ) | (8,482 | ) | ||||||||||||||||||||||
|
Stock based compensation expense |
— | — | — | — | 893 | — | — | 893 | ||||||||||||||||||||||||
|
Foreign currency translation adjustment |
— | — | — | — | — | (463 | ) | — | (463 | ) | ||||||||||||||||||||||
|
Unrealized gain on investments |
— | — | — | — | — | (1 | ) | — | (1 | ) | ||||||||||||||||||||||
|
Round-Up shares from the 1-for-80 reverse split effective April 23, 2024 |
— | — | 83 | — | — | — | — | — | ||||||||||||||||||||||||
|
Balance at June 30, 2024 |
16 | $ | — | 1,320 | $ | 1 | $ | 342,586 | $ | (436 | ) | $ | (323,446 | ) | $ | 18,705 | ||||||||||||||||
CONDENSED CONSOLIDATED STATEMENTS OF CASH FLOWS
(unaudited, in thousands)
|
Six Months Ended June 30, |
||||||||
|
2024 |
2023 |
|||||||
|
Operating activities: |
||||||||
|
Net loss |
$ | (8,482 | ) | $ | (9,207 | ) | ||
|
Adjustments to reconcile net loss to net cash used in operating activities: |
||||||||
|
Depreciation and amortization (accretion) |
(30 | ) | 1,363 | |||||
|
Right-of-use asset accretion |
— | (67 | ) | |||||
|
Stock-based compensation |
893 | 1,932 | ||||||
|
Gain on sale of fixed assets |
(219 | ) | — | |||||
|
Prepaid rent |
1,112 | — | ||||||
|
Realized investment losses |
— | (53 | ) | |||||
|
Other non-cash transactions |
21 | (110 | ) | |||||
|
Changes in operating assets and liabilities |
(1,771 | ) | (18,285 | ) | ||||
|
Net cash used in operating activities |
(8,476 | ) | (24,427 | ) | ||||
|
Investing activities: |
||||||||
|
Purchases of property and equipment |
— | (115 | ) | |||||
|
Proceeds from maturity of investments |
9,000 | 18,895 | ||||||
|
Proceeds on sale of fixed assets |
2,176 | — | ||||||
|
Purchases of investments |
— | (8,243 | ) | |||||
|
Net cash provided by investing activities |
11,176 | 10,537 | ||||||
|
Financing activities: |
||||||||
|
Proceeds from employee stock purchase plan |
— | 52 | ||||||
|
Proceeds from issuance of common stock resulting from ATM sales, net of $0.7 million in transaction costs, respectively |
— | 19,729 | ||||||
|
Net cash provided by financing activities |
— | 19,781 | ||||||
|
Effect of exchange rate change on cash and cash equivalents |
(365 | ) | 412 | |||||
|
Net increase in cash and cash equivalents |
2,335 | 6,303 | ||||||
|
Cash and cash equivalents at beginning of period |
17,396 | 38,635 | ||||||
|
Cash and cash equivalents at end of period |
$ | 19,731 | $ | 44,938 | ||||
|
Supplemental cash flow disclosures: |
||||||||
|
Net unrealized gain (loss) on investments |
$ | (1 | ) | $ | 72 | |||
|
Property and equipment included in accounts payable |
$ | — | $ | 74 | ||||
The accompanying notes are an integral part of these condensed consolidated financial statements.
NOTES TO THE CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
(unaudited)
1. Corporate Information
Pieris Pharmaceuticals, Inc., or the Company or Pieris, was founded in May 2013, and acquired 100% interest in Pieris Pharmaceuticals GmbH (formerly Pieris AG, a German company which was founded in 2001) in December 2014. Pieris Pharmaceuticals, Inc. and its wholly-owned subsidiaries, hereinafter collectively Pieris, or the Company, is a biopharmaceutical company that, prior to July of 2023, discovered and developed Anticalin-based drugs to target validated disease pathways in unique and transformative ways. Pieris’ clinical pipeline consists of immuno-oncology, or IO, programs partnered with several major multi-national pharmaceutical companies. Pieris' corporate headquarters is located in Boston, Massachusetts. Pieris also maintains office space in Hallbergmoos, Germany. The Company’s core Anticalin technology and platform was developed in Germany.
On July 18, 2023, the Company announced its intention to explore engaging in one or more strategic transactions, including mergers, reverse mergers, acquisitions, other business combinations or sales of assets, or other strategic transactions. This decision was related to events that impacted the Company’s inhaled respiratory franchise in connection with AstraZeneca's discontinuation of enrollment of the Phase 2a study for elarekibep, an inhaled IL-4Rα antagonist Anticalin protein to treat uncontrolled asthma. As part of this initiative, the Company engaged Stifel, Nicolaus & Company, Incorporated to serve as its advisor in its review of strategic transactions.
Also on July 18, 2023, the Company’s Board of Directors approved a reduction in the Company’s workforce by approximately 70%. Since July of 2023, and through June 30, 2024, the Company took additional steps to reduce its operating footprint including terminating its remaining lease obligations in Germany and winding down its proprietary inhaled respiratory programs. The Company also has opted out and terminated programs where possible to reduce operating costs. Further reductions in the workforce have occurred based upon these actions. As a result, the Company has incurred approximately $7.5 million of severance costs and other related termination benefits in 2023 as the service period to earn such benefits is considered complete. The Company expects termination benefits to be paid through the end of 2024.
On March 27, 2024, the Company announced the implementation of a new strategy along with relevant cost-saving measures that are expected to extend its cash runway into at least 2027, while maximizing its ability to capture the potential milestones from its partnered 4-1BB bispecific Mabcalin™ protein IO assets. The Company may be entitled to aggregate milestones of up to approximately $15.0 million upon first patient dosed in the Phase 2 trials for SGN-BB228 and BOS-342, which are currently in Phase 1 clinical development, and up to approximately $40.0 million upon first patient dosed in pivotal clinical trials for SGN-BB228 and BOS-342.
On July 23, 2024, the Company and its wholly-owned subsidiary, Polo Merger Sub, Inc. ("Merger Sub") entered into an Agreement and Plan of Merger (the “Merger Agreement”) with Palvella Therapeutics, Inc. ("Palvella"), discussed further in Note 11, whereby Merger Sub will merge with and into Palvella, with Palvella continuing as a wholly-owned subsidiary of the Company and the surviving corporation of the merger (the "Merger"). Subject to the terms and conditions of the Merger Agreement, at the effective time of the Merger, (i) each then-outstanding share of Palvella capital stock will be converted into the right to receive a number of shares of the Company's common stock equal to the Exchange Ratio as defined in the Merger Agreement; and (ii) each then-outstanding Palvella stock option to purchase Palvella common stock will be assumed by the Company. Each of the Company and Palvella has agreed to customary representations, warranties and covenants in the Merger Agreement, including, among others, covenants relating to (1) using reasonable best efforts to obtain the requisite approvals of their respective stockholders, (2) non-solicitation of alternative acquisition proposals, (3) the conduct of their respective businesses during the period between the date of signing the Merger Agreement and the closing of the Merger, (4) the Company using its commercially reasonable efforts to maintain the existing listing of the Company's common stock on Nasdaq and the Company causing the shares of the Company's common stock to be issued in connection with the Merger to be approved for listing on Nasdaq prior to the closing of the Merger and (5) the Company filing with the U.S. Securities and Exchange Commission and causing to become effective a registration statement to register the shares of the Company's common stock to be issued in connection with the Merger.
On August 7, 2024, the Company entered into a Subscription and Investment Representation Agreement with James Geraghty, Chairman of the Company’s Board of Directors (the "Purchaser"), pursuant to which the Company agreed to issue and sell one (1) share of the Company’s Series F Preferred Stock, par value $0.001 per share, to the Purchaser for $1.00 cash. The sale closed on August 7, 2024. The Series F Preferred Stock have no voting rights other than the right to vote on a proposed amendment to the Company’s amended and restated articles of incorporation to effect an increase in the number of authorized shares of the Company’s common stock (the "Authorized Share Increase Proposal"). Each share of Series F Preferred Stock outstanding on the record date entitles the holder thereof to 25,000,000 votes on the Authorized Share Increase Proposal, and all shares of Series F Preferred Stock held by such holder must and will be voted, without further action by such holder, in the same proportion as the aggregate shares of Pieris common stock (excluding any shares of Pieris common stock that are not voted) that are voted on the Authorized Share Increase Proposal.
As of June 30, 2024, cash and cash equivalents were $19.7 million. For the three months ended June 30, 2024 and 2023, the Company had a net loss of $3.6 million and net income of $4.0 million, respectively. For the six months ended June 30, 2024 and 2023, the Company had net losses of $8.5 million and $9.2 million, respectively. The Company has incurred net losses since inception and had an accumulated deficit of $323.4 million as of June 30, 2024. Net losses and negative cash flows from operations have had, and will continue to have, an adverse effect on the Company’s stockholders’ equity and working capital. The Company expects to continue to incur operating losses for the foreseeable future.
The Company has historically devoted substantially all of its financial resources and efforts to research and development and general and administrative expenses to support the discovery and development of Anticalin-based drugs. Going forward, as part of the Company's previous decision to implement measures to maximize its ability to capture potential milestones from its partnered programs with Pfizer and Boston Pharmaceuticals (all as defined in Note 3 below) and the Company's plan to consummate the potential Merger, subject to stockholder approval, the Company has discontinued all research and development efforts and continues to reduce discretionary expenditures and other fixed or variable personnel costs. The Company believes that its currently available funds will be sufficient to fund its operations through at least the next twelve months from the issuance of this proxy statement/prospectus. The Company's belief with respect to its ability to fund operations is based on estimates that are subject to risks and uncertainties. If actual results are different from management’s estimates, the Company may need to seek additional funding.
2. Summary of Significant Accounting Policies
The Company’s significant accounting policies are described in Note 2—Summary of Significant Accounting Policies, in the Company’s Annual Report on Form 10-K for the fiscal year ended December 31, 2023. There have been no material additions to the significant accounting policies for the six months ended June 30, 2024.
Unaudited Interim Financial Information
The accompanying unaudited condensed consolidated financial statements included herein have been prepared by the Company in accordance with accounting principles generally accepted in the United States, or U.S. GAAP, for interim financial information and pursuant to the rules and regulations of the SEC. Accordingly, certain information and footnote disclosures normally included in financial statements prepared in accordance with U.S. GAAP have been condensed or omitted pursuant to such rules and regulations. In the opinion of management, all adjustments, consisting of normal recurring adjustments, and disclosures considered necessary for a fair presentation of interim period results have been included. Interim results for the three and six months ended June 30, 2024 are not necessarily indicative of results that may be expected for the year ending December 31, 2024. For further information, refer to the financial statements and footnotes thereto included in the Company’s Annual Report on Form 10-K for the fiscal year ended December 31, 2023, which was filed with the SEC on March 29, 2024.
Basis of Presentation and Use of Estimates
The accompanying unaudited condensed consolidated financial statements of Pieris Pharmaceuticals, Inc. and its wholly-owned subsidiaries were prepared in accordance with U.S. GAAP. The unaudited condensed consolidated financial statements include the accounts of all subsidiaries. All intercompany balances and transactions have been eliminated.
Effective at 5:00 p.m. Eastern Time on April 22, 2024, the Company effected a 1-for-80 reverse stock split of its common stock, or the Reverse Split, with any fractional shares resulting from the Reverse Split rounded up to the next whole share of common stock. All references to shares of common stock outstanding, average number of shares outstanding and per share amounts in this proxy statement/prospectus have been restated to reflect the Reverse Split on a retroactive basis.
The preparation of the financial statements in accordance with U.S. GAAP requires management to make estimates, judgments and assumptions that affect the reported amounts of assets, liabilities, revenues, expenses and the related disclosures at the date of the financial statements and during the reporting period. Significant estimates are used for, but are not limited to, revenue recognition; deferred tax assets, deferred tax liabilities and valuation allowances; beneficial conversion features; fair value of stock options, preferred stock, and warrants; fair value of assets held for sale; and prepaid and accrued clinical trial expenses. Management evaluates its estimates on an ongoing basis. Actual results and outcomes could differ materially from management’s estimates, judgments and assumptions.
Cash, Cash Equivalents and Investments
The Company determines the appropriate classification of its investments at the time of purchase. All liquid investments with original maturities of 90 days or less from the purchase date and for which there is an active market are considered to be cash equivalents. The Company’s investments are comprised of money market, asset backed securities, government treasuries and corporate bonds that are classified as available-for-sale in accordance with Financial Accounting Standards Board, or FASB, Accounting Standards Codification, or ASC, Topic 320, Investments Debt and Equity Securities. The Company classifies investments available to fund current operations as current assets on its balance sheets.
Available-for-sale investments are recorded at fair value, with unrealized gains or losses included in accumulated other comprehensive loss on the Company’s balance sheets. Realized gains and losses are determined using the specific identification method and are included as a component of other income.
The Company reviews investments for other-than-temporary impairment whenever the fair value of an investment is less than the amortized cost and evidence indicates that an investment’s carrying amount is not recoverable within a reasonable period of time. To determine whether an impairment is other-than temporary, the Company considers its intent to sell or whether it is more likely than not that the Company will be required to sell the investment before recovery of the investment’s amortized cost basis. Evidence considered in this assessment includes reasons for the impairment, the severity and the duration of the impairment and changes in value subsequent to period end.
Concentration of Credit Risk and Off-Balance Sheet Risk
The Company has no financial instruments with off-balance sheet risk such as foreign exchange contracts, option contracts or other foreign hedging arrangements. Financial instruments that subject Pieris to concentrations of credit risk include cash and cash equivalents, investments, and accounts receivable. The Company’s cash, cash equivalents, and investments are held in accounts with financial institutions that management believes are creditworthy. The Company’s investment policy includes guidelines on the quality of the institutions and financial instruments and defines allowable investments that the Company believes minimize the exposure to concentration of credit risk. The Company has not experienced any credit losses in such accounts and does not believe it is exposed to any significant credit risk on these funds. Accounts receivable primarily consist of amounts due under strategic partnership and other license agreements with major multi-national pharmaceutical companies for which the Company does not obtain collateral.
Fair Value Measurement
The Company is required to disclose information on all assets and liabilities reported at fair value that enables an assessment of the inputs used in determining the reported fair values. FASB ASC Topic 820, Fair Value Measurement and Disclosures, established a hierarchy of inputs used in measuring fair value that maximizes the use of observable inputs and minimizes the use of unobservable inputs by requiring that the observable inputs be used when available. Observable inputs are inputs that market participants would use in pricing the financial instrument based on market data obtained from sources independent of the Company. Unobservable inputs are inputs that reflect the Company’s assumptions about the inputs that market participants would use in pricing the financial instrument and are developed based on the best information available in the circumstances. The fair value hierarchy applies only to the valuation inputs used in determining the reported or disclosed fair value of the financial instruments and is not a measure of the investment credit quality. Fair value measurements are classified and disclosed in one of the following three categories:
|
• |
Level 1 inputs are quoted prices in active markets for identical assets or liabilities that the reporting entity has the ability to access at the measurement date. |
|
• |
Level 2 utilizes quoted market prices in markets that are not active, broker or dealer quotations or alternative pricing sources with reasonable levels of price transparency. |
|
• |
Level 3 inputs are unobservable inputs for the asset or liability in which there is little, if any, market activity for the asset or liability at the measurement date. |
To the extent that valuation is based on models or inputs that are less observable or unobservable in the market, the determination of fair value requires more judgment. Accordingly, the degree of judgment exercised by the Company in determining fair value is greatest for instruments categorized in Level 3. A financial instrument’s level within the fair value hierarchy is based on the lowest level of any input that is significant to the fair value measurement.
Financial instruments measured at fair value on a recurring basis include cash equivalents and investments (see Note 5).
An entity may elect to measure many financial instruments and certain other items at fair value at specified election dates. Subsequent unrealized gains and losses on items for which the fair value option has been elected will be reported in net loss. The Company did not elect to measure any additional financial instruments or other items at fair value.
Property and Equipment
Property and equipment are recorded at acquisition cost, less accumulated depreciation and impairment. Depreciation on property and equipment is calculated using the straight-line method over the remaining estimated useful lives of the assets. Maintenance and repairs to these assets are charged to expenses as occurred. The estimated useful life of the different groups of property and equipment is as follows:
|
Asset Classification |
Estimated useful life (in years) |
||
|
Leasehold improvements |
shorter of useful life or remaining life of the lease |
||
|
Laboratory furniture and equipment |
8 - 14 | ||
|
Office furniture and equipment |
5 - 13 | ||
|
Computer and equipment |
3 - 7 |
If the criteria in ASC Topic 360 Property, Plant and Equipment are met, a long-lived asset is classified as held for sale. The long-lived asset is reported at the lower of its carrying value or fair value less cost to sell beginning in the period the held for sale criteria are met. The carrying amount of the asset will be adjusted each reporting period for subsequent changes in fair value less costs to sell. A loss is recognized for any subsequent write-down to fair value less cost to sell. A gain is recognized for any subsequent increase in fair value less cost to sell, but not in excess of the cumulative loss previously recognized. Once classified as held for sale, depreciation and amortization are no longer recorded for any long-lived assets included in the disposal group.
Impairment of Long-lived Assets
The Company reviews its long-lived assets to be held and used for impairment whenever events or changes in business circumstances indicate that the carrying amount of the assets may not be fully recoverable. The Company evaluates the realizability of its long-lived assets based on profitability and cash flow expectations for the related asset. Any write-downs are treated as permanent reductions in the carrying amount of the assets.
Revenue Recognition
Pieris has entered into several licensing agreements with collaboration partners for the development of Anticalin therapeutics against a variety of targets. The terms of these agreements provide for the transfer of multiple goods or services which may include: (i) licenses, or options to obtain licenses, to Pieris’ Anticalin technology and/or specific programs and (ii) research and development activities to be performed on behalf of or with a collaborative partner. Payments to Pieris under these agreements may include upfront fees (which include license and option fees), payments for research and development activities, payments based upon the achievement of certain milestones, and royalties on product sales. There are no performance, cancellation, termination or refund provisions in any of the arrangements that could result in material financial consequences to Pieris. As the Company's intellectual property assets are considered to be located in Germany, the Company records all consolidated revenue in its subsidiary, Pieris Pharmaceuticals GmbH.
Collaborative Arrangements
The Company considers the nature and contractual terms of an arrangement and assesses whether the arrangement involves a joint operating activity pursuant to which it is an active participant and exposed to significant risks and rewards with respect to the arrangement. If the Company is an active participant and exposed to the significant risks and rewards with respect to the arrangement, it accounts for these arrangements pursuant to ASC Topic 808, Collaborative Arrangements, or ASC 808, and applies a systematic and rational approach to recognize revenue. The Company classifies payments received as revenue and payments made as a reduction of revenue in the period in which they are earned. Revenue recognized under a collaborative arrangement involving a participant that is not a customer is presented as Collaboration Revenue in the condensed consolidated statement of operations.
Revenue from Contracts with Customers
In accordance with ASC Topic 606, revenue is recognized when a customer obtains control of promised goods or services. The amount of revenue recognized reflects the consideration to which the Company expects to be entitled in exchange for these goods and services. To achieve this core principle, the Company applies the following five steps: 1) identify the customer contract; 2) identify the contract’s performance obligations; 3) determine the transaction price; 4) allocate the transaction price to the performance obligations; and 5) recognize revenue when or as a performance obligation is satisfied.
The Company evaluates all promised goods and services within a customer contract and determines which of such goods and services are separate performance obligations. This evaluation includes an assessment of whether the good or service is capable of being distinct and whether the good or service is separable from other promises in the contract. In assessing whether promised goods or services are distinct, the Company considers factors such as the stage of development of the underlying intellectual property and the capabilities of the customer to develop the intellectual property on their own or whether the required expertise is readily available.
Licensing arrangements are analyzed to determine whether the promised goods or services, which often include licenses, research and development services and governance committee services, are distinct or whether they must be accounted for as part of a combined performance obligation. If the license is considered not to be distinct, the license would then be combined with other promised goods or services as a combined performance obligation. If the Company is involved in a governance committee, it assesses whether its involvement constitutes a separate performance obligation. When governance committee services are determined to be separate performance obligations, the Company determines the fair value to be allocated to this promised service.
Certain contracts contain optional and additional items, which are considered marketing offers and are accounted for as separate contracts with the customer if such option is elected by the customer, unless the option provides a material right which would not be provided without entering into the contract. An option that is considered a material right is accounted for as a separate performance obligation.
The transaction price is determined based on the consideration to which the Company will be entitled in exchange for transferring goods and services to the customer. A contract may contain variable consideration, including potential payments for both milestone and research and development services. For certain potential milestone payments, the Company estimates the amount of variable consideration by using the most likely amount method. In making this assessment, the Company evaluates factors such as the clinical, regulatory, commercial and other risks that must be overcome to achieve the milestone. Each reporting period the Company re-evaluates the probability of achievement of such variable consideration and any related constraints. The Company will include variable consideration, without constraint, in the transaction price to the extent it is probable that a significant reversal in the amount of cumulative revenue recognized will not occur when the uncertainty associated with the variable consideration is subsequently resolved. For potential research and development service payments, the Company estimates the amount of variable consideration by using the expected value method, including any approved budget updates arising from additional research or development services.
If the contract contains a single performance obligation, the entire transaction price is allocated to the single performance obligation. Contracts that contain multiple performance obligations require an allocation of the transaction price among the performance obligations on a relative standalone selling price basis unless a portion of the transaction price is variable and meets the criteria to be allocated entirely to a performance obligation or to a distinct good or service that forms part of a single performance obligation.
The Company allocates the transaction price based on the estimated standalone selling price of the underlying performance obligations or, in the case of certain variable consideration, to one or more performance obligations. The Company must develop assumptions that require judgment to determine the stand-alone selling price for each performance obligation identified in the contract. The Company utilizes key assumptions to determine the stand-alone selling price, which may include other comparable transactions, pricing considered in negotiating the transaction and the estimated costs to complete the respective performance obligation. Certain variable consideration is allocated specifically to one or more performance obligations in a contract when the terms of the variable consideration relate to the satisfaction of the performance obligation and the resulting amounts allocated to each performance obligation are consistent with the amount the Company would expect to receive for each performance obligation.
When a performance obligation is satisfied, revenue is recognized for the amount of the transaction price, excluding estimates of variable consideration that are constrained, that is allocated to that performance obligation on a relative standalone selling price basis. Significant management judgment is required in determining the level of effort required under an arrangement and the period over which the Company is expected to complete its performance obligations under an arrangement.
For performance obligations consisting of licenses and other promises, the Company utilizes judgment to assess the nature of the combined performance obligation to determine whether the combined performance obligation is satisfied over time or at a point in time and, if over time, the appropriate method of measuring progress for purposes of recognizing revenue from non-refundable, up-front fees. The Company evaluates the measure of progress each reporting period and, if necessary, adjusts the measure of performance and related revenue recognition. If the license to the Company’s intellectual property is determined to be distinct from the other performance obligations identified in the arrangement, the Company will recognize revenue from non-refundable, up-front fees allocated to the license when the license is transferred to the customer and the customer is able to use and benefit from the license.
Revenue recognized under an arrangement involving a participant that is a customer is presented as Customer Revenue.
Milestones and Royalties
The Company aggregates milestones into four categories: (i) research milestones, (ii) development milestones, (iii) commercial milestones, and (iv) sales milestones. Research milestones are typically achieved upon reaching certain success criteria as defined in each agreement related to developing an Anticalin protein against the specified target. Development milestones are typically reached when a compound reaches a defined phase of clinical research or passes such phase, or upon gaining regulatory approvals. Commercial milestones are typically achieved when an approved pharmaceutical product reaches the status for commercial sale, including regulatory approval. Sales milestones are certain defined levels of net sales by the licensee, such as when a product first achieves global sales or annual sales of a specified amount.
There is uncertainty that the events to obtain the research and development milestones will be achieved given the nature of clinical development and the stage of the Company’s technology. The Company has thus determined that all research and development milestones will be constrained until it is deemed probable that a significant revenue reversal will not occur. For revenues from research and development milestones, payments will be recognized consistent with the recognition pattern of the performance obligation to which they relate.
For arrangements that include sales-based royalties, including milestone payments based on the level of sales, and for which the license is deemed to be the predominant item to which the royalties relate, the Company recognizes revenue at the later of (i) when the related sales occur, or (ii) when the performance obligation to which some or all of the royalty has been allocated has been satisfied (or partially satisfied). Commercial milestones and sales royalties are determined by sales or usage-based thresholds and will be accounted for under the royalty recognition constraint as constrained variable consideration.
The Company calculates the maximum amount of potential milestones achievable under each collaboration agreement and discloses such potential future milestones for all current collaborations using such a maximum calculation.
Contract Balances
The Company recognizes a contract asset when the Company transfers goods or services to a customer before the customer pays consideration or before payment is due, excluding any amounts presented as a receivable (i.e., accounts receivable). A contract asset is an entity’s right to consideration in exchange for goods or services that the entity has transferred to a customer. The contract liabilities (i.e., deferred revenue) primarily relate to contracts where the Company has received payment but has not yet satisfied the related performance obligations.
In the event of an early termination of a collaboration agreement, any contract liabilities would be recognized in the period in which all Company obligations under the agreement have been fulfilled.
Costs to Obtain and Fulfill a Contract with a Customer
Certain costs to obtain customer contracts, including success-based fees paid to third-party service providers, and costs to fulfill customer contracts are capitalized in accordance with FASB ASC Topic 340, Other Assets and Deferred Costs, or ASC 340. These costs are amortized to expense on a systemic basis that is consistent with the transfer to the customer of the goods or services to which the asset relates. The Company will expense the amortization of costs to obtain customer contracts to general and administrative expense and costs to fulfill customer contracts to research and development expense.
Government Grants
The Company recognizes grants from governmental agencies when there is reasonable assurance that the Company will comply with the conditions attached to the grant arrangement and the grant will be received. The Company evaluates the conditions of each grant as of each reporting period to evaluate whether the Company has reached reasonable assurance of meeting the conditions of each grant arrangement and that it is expected that the grant will be received as a result of meeting the necessary conditions. Grants are recognized in the condensed consolidated statements of operations on a systematic basis over the periods in which the Company recognizes the related costs for which the government grant is intended to compensate. Specifically, grant income related to research and development costs is recognized as such expenses are incurred. Grant income is included as a separate caption within Other income (expense) in the condensed consolidated statements of operations.
Leases
In accordance with accounting standards update, or ASU, No. 2016-2, Leases (Topic 842), or ASC 842, and for each of the Company’s leases, the following is recognized: (i) a lease liability, which is a lessee’s obligation to make lease payments arising from a lease, measured on a discounted basis and (ii) a right-of-use asset, which is an asset that represents the lessee’s right to use, or control the use of, a specified asset for the lease term for all leases (with the exception of short-term leases) at the commencement date.
The Company determines if an arrangement is a lease at inception. The Company’s contracts are determined to contain a lease within the scope of ASC 842 when all of the following criteria based on the specific circumstances of the arrangement are met: (1) there is an identified asset for which there are no substantive substitution rights; (2) the Company has the right to obtain substantially all of the economic benefits from the identified asset; and (3) the Company has the right to direct the use of the identified asset.
At the commencement date, operating lease liabilities and their corresponding right-of-use assets are recorded based on the present value of future lease payments over the expected lease term. The Company’s lease agreements do not provide an implicit rate. As a result, the Company utilizes an estimated incremental borrowing rate to discount lease payments, which is based on the rate of interest the Company would have to pay to borrow a similar amount on a collateralized basis over a similar term and based on observable market data points. Certain adjustments to the right-of-use asset may be required for items such as initial direct costs paid or lease incentives received. Operating lease cost is recognized over the expected term on a straight-line basis.
The Company typically only includes an initial lease term in its assessment of a lease agreement. Options to renew a lease are not included in the Company’s assessment unless there is reasonable certainty that the Company will renew. The expected lease term includes noncancellable lease periods and, when applicable, periods covered by an option to extend the lease if the Company is reasonably certain to exercise that option, as well as periods covered by an option to terminate the lease if the Company is reasonably certain not to exercise that option.
Assumptions made by the Company at the commencement date are re-evaluated upon occurrence of certain events, including a lease modification. A lease modification results in a separate contract when the modification grants the lessee an additional right of use not included in the original lease and when lease payments increase commensurate with the standalone price for the additional right of use. When a lease modification results in a separate contract, it is accounted for in the same manner as a new lease.
When a lease is terminated in its entirety, the corresponding lease liability and right-of-use asset are adjusted to zero. Any difference between the carrying amounts of the right-of-use asset and lease liability as compared to the termination payment is recorded in the statement of operations as a gain or loss.
Recent Accounting Pronouncements Not Yet Adopted
On December 14, 2023, the FASB issued ASU 2023-09, or ASU 2023-09, Improvements to Income Tax Disclosures. The standard requires disaggregated information about a reporting entity’s effective tax rate reconciliation as well as information on income taxes paid. The standard is intended to benefit investors by providing more detailed income tax disclosures that would be useful in making capital allocation decisions. ASU 2023-09 applies to all entities subject to income taxes. For public business entities, the new requirements will be effective for annual periods beginning after December 15, 2024. For entities other than public business entities, the requirement will be effective for annual periods beginning after December 15, 2025. The Company is currently evaluating the effect on the unaudited condensed consolidated financial statements.
3. Revenue
General
The Company has not generated revenue from product sales. The Company has generated revenue from contracts with customers (option, license and collaboration agreements), which include upfront payments for licenses or options to obtain licenses, payments for research and development services and milestone payments.
The Company recognized revenue from the following strategic partnerships and other license agreements (in thousands):
|
Three Months Ended June 30, |
Six Months Ended June 30, |
|||||||||||||||
|
2024 |
2023 |
2024 |
2023 |
|||||||||||||
|
Pfizer |
$ | — | $ | 3,486 | $ | 6 | $ | 4,909 | ||||||||
|
AstraZeneca |
— | 4,056 | — | 4,490 | ||||||||||||
|
Servier |
— | (31 | ) | 47 | (105 | ) | ||||||||||
|
Genentech |
— | 12,544 | — | 12,697 | ||||||||||||
|
Total Revenue |
$ | — | $ | 20,055 | $ | 53 | $ | 21,991 | ||||||||
As of June 30, 2024, under the Company’s existing strategic partnerships and other license agreements, the Company could receive the following potential milestone payments (in millions):
|
Research, Development, Regulatory & Commercial Milestones |
Sales Milestones |
|||||||
|
Pfizer |
$ | 759 | $ | 450 | ||||
|
Boston Pharmaceuticals |
85 | 265 | ||||||
|
Total potential milestone payments |
$ | 844 | $ | 715 | ||||
Strategic Partnerships
Genentech
On May 19, 2021, the Company and Genentech, Inc., or Genentech, entered into a Research Collaboration and License Agreement, or the Genentech Agreement, to discover, develop and commercialize locally delivered respiratory and ophthalmology therapies that leverage the Company’s proprietary Anticalin technology. Upon signing the Genentech Agreement, Genentech paid the Company a $20 million upfront fee.
Under the terms of the Genentech Agreement, the Company was responsible for discovery and preclinical development of two initial programs. In April and May 2023, Genentech and the Company decided to discontinue the discovery-stage programs in ophthalmology and respiratory, respectively, for scientific reasons. Pursuant to this decision, the material right performance obligations related to the target swaps for these programs also expired. Based on these decisions, there are no more active performance obligations remaining under the collaboration and the Company recognized all remaining revenue, or $12.5 million, under the collaboration in the three months ended June 30, 2023.
Genentech also had options to select two additional programs with the payment of a fee, which expired in May 2024. With the expiration of these options and no programs active or ongoing, the Genentech Agreement also expired.
Boston Pharmaceuticals
On April 24, 2021, the Company and BP Asset XII, Inc., or Boston Pharmaceuticals, a subsidiary of Boston Pharma Holdings, LLC, entered into an Exclusive Product License Agreement, or the BP Agreement, to develop BOS-342, also referred to as PRS-342, a 4-1BB/GPC3 preclinical immuno-oncology Mabcalin® (antibody-Anticalin fusion) protein.
Under the terms of the BP Agreement, Boston Pharmaceuticals exclusively licensed worldwide rights to BOS-342. The Company received an upfront payment and is further entitled to receive development, regulatory and sales-based milestone payments, tiered royalties up to low double-digits on sales of BOS-342 and a percentage of consideration received by Boston Pharmaceuticals in the event of a sublicense of a program licensed under the BP Agreement or a change of control of Boston Pharmaceuticals.
The Company recognized the full transaction price as revenue in 2021 and has no remaining obligations. In August 2023, the first patient was dosed in the Boston Pharmaceuticals sponsored Phase 1/2 study of PRS-342/BOS-342 in hepatocellular carcinoma (HCC), for which the Company received a milestone payment of $2.5 million.
Pfizer (formerly Seagen)
On February 8, 2018, the Company entered into a license and collaboration agreement, or the Pfizer Collaboration Agreement, and a non-exclusive Anticalin platform technology license agreement, or the Pfizer Platform License, and together with the Pfizer Collaboration Agreement, the Pfizer Agreements, with Pfizer Inc., or Pfizer, pursuant to which the parties agreed to develop multiple targeted bispecific IO treatments for solid tumors and blood cancers.
Under the terms of the Pfizer Agreements, the companies agreed to pursue multiple antibody-Anticalin fusion proteins during the research phase. The Pfizer Agreements provide Pfizer an option to select up to three programs for further development, which Pfizer did, and Pfizer is responsible for developing, funding and commercializing each of these programs.
On March 24, 2021, the Company entered into a Second Pfizer Amendment (formerly the Second Seagen Amendment), to amend the existing immuno-oncology collaboration agreement relating to joint development and commercial rights for one program in the alliance. Under the Second Pfizer Amendment, the Company retains a co-promotion option in the United States for one program, while Pfizer remains solely responsible for the development and overall commercialization of that program. The Company will also be entitled to increased royalties from that program if it chooses to exercise the co-promotion option.
Under the Pfizer Agreements, the Company is eligible to receive other various research, development, commercial and sales milestones. There is uncertainty that the events to obtain the research and development milestones will be achieved given the nature of clinical development and the stage of the Company’s technology. The Company has thus determined that all research and development milestones will be constrained until it is deemed probable that a significant revenue reversal will not occur, with the exception of the $5.0 million milestone as described in the following paragraph.
In January 2023, the Company achieved a milestone for the first program in the Pfizer collaboration for $5.0 million. The Company evaluated the recognition of the milestone under ASC 606 and concluded that the constraints on the milestone no longer existed as of December 31, 2022 and therefore recorded the full $5.0 million as revenue for the year ended December 31, 2022.
In September 2023, Pfizer and the Company entered into an amendment of the Second Pfizer Amendment that provides Pfizer with collaboration product licenses and no changes to the amounts achievable under the collaboration agreement. The effect of the September 2023 amendment was to transfer responsibility for substantially all activities previously performed by the Company to Pfizer. Subsequently, in December 2023, the transfer of the programs was fully approved by the combined joint steering committee. Accordingly, the Company recognized revenue of approximately $10.1 million for the delivery on its performance obligations related to the two programs for the year ended December 31, 2023. With this amendment, the Company has satisfied all remaining obligations under the collaboration.
AstraZeneca
On May 2, 2017, the Company entered into a license and collaboration agreement, or the AstraZeneca Collaboration Agreement, and a non-exclusive Anticalin platform technology license agreement, or AstraZeneca Platform License, and together with the AstraZeneca Collaboration Agreement, the AstraZeneca Agreements, with AstraZeneca AB, or AstraZeneca, which became effective on June 10, 2017, following expiration of the waiting period under the Hart-Scott-Rodino Antitrust Improvements Act of 1976.
In addition to elarekibep (formerly known as PRS-060/AZD1402), or the AstraZeneca Lead Product, the Company and AstraZeneca agreed to collaborate, under the original terms of the AstraZeneca Collaboration Agreement, to progress four additional novel Anticalin proteins against undisclosed targets for respiratory diseases, or the AstraZeneca Collaboration Products, and together with the AstraZeneca Lead Product, the AstraZeneca Products. The first two discovery-stage programs were discontinued in 2022. The third discovery-stage program was discontinued in the second quarter of 2023, which led to recognition of $4.0 million of revenue in that same quarter.
In June 2023, based on non-clinical safety findings in a 13-week toxicology study of elarekibep in non-human primates previously disclosed by the Company, AstraZeneca notified us of its decision to discontinue and cease dosing in the ongoing clinical studies of elarekibep. There was no effect to revenue as a result of the discontinuation of this program.
On July 17, 2023, as a result of the non-clinical safety finding in the 13-week toxicology study of elarekibep in non-human primates, AstraZeneca notified the Company of its intention to terminate the AstraZeneca Collaboration Agreement and the AstraZeneca Platform License, effective October 15, 2023. As a result of this, the remaining amount of current deferred revenue, or $3.5 million, related to the fourth discovery-stage program was recognized in revenue in the third quarter of 2023. With the termination of the AstraZeneca Agreements, there are no more active programs or performance obligations related to the collaboration. Following the termination date, the Company determined that it would not continue development of the programs under the AstraZeneca Agreements.
Servier
In 2017, the Company entered into a license and collaboration agreement, or Servier Collaboration Agreement, and a non-exclusive Anticalin platform license agreement, or Servier Platform License Agreement, and together with the Servier Collaboration Agreement, the Servier Agreements, with Les Laboratoires Servier and Institut de Recherches Internationales Servier, or Servier, pursuant to which the Company and Servier agreed to initially pursue five bispecific therapeutic programs. The intention of the collaboration and defined programs was to combine antibodies from the Servier portfolio with one or more Anticalin proteins based on the Company’s proprietary platform to generate innovative IO bispecific drug candidates.
Since inception, four of the five initially committed programs have been discontinued by Servier. The Company does not presently intend to continue development of the four discontinued programs but retains full rights to advance the development and commercialization of those products on a world-wide basis in the future.
On June 28, 2024, Servier provided the Company with a written notice of termination of the Servier Collaboration Agreement. Pursuant to the Servier Platform License Agreement, the Servier Platform License Agreement terminates upon termination of the Servier Collaboration Agreement. The Servier Collaboration Agreement and Servier Platform License Agreements will terminate effective December 27, 2024, or 180 days from the date on which Servier notified the Company of its intent to terminate both agreements.
With this notice, Servier will discontinue and cease dosing in the Phase 1 clinical study of S095012. Servier's decision to terminate both agreements was based on a potential safety concern in S095012 Phase 1 clinical studies. The Company intends to review the safety data from the S095012 Phase 1 clinical study to understand the implications of the data. The Company does not intend to pursue any further development of S095012.
The Company receives payments from its collaboration partners based on payments established in each contract. Upfront payments and fees are recorded as deferred revenue upon receipt or when due until such time as the Company satisfies its performance obligations under each arrangement. A contract asset is a conditional right to consideration in exchange for goods or services that the Company has transferred to a customer. Amounts are recorded as accounts receivable when the Company’s right is unconditional.
There were no additions to deferred revenue during the three and six months ended June 30, 2024. There were no reductions to deferred revenue for the three and six months ended June 30, 2024 and reductions to deferred revenue were $19.9 million and $21.6 million for the three and six months ended June 30, 2023 respectively.
4. Grant Income
One of the Company's proprietary respiratory assets, PRS-220, an oral inhaled Anticalin protein targeting connective tissue growth factor, or CTGF, was being developed as a local treatment for idiopathic pulmonary fibrosis, and other forms for fibrotic lung disorders. In June 2021, the Company received a €14.2 million (approximately $17.0 million) grant from the Bavarian Ministry of Economic Affairs, Regional Development and Energy (the Bavarian Grant) supporting research and development for post-acute sequelae of SARS-CoV-2 infection (PASC) pulmonary fibrosis, or PASC-PF, also known as post-COVID-19 syndrome pulmonary fibrosis, or “long COVID”.
The Bavarian Grant provides partial reimbursement for qualifying research and development activities on PRS-220, including drug manufacturing costs, activities and costs to support an IND filing, and Phase 1 clinical trials costs. The Bavarian Grant provides reimbursement of qualifying costs incurred through December 2023, with submission for reimbursements allowed through February 2024, which was successfully completed by the Company. The timing of reimbursements follows the expected development timeline of this program. Qualifying costs incurred may exceed the annual grant funding thresholds.
In addition, the Company is required to communicate if there is a change in control or other event that would impact the continuation of PRS-220 to the Bavarian project agency, in which case the Company may be required to refund some or all amounts received under the grant.
5. Cash, cash equivalents and investments
As of June 30, 2024 and December 31, 2023, cash, cash equivalents and investments comprised funds in depository, money market accounts and U.S. treasury securities. The following tables present the cash equivalents and investments carried at fair value in accordance with the hierarchy defined in Note 2 at June 30, 2024 and December 31, 2023.
|
Total |
Quoted prices in active markets (Level 1) |
Significant other observable inputs (Level 2) |
Significant unobservable inputs (Level 3) |
|||||||||||||
|
June 30, 2024 |
||||||||||||||||
|
Money market funds, included in cash equivalents |
$ | 14,138 | $ | 14,138 | $ | — | $ | — | ||||||||
|
Total |
$ | 14,138 | $ | 14,138 | $ | — | $ | — | ||||||||
|
Total |
Quoted prices in active markets (Level 1) |
Significant other observable inputs (Level 2) |
Significant unobservable inputs (Level 3) |
|||||||||||||
|
December 31, 2023 |
||||||||||||||||
|
Money market funds, included in cash equivalents |
$ | 13,224 | $ | 13,224 | $ | — | $ | — | ||||||||
|
Investments - US treasuries |
8,970 | 8,970 | — | — | ||||||||||||
|
Total |
$ | 22,194 | $ | 22,194 | $ | — | $ | — | ||||||||
Cash equivalents and marketable securities have been initially valued at the transaction price and subsequently valued, at the end of each reporting period, utilizing third party pricing services or other market observable data. The pricing services utilize industry standard valuation models, including both income and market-based approaches and observable market inputs to determine value. The Company validates the prices provided by its third-party pricing services by reviewing their pricing methods and obtaining market values from other pricing sources, as needed. After completing its validation procedures, the Company did not adjust any fair value measurements provided by the pricing services as of June 30, 2024.
The Company recorded no realized gains or losses from the maturity of available-for-sale securities during the three and six months ended June 30, 2024 and recorded no realized gains or losses and $0.1 million in realized losses from the maturity of available-for-sale securities during the three and six months ended June 30, 2023, respectively.
6. Assets Held for Sale
As of June 30, 2024 and December 31, 2023, assets held for sale are summarized as follows (in thousands):
|
June 30, |
December 31, |
|||||||
|
2024 |
2023 |
|||||||
|
Laboratory furniture and equipment |
$ | — | $ | 1,967 | ||||
|
Office furniture and equipment |
— | 221 | ||||||
|
Assets held for sale |
$ | — | $ | 2,188 | ||||
At the end of the third quarter of 2023, as part of the Company's strategic process for maximizing the value of assets, the Company committed to a plan to prepare and sell all property and equipment held at the Hallbergmoos, Germany location. The sale of the assets was deemed probable as a result of management's decision, including the estimated timing of sale which was determined to be within a year of the decision. As a result of this decision, the property and equipment met the criteria for held-for-sale accounting.
The net book value of its long-lived assets, as of December 31, 2023 represents the Company's best estimate of the fair value less costs to sell that could be recovered related to lab and office equipment and furniture as part of the Company's initiative to monetize all remaining assets. As the estimated selling price less costs to sell are based primarily on unobservable inputs as they relate to the location and condition of the specific lab equipment and furniture, they are classified in Level 3 in the fair value hierarchy. In the six months ended June 30, 2024, the Company conducted an auction, with the assistance of a third party, of its assets held for sale. After the conclusion of the auction, the Company recovered the total net book value of the assets held for sale and recorded a gain on the sale of the assets of $0.2 million within "Other income (loss)" in the accompanying condensed consolidated statements of operations and comprehensive loss for the three and six months ended June 30, 2024.
7. Accrued Expenses
Accrued expenses and other current liabilities consisted of the following (in thousands):
|
June 30, |
December 31, |
|||||||
|
2024 |
2023 |
|||||||
|
Compensation expense |
$ | 3,108 | $ | 6,448 | ||||
|
Research and development fees |
528 | 968 | ||||||
|
Accrued accounts payable |
701 | 558 | ||||||
|
Other current liabilities |
1,013 | 363 | ||||||
|
Accrued license obligations |
27 | 213 | ||||||
|
Total |
$ | 5,377 | $ | 8,550 | ||||
The compensation expense line item in the above table includes both severance and benefit costs associated with the Company's corporate restructuring actions announced in 2023, inclusive of those employees retained as the service period to earn such benefits is considered complete. The Company recognized restructuring expenses consisting of one-time cash severance payments and other employee-related costs. Severance pay and related costs for certain retained employees are estimated to be paid through the end of 2024. The Company recorded these restructuring charges based on each employee’s role to the respective research and development and general and administrative operating expense categories on its condensed consolidated statements of operations and comprehensive loss.
The following tables includes a roll forward of the restructuring activity and payments recorded for the three and six months ended June 30, 2024 (in thousands):
|
Severance and Benefits Costs |
||||
|
Balance at March 31, 2024 |
$ | 3,634 | ||
|
Adjustments to restructuring charges |
$ | 17 | ||
|
Cash payments |
(801 | ) | ||
|
Balance at June 30, 2024 |
$ | 2,850 | ||
|
Severance and Benefits Costs |
||||
|
Balance at December 31, 2023 |
$ | 5,105 | ||
|
Adjustments to restructuring charges |
$ | (269 | ) | |
|
Cash payments |
(1,986 | ) | ||
|
Balance at June 30, 2024 |
$ | 2,850 | ||
8. Net Income (Loss) per Share
Basic net loss per share is calculated by dividing net income (loss) by the weighted average shares outstanding during the period, without consideration for common stock equivalents. Diluted net loss per share is calculated by adjusting weighted average shares outstanding for the dilutive effect of common stock equivalents outstanding for the period, determined using the treasury-stock and if-converted methods. For purposes of the diluted net loss per share calculation, preferred stock, stock options and warrants are considered to be common stock equivalents but have been excluded from the calculation of diluted net loss per share, as their effect would be anti-dilutive for all periods presented, with the exception of the three months ended June 30, 2023.
A reconciliation of basic and diluted net income (loss) per share is as follows (in thousands, except for per share data):
|
Three Months Ended June 30, |
Six Months Ended June 30, |
|||||||||||||||
|
2024 |
2023 |
2024 |
2023 |
|||||||||||||
|
Net income (loss) |
$ | (3,590 | ) | $ | 3,976 | $ | (8,482 | ) | $ | (9,207 | ) | |||||
|
Basic weighted average common shares outstanding |
1,299 | 1,095 | 1,268 | 1,014 | ||||||||||||
|
Weighted average common equivalent shares |
— | 2 | — | — | ||||||||||||
|
Diluted weighted average common shares outstanding |
1,299 | 1,098 | 1,268 | 1,014 | ||||||||||||
|
Basic net income (loss) per share |
$ | (2.76 | ) | $ | 3.63 | $ | (6.69 | ) | $ | (9.08 | ) | |||||
|
Diluted net income (loss) per share |
$ | (2.76 | ) | $ | 3.62 | $ | (6.69 | ) | $ | (9.08 | ) | |||||
As of June 30, 2024 and 2023, and as calculated using the treasury stock method, approximately 0.5 million of weighted average shares, were excluded from the calculation of diluted weighted average shares outstanding as their effect was antidilutive.
9. Stockholders’ Equity
Effective at 5:00 p.m. Eastern Time on April 22, 2024, the Company effected a 1-for-80 Reverse Split of its common stock. All references to shares of common stock outstanding, average number of shares outstanding and per share amounts in this proxy statement/prospectus have been restated to reflect the Reverse Split on a retroactive basis.
The Company had 3,750,000 shares authorized and 1,320,240 shares and 1,236,688 shares of common stock issued and outstanding as of June 30, 2024 and December 31, 2023, respectively, with a par value of $0.001 per share.
The Company had 10,000,000 shares authorized and 15,617 shares of preferred stock issued and outstanding as of June 30, 2024 and December 31, 2023. Preferred stock has a par value of $0.001 per share, converts on a factor of 13.34 common shares for each preferred share, and consists of the following:
|
• |
Series A Convertible, 85 shares issued and outstanding at June 30, 2024 and December 31, 2023, respectively. |
|
• |
Series B Convertible, 4,026 shares issued and outstanding at June 30, 2024 and December 31, 2023, respectively. |
|
• |
Series C Convertible, 3,506 shares issued and outstanding at June 30, 2024 and December 31, 2023, respectively. |
|
• |
Series D Convertible, 3,000 shares issued and outstanding at June 30, 2024 and December 31, 2023, respectively. |
|
• |
Series E Convertible, 5,000 shares issued and outstanding at June 30, 2024 and December 31, 2023, respectively. |
2020 Employee, Director and Consultant Equity Incentive Plan
At the 2020 Annual Meeting of Stockholders, the Company's stockholders approved the 2020 Employee, Director and Consultant Equity Incentive Plan, or the 2020 Plan. The 2020 Plan permits the Company to issue up to 43,750 shares of common stock pursuant to awards granted under the 2020 Plan. Upon approval of the 2020 Plan, the 2019 Employee, Director and Consultant Equity Incentive Plan, or the 2019 Plan, was terminated; all unissued options were canceled and no additional awards will be made thereunder. All outstanding awards under the 2019 Plan will remain in effect and any awards forfeited from the outstanding awards will be allocated back into the 2020 Plan. There were approximately 19,746 shares remaining and available for grant under the 2019 Plan that terminated upon original approval of the 2020 Plan.
At the 2021 Annual Meeting of Stockholders, held on June 25, 2021, the Company’s stockholders approved the first amendment to the 2020 Plan to add 28,125 shares for issuance under the 2020 Plan. At the 2022 Annual Meeting of Stockholders held on June 22, 2022, the Company’s stockholders approved a second amendment to the 2020 Plan to add 37,500 shares of common stock for issuance under the 2020 Plan. At the 2023 Annual Meeting of Stockholders held on June 21, 2023, the Company’s stockholders approved a third amendment to the 2020 Plan to add 75,000 shares of common stock for issuance under the 2020 Plan.
2023 Employee Stock Purchase Plan
At the 2023 Annual Meeting of Stockholders, the Company’s stockholders approved the 2023 Employee Stock Purchase Plan, or the 2023 ESPP. The 2023 ESPP provides eligible employees with the opportunity to purchase shares of the Company's common stock at a discount, on a tax-favored basis, through regular payroll deductions in compliance with federal tax regulations. The Company has reserved 9,375 shares of common stock for issuance under the 2023 ESPP.
Open Market Sales Agreements
In August 2021, the Company established an at-the-market program, or ATM Program, under a sales agreement with Jefferies LLC, pursuant to which the Company may offer and sell shares of its common stock from time to time, up to an aggregate amount of gross sales proceeds of $50.0 million. The ATM Program is offered under a shelf registration statement on Form S-3 that was filed with and declared effective by the SEC in August 2021. In November 2022, the sales agreement was amended to provide for an increase in the aggregate offering amount, such that under the ATM Program, as amended, the Company may offer and sell shares of its common stock, from time to time, up to an aggregate amount of gross sales proceeds of $75.0 million.
For the six months ended June 30, 2024, the Company did not sell any shares under the ATM program. For the six months ended June 30, 2023, the Company sold 0.3 million shares for gross proceeds of $20.3 million under the ATM program at an average stock price of $67.20 per share.
The Company is currently subject to the SEC general instructions of Form S-3 known as the “baby shelf rules.” Under these instructions, the amount of funds the Company can raise through primary public offerings of securities in any 12-month period using its registration statement on Form S-3 is limited to one-third of the aggregate market value of the shares of the Company’s common stock held by non-affiliates. Therefore, the Company will be limited in the amount of proceeds it is able to raise by selling shares of its common stock using its Form S-3, including under the ATM Program, until such time as its public float exceeds $75 million.
10. Leases
The Company generally conducts its operational functions in the United States remotely.
In October 2018, Pieris Pharmaceuticals GmbH entered into a lease for office and laboratory space located in Hallbergmoos, Germany, or the Hallbergmoos Lease. The Hallbergmoos Lease was subsequently amended in May 2019 and February 2020. The Hallbergmoos Lease, as amended, provided an initial rental term of 12.5 years, and a rental area of approximately 105,000 square feet.
In December 2023, Pieris Pharmaceuticals GmbH entered into an agreement to terminate the Hallbergmoos Lease, or the Lease Termination Agreement. Under the terms of the Lease Termination Agreement, Pieris Pharmaceuticals GmbH terminated the Hallbergmoos Lease in exchange for a termination fee of approximately €9.7 million, and vacated the majority of the premises by December 31, 2023, while continuing to occupy, through June 2024, a limited portion of the office space and using another portion of the former lab space to house its assets being held for sale.
There was no cash paid for amounts included in the measurement of the lease liabilities for the three and six months ended June 30, 2024. Cash paid for amounts included in the measurement of the lease liabilities was $0.5 million and $1.1 million for the three and six months ended June 30, 2023, respectively.
The following table summarizes operating lease costs included in operating expenses (in thousands):
|
Three Months Ended June 30, |
Six Months Ended June 30, |
|||||||||||||||
|
2024 |
2023 |
2024 |
2023 |
|||||||||||||
|
Operating lease costs |
$ | — | $ | 287 | $ | — | $ | 574 | ||||||||
|
Variable lease costs (1) |
— | 204 | — | 388 | ||||||||||||
|
Total lease cost |
$ | — | $ | 491 | $ | — | $ | 962 | ||||||||
|
(1) Variable lease costs include certain additional charges for operating costs, including insurance, maintenance, taxes, utilities, and other costs incurred, which are billed based on both usage and as a percentage of the Company’s share of total square footage. The variable costs for the three and six months ended June 30, 2024 were immaterial, as the Company continues to occupy a limited portion of the space. |
11. Subsequent Events
Merger with Palvella Therapeutics, Inc.
On July 23, 2024, the Company and its wholly-owned subsidiary, Merger Sub entered into the Merger Agreement with Palvella whereby Merger Sub will merge with and into Palvella, with Palvella continuing as a wholly-owned subsidiary of the Company and the surviving corporation of the Merger. If the Merger is completed, the business of Palvella will continue as the business of the combined organization. Consummation of the Merger is contingent on certain closing conditions as identified in the Merger Agreement, including among others, (1) approval by the Company's stockholders of the Required Voting Proposals, as defined in the Merger Agreement, (2) approval by the Palvella stockholders of the adoption of the Merger Agreement, (3) Nasdaq’s approval of the listing of the shares of the Company's common stock to be issued in connection with the Merger, (4) the effectiveness of the Registration Statement, and (5) consummation of the PIPE Financing, all in accordance with the terms of the Purchase Agreement.
Each party’s obligation to consummate the Merger is also subject to other specified customary conditions, including the representations and warranties of the other party being true and correct as of the date of the Merger Agreement and as of the closing date of the Merger, generally subject to an overall material adverse effect qualification, the performance in all material respects by the other party of its obligations under the Merger Agreement required to be performed on or prior to the date of the closing of the Merger and the absence of any material adverse effect affecting the other party that is continuing on the closing date.
Subject to the terms and conditions of the Merger Agreement, at the Effective Time, as defined in the Merger Agreement, (i) each then-outstanding share of Palvella capital stock will be converted into the right to receive a number of shares of the Company's common stock equal to the Exchange Ratio as defined in the Merger Agreement; and (ii) each then-outstanding Palvella stock option to purchase Palvella common stock will be assumed by the Company, subject to adjustment as set forth in the Merger Agreement. In connection with the Merger, and contingent on the approval of the Company's stockholders, the Company intends to amend the amended and restated articles of incorporation of the Company to increase the number of shares of authorized common stock, change the corporate name of the Company to “Palvella Therapeutics, Inc.” and adopt a new 2024 equity incentive plan. The provisions for calculating the Exchange Ratio are set forth in the Merger Agreement, and assume a valuation for Palvella equal to $95 million, and a valuation for Pieris equal to $21 million, provided, that (a) if Pieris’ net cash as of the closing is greater than $11 million, then Pieris’ valuation will be adjusted upwards on a dollar-for-dollar basis by the difference of: (i) Pieris' net cash, minus (ii) $11 million, and (b) if Pieris’ net cash is less than $11 million, then Pieris’ valuation will be adjusted downwards on a dollar-for-dollar basis by the difference of: (i) $11 million, minus (ii) Pieris’ net cash.
Under the terms of the Merger Agreement, on a pro forma basis, it is expected that upon the closing of the Merger, pre-Merger Company stockholders will own approximately 18% of the combined company and pre-Merger Palvella stockholders will own approximately 82% of the combined company, based on the number of shares of the Company's common stock expected to be issued in connection with the Merger, in each case, prior to the issuance of shares under a proposed concurrent private financing. The percentage of the combined company that pre-merger Palvella stockholders and pre-merger Pieris stockholders will own upon the closing of the merger is subject to further adjustment based on the amount of Pieris' net cash at the time of closing.
Each of Pieris and Palvella has agreed to customary representations, warranties and covenants in the Merger Agreement, including, among others, covenants relating to (1) using reasonable best efforts to obtain the requisite approvals of their respective stockholders, (2) non-solicitation of alternative acquisition proposals, (3) the conduct of their respective businesses during the period between the date of signing the Merger Agreement and the closing of the Merger, (4) Pieris using its commercially reasonable efforts to maintain the existing listing of the Pieris common stock on Nasdaq and Pieris causing the shares of Pieris common stock to be issued in connection with the Merger to be approved for listing on Nasdaq prior to the closing of the Merger and (5) Pieris filing with the U.S. Securities and Exchange Commission (the “SEC”) and causing to become effective a registration statement to register the shares of Pieris common stock to be issued in connection with the Merger (the “Registration Statement”). The Registration Statement related to the Merger was included on the Form S-4 filed by the Company with the SEC on August 9, 2024.
The transaction is expected to close in the fourth quarter of 2024 and remains subject to stockholder approval.
Contingent Value Rights
At or prior to the Effective Time, Pieris will enter into a Contingent Value Rights Agreement (the “CVR Agreement”) with a rights agent (“Rights Agent”) pursuant to which Pieris’ pre Merger capital stockholders will receive one contingent value right (each, a “CVR”) for each outstanding share of Pieris common stock held by such stockholder, or share of common stock underlying preferred stock held by such stockholder on such date.
Each CVR will represent the contractual right to receive payments upon the receipt of payments by Pieris or any of its affiliates under certain strategic partner agreements, including existing collaboration agreements pursuant to which Pieris may be entitled to milestones and royalties in the future and other out-licensing agreements for certain of Pieris’ legacy assets, and upon the receipt of certain research and development tax credits in favor of Pieris or any of its affiliates, in each case as set forth in, and subject to and in accordance with the terms and conditions of, the CVR Agreement.
The contingent payments under the CVR Agreement, if they become payable, will become payable to the Rights Agent for subsequent distribution to the holders of the CVRs. In the event that no such proceeds are received, holders of the CVRs will not receive any payment pursuant to the CVR Agreement. There can be no assurance that holders of CVRs will receive any amounts with respect thereto. The right to the contingent payments contemplated by the CVR Agreement is a contractual right only and will not be transferable, except in the limited circumstances specified in the CVR Agreement. The CVRs will not be evidenced by a certificate or any other instrument and will not be registered with the SEC. The CVRs will not have any voting or dividend rights and will not represent any equity or ownership interest in Pieris or any of its affiliates. No interest will accrue on any amounts payable in respect of the CVRs.
Termination Fees
The Merger Agreement contains certain termination rights of each of Pieris and Palvella, including, subject to compliance with the applicable terms of the Merger Agreement, the right of each party to terminate the Merger Agreement to enter into a definitive agreement for a superior proposal. Upon termination of the Merger Agreement under specified circumstances, Pieris may be required to pay Palvella a termination fee of $1.0 million and Palvella may be required to pay Pieris a termination fee of $2.0 million.
Securities Purchase Agreement
On July 23, 2024, and in connection with the executed Merger Agreement, Pieris entered into a securities purchase agreement (the “Purchase Agreement”) with certain investors, including BVF Partners, L.P., an existing stockholder of Pieris (the “PIPE Investors”), pursuant to which, among other things, the PIPE Investors have agreed to subscribe for and purchase (either for cash or in exchange for the termination and cancellation of outstanding convertible notes issued by Palvella), and Pieris agreed to issue and sell to the PIPE Investors, an aggregate of approximately 3,154,241 of shares of Pieris common stock at a price per share equal to $13.7299 multiplied by (x) 0.315478 divided by (y) the Exchange Ratio (the “Purchase Price”), subject to adjustment as set forth in the Purchase Agreement, and/or in lieu of Pieris common stock to certain purchasers who so choose, pre-funded warrants (the “Pre-Funded Warrants”) to purchase up to 2,592,585 shares of Pieris common stock at a purchase price per Pre-Funded Warrant equal to the Purchase Price minus $0.001, subject to adjustment as set forth in the Purchase Agreement, (the “PIPE Financing”). The Purchase Agreement contains customary representations and warranties of Pieris, on the one hand, and the PIPE Investors, on the other hand, and customary conditions to closing, including the consummation of the Merger. The gross proceeds from the PIPE Financing are expected to be approximately $78.9 million, before paying estimated expenses. The closing of the PIPE Financing is expected to occur in connection with and immediately following the consummation of the Merger.
At or prior to the closing of the PIPE Financing, Pieris will enter into a registration rights agreement (the “Registration Rights Agreement”) with the PIPE Investors pursuant to which the PIPE Investors will be entitled to certain resale registration rights with respect to shares of Pieris common stock issued to the PIPE Investors and any shares of Pieris common stock issued upon exercise of the Pre-Funded Warrants. Pursuant to the Registration Rights Agreement, the Company will be required to prepare and file a resale registration statement with the SEC within 30 days following the closing of the PIPE Financing. The Company shall use its commercially reasonable efforts to cause this registration statement to be declared effective by the SEC within 90 days following the closing of the PIPE Financing (or within 120 days following the PIPE Financing if the SEC reviews such resale registration statement).
Subscription and Investment Representation Agreement (Series F Preferred Stock)
On August 7, 2024, the Company entered into a Subscription and Investment Representation Agreement with James Geraghty, Chairman of the Company’s Board of Directors (the "Purchaser"), pursuant to which the Company agreed to issue and sell one (1) share of the Company’s Series F Preferred Stock, par value $0.001 per share, to the Purchaser for $1.00 cash. The sale closed on August 7, 2024.
Voting Rights
The Series F Preferred Stock have no voting rights other than the right to vote on a proposed amendment to the Company’s amended and restated articles of incorporation to effect an increase in the number of authorized shares of the Company’s common stock (the "Authorized Share Increase Proposal"). Each share of Series F Preferred Stock outstanding on the record date entitles the holder thereof to 25,000,000 votes on Authorized Share Increase Proposal, and all shares of Series F Preferred Stock held by such holder must and will be voted, without further action by such holder, in the same proportion as the aggregate shares of Pieris common stock (excluding any shares of Pieris common stock that are not voted) that are voted on the Authorized Share Increase Proposal. As an example, if 70% of the aggregate votes cast by Pieris common stock voting on the Authorized Share Increase Proposal are voted in favor thereof and 30% of the aggregate votes cast by Pieris common stock voting on the Authorized Share Increase Proposal are voted against such Proposal, then 70% of the votes entitled to be cast by the Series F Preferred Stock will be cast in favor of the Proposal and 30% of such votes will be cast against the Proposal. For purposes of the foregoing, abstentions and broker non-votes will not be considered votes cast.
Conversion and Redemption
Shares of the Series F Preferred Stock are not convertible into any other security, and are redeemable by the Company upon the earlier to occur of: (i) the order of the Pieris board of directors in its sole discretion, automatically and effective at such date and time as is determined and specified by the Pieris board of directors in its sole discretion and (ii) automatically and effective immediately after the effectiveness of the increase in the number of authorized shares of Pieris common stock proposed in the Authorized Share Increase Proposal. Upon redemption, the holder of the Series F Preferred Stock will receive cash consideration of $0.01 per share. Shares of the Series F Preferred Stock may not be transferred prior to their redemption without the prior written consent of the Pieris board of directors.
Other Rights and Restrictions
Each holder of Series F Preferred Stock has entered into a written agreement with the Company to attend the Pieris special meeting, to vote all shares of Series F Preferred Stock with regard to the Authorized Share Increase Proposal in the same proportion as the aggregate shares of Pieris common stock (excluding any shares of Pieris common stock that are not voted) are voted on the Authorized Share Increase Proposal and, upon request by the Company, to grant a designee of the Company an irrevocable proxy to vote the shares of Series F Preferred Stock in accordance with the foregoing.
PIERIS PHARMACEUTICALS, INC.
Table of Contents
Report of Independent Registered Public Accounting Firm
To the Stockholders and the Board of Directors of Pieris Pharmaceuticals, Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Pieris Pharmaceuticals, Inc. (the Company) as of December 31, 2023 and 2022, the related consolidated statements of operations and comprehensive loss, changes in stockholders’ equity, and cash flows for each of the two years in the period ended December 31, 2023, and the related notes (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company at December 31, 2023 and 2022, and the results of its operations and its cash flows for each of the two years in the period ended December 31, 2023, in conformity with U.S. generally accepted accounting principles.
Basis for Opinion
These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matter
The critical audit matter communicated below is a matter arising from the current period audit of the financial statements that was communicated or required to be communicated to the audit committee and that: (1) relates to accounts or disclosures that are material to the financial statements and (2) involved our especially challenging, subjective or complex judgments. The communication of the critical audit matter does not alter in any way our opinion on the consolidated financial statements, taken as a whole, and we are not, by communicating the critical audit matter below, providing a separate opinion on the critical audit matter or on the accounts or disclosures to which it relates.
|
Accrued and Prepaid Clinical Trial Expenses |
||
|
Description of the Matter |
As discussed in Note 2 to the consolidated financial statements, the Company records research and development expenses, which include expenses related to clinical trials, as incurred. The Company’s determination of clinical trial costs incurred, as well as the related accrued and prepaid expenses at each reporting period incorporates judgment and utilizes various assumptions. Such judgments and assumptions include an evaluation of the information provided to the Company by third parties on actual costs incurred but not yet billed, estimated project timelines and patient enrollment. Payments for these activities are based on the terms of the individual arrangements, which differ from the pattern of costs incurred.
Auditing the Company’s accrued and prepaid clinical trial expenses was especially challenging due to the large volume of information received from multiple sources that perform service on the Company’s behalf. While the Company’s estimates of accrued and prepaid clinical trial expenses are primarily based on information received related to each study from its vendors, the Company may need to make an estimate for additional costs incurred based on management judgment. Additionally, due to the duration of the work performed under clinical trials and the timing of invoices received from vendors, the actual amounts incurred are not typically known at the time the financial statements are issued. |
|
|
How We Addressed the Matter in Our Audit |
To evaluate the accrued and prepaid clinical trial expenses, our audit procedures included, among others, testing the accuracy and completeness of the underlying data used in the estimates and evaluating the significant assumptions that are used by management to estimate the recorded accruals and prepayments. We corroborated the progress of research and development activities associated with clinical trials through discussion with the Company’s research and development personnel that oversee the research and development activities. We inspected the Company’s third-party contracts, amendments, and any pending change orders to assess the impact on amounts recorded. We also reviewed information received by the Company directly from vendors, which indicated the vendors' estimate of costs incurred to date. In addition, we performed analytics over fluctuations in accruals and prepaids by vendor throughout the period subject to audit and compared subsequent invoices received from third parties to amounts accrued. |
|
/s/ Ernst & Young LLP |
|
We have served as the Company's auditor since 2016. |
|
Boston, Massachusetts |
|
March 29, 2024, except Note 14, as to which the date is August 9, 2024 |
PIERIS PHARMACEUTICALS, INC.
(In thousands, except share and per share data)
|
December 31, |
December 31, |
|||||||
|
2023 |
2022 |
|||||||
|
Assets |
||||||||
|
Current assets: |
||||||||
|
Cash and cash equivalents |
$ | 17,396 | $ | 38,635 | ||||
|
Short term investments |
8,970 | 20,534 | ||||||
|
Accounts receivable |
572 | 5,810 | ||||||
|
Receivable from public grants |
3,141 | 4,771 | ||||||
|
Other receivables |
2,326 | 462 | ||||||
|
Assets held for sale, property and equipment |
2,188 | — | ||||||
|
Prepaid expenses and other current assets |
4,087 | 3,212 | ||||||
|
Total current assets |
38,680 | 73,424 | ||||||
|
Property and equipment, net |
— | 16,992 | ||||||
|
Operating lease right-of-use assets, non-current |
— | 3,705 | ||||||
|
Other non-current assets |
— | 1,369 | ||||||
|
Total assets |
$ | 38,680 | $ | 95,490 | ||||
|
Liabilities and stockholders’ equity |
||||||||
|
Current liabilities: |
||||||||
|
Accounts payable |
$ | 3,372 | $ | 4,154 | ||||
|
Operating lease liabilities, current |
— | 859 | ||||||
|
Accrued expenses and other current liabilities |
8,550 | 10,746 | ||||||
|
Deferred revenues, current portion |
— | 20,824 | ||||||
|
Total current liabilities |
11,922 | 36,583 | ||||||
|
Deferred revenue, net of current portion |
— | 18,734 | ||||||
|
Operating lease liabilities, non-current |
— | 12,244 | ||||||
|
Total liabilities |
11,922 | 67,561 | ||||||
|
Commitments and Contingencies |
||||||||
|
Stockholders’ equity: |
||||||||
|
Convertible preferred stock, $0.001 par value per share, 10,000,000 shares authorized and 15,617 shares issued and outstanding at December 21, 2023 and 2022 |
— | — | ||||||
|
Common stock, $0.001 par value per share, 3,750,000 shares authorized and 1,236,688 and 931,489 shares issued and outstanding at December 21, 2023 and 2022, respectively |
1 | 1 | ||||||
|
Additional paid-in capital |
341,693 | 318,603 | ||||||
|
Accumulated other comprehensive income |
28 | (254 | ) | |||||
|
Accumulated deficit |
(314,964 | ) | (290,421 | ) | ||||
|
Total stockholders’ equity |
26,758 | 27,929 | ||||||
|
Total liabilities and stockholders’ equity |
$ | 38,680 | $ | 95,490 | ||||
The accompanying notes are an integral part of these consolidated financial statements.
PIERIS PHARMACEUTICALS, INC.
CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS
(In thousands, except per share data)
|
Year Ended December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
Revenue |
||||||||
|
Customer revenue |
$ | 38,711 | $ | 25,469 | ||||
|
Collaboration revenue |
4,099 | 433 | ||||||
|
Total revenue |
42,810 | 25,902 | ||||||
|
Operating expenses |
||||||||
|
Research and development |
41,801 | 52,982 | ||||||
|
General and administrative |
16,853 | 16,394 | ||||||
|
Asset impairment |
13,912 | — | ||||||
|
Total operating expenses |
72,566 | 69,376 | ||||||
|
Loss from operations |
(29,756 | ) | (43,474 | ) | ||||
|
Other income (expense) |
||||||||
|
Interest income |
1,851 | 721 | ||||||
|
Grant income |
3,612 | 8,173 | ||||||
|
Other (expense) income |
(250 | ) | 1,303 | |||||
|
Net loss |
$ | (24,543 | ) | $ | (33,277 | ) | ||
|
Other comprehensive (loss) income: |
||||||||
|
Foreign currency translation |
208 | (1,010 | ) | |||||
|
Unrealized gain (loss) on available-for-sale securities |
74 | (73 | ) | |||||
|
Comprehensive loss |
$ | (24,261 | ) | $ | (34,360 | ) | ||
|
Net loss per share |
||||||||
|
Basic and diluted |
$ | (21.80 | ) | $ | (35.90 | ) | ||
|
Weighted average number of common shares outstanding |
||||||||
|
Basic and diluted |
1,126 | 927 | ||||||
The accompanying notes are an integral part of these consolidated financial statements.
PIERIS PHARMACEUTICALS, INC.
CONSOLIDATED STATEMENTS OF CHANGES IN STOCKHOLDERS’ EQUITY
(In thousands)
|
Accumulated |
||||||||||||||||||||||||||||||||
|
Preferred shares |
Common shares |
Additional |
other |
Total |
||||||||||||||||||||||||||||
|
No. of |
Share |
No. of |
Share |
paid-in |
comprehensive |
Accumulated |
Stockholders’ |
|||||||||||||||||||||||||
|
shares |
capital |
shares |
capital |
capital |
income (loss) |
deficit |
equity |
|||||||||||||||||||||||||
|
Balance as of January 1, 2022 |
16 | $ | — | 903 | $ | 1 | $ | 307,069 | $ | 829 | $ | (257,144 | ) | $ | 50,755 | |||||||||||||||||
|
Net loss |
— | — | — | — | — | — | (33,277 | ) | (33,277 | ) | ||||||||||||||||||||||
|
Foreign currency translation adjustment |
— | — | — | — | — | (1,010 | ) | — | (1,010 | ) | ||||||||||||||||||||||
|
Unrealized loss on investments |
— | — | — | — | — | (73 | ) | — | (73 | ) | ||||||||||||||||||||||
|
Stock based compensation expense |
— | — | — | — | 4,402 | — | — | 4,402 | ||||||||||||||||||||||||
|
Issuance of common stock resulting from exercise of stock options |
— | — | 1 | — | 95 | — | — | 95 | ||||||||||||||||||||||||
|
Issuance of common stock resulting from purchase of employee stock purchase plan shares |
— | — | 2 | — | 197 | — | — | 197 | ||||||||||||||||||||||||
|
Issuance of common stock pursuant to ATM offering program, net of $0.3 million in offering costs |
— | — | 25 | — | 6,840 | — | — | 6,840 | ||||||||||||||||||||||||
|
Balance at December 31, 2022 |
16 | $ | — | 931 | $ | 1 | $ | 318,603 | $ | (254 | ) | $ | (290,421 | ) | $ | 27,929 | ||||||||||||||||
|
Net loss |
— | — | — | — | — | — | (24,543 | ) | (24,543 | ) | ||||||||||||||||||||||
|
Foreign currency translation adjustment |
— | — | — | — | — | 208 | — | 208 | ||||||||||||||||||||||||
|
Unrealized gain on investments |
— | — | — | — | — | 74 | — | 74 | ||||||||||||||||||||||||
|
Stock based compensation expense |
— | — | — | — | 3,349 | — | — | 3,349 | ||||||||||||||||||||||||
|
Issuance of common stock resulting from purchase of employee stock purchase plan shares |
— | — | 2 | — | 66 | — | — | 66 | ||||||||||||||||||||||||
|
Issuance of common stock pursuant to ATM offering program, net of $0.7 million in offering costs |
— | — | 304 | — | 19,675 | — | — | 19,675 | ||||||||||||||||||||||||
|
Balance at December 31, 2023 |
16 | $ | — | 1,237 | $ | 1 | $ | 341,693 | $ | 28 | $ | (314,964 | ) | $ | 26,758 | |||||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
PIERIS PHARMACEUTICALS, INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
(In thousands)
|
Year Ended December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
Operating activities: |
||||||||
|
Net loss |
$ | (24,543 | ) | $ | (33,277 | ) | ||
|
Adjustments to reconcile net loss to net cash used in operating activities: |
||||||||
|
Depreciation and amortization (accretion) |
1,904 | 2,770 | ||||||
|
Right-of-use asset (accretion) amortization |
(123 | ) | 10 | |||||
|
Stock-based compensation |
3,349 | 4,402 | ||||||
|
Asset impairment |
13,912 | — | ||||||
|
Realized investment gains |
(53 | ) | (376 | ) | ||||
|
Other non-cash transactions |
(124 | ) | (91 | ) | ||||
|
Changes in operating assets and liabilities: |
||||||||
|
Accounts receivable |
5,307 | (2,624 | ) | |||||
|
Prepaid expenses and other assets |
1,018 | (1,358 | ) | |||||
|
Deferred revenue |
(39,967 | ) | (20,185 | ) | ||||
|
Accounts payable |
(789 | ) | (4,208 | ) | ||||
|
Accrued expenses and other current liabilities |
(2,336 | ) | (4,005 | ) | ||||
|
Lease liability, prior to operating lease termination |
(868 | ) | (990 | ) | ||||
|
Change in lease liability due to termination of operating lease |
(10,506 | ) | — | |||||
|
Net cash used in operating activities |
(53,819 | ) | (59,932 | ) | ||||
|
Investing activities: |
||||||||
|
Purchases of property and equipment |
(171 | ) | (1,041 | ) | ||||
|
Proceeds from maturity of investments |
35,008 | 28,200 | ||||||
|
Purchases of investments |
(22,835 | ) | (48,395 | ) | ||||
|
Net cash provided by (used in) investing activities |
12,002 | (21,236 | ) | |||||
|
Financing activities: |
||||||||
|
Proceeds from exercise of stock options |
— | 95 | ||||||
|
Proceeds from employee stock purchase plan |
66 | 197 | ||||||
|
Proceeds from issuance of common stock resulting from ATM sales, net of $0.7 million and $0.3 million in transaction costs, respectively |
19,729 | 6,922 | ||||||
|
Net cash provided by financing activities |
19,795 | 7,214 | ||||||
|
Effect of exchange rate change on cash and cash equivalents |
783 | (5,175 | ) | |||||
|
Net decrease in cash and cash equivalents |
(21,239 | ) | (79,129 | ) | ||||
|
Cash and cash equivalents at beginning of period |
38,635 | 117,764 | ||||||
|
Cash and cash equivalents at end of period |
$ | 17,396 | $ | 38,635 | ||||
|
Supplemental cash flow disclosures: |
||||||||
|
Net unrealized gain (loss) on investments |
$ | 74 | $ | (73 | ) | |||
|
Property and equipment included in accounts payable |
$ | — | $ | 193 | ||||
The accompanying notes are an integral part of these consolidated financial statements.
PIERIS PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
|
1. |
Corporate Information |
Pieris Pharmaceuticals, Inc., or the Company or Pieris, was founded in May 2013, and acquired 100% interest in Pieris Pharmaceuticals GmbH (formerly Pieris AG, a German company which was founded in 2001) in December 2014. Pieris Pharmaceuticals, Inc. and its wholly-owned subsidiaries, hereinafter collectively Pieris, or the Company, is a biopharmaceutical company that, prior to July of 2023, discovered and developed Anticalin-based drugs to target validated disease pathways in unique and transformative ways. Pieris’ clinical pipeline consists of immuno-oncology, or IO, programs partnered with several major multi-national pharmaceutical companies. Pieris' corporate headquarters is located in Boston, Massachusetts. Pieris also maintains office space in Hallbergmoos, Germany.
The Company’s core Anticalin technology and platform was developed in Germany.
On July 18, 2023, the Company announced its intention to explore engaging in one or more strategic transactions, including mergers, reverse mergers, acquisitions, other business combinations or sales of assets, or other strategic transactions. This decision was related to events that impacted the Company’s inhaled respiratory franchise, based upon AstraZeneca's discontinuation of enrollment of the Phase 2a study for elarekibep, an inhaled IL-4Rα antagonist Anticalin protein to treat uncontrolled asthma. As part of this initiative, the Company engaged Stifel, Nicolaus & Company, Incorporated to serve as its advisor in its review of strategic transactions.
Also on July 18, 2023, the Company’s board of directors approved a reduction in the Company’s workforce by approximately 70%. Since July of 2023, and through December 31, 2023, the Company took additional steps to reduce its operating footprint including terminating its remaining lease obligations in Germany and winding down its proprietary inhaled respiratory programs. The Company also has opted out and terminated programs where possible to reduce operating costs. Further reductions in the workforce have occurred based upon these actions. As a result, the Company has incurred approximately $7.5 million of severance costs and other related termination benefits in 2023 as the service period to earn such benefits is considered complete. The Company expects termination benefits to be paid through the end of 2024.
On March 27, 2024, the Company announced the implementation of a new strategy along with relevant cost-saving measures that are expected to extend its cash runway into at least 2027, while maximizing its ability to capture the potential milestones from its partnered 4-1BB bispecific Mabcalin protein IO assets. The Company may be entitled to aggregate milestones of up to $20 million upon first patient dosed in the phase 2 trials for SGN-BB228, S095012 (formerly PRS-344) and BOS-342, which are all currently in phase 1 clinical development, and up to $55 million upon first patient dosed in pivotal clinical trials for SGN-BB228, S095012 and BOS-342. To support this new strategy, the Company plans to discontinue all of its research and development efforts which it expects to complete by the middle of 2024, implement a workforce reduction that will impact additional employees and the executive leadership team which is expected to be implemented in the second quarter of 2024, and reduce the size of its Board of Directors, which is also expected to have implemented in the second quarter of 2024. In addition to the alliance management activities for its partnered programs, the Company remains committed to obtaining value for its products in prior development, including cinrebafusp alfa, as well as its proprietary platform capabilities by pursuing potential out-licensing or sales transactions. In addition to these potential transactions, the Company may also, from time-to-time, consider strategic opportunities that it believes may increase stockholder value.
As of December 31, 2023, cash, cash equivalents, and investments were $26.4 million. The Company’s net loss was $24.5 million and $33.3 million for the years ended December 31, 2023 and 2022, respectively. The Company has incurred net losses since inception and had an accumulated deficit of $315.0 million as of December 31, 2023. Net losses and negative cash flows have had, and will continue to have, an adverse effect on the Company’s stockholders’ equity and working capital. The Company expects to continue to incur operating losses for the foreseeable future.
The Company has historically devoted substantially all of its financial resources and efforts to research and development and general and administrative expenses to support the discovery and development of Anticalin-based drugs. Going forward, as part of the Company's decision to implement measures to maximize its ability to capture potential milestones from its partnered programs with Pfizer, Boston Pharmaceuticals, and Servier, the Company plans to discontinue all research and development efforts and reduce discretionary expenditures and other fixed or variable personnel costs. The Company believes that its currently available funds will be sufficient to fund its operations through at least the next twelve months from the issuance of this Annual Report on Form 10-K. The Company's belief with respect to its ability to fund operations is based on estimates that are subject to risks and uncertainties. If actual results are different from management’s estimates, the Company may need to seek additional funding.
|
2. |
Summary of Significant Accounting Policies |
Basis of Presentation and Use of Estimates
The accompanying consolidated financial statements of Pieris Pharmaceuticals, Inc. and its wholly-owned subsidiaries were prepared in accordance with U.S. GAAP. The consolidated financial statements include the accounts of all subsidiaries. All intercompany balances and transactions have been eliminated.
The preparation of the financial statements in accordance with U.S. GAAP requires management to make estimates, judgments, and assumptions that affect the reported amounts of assets, liabilities, revenues, expenses and the related disclosures at the date of the financial statements and during the reporting period. Significant estimates are used for, but are not limited to, revenue recognition; deferred tax assets, deferred tax liabilities and valuation allowances; fair value of held for sale assets; beneficial conversion features; fair value of stock options, preferred stock, and warrants; and prepaid and accrued clinical trial expenses. Management evaluates its estimates on an ongoing basis. Actual results and outcomes could differ materially from management’s estimates, judgments, and assumptions.
Foreign Currency Translation
The financial statements of the Company’s foreign subsidiaries are translated from local currency into reporting currency, which is U.S. dollars, using the current exchange rate at the balance sheet date for assets and liabilities, and the weighted average exchange rate prevailing during the period for revenues and expenses. The functional currency for Pieris’ foreign subsidiaries is considered to be the local currency for each entity and, accordingly, translation adjustments for these subsidiaries are included in accumulated other comprehensive loss within stockholders’ equity.
Realized and unrealized gains and losses resulting from foreign currency transactions denominated in currencies other than the functional currency are reflected as other (expense) income, net in the consolidated statements of operations. Foreign currency gains and losses on available-for-sale investment transactions are recorded to other comprehensive income (loss) on the Company's balance sheet per Financial Accounting Standards Board, or FASB, Accounting Standards Codification, or ASC, Topic 830, Foreign Currency Matters.
Cash, Cash Equivalents and Investments
The Company determines the appropriate classification of its investments at the time of purchase. All liquid investments with original maturities of 90 days or less from the purchase date and for which there is an active market are considered to be cash equivalents. The Company’s investments are comprised of money market, asset backed securities, government treasuries, and corporate bonds that are classified as available-for-sale in accordance with FASB ASC 320, Investments—Debt and Equity Securities. The Company classifies investments available to fund current operations as current assets on its balance sheets.
Available-for-sale investments are recorded at fair value, with unrealized gains or losses included in accumulated other comprehensive income (loss) on the Company’s balance sheets. Realized gains and losses are determined using the specific identification method and are included as a component of other income (expense).
The Company reviews investments for other-than-temporary impairment whenever the fair value of an investment is less than the amortized cost and evidence indicates that an investment’s carrying amount is not recoverable within a reasonable period of time. To determine whether an impairment is other-than temporary, the Company considers its intent to sell, or whether it is more likely than not that the Company will be required to sell the investment before recovery of the investment’s amortized cost basis. Evidence considered in this assessment includes reasons for the impairment, the severity and the duration of the impairment, and changes in value subsequent to period end.
Concentration of Credit Risk and Off-Balance Sheet Risk
The Company has no financial instruments with off‑balance sheet risk such as foreign exchange contracts, option contracts, or other foreign hedging arrangements. Financial instruments that subject Pieris to concentrations of credit risk include cash and cash equivalents, investments, and accounts receivable. The Company’s cash, cash equivalents, and investments are held in accounts with financial institutions that management believes are creditworthy. The Company’s investment policy includes guidelines on the quality of the institutions and financial instruments and defines allowable investments that the Company believes minimizes the exposure to concentration of credit risk. The Company has not experienced any credit losses in such accounts and does not believe it is exposed to any significant credit risk on these funds. Accounts receivable primarily consist of amounts due under strategic partnership and other license agreements with major multi-national pharmaceutical companies for which the Company does not obtain collateral.
Fair Value Measurement
The Company is required to disclose information on all assets and liabilities reported at fair value that enables an assessment of the inputs used in determining the reported fair values. FASB ASC Topic 820, Fair Value Measurement and Disclosures, or ASC 820, established a hierarchy of inputs used in measuring fair value that maximizes the use of observable inputs and minimizes the use of unobservable inputs by requiring that the observable inputs be used when available. Observable inputs are inputs that market participants would use in pricing the financial instrument based on market data obtained from sources independent of the Company. Unobservable inputs are inputs that reflect the Company’s assumptions about the inputs that market participants would use in pricing the financial instrument and are developed based on the best information available in the circumstances. The fair value hierarchy applies only to the valuation inputs used in determining the reported or disclosed fair value of the financial instruments and is not a measure of the investment credit quality. Fair value measurements are classified and disclosed in one of the following three categories:
|
• |
Level 1 inputs are quoted prices in active markets for identical assets or liabilities that the reporting entity has the ability to access at the measurement date. |
|
• |
Level 2 utilizes quoted market prices in markets that are not active, broker or dealer quotations, or alternative pricing sources with reasonable levels of price transparency. |
|
• |
Level 3 inputs are unobservable inputs for the asset or liability in which there is little, if any, market activity for the asset or liability at the measurement date. |
To the extent that valuation is based on models or inputs that are less observable or unobservable in the market, the determination of fair value requires more judgment. Accordingly, the degree of judgment exercised by the Company in determining fair value is greatest for instruments categorized in Level 3. A financial instrument’s level within the fair value hierarchy is based on the lowest level of any input that is significant to the fair value measurement.
Financial instruments measured at fair value on a recurring basis include cash equivalents and investments, if any (Note 5).
An entity may elect to measure many financial instruments and certain other items at fair value at specified election dates. Subsequent unrealized gains and losses on items for which the fair value option has been elected will be reported in net loss. The Company did not elect to measure any additional financial instruments or other items at fair value.
Fair Values of Financial Instruments
The fair value of cash, accounts receivable, and accounts payable approximates the carrying value of these financial instruments because of the short-term nature of any maturities. The Company determines the estimated fair values of other financial instruments, using available market information and valuation methodologies, primarily input from independent third party pricing sources.
Accounts Receivable
Accounts receivable are recorded net of allowances for credit losses and represent amounts due from strategic partners. The Company monitors and evaluates collectability of receivables on an ongoing basis and considers whether an allowance for credit losses is necessary. The Company determined that no such reserve is needed as of December 31, 2023 and 2022. Historically, the Company has not had collectability issues.
Property and Equipment
Property and equipment are recorded at acquisition cost, less accumulated depreciation and impairment. Depreciation on property and equipment is calculated using the straight-line method over the remaining estimated useful lives of the assets. Maintenance and repairs to these assets are charged to expenses as occurred. Substantially all of the Company's fixed assets are located in Germany. The estimated useful life of the different groups of property and equipment is as follows:
|
Asset Classification |
Estimated useful life (in years) |
||
|
Leasehold improvements |
shorter of useful life or remaining life of the lease |
||
|
Laboratory furniture and equipment |
8-14 |
||
|
Office furniture and equipment |
5-13 |
||
|
Computer and equipment |
3 - 7 |
If the criteria in ASC 360 Property, Plant and Equipment are met, a long-lived asset is classified as held for sale. The long-lived asset is reported at the lower of its carrying value or fair value less cost to sell beginning in the period the held for sale criteria are met. The carrying amount of the asset will be adjusted each reporting period for subsequent changes in fair value less cost to sell. A loss is recognized for any subsequent write-down to fair value less cost to sell. A gain is recognized for any subsequent increase in fair value less cost to sell, but not in excess of the cumulative loss previously recognized. Once classified as held for sale, depreciation and amortization are no longer recorded for any long-lived assets included in the disposal group.
Impairment of Long-lived Assets
The Company reviews its long-lived assets to be held and used for impairment whenever events or changes in business circumstances indicate that the carrying amount of the assets may not be fully recoverable. The Company evaluates the realizability of its long-lived assets based on profitability and cash flow expectations for the related asset. Any write-downs are treated as permanent reductions in the carrying amount of the assets.
Revenue Recognition
Pieris has entered into several licensing agreements with collaboration partners for the development of Anticalin therapeutics against a variety of targets. The terms of these agreements provide for the transfer of multiple goods or services which may include: (i) licenses, or options to obtain licenses, to Pieris’ Anticalin technology and/or specific programs and (ii) research and development activities to be performed on behalf of or with a collaborative partner. Payments to Pieris under these agreements may include upfront fees (which include license and option fees), payments for research and development activities, payments based upon the achievement of certain milestones, and royalties on product sales. There are no performance, cancellation, termination or refund provisions in any of the arrangements that could result in material financial consequences to Pieris. As the Company's intellectual property assets are considered to be located in Germany, the Company records all consolidated revenue in its subsidiary, Pieris Pharmaceuticals, GmbH.
Collaborative Arrangements
The Company considers the nature and contractual terms of an arrangement and assesses whether the arrangement involves a joint operating activity pursuant to which it is an active participant and exposed to significant risks and rewards with respect to the arrangement. If the Company is an active participant and exposed to the significant risks and rewards with respect to the arrangement, it accounts for these arrangements pursuant to ASC 808, Collaborative Arrangements, or ASC 808, and applies a systematic and rational approach to recognize revenue. The Company classifies payments received as revenue and payments made as a reduction of revenue in the period in which they are earned. Revenue recognized under a collaborative arrangement involving a participant that is not a customer is presented as Collaboration Revenue in the Statement of Operations.
Revenue from Contracts with Customers
In accordance with ASC 606, revenue is recognized when a customer obtains control of promised goods or services. The amount of revenue recognized reflects the consideration to which the Company expects to be entitled in exchange for these goods and services. To achieve this core principle, the Company applies the following five steps: 1) identify the customer contract; 2) identify the contract’s performance obligations; 3) determine the transaction price; 4) allocate the transaction price to the performance obligations; and 5) recognize revenue when or as a performance obligation is satisfied.
The Company evaluates all promised goods and services within a customer contract and determines which of such goods and services are separate performance obligations. This evaluation includes an assessment of whether the good or service is capable of being distinct and whether the good or service is separable from other promises in the contract. In assessing whether promised goods or services are distinct, the Company considers factors such as the stage of development of the underlying intellectual property and the capabilities of the customer to develop the intellectual property on their own or whether the required expertise is readily available.
Licensing arrangements are analyzed to determine whether the promised goods or services, which often include licenses, research and development services and governance committee services, are distinct or whether they must be accounted for as part of a combined performance obligation. If the license is considered not to be distinct, the license would then be combined with other promised goods or services as a combined performance obligation. If the Company is involved in a governance committee, it assesses whether its involvement constitutes a separate performance obligation. When governance committee services are determined to be separate performance obligations, the Company determines the fair value to be allocated to this promised service.
Certain contracts contain optional and additional items, which are considered marketing offers and are accounted for as separate contracts with the customer if such option is elected by the customer, unless the option provides a material right which would not be provided without entering into the contract. An option that is considered a material right is accounted for as a separate performance obligation.
The transaction price is determined based on the consideration to which the Company will be entitled in exchange for transferring goods and services to the customer. A contract may contain variable consideration, including potential payments for both milestone and research and development services. For certain potential milestone payments, the Company estimates the amount of variable consideration by using the most likely amount method. In making this assessment, the Company evaluates factors such as the clinical, regulatory, commercial and other risks that must be overcome to achieve the milestone. Each reporting period the Company re-evaluates the probability of achievement of such variable consideration and any related constraints. Pieris will include variable consideration, without constraint, in the transaction price to the extent it is probable that a significant reversal in the amount of cumulative revenue recognized will not occur when the uncertainty associated with the variable consideration is subsequently resolved. For potential research and development service payments, the Company estimates the amount of variable consideration by using the expected value method, including any approved budget updates arising from additional research or development services.
If the contract contains a single performance obligation, the entire transaction price is allocated to the single performance obligation. Contracts that contain multiple performance obligations require an allocation of the transaction price among the performance obligations on a relative standalone selling price basis unless a portion of the transaction price is variable and meets the criteria to be allocated entirely to a performance obligation or to a distinct good or service that forms part of a single performance obligation.
The Company allocates the transaction price based on the estimated standalone selling price of the underlying performance obligations or in the case of certain variable consideration to one or more performance obligations. The Company must develop assumptions that require judgment to determine the stand-alone selling price for each performance obligation identified in the contract. The Company utilizes key assumptions to determine the stand-alone selling price, which may include other comparable transactions, pricing considered in negotiating the transaction and the estimated costs to complete the respective performance obligation. Certain variable consideration is allocated specifically to one or more performance obligations in a contract when the terms of the variable consideration relate to the satisfaction of the performance obligation and the resulting amounts allocated to each performance obligation are consistent with the amount the Company would expect to receive for each performance obligation.
When a performance obligation is satisfied, revenue is recognized for the amount of the transaction price, excluding estimates of variable consideration that are constrained, that is allocated to that performance obligation on a relative standalone selling price basis. Significant management judgment is required in determining the level of effort required under an arrangement and the period over which the Company is expected to complete its performance obligations under an arrangement.
For performance obligations consisting of licenses and other promises, the Company utilizes judgment to assess the nature of the combined performance obligation to determine whether the combined performance obligation is satisfied over time or at a point in time and, if over time, the appropriate method of measuring progress for purposes of recognizing revenue from non- refundable, up-front fees. The Company evaluates the measure of progress each reporting period and, if necessary, adjusts the measure of performance and related revenue recognition. If the license to the Company’s intellectual property is determined to be distinct from the other performance obligations identified in the arrangement, the Company will recognize revenue from non-refundable, up-front fees allocated to the license when the license is transferred to the customer and the customer is able to use and benefit from the license.
Revenue recognized under an arrangement involving a participant that is a customer is presented as Customer Revenue.
Milestones and Royalties
The Company aggregates milestones into four categories: (i) research milestones, (ii) development milestones, (iii) commercial milestones and (iv) sales milestones. Research milestones are typically achieved upon reaching certain success criteria as defined in each agreement related to developing an Anticalin protein against the specified target. Development milestones are typically reached when a compound reaches a defined phase of clinical research or passes such phase, or upon gaining regulatory approvals. Commercial milestones are typically achieved when an approved pharmaceutical product reaches the status for commercial sale, including regulatory approval. Sales milestones are certain defined levels of net sales by the licensee, such as when a product first achieves global sales or annual sales of a specified amount.
There is uncertainty that the events to obtain the research and development milestones will be achieved given the nature of clinical development and the stage of the Company’s technology. The Company has thus determined that all research and development milestones will be constrained until it is deemed probable that a significant revenue reversal will not occur. For revenues from research and development milestones, payments will be recognized consistent with the recognition pattern of the performance obligation to which they relate.
For arrangements that include sales-based royalties, including milestone payments based on the level of sales, and for which the license is deemed to be the predominant item to which the royalties relate, the Company recognizes revenue at the later of (i) when the related sales occur, or (ii) when the performance obligation to which some or all of the royalty has been allocated has been satisfied (or partially satisfied). Commercial milestones and sales royalties are determined by sales or usage-based thresholds and will be accounted for under the royalty recognition constraint as constrained variable consideration.
The Company calculates the maximum amount of potential milestones achievable under each collaboration agreement and discloses such potential future milestones for all current collaborations using such a maximum calculation.
Contract Balances
The Company recognizes a contract asset when the Company transfers goods or services to a customer before the customer pays consideration or before payment is due, excluding any amounts presented as a receivable (i.e., accounts receivable). A contract asset is an entity’s right to consideration in exchange for goods or services that the entity has transferred to a customer. The contract liabilities (i.e., deferred revenue) primarily relate to contracts where the Company has received payment but has not yet satisfied the related performance obligations.
In the event of an early termination of a collaboration agreement, any contract liabilities would be recognized in the period in which all Company obligations under the agreement have been fulfilled.
Costs to Obtain and Fulfill a Contract with a Customer
Certain costs to obtain customer contracts, including success-based fees paid to third-party service providers, and costs to fulfill customer contracts are capitalized in accordance with FASB ASC 340, Other Assets and Deferred Costs, or ASC 340. These costs are amortized to expense on a systemic basis that is consistent with the transfer to the customer of the goods or services to which the asset relates. The Company will expense the amortization of costs to obtain customer contracts to general and administrative expense and costs to fulfill customer contracts to research and development expense.
Research and Development
Research and development expenses are charged to the statement of operations as incurred. Research and development expenses are comprised of costs incurred in performing research and development activities, including salaries and benefits, facilities costs, pre-clinical and clinical costs, contract services, consulting, depreciation and amortization expense, and other related costs. Costs associated with acquired technology, in the form of upfront fees or milestone payments, are charged to research and development expense as incurred.
Income Taxes
The Company applies ASC Topic 740 Income Taxes, which established financial accounting and reporting requirements for the effects of income taxes that result from the Company’s activities during the current and preceding years. Deferred tax assets and liabilities are recognized for the future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax bases, and operating losses and tax credit carry forwards. Deferred tax assets and liabilities are measured using enacted statutory tax rates expected to apply to taxable income in the jurisdictions and years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in income in the period that includes the enactment date. Where the Company determines that it is more likely than not that some portion or all of the deferred tax assets will not be realized in the future, the deferred tax assets are reduced by a valuation allowance. The Company records interest and penalties related to uncertain tax positions as part of income tax expense.
The Tax Cuts and Jobs Act (TCJA) subjects a U.S. shareholder to tax on global-intangible low tax income (GILTI) earned by certain foreign subsidiaries. The Company has made an accounting policy election to provide for the tax expense related to GILTI in the year the tax is incurred as a period expense only.
Stock-based Compensation
The Company measures share-based payments in accordance with ASC Topic 718, Stock Compensation. Pieris records its stock-based compensation expense over the requisite service period and records forfeitures as they occur. Determining the appropriate fair value model and related assumptions requires judgment, including estimating share price volatility and expected terms of the awards. For employee options, the fair value measurement date is generally on the date of grant and the related compensation expense is recognized on a straight-line basis over the requisite service period of the awards, less expense for actual forfeitures.
The Company uses the Black-Scholes option pricing model to determine the estimated fair value for stock-based awards. Option-pricing models require the input of various subjective assumptions, including the option’s expected life, expected dividend yield, price volatility, risk free interest rate and forfeitures of the underlying stock. Due to the limited operating history of the Company as a public entity and a lack of company specific historical and implied volatility data, the Company has based its estimate of expected volatility on the historical volatility of a group of similar companies that are publicly traded. When selecting these public companies on which it has based its expected stock price volatility, the Company selected companies with comparable characteristics to it, including enterprise value, risk profiles, position within the industry, and with historical share price information sufficient to meet the expected term of the stock-based awards. The Company computes historical volatility data using the daily closing prices for the selected companies’ shares during the equivalent period of the calculated expected term of the stock-based awards. The Company will continue to apply this process until a sufficient amount of historical information regarding the volatility of its own stock price becomes available.
Due to the lack of Company specific historical option activity, the Company has estimated the expected term of its employee stock options using the “simplified” method, whereby, the expected term equals the arithmetic average of the vesting term and the original contractual term of the option. The expected term for non-employee awards is the remaining contractual term of the option. The risk-free interest rates are based on the U.S. Treasury securities with a maturity date commensurate with the expected term of the associated award. The Company has never paid dividends, but may elect pay out dividends to stockholders in the future if we determine that there is sufficient cash and investments to achieve our near and long-term objectives.
All excess tax benefits and tax deficiencies are recorded as income tax expense or benefit in the Company's statement of operations and comprehensive loss. For the years ended December 31, 2023 and 2022, the Company did not record an income statement benefit for excess tax benefits as a valuation allowance is also required on these amounts.
Government Grants
The Company recognizes grants from governmental agencies when there is reasonable assurance that the Company will comply with the conditions attached to the grant arrangement and the grant will be received. The Company evaluates the conditions of each grant as of each reporting period to evaluate whether the Company has reached reasonable assurance of meeting the conditions of each grant arrangement and that it is expected that the grant will be received as a result of meeting the necessary conditions. Grants are recognized in the consolidated statements of operations on a systematic basis over the periods in which the Company recognizes the related costs for which the government grant is intended to compensate. Specifically, grant income related to research and development costs is recognized as such expenses are incurred. Grant income is included as a separate caption within Other income (expense), net in the consolidated statements of operations.
Leases
The Company accounts for leases pursuant to ASC 842 Leases (Topic 842), or ASC 842. As a lessee, the Company is required to recognize (i) a lease liability, which is a lessee's obligation to make lease payments arising from a lease, measured on a discounted basis; and (ii) a right-of-use asset, which is an asset that represents the lessee’s right to use, or control the use of, a specified asset for the lease term for all leases (with the exception of short-term leases) at the commencement date.
The Company determines if an arrangement is a lease at inception. The Company’s contracts are determined to contain a lease within the scope of ASC 842 when all of the following criteria based on the specific circumstances of the arrangement are met: (1) there is an identified asset for which there are no substantive substitution rights; (2) the Company has the right to obtain substantially all of the economic benefits from the identified asset; and (3) the Company has the right to direct the use of the identified asset. In addition, the Company does not apply the recognition requirements in the lease standard to short-term leases (a lease that at commencement date has a lease term of 12 months or less and does not contain a purchase option that it is reasonably certain to exercise) and does not separate lease and non-lease components for all asset classes. Any variable components of lease costs are excluded from lease payments and are recognized in the period incurred, including increases to rent based on German Consumer Price Index, or CPI.
At the commencement date, operating lease liabilities and their corresponding right-of-use assets are recorded based on the present value of future lease payments over the expected lease term. The Company’s lease agreements do not provide an implicit rate. As a result, the Company utilizes an estimated incremental borrowing rate to discount lease payments, which is based on the rate of interest the Company would have to pay to borrow a similar amount on a collateralized basis over a similar term and based on observable market data points. Certain adjustments to the right-of-use asset may be required for items such as initial direct costs paid or lease incentives received. Operating lease cost is recognized over the expected term on a straight-line basis.
The Company typically only includes an initial lease term in its assessment of a lease agreement. Options to renew a lease are not included in the Company’s assessment unless there is reasonable certainty that the Company will renew. The expected lease term includes noncancelable lease periods and, when applicable, periods covered by an option to extend the lease if the Company is reasonably certain to exercise that option, as well as periods covered by an option to terminate the lease if the Company is reasonably certain not to exercise that option.
Assumptions made by the Company at the commencement date are re-evaluated upon occurrence of certain events, including a lease modification. A lease modification results in a separate contract when the modification grants the lessee an additional right of use not included in the original lease and when lease payments increase commensurate with the standalone price for the additional right of use. When a lease modification results in a separate contract, it is accounted for in the same manner as a new lease.
When a lease is terminated in its entirety, the corresponding lease liability and right-of-use asset are adjusted to zero. Any difference between the carrying amounts of the right-of-use asset and lease liability as compared to the termination payment is recorded in the statement of operations as a gain or loss.
Contingencies
Accruals are recorded for loss contingencies when it is probable that a liability has been incurred and the amount of the related loss can be reasonably estimated. The Company evaluates, on a quarterly basis, developments in legal proceedings and other matters that could cause an increase or decrease in the amount of the liability that has been accrued previously. Considering facts known at the time of the assessment, the Company determines whether potential losses are considered reasonably possible or probable and whether they are estimable. Based upon this assessment, the Company carries out an evaluation of disclosure requirements and considers possible accruals in the financial statements.
Segment Reporting
Operating segments are identified as components of an enterprise where separate discrete financial information is evaluated by the chief operating decision maker in making decisions on how to allocate resources and assess performance. The Company operates as a single segment dedicated to the discovery and development of biotechnological applications and the Company’s chief operating decision maker, or CODM, makes decisions based on the Company as a whole. The Company has determined that its CODM is its Chief Executive Officer.
Earnings per Share
Basic earnings per share attributable to common stockholders is calculated by dividing net loss attributable to common stockholders by the weighted average shares outstanding during the period, without consideration for common stock equivalents.
Diluted earnings per share attributable to common stockholders is calculated by adjusting weighted average shares outstanding for the dilutive effect of common stock equivalents outstanding for the period, determined using the treasury-stock and if-converted methods. For purposes of the diluted net loss per share attributable to common stockholders' calculation, preferred stock, stock options, unvested restricted stock, and warrants are considered to be common stock equivalents but have been excluded from the calculation of diluted net loss per share attributable to common stockholders, as their effect would be anti-dilutive for all periods presented. Therefore, basic and diluted net loss per share were the same for all periods presented.
Recent Accounting Pronouncements Not Yet Adopted
The Company has considered recent accounting pronouncements and concluded that they are either not applicable to the business, or that the effect is not expected to be material to the audited consolidated financial statements as a result of future adoption.
|
3. |
Revenue |
General
The Company has not generated revenue from product sales. The Company has generated revenue from contracts with customers (option, license and collaboration agreements), which include upfront payments for licenses or options to obtain licenses, payments for research and development services and milestone payments.
During the years ended December 31, 2023 and 2022, respectively, the Company recognized revenue from the following strategic partnerships and other license agreements (in thousands):
|
Year Ended December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
AstraZeneca |
$ | 8,399 | $ | 9,117 | ||||
|
Pfizer |
15,134 | 8,287 | ||||||
|
Servier |
4,099 | 5,359 | ||||||
|
Genentech |
12,697 | 3,139 | ||||||
|
Boston Pharmaceuticals |
2,481 | — | ||||||
|
Total Revenue |
$ | 42,810 | $ | 25,902 | ||||
Under the Company´s existing strategic partnerships and other license agreements, the Company could receive the following potential milestone payments (in millions) as of December 31, 2023:
|
Research, Development, Regulatory & Commercial Milestones |
Sales Milestones |
|||||||
|
Pfizer |
$ | 759 | $ | 450 | ||||
|
Servier |
107 | 99 | ||||||
|
Boston Pharmaceuticals |
85 | 265 | ||||||
|
Total potential milestone payments |
$ | 951 | $ | 814 | ||||
Strategic Partnerships
Genentech
On May 19, 2021, the Company and Genentech, Inc., or Genentech, entered into a Research Collaboration and License Agreement, or the Genentech Agreement, to discover, develop and commercialize locally delivered respiratory and ophthalmology therapies that leverage the Company’s proprietary Anticalin technology. Upon signing the Genentech Agreement, Genentech paid the Company a $20 million upfront fee. In addition, the Company may be eligible to receive additional milestone payments across multiple programs, as well as tiered royalty payments on net sales at percentages ranging from the mid-single to low double-digits, subject to certain standard reductions and offsets.
Under the terms of the Genentech Agreement, the Company was responsible for discovery and preclinical development of two initial programs. In April and May 2023, Genentech and the Company decided to discontinue the discovery-stage programs in ophthalmology and respiratory, respectively, for scientific reasons. Pursuant to this decision, the material right performance obligations related to the target swaps for these programs also expired. Based on these decisions, there aren't any active performance obligations remaining under the collaboration and the Company recognized all remaining revenue, or $12.5 million, under the collaboration in the second quarter of 2023.
The Genentech Agreement also provided an option to select additional programs, at Genentech's discretion, for a fee and this option expires in May 2024. If Genentech exercises its option to start additional programs, the Company would be eligible to receive additional milestone payments, as well as tiered royalty payments on net sales, subject to certain standard reductions and offsets. Genentech’s options to nominate two additional collaboration targets of their choosing is subject to the legal availability of the target to be researched. As of December 31, 2023, any variable consideration related to the exercise of such options is considered fully constrained.
Boston Pharmaceuticals
On April 24, 2021, the Company and BP Asset XII, Inc., or Boston Pharmaceuticals, a subsidiary of Boston Pharma Holdings, LLC, entered into an Exclusive Product License Agreement, or the BP Agreement, to develop PRS-342, a 4-1BB/GPC3 preclinical immuno-oncology Anticalin-antibody bispecific fusion protein.
Under the terms of the BP Agreement, Boston Pharmaceuticals exclusively licensed worldwide rights to PRS-342. The Company received an upfront payment of $10.0 million and is further entitled to receive development, regulatory and sales-based milestone payments, tiered royalties up to low double-digits on sales of PRS-342 and a percentage of consideration received by Boston Pharmaceuticals in the event of a sublicense of a program licensed under the BP Agreement or a change of control of Boston Pharmaceuticals. The Company also contributed $4.0 million toward manufacturing activities.
The Company completed all performance obligations in 2021, at which point the revenue was recorded from the upfront payment. In August 2023, the first patient was dosed in the Boston Pharmaceuticals sponsored Phase 1/2 study for BOS-342 in hepatocellular carcinoma, or HCC, for which the Company received a milestone payment.
Pfizer
On February 8, 2018, the Company entered into a license and collaboration agreement, or the Pfizer Collaboration Agreement (formerly the Seagen Collaboration Agreement), and a non-exclusive Anticalin platform technology license agreement, or the Pfizer Platform License (formerly the Seagen Platform License), and together with the Pfizer Collaboration Agreement, the Pfizer Agreements (formerly the Seagen Agreements), with Pfizer (formerly Seagen), pursuant to which they agreed to develop multiple targeted bispecific IO treatments for solid tumors and blood cancers.
Under the terms of the Pfizer Agreements, the companies pursued multiple Anticalin-antibody fusion proteins during the research phase. The Pfizer Agreements provide Pfizer a base option to select up to three programs for further development. Prior to the initiation of a pivotal trial, the Company may opt into global co-development and U.S. commercialization of the second program and share in global costs and profits on an equal basis. Pfizer will solely develop, fund and commercialize the other two programs. Pfizer may also decide to select additional candidates from the initial research phase for further development in return for the payment to us of additional fees, milestone payments, and royalties.
On March 24, 2021, the Company announced that Pfizer made a strategic equity investment in Pieris, and that the companies had entered into a Second Pfizer Amendment (formerly Second Seagen Amendment), in which their existing immuno-oncology collaboration agreement has been amended relating to joint development and commercial rights for one program in the alliance. Under the Second Pfizer Amendment, Pieris’ option to co-develop and co-commercialize one of three programs in the collaboration was converted to a co-promotion option in the United States, with Pfizer solely responsible for the development and overall commercialization of that program. Pieris will also be entitled to increased royalties from that program in the event that it chooses to exercise the co-promotion option. In connection with the agreements described above, the Company and Pfizer entered into a subscription agreement, or the Pfizer Subscription Agreement (formerly the Seagen Subscription Agreement), pursuant to which the Company agreed to issue to Pfizer, and Pfizer agreed to acquire from the Company, 46,327 shares of the Company’s common stock for a total purchase price of $13.0 million, or $280.80 per share, in a private placement transaction pursuant to Section 4(a)(2) of the Securities Act of 1933, as amended. The Pfizer Subscription Agreement includes a provision to the effect that Pfizer may ask the Company to file a registration statement to register the resale of the shares issued to Pfizer, at any time beginning on the date that is 60 calendar days from the date of issuance of the shares. The Company assessed the ASC 606 implications of the Pfizer Subscription Agreement and concluded that the fair value of the shares on a per share basis was $208.80 per share as of the transaction date. This resulted in a premium paid for the shares of $3.3 million, all of which was recorded in deferred revenue upon contract execution and allocated to the remaining performance obligations.
In the second quarter of 2022, the Company recorded approximately $1.5 million in revenue related to completion of the performance obligation for the expiration of the target swap under the second program in the collaboration.
Under the Pfizer Agreements, the Company is eligible to receive various research, development, commercial and sales milestones. There is uncertainty that the events to obtain the research and development milestones will be achieved given the nature of clinical development and the stage of the Company’s technology. The Company has thus determined that all research and development milestones will be constrained until it is deemed probable that a significant revenue reversal will not occur, with the exception of the $5.0 million milestone as described in the following paragraph.
In January 2023, the Company achieved a milestone for the first program in the collaboration for $5.0 million. The Company evaluated the recognition of the milestone under ASC 606 and concluded that the constraints on the milestone no longer existed as of December 31, 2022 and therefore recorded the full $5.0 million as revenue for the year ended December 31, 2022.
In September 2023, Pfizer and the Company entered into an amendment of the Second Pfizer Amendment that provides Pfizer with collaboration product licenses and no changes to the amounts achievable under the collaboration agreement. The effect of the September 2023 amendment was to transfer responsibility for substantially all activities previously performed by the Company to Pfizer. Subsequently, in December 2023, the transfer of the programs was fully approved by the combined joint steering committee. Accordingly, the Company recognized revenue of approximately $10.1 million for the delivery on its performance obligations related to the two programs for the year ended December 31, 2023. With this amendment, the Company has satisfied all remaining obligations under the collaboration.
AstraZeneca
On May 2, 2017, the Company entered into a license and collaboration agreement, or the AstraZeneca Collaboration Agreement, and a non-exclusive Anticalin platform technology license agreement, or AstraZeneca Platform License, and together with the AstraZeneca Collaboration Agreement, the AstraZeneca Agreements with AstraZeneca AB, or AstraZeneca, which became effective on June 10, 2017, following expiration of the waiting period under the Hart-Scott-Rodino Antitrust Improvements Act of 1976. Under the AstraZeneca Agreements the parties agreed to advance several novel inhaled Anticalin proteins.
In addition to the Company’s former lead inhaled drug candidate, PRS-060/AZD1402, or the AstraZeneca Lead Product, the Company and AstraZeneca, under the original terms of the AstraZeneca Collaboration Agreement, would also collaborate to progress four additional novel Anticalin proteins against undisclosed targets for respiratory diseases, or the AstraZeneca Collaboration Products, and together with the AstraZeneca Lead Product, the AstraZeneca Products. As of June 2023, the AstraZeneca Lead Product and three of the four AstraZeneca Collaboration Products had been discontinued. The first two discovery-stage programs were previously discontinued in 2022, which led to approximately $9.7 million in revenue recognized due to these discontinuations. Elarekibep and the third discovery-stage program were discontinued in the second quarter of 2023. The discontinuation of the third discovery program led to recognition of $4.0 million of revenue in the quarter ended June 30, 2023, while there was no revenue recognized as a result of the discontinuation of elarekibep.
The Company was responsible for advancing the AstraZeneca Lead Product through its phase 1 study, with the associated costs funded by AstraZeneca. The parties would collaborate thereafter to conduct a phase 2a study in asthma patients, with AstraZeneca continuing to fund development costs. After completion of a phase 2a study, Pieris had the option to co-develop the AstraZeneca Lead Product and also had a separate option to co-commercialize the AstraZeneca Lead Product in the United States. For the AstraZeneca Collaboration Products, the Company was responsible for the initial discovery of the novel Anticalin proteins, after which AstraZeneca would take the lead on continued development of the AstraZeneca Collaboration Products. The Company had the option to co-develop two of the four AstraZeneca Collaboration Products beginning at a pre-defined preclinical stage and would also have the option to co-commercialize these two programs in the United States, while AstraZeneca would be responsible for development and commercialization of the other programs worldwide.
On July 17, 2023, AstraZeneca notified the Company of its intention to terminate the AstraZeneca Collaboration Agreement and the AstraZeneca Platform License, effective October 15, 2023. AstraZeneca’s decision to terminate the AstraZeneca Agreements was based on non-clinical safety findings in a 13-week toxicology study of elarekibep in non-human primates previously disclosed by the Company. As a result of this, the remaining amount of current deferred revenue, or $3.5 million, related to the fourth discovery-stage program was recognized in revenue as of September 30, 2023. With the termination of the AstraZeneca Agreements, there are no more active programs or performance obligations related to the collaboration. Following the termination date, the Company determined that it would not continue development of the programs under the AstraZeneca Agreements.
The Company incurred $1.6 million of third-party success fees to obtain the contract with AstraZeneca. Upon adoption of ASC 606, the Company capitalized $1.1 million in accordance with ASC 340. In accordance with the termination of the AstraZeneca Agreements and recognition of remaining revenue, the Company also amortized the remaining deferred transactions costs to obtain the contract, or $0.3 million. Amortization for the year ended December 31, 2022 was $0.3 million.
Servier
In 2017, the Company entered into a license and collaboration agreement, or Servier Collaboration Agreement, and a non-exclusive Anticalin platform license agreement, or Servier Platform License, and together with the Servier Collaboration Agreement, the Servier Agreements with Les Laboratoires Servier and Institut de Recherches Internationales Servier, or Servier, pursuant to which the Company and Servier agreed to initially pursue five bispecific therapeutic programs. The intention of the collaboration and defined programs was to combine antibodies from the Servier portfolio with one or more Anticalin proteins based on the Company's proprietary platform to generate innovative IO bispecific drug candidates, or the Collaboration products.
In the first quarter of 2022, the Company satisfied the performance obligation related to the material right for S095025 (PRS-352), which led to point-in-time recognition of revenue for $4.9 million of revenue previously deferred. In the fourth quarter of 2022, Servier discontinued development of S095025 based upon a strategic portfolio review. Since inception, four of the five initially committed programs have been discontinued by Servier. The Company does not presently intend to continue development of the four discontinued programs but retains full rights to advance the development and commercialization of those products on a world-wide basis in the future.
In July 2023, the Company notified Servier of its decision to opt out of co-development and commercialization of S095012 (PRS-344), a 4- 1BB/PD- L1 bispecific Mabcalin protein, in the U.S. Servier retains exclusive, even as to the Company, worldwide rights to the program, including the right to continue to advance development and potential commercialization of S095012 (PRS-344) in the U.S. As a result of the Company's decision to opt out of co-development, the Company will be entitled to increased royalty rates and potential royalties and milestones, if any, for S095012 (PRS-344) under the terms of the Servier Agreement. With the decision to opt out of co-development of S095012 (PRS-344), the Company recognized the remaining revenue under the collaboration, or $4.7 million, in 2023 and there are no more active co-development programs under the collaboration.
Contract Balances
The Company receives payments from its collaboration partners based on payments established in each contract. Upfront payments and fees are recorded as deferred revenue upon receipt or when due until such time as the Company satisfies its performance obligations under each arrangement. A contract asset is a conditional right to consideration in exchange for goods or services that the Company has transferred to a customer. Amounts are recorded as accounts receivable when the Company’s right is unconditional.
There were no additions to deferred revenue during the year ended December 31, 2023 and reductions to deferred revenue were $39.7 million for the year ended December 31, 2023.
|
4. |
Grant Income |
One of the Company's proprietary respiratory assets is PRS-220, an oral inhaled Anticalin protein targeting connective tissue growth factor, or CTGF, was being developed as a local treatment for idiopathic pulmonary fibrosis, and other forms for fibrotic lung disorders. In June 2021, the Company received a €14.2 million (approximately $17.0 million) grant from the Bavarian Ministry of Economic Affairs, Regional Development and Energy (the Bavarian Grant) supporting research and development for post-acute sequelae of SARS-CoV-2 infection (PASC) pulmonary fibrosis, or PASC-PF, also known as post-COVID-19 syndrome pulmonary fibrosis, or “long COVID”.
The Bavarian Grant provides partial reimbursement for qualifying research and development activities on PRS-220, including drug manufacturing costs, activities and costs to support an IND filing, and phase 1 clinical trials costs. The Bavarian Grant provides reimbursement of qualifying costs incurred through December 2023, with submission for reimbursements allowed through February 2024, which was successfully completed by the Company. The timing follows the expected development timeline of this program. Qualifying costs incurred may exceed the annual grant funding thresholds.
In addition, the Company is required to communicate if there is a change in control or other event that would impact the continuation of PRS-220 to the Bavarian project agency, in which case the Company may be required to refund some or all amounts received under the grant.
|
5. |
Cash, Cash Equivalents and Investments |
As of December 31, 2023, cash, cash equivalents and investments comprised funds in depository, money market accounts and U.S. treasury securities. As of December 31, 2022, cash, cash equivalents and investments comprised funds in depository, money market accounts, U.S. and foreign treasury securities, asset-backed securities and corporate bonds. The following table presents the cash equivalents and investments carried at fair value in accordance with the hierarchy defined in Note 2 at December 31, 2023.
|
Total |
Quoted prices in active markets (Level 1) |
Significant other observable inputs (Level 2) |
Significant unobservable inputs (Level 3) |
|||||||||||||
|
December 31, 2023 |
||||||||||||||||
|
Money market funds, included in cash equivalents |
$ | 13,224 | $ | 13,224 | $ | — | $ | — | ||||||||
|
Investments - US treasuries |
8,970 | 8,970 | — | — | ||||||||||||
|
Total |
$ | 22,194 | $ | 22,194 | $ | — | $ | — | ||||||||
|
Total |
Quoted prices in active markets (Level 1) |
Significant other observable inputs (Level 2) |
Significant unobservable inputs (Level 3) |
|||||||||||||
|
December 31, 2022 |
||||||||||||||||
|
Money market funds, included in cash equivalents |
$ | 17,618 | $ | 17,618 | $ | — | $ | — | ||||||||
|
Investments - US treasuries |
3,573 | 3,573 | — | — | ||||||||||||
|
Investments - Foreign treasuries |
896 | 896 | — | — | ||||||||||||
|
Investments - Asset-backed securities |
499 | — | 499 | — | ||||||||||||
|
Investments - Corporate bonds |
15,566 | — | 15,566 | — | ||||||||||||
|
Total |
$ | 38,152 | $ | 22,087 | $ | 16,065 | $ | — | ||||||||
Cash equivalents and marketable securities have been initially valued at the transaction price and subsequently valued, at the end of each reporting period, utilizing third party pricing services or other market observable data. The pricing services utilize industry standard valuation models, including both income and market-based approaches and observable market inputs to determine value. The Company validates the prices provided by its third-party pricing services by reviewing their pricing methods and obtaining market values from other pricing sources, as needed. After completing its validation procedures, the Company did not adjust any fair value measurements provided by the pricing services as of December 31, 2023.
Investments at December 31, 2023 consist of the following (in thousands):
|
Contractual maturity |
||||||||||||||||||||
|
(in days) |
Amortized Cost |
Unrealized gains |
Unrealized losses |
Fair Value |
||||||||||||||||
|
Investments |
||||||||||||||||||||
|
US treasuries |
4-51 | $ | 8,969 | $ | 1 | $ | — | $ | 8,970 | |||||||||||
|
Total |
$ | 8,969 | $ | 1 | $ | — | $ | 8,970 | ||||||||||||
The Company recorded realized losses from the maturity of available-for-sale securities of $0.1 million and realized gains of $0.4 million for the years ended December 31, 2023 and 2022, respectively.
As of December 31, 2023, there were no investments with a fair value that was significantly lower than the amortized cost basis or any investments that had been in an unrealized loss position for a significant period.
|
6. |
Assets Held for Sale, Property and Equipment |
As of December 31, 2023, assets held for sale are summarized as follows (in thousands):
|
December 31, |
||||
|
2023 |
||||
|
Laboratory furniture and equipment |
$ | 1,967 | ||
|
Office furniture and equipment |
221 | |||
|
Assets held for sale |
$ | 2,188 | ||
At the end of the third quarter of 2023, as part of the Company's strategic process for maximizing the value of assets, the Company committed to a plan to prepare and sell all property and equipment held at the Hallbergmoos, Germany location. The sale of the assets was deemed probable as a result of management's decision, including the estimated timing of sale which was determined to be within a year of the decision. As a result of this decision, the property and equipment met the criteria for held-for-sale accounting.
The Company recorded impairment charges totaling $13.9 million, of which $1.8 million related to impairment of its right-of-use asset under the Hallbergmoos Lease (see Note 13) with the remaining related to a complete write-off of leasehold improvements and a partial impairment of the Company's other long-lived assets. The remaining $2.2 million in net book value of its long-lived assets represents the Company's best estimate of the fair value less costs to sell that could be recovered related to lab and office equipment and furniture as part of the Company's initiative to monetize all remaining assets. As the estimated selling price less costs to sell are based primarily on unobservable inputs as they relate to the location and condition of the specific lab equipment and furniture, they are classified in Level 3 in the fair value hierarchy. In the first quarter of 2024, the Company conducted an auction, with the assistance of a third party, of its assets held for sale. After the conclusion of the auction, the Company has recovered substantially all of the total net book value of the assets held for sale. The Company has further plans to sell all remaining assets in the second quarter of 2024.
As of December 31, 2022, property and equipment are summarized as follows (in thousands):
|
December 31, |
||||
|
2022 |
||||
|
Laboratory furniture and equipment |
$ | 11,970 | ||
|
Office furniture and equipment |
1,861 | |||
|
Computer equipment |
364 | |||
|
Leasehold improvements |
12,444 | |||
|
Property and equipment, cost |
26,639 | |||
|
Accumulated depreciation |
(9,647 | ) | ||
|
Property and equipment, net |
$ | 16,992 | ||
Depreciation expense was $1.8 million and $2.3 million for the years ended December 31, 2023 and 2022, respectively. There were no other changes in accumulated depreciation other than the foreign currency impact.
|
7. |
Accrued Expenses |
Accrued expenses and other current liabilities consisted of the following (in thousands):
|
Years Ended December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
Compensation expense |
$ | 6,448 | $ | 3,015 | ||||
|
Research and development fees |
968 | 5,758 | ||||||
|
Accrued accounts payable |
558 | 1,245 | ||||||
|
Other current liabilities |
363 | 483 | ||||||
|
Accrued license obligations |
213 | 245 | ||||||
|
Total |
$ | 8,550 | $ | 10,746 | ||||
The compensation expense line item in the above table includes both severance and benefit costs associated with the Company's corporate restructuring actions announced in 2023, inclusive of those employees retained as the service period to earn such benefits is considered complete. The Company recognized restructuring expenses consisting of one-time cash severance payments and other employee-related costs. Severance pay and related costs for certain retained employees are estimated to be paid through the end of 2024. The Company recorded these restructuring charges based on each employee’s role to the respective research and development and general and administrative operating expense categories on its consolidated statements of operations and comprehensive loss.
The following table includes a roll forward of the restructuring activity and payments recorded for the year ended December 31, 2023 (in thousands):
|
Severance and Benefits Costs |
||||
|
Restructuring expenses |
$ | 7,523 | ||
|
Cash payments |
$ | (2,418 | ) | |
|
Balance at December 31, 2023 |
$ | 5,105 | ||
|
8. |
Income Taxes |
The Company reported a loss before income taxes consisting of the following (in thousands):
|
Years Ended December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
Domestic |
$ | (9,818 | ) | $ | (11,765 | ) | ||
|
Foreign |
(14,726 | ) | (21,512 | ) | ||||
|
Loss before income taxes |
$ | (24,544 | ) | $ | (33,277 | ) | ||
The components of the provision for income taxes are as follows (in thousands):
|
Years Ended December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
Current: |
||||||||
|
Federal |
$ | — | $ | — | ||||
|
State |
— | — | ||||||
|
Foreign |
— | — | ||||||
|
Total current |
— | — | ||||||
|
Deferred: |
||||||||
|
Federal |
— | — | ||||||
|
State |
— | — | ||||||
|
Foreign |
— | — | ||||||
|
Total deferred |
— | — | ||||||
|
Provision for income taxes |
$ | — | $ | — | ||||
The reconciliation of the federal statutory rate to the Company’s effective tax rate is as follows:
|
2023 |
2022 |
|||||||
|
Federal income tax rate |
21.0 | % | 21.0 | % | ||||
|
Foreign rate differential |
0.7 | % | 5.0 | % | ||||
|
State tax, net of federal benefit |
3.8 | % | 2.0 | % | ||||
|
Share-based awards compensation |
(2.1 | )% | (2.2 | )% | ||||
|
Permanent items |
(2.1 | )% | 0.3 | % | ||||
|
Other |
(1.0 | )% | 1.0 | % | ||||
|
Release of uncertain tax position |
22.7 | % | — | % | ||||
|
Credits |
0.8 | % | 1.2 | % | ||||
|
Change in valuation allowance |
(43.8 | )% | (28.3 | )% | ||||
|
Effective income tax rate |
— | % | — | % | ||||
The components of deferred tax assets and liabilities related to net tax effects of temporary differences between the carrying amount of assets and liabilities for financial reporting purposes and the amounts used for income taxes purposes were as follows (in thousands):
|
Years Ended December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
Deferred tax assets: |
||||||||
|
Net operating loss carryforwards |
$ | 67,496 | $ | 54,845 | ||||
|
Share-based awards compensation |
2,624 | 3,112 | ||||||
|
Accrued expenses |
461 | 216 | ||||||
|
R&D Credits |
644 | 413 | ||||||
|
Depreciation and other |
479 | 384 | ||||||
|
Unrealized foreign currency |
(377 | ) | 359 | |||||
|
Capitalized R&D |
1,165 | 952 | ||||||
|
Lease liability |
— | 3,541 | ||||||
|
Total deferred tax assets |
72,492 | 63,822 | ||||||
|
Deferred tax liabilities: |
||||||||
|
Right-of-use asset |
— | (3,270 | ) | |||||
|
Accrued expenses |
— | — | ||||||
|
Total deferred tax liabilities |
— | (3,270 | ) | |||||
|
Less: valuation allowance: |
(72,492 | ) | (60,552 | ) | ||||
|
Net deferred tax asset |
$ | — | $ | — | ||||
The Company operates in multiple jurisdictions. Accordingly, the Company files U.S. federal and state income tax returns as well as returns in multiple foreign jurisdictions. In assessing the realizability of deferred tax assets, management considers whether it is more likely than not that some portion or all deferred tax assets will not be realized. The ultimate realization of deferred tax assets is dependent upon the generation of future taxable income during the periods in which those temporary differences become deductible. Management considers the scheduled reversal of deferred tax liabilities, projected future taxable income, and tax-planning strategies in making this assessment. Management believes it is more likely than not that the results of future operations will not generate sufficient taxable income in the United States or in its foreign jurisdictions to realize the full benefits of its deferred tax assets. As of December 31, 2023, the Company continues to maintain a full valuation allowance against all net deferred tax assets.
The cumulative amount of earnings of our foreign subsidiaries are expected to be permanently invested in the foreign subsidiaries. Deferred taxes have not been provided on the excess of book basis over tax basis, or the excess tax basis over book basis in the shares of our foreign subsidiaries because these basis differences are not expected to reverse in the foreseeable future and are essentially permanent in duration. Our intention is to reinvest the earnings of the foreign subsidiaries indefinitely.
The increase in the valuation allowance of deferred tax assets of $11.9 million for the year ended December 31, 2023 was primarily a result of the operating losses generated in current tax year.
As of December 31, 2023, the Company had net operating loss carryforwards for U.S. federal income tax purposes of $43.4 million and net operating loss carryforwards for state income tax purposes of $46.7 million. Federal tax loss carryforwards that were created prior to December 31, 2017 expire through 2037 and federal losses created after that date do not expire. State loss carryforwards expire starting in 2035. Pursuant to Section 382 of the Internal Revenue Code of 1986, or the Code, and similar state tax law, certain substantial changes in the Company’s ownership may result in a limitation on the amount of net operating loss and tax credit carryforwards that may be used in future years. Utilization of the net operating loss and tax credit carryforwards may be subject to an annual limitation under Section 382 of the Code due to ownership change limitations that have occurred previously or that could occur in the future. These ownership changes may limit the amount of net operating loss and tax credit carryforwards that can be utilized annually to offset future taxable income and tax, respectively. The Company completed a Section 382 study through December 31, 2020. Based on the study, the Company underwent an ownership change for Section 382 purposes which occurred in February 2018. As a result of the ownership change, the Company’s net operating loss and tax credit carryforwards as of the ownership change dates are subject to limitation under Section 382; however, these limitations are not expected to result in any of the impacted net operating loss and tax credit carryforwards to expire unutilized. Any net operating losses or tax credits generated after the February 2018 change are not subject to this annual limitation. However, subsequent ownership changes, as defined by Section 382, may potentially further limit the amount of net operating loss and tax credit carryforwards that could be utilized to offset future taxable income and tax. The Company is currently open to examination under the statute of limitations by the Internal Revenue Service and state jurisdictions for the tax years ended 2019 through the current year. Carryforward tax attributes generated in years past may still be adjusted upon future examination if they have or will be used in a future period. The Company is not under examination by the Internal Revenue Service or any other jurisdictions for any tax years.
As of December 31, 2023, the Company had German corporate income tax and trade tax net operating loss carryforwards of approximately $187.6 million and $183.7 million respectively. Under current German laws, tax loss carryforwards may only be used to offset any relevant later assessment period (calendar year) of $1.2 million plus 60% of the exceeding taxable income and trade profit of such period and do not expire. In addition, certain transactions, including transfers of shares or interest in the loss holding entity, may result in the partial or total forfeiture of tax losses existing at that date. Partial or total forfeiture of tax losses may further occur in corporate reorganizations of the loss holding entity.
As of December 31, 2023, the Company had gross U.S. federal and state research and development and other tax credit carryforwards of $0.5 million and $0.1 million, respectively, which may be available to offset future tax liabilities and begin to expire in 2042 and 2037, respectively. As of December 31, 2022, the Company had gross U.S. federal and state research and development and other tax credit carryforwards of $0.3 million and $0.1 million, respectively, which may be available to offset future tax liabilities and begin to expire in 2042 and 2037, respectively.
The Company accounts for uncertain tax positions pursuant to ASC 740, Income Taxes, which prescribes a recognition threshold and measurement process for financial statement recognition of uncertain tax positions taken or expected to be taken in a tax return. If the tax position meets this threshold, the benefit to be recognized is measured at the largest amount of benefit that is more likely than not (determined by cumulative probability) of being realized upon ultimate settlement with the taxing authority. The Company recognizes interest and penalties, if any, related to uncertain tax positions in income tax expense. No interest and penalties related to uncertain tax positions were accrued at December 31, 2023 and December 31, 2022.
On December 22, 2017, the Tax Cuts and Jobs Act, or the TCJA, was signed into law. Under the TCJA provisions, effective with tax years beginning on or after January 1, 2022, taxpayers can no longer immediately expense qualified research and development expenditures. Taxpayers are now required to capitalize and amortize these costs over five years for research conducted within the United States or 15 years for research conducted abroad.
The following table sets forth a reconciliation of the beginning and ending amounts of unrecognized tax benefits, excluding the impact of interest and penalties, for the year ended December 31, 2023 (in thousands):
|
Unrecognized tax benefits at December 31, 2022 |
$ | 5,363 | ||
|
Decrease as a result of a lapse of the applicable statute of limitations |
(5,363 | ) | ||
|
Unrecognized tax benefits at December 31, 2023 |
$ | — |
The Company does not expect unrecognized tax benefits to change significantly over the next twelve months. The full amount of unrecognized tax benefits would impact the effective rate, subject to valuation allowance considerations, if recognized.
|
9. |
Stockholders’ equity |
The Company had 3,750,000 shares authorized and 1,236,688 and 931,489 shares of common stock issued and outstanding as of December 31, 2023 and December 31, 2022, respectively, with a par value of $0.001 per share.
The Company had 10,000,000 shares authorized and 15,617 shares of preferred stock issued and outstanding as of December 31, 2023 and 2022. Preferred stock has a par value of $0.001 per share, and consists of the following tranches:
|
• |
Series A Convertible, 85 shares issued and outstanding at December 31, 2023 and 2022 |
|
• |
Series B Convertible, 4,026 shares issued and outstanding at December 31, 2023 and 2022 |
|
• |
Series C Convertible, 3,506 shares issued and outstanding at December 31, 2023 and 2022 |
|
• |
Series D Convertible, 3,000 shares issued and outstanding at December 31, 2023 and 2022 |
|
• |
Series E Convertible, 5,000 shares issued and outstanding at December 31, 2023 and 2022 |
Common Stock
Each share of the Company’s common stock is entitled to one vote and all shares rank equally as to voting and other matters. Dividends may be declared and paid on the common stock from funds legally available therefore, if, as and when determined by the Board of Directors.
Preferred Stock
The Company has issued multiple series (Series A through E) of preferred stock to certain entities affiliated with Biotechnology Value Fund, L.P., or BVF. In each case, each share Preferred Stock is convertible into 13.34 shares of the Company's common stock (subject to adjustment as provided in the Certificate of Designation for each series) at any time at the option of the holder, provided that the holder is prohibited from converting the Preferred Stock into shares of the Company's common stock if, as a result of such conversion, the holder, together with its affiliates, would own more than 9.99% of the total number of shares of Common Stock then issued and outstanding, or the Beneficial Ownership Limitation. The holder may reset the Beneficial Ownership Limitation to a higher or lower number, not to exceed 19.99% of the total number of common shares issued and outstanding immediately after giving effect to a conversion, upon providing written notice to the Company. Any such notice providing for an increase to the Beneficial Ownership Limitation will be effective 61 days after delivery to the Company.
Series A, Series B, Series C, Series D and Series E Preferred Stock rank senior to the Company’s common stock; senior to any class or series of capital stock of the Company created after the designation of and specifically ranking by its terms as junior to the five series of Preferred Stock; in parity with each other and with any class or series of capital stock of the Company created after the designation of and specifically ranking by its terms as in parity with the existing five series of Preferred Stock; and junior to any class or series of capital stock of the Company created after the designation of and specifically ranking by its terms as senior to the existing five series of Preferred Stock. In the event of the Company’s liquidation, dissolution or winding up, subject to the rights of holders of, holders are entitled to receive a payment equal to $0.001 per share of Preferred Stock pursuant to the rights and preferences discussed above, plus an additional amount equal to any dividends declared but unpaid on such shares. However, if the assets of the Company are insufficient to comply with the preceding sentence, then all remaining assets of the Company shall be distributed ratably to holders of the shares of the existing five series of Preferred Stock.
For each series of Preferred Stock, the Company designated the requisite number of shares of its authorized and unissued preferred stock as a specific series of Preferred Stock and filed a Certificate of Designation with the Nevada Secretary of State.
Shares of Preferred Stock generally have no voting rights, except as required by law and except that the consent of holders of a majority of the then outstanding Preferred Stock is required to amend the terms of the Certificate of Designation for each respective series of Preferred Stock. Holders of Preferred Stock are entitled to receive any dividends payable to holders of the Company's common stock subject to the rights and preferences discussed above, in each case, as to distributions of assets upon the Company’s liquidation, dissolution or winding up whether voluntarily or involuntarily and/or the right to receive dividends.
Series A Preferred Stock
In June 2016, the Company entered into a securities purchase agreement for a private placement of the Company’s securities with a select group of institutional investors, or the 2016 PIPE. The 2016 PIPE sale transaction, by the Company, consisted of 8,188,804 units at a price of $2.015 per unit for gross proceeds, to the Company, of approximately $16.5 million. After deducting for placement agent fees and offering expenses, the aggregate net proceeds from the private placement was approximately $15.3 million. In connection with the 2016 PIPE, the Company issued 40,323 shares of common stock and 4,963 shares of Series A Preferred Stock to the 2016 PIPE investors.
Series B Preferred Stock
On January 30, 2019, the Company and certain entities affiliated with BVF entered into an exchange agreement pursuant to which BVF agreed to exchange an aggregate of 62,500 shares of the Company’s common stock owned by BVF for an aggregate of 5,000 shares of Series B Preferred Stock.
Series C and 2019 Private Placement
In November 2019, the Company entered into a securities purchase agreement for a private placement, or the Purchase Agreement with a select group of institutional investors, including lead investor BVF as well as existing and new investors, or Investors. The private placement consisted of 9,014,960 units, at a price of $3.55 per unit, or the Financing, for gross proceeds of approximately $32.0 million, and net proceeds to the Company of approximately $31.0 million. Each unit consists of (i) one share of the Company’s common stock or 0.001 shares of non-voting convertible preferred stock, or Series C Preferred Shares, and together with the Common Shares, or Shares, and (ii) one immediately-exercisable warrant to purchase one share of the Company’s common stock with an exercise price of $568.00, or Exercise Price.
If (i) the initial public disclosure of the phase 2a Study of elarekibep that includes the “p” value achieved for the primary endpoint of such study reveals top-line data on the primary efficacy endpoint in the phase 2a Study with a “p” value below 0.05 (i.e., p < 0.05) in at least one dose level; and (ii) the 10-day volume weighted average stock price commencing on the trading day immediately after the initial public disclosure is at least three percent more than the Exercise Price, ((i) and (ii), collectively, the “Performance Condition”), then the warrants will be exercisable for a period of 60 days from the date of the initial data disclosure and may only be exercised for cash. Otherwise, the warrants will be exercisable for a period of five years from the date of issuance, or Exercise Date. If the Performance Condition has not been met and the last reported sale price of the Company’s common stock immediately prior to the Expiration Date was greater than the Exercise Price, then the warrants shall be automatically deemed exercised on a cashless basis on the Expiration Date.
Upon issuance, each Series C Preferred Share included an embedded beneficial conversion feature as the market price of the Company’s common stock on the date of issuance of the Series C convertible Preferred Stock was $274.40 per share. As a result, the Company recorded the intrinsic value of the beneficial conversion feature of $2.8 million as a discount on the Series C convertible preferred stock at issuance. As the Series C Preferred Shares are immediately convertible upon issuance and do not include a stated redemption date, the discount was immediately accreted as a deemed dividend.
Series D Preferred Stock Conversion
On March 31, 2020, the Company and certain entities affiliated with BVF entered into an exchange agreement pursuant to which, on April 1, 2020, BVF exchanged an aggregate of 37,500 shares of the Company’s common stock owned by BVF for an aggregate of 3,000 shares of Series D Preferred Stock.
Series E Preferred Stock Conversion
On May 20, 2021, the Company and certain entities affiliated with BVF entered into an exchange agreement pursuant to which, BVF exchanged an aggregate of 62,500 shares of the Company’s common stock owned by BVF for an aggregate of 5,000 shares of Series E Preferred Stock.
Open Market Sales Agreement
In August 2021, the Company established an at-the-market program, or ATM Program, under a sales agreement with Jefferies LLC, pursuant to which the Company may offer and sell shares of its common stock, par value $0.001 per share, from time to time, up to an aggregate amount of gross sales proceeds of $50.0 million. In November 2022, the sales agreement was amended to provide for an increase in the aggregate offering amount, such that under the ATM Program, as amended, the Company may offer and sell shares of its common stock, from time to time, up to an aggregate amount of gross sales proceeds of $75.0 million. The ATM Program, as amended, is offered under a shelf registration statement on Form S-3 that was filed with and declared effective by the SEC in August 2021. For the year ended December 31, 2023, the Company sold 0.3 million shares for gross proceeds of $20.3 million under the ATM Program at an average stock price of $67.07. For the year ended December 31, 2022, the Company sold 25,000 shares for gross proceeds of $7.2 million under the ATM Programs and the predecessor ATM program at an average stock price of $276.51.
As of the filing of this Annual Report on Form 10-K, the Company will be subject to the SEC general instructions of Form S-3 known as the “baby shelf rules.” Under these instructions, the amount of funds the Company can raise through primary public offerings of securities in any 12-month period using its registration statement on Form S-3 is limited to one-third of the aggregate market value of the shares of the Company’s common stock held by non-affiliates. Therefore, the Company will be limited in the amount of proceeds it is able to raise by selling shares of its common stock using its Form S-3, including under the ATM Program, until such time as its public float exceeds $75 million.
|
10. |
Net Loss per Share |
Basic net loss per share is calculated by dividing net income (loss) by the weighted average shares outstanding during the period, without consideration for common stock equivalents. Diluted net loss per share is calculated by adjusting weighted average shares outstanding for the dilutive effect of common stock equivalents outstanding for the period, determined using the treasury-stock and if-converted methods. For purposes of the diluted net loss per share calculation, preferred stock, stock options, and warrants are considered to be common stock equivalents but have been excluded from the calculation of diluted net loss per share, as their effect would be anti-dilutive for all periods presented. Therefore, basic and diluted net loss per share were the same for all periods presented.
For the years ended December 31, 2023 and 2022, and as calculated using the treasury stock method, approximately 0.5 million and 0.5 million of weighted average shares, respectively, were excluded from the calculation of diluted weighted average shares outstanding as their effect was antidilutive.
|
11. |
Stock and Employee Benefit Plans |
Employee, Director and Consultant Equity Incentive Plans
At the 2020 Annual Shareholder Meeting, held on June 23, 2020, the stockholders approved the 2020 Employee, Director and Consultant Equity Incentive Plan, or the 2020 Plan. The 2020 Plan originally permitted the Company to issue up to 43,750 shares of common stock pursuant to awards granted under the 2020 Plan. Upon approval of the 2020 Plan, the 2019 Employee, Director and Consultant Equity Incentive Plan, or the 2019 Plan, was terminated; all unissued options were canceled and no additional awards will be made thereunder. All outstanding awards under the 2019 Plan will remain in effect and any awards forfeited from the outstanding awards will be allocated back into the 2020 Plan. The 2020 Plan, similar to the 2019 Plan, provided for the grant of stock options, restricted and unrestricted stock awards, and other stock-based awards to employees of the Company, non-employee directors of the Company, and certain other consultants performing services for the Company as designated by either the Board of Directors or the compensation committee of the Board of Directors. At the 2021 Annual Meeting of Stockholders, held on June 25, 2021, the Company’s stockholders approved the first amendment to the 2020 Plan to add 28,125 shares for issuance under the 2020 Plan, which increased the total permitted for issuance under the 2020 Plan to 71,875. At the 2022 Annual Meeting of Stockholders, held on June 22, 2022, the Company’s stockholders approved the second amendment to the 2020 Plan to add an additional 37,500 shares for issuance under the 2020 Plan. At the 2023 Annual Meeting of Stockholders held on June 21, 2023, the Company's stockholders approved a third amendment to the 2020 Plan to add 75,000 shares of common stock for issuance under the 2020 Plan, which increased the total permitted for issuance under the 2020 Plan to 184,375. The 2020 Plan permits the Company to issue up to 184,375 shares reserved for issuance pursuant to the 2020 Plan and any additional shares which may be issued if awards outstanding under the Company’s 2014, 2016, 2018 and 2019 Plans are canceled or expire.
The Company’s stock options have a maximum term of 10 years from the date of grant. Stock options granted may be either incentive stock options or nonqualified stock options and the exercise price of stock options must be at least equal to the fair market value of the common stock on the date of grant. The Company’s general policy is to issue shares of common stock upon the exercise of stock options.
The Company estimates the fair value of each stock award on the grant date using the Black-Scholes option-pricing model based on the following assumptions:
|
Years Ended December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
Risk free interest rate |
3.33% - 4.11 | % | 1.43% - 3.39 | % | ||||
|
Expected term (in years) |
5.5 - 5.73 | 5.5 - 5.73 | ||||||
|
Dividend yield |
— | — | ||||||
|
Expected volatility |
79.5% - 98.6 | % | 79.9% - 81.1 | % | ||||
The weighted-average fair value of the 46,677 and 38,498 options granted during the years ended December 31, 2023 and 2022 was $97.34 and $230.29, respectively. As of December 31, 2023, there were 116,060 shares available for future grant under the 2020 Plan.
The following table summarizes stock option activity for employees and non-employees:
|
Weighted- |
||||||||||||||||
|
Weighted- |
Average |
Aggregate |
||||||||||||||
|
Average |
Remaining |
Intrinsic |
||||||||||||||
|
Number of |
Exercise |
Contractual |
Value |
|||||||||||||
|
Options |
Price |
Life (in years) |
(in thousands) |
|||||||||||||
|
Outstanding, December 31, 2022 |
165,849 |
$ |
256.93 |
$ |
— |
|||||||||||
|
Granted |
46,677 |
97.34 |
— |
|||||||||||||
|
Canceled/Forfeited |
34,345 |
208.95 |
— |
|||||||||||||
|
Outstanding, December 31, 2023 |
178,181 |
$ |
224.37 |
5.90 |
$ |
— |
||||||||||
|
Vested or expected to vest, December 31, 2023 |
178,181 |
$ |
224.37 |
5.90 |
$ |
— |
||||||||||
|
Exercisable, December 31, 2023 |
126,179 |
$ |
257.26 |
4.75 |
$ |
— |
||||||||||
Periodically, the Company grants inducement options, which are awards outside of stockholder-approved stock option plans, and which are awarded as an inducement material to the executive officers or other personnel entering senior leadership roles with the Company. The terms of inducement option awards were substantially the same as those issued under our 2020 Plan. These awards are excluded from the table above. The following table summarizes stock option activity for these inducement options (in thousands):
|
Weighted- |
||||||||||||||||
|
Weighted- |
Average |
Aggregate |
||||||||||||||
|
Average |
Remaining |
Intrinsic |
||||||||||||||
|
Number of |
Exercise |
Contractual |
Value |
|||||||||||||
|
Options |
Price |
Life (in years) |
(in thousands) |
|||||||||||||
|
Outstanding, December 31, 2022 |
3,750 |
$ |
372.00 |
$ |
— |
|||||||||||
|
Granted |
— |
$ |
— |
$ |
— |
|||||||||||
|
Canceled/Forfeited |
— |
$ |
— |
$ |
— |
|||||||||||
|
Outstanding, December 31, 2023 |
3,750 |
$ |
372.00 |
5.67 |
$ |
— |
||||||||||
|
Vested or expected to vest, December 31, 2023 |
3,750 |
$ |
372.00 |
5.67 |
$ |
— |
||||||||||
|
Exercisable, December 31, 2023 |
3,750 |
$ |
372.00 |
5.67 |
$ |
— |
||||||||||
Employee Stock Purchase Plans
At the 2023 Annual Meeting of Stockholders, the Company’s stockholders approved the 2023 Employee Stock Purchase Plan, or the 2023 ESPP, which replaces the former 2018 Employee Stock Purchase Plan, or 2018 ESPP. The 2023 ESPP provides eligible employees with the opportunity to purchase shares of the Company's common stock at a discount of 85% of the lower closing market price of the common stock at the beginning date or ending date of each purchase period, on a tax-favored basis, through regular payroll deductions in compliance with federal tax regulations. The Company has reserved 9,375 shares of common stock for issuance under the 2023 ESPP.
Total shares purchased under the 2023 ESPP and 2018 ESPP plan were 1,933 and 2,268 for the years ended December 31, 2023 and 2022, respectively.
Total Stock-based Compensation Expense
Total stock-based compensation expense is recorded in operating expenses based upon the functional responsibilities of the individuals holding the respective options as follows (in thousands):
|
Years Ended December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
Research and development |
$ | 1,190 | $ | 1,905 | ||||
|
General and administrative |
2,159 | 2,497 | ||||||
|
Total stock-based compensation |
$ | 3,349 | $ | 4,402 | ||||
As of December 31, 2023, the total unrecognized compensation cost related to all non-vested awards was $4.3 million. The unrecognized compensation cost would be recognized over a remaining weighted-average period of 2.24 years.
|
12. |
License Agreement |
TUM License
The Company and the Technical University of Munich, or TUM, initiated discussions in the second quarter of 2018 to clarify, expand and restructure the research and licensing agreement with TUM, the TUM License, including the parties’ obligations under the TUM License. The TUM License assigns or exclusively licenses to the Company certain intellectual property related to the Company's Anticalin platform technology. The parties' discussions relate to revised commercial terms and to re-initiating additional collaborations between faculty at TUM and Pieris. While an amended and restated license agreement has not yet been completed, the Company may enter into an amendment reflecting the parties discussions. These discussions may also lead to an increase in the Company's collaborative research activities with TUM.
|
13. |
Leases |
In August 2015, the Company entered into a sublease to lease approximately 3,950 square feet in Boston, Massachusetts, which expired on December 31, 2022. The Company did not extend the sublease. The Company generally conducts its operational functions in the United States remotely.
In October 2018, Pieris Pharmaceuticals GmbH entered into a lease for office and laboratory space located in Hallbergmoos, Germany, or the Hallbergmoos Lease. Pieris GmbH moved its operations to the Hallbergmoos facility in February 2020. The Hallbergmoos Lease was subsequently amended in May 2019 and February 2020. The Hallbergmoos Lease, as amended, provided an initial rental term of 12.5 years, and a rental area of approximately 105,000 square feet.
In December 2023, Pieris Pharmaceuticals GmbH entered into an agreement to terminate the Hallbermoos Lease, or the Lease Termination Agreement. Under the terms of the Lease Termination Agreement, Pieris Pharmaceuticals GmbH terminated the Hallbergmoos Lease in exchange for a termination fee of approximately €9.7 million, and vacated the majority of the premises by December 31, 2023, while continuing to occupy, through June 2024, a limited portion of the office space and using another portion of the former lab space to house its assets being held for sale.
Cash paid for amounts included in the measurement of the lease liabilities were $2.2 million and $2.4 million for the years ended December 31, 2023 and 2022, respectively, all of which were incurred prior to the lease termination.
The Hallbergmoos Lease included $11.5 million of tenant improvements allowance for normal tenant improvements which was incurred prior to commencing the lease. The Company capitalized the leasehold incentives which were included in Property and equipment, net on the Consolidated Balance Sheet and were amortized on a straight-line basis over the shorter of the useful life or the remaining lease term. The leasehold improvement were subsequently fully impaired in the third quarter of 2023.
The following table summarizes operating lease costs included in operating expenses (in thousands):
|
Year Ended December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
Operating lease costs |
$ |
1,169 |
$ |
1,356 |
||||
|
Variable lease costs (1) |
679 |
737 |
||||||
|
Total lease cost |
$ |
1,848 |
$ |
2,093 |
||||
(1) Variable lease costs include certain additional charges for operating costs, including insurance, maintenance, taxes, utilities and other costs incurred, which are billed based on both usage and as a percentage of the Company’s share of total square footage. These costs also included costs associated with increases rent expense based on CPI.
|
14. |
Subsequent Event |
The accompanying consolidated financial statements reflect the 1-for-80 reverse split of the Company’s common stock that was approved by the Company’s Board of Directors and made effective on April 22, 2024. All share and per share information herein that relates to common stock prior to the effective date has been retroactively restated to reflect the reverse stock split.
FINANCIAL STATEMENTS (UNAUDITED)
Palvella Therapeutics, Inc.
For the Quarterly Period Ended June 30, 2024
|
Statements of Operations for the Three and Six Months Ended June 30, 2024 and 2023 |
||
|
Statements of Cash Flows for the Three Months Ended June 30, 2024 and 2023 |
||
PALVELLA THERAPEUTICS, INC.
(in thousands, except share and per share amounts)
|
June 30, 2024 |
December 31, 2023 |
|||||||
|
(unaudited) |
||||||||
|
Assets |
||||||||
|
Current assets: |
||||||||
|
Cash and cash equivalents |
$ | 14,537 | $ | 7,350 | ||||
|
Deferred transaction costs |
500 | — | ||||||
|
Prepaid expenses and other current assets |
307 | 198 | ||||||
|
Total current assets |
15,344 | 7,548 | ||||||
|
Total assets |
$ | 15,344 | $ | 7,548 | ||||
|
Liabilities and stockholders’ deficit |
||||||||
|
Current liabilities: |
||||||||
|
Accounts payable |
$ | 1,446 | $ | 936 | ||||
|
Accrued expenses and other current liabilities |
2,949 | 1,424 | ||||||
|
Total current liabilities |
4,395 | 2,360 | ||||||
|
Royalty agreement liability |
9,802 | 8,054 | ||||||
|
Derivative liabilities – royalty agreement |
1,343 | 1,014 | ||||||
|
Convertible notes payable |
10,029 | — | ||||||
|
Total liabilities |
25,569 | 11,428 | ||||||
|
Commitments and contingencies (Note 10) |
||||||||
|
Convertible preferred stock, $0.00001 par value; 20,655,895 shares authorized; 15,360,787 shares issued and outstanding at June 30, 2024 and December 31, 2023; aggregate liquidation value of $65,839 at June 30, 2024 |
70,603 | 70,603 | ||||||
|
Stockholders’ deficit: |
||||||||
|
Common stock, $0.00001 par value; 29,000,000 (25,500,000 voting and 3,500,000 non-voting) shares authorized; 5,720,009 (5,050,000 voting and 670,009 non-voting) shares issued and outstanding at June 30, 2024 and December 31, 2023 |
— | — | ||||||
|
Additional paid-in capital |
2,181 | 1,818 | ||||||
|
Accumulated deficit |
(83,009 | ) | (76,301 | ) | ||||
|
Total stockholders’ deficit |
(80,828 | ) | (74,483 | ) | ||||
|
Total liabilities, convertible preferred stock and stockholders’ deficit |
$ | 15,344 | $ | 7,548 | ||||
The accompanying notes are an integral part of these financial statements.
|
Three Months Ended June 30, |
Six Months Ended June 30, |
|||||||||||||||
|
2024 |
2023 |
2024 |
2023 |
|||||||||||||
|
Operating expenses: |
||||||||||||||||
|
Research and development |
$ | 1,442 | $ | 3,382 | $ | 2,426 | $ | 6,998 | ||||||||
|
General and administrative |
1,466 | 978 | 2,241 | 1,902 | ||||||||||||
|
Total operating expenses |
2,908 | 4,360 | 4,667 | 8,900 | ||||||||||||
|
Operating loss |
(2,908 | ) | (4,360 | ) | (4,667 | ) | (8,900 | ) | ||||||||
|
Other (expense) income: |
||||||||||||||||
|
Interest (expense) income - royalty agreement |
(942 | ) | 11,284 | (1,747 | ) | 8,705 | ||||||||||
|
Interest expense – convertible notes payable |
(29 | ) | — | (29 | ) | — | ||||||||||
|
Fair value adjustments on derivative liabilities - royalty agreement |
(271 | ) | 678 | (329 | ) | 593 | ||||||||||
|
Other (expense) income, net |
(22 | ) | 94 | 64 | 586 | |||||||||||
|
Net (loss) income |
$ | (4,172 | ) | $ | 7,696 | $ | (6,708 | ) | $ | 984 | ||||||
|
Less: Cumulative Series D preferred dividends |
— | (194 | ) | — | (388 | ) | ||||||||||
|
Net (loss) income attributable to common stockholders |
$ | (4,172 | ) | $ | 7,502 | $ | (6,708 | ) | $ | 596 | ||||||
|
Net (loss) income per share: |
||||||||||||||||
|
Basic and diluted |
$ | (0.76 | ) | $ | 0.31 | $ | (1.24 | ) | $ | 0.00 | ||||||
|
Weighted-average shares used in computing net (loss) income per share: |
||||||||||||||||
|
Basic |
5,720,009 | 5,720,009 | 5,720,009 | 5,720,009 | ||||||||||||
|
Diluted |
5,720,009 | 5,836,458 | 5,720,009 | 5,720,009 | ||||||||||||
The accompanying notes are an integral part of these financial statements.
|
Convertible |
Additional |
Total |
||||||||||||||||||||||||||
|
Preferred Stock |
Common Stock |
Paid-in |
Accumulated |
Stockholders' |
||||||||||||||||||||||||
|
Shares |
Amount |
Shares |
Amount |
Capital |
Deficit |
Deficit |
||||||||||||||||||||||
|
Balance at March 31, 2023 |
15,360,787 | $ | 70,603 | 5,720,009 | $ | — | $ | 1,356 | $ | (101,704 | ) | $ | (100,348 | ) | ||||||||||||||
|
Stock-based compensation |
— | — | — | — | 159 | — | 159 | |||||||||||||||||||||
|
Net income |
— | — | — | — | — | 7,696 | 7,696 | |||||||||||||||||||||
|
Balance at June 30, 2023 |
15,360,787 | $ | 70,603 | 5,720,009 | $ | — | $ | 1,515 | $ | (94,008 | ) | $ | (92,493 | ) | ||||||||||||||
|
Balance at March 31, 2024 |
15,360,787 | $ | 70,603 | 5,720,009 | $ | — | $ | 1,964 | $ | (78,837 | ) | $ | (76,873 | ) | ||||||||||||||
|
Stock-based compensation |
— | — | — | — | 217 | — | 217 | |||||||||||||||||||||
|
Net loss |
— | — | — | — | — | (4,172 | ) | (4,172 | ) | |||||||||||||||||||
|
Balance at June 30, 2024 |
15,360,787 | $ | 70,603 | 5,720,009 | $ | — | $ | 2,181 | $ | (83,009 | ) | $ | (80,828 | ) | ||||||||||||||
The accompanying notes are an integral part of these financial statements.
|
Convertible |
Additional |
Total |
||||||||||||||||||||||||||
|
Preferred Stock |
Common Stock |
Paid-in |
Accumulated |
Stockholders' |
||||||||||||||||||||||||
|
Shares |
Amount |
Shares |
Amount |
Capital |
Deficit |
Deficit |
||||||||||||||||||||||
|
Balance at January 1, 2023 |
15,360,787 | $ | 70,603 | 5,720,009 | $ | — | $ | 1,215 | $ | (94,992 | ) | $ | (93,777 | ) | ||||||||||||||
|
Stock-based compensation |
— | — | — | — | 300 | — | 300 | |||||||||||||||||||||
|
Net income |
— | — | — | — | — | 984 | 984 | |||||||||||||||||||||
|
Balance at June 30, 2023 |
15,360,787 | $ | 70,603 | 5,720,009 | $ | — | $ | 1,515 | $ | (94,008 | ) | $ | (92,493 | ) | ||||||||||||||
|
Balance at January 1, 2024 |
15,360,787 | $ | 70,603 | 5,720,009 | $ | — | $ | 1,818 | $ | (76,301 | ) | $ | (74,483 | ) | ||||||||||||||
|
Stock-based compensation |
— | — | — | — | 363 | — | 363 | |||||||||||||||||||||
|
Net loss |
— | — | — | — | — | (6,708 | ) | (6,708 | ) | |||||||||||||||||||
|
Balance at June 30, 2024 |
15,360,787 | $ | 70,603 | 5,720,009 | $ | — | $ | 2,181 | $ | (83,009 | ) | $ | (80,828 | ) | ||||||||||||||
The accompanying notes are an integral part of these financial statements.
|
Six Months Ended June 30, |
||||||||
|
2024 |
2023 |
|||||||
|
Cash flows from operating activities |
||||||||
|
Net (loss) income |
$ | (6,708 | ) | $ | 984 | |||
|
Adjustments to reconcile net loss to net cash used in operating activities: |
||||||||
|
Non-cash interest (income) expense – royalty agreement |
1,748 | (8,704 | ) | |||||
|
Non-cash interest expense – convertible note |
29 | — | ||||||
|
Change in fair value of derivative liabilities - royalty agreement |
329 | (593 | ) | |||||
|
Stock-based compensation |
363 | 300 | ||||||
|
Costs to issue convertible notes |
108 | — | ||||||
|
Change in operating assets and liabilities: |
||||||||
|
Prepaid expenses and other current assets |
(109 | ) | 250 | |||||
|
Accounts payable |
510 | 45 | ||||||
|
Accrued expenses and other current liabilities |
1,025 | (440 | ) | |||||
|
Net cash used in operating activities |
(2,705 | ) | (8,158 | ) | ||||
|
Cash flows from financing activities |
||||||||
|
Proceeds from the issuance of convertible notes payable |
10,000 | — | ||||||
|
Costs to issue convertible notes |
(108 | ) | — | |||||
|
Net cash provided by financing activities |
9,892 | — | ||||||
|
Net increase (decrease) in cash and cash equivalents |
7,187 | (8,158 | ) | |||||
|
Cash and cash equivalents at beginning of year |
7,350 | 16,053 | ||||||
|
Cash and cash equivalents at end of period |
$ | 14,537 | $ | 7,895 | ||||
|
Supplementary schedule of non-cash financing activities: |
||||||||
|
Deferred transaction costs, accrued but not paid |
$ | 500 | $ | — | ||||
The accompanying notes are an integral part of these financial statements.
1. Description of Business, Organization and Liquidity
Business Risks and Liquidity
Palvella Therapeutics, Inc. (the “Company”) is a late clinical-stage biopharmaceutical company committed to serving individuals suffering from serious, rare genetic skin diseases without approved therapies. The Company’s lead product candidate, QTORIN 3.9% rapamycin anhydrous gel (“QTORIN rapamycin”), is based on the Company’s patented QTORIN platform. QTORIN rapamycin is in clinical development for two rare genetic skin disorders. Since inception, the Company has devoted substantially all of its time to identifying, researching and conducting preclinical and clinical activities for its product candidates, acquiring and developing its platform technology, organizing and staffing the Company, business planning, raising capital and establishing its intellectual property portfolio. The Company’s principal executive offices are located in Wayne, Pennsylvania.
Liquidity
Since inception, the Company has incurred net losses and negative cash flows from operations. During the three and six months ended June 30, 2024, the Company reported net loss of $4.2 million and $6.7 million, respectively, and net cash used in operating activities of $2.8 million. At June 30, 2024, the Company had an accumulated deficit of $83.0 million.
The Company has financed its operations to date primarily through the sale of its convertible preferred stock, funding received under a royalty agreement, and entering into a convertible note purchase agreement that are convertible into the Company’s common stock based on certain conditions and events. $10 million of the convertible note purchase agreements has been issued as of June 30, 2024 and $12.4 million in convertible note purchase agreements has been issued in total through September 18, 2024. Management does not expect to generate commercial revenue or operating cash flows for at least the next several years. The Company’s ability to continue as a going concern in the near term is largely dependent on its ability to obtain additional sources of financing in order to fund operating expenses, complete development of its product candidates, obtain regulatory approvals, launch, and commercialize its product candidates, and continue research and development programs. The Company’s forecasted cash required to fund operations indicates that the Company does not have sufficient funds to support operations through the one-year period from the issuance date of these financial statements. Accordingly, there is substantial doubt about the Company’s ability to continue as a going concern within one year after the date that these financial statements are issued.
Management’s plans to address this going concern uncertainty include raising additional financing through public or private equity offerings, debt financings, collaborations and licensing arrangements, strategic transactions, or other sources to fund its operations; however, there can be no assurance that the Company will be able to obtain such funding on terms acceptable to the Company, on a timely basis or at all. The failure of the Company to obtain sufficient funds on acceptable terms when needed would have a material adverse effect on the Company’s business, results of operations and financial condition.
The accompanying financial statements have been prepared on a going-concern basis, which contemplates the realization of assets and satisfaction of liabilities in the normal course of business. The financial statements do not include any adjustments related to the recoverability and classification of recorded asset amounts or the amounts of classification of liabilities that might result from the outcome of this uncertainty.
2. Summary of Significant Accounting Policies
Basis of presentation
The accompanying unaudited financial statements have been prepared in conformity with accounting principles generally accepted in the United States (“GAAP”) for interim reporting. Any references in these notes to applicable guidance is meant to refer to GAAP as found in the Accounting Standards Codification (“ASC”) and Accounting Standards Updates (“ASU”) of the Financial Accounting Standards Board (“FASB”).
Accordingly, these interim Financial Statements do not include all disclosures required by U.S. GAAP for annual financial statements pursuant to the rules and regulations of the U.S. Securities and Exchange Commission, or SEC. In management’s opinion, the unaudited interim Financial Statements have been prepared on the same basis as the annual financial statements and include all adjustments, which include only normal recurring adjustments, necessary for the fair presentation of the Company’s financial position as of June 30, 2024, the Company’s results of operations for the three and six months ended June 30, 2024 and 2023, and cash flows for the six months ended June 30, 2024 and 2023. The results of operations for the three and six months ended June 30, 2024 are not necessarily indicative of the results to be expected for the full fiscal year or any other future interim or annual periods.
The information contained within the unaudited interim financial statements should be read in conjunction with the audited financial statements and accompanying notes as of and for the year ended December 31, 2023.
Use of estimates
The preparation of financial statements in accordance with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and reported amounts of revenues and expenses during the reporting period. Management considers many factors in selecting appropriate financial accounting policies and controls and in developing the estimates and assumptions that are used in the preparation of these financial statements. Management must apply significant judgment in this process and actual results could differ materially from those estimates.
Concentration of credit risk and other risks and uncertainties
Financial instruments that potentially expose the Company to concentrations of credit risk consist primarily of cash and cash equivalents. The Company holds all cash at two accredited financial institutions in amounts that exceed federally insured limits. The Company does not believe that it is subject to unusual credit risk beyond the normal credit risk associated with commercial banking relationships. The Company has not experienced any losses on its deposits of cash and cash equivalents.
The Company is dependent on contract manufacturing organizations (“CMOs”) to supply products for research and development of its product candidates, including pre-clinical and clinical studies, and for commercialization of its product candidates, if approved. The Company’s development programs could be adversely affected by any significant interruption in its CMOs’ operations or by a significant interruption in the supply of active pharmaceutical ingredients and other components.
Products developed by the Company require approval from the U.S. Food and Drug Administration (“FDA”) or other international regulatory agencies prior to commercial sales. There can be no assurance the Company’s product candidates will receive the necessary approvals. If the Company is denied approvals, approvals are delayed, or it is unable to maintain approvals received, such events could have a materially adverse impact on the Company.
Cash and cash equivalents
Cash and cash equivalents are held in accounts at two independent financial institutions. Cash equivalents are defined as money market funds with maturity from date of purchase of 90 days or less that are readily convertible into cash.
Fair value measurements
Fair value is defined as the exchange price that would be received for an asset or paid to transfer a liability in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants on the measurement date. Valuation techniques used to measure fair value must maximize the use of observable inputs and minimize the use of unobservable inputs. Financial assets and liabilities carried at fair value are to be classified and disclosed in one of the following three levels of the fair value hierarchy, of which the first two are considered observable and the last is considered unobservable:
|
● |
Level 1 – Quoted prices in active markets for identical assets or liabilities. |
|
● |
Level 2 – Observable inputs (other than Level 1 quoted prices), such as quoted prices in active markets for similar assets or liabilities, quoted prices in markets that are not active for identical or similar assets or liabilities, or other inputs that are observable or can be corroborated by observable market data. |
|
● |
Level 3 – Unobservable inputs that are supported by little or no market activity and that are significant to determining the fair value of the assets or liabilities, including pricing models, discounted cash flow methodologies, and similar techniques. |
At June 30, 2024 and December 31, 2023, the carrying amounts of financial instruments, which include cash and cash equivalents, accounts payable, and accrued expenses and other liabilities, approximate their fair value due to their short maturities. The Company records its derivative liabilities and convertible notes payable at fair value. At June 30, 2024 and December 31, 2023, the fair value of the royalty agreement liability, which is based on Level 3 inputs (including probability-weighted cash flow estimates of the Company’s potential future royalty payments and a weighted-average cost of capital of 24.0%) is approximately $10.7 million and $8.0 million, respectively.
Derivative instruments
The Company had milestone payments which may be required in connection with the royalty agreement (see Note 4) that were determined to be derivative liabilities. The valuation of the derivative liabilities is based on unobservable inputs and, therefore, represent Level 3 financial liabilities. The fair value of the derivative liabilities – royalty agreement was calculated using the present value of the potential payments using a weighted-average cost of capital and an assessment of the probability of the achievement of the milestones as well as an assessment of the timing of the potential milestone payments.
The derivative liabilities – royalty agreement was initially recorded at fair value, with gains and losses arising for changes in fair value of the derivative liabilities – royalty agreement recognized within the statements of operations as fair value adjustments on the derivative liabilities at each financial reporting period.
Convertible Notes
The fair value of the Convertible Notes was based on a probability-weighted expected return model ("PWERM"), which represents Level 3 measurements. The valuation utilized unobservable inputs, including estimates of the probability and riming of future commercialization of products not yet approved by the FDA or other regulatory agencies. Other significant assumptions include the discount rate, the fair value of our common stock, volatility, probability of the Convertible Notes being held to maturity, the probabilities of certain exit events, including a qualified financing, non-qualified financing, or corporate transaction.
As permitted under FASB ASC Topic 825, Financial Instruments (“ASC 825”), the Company elected the fair value option to account for its June 2024 convertible notes (collectively, the “Convertible Notes”). In accordance with ASC 825, the Company records these convertible notes at fair value with changes in fair value recorded in the Statement of Operations. As a result of applying the fair value option, direct costs and fees of $0.1 million related to the convertible notes were expensed as incurred and were not deferred. See Note 6.
Research and development expenses
Research and development costs are charged to expense as incurred. Research and development expenses include, among other costs, salaries and benefits of scientific personnel and the external cost of producing and testing the clinical material for clinical trials.
The Company has entered various research and development and clinical trial-related contracts. The Company defers and capitalizes prepaid nonrefundable advance research and development payments to third parties for goods and services to be used in future research and development activities and recognizes to research and development expense over the period that the research and development activities are performed or the services are provided. These agreements are generally cancelable, and related payments are recorded as research and development expenses as incurred. The Company records accruals for estimated ongoing research and clinical trial costs. When determining the accruals, at the end of a reporting period, the Company analyzes progress of its studies and clinical trials, including the phase or completion of events, invoices received and contracted costs. Actual results could differ from the Company’s estimates.
Stock-based compensation
The Company measures all stock options and other stock-based awards granted to employees, directors, consultants, and other nonemployees based on the fair value on the date of the grant and recognizes compensation expense of those awards over the requisite service period on a straight-line basis, which is generally the vesting period of the respective award. The Company recognizes forfeitures at the time forfeitures occur.
The Company classifies stock-based compensation expense in its statements of operations in the same way the payroll costs or service payments are classified for the related stock-based award recipient.
The fair value of each stock option grant is estimated on the date of grant using the Black-Scholes option pricing model (“Black-Scholes”). Black-Scholes requires inputs based on certain subjective assumptions, including (i) the expected stock price volatility, (ii) the expected term of the award, (iii) the risk-free interest rate and (iv) expected dividends. Due to the lack of a public market for the Company’s common stock and lack of company-specific historical and implied volatility data, the Company has based its computation of expected volatility on the historical volatility of a representative group of public companies with similar characteristics to the Company, including stage of product development and life science industry focus. The historical volatility is calculated based on a period of time commensurate with expected term assumption. The Company uses the simplified method to calculate the expected term for options granted whereby the expected term equals the arithmetic average of the vesting term and the original contractual term of the options due to its lack of sufficient historical data. The risk-free interest rate is based on U.S. Treasury securities with a maturity date commensurate with the expected term of the associated award. The expected dividend yield is assumed to be zero as the Company has never paid dividends and has no current plans to pay any dividends on its common stock.
Income taxes
The Company accounts for income taxes using the asset and liability method, which requires the recognition of deferred taxes using an asset and liability approach. The Company recognizes deferred tax assets and liabilities for the expected future tax consequences of events that have been included in the financial statements or tax returns. Deferred income tax assets and liabilities are determined based on differences between the financial statement reporting and tax bases of assets and liabilities and are measured using the enacted tax rates and laws that will be in effect when the differences are expected to reverse. Deferred income tax assets are reduced, as necessary, by a valuation allowance when management determines it is more likely than not that some or all the tax benefits will not be realized. The effect on deferred income tax assets and liabilities of a change in tax rates is recognized in the period that such tax rate changes are enacted.
The Company may recognize the tax benefit from an uncertain tax position only if it is more likely than not to be sustained upon examination based on the technical merits of the position. The amount of the accrued liability for which an exposure exists is measured as the largest amount of benefit determined on a cumulative probability basis that the Company believes is more likely than not to be realized upon ultimate settlement of the position. The Company has elected to treat interest and penalties, to the extent they arise, as a component of income taxes.
Related party transactions
The Company’s board of directors reviews and approves transactions with directors, officers, and holders of 5% or more of its voting securities and their affiliates, each a related party. The material facts as to the related party’s relationship or interest in the transaction are disclosed to its board of directors prior to their consideration of such transaction, and the transaction is not considered approved by its board of directors unless a majority of the directors who are not interested in the transaction approve the transaction.
Segments
Operating segments are defined as components of an entity for which separate financial information is available and that is regularly reviewed by the chief operating decision maker (“CODM”), in deciding how to allocate resources to an individual segment and in assessing performance. The Company’s CODM is its chief executive officer. The Company has determined it operates in one segment.
Net (loss) income per share
The Company’s convertible preferred stock are participating securities. Accordingly, in any period in which the Company reports net income, basic earnings per share is computed using the “two-class” method which includes the weighted- average number of shares of common stock outstanding during the period and other securities that participate in dividends (a participating security). During the periods where the Company incurs net losses, the Company allocates no loss to participating securities because these securities have no contractual obligation to share in the losses of the Company.
Basic net loss per share of common stock is computed by dividing net loss by the weighted-average number of shares of common stock outstanding during each period. Diluted net loss per share of common stock includes the effect, if any, from the potential exercise or conversion of securities, such as convertible preferred stock and stock options, which would result in the issuance of incremental shares of common stock. For diluted net loss per share, the weighted-average number of shares of common stock is the same for basic net loss per share due to the fact that when a net loss exists, dilutive securities are not included in the calculation as the impact is anti-dilutive. For the three and six months ended June 30, 2024 and 2023 basic and diluted net loss per share are the same.
Recently issued accounting standards
In December 2023, the FASB issued ASU No. 2023-09, Improvements to Income Tax Disclosures which requires disclosure of disaggregated income taxes paid, prescribes standard categories for the components of the effective tax rate reconciliation, and modifies other income tax-related disclosures. ASU No. 2023-09 is effective for fiscal years beginning after December 15, 2024 for public companies and December 15, 2025 for private companies and allows for adoption on a prospective basis, with a retrospective option. Early adoption is permitted. The Company is currently evaluating the impact of the ASU on the income tax disclosures within its financial statements.
In November 2023, the FASB issued ASU No. 2023-07, Improvements to Reportable Segment Disclosures which requires that a public entity provide additional segment disclosures on an interim and annual basis. The amendments in this ASU should be applied retrospectively to all prior periods presented in the financial statements, unless impracticable. Upon transition, the segment expense categories and amounts disclosed in the prior periods should be based on the significant segment expense categories identified and disclosed in the period of adoption. For public companies the ASU is effective for fiscal years beginning after December 15, 2023 and interim periods within fiscal years beginning after December 15, 2024. For private companies the ASU is effective for fiscal years starting after December 15, 2024, and interim periods within those fiscal periods. Early adoption is permitted. The Company is currently evaluating the impact of the adoption on the Company’s segment disclosures within its financial statements.
3. Fair Value Measurements
The following table presents information about the Company’s financial assets and liabilities measured at fair value on a recurring basis and indicate the level of the fair value hierarchy utilized to determine such fair values (in thousands):
|
June 30, 2024 |
||||||||||||||||
|
Level 1 |
Level 2 |
Level 3 |
Total |
|||||||||||||
|
Current assets: |
||||||||||||||||
|
Money market funds |
$ | 14,370 | $ | — | $ | — | $ | 14,370 | ||||||||
|
Total assets measured at fair value |
$ | 14,370 | $ | — | $ | — | $ | 14,370 | ||||||||
|
Liabilities: |
||||||||||||||||
|
Derivative liabilities - royalty agreement |
$ | — | $ | — | $ | 1,343 | $ | 1,343 | ||||||||
|
Convertible notes payable |
— | — | 10,029 | 10,029 | ||||||||||||
|
Total liabilities measured at fair value |
$ | — | $ | — | $ | 11,372 | $ | 11,372 | ||||||||
|
December 31, 2023 |
||||||||||||||||
|
Level 1 |
Level 2 |
Level 3 |
Total |
|||||||||||||
|
Current assets: |
||||||||||||||||
|
Money market funds |
$ | 7,203 | $ | — | $ | — | $ | 7,203 | ||||||||
|
Total assets measured at fair value |
$ | 7,203 | $ | — | $ | — | $ | 7,203 | ||||||||
|
Liabilities: |
||||||||||||||||
|
Derivative liabilities - royalty agreement |
$ | — | $ | — | $ | 1,014 | $ | 1,014 | ||||||||
|
Total liabilities measured at fair value |
$ | — | $ | — | $ | 1,014 | $ | 1,014 | ||||||||
Money market funds are highly liquid investments and are actively traded. The pricing information on the Company’s money market funds is based on quoted prices in active markets for identical securities. This approach results in a classification of these securities as Level 1 of the fair value hierarchy. Money market funds are cash equivalents and are included in cash and cash equivalents on the Company’s balance sheet as of June 30, 2024 and December 31, 2023.
The Company measures the Convertible Notes and warrant liabilities at fair value based on significant inputs not observable in the market, which causes them to be classified as a Level 3 measurement within the fair value hierarchy. These valuations use assumptions and estimates the Company believes would be made by a market participant in making the same valuation. The Company assesses these assumptions and estimates on an on-going basis as additional data impacting the assumptions and estimates are obtained. Changes in the fair value of the Convertible Notes and warrant liabilities related to updated assumptions and estimates are recognized within the Statements of Operations.
The fair value of the Convertible Notes and warrant liabilities may change significantly as additional data is obtained, impacting the Company’s assumptions regarding probabilities of outcomes used to estimate the fair value of the liabilities. The estimates of fair value may not be indicative of the amounts that could be realized in a current market exchange. Accordingly, the use of different market assumptions and/or different valuation techniques may have a material effect on the estimated fair value amounts, and such changes could materially impact the Company’s results of operations in future periods.
The key assumptions used to determine the fair value of the derivative liabilities – royalty agreement at June 30, 2024 and December 31, 2023 are as follows:
|
June 30, 2024 |
December 31, 2023 |
|||||||
|
Discount rate |
24.0 | % | 25.0 | % | ||||
|
Probability rate of achieving FDA approval of a product |
56.6 | % | 50.0 | % | ||||
|
Expected term to FDA regulatory approval of a product (in years) |
2.92 | 3.50 | ||||||
The following assumptions were used in determining the fair value of the Convertible Notes as of June 30, 2024:
|
June 30, 2024 |
||||
|
Risk-free interest rate |
5.2 | % | ||
|
Volatility |
76.4 | % | ||
|
Dividend yield |
0.0 | % | ||
|
Probability-weighted remaining term (years) |
0.8 | |||
|
Stock price |
$ | 3.67 | ||
The following tables provide reconciliations of the liabilities measured at fair value on a recurring basis using significant unobservable inputs (Level 3) at June 30, 2024 (in thousands):
|
Three Months Ended June 30, 2024 |
Six Months Ended June 30, 2024 |
|||||||
|
Balance, beginning of period |
$ | 1,072 | $ | 1,014 | ||||
|
Fair value adjustments |
271 | 329 | ||||||
|
Balance, end of the period |
$ | 1,343 | $ | 1,343 | ||||
Convertible Notes Payable
|
Three Months Ended June 30, 2024 |
Six Months Ended June 30, 2024 |
|||||||
|
Balance, beginning of period |
$ | - | $ | - | ||||
|
Initial fair value at issuance |
10,000 | 10,000 | ||||||
|
Accrued interest expense |
29 | 29 | ||||||
|
Fair value adjustments |
- | - | ||||||
|
Balance, end of the period |
$ | 10,029 | $ | 10,029 | ||||
Due to the issuance of the convertible notes in close proximity to the periods ended June 30, 2024, there was no change in fair value.
4. Strategic Agreements
Ligand Development Funding Agreement
In December 2018, the Company entered into the Ligand Agreement with Ligand, whereby Ligand agreed to make a one-time payment of $10.0 million to fund the development of QTORIN rapamycin. As partial consideration for the one-time payment, the Company granted Ligand the right to receive up to $8.0 million in milestone payments upon the achievement of certain corporate, financing and regulatory milestones by the Company related to QTORIN rapamycin for the treatment of any and all indications. In addition, the Company agreed to pay to Ligand tiered royalties from 5.0% to 9.8% based on any aggregate annual worldwide net product sales of any products based on QTORIN rapamycin. On a licensed product-by-licensed product and country-by-country basis, the royalty period is from the date of first commercial sale of such licensed product in a country until the latest of (i) the expiration of the last valid claim within the licensed patent rights covering such licensed product in the country in which such licensed product is made, used or sold, (ii) the expiration of the regulatory exclusivity term conferred by the applicable regulatory authority in such country with respect to such licensed product, and (iii) the fifteenth anniversary of the first commercial sale of such licensed product in such country. In certain circumstances, the Company has the right to reduce the royalty rates under the Ligand Agreement by making payments (“Royalty Buy Down Payments”). Specifically, once the Company has made royalty payments to Ligand equal to certain specified amounts in the mid eight figures, the Company has the option to make Royalty Buy Down Payments at any time during the remainder of the term of the Ligand Agreement to reduce its certain royalty tier percentages on annual worldwide net sales of any products by one or two percentage points. Such Royalty Buy Down Payments range in size from the low seven figures to the low eight figures.
Ligand may terminate the agreement for any or no reason upon a 90-day notice to the Company. Ligand may also terminate the agreement for cause in connection with a material breach that the Company does not cure within a certain period of time.
The total amount of potential future milestone payments remaining under the arrangement were $5.0 million as of June 30, 2024 and December 31, 2023. The potential future milestone payments represent derivative liabilities with a fair value of $1.3 million and $1.0 million as of June 30, 2024 and December 31, 2023, respectively, which are classified as derivative liabilities – royalty agreement on the accompanying balance sheets. See Note 3 for fair value measurements.
The Company’s obligation to pay tiered royalties under the Ligand Agreement was determined to be a debt instrument based on the likelihood of repaying the amounts provided to fund the development of QTORIN rapamycin and that the Company has significant continuing involvement in the generation of the cash flows potentially due to Ligand. This obligation is reflected as royalty agreement liability which is classified as a long-term liability on the accompanying balance sheets. Interest expense with respect to the royalty agreement liability is determined using the effective interest method based upon probability-adjusted cash flow estimates of the Company’s potential future royalty payments under the Ligand Agreement, yielding an effective interest rate of 39.9% and 20.2% for the three months ending June 30, 2024 and 2023, respectively. Changes in these estimates impact the amount of interest expense recognized through the accompanying statements of operations. During the second quarter 2023, the Company received data from certain of its clinical trials that reduced the projected net product sales related to QTORIN rapamycin and the corresponding probabilities of successful commercialization resulting in a significant reduction in the future royalty agreement liability. In the second quarter of 2024, the Company received data that adjusted the projected net product sales related to QTORIN rapamycin resulting an increase to the future royalty agreement liability. The Company incurred non-cash interest expense of $0.9 million for the three months ended June 30, 2024 and interest income of $11.3 million for the three months ended June 30, 2023, all of which is a component of the royalty agreement liability on the accompanying balance sheets.
In November 2023, the Ligand Agreement was amended (the “Amended Ligand Agreement”), whereby Ligand paid the Company an additional $5.0 million in return for an increase in the future tiered royalties to 8.0% to 9.8% of any aggregate annual worldwide net product sales of any products based on QTORIN rapamycin. The Royalty Buy Down Payments, and the associated rate modifications, in the original agreement were eliminated as part of the amendment. The Company determined that the original Ligand Agreement was extinguished with the newly Amended Ligand Agreement recorded at the estimated fair value of the royalty agreement liability on the date of the amendment. This resulted in a one-time, non-cash gain on extinguishment of approximately $23.1 million for the quarter ended December 31, 2023.
The Ligand Agreement requires the Company to make certain estimates and assumptions about the future development, FDA approval, commercialization, and net sales of any product containing QTORIN rapamycin. These estimates and assumptions are subject to significant variability and are thus subject to significant uncertainty. Therefore, these estimates and assumptions are likely to change as the Company develops and commercializes products containing QTORIN rapamycin that may result in future adjustments to the royalty agreement liability, the derivative liabilities, and the accretion of interest expense.
5. Accrued Expenses and Other Current Liabilities
Accrued expenses and other current liabilities consist of the following (in thousands):
|
June 30, |
December 31, |
|||||||
|
2024 |
2023 |
|||||||
|
Professional fees |
$ | 1,623 | $ | 960 | ||||
|
Compensation expense |
833 | 175 | ||||||
|
Research and development expenses |
262 | 120 | ||||||
|
Other |
231 | 169 | ||||||
|
Total accrued expenses and other current liabilities |
$ | 2,949 | $ | 1,424 | ||||
6. Convertible Notes Payable
To facilitate the ongoing operations of the Company, the Company entered into the following convertible promissory notes during the periods ended June 30, 2024:
|
Issuance Date |
Original Issuance Amount |
Maturity Date |
Interest |
Fair Value as of June 30, 2024 |
|||||||||
|
Note 1 |
6/6/2024 |
$ | 5,000,000 |
6/6/2027 |
SOFR + 2 |
% |
$ | 5,000,000 | |||||
|
Note 2 |
6/26/2024 |
2,500,000 |
6/6/2027 |
SOFR + 2 |
% |
2,500,000 | |||||||
|
Note 3 |
6/26/2024 |
2,500,000 |
6/6/2027 |
SOFR + 2 |
% |
2,500,000 | |||||||
| $ | 10,000,000 | $ | 10,000,000 | ||||||||||
Total interest expense incurred on the Convertible Notes during the six months ended June 30, 2024 totaled $29,000. As of and for the periods ended June 30, 2024, the interest rate for the Convertible Notes was 7.35%.
Upon a Qualified Financing, defined as either the earlier to occur of a) issuance of shares of preferred stock resulting in aggregate gross proceeds of at least $20,000,000 or b) an initial public offering, in each case on or before the maturity date, the principal and accrued interest on the Convertible Notes shall automatically convert into shares of the Company. In the case of the Qualified Financing being an issuance of preferred stock resulting in aggregate gross proceeds of at least $20,000,000, the Convertible Notes shall convert into shares of preferred stock having identical rights, privileges, preferences and restrictions as those issued to the investors in the Qualified Financing. In the case of the Qualified Financing being an initial public offering, the Convertible Notes shall convert into shares of common stock.
The Qualified Financing Conversion Price is equal to the lesser of (a) 80% of the price paid per share by the investors in the Qualified Financing or (b) the price per share as calculated by dividing $126,188,357 by the number of shares of common stock outstanding on an as-converted basis immediately prior to the Qualified Financing. If the Company consummates a reverse merger within 12 months of the issuance date, then the Qualified Financing Conversion Price shall be equal to the lowest cash price per share paid by the purchasers of the equity securities in connection with the private investment in public entity (the “PIPE”) or other related financing transaction consummated concurrently with the reverse merger.
Upon a Non-Qualified Financing, defined as any issuance of preferred stock to investors on or before the maturity date with the purpose of raising capital that does not meet the definition of a Qualified Financing, the holders of the Convertible Notes have the option to convert into shares of preferred stock having identical rights, privileges, preferences and restrictions as those issued to the investors in the Non-Qualified Financing.
The Non-Qualified Financing Conversion Price is equal to the lesser of (a) 80% of the price paid per share by the investors in the Non-Qualified Financing or (b) the price per share as calculated by dividing $126,188,357 by the number of shares of common stock outstanding on an as-converted basis immediately prior to the Qualified Financing.
Upon a Corporate Transaction, as defined as a) the closing of the sale, transfer, or other disposition of all or substantially all of the Company’s assets, b) the consummation of a merger with or into another entity (except for a reverse merger), or c) a liquidation or dissolution of the company, the holders will receive the greater of 1) 1.5 times the outstanding principal and accrued interest, 2) the amount the holders would have been entitled to receive had the outstanding principal and accrued interest been converted into shares of common stock at a price per share as calculated by dividing $126,188,357 by the number of shares outstanding on an as-converted basis immediately prior to the Corporate Transaction or 3) the amount the holders would have been entitled to receive had the outstanding principal and accrued interest been converted into shares of common stock immediately prior to the Corporate Transaction, at a price per share equal to 80% of the cash price per share paid or valued by the counterparty to the Company in a Corporate Transaction.
If the shares are neither repaid nor converted in connection with a Qualified Financing, Non-Qualified Financing, or Corporate Transaction, the outstanding principal and accrued interest of the Convertible Notes shall be due and payable within 30 days of the earlier of a) the date the Company receives approval of a new drug Application (NDA) by the United States Food and Drug Administration of QTORIN rapamycin b) June 6, 2027 or c) an event of default.
The Company has elected the fair value measurement option to account for the Convertible Notes. Under this method, changes in fair value are reported in the statement of operations. There were no changes in instrument-specific credit risk for the Notes for the periods ended June 30, 2024.
7. Convertible Preferred Stock
The Company amended and restated its certificate of incorporation (as amended, the “Amended Certificate”) such that it is authorized to issue 29,000,000 shares of common stock (25,500,000 voting and 3,500,000 non-voting) and 20,655,895 shares of preferred stock, with 2,241,903 shares designated as Series A-1 Convertible Preferred stock (Series A-1 Preferred”), 1,240,134 shares designated as Series A-2 Convertible Preferred stock (“Series A-2 Preferred”), 1,533,528 shares designated as Series B Convertible Preferred stock (“Series B Preferred”), 8,509,995 shares designated as Series C Convertible Preferred stock (“Series C Preferred”) and 7,130,335 shares designated as Series D Preferred.
The following table summarizes outstanding convertible preferred stock (in thousands, except share and per share amounts):
|
June 30, 2024 and December 31, 2023 |
June 30, 2024 |
|||||||||||||||
|
Issued and |
||||||||||||||||
|
Original Issue |
Authorized |
Outstanding |
Liquidation |
|||||||||||||
|
Price Per Share |
Shares |
Shares |
Preference |
|||||||||||||
|
Series A-1 Preferred |
$ | 1.31 | 2,241,903 | 2,241,903 | $ | 2,937 | ||||||||||
|
Series A-2 Preferred |
$ | 1.64 | 1,240,134 | 1,240,134 | 2,034 | |||||||||||
|
Series B Preferred |
$ | 3.19 | 1,533,528 | 1,533,528 | 4,892 | |||||||||||
|
Series C Preferred |
$ | 5.29 | 8,509,995 | 8,509,995 | 45,000 | |||||||||||
|
Series D Preferred |
$ | 5.29 | 7,130,335 | 1,835,227 | 10,976 | |||||||||||
| 20,655,895 | 15,360,787 | $ | 65,839 | |||||||||||||
The rights and preferences of the Series A‑1 Preferred, Series A‑2 Preferred, Series B Preferred, Series C Preferred and Series D Preferred, collectively Preferred Stock, under the Amended Certificate are as follows:
Dividends
The Series D Preferred holders, in preference to holders of any other series of the Company’s stock, are entitled to cumulative dividends in an amount in cash equal to 8% of the applicable Series D Preferred original issue price of $5.2879 per annum on each outstanding share of such Series D Preferred calculated from the date of issuance of such share, if and when declared by the Company’s board of directors. The Series C Preferred holders, in preference to holders of any other series of the Company’s stock other than the Series D Preferred, are entitled to non-cumulative dividends in an amount in cash equal to 8% of the applicable Series C Preferred original issue price of $5.2879 per annum on each outstanding share of such Series C Preferred calculated from the date of issuance of such share, if and when declared by the Company’s board of directors. The holders of Preferred Stock and Common Stock are entitled to participate in the distribution of the dividend as they would have received if all outstanding shares of Preferred Stock had been converted into common stock on the date of such event, after all holders of the Series D Preferred and the Series C Preferred have received such dividend in full. No dividends were declared or paid as of June 30, 2024. The Series D Preferred cumulative preferred stock dividends in arrears were approximately $1.3 million and $0.8 million as of June 30, 2024 and December 31, 2023, respectively.
Liquidation
In the event of any voluntary or involuntary liquidation, dissolution, or winding up of the Company, holders of the Series D Preferred shall be entitled to be paid out of the assets of the Company available for distribution to its stockholders, and in the event of a deemed liquidation event, as defined in the Amended Certificate, the holders of Series D Preferred then outstanding shall be entitled to be paid out of the consideration payable to stockholders in such deemed liquidation event or out of the available proceeds, as applicable, before any payment shall be made to the holders of the Series A-1 Preferred, Series A-2 Preferred, Series B Preferred, Series C Preferred and Common Stock by reason of their ownership thereof, an amount per share equal to the greater of (i) the applicable original issue price plus and accrued but unpaid cumulative dividends, or (ii) such amount per share as would have been payable had all shares of Series D Preferred been converted into Common Stock immediately prior to such liquidation, dissolution, winding up, or deemed liquidation event (“Series D Liquidation Amount”).
In the event of any voluntary or involuntary liquidation, dissolution, or winding up of the Company, holders of the Series C Preferred shall be entitled to be paid out of the assets of the Company available for distribution to its stockholders, and in the event of a deemed liquidation event, as defined in the Amended Certificate, the holders of Series C Preferred then outstanding shall be entitled to be paid out of the consideration payable to stockholders in such deemed liquidation event or out of the available proceeds, as applicable, before any payment shall be made to the holders of the Series A-1 Preferred, Series A-2 Preferred, Series B Preferred and Common Stock by reason of their ownership thereof, an amount per share equal to the greater of (i) the applicable original issue price, or (ii) such amount per share as would have been payable had all shares of Series C Preferred been converted into Common Stock immediately prior to such liquidation, dissolution, winding up, or deemed liquidation event (“Series C Liquidation Amount”).
In the event of any voluntary or involuntary liquidation, dissolution, or winding up of the Company, after payment in full of the Series D Liquidation Amount to the holders of Series D Preferred and the Series C Liquidation Amount to the holders of the Series C Preferred, holders of shares of Series A-1 Preferred, Series A-2 Preferred, and Series B Preferred, then outstanding shall be entitled to be paid out of the assets of the Company available for distribution to its stockholders, and in the event of a deemed liquidation event, as defined in the Amended Certificate, the holders of shares of Series A-1 Preferred, Series A-2 Preferred, and Series B Preferred then outstanding shall be entitled to be paid out of the consideration payable to stockholders in such deemed liquidation event or out of the available proceeds, as applicable, before any payment shall be made to the holders of Common Stock by reason of their ownership thereof, an amount per share equal to the greater of (i) the applicable original issue price, or (ii) such amount per share as would have been payable had all shares of Series A-1 Preferred, Series A-2 Preferred, and Series B Preferred, been converted into common stock immediately prior to such liquidation, dissolution, winding up, or deemed liquidation event. If upon any such liquidation, dissolution, or winding up of the Company or deemed liquidation event, the assets of the Company available for distribution to its stockholders shall be insufficient to pay the holders of shares of Series A-1 Preferred, Series A-2 Preferred, and Series B Preferred, the full amount to which they shall be entitled to the holders of shares of Series A-1 Preferred, Series A-2 Preferred, and Series B Preferred shall share ratably in any distribution of the assets available for distribution in proportion to the respective amounts which would otherwise be payable in respect of the shares held by them upon such distribution if all amounts payable on or with respect to such shares were paid in full.
Voting
The holders of outstanding shares of Preferred Stock are entitled to vote on all matters and shall be entitled to vote based on the number of shares of common stock into which each share of the preferred stock is convertible.
Redemption
Preferred Stock is not subject to mandatory redemption. The Preferred Stock is subject to redemption under certain deemed liquidation events not solely within the control of the Company, as defined, and as such are considered contingently redeemable for accounting purposes and are classified as temporary equity in the Company’s balance sheets. As a result, the Preferred Stock is not currently redeemable and the Company has determined that the Preferred Stock is not considered probable to become redeemable.
Conversion
Preferred Stock is convertible, at the option of the holder thereof, at any time and from time to time, and without the payment of additional consideration by the holder thereof, into such number of fully paid and nonassessable shares of Common Stock as is determined by dividing (a) the applicable original issue price by (b) the applicable conversion price in effect at the time of conversion.
Preferred Stock automatically converts upon the closing of a firm commitment underwritten initial public offering of common stock, in which the price per share is at least two times the Series D original issue price, subject to adjustment, resulting in gross proceeds of at least $50.0 million to the Company.
| Preferred Conversion Price to | ||||
| Preferred Stock | Common Stock | |||
|
Series A-1 Preferred |
$ | 1.31 | ||
|
Series A-2 Preferred |
$ | 1.64 | ||
|
Series B Preferred |
$ | 3.19 | ||
|
Series C Preferred |
$ | 5.29 | ||
|
Series D Preferred |
$ | 5.29 | ||
For Preferred Stock, the preferred conversion price and the rate at which applicable shares may be converted is subject to adjustment upon the occurrence of certain events. As of June 30, 2024 and December 31, 2023, the effective conversion ratio for all Preferred Stock is one for one.
8. 2019 Equity Incentive Plan
In March 2019, the Company adopted the 2019 Equity Incentive Plan (the “Plan”), which provides employees, consultants and advisors, and non-employee members of the Board of Directors and its affiliates with the opportunity to receive grants of incentive stock options, nonqualified stock options, and stock awards. In March 2024, the Company amended the 2019 Plan to include an additional 1,171,768 shares available for awards under the Plan. A total of 2,782,809 shares of the Company’s non-voting common stock may be issued for grants under the Plan. As of June 30, 2024, there were 2,149,138 options granted and 633,671 were available for grant.
For incentive stock options and non-statutory stock options, the option exercise price may not be less than 100% of the estimated fair value on the date of grant. Options granted to employees typically vest over a four-year period but may be granted with different vesting terms. The options expire ten years from the grant date.
A summary of activity under the Plan for the six months ended June 30, 2024 as follows:
|
Common |
Weighted |
Weighted |
|||||||||
|
Shares |
Average |
Average |
|||||||||
|
Underlying |
Exercise |
Remaining |
|||||||||
|
Stock |
Price per |
Contractual |
|||||||||
|
Options |
Share |
Life (Years) |
|||||||||
|
Outstanding at December 31, 2023 |
1,581,041 | $ | 2.72 | 7.3 | |||||||
|
Granted |
568,097 | 2.33 | |||||||||
|
Exercised |
— | ||||||||||
|
Forfeited / Cancelled |
— | ||||||||||
|
Outstanding at June 30, 2024 |
2,149,138 | $ | 2.62 | 7.6 | |||||||
|
Exercisable at June 30, 2024 |
1,184,898 | $ | 2.60 | 6.3 | |||||||
The aggregate intrinsic value for both options outstanding and options exercisable as of June 30, 2024 was $69,000.
During the three months ended June 30, 2024 and 2023, the Company recognized $217,000 and $158,000, respectively, of stock-based compensation expense, of which $60,000 and $14,000, respectively, was recorded as general and administrative expense and $157,000 and $144,000, respectively, was recorded as research and development expense in the accompanying statements of operations.
During the six months ended June 30, 2024 and 2023, the Company recognized $363,000 and $300,000, respectively, of stock-based compensation expense, of which $74,000 and $29,000, respectively, was recorded as general and administrative expense and $289,000 and $271,000, respectively, was recorded as research and development expense in the accompanying statements of operations.
As of June 30, 2024, there was approximately $1.8 million of unrecognized compensation cost related to non-vested stock options, which is expected to be recognized over a remaining weighted-average service period of 3.1 years.
The weighted average fair value of stock options granted during the six months ended June 30, 2024 was $1.68 per share which was estimated using the Black-Scholes option pricing model with the following weighted average assumptions:
|
Expected volatility |
81.91 | % | ||
|
Risk-free interest |
4.56 | % | ||
|
Expected term (years) |
5.92 | |||
|
Expected dividend yield |
— |
9. Income Taxes
The Company recorded no provision for income taxes for both the periods ended June 30, 2024 and 2023.
In assessing the realizability of the net deferred tax asset, the Company considers all relevant positive and negative evidence in determining whether it is more likely than not that some portion or all of the deferred income tax assets will not be realized. The realization of the gross deferred tax assets is dependent on several factors, including the generation of sufficient taxable income prior to the expiration of the NOLs. Management believes that it is more likely than not that the Company’s deferred income tax assets will not be realized. As such, there is a full valuation allowance against the net deferred tax assets as of June 30, 2024 and December 31, 2023.
The Company recognizes interest and penalties related to uncertain tax positions in income tax expense. As of June 30, 2024 and December 31, 2023, the Company reported no liabilities for unrecognized tax benefits along with no related interest and penalty exposure as accrued income tax on the accompanying balance sheets. Income tax returns for the tax years 2020 and later remain subject to examination by the taxing authority jurisdictions.
10. Commitments and Contingencies
Lease
The Company leases office space in Wayne, Pennsylvania, under a lease agreement, as amended, expiring on October 31, 2024 that had an initial term of less than 12 months. The minimum lease payments due under this lease are as follows as of June 30, 2024 (in thousands):
|
Year ended December 31, |
||||
| 2024 | $ | 27 | ||
|
Total future minimum payments |
$ | 27 |
Rent expense recorded during the three months ended June 30, 2024 and 2023 was $20,000. Rent expense recorded during the six months ended June 30, 2024, and 2023 was $40,000.
Litigation
Liabilities for loss contingencies arising from claims, assessments, ligation, fines, penalties, and other sources are recorded when it is probable that a liability has been incurred and the amount can be reasonably estimated. There are no matters currently outstanding.
11. Net Loss Per Share
Basic and diluted net loss per share is calculated as follows (in thousands, except share and per share amounts):
|
Three Months Ended June 30, |
Six Months Ended June 30, |
|||||||||||||||
|
2024 |
2023 |
2024 |
2023 |
|||||||||||||
|
Net income (loss) per share of common stock |
||||||||||||||||
|
Numerator: |
||||||||||||||||
|
Net loss |
$ | (4,172 | ) | $ | 7,696 | $ | (6,708 | ) | $ | 985 | ||||||
|
Less cumulative preferred Series D dividends |
(194 | ) | (194 | ) | (388 | ) | (388 | ) | ||||||||
|
Less non-cumulative preferred Series C dividends |
— | (900 | ) | — | (597 | ) | ||||||||||
|
Less: allocation of undistributed earnings to participating securities - preferred stock |
— | (4,811 | ) | — | — | |||||||||||
|
Net income (loss) available to common shareholders – basic and diluted |
$ | (4,366 | ) | 1,791 | $ | (7,096 | ) | — | ||||||||
|
Denominator: |
||||||||||||||||
|
Weighted-average number of shares outstanding used in computing net loss per share, basic |
5,720,009 | 5,720,009 | 5,720,009 | 5,720,009 | ||||||||||||
|
Effect of dilutive securities: |
||||||||||||||||
|
Stock options |
— | 116,449 | — | — | ||||||||||||
|
Denominator for diluted net income (loss) per share –adjusted weighted average shares |
5,720,009 | 5,836,458 | 5,720,009 | 5,720,009 | ||||||||||||
|
Net loss per share, basic |
$ | (0.76 | ) | $ | 0.31 | $ | (1.24 | ) | $ | 0.00 | ||||||
|
Net loss per share, diluted |
$ | (0.76 | ) | $ | 0.31 | $ | (1.24 | ) | $ | 0.00 | ||||||
The following potentially dilutive securities have been excluded from the calculation of diluted weighted-average shares of common stock outstanding, as they would be anti-dilutive:
|
Three Months Ended June 30, |
Six Months Ended June 30, |
|||||||||||||||
|
2024 |
2023 |
2024 |
2023 |
|||||||||||||
|
Convertible preferred stock |
15,360,787 | 15,360,787 | 15,360,787 | 15,360,787 | ||||||||||||
|
Stock options to purchase common stock |
2,149,138 | 1,179,732 | 2,149,138 | 1,611,041 | ||||||||||||
Amounts in the above table reflect the common stock equivalent.
12. Subsequent Events
On July 23, 2024, Pieris Pharmaceuticals, Inc., a Nevada corporation (“Pieris”), Polo Merger Sub, Inc., a Delaware corporation and a wholly-owned subsidiary of Pieris (“Merger Sub”), and the Company, entered into an Agreement and Plan of Merger (the “Merger Agreement”), pursuant to which the Merger Sub will merge with and into the Company, with the Company continuing as a wholly-owned subsidiary of Pieris and the surviving corporation of the merger (the “Merger”). The Merger is intended to qualify for federal income tax purposes as a tax-free reorganization and as a non-taxable exchange of shares of the Company’s capital stock for Pieris common stock.
Subject to the terms and conditions of the Merger Agreement (a) each then-outstanding share of the Company’s capital stock will be converted into the right to receive a number of shares of Pieris common stock (subject to the payment of cash in lieu of fractional shares) calculated in accordance with the Merger Agreement; and (b) each then-outstanding the Company’s stock option to purchase the Company’s common stock will be assumed by Pieris, subject to adjustment as set forth in the Merger Agreement.
It is currently anticipated that upon the closing of the Merger, pre-Merger Pieris stockholders will own approximately 18% of the combined company and pre-Merger Palvella stockholders will own approximately 82% of the combined company on a pro forma basis, based on the number of shares of Pieris common stock expected to be issued in connection with the Merger. The shares of the combined company purchased by the PIPE Investors in the PIPE Financing (as such terms are defined below) are not reflected in the foregoing percentages.
The provisions for calculating the conversion ratio “the Exchange Ratio” are set forth in the Merger Agreement and assume a valuation for the Company equal to $95.0 million and a valuation for Pieris equal to $21.0 million, subject to adjustment based on Pieris’ net cash as of the date immediately preceding the anticipated closing date, as set forth in the Merger Agreement. The Exchange Ratio is also based on the relative capitalizations and the total number of shares of capital stock of such company issued and outstanding immediately prior to the Merger for Pieris and the Company, among other provisions as further described in the Merger Agreement.
Consummation of the Merger is subject to certain closing conditions, including, among other things, (1) approval by Pieris stockholders of the Required Pieris Voting Proposals, (2) approval by the Company’s stockholders of the adoption of the Merger Agreement, and (3) Nasdaq’s approval of the listing of the shares of Pieris common stock to be issued in connection with the Merger.
The Merger Agreement contains certain termination rights of each of Pieris and the Company, including the right of each party to terminate the Merger Agreement to enter into a definitive agreement for a superior proposal. Upon termination of the Merger Agreement under specified circumstances, Pieris may be required to pay the Company a termination fee of $1.0 million and the Company may be required to pay Pieris a termination fee of $2.0 million.
In July and August 2024, the Company received an additional $2.4 million in exchange for a convertible promissory note as of the date of issuance of the financial statements. The Convertible Note bears an annual interest of 2.0% plus SOFR and shall be due and payable upon the earlier to occur of June 2027 or certain events defined in the Convertible Note. Under certain circumstances, the Convertible Note is convertible at the option of requisite holders into the Company’s equity securities at defined conversion prices.
Report of Independent Registered Public Accounting Firm
To the Shareholders and the Board of Directors of Palvella Therapeutics, Inc.
Opinion on the Financial Statements
We have audited the accompanying balance sheets of Palvella Therapeutics, Inc, (the Company) as of December 31, 2023 and 2022, and the related statements of operations, changes in convertible preferred stock and stockholders’ deficit, and cash flows for the years then ended, and the related notes (collectively referred to as the “financial statements”). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company at December 31, 2023 and 2022, and the results of its operations and its cash flows for the years then ended in conformity with U.S. generally accepted accounting principles.
The Company’s Ability to Continue as a Going Concern
The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 1 to the financial statements, the Company has suffered recurring net losses from operations, incurred negative cash flows from operations, and has stated that substantial doubt exists about the Company’s ability to continue as a going concern. Management's evaluation of the events and conditions and management’s plans regarding these matters are also described in Note 1. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB and in accordance with auditing standards generally accepted in the United States of America. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company's internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matters
The critical audit matters communicated below are matters arising from the current period audit of the financial statements that were communicated or required to be communicated to the audit committee and that: (1) relate to accounts or disclosures that are material to the financial statements and (2) involved our especially challenging, subjective or complex judgments. The communication of critical audit matters does not alter in any way our opinion on the consolidated financial statements, taken as a whole, and we are not, by communicating the critical audit matters below, providing separate opinions on the critical audit matters or on the accounts or disclosures to which they relate.
|
Royalty agreement liability and related interest income |
|||
|
Description of the Matter |
As described in Note 4 to the financial statements, the Company is party to a development funding agreement with Ligand Pharmaceuticals, Inc. (Ligand) (the Ligand Agreement). Pursuant to the Ligand Agreement, as partial consideration for the upfront payment received from Ligand, the Company agreed to pay to Ligand tiered future royalties based on any aggregate annual worldwide net product sales of any products based on QTORIN rapamycin. The Company recorded its obligation to pay tiered royalties under the Ligand Agreement as a debt instrument (royalty agreement liability) on the balance sheet at a carrying value of $8.1 million as of December 31, 2023 and has recognized imputed interest income of $6.3 million for the year ended December 31, 2023 using the effective interest rate method. The effective interest rate is estimated at each balance sheet date based on the rate that would enable the liability to be repaid in full over the anticipated life of the arrangement. The effective interest rate may vary during the term of the Ligand Agreement based on changes in the probability-adjusted cash flow estimates of the Company’s potential future royalty payments under the Ligand Agreement.
Auditing the interest income associated with the royalty agreement liability involved complex and subjective auditor judgment due to the estimation uncertainty involved in determining the probability-adjusted cash flow estimates of the Company’s potential future royalty payments. The Company’s effective interest rate calculation includes probability-adjusted revenue projections for which future royalties will be paid, which are sensitive to significant assumptions including the size of the addressable patient population, the anticipated pricing of the Company’s products, and the probability of successful development and commercialization, among others. |
||
|
How We Addressed the Matter in Our Audit |
To test the interest income associated with the royalty agreement liability, our audit procedures included, among others, testing the significant assumptions used to develop the estimates and evaluating the completeness and accuracy of the underlying data used by the Company in its effective interest rate calculation. For example, we compared the estimated size of the addressable patient population to a third-party prevalence study and government census data, and we compared the anticipated pricing information to a third-party market analysis. We compared the probability of achieving development and commercial success to studies published in medical journals evaluating clinical advancement and approval rates for similar products. We tested that the revenue projections were updated based on the most recent clinical trial data received in 2023 and recalculated the current year interest income. |
||
|
Fair value of royalty agreement liability recorded upon extinguishment |
|||
|
Description of the Matter |
As described in Note 3 and 4 to the financial statements, the Ligand Agreement was amended in November 2023 (the Amended Ligand Agreement), resulting in an extinguishment of the royalty agreement liability and recording of a new royalty agreement liability at its estimated fair value of $7.8 million on the date of the amendment. This resulted in a non-cash gain on extinguishment of approximately $23.1 million being recorded in the statement of operations during the year ended December 31, 2023. The Company estimated the fair value of the royalty agreement liability utilizing a Monte Carlo valuation model. Auditing the estimated fair value of the royalty agreement liability was complex and involved a high degree of subjectivity as the fair value is based on various inputs and assumptions, such as the net sales discount rate and the continuous counterparty discount rate. |
||
|
How We Addressed the Matter in Our Audit |
To test the estimated fair value of the royalty agreement liability, our audit procedures included, among others, testing the Monte Carlo valuation model and assessing the reasonableness of the significant assumptions used in the model. We involved valuation specialists to assess the appropriateness of the valuation model and to perform comparative calculations to corroborate the accuracy of the output from the Company’s model. With the assistance of our valuation specialists, we compared the key assumptions used in developing the discount rates to available market data and comparable company information. |
/s/ Ernst & Young LLP
We have served as the Company’s auditor since 2018.
Philadelphia, Pennsylvania
August 9, 2024
|
December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
Assets |
||||||||
|
Current assets: |
||||||||
|
Cash and cash equivalents |
$ | 7,350 | $ | 16,053 | ||||
|
Prepaid expenses and other current assets |
198 | 471 | ||||||
|
Total current assets |
7,548 | 16,524 | ||||||
|
Total assets |
$ | 7,548 | $ | 16,524 | ||||
|
Liabilities and stockholders’ deficit |
||||||||
|
Current liabilities: |
||||||||
|
Accounts payable |
$ | 936 | $ | 3,181 | ||||
|
Accrued expenses and other current liabilities |
1,424 | 2,601 | ||||||
|
Total current liabilities |
2,360 | 5,782 | ||||||
|
Royalty agreement liability |
8,054 | 32,417 | ||||||
|
Derivative liabilities – royalty agreement |
1,014 | 1,499 | ||||||
|
Total liabilities |
11,428 | 39,698 | ||||||
|
Commitments and contingencies (Note 9) |
||||||||
|
Convertible preferred stock, $0.00001 par value; 20,655,895 shares authorized; 15,360,787 shares issued and outstanding at December 31, 2023 and 2022; aggregate liquidation value of $65,377 at December 31, 2023 |
70,603 | 70,603 | ||||||
|
Stockholders’ deficit: |
||||||||
|
Common stock, $0.00001 par value; 29,000,000 (25,500,000 voting and 3,500,000 non-voting) shares authorized; 5,720,009 (5,050,000 voting and 670,009 non-voting) shares issued and outstanding at December 31, 2023 and 2022 |
— | — | ||||||
|
Additional paid-in capital |
1,818 | 1,215 | ||||||
|
Accumulated deficit |
(76,301 | ) | (94,992 | ) | ||||
|
Total stockholders’ deficit |
(74,483 | ) | (93,777 | ) | ||||
|
Total liabilities, convertible preferred stock and stockholders’ deficit |
$ | 7,548 | $ | 16,524 | ||||
The accompanying notes are an integral part of these financial statements.
|
Year Ended December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
Operating expenses: |
||||||||
|
Research and development |
$ | 8,793 | $ | 13,884 | ||||
|
General and administrative |
3,076 | 4,156 | ||||||
|
Total operating expenses |
11,869 | 18,040 | ||||||
|
Operating loss |
(11,869 | ) | (18,040 | ) | ||||
|
Other income (expense): |
||||||||
|
Interest income (expense) - royalty agreement |
6,265 | (10,364 | ) | |||||
|
Fair value adjustments on derivative liabilities - royalty agreement |
485 | (300 | ) | |||||
|
Gain on extinguishment – royalty agreement |
23,098 | — | ||||||
|
Other income, net |
712 | 126 | ||||||
|
Income (loss) before income taxes |
18,691 | (28,578 | ) | |||||
|
Income tax benefit |
— | 1,026 | ||||||
|
Net income (loss) |
$ | 18,691 | $ | (27,552 | ) | |||
|
Less: Cumulative Series D preferred dividends |
(776 | ) | — | |||||
|
Net income (loss) attributable to common stockholders |
17,915 | (27,552 | ) | |||||
|
Net income (loss) per share of common stock: |
||||||||
|
Basic |
$ | 0.68 | $ | (4.82 | ) | |||
|
Diluted |
$ | 0.67 | $ | (4.82 | ) | |||
|
Weighted-average shares used in computing net income (loss) per share of common stock: |
||||||||
|
Basic |
5,720,009 | 5,718,926 | ||||||
|
Diluted |
5,796,956 | 5,718,926 | ||||||
The accompanying notes are an integral part of these financial statements.
|
Convertible |
Additional |
Total |
||||||||||||||||||||||||||
|
Preferred Stock |
Common Stock |
Paid-in |
Accumulated |
Stockholders’ |
||||||||||||||||||||||||
|
Shares |
Amount |
Shares |
Amount |
Capital |
Deficit |
Deficit |
||||||||||||||||||||||
|
Balance at December 31, 2021 |
13,525,560 | $ | 61,071 | 5,708,030 | $ | — | $ | 754 | $ | (67,440 | ) | $ | (66,686 | ) | ||||||||||||||
|
Issuance of Series D preferred stock, net of issuance costs of $173 |
1,835,227 | 9,532 | — | — | — | — | — | |||||||||||||||||||||
|
Stock options exercised |
— | — | 11,979 | — | 34 | — | 34 | |||||||||||||||||||||
|
Stock-based compensation |
— | — | — | — | 427 | — | 427 | |||||||||||||||||||||
|
Net loss |
— | — | — | — | — | (27,552 | ) | (27,552 | ) | |||||||||||||||||||
|
Balance at December 31, 2022 |
15,360,787 | $ | 70,603 | 5,720,009 | $ | — | $ | 1,215 | $ | (94,992 | ) | $ | (93,777 | ) | ||||||||||||||
|
Stock-based compensation |
— | — | — | — | 603 | — | 603 | |||||||||||||||||||||
|
Net income |
— | — | — | — | — | 18,691 | 18,691 | |||||||||||||||||||||
|
Balance at December 31, 2023 |
15,360,787 | $ | 70,603 | 5,720,009 | $ | — | $ | 1,818 | $ | (76,301 | ) | $ | (74,483 | ) | ||||||||||||||
The accompanying notes are an integral part of these financial statements.
|
Year Ended December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
Cash flows from operating activities |
||||||||
|
Net income (loss) |
$ | 18,691 | $ | (27,552 | ) | |||
|
Adjustments to reconcile net loss to net cash used in operating activities: |
||||||||
|
Non-cash interest (income) expense – royalty agreement |
(6,265 | ) | 10,364 | |||||
|
Change in fair value of derivative liabilities – royalty agreement |
(485 | ) | 300 | |||||
|
Gain on extinguishment – royalty agreement |
(23,098 | ) | — | |||||
|
Stock-based compensation |
603 | 427 | ||||||
|
Change in operating assets and liabilities: |
||||||||
|
Prepaid expenses and other assets |
273 | 695 | ||||||
|
Accounts payable |
(2,245 | ) | 1,980 | |||||
|
Accrued expenses and other current liabilities |
(1,177 | ) | (1,054 | ) | ||||
|
Net cash used in operating activities |
(13,703 | ) | (14,840 | ) | ||||
|
Cash flows from financing activities |
||||||||
|
Proceeds from issuance of Series D preferred stock, net |
— | 9,532 | ||||||
|
Proceeds from amendment to royalty agreement |
5,000 | — | ||||||
|
Exercise of stock options |
— | 34 | ||||||
|
Net cash provided by financing activities |
5,000 | 9,566 | ||||||
|
Net decrease in cash and cash equivalents |
(8,703 | ) | (5,274 | ) | ||||
|
Cash and cash equivalents at beginning of year |
16,053 | 21,327 | ||||||
|
Cash and cash equivalents at end of year |
$ | 7,350 | $ | 16,053 | ||||
The accompanying notes are an integral part of these financial statements.
1. Description of Business, Organization and Liquidity
Business
Palvella Therapeutics, Inc. (the “Company”) is a late clinical-stage biopharmaceutical company committed to serving individuals suffering from serious, rare genetic skin diseases without approved therapies. The Company’s lead product candidate, QTORIN 3.9% rapamycin anhydrous gel (“QTORIN rapamycin”), is based on the Company’s patented QTORIN platform. QTORIN rapamycin is in clinical development for two rare genetic skin disorders. Since inception, the Company has devoted substantially all of its time to identifying, researching and conducting preclinical and clinical activities for its product candidates, acquiring and developing its platform technology, organizing and staffing the Company, business planning, raising capital and establishing its intellectual property portfolio. The Company’s principal executive offices are located in Wayne, Pennsylvania.
Liquidity
Since inception, the Company has incurred net losses and negative cash flows from operations. During the year ended December 31, 2023, the Company reported net income of $18.7 million due primarily to a $23.1 million non-cash gain on extinguishment of the royalty agreement (see Note 4) but has incurred net losses in all previous years. During the year ended December 31, 2023, the Company had net cash used in operating activities of $13.7 million and had an accumulated deficit of $76.3 million as of December 31, 2023.
The Company has financed its operations to date primarily through the sale of its convertible preferred stock, funding received under a royalty agreement, and entering into a convertible note purchase agreement under which the Company raised $11.9 million of gross proceeds from the issuance of convertible promissory notes through the issuance date of these financial statements that are convertible into the Company’s common stock based on certain conditions and events. Management does not expect to generate commercial revenue or operating cash flows for at least the next several years. The Company’s ability to continue as a going concern in the near term is largely dependent on its ability to obtain additional sources of financing in order to fund operating expenses, complete development of its product candidates, obtain regulatory approvals, launch, and commercialize its product candidates, and continue research and development programs. The Company’s forecasted cash required to fund operations indicates that the Company does not have sufficient funds to support operations through the one-year period from the issuance date of these financial statements. Accordingly, there is substantial doubt about the Company’s ability to continue as a going concern within one year after the date that these financial statements are issued.
Management’s plans to address this going concern uncertainty include raising additional financing through public or private equity offerings, debt financings, collaborations and licensing arrangements, strategic transactions, or other sources to fund its operations; however, there can be no assurance that the Company will be able to obtain such funding on terms acceptable to the Company, on a timely basis or at all. The failure of the Company to obtain sufficient funds on acceptable terms when needed would have a material adverse effect on the Company’s business, results of operations and financial condition.
The accompanying financial statements have been prepared on a going-concern basis, which contemplates the realization of assets and satisfaction of liabilities in the normal course of business. The financial statements do not include any adjustments related to the recoverability and classification of recorded asset amounts or the amounts of classification of liabilities that might result from the outcome of this uncertainty.
2. Summary of Significant Accounting Policies
Basis of presentation
The accompanying financial statements have been prepared in conformity with accounting principles generally accepted in the United States (“GAAP”). Any references in these notes to applicable guidance is meant to refer to GAAP as found in the Accounting Standards Codification (“ASC”) and Accounting Standards Updates (“ASU”) of the Financial Accounting Standards Board (“FASB”).
Use of estimates
The preparation of financial statements in accordance with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and reported amounts of revenues and expenses during the reporting period. Management considers many factors in selecting appropriate financial accounting policies and controls and in developing the estimates and assumptions that are used in the preparation of these financial statements. Management must apply significant judgment in this process and actual results could differ materially from those estimates.
Concentration of credit risk and other risks and uncertainties
Financial instruments that potentially expose the Company to concentrations of credit risk consist primarily of cash and cash equivalents. The Company holds all cash at two accredited financial institutions in amounts that exceed federally insured limits. The Company does not believe that it is subject to unusual credit risk beyond the normal credit risk associated with commercial banking relationships. The Company has not experienced any losses on its deposits of cash and cash equivalents.
The Company is dependent on contract manufacturing organizations (“CMOs”) to supply products for research and development of its product candidates, including pre-clinical and clinical studies, and for commercialization of its product candidates, if approved. The Company’s development programs could be adversely affected by any significant interruption in its CMOs’ operations or by a significant interruption in the supply of active pharmaceutical ingredients and other components.
Products developed by the Company require approval from the U.S. Food and Drug Administration (“FDA”) or other international regulatory agencies prior to commercial sales. There can be no assurance the Company’s product candidates will receive the necessary approvals. If the Company is denied approvals, approvals are delayed, or it is unable to maintain approvals received, such events could have a materially adverse impact on the Company.
Cash and cash equivalents
Cash and cash equivalents are held in accounts at two independent financial institutions. Cash equivalents are defined as all highly liquid investments with maturity from date of purchase of 90 days or less that are readily convertible into cash. Cash equivalents include money market funds that invest primarily in U.S. Treasury securities.
Fair value measurements
Fair value is defined as the exchange price that would be received for an asset or paid to transfer a liability in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants on the measurement date. Valuation techniques used to measure fair value must maximize the use of observable inputs and minimize the use of unobservable inputs. Financial assets and liabilities carried at fair value are to be classified and disclosed in one of the following three levels of the fair value hierarchy, of which the first two are considered observable and the last is considered unobservable:
|
● |
Level 1 – Quoted prices in active markets for identical assets or liabilities. |
|
● |
Level 2 – Observable inputs (other than Level 1 quoted prices), such as quoted prices in active markets for similar assets or liabilities, quoted prices in markets that are not active for identical or similar assets or liabilities, or other inputs that are observable or can be corroborated by observable market data. |
|
● |
Level 3 – Unobservable inputs that are supported by little or no market activity and that are significant to determining the fair value of the assets or liabilities, including pricing models, discounted cash flow methodologies, and similar techniques. |
At December 31, 2023 and 2022, the carrying amounts of financial instruments, which include cash and cash equivalents, accounts payable, and accrued expenses and other liabilities, approximate their fair value due to their short maturities. The Company records its derivative liabilities at fair value. At December 31, 2023, the fair value of the royalty agreement liability approximates its carrying value since the royalty agreement liability was remeasured at fair value in connection with the November 2023 amendment to its development funding and royalties agreement with Ligand Pharmaceuticals, Inc. ("Ligand”) (the “Ligand Agreement”) (see Note 4).
Derivative instruments
The Company has milestone payments which may be required in connection with the royalty agreement (see Note 4) that were determined to be derivative liabilities. The valuation of the derivative liabilities is based on unobservable inputs and, therefore, represent Level 3 financial liabilities. The fair value of the derivative liabilities – royalty agreement was calculated using the present value of the potential payments using a weighted-average cost of capital and an assessment of the probability of the achievement of the milestones as well as an assessment of the timing of the potential milestone payments.
The derivative liabilities – royalty agreement was initially recorded at fair value, with gains and losses arising for changes in fair value of the derivative liabilities – royalty agreement recognized within the statements of operations as fair value adjustments on the derivative liabilities at each financial reporting period.
Research and development expenses
Research and development costs are charged to expense as incurred. Research and development expenses include, among other costs, salaries and benefits of scientific personnel and the external cost of producing and testing the clinical material for clinical trials.
The Company has entered various research and development and clinical trial-related contracts. The Company defers and capitalizes prepaid nonrefundable advance research and development payments to third parties for goods and services to be used in future research and development activities and recognizes to research and development expense over the period that the research and development activities are performed or the services are provided. These agreements are generally cancelable, and related payments are recorded as research and development expenses as incurred. The Company records accruals for estimated ongoing research and clinical trial costs. When determining the accruals, at the end of a reporting period, the Company analyzes progress of its studies and clinical trials, including the phase or completion of events, invoices received and contracted costs. Actual results could differ from the Company’s estimates.
Stock-based compensation
The Company measures all stock options and other stock-based awards granted to employees, directors, consultants, and other nonemployees based on the fair value on the date of the grant and recognizes compensation expense of those awards over the requisite service period on a straight-line basis, which is generally the vesting period of the respective award. The Company recognizes forfeitures at the time forfeitures occur.
The Company classifies stock-based compensation expense in its statements of operations in the same way the payroll costs or service payments are classified for the related stock-based award recipient.
The fair value of each stock option grant is estimated on the date of grant using the Black-Scholes option pricing model (“Black-Scholes”). Black-Scholes requires inputs based on certain subjective assumptions, including (i) the expected stock price volatility, (ii) the expected term of the award, (iii) the risk-free interest rate and (iv) expected dividends. Due to the lack of a public market for the Company’s common stock and lack of company-specific historical and implied volatility data, the Company has based its computation of expected volatility on the historical volatility of a representative group of public companies with similar characteristics to the Company, including stage of product development and life science industry focus. The historical volatility is calculated based on a period of time commensurate with the expected term assumption. The Company uses the simplified method to calculate the expected term for options granted whereby the expected term equals the arithmetic average of the vesting term and the original contractual term of the options due to its lack of sufficient historical data. The risk-free interest rate is based on U.S. Treasury securities with a maturity date commensurate with the expected term of the associated award. The expected dividend yield is assumed to be zero as the Company has never paid dividends and has no current plans to pay any dividends on its common stock.
As the Company’s common stock has not been publicly traded, the Company periodically estimated the fair value of the Company’s common stock considering, among other things, contemporaneous valuations of its common stock prepared by an unrelated third-party valuation firm in accordance with the guidance provided by the American Institute of Certified Public Accountants (“AICPA”), Valuation of Privately-Held-Company Equity Securities Issued as Compensation. The Company’s stock valuations were prepared using either a hybrid method, where the equity value in one or more of the scenarios is calculated using an option-pricing method, or a probability-weighted expected return method, or PWERM, where the fair value of common stock is estimated based upon an analysis of future values for the Company, assuming various outcomes. Under the PWERM, the common stock value is based on the probability-weighted present value of expected future investment returns considering each of the possible outcomes available as well as the rights of each class of stock. The future value of the common stock under each outcome is discounted back to the valuation date at an appropriate risk-adjusted discount rate and probability weighted to arrive at an indication of value for the common stock. In addition to considering the results of these third-party valuations, the Company considered various objective and subjective factors to determine the price of its common stock as of each grant date, which may be as of a date later than the most recent third-party valuation date.
Income taxes
The Company accounts for income taxes using the asset and liability method, which requires the recognition of deferred taxes using an asset and liability approach. The Company recognizes deferred tax assets and liabilities for the expected future tax consequences of events that have been included in the financial statements or tax returns. Deferred income tax assets and liabilities are determined based on differences between the financial statement reporting and tax bases of assets and liabilities and are measured using the enacted tax rates and laws that will be in effect when the differences are expected to reverse. Deferred income tax assets are reduced, as necessary, by a valuation allowance when management determines it is more likely than not that some or all the tax benefits will not be realized. The effect on deferred income tax assets and liabilities of a change in tax rates is recognized in the period that such tax rate changes are enacted.
The Company may recognize the tax benefit from an uncertain tax position only if it is more likely than not to be sustained upon examination based on the technical merits of the position. The amount of the accrued liability for which an exposure exists is measured as the largest amount of benefit determined on a cumulative probability basis that the Company believes is more likely than not to be realized upon ultimate settlement of the position. The Company has elected to treat interest and penalties, to the extent they arise, as a component of income taxes.
Related party transactions
The Company’s board of directors reviews and approves transactions with directors, officers, and holders of 5% or more of its voting securities and their affiliates, each a related party. The material facts as to the related party’s relationship or interest in the transaction are disclosed to its board of directors prior to their consideration of such transaction, and the transaction is not considered approved by its board of directors unless a majority of the directors who are not interested in the transaction approve the transaction.
Segments
Operating segments are defined as components of an entity for which separate financial information is available and that is regularly reviewed by the chief operating decision maker (“CODM”), in deciding how to allocate resources to an individual segment and in assessing performance. The Company’s CODM is its chief executive officer. The Company has determined it operates in one segment.
Net income (loss) per share
The Company’s convertible preferred stock are participating securities. Accordingly, in any period in which the Company reports net income attributable to common stockholders, basic earnings per share is computed using the “two-class” method. Under this method, net income is reduced by any dividends earned and the remaining earnings (undistributed earnings) are allocated to common stock and each series of convertible preferred stock to the extent that each preferred security may share in earnings as if all of the earnings for the period had been distributed. The total earnings allocated to common stock is then divided by the number of outstanding shares to which the earnings are allocated to determine the earnings per share. The two-class method is not applicable during periods with a net loss, as the holders of the convertible preferred stock have no obligation to fund losses. Diluted net income (loss) per common share is computed under the two-class method by using the weighted-average number of shares of common stock outstanding, plus, for periods with net income attributable to common shareholders, the potential dilutive effects of outstanding stock options. In addition, the Company analyzes the potential dilutive effect of the outstanding convertible preferred stock under the “if-converted” method when calculating diluted earnings per share, in which it is assumes that the outstanding convertible preferred stock converts into common stock at the beginning of the period or when issued if later. The Company reports the more dilutive of the approaches (two class or “if-converted”) as their diluted net income per share during the period. For years in which a net loss exists, the weighted-average number of shares of common stock is the same for basic and diluted net loss per share due to the fact that when a net loss exists, dilutive securities are not included in the calculation as the impact is anti-dilutive. See Note 10 for further details on the Company’s net income (loss) per share calculations.
Recently adopted accounting standards
In June 2016, the FASB issued ASU 2016-13, Financial Instruments-Credit Losses: Measurement of Credit Losses on Financial Instruments (Topic 326) (“ASU 2016-13”). ASU 2016-13 requires measurement and recognition of expected credit losses for financial instruments based on historical experience, current conditions, and reasonable and supportable forecasts. ASU 2016-13 was adopted by the Company on January 1, 2023 and it did not have a material effect on its financial statements or related disclosures.
Recently issued accounting standards
In December 2023, the FASB issued ASU No. 2023-09, Improvements to Income Tax Disclosures which requires disclosure of disaggregated income taxes paid, prescribes standard categories for the components of the effective tax rate reconciliation, and modifies other income tax-related disclosures. ASU No. 2023-09 is effective for fiscal years beginning after December 15, 2024 for public companies and December 15, 2025 for private companies and allows for adoption on a prospective basis, with a retrospective option. Early adoption is permitted. The Company is currently evaluating the impact of the ASU on the income tax disclosures within its financial statements.
In November 2023, the FASB issued ASU No. 2023-07, Improvements to Reportable Segment Disclosures which requires that a public entity provide additional segment disclosures on an interim and annual basis. The amendments in this ASU should be applied retrospectively to all prior periods presented in the financial statements, unless impracticable. Upon transition, the segment expense categories and amounts disclosed in the prior periods should be based on the significant segment expense categories identified and disclosed in the period of adoption. For public companies the ASU is effective for fiscal years beginning after December 15, 2023 and interim periods within fiscal years beginning after December 15, 2024. For private companies the ASU is effective for fiscal years starting after December 15, 2024, and interim periods within those fiscal periods. Early adoption is permitted. The Company is currently evaluating the impact of the adoption on the Company’s segment disclosures within its financial statements.
3. Fair Value Measurements
The following table presents information about the Company’s financial assets and liabilities measured at fair value on a recurring basis and indicate the level of the fair value hierarchy utilized to determine such fair values (in thousands):
|
December 31, 2023 |
||||||||||||||||
|
Level 1 |
Level 2 |
Level 3 |
Total |
|||||||||||||
|
Current assets: |
||||||||||||||||
|
Money market funds |
$ | 7,203 | $ | — | $ | — | $ | 7,203 | ||||||||
|
Total assets measured at fair value |
$ | 7,203 | $ | — | $ | — | $ | 7,203 | ||||||||
|
Liabilities: |
||||||||||||||||
|
Derivative liabilities – royalty agreement |
$ | — | $ | — | $ | 1,014 | $ | 1,014 | ||||||||
|
Total liabilities measured at fair value |
$ | — | $ | — | $ | 1,014 | $ | 1,014 | ||||||||
|
December 31, 2022 |
||||||||||||||||
|
Level 1 |
Level 2 |
Level 3 |
Total |
|||||||||||||
|
Current assets: |
||||||||||||||||
|
Money market funds |
$ | 15,203 | $ | — | $ | — | $ | 15,203 | ||||||||
|
Total assets measured at fair value |
$ | 15,203 | $ | — | $ | — | $ | 15,203 | ||||||||
|
Liabilities: |
||||||||||||||||
|
Derivative liabilities – royalty agreement |
$ | — | $ | — | $ | 1,499 | $ | 1,499 | ||||||||
|
Total liabilities measured at fair value |
$ | — | $ | — | $ | 1,499 | $ | 1,499 | ||||||||
Money market funds are highly liquid investments and are actively traded. The pricing information on the Company’s money market funds is based on quoted prices in active markets for identical securities. This approach results in a classification of these securities as Level 1 of the fair value hierarchy. Money market funds are cash equivalents and are included in cash and cash equivalents on the Company’s balance sheet as of December 31, 2023 and 2022.
The key assumptions used to determine the fair value of the derivative liabilities – royalty agreement at December 31, 2023 and 2022 are as follows:
|
December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
Discount rate |
25.0 | % | 25.0 | % | ||||
|
Probability rate of achieving FDA approval of a product |
50 | % | 50 | % | ||||
|
Expected term to FDA regulatory approval of a product (in years) |
3.50 | 1.75 | ||||||
The following table provides a reconciliation of the liabilities measured at fair value on a recurring basis using significant unobservable inputs (Level 3) (in thousands):
|
2023 |
2022 |
|||||||
|
Derivative liabilities – royalty agreement |
||||||||
|
Balance at January 1 |
$ | 1,499 | $ | 1,199 | ||||
|
Fair value adjustments on derivative liabilities |
(485 | ) | 300 | |||||
|
Balance at December 31 |
$ | 1,014 | $ | 1,499 | ||||
The derivative liabilities – royalty agreement is classified as long term on the Company’s balance sheets according to the estimated timing of the occurrence of the potential payments.
As discussed in Note 4 below, the Amended Ligand Agreement was initially measured at its estimated fair value. This nonrecurring fair value measurement was based upon Level 3 inputs. The Company estimated the fair value of the royalty agreement liability on the amendment date using a Monte Carlo valuation model. The Company derived risk-adjusted quarterly net sales forecasts by applying a net sales discount rate of 21% to its forecasted net sales over the royalty term, and then simulated the risk-adjusted net sales using a Monte Carlo simulation. Based on the simulated net sales, the Company estimated the royalty payments within each simulation path based on the contractual royalty rates, and then present valued the royalty payments using a discount rate of 22%, which was based on its estimated discrete weighted average cost of capital of 24.5% as of the amendment date, adjusted to reflect the continuously compounded nature of the analysis. The Company then averaged across all simulation paths to derive the fair value of the royalty agreement liability on the date of amendment of $7.8 million.
4. Strategic Agreements
Ligand Development Funding Agreement
In December 2018, the Company entered into the Ligand Agreement with Ligand, whereby Ligand agreed to make a one-time payment of $10.0 million to fund the development of QTORIN rapamycin. As partial consideration for the one-time payment, the Company granted Ligand the right to receive up to $8.0 million in milestone payments upon the achievement of certain corporate, financing and regulatory milestones by the Company related to QTORIN rapamycin for the treatment of any and all indications. In addition, the Company agreed to pay to Ligand tiered royalties from 5.0% to 9.8% based on any aggregate annual worldwide net product sales of any products based on QTORIN rapamycin. On a licensed product-by-licensed product and country-by-country basis, the royalty period is from the date of first commercial sale of such licensed product in a country until the latest of (i) the expiration of the last valid claim within the licensed patent rights covering such licensed product in the country in which such licensed product is made, used or sold, (ii) the expiration of the regulatory exclusivity term conferred by the applicable regulatory authority in such country with respect to such licensed product, and (iii) the fifteenth anniversary of the first commercial sale of such licensed product in such country. In certain circumstances, the Company has the right to reduce the royalty rates under the Ligand Agreement by making payments (“Royalty Buy Down Payments”). Specifically, once the Company has made royalty payments to Ligand equal to certain specified amounts in the mid eight figures, the Company has the option to make Royalty Buy Down Payments at any time during the remainder of the term of the Ligand Agreement to reduce its certain royalty tier percentages on annual worldwide net sales of any products by one or two percentage points. Such Royalty Buy Down Payments range in size from the low seven figures to the low eight figures.
Ligand may terminate the agreement for any or no reason upon a 90-day notice to the Company. Ligand may also terminate the agreement for cause in connection with a material breach that the Company does not cure within a certain period of time.
The total amount of potential future milestone payments remaining under the arrangement were $5.0 million as of December 31, 2023 and 2022. The potential future milestone payments represent derivative liabilities with a fair value of $1.0 million and $1.5 million as of December 31, 2023 and 2022, respectively, which are classified as derivative liabilities – royalty agreement on the accompanying balance sheets. See Note 3 for fair value measurements.
The Company’s obligation to pay tiered royalties under the Ligand Agreement was determined to be a debt instrument based on the likelihood of repaying the amounts provided to fund the development of QTORIN rapamycin and that the Company has significant continuing involvement in the generation of the cash flows potentially due to Ligand. This obligation is reflected as royalty agreement liability which is classified as a long-term liability on the accompanying balance sheets. Interest expense with respect to the royalty agreement liability is determined using the effective interest method based upon probability-adjusted cash flow estimates of the Company’s potential future royalty payments under the Ligand Agreement, yielding an effective interest rate of 38.9% and 30.3% as of December 31, 2023 and 2022, respectively. The effective interest rate is estimated at each balance sheet date based on the rate that would enable the liability to be repaid in full over the anticipated life of the arrangement. Changes in these estimates impact the amount of interest expense recognized through the accompanying statements of operations. During the second quarter of 2023, the Company received data from certain of its clinical trials that reduced the projected net product sales related to QTORIN rapamycin and the corresponding probabilities of successful commercialization, resulting in a significant reduction in the expected future royalty payments and a corresponding reduction in the royalty agreement liability as of December 31, 2023. The Company incurred non-cash interest income (expense) of $6.3 million and ($10.4) million for the years ended December 31, 2023 and 2022, respectively, all of which is a component of the royalty agreement liability on the accompanying balance sheets.
In November 2023, the Ligand Agreement was amended (the “Amended Ligand Agreement”), whereby Ligand paid the Company an additional $5.0 million in return for an increase in the future tiered royalties to 8.0% to 9.8% of any aggregate annual worldwide net product sales of any products based on QTORIN rapamycin The Royalty Buy Down Payments, and the associated rate modifications, in the original agreement were eliminated as part of the amendment. The Amended Ligand Agreement also replaced the termination provision so that the agreement may be terminated by the earlier of a mutual written agreement of the parties or when the royalties contemplated by the agreement are paid to Ligand. The Company evaluated the accounting for the Amended Ligand Agreement under ASC 470, Debt, and concluded that the present value of the cash flows under the Amended Ligand Agreement differed by more than 10% from the present value of the cash flows under the original Ligand Agreement. As such, the Ligand Agreement was extinguished and the Amended Ligand Agreement was recorded at the estimated fair value of the royalty agreement liability on the date of the amendment. This resulted in a non-cash gain on extinguishment of approximately $23.1 million being recorded in the accompanying statement of operations related to the difference between the carrying value of the liability and its estimated fair value of $7.8 million on the date of amendment. See Note 3 for fair value measurements.
The Amended Ligand Agreement includes an option for Ligand to purchase additional product revenue participation rights from Palvella over a 10-year period. The option allows Ligand, for each product developed on the QTORIN platform that completes the first human clinical trial in the United States, the opportunity to make an upfront payment to Palvella (as set forth in the Amended Ligand Agreement) in return for a royalty rate (as set forth in the Amended Ligand Agreement). The Company determined that the option to purchase additional product revenue participation rights was not a freestanding financial instrument as it was not separately exercisable and legally detachable. The Company also determined that the option did not meet the definition of a derivative subject to bifurcation as it does not have a net settlement characteristic.
The Ligand Agreement requires the Company to make certain estimates and assumptions about the timing and probability of FDA approval and commercialization, and the amount of future net sales for any product containing QTORIN rapamycin. The estimated future net sales are based on subjective assumptions that include the estimated size of the addressable patient population and the anticipated pricing of the Company’s products. These estimates and assumptions are subject to significant variability and are thus subject to significant uncertainty. Therefore, these estimates and assumptions are likely to change as the Company develops and commercializes products containing QTORIN rapamycin that may result in significant future adjustments to the royalty agreement liability, the derivative liabilities, and the accretion of interest expense.
5. Accrued Expenses and Other Current Liabilities
Accrued expenses and other current liabilities consist of the following (in thousands):
|
December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
Professional fees |
$ | 960 | $ | 997 | ||||
|
Compensation expense |
175 | 762 | ||||||
|
Research and development expenses |
120 | 683 | ||||||
|
Other |
169 | 159 | ||||||
|
Total accrued expenses and other current liabilities |
$ | 1,424 | $ | 2,601 | ||||
6. Convertible Preferred Stock
In December 2022, the Company issued 1,835,227 shares of Series D Convertible Preferred Stock (“Series D Preferred”) at a price of $5.2879 per share. The Series D Preferred also contains a Milestone Closing option for additional shares to be issued following the Company’s receipt of clinical data for the top line Phase 3 data results for QTORIN rapamycin for pachyonychia congenita. Under the Milestone Closing, each Purchaser of the Series D Preferred shall have the right to purchase, and the Company agrees to sell and issue to each Purchaser at the Milestone Closing, up to that portion of 4,727,775 shares of Series D Preferred which equals the proportion that the number of shares of Series D Preferred then held by such Purchaser bears to the total number of shares of Series D Preferred outstanding immediately prior to the Milestone Closing, at a purchase price of $5.2879 per share. The Company determined that the future tranche right to purchase additional shares of Series D Preferred was not a freestanding financial instrument as it was not separately exercisable and legally detachable. The future tranche right was evaluated as an embedded derivative and was not bifurcated from the Series D Preferred shares since it did not have a net settlement characteristic and therefore did not meet the definition of a derivative. The future tranche right was cancelled in July 2024.
In connection with the issuance of the Series D Preferred, the Company amended and restated its certificate of incorporation (as amended, the “Amended Certificate”) such that it is authorized to issue 29,000,000 shares of common stock (25,500,000 voting and 3,500,000 non-voting) and 20,655,895 shares of preferred stock, with 2,241,903 shares designated as Series A-1 Convertible Preferred stock (Series A-1 Preferred”), 1,240,134 shares designated as Series A-2 Convertible Preferred stock (“Series A-2 Preferred”), 1,533,528 shares designated as Series B Convertible Preferred stock (“Series B Preferred”), 8,509,995 shares designated as Series C Convertible Preferred stock (“Series C Preferred”) and 7,130,335 shares designated as Series D Preferred.
The following tables summarize outstanding convertible preferred stock (in thousands, except share and per share amounts):
|
December 31, 2023 and 2022 |
December 31, 2023 |
|||||||||||||||
|
Issued and |
||||||||||||||||
|
Original Issue |
Authorized |
Outstanding |
Liquidation |
|||||||||||||
|
Price Per Share |
Shares |
Shares |
Preference |
|||||||||||||
|
Series A-1 Preferred |
$ | 1.31 | 2,241,903 | 2,241,903 | $ | 2,937 | ||||||||||
|
Series A-2 Preferred |
$ | 1.64 | 1,240,134 | 1,240,134 | 2,034 | |||||||||||
|
Series B Preferred |
$ | 3.19 | 1,533,528 | 1,533,528 | 4,892 | |||||||||||
|
Series C Preferred |
$ | 5.29 | 8,509,995 | 8,509,995 | 45,000 | |||||||||||
|
Series D Preferred |
$ | 5.29 | 7,130,335 | 1,835,227 | 10,514 | |||||||||||
| 20,655,895 | 15,360,787 | $ | 65,377 | |||||||||||||
The rights and preferences of the Series A‑1 Preferred, Series A‑2 Preferred, Series B Preferred, Series C Preferred and Series D Preferred, collectively Preferred Stock, under the Amended Certificate are as follows:
Dividends
The Series D Preferred holders, in preference to holders of any other series of the Company’s stock, are entitled to cumulative dividends in an amount in cash equal to 8% of the applicable Series D Preferred original issue price of $5.2879 per annum on each outstanding share of such Series D Preferred calculated from the date of issuance of such share, if and when declared by the Company’s board of directors. The Series C Preferred holders, in preference to holders of any other series of the Company’s stock other than the Series D Preferred, are entitled to non-cumulative dividends in an amount in cash equal to 8% of the applicable Series C Preferred original issue price of $5.2879 per annum on each outstanding share of such Series C Preferred calculated from the date of issuance of such share, if and when declared by the Company’s board of directors. The holders of Preferred Stock and Common Stock are entitled to participate in the distribution of the dividend as they would have received if all outstanding shares of Preferred Stock had been converted into common stock on the date of such event, after all holders of the Series D Preferred and the Series C Preferred have received such dividend in full. No dividends were declared or paid as of December 31, 2023. The Series D Preferred cumulative preferred stock dividends in arrears were approximately $0.8 million as of December 31, 2023 and were de minimis as of December 31, 2022.
Liquidation
In the event of any voluntary or involuntary liquidation, dissolution, or winding up of the Company, holders of the Series D Preferred shall be entitled to be paid out of the assets of the Company available for distribution to its stockholders, and in the event of a deemed liquidation event, as defined in the Amended Certificate, the holders of Series D Preferred then outstanding shall be entitled to be paid out of the consideration payable to stockholders in such deemed liquidation event or out of the available proceeds, as applicable, before any payment shall be made to the holders of the Series A-1 Preferred, Series A-2 Preferred, Series B Preferred, Series C Preferred and Common Stock by reason of their ownership thereof, an amount per share equal to the greater of (i) the applicable original issue price plus and accrued but unpaid cumulative dividends, or (ii) such amount per share as would have been payable had all shares of Series D Preferred been converted into Common Stock immediately prior to such liquidation, dissolution, winding up, or deemed liquidation event (“Series D Liquidation Amount”).
In the event of any voluntary or involuntary liquidation, dissolution, or winding up of the Company, holders of the Series C Preferred shall be entitled to be paid out of the assets of the Company available for distribution to its stockholders, and in the event of a deemed liquidation event, as defined in the Amended Certificate, the holders of Series C Preferred then outstanding shall be entitled to be paid out of the consideration payable to stockholders in such deemed liquidation event or out of the available proceeds, as applicable, before any payment shall be made to the holders of the Series A-1 Preferred, Series A-2 Preferred, Series B Preferred and Common Stock by reason of their ownership thereof, an amount per share equal to the greater of (i) the applicable original issue price, or (ii) such amount per share as would have been payable had all shares of Series C Preferred been converted into Common Stock immediately prior to such liquidation, dissolution, winding up, or deemed liquidation event (“Series C Liquidation Amount”).
In the event of any voluntary or involuntary liquidation, dissolution, or winding up of the Company, after payment in full of the Series D Liquidation Amount to the holders of Series D Preferred and the Series C Liquidation Amount to the holders of the Series C Preferred, holders of shares of Series A-1 Preferred, Series A-2 Preferred, and Series B Preferred, then outstanding shall be entitled to be paid out of the assets of the Company available for distribution to its stockholders, and in the event of a deemed liquidation event, as defined in the Amended Certificate, the holders of shares of Series A-1 Preferred, Series A-2 Preferred, and Series B Preferred then outstanding shall be entitled to be paid out of the consideration payable to stockholders in such deemed liquidation event or out of the available proceeds, as applicable, before any payment shall be made to the holders of Common Stock by reason of their ownership thereof, an amount per share equal to the greater of (i) the applicable original issue price, or (ii) such amount per share as would have been payable had all shares of Series A-1 Preferred, Series A-2 Preferred, and Series B Preferred, been converted into common stock immediately prior to such liquidation, dissolution, winding up, or deemed liquidation event. If upon any such liquidation, dissolution, or winding up of the Company or deemed liquidation event, the assets of the Company available for distribution to its stockholders shall be insufficient to pay the holders of shares of Series A-1 Preferred, Series A-2 Preferred, and Series B Preferred, the full amount to which they shall be entitled to the holders of shares of Series A-1 Preferred, Series A-2 Preferred, and Series B Preferred shall share ratably in any distribution of the assets available for distribution in proportion to the respective amounts which would otherwise be payable in respect of the shares held by them upon such distribution if all amounts payable on or with respect to such shares were paid in full.
Voting
The holders of outstanding shares of Preferred Stock are entitled to vote on all matters and shall be entitled to vote based on the number of shares of common stock into which each share of the preferred stock is convertible.
Redemption
Preferred Stock is not subject to mandatory redemption. The Preferred Stock is subject to redemption under certain deemed liquidation events not solely within the control of the Company, as defined, and as such are considered contingently redeemable for accounting purposes and are classified as temporary equity in the Company’s balance sheets. The Preferred Stock is not considered probable to become redeemable as no deemed liquidation events are expected to occur.
Conversion
Preferred Stock is convertible, at the option of the holder thereof, at any time and from time to time, and without the payment of additional consideration by the holder thereof, into such number of fully paid and nonassessable shares of Common Stock as is determined by dividing (a) the applicable original issue price by (b) the applicable conversion price in effect at the time of conversion.
Preferred Stock automatically converts upon the closing of a firm commitment underwritten initial public offering of common stock, in which the price per share is at least two times the Series D original issue price, subject to adjustment, resulting in gross proceeds of at least $50.0 million to the Company.
| Preferred Conversion Price to | ||||
| Preferred Stock | Common Stock | |||
|
Series A-1 Preferred |
$ | 1.31 | ||
|
Series A-2 Preferred |
$ | 1.64 | ||
|
Series B Preferred |
$ | 3.19 | ||
|
Series C Preferred |
$ | 5.29 | ||
|
Series D Preferred |
$ | 5.29 | ||
For Preferred Stock, the preferred conversion price and the rate at which applicable shares may be converted is subject to adjustment upon the occurrence of certain events. As of December 31, 2023 and 2022, the effective conversion ratio for all Preferred Stock is one for one.
7. 2019 Equity Incentive Plan
In March 2019, the Company adopted the 2019 Equity Incentive Plan (the “Plan”), which provides employees, consultants and advisors, and non-employee members of the Board of Directors and its affiliates with the opportunity to receive grants of incentive stock options, nonqualified stock options, and stock awards. In May 2020, the Company amended the 2019 Plan to include an additional 1,000,000 shares available for awards under the Plan. A total of 1,690,000 shares of the Company’s non-voting common stock may be issued for the grants under the Plan. As of December 31, 2023, there were 30,000 shares available for future grants under the Plan.
For incentive stock options and non-statutory stock options, the option exercise price may not be less than 100% of the estimated fair value on the date of grant. Options granted to employees typically vest over a four-year period but may be granted with different vesting terms. The options expire ten years from the grant date.
A summary of activity under the Plan for the year ended December 31, 2023 as follows:
|
Common |
Weighted |
Weighted |
||||||||||
|
Shares |
Average |
Average |
||||||||||
|
Underlying |
Exercise |
Remaining |
||||||||||
|
Stock |
Price per |
Contractual |
||||||||||
|
Options |
Share |
Life (Years) |
||||||||||
|
Outstanding at January 1, 2023 |
1,024,628 | $ | 2.53 | 7.1 | ||||||||
|
Granted |
608,388 | 3.03 | ||||||||||
|
Exercised |
— | |||||||||||
|
Forfeited / Cancelled |
(51,975 | ) | 2.64 | |||||||||
|
Outstanding at December 31, 2023 |
1,581,041 | $ | 2.72 | 7.3 | ||||||||
|
Exercisable at December 31, 2023 |
1,009,011 | $ | 2.56 | 6.5 | ||||||||
The aggregate intrinsic value of options outstanding and options exercisable as of December 31, 2023 was $494,000 and $470,000, respectively.
During the year ended December 31, 2023 and 2022, the Company recognized $603,000 and $427,000, respectively, of stock-based compensation expense, of which $55,000 and $80,000, respectively, was recorded as general and administrative expense and $548,000 and $347,000, respectively, was recorded as research and development expense in the accompanying statements of operations. As of December 31, 2023, there was approximately $1.2 million of unrecognized compensation cost related to non-vested stock options, which is expected to be recognized over a remaining weighted-average service period of 2.7 years.
The weighted average fair value of stock options granted during the year ended December 31, 2023 was $2.16 per share which was estimated using the Black-Scholes option pricing model with the following weighted average assumptions:
|
Expected volatility |
80.14% | - | 81.01 | % | ||
|
Risk-free interest |
4.05% | - | 4.08 | % | ||
|
Expected term (years) |
5.4 | - | 6.0 | |||
|
Expected dividend yield |
— |
There were no stock options granted during the year ended December 31, 2022.
8. Income Taxes
The Company had no provision for income taxes for the year ended December 31, 2023. The Company recorded a benefit for income taxes of $1.0 million for the year ended December 31, 2022 which consisted of approximately $0.1 million of current federal tax benefit and $0.9 million of current state tax benefit. The 2022 tax benefit is attributed to the reversal of the Company's uncertain tax position due to the lapse of the 2018 Pennsylvania statute of limitations concerning the timing of the initial upfront payment received under the Ligand Agreement.
A reconciliation of the statutory U.S. federal income tax rate to the Company’s effective tax rate is as follows:
|
December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
U.S. federal statutory rate |
21.0 | % | 21.0 | % | ||||
|
Uncertain tax position and interest |
0.0 | 3.8 | ||||||
|
State and local taxes |
4.0 | (3.9 | ) | |||||
|
Permanent items |
0.3 | (1.0 | ) | |||||
|
Change in valuation allowance |
(25.4 | ) | (16.3 | ) | ||||
|
Other |
0.1 | 0.0 | ||||||
|
Effective income tax rate |
0.0 | % | 3.6 | % | ||||
The tax effects of temporary differences that gave rise to significant portions of the deferred tax assets were as follows (in thousands):
| December 31, | ||||||||
|
2023 |
2022 |
|||||||
|
Deferred tax assets: |
||||||||
|
Royalty agreement liabilities |
$ | 1,587 | $ | 9,080 | ||||
|
Net operating loss |
9,179 | 7,850 | ||||||
|
Section 174 R&D capitalization |
4,548 | 3,152 | ||||||
|
Accrued expenses and other |
406 | 364 | ||||||
|
Orphan drug credit |
199 | 199 | ||||||
|
Startup costs |
95 | 104 | ||||||
|
Net deferred tax assets |
16,014 | 20,749 | ||||||
|
Valuation allowance |
(16,014 | ) | (20,749 | ) | ||||
|
Net deferred tax assets |
$ | — | $ | — | ||||
|
Decrease (increase) in valuation allowance |
$ | 4,735 | $ | (4,637 | ) | |||
In assessing the realizability of the net deferred tax asset, the Company considers all relevant positive and negative evidence in determining whether it is more likely than not that some portion or all of the deferred income tax assets will not be realized. The realization of the gross deferred tax assets is dependent on several factors, including the generation of sufficient taxable income prior to the expiration of the NOLs. Management believes that it is more likely than not that the Company’s deferred income tax assets will not be realized. As such, there is a full valuation allowance in the amount of $16.0 million and $20.7 million against the net deferred tax assets as of December 31, 2023 and 2022, respectively.
The Company recognizes interest and penalties related to uncertain tax positions in income tax expense. As of December 31, 2023, the Company reported no liabilities for unrecognized tax benefits along with no related interest and penalty exposure as accrued income tax on the accompanying balance sheets. Income tax returns for the tax years 2019 through 2023 remain subject to examination by the taxing authority jurisdictions.
At December 31, 2023, the Company had NOLs for federal income tax purposes of $36.7 million, which are available to reduce future federal taxable income and have an indefinite carryforward. The Company has NOLs for state income tax purposes of $37.6 million, which are available to reduce future state taxable income through 2038. In addition, the Company has orphan drug credits of $0.2 million to reduce future federal taxes through 2039.
9. Commitments and Contingencies
Lease
The Company leases office space in Wayne, Pennsylvania, under a lease agreement, as amended, expiring on October 31, 2024 that had an initial term of less than 12 months. The minimum lease payments due under this lease are as follows as of December 31, 2023 (in thousands):
|
Year ended December 31, |
||||
|
2024 |
$ | 67 | ||
|
Total future minimum payments |
$ | 67 |
Rent expense recorded during the years ended December 31, 2023 and 2022 was $80,000.
Litigation
Liabilities for loss contingencies arising from claims, assessments, ligation, fines, penalties, and other sources are recorded when it is probable that a liability has been incurred and the amount can be reasonably estimated. There are no matters currently outstanding.
10. Net Income (Loss) Per Share
Basic and diluted net income (loss) per share is calculated as follows (in thousands, except share and per share amounts):
|
Year Ended December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
Net income (loss) per share of common stock |
||||||||
|
Numerator: |
||||||||
|
Net income (loss) attributable to common stockholders |
$ | 17,915 | $ | (27,552 | ) | |||
|
Less: non-cumulative preferred Series C dividends |
(3,600 | ) | — | |||||
|
Less: allocation of undistributed earnings to participating securities - preferred stock |
(10,431 | ) | — | |||||
|
Net income (loss) attributable to common stockholders – basic and diluted |
$ | 3,884 | $ | (27,552 | ) | |||
|
Denominator: |
||||||||
|
Weighted-average number of shares outstanding used in computing net income (loss) per share - basic |
5,720,009 | 5,718,926 | ||||||
|
Effect of dilutive securities: |
||||||||
|
Stock options |
76,947 | — | ||||||
|
Denominator for diluted net income (loss) per share – adjusted weighted average shares |
5,796,956 | 5,718,926 | ||||||
|
Net income (loss) per share, basic |
$ | 0.68 | $ | (4.82 | ) | |||
|
Net income (loss) per share, diluted |
$ | 0.67 | $ | (4.82 | ) | |||
The following potentially dilutive securities have been excluded from the calculation of diluted weighted-average shares of common stock outstanding, as they would be anti-dilutive:
|
December 31, |
||||||||
|
2023 |
2022 |
|||||||
|
Convertible preferred stock |
15,360,787 | 15,360,787 | ||||||
|
Stock options to purchase common stock |
1,119,732 | 1,024,628 | ||||||
Amounts in the above table reflect the common stock equivalent.
11. Subsequent Events
On July 23, 2024, Pieris Pharmaceuticals, Inc., a Nevada corporation (“Pieris”) that is listed on the Nasdaq exchange, Polo Merger Sub, Inc., a Delaware corporation and a wholly-owned subsidiary of Pieris (“Merger Sub”), and the Company, entered into an Agreement and Plan of Merger (the “Merger Agreement”), pursuant to which the Merger Sub will merge with and into the Company, with the Company continuing as a wholly-owned subsidiary of Pieris and the surviving corporation of the merger (the “Merger”). The Merger is intended to qualify for federal income tax purposes as a tax-free reorganization and as a non-taxable exchange of shares of the Company’s capital stock for Pieris common stock.
Subject to the terms and conditions of the Merger Agreement (a) each then-outstanding share of the Company’s capital stock will be converted into the right to receive a number of shares of Pieris common stock (subject to the payment of cash in lieu of fractional shares) calculated in accordance with the Merger Agreement; and (b) each then-outstanding stock option to purchase the Company’s common stock will be assumed by Pieris, subject to adjustment as set forth in the Merger Agreement.
Consummation of the Merger is subject to certain closing conditions, including, among other things, (1) approval by Pieris stockholders, (2) approval by the Company’s stockholders, and (3) Nasdaq’s approval of the listing of the shares of Pieris common stock to be issued in connection with the Merger.
The Merger Agreement contains certain termination rights of each of Pieris and the Company, including the right of each party to terminate the Merger Agreement to enter into a definitive agreement for a superior proposal. Upon termination of the Merger Agreement under specified circumstances, Pieris may be required to pay the Company a termination fee of $1.0 million and the Company may be required to pay Pieris a termination fee of $2.0 million.
In June 2024, the Company entered into a Convertible Note Purchase Agreement (the “Convertible Note”) with certain existing and new investors for the issuance of up to $20 million of convertible promissory notes. Through the issuance date of these financial statements, the Company has received $11.9 million of gross proceeds in exchange for convertible promissory notes issued. The Convertible Note bears an annual interest of 2.0% plus SOFR and shall be due and payable upon the earlier to occur of June 2027 or certain events defined in the Convertible Note. Under certain circumstances, the Convertible Note is convertible at the option of requisite holders into the Company’s equity securities at defined conversion prices. The terms of the Convertible Note specify that upon the consummation of the Merger, all outstanding principal and any unpaid accrued interest on the notes shall be automatically converted into common stock of Palvella.
In March 2024, the Company amended the 2019 Plan to include an additional 1,171,768 shares available for awards under the Plan.
Subsequent events have been evaluated through August 9, 2024, which is the date the accompanying financial statements were issued.
ANNEX A
EXECUTION VERSION
AGREEMENT AND PLAN OF MERGER
by and among
PIERIS PHARMACEUTICALS, INC.,
POLO MERGER SUB, INC.
and
PALVELLA THERAPEUTICS, INC.
Dated as of July 23, 2024
TABLE OF CONTENTS
Page
|
ARTICLE I THE MERGER |
2 |
|
|
1.1. |
Effective Time of the Merger |
2 |
|
1.2. |
Closing |
2 |
|
1.3. |
Effects of the Merger |
3 |
|
1.4. |
Directors and Officers of the Surviving Corporation |
3 |
|
1.5. |
Public Company Matters |
3 |
|
ARTICLE II CONVERSION OF SECURITIES |
4 |
|
|
2.1. |
Conversion of Capital Stock |
4 |
|
2.2. |
Exchange of Certificates |
7 |
|
2.3. |
Merger Partner Stock Plans |
9 |
|
2.4. |
Dissenting Shares |
10 |
|
ARTICLE III REPRESENTATIONS AND WARRANTIES OF MERGER PARTNER |
10 |
|
|
3.1. |
Organization, Standing and Power |
11 |
|
3.2. |
Capitalization |
11 |
|
3.3. |
Subsidiaries |
12 |
|
3.4. |
Authority; No Conflict; Required Filings and Consents |
12 |
|
3.5. |
Financial Statements; Information Provided |
14 |
|
3.6. |
No Undisclosed Liabilities |
15 |
|
3.7. |
Absence of Certain Changes or Events |
15 |
|
3.8. |
Taxes |
15 |
|
3.9. |
Owned and Leased Real Properties |
17 |
|
3.10. |
Intellectual Property |
18 |
|
3.11. |
Contracts |
21 |
|
3.12. |
Litigation |
22 |
|
3.13. |
Environmental Matters |
22 |
|
3.14. |
Employee Benefit Plans |
23 |
|
3.15. |
Compliance With Laws |
25 |
|
3.16. |
Permits and Regulatory Matters |
25 |
|
3.17. |
Employees |
27 |
|
3.18. |
Insurance |
27 |
|
3.19. |
Brokers; Fees and Expenses |
28 |
|
3.20. |
Certain Business Relationships with Affiliates |
28 |
|
3.21. |
Controls and Procedures, Certifications and Other Matters |
28 |
|
3.22. |
Books and Records |
28 |
|
3.23. |
Ownership of Public Company Common Stock |
28 |
|
3.24. |
Privacy and Data Protection |
29 |
|
3.25. |
No Other Representations or Warranties |
29 |
|
ARTICLE IV REPRESENTATIONS AND WARRANTIES OF PUBLIC COMPANY AND THE MERGER SUB |
29 |
|
|
4.1. |
Organization, Standing and Power |
30 |
|
4.2. |
Capitalization |
30 |
|
4.3. |
Subsidiaries |
32 |
|
4.4. |
Authority; No Conflict; Required Filings and Consents |
32 |
|
4.5. |
SEC Filings; Financial Statements; Information Provided |
34 |
|
4.6. |
No Undisclosed Liabilities |
35 |
|
4.7. |
Absence of Certain Changes or Events |
35 |
|
4.8. |
Taxes |
35 |
|
4.9. |
Owned and Leased Real Properties |
37 |
|
4.10. |
Intellectual Property |
38 |
|
4.11. |
Contracts |
40 |
|
4.12. |
Litigation |
41 |
|
4.13. |
Environmental Matters |
41 |
|
4.14. |
Employee Benefit Plans |
42 |
|
4.15. |
Compliance With Laws |
43 |
|
4.16. |
Permits and Regulatory Matters |
43 |
|
4.17. |
Employees |
45 |
|
4.18. |
Insurance |
46 |
|
4.19. |
Opinion of Financial Advisor |
46 |
|
4.20. |
Valid Issuance |
46 |
|
4.21. |
Nevada “Combinations with Interested Stockholders” Statutes |
46 |
|
4.22. |
Brokers; Fees and Expenses |
46 |
|
4.23. |
Operations of Merger Sub |
46 |
|
4.24. |
Certain Business Relationships with Affiliates |
47 |
|
4.25. |
Controls and Procedures, Certifications and Other Matters |
47 |
|
4.26. |
Books and Records |
47 |
|
4.27. |
Privacy and Data Protection |
47 |
|
4.28. |
No Other Representations or Warranties |
48 |
|
ARTICLE V CONDUCT OF BUSINESS |
48 |
|
|
5.1. |
Covenants of Merger Partner |
48 |
|
5.2. |
Covenants of Public Company |
50 |
|
5.3. |
Confidentiality |
53 |
|
5.4. |
Closing Dividend; Legacy Asset Dispositions |
53 |
|
ARTICLE VI ADDITIONAL AGREEMENTS |
54 |
|
|
6.1. |
No Solicitation |
54 |
|
6.2. |
Proxy Statement/Prospectus; Registration Statement |
58 |
|
6.3. |
Nasdaq Listing |
59 |
|
6.4. |
Access to Information |
59 |
|
6.5. |
Stockholder Approval |
60 |
|
6.6. |
Legal Conditions to Merger |
61 |
|
6.7. |
Public Disclosure |
62 |
|
6.8. |
Section 368(a) Reorganization; Section 351 Exchange |
62 |
|
6.9. |
Affiliate Legends |
63 |
|
6.10. |
D&O Indemnification |
63 |
|
6.11. |
Notification of Certain Matters |
65 |
|
6.12. |
Employee Communications |
65 |
|
6.13. |
FIRPTA Tax Certificates |
65 |
|
6.14. |
State Takeover Laws |
65 |
|
6.15. |
Security Holder Litigation |
66 |
|
6.16. |
Section 16 Matters |
66 |
|
6.17. |
Calculation of Public Company Net Cash and Exchange Ratio |
66 |
|
6.18. |
Termination of Section 401(k) Plans |
70 |
|
6.19. |
Merger Partner Financials |
70 |
|
6.20. |
New Public Company Equity Plan; Termination of Public Company ESPP |
71 |
|
ARTICLE VII CONDITIONS TO MERGER |
71 |
|
|
7.1. |
Conditions to Each Party’s Obligation to Effect the Merger |
71 |
|
7.2. |
Additional Conditions to the Obligations of Public Company and Merger Sub |
72 |
|
7.3. |
Additional Conditions to the Obligations of Merger Partner |
73 |
|
ARTICLE VIII TERMINATION AND AMENDMENT |
74 |
|
|
8.1. |
Termination |
74 |
|
8.2. |
Effect of Termination |
76 |
|
8.3. |
Fees and Expenses |
76 |
|
ARTICLE IX MISCELLANEOUS |
78 |
|
|
9.1. |
Non-survival of Representations, Warranties and Agreements |
78 |
|
9.2. |
Notices |
78 |
|
9.3. |
Entire Agreement |
79 |
|
9.4. |
Amendment |
79 |
|
9.5. |
Extension; Waiver |
79 |
|
9.6. |
Procedure for Termination, Amendment, Extension or Waiver |
79 |
|
9.7. |
No Third Party Beneficiaries |
80 |
|
9.8. |
Assignment |
80 |
|
9.9. |
Severability |
80 |
|
9.10. |
Counterparts and Signature |
80 |
|
9.11. |
Interpretation |
80 |
|
9.12. |
Governing Law |
81 |
|
9.13. |
Remedies |
81 |
|
9.14. |
Submission to Jurisdiction |
81 |
|
9.15. |
WAIVER OF JURY TRIAL |
82 |
|
9.16. |
Disclosure Schedule |
82 |
|
9.17. |
Certain Defined Terms |
82 |
|
Exhibit A-1 |
Form of Merger Partner Support Agreement |
|
Exhibit A-2 |
Form of Public Company Support Agreement |
|
Exhibit A-3 |
Form of Lock-Up Agreement |
|
Exhibit B-1 |
Form of Surviving Corporation Certificate of Incorporation |
|
Exhibit B-2 |
Form of Surviving Corporation Bylaws |
|
Exhibit C |
Form of CVR Agreement |
|
Exhibit D |
Form of Subscription Agreement |
|
Annex A |
Illustration of Exchange Ratio |
|
Annex B |
Illustration of Public Company Net Cash |
|
Annex C |
Director |
THIS AGREEMENT AND PLAN OF MERGER (this “Agreement”), dated as of July 23, 2024, is entered into by and among Pieris Pharmaceuticals, Inc., a Nevada corporation (“Public Company”), Polo Merger Sub, Inc., a Delaware corporation and a wholly-owned subsidiary of Public Company (the “Merger Sub”), and Palvella Therapeutics, Inc. a Delaware corporation (“Merger Partner”).
WHEREAS, the Board of Directors of Public Company (the “Public Company Board”) and the Board of Directors of Merger Partner (the “Merger Partner Board”) have each (i) determined that the Merger is fair to, and in the best interests of, their respective corporations and stockholders, (ii) approved and declared advisable this Agreement, the Merger and the actions contemplated by this Agreement and (iii) determined to recommend that the stockholders of their respective corporations vote to approve such matters as are contemplated by this Agreement, including, in the case of Merger Partner, the adoption of this Agreement and, in the case of Public Company, (i) the approval of the issuance of shares of common stock, $0.001 par value per share, of Public Company (the “Public Company Common Stock”) pursuant to this Agreement and the Concurrent Financing (as defined below), in each case pursuant to Nasdaq Listing Rule 5635 (the “Share Issuances”), (ii) approval, to the extent required under applicable Nevada Revised Statutes (“NRS”) statutes, of the Public Company Charter Amendments (the “Charter Amendment Proposal”) and (iii) the adoption of the 2024 Equity Incentive Plan (the “2024 Equity Incentive Plan Proposal”);
WHEREAS, the combination of Public Company and Merger Partner shall be effected through a merger (the “Merger”) of Merger Sub with and into Merger Partner in accordance with the terms of this Agreement and the General Corporation Law of the State of Delaware (the “DGCL”), as a result of which Merger Partner will become a wholly-owned subsidiary of Public Company;
WHEREAS, concurrently with the execution and delivery of this Agreement and as a condition and inducement to Public Company’s willingness to enter into this Agreement, each of the stockholders, directors and officers of Merger Partner named in Schedule A of the Merger Partner Disclosure Schedule has entered into a support agreement, dated as of the date of this Agreement, in substantially the form attached hereto as Exhibit A-1 (the “Merger Partner Support Agreements”);
WHEREAS, concurrently with the execution and delivery of this Agreement and as a condition and inducement to Merger Partner’s willingness to enter into this Agreement, each of the stockholders of the Public Company named in Schedule A of the Public Company Disclosure Schedule have entered into a support agreement, dated as of the date of this Agreement, in substantially the form attached hereto as Exhibit A-2 (the “Public Company Support Agreement”);
WHEREAS, concurrently with the execution and delivery of this Agreement and as a condition and inducement to Public Company’s and Merger Partner’s willingness to enter into this Agreement, each of (i) the officers and directors of the Public Company immediately prior to the Effective Time and listed on Schedule A of the Public Company Disclosure Schedule, (ii) the individuals named on Section 1.4(b) of the Merger Partner Disclosure Schedule, (iii) the stockholders, directors and officers of Merger Partner named in Schedule A of the Merger Partner Disclosure Schedule and (iv) the stockholders of the Public Company named in Schedule A of the Public Company Disclosure Schedule have entered into a lock-up agreement in substantially the form attached hereto as Exhibit A-3 (the “Lock-Up Agreements”);
WHEREAS, for United States federal income tax purposes, it is intended that (i) the Merger shall qualify as a “reorganization” within the meaning of Section 368(a) of the Internal Revenue Code of 1986, as amended (the “Code”), and in the event that the former shareholders of Merger Partner, including shareholders that participate in the Concurrent Financing (as defined below) are in “control” of Public Company immediately after the Effective Time within the meaning of Section 368(c) of the Code (the “Control Requirement”) shall also qualify as a non-taxable exchange of shares of voting common stock, par value $0.0001 per share of Merger Partner (“Merger Partner Voting Common Stock”), non-voting common stock, $0.001 per share of Merger Partner (“Merger Partner Non-Voting Common Stock” and together with Merger Partner Voting Common Stock, “Merger Partner Common Stock”) and preferred stock, $0.001 par value per share, of Merger Partner (“Merger Partner Preferred Stock,” and together with Merger Partner Common Stock, “Merger Partner Capital Stock”) for shares of Public Company Common Stock within the meaning of Section 351(a) of the Code and (ii) this Agreement shall constitute a “plan of reorganization” within the meaning of Treasury Regulations Sections 1.368-2(g) and 1.368-3(a); and
WHEREAS, concurrently with the execution and delivery of this Agreement, certain investors shall have entered into a subscription agreement, substantially in the form attached hereto as Exhibit D (the “Subscription Agreement”), representing an aggregate commitment of not less than $30,000,000 (including up to an aggregate principal amount of $20,000,000 from the conversion of convertible notes of Merger Partner made pursuant to that certain Convertible Note Purchase Agreement made as of June 6, 2024 (the “Merger Partner Notes”) that are outstanding as of the date hereof), pursuant to which such investors have agreed to purchase the number of shares of Public Company Common Stock set forth therein immediately following to the Effective Time (the “Concurrent Financing”).
NOW, THEREFORE, in consideration of the foregoing and the respective representations, warranties, covenants and agreements set forth below, Public Company, Merger Sub and Merger Partner agree as follows:
ARTICLE I
THE MERGER
1.1. Effective Time of the Merger. Upon the terms and subject to the conditions set forth in this Agreement, on the Closing Date the parties hereto will cause the Merger to be consummated by executing and filing a certificate of merger (the “Certificate of Merger”) in accordance with the relevant provisions of the DGCL. The Merger shall become effective upon the filing of the Certificate of Merger with the Secretary of State of the State of Delaware or at such subsequent time or date as Public Company and Merger Partner shall agree and specify in the Certificate of Merger (the “Effective Time”).
1.2. Closing. Subject to the satisfaction or (to the extent permitted by Law) waiver of the conditions set forth in Article VII, the closing of the Merger (the “Closing”) will take place at 10:00 a.m., Eastern time (or at such other time as Public Company and Merger Partner mutually agree upon, orally or in writing), on a date to be specified by Public Company and Merger Partner (the “Closing Date”), which shall be no later than the second Business Day after satisfaction or (to the extent permitted by Law) waiver of the conditions set forth in Article VII (other than those conditions that by their nature are to be satisfied at the Closing, but subject to the satisfaction or (to the extent permitted by Law) waiver of such conditions by remote exchange of electronic documents, unless another date or time is agreed to in writing by Public Company and Merger Partner). For the purposes of this Agreement, the term “Business Day” shall mean any day other than a Saturday, Sunday or other day on which commercial banking institutions in New York, New York, Boston, Massachusetts, Philadelphia, Pennsylvania or Wilmington, Delaware are required or permitted by Law to be closed or other day on which the Delaware Secretary of State is closed.
1.3. Effects of the Merger. At the Effective Time, (i) Merger Sub shall be merged with and into Merger Partner (Merger Partner as the surviving corporation following the Merger is sometimes referred to herein as the “Surviving Corporation”) and the separate existence of Merger Sub shall cease and (ii) the certificate of incorporation of Merger Partner as in effect as of immediately prior to the Effective Time shall be amended and restated in its entirety to read as set forth on Exhibit B-1, and, as so amended and restated, shall be the certificate of incorporation of the Surviving Corporation until thereafter amended as provided therein and in accordance with the applicable provisions of the DGCL. In addition, the bylaws of Merger Partner, as in effect immediately prior to the Effective Time, shall be amended and restated to read as set forth on Exhibit B-2, and, as so amended, shall be the bylaws of the Surviving Corporation until thereafter amended as provided therein and in accordance with the applicable provisions of the DGCL. The Merger shall have the effects set forth in this Agreement and the applicable provisions of the DGCL.
1.4. Directors and Officers of the Surviving Corporation.
(a) The individuals named on Section 1.4(a) of the Merger Partner Disclosure Schedule shall be and constitute all of the directors of the Surviving Corporation as of the Effective Time, each to hold office until his or her respective successor has been duly elected or appointed and qualified or until his or her earlier death, resignation or removal in accordance with the certificate of incorporation and bylaws of the Surviving Corporation.
(b) The individuals named on Section 1.4(b) of the Merger Partner Disclosure Schedule (or such other executive officers of Merger Partner designated by Merger Partner prior to the Effective Time) shall be and constitute all of the officers of the Surviving Corporation as of the Effective Time, each to hold office until his or her respective successor has been duly elected or appointed and qualified or until his or her earlier death, resignation or removal in accordance with the certificate of incorporation and bylaws of the Surviving Corporation.
1.5. Public Company Matters.
(a) Board of Directors. Public Company shall use reasonable best efforts to take such actions (including, to the extent necessary, procuring the resignation or removal of any directors on the Public Company Board) so that, immediately after the Effective Time, the number of directors that comprise the full Public Company Board shall be five (5) and shall consist of (i) four (4) of the directors on the Merger Partner Board (or if any such individual is unable or unwilling to serve as a director on the Public Company Board immediately following the Effective Time, then another individual designated by Merger Partner prior to the Effective Time), and (ii) the designated director from the Public Company Board listed on Annex C (or if such individual is unable or unwilling to serve as a director of the Public Company Board immediately following the Effective Time, then another individual that is designated by Public Company prior to the Effective Time).
(b) Officers. Public Company shall use reasonable best efforts to take such actions (including, to the extent necessary, procuring the resignation (to the extent limited to positions held by such officers and not employment) or removal of any officer of Public Company) so that the officers of Merger Partner immediately prior to the Effective Time shall constitute all of the officers of Public Company immediately after the Effective Time (or if such individual is unable or unwilling to serve as an officer of the Public Company Board immediately following the Effective Time, then another individual that is designated by Merger Partner prior to the Effective Time), each having the same title as he or she had as an officer of Merger Partner immediately prior to the Effective Time.
(c) Lock-Up Agreements. Public Company and Merger Partner shall use reasonable best efforts to have each individual who will serve as a director or officer of Public Company following the Closing execute and deliver a Lock Up Agreement prior to Closing.
ARTICLE II
CONVERSION OF SECURITIES
2.1. Conversion of Capital Stock. As of the Effective Time, by virtue of the Merger and without any action on the part of the holder of any shares of Merger Partner Capital Stock or any shares of capital stock of Merger Sub:
(a) Capital Stock of Merger Sub. Each share of the common stock, $0.001 par value per share, of Merger Sub issued and outstanding immediately prior to the Effective Time shall be converted into and become one fully paid and nonassessable share of common stock, $0.001 par value per share, of the Surviving Corporation.
(b) Cancellation of Treasury Stock and Public Company Owned Stock. All shares of Merger Partner Common Stock that are held in treasury and any shares of Merger Partner Common Stock owned by Public Company, Merger Sub or any other subsidiary of Public Company immediately prior to the Effective Time shall be cancelled and shall cease to exist and no stock of Public Company or other consideration shall be delivered in exchange therefor.
(c) Conversion of Merger Partner Capital Stock. Subject to Section 2.2, each share of Merger Partner Capital Stock, other than shares to be cancelled in accordance with Section 2.1(b) and any Dissenting Shares, issued and outstanding immediately prior to the Effective Time shall be automatically converted into the right to receive a number of shares of Public Company Common Stock equal to the Exchange Ratio. As of the Effective Time, all such shares of Merger Partner Capital Stock shall cease to be outstanding and shall automatically be cancelled and shall cease to exist, and each holder of a certificate or non-certificated book entry representing any such shares of Merger Partner Capital Stock shall cease to have any rights with respect thereto, except the right to receive the shares of Public Company Common Stock pursuant to this Section 2.1(c) and any cash in lieu of fractional shares of Public Company Common Stock to be issued or paid in consideration therefor and any amounts payable pursuant to Section 2.2(d) upon the surrender of such certificate in accordance with Section 2.2, without interest. For purposes of this Agreement, “Exchange Ratio” means the quotient obtained by dividing (x) the number of Merger Partner Merger Shares by (y) the number of Merger Partner Outstanding Shares, in which:
(i) “Aggregate Valuation” means the sum of (a) the Merger Partner Valuation, plus (b) the Public Company Valuation.
(ii) “Merger Partner Allocation Percentage” the quotient determined by dividing (i) the Merger Partner Valuation by (ii) the Aggregate Valuation.
(iii) “Merger Partner Merger Shares” means the product determined by multiplying (i) the Post-Closing Public Company Shares by (ii) the Merger Partner Allocation Percentage.
(iv) “Merger Partner Outstanding Shares” means the total number of shares of Merger Partner Capital Stock outstanding immediately prior to the Effective Time, expressed on a fully-diluted and as-converted to Merger Partner Common Stock basis calculated using the Treasury Stock Method and assuming, without duplication, (A) the issuance of all shares of Merger Partner Common Stock that would be issued assuming the acceleration and exercise of all Merger Partner Stock Options outstanding as of immediately prior to the Effective Time, and (B) the conversion of all Merger Partner Preferred Stock outstanding as of immediately prior to the Effective Time (on an as-converted to Merger Partner Common Stock basis). Notwithstanding the foregoing, any Merger Partner Stock Options granted on or after the date of this Agreement would be included in the total number of Merger Partner Outstanding Shares on a fully-diluted and as-converted to Merger Partner Common Stock basis without applying the Treasury Stock Method. For the avoidance of doubt, other than with respect to Merger Partner Common Stock underlying outstanding Merger Partner Stock Options, shares of Merger Partner Common Stock reserved for issuance under the Merger Partner Stock Plans as of immediately prior to the Effective Time shall not be included in the total number of shares of Merger Partner Common Stock outstanding for purposes of determining the Merger Partner Outstanding Shares.
(v) “Merger Partner Valuation” means $95,000,000.
(vi) “Post-Closing Public Company Shares” means the quotient determined by dividing (i) the Public Company Outstanding Shares by (ii) the Public Company Allocation Percentage.
(vii) “Public Company Allocation Percentage” means the quotient determined by dividing (i) the Public Company Valuation by (ii) the Aggregate Valuation.
(viii) “Public Company Closing Price” means the volume weighted average closing trading price of a share of Public Company Common Stock on Nasdaq for the five (5) consecutive trading days ending five (5) trading days immediately prior to the date upon which the Effective Time occurs.
(ix) “Public Company Outstanding Shares” means the total number of shares of Public Company Common Stock that are issued and outstanding immediately prior to the Effective Time, expressed on a fully-diluted and as-converted to Public Company Common Stock basis, calculated using the Treasury Stock Method and assuming, without duplication, (A) the acceleration and exercise of all Public Company Stock Options and Public Company Warrants outstanding as of immediately prior to the Effective Time with an exercise price less than the Public Company Closing Price, and (B) the conversion of all Public Company Preferred Stock outstanding as of immediately prior to the Effective Time (on an as-converted to Public Company Common Stock basis). For the avoidance of doubt, (1) no Public Company Stock Options outstanding as of immediately prior to the Effective Time with an exercise price equal to or greater than the Public Company Closing Price shall be included in the total number of shares of Public Company Common Stock outstanding for purposes of determining the Public Company Outstanding Shares, and (2) other than with respect to Public Company Common Stock underlying outstanding Public Company Stock Options and Public Company Warrants with an exercise price less than the Public Company Closing Price, shares of Public Company Common Stock reserved for issuance under the Public Company Stock Plans as of immediately prior to the Effective Time shall not be included in the total number of shares of Public Company Common Stock outstanding for purposes of determining the Public Company Outstanding Shares.
(x) “Public Company Valuation” means $21,000,000; provided, that (A) if the Final Public Company Net Cash is greater than $11,000,000, then the Public Company Valuation will be adjusted upwards on a dollar-for-dollar basis by the difference of: (i) the Final Public Company Net Cash, minus (ii) $11,000,000, and (B) if the Final Public Company Net Cash is less than $11,000,000, then the Public Company Valuation will be adjusted downwards on a dollar-for-dollar basis by the difference of: (i) $11,000,000, minus (ii) the Final Public Company Net Cash.
(xi) “Treasury Stock Method” means, with respect to Public Company or Merger Partner, as the case may be, a calculation that assumes on a pro forma basis that all outstanding and unexercised Public Company Stock Options or Merger Partner Stock Options (as defined below), as the case may be, with an exercise price less than the Public Company Closing Price, are fully-vested and exercised on a cashless basis (i.e. that the proceeds from such exercises are used to repurchase shares of Public Company Common Stock or Merger Partner Capital Stock, as the case may be, at the Public Company Closing Price, thereby reducing the number of shares outstanding, with the net effect representing the potential dilution from the vesting and exercise of all such Public Company Stock Options or Merger Partner Stock Options, as the case may be). For the avoidance of doubt, the Treasury Stock Method shall exclude entirely any Public Company Stock Options or Merger Partner Stock Options with a per-share exercise price greater than the Public Company Closing Price.
For the avoidance of doubt, neither the proceeds from the Concurrent Financing nor the shares of Public Company Common Stock to be issued in connection therewith shall be included in the calculation or determination of the Exchange Ratio or any component thereof. For illustrative purposes only, a sample Exchange Ratio calculation is attached hereto as Annex A.
(d) Unvested Stock. At the Effective Time, any shares of Public Company Common Stock issued in accordance with Section 2.1(c) with respect to shares of Merger Partner Common Stock held by any employee, director or consultant pursuant to any of Merger Partner’s plans or arrangements that, immediately prior to the Effective Time, are subject to a repurchase option or otherwise “unvested” (“Merger Partner Restricted Stock”) shall remain subject to the same terms, restrictions and vesting schedule as in effect immediately prior to the Effective Time, except to the extent by their terms such shares of Merger Partner Restricted Stock vest at the Effective Time and except for such changes necessary to give effect to the Exchange Ratio and the conversion to Public Company Common Stock. All outstanding rights that Merger Partner may hold immediately prior to the Effective Time to repurchase shares of Merger Partner Restricted Stock are, effective as of immediately after the Effective Time, hereby assigned to Public Company and shall thereafter be exercisable by Public Company upon the same terms and conditions in effect immediately prior to the Effective Time, except that the shares purchasable pursuant to such rights and the purchase price payable per share shall be appropriately adjusted to reflect the Exchange Ratio. Merger Partner shall, prior to the Closing, take all steps necessary to cause the foregoing provisions of this Section 2.1(d) to occur.
(e) Equitable Adjustments. In the event of any stock split, reverse stock split, stock dividend (including any dividend or distribution of securities convertible into capital stock), reorganization, reclassification, combination, recapitalization or other like change with respect to the Merger Partner Capital Stock, Public Company Common Stock or Public Company Preferred Stock occurring after the date hereof and prior to the Effective Time, all references herein to specified numbers of shares of any class or series affected thereby, and all calculations provided for that are based upon numbers of shares of any class or series (or prices therefor) affected thereby, shall be equitably adjusted to the extent necessary to provide the parties the same economic effect as contemplated by this Agreement prior to such stock split, reverse stock split, stock dividend, reorganization, reclassification, combination, capitalization or other like change. Nothing in this Section 2.1(e) shall be construed to permit Merger Partner, Public Company or any subsidiary of Public Company to take any action with respect to its securities that is prohibited by the terms of this Agreement.
2.2. Exchange of Certificates. The procedures for exchanging outstanding shares of Merger Partner Capital Stock for Public Company Common Stock pursuant to the Merger are as follows:
(a) Exchange Agent. At the Effective Time, Public Company shall deposit with Computershare Trust Company, N.A. or another bank or trust company designated by Public Company and reasonably acceptable to Merger Partner (the “Exchange Agent”), for the benefit of the holders of shares of Merger Partner Capital Stock, for exchange in accordance with this Section 2.2, through the Exchange Agent, (i) certificates or non-certificated book entries representing the shares of Public Company Common Stock (such shares of Public Company Common Stock, together with any dividends or distributions with respect thereto with a record date after the Effective Time, being hereinafter referred to as the “Exchange Fund”) issuable pursuant to Section 2.1 in exchange for outstanding shares of Merger Partner Capital Stock, (ii) cash in an amount sufficient to make payments for fractional shares required pursuant to Section 2.2(c), and (iii) any dividends or distributions to which holders of certificates or non-certificated book entries that, as of immediately prior to the Effective Time, represented outstanding shares of Merger Partner Capital Stock (collectively, “Certificates”), whose shares were converted pursuant to Section 2.1 into the right to receive shares of Public Company Common Stock, may be entitled pursuant to Section 2.2(d).
(b) Exchange Procedures. As soon as reasonably practicable after the Effective Time, the Exchange Agent shall mail to each holder of record of a Certificate (i) a letter of transmittal in customary form reasonably acceptable to Public Company and the Exchange Agent specifying that delivery shall be effected, and risk of loss and title to the Certificates shall pass, only upon delivery of the Certificates to the Exchange Agent or, in the case of Certificates that are non-certificated book entries, upon adherence to the procedures set forth in the letter of transmittal, and (ii) instructions for use in effecting the surrender of the Certificates in exchange for certificates representing shares of Public Company Common Stock (plus cash in lieu of fractional shares, if any, of Public Company Common Stock and any dividends or distributions as provided below). Upon surrender of a Certificate (or an affidavit of loss in lieu of such Certificate in accordance with Section 2.2(i)) for cancellation to the Exchange Agent or to such other agent or agents as may be appointed by Public Company, together with such letter of transmittal, duly executed, and such other documents as may reasonably be required by the Exchange Agent and Public Company, the holder of such Certificate shall be entitled to receive in exchange therefor a certificate or book entry account representing that number of whole shares of Public Company Common Stock which such holder has the right to receive pursuant to the provisions of this Article II, plus cash in lieu of fractional shares pursuant to Section 2.2(c) and any dividends or distributions then payable pursuant to Section 2.2(d), and the Certificate so surrendered shall immediately be cancelled. In the event of a transfer of ownership of Merger Partner Capital Stock which is not registered in the transfer records of Merger Partner, a certificate representing the proper number of whole shares of Public Company Common Stock, plus cash in lieu of fractional shares pursuant to Section 2.2(c) and any dividends or distributions pursuant to Section 2.2(d) may be issued or paid to a Person other than the Person in whose name the Certificate so surrendered is registered, only if such Certificate is presented to the Exchange Agent, accompanied by all documents required to evidence and effect such transfer and by evidence that any applicable stock transfer taxes have been paid. Until surrendered as contemplated by this Section 2.2, each Certificate shall be deemed at any time after the Effective Time to represent only the right to receive shares of Public Company Common Stock pursuant to the provisions of this Article II, plus cash in lieu of fractional shares pursuant to Section 2.2(c) and any dividends or distributions then payable pursuant to Section 2.2(d) as contemplated by this Section 2.2.
(c) No Fractional Shares. No certificate or scrip representing fractional shares of Public Company Common Stock shall be issued upon the surrender for exchange of Certificates, and such fractional share interests shall not entitle the owner thereof to vote or to any other rights of a stockholder of Public Company. Notwithstanding any other provision of this Agreement, each holder of shares of Merger Partner Capital Stock converted pursuant to the Merger who would otherwise have been entitled to receive a fraction of a share of Public Company Common Stock (after taking into account all Certificates delivered by such holder and the aggregate number of shares of Merger Partner Capital Stock represented thereby) shall receive, in lieu thereof, cash (without interest and subject to applicable Tax withholding) in an amount equal to such fractional part of a share of Public Company Common Stock multiplied by the last reported sale price of Public Company Common Stock at the 4:00 p.m., Eastern time, end of regular trading hours on The Nasdaq Capital Market (“Nasdaq”) on the last trading day prior to the Effective Time.
(d) Distributions with Respect to Unexchanged Shares. No dividends or other distributions declared or made after the Effective Time with respect to Public Company Common Stock with a record date after the Effective Time shall be paid to the holder of any unsurrendered Certificate until the holder of record of such Certificate shall surrender such Certificate (or an affidavit of loss in lieu of such Certificate in accordance with Section 2.2(i)) in accordance with this Section 2.2. Subject to the effect of applicable Laws, following surrender of any such Certificate (or an affidavit of loss in lieu of such Certificate in accordance with Section 2.2(i)), there shall be issued and paid to the record holder of the Certificate, at the time of such surrender the amount of dividends or other distributions with a record date after the Effective Time previously paid with respect to such whole shares of Public Company Common Stock, without interest, and at the appropriate payment date, the amount of dividends or other distributions having a record date after the Effective Time, but prior to surrender and a payment date subsequent to surrender that are payable with respect to such whole shares of Public Company Common Stock.
(e) No Further Ownership Rights in Merger Partner Capital Stock. All shares of Public Company Common Stock issued upon the surrender for exchange of Certificates in accordance with the terms hereof (including any cash or dividends or other distributions paid pursuant to Section 2.2(c) or 2.2(d)) shall be deemed to have been issued (and paid) in full satisfaction of all rights pertaining to such shares of Merger Partner Capital Stock, and from and after the Effective Time there shall be no further registration of transfers on the stock transfer books of the Surviving Corporation of the shares of Merger Partner Capital Stock that were outstanding immediately prior to the Effective Time. If, after the Effective Time, Certificates are presented to the Surviving Corporation or the Exchange Agent for any reason, they shall be cancelled and exchanged as provided in this Article II, subject to applicable Law in the case of Dissenting Shares.
(f) Termination of Exchange Fund. Any portion of the Exchange Fund that remains undistributed to the holders of Merger Partner Capital Stock for one year after the Effective Time shall be delivered to Public Company, upon demand, and any holder of Merger Partner Capital Stock immediately prior to the Effective Time who has not previously complied with this Section 2.2 shall thereafter look only to Public Company, as a general unsecured creditor, for payment of its claim for Public Company Common Stock, any cash in lieu of fractional shares of Public Company Common Stock and any dividends or distributions with respect to Public Company Common Stock.
(g) No Liability. To the extent permitted by applicable Law, none of Public Company, Merger Sub, Merger Partner, the Surviving Corporation or the Exchange Agent shall be liable to any holder of shares of Merger Partner Capital Stock or Public Company Common Stock, as the case may be, for such shares or any cash amounts required to be delivered to a public official pursuant to any applicable abandoned property, escheat or similar Law. If any Certificate shall not have been surrendered immediately prior to such date on which any shares of Public Company Common Stock, and any cash payable to the holder of such Certificate or any dividends or distributions payable to the holder of such Certificate pursuant to this Article II would otherwise escheat to or become the property of any Governmental Entity, such Certificate and any such shares of Public Company Common Stock or cash, dividends or distributions in respect of such Certificate shall, to the maximum extent permitted by applicable Law, become the property of the Surviving Corporation, free and clear of all claims or interest of any Person previously entitled thereto.
(h) Withholding Rights. Each of the Exchange Agent, Public Company and the Surviving Corporation shall be entitled to deduct and withhold from the amounts otherwise payable pursuant to, or contemplated by, this Agreement to any holder of shares of Merger Partner Capital Stock and any other recipient of payments hereunder such amounts as it reasonably determines that it is required to deduct and withhold with respect to the making of such payment under the Code, or any other applicable provision of Law. The applicable withholding agent shall use commercially reasonable efforts to provide prior notice to any holder of shares of Merger Partner Capital Stock of its intent to deduct or withhold Taxes on payments for Merger Partner Capital Stock and shall reasonably cooperate with such holder in obtaining any available exemption or reduction of such withholding. Any amounts so deducted or withheld shall be timely paid over to the appropriate Governmental Entity. To the extent that amounts are so deducted or withheld and paid over to the appropriate Governmental Entity by the Surviving Corporation or Public Company, as the case may be, such deducted or withheld amounts shall be treated for all purposes of this Agreement as having been paid to the holder of the shares of Merger Partner Capital Stock or other recipient of payments hereunder in respect of which such deduction and withholding was made by the Surviving Corporation or Public Company, as the case may be.
(i) Lost Certificates. If any Certificate shall have been lost, stolen or destroyed, upon the making of an affidavit (in form and substance satisfactory to Public Company and the Exchange Agent) of that fact by the Person claiming such Certificate to be lost, stolen or destroyed and, if required by the Public Company, the posting by such Person of a bond in such reasonable amount as the Public Company may direct as indemnity against any claim that may be made against it with respect to such Certificate, the Exchange Agent shall issue in exchange for such lost, stolen or destroyed Certificate the shares of Public Company Common Stock and any cash in lieu of fractional shares, and unpaid dividends and distributions on shares of Public Company Common Stock deliverable in respect thereof pursuant to this Agreement.
2.3. Merger Partner Stock Plans.
(a) At the Effective Time, each outstanding option to purchase Merger Partner Common Stock (each, a “Merger Partner Stock Option” and collectively, the “Merger Partner Stock Options”), whether vested or unvested, and all stock or equity-related plans, agreements or arrangements of Merger Partner (the “Merger Partner Stock Plans”) themselves, insofar as they relate to outstanding Merger Partner Stock Options, shall be assumed by Public Company and shall be exchanged for an option to acquire, on the same terms and conditions as were applicable under such Merger Partner Stock Option immediately prior to the Effective Time, such number of shares of Public Company Common Stock as is equal to the number of shares of Merger Partner Common Stock subject to the unexercised portion of such Merger Partner Stock Option immediately prior to the Effective Time multiplied by the Exchange Ratio (rounded down to the nearest whole share number), with an exercise price per share for the options equal to the exercise price per share of such Merger Partner Stock Option immediately prior to the Effective Time divided by the Exchange Ratio (rounded up to the nearest whole cent); provided, that in the case of any Merger Partner Stock Option to which Section 421 of the Code applies as of the Effective Time by reason of its qualification under Section 422 of the Code, the exercise price, the number of shares of Public Company Common Stock subject to such option, and the terms and conditions of exercise of such option shall be determined in a manner consistent with the requirements of Section 424(a) of the Code; provided further, that in the case of any Merger Partner Stock Option to which an exemption to Section 409A of the Code applies as of the Effective Time, the exercise price, the number of shares of Public Company Common Stock subject to such option, and the terms and conditions of exercise of such option shall be determined in a manner consistent with the requirements of Section 409A of the Code. Such Merger Partner Stock Options shall continue in effect on the same terms and conditions to which they are currently subject (subject to the adjustments required by this Section 2.3 after giving effect to the Merger). Merger Partner shall, prior to the Effective Time, take all actions necessary or appropriate to give effect to the treatment of Merger Partner Stock Options contemplated by this Section 2.3(a) and to ensure that there will be no accelerated vesting of Merger Partner Stock Options in connection with the Merger.
(b) As soon as practicable after the Effective Time, Public Company shall deliver to the participants in Merger Partner Stock Plans an appropriate notice setting forth such participants’ rights pursuant to Merger Partner Stock Options, as provided in this Section 2.3.
(c) Public Company shall take all corporate action necessary to reserve for issuance a sufficient number of shares of Public Company Common Stock for delivery upon exercise of Merger Partner Stock Options assumed in accordance with this Section 2.3. To the extent permitted under applicable Law, as promptly as practicable after the Effective Time, Public Company shall file a registration statement on Form S-8 (or any successor form) with respect to the shares of Public Company Common Stock subject to such options, to the extent so registrable, and shall use commercially reasonable efforts to maintain the effectiveness of such registration statement or registration statements (and maintain the current status of the prospectus or prospectuses contained therein) for so long as such options remain outstanding.
(d) As of the Effective Time, Public Company shall assume each of the Merger Partner Stock Plans to the extent of the outstanding Merger Partner Stock Options. Prior to the Closing, Merger Partner shall take all corporate action necessary to terminate as of the Effective Time (or otherwise limit the ability to grant new awards or issue shares under) each Merger Partner Stock Plan except with respect to the issuance of shares upon the exercise of any outstanding Merger Partner Stock Option.
2.4. Dissenting Shares.
(a) For purposes of this Agreement, “Dissenting Shares” shall mean shares of Merger Partner Capital Stock issued and outstanding immediately prior to the Effective Time that are held as of the Effective Time by a holder who has not voted in favor of the Merger or consented thereto in writing and who has made a proper demand for appraisal of such shares in accordance with Section 262 of the DGCL (until such time as such holder fails to perfect or otherwise loses such holder’s appraisal rights under the DGCL with respect to such shares, at which time such shares shall cease to be Dissenting Shares). Dissenting Shares will only entitle the holder thereof to such rights as are granted by the DGCL to a holder thereof and shall not be converted into or represent the right to receive Public Company Common Stock unless the stockholder holding such Dissenting Shares shall have forfeited his, her or its right to appraisal under the DGCL or properly withdrawn his, her or its demand for appraisal. If such stockholder has so forfeited or withdrawn his, her or its right to appraisal of Dissenting Shares, then (i) as of the occurrence of such event, such holder’s Dissenting Shares shall cease to be Dissenting Shares and shall be deemed to have been converted, as of the Effective Time, into and represent the right to receive Public Company Common Stock issuable in respect of such Merger Partner Capital Stock pursuant to Section 2.1(c) or Section 2.1(d), as the case may be, without interest, and (ii) promptly following the occurrence of such event, Public Company shall deliver to the Exchange Agent a certificate representing Public Company Common Stock to which such stockholder is entitled pursuant to Section 2.1(c) or Section 2.1(d), as well as any cash, dividend or other distributions to which such holder of Merger Partner Capital Stock may be entitled under this Article II if not previously delivered to the Exchange Agent.
(b) Merger Partner shall give Public Company (i) prompt notice of any written demands for appraisal of any Merger Partner Capital Stock, withdrawals of such demands and any other instruments that relate to such demands received by Merger Partner and (ii) the opportunity to participate in all negotiations and proceedings with respect to demands for appraisal under the DGCL. Merger Partner shall not, except with the prior written consent of Public Company, which shall not be unreasonably withheld, conditioned or delayed, make any payment with respect to any demands for appraisal of Merger Partner Capital Stock or settle or offer to settle any such demands.
ARTICLE III
REPRESENTATIONS AND WARRANTIES OF MERGER PARTNER
Except as set forth herein or in the disclosure schedule delivered or made available by Merger Partner to Public Company and Merger Sub on the date of this Agreement (the “Merger Partner Disclosure Schedule”), Merger Partner represents and warrants to Public Company and Merger Sub as follows:
3.1. Organization, Standing and Power. Merger Partner is a corporation duly organized, validly existing and in good standing under the Laws of the jurisdiction of its incorporation, has all requisite corporate power and authority to own, lease and operate its properties and assets and to carry on its business as currently conducted, and is duly qualified to do business and is in good standing (to the extent applicable in such jurisdiction), under the Laws of all jurisdictions in which the character of the properties it owns, operates or leases or the nature of its activities makes such qualification necessary, except for such failures to be so qualified or in good standing, individually or in the aggregate, that have not had, and are not reasonably likely to have, a Merger Partner Material Adverse Effect. Merger Partner has made available to Public Company complete and accurate copies of its certificate of incorporation and bylaws, and copies of any amendments thereto, existing as of the date of this Agreement and is not in material violation of any provision of either such document.
3.2. Capitalization.
(a) As of the date of this Agreement, the authorized capital stock of Merger Partner consists of 29,000,000 shares of Merger Partner Common Stock and 20,655,895 shares of Merger Partner Preferred Stock. The rights and privileges of each class of Merger Partner’s capital stock are as set forth in Merger Partner’s certificate of incorporation. As of the date of this Agreement, (i) 5,050,000 shares of Merger Partner Voting Common Stock are issued and outstanding and 670,009 shares of Merger Partner Non-Voting Common Stock are issued and outstanding, (ii) no shares of Merger Partner Common Stock are held in the treasury of Merger Partner and (iii) 2,241,903 shares of Merger Partner Preferred Stock are designated as Series A-1 Preferred Stock are issued or outstanding, 1,240,134 shares of Merger Partner Preferred Stock designated as Series A-2 Preferred Stock are issued or outstanding, 1,533,528 shares of Merger Partner Preferred Stock designated as Series B Preferred Stock are issued or outstanding, 8,509,995 shares of Merger Partner Preferred Stock designated as Series C Preferred Stock are issued or outstanding and 1,835,227 shares of Merger Partner Preferred Stock designated as Series D Preferred Stock are issued or outstanding.
(b) As of the date of this Agreement, there are outstanding 78,959 shares of Merger Partner Non-Voting Common Stock that were issued upon the previous exercise of Merger Partner Stock Options, there are outstanding Merger Partner Stock Options with respect to which 2,149,138 shares of Merger Partner Non-Voting Common Stock are issuable, there are 633,671 shares of Merger Partner Non-Voting Common Stock reserved for future issuance under the Merger Partner Stock Plan. Merger Partner has made available to Public Company complete and accurate copies of all Merger Partner Stock Plans and the forms of all award agreements evidencing Merger Partner Stock Options. With respect to each Merger Partner Stock Option (whether outstanding or previously exercised or vested and/or settled, as applicable) (i) each grant of a Merger Partner Stock Option was duly authorized no later than the date on which the grant of such Merger Partner Stock Option was by its terms to be effective by all necessary corporate action, including, as applicable, approval by the Merger Partner’s Board of Directors (or a duly constituted and authorized committee thereof), or a duly authorized delegate thereof, and any required stockholder approval by the necessary number of votes or written consents, (ii) each such grant was made in accordance with the terms of the applicable Merger Partner Stock Plan, the Securities Act of 1933, as amended (the “Securities Act”), the Securities Exchange Act of 1934, as amended (the “Exchange Act”), to the extent applicable, and all other applicable Laws and are not and have not been the subject of any internal investigation, review or inquiry.
(c) Except (i) as set forth in this Section 3.2, (ii) as reserved for future grants under Merger Partner Stock Plans, and (iii) as set forth on Section 3.2(c) of the Merger Partner Disclosure Schedules, as of the date of this Agreement, (A) there are no equity securities of any class of Merger Partner, or any security exchangeable into or exercisable for such equity securities, issued, reserved for issuance or outstanding and (B) there are no options, warrants, equity securities, calls, rights, commitments or agreements of any character to which Merger Partner is a party or by which Merger Partner is bound obligating Merger Partner to issue, exchange, transfer, deliver or sell, or cause to be issued, exchanged, transferred, delivered or sold, additional shares of capital stock or other equity interests of Merger Partner or any security or rights convertible into or exchangeable or exercisable for any such shares or other equity interests, or obligating Merger Partner to grant, extend, accelerate the vesting of, otherwise modify or amend or enter into any such option, warrant, equity security, call, right, commitment or agreement. Other than the Merger Partner Support Agreement or pursuant to any Merger Partner Stock Plan, Merger Partner is not a party to or is bound by any, and to the knowledge of Merger Partner, there are no, agreements or understandings with respect to the voting (including voting trusts and proxies) or sale or transfer (including agreements imposing transfer restrictions) of any shares of capital stock or other equity interests of Merger Partner. For purposes of this Agreement, the term “Affiliate” when used with respect to any party shall mean any Person who is an “affiliate” of that party within the meaning of Rule 405 promulgated under the Securities Act. Except as contemplated by this Agreement or described in this Section 3.2(c), there are no registration rights to which Merger Partner is a party or by which it or they are bound with respect to any equity security of any class of Merger Partner.
(d) All outstanding shares of Merger Partner Capital Stock are, and all shares of Merger Partner Non-Voting Common Stock subject to issuance as specified in Sections 3.2(b) and 3.2(c) upon issuance on the terms and conditions specified in the instruments pursuant to which they are issuable, will be, duly authorized, validly issued, fully paid and nonassessable and not subject to or issued in violation of any purchase option, call option, right of first refusal, preemptive right, subscription right or any similar right under any provision of the DGCL, Merger Partner’s certificate of incorporation or bylaws or any agreement to which Merger Partner is a party or is otherwise bound. There are no obligations, contingent or otherwise, of Merger Partner to repurchase, redeem or otherwise acquire any shares of Merger Partner Capital Stock. All outstanding shares of Merger Partner Capital Stock have been offered, issued and sold by Merger Partner in compliance with all applicable federal and state securities Laws.
(e) No consent of the holders of Merger Partner Stock Options is required in connection with the actions contemplated by Section 2.3.
3.3. Subsidiaries. Merger Partner does not have any subsidiaries and does not otherwise own any shares of capital stock or any interest in any other Person. Merger Partner does not control directly or indirectly or have any direct or indirect equity participation or similar interest in any Person.
3.4. Authority; No Conflict; Required Filings and Consents.
(a) Merger Partner has all requisite corporate power and authority to enter into this Agreement and, subject only to the adoption of this Agreement (the “Merger Partner Voting Proposal”) by Merger Partner’s stockholders under the DGCL and the certificate of incorporation of Merger Partner (the “Merger Partner Stockholder Approval”) to consummate the transactions contemplated by this Agreement. Without limiting the generality of the foregoing, the Merger Partner Board, at a duly called meeting at which all directors were present, by a unanimous vote, or via unanimous written consent (i) determined that the Merger is fair to, and in the best interests of, Merger Partner and its stockholders, (ii) approved this Agreement, the Merger and the actions contemplated by this Agreement in accordance with the provisions of the DGCL, (iii) declared this Agreement advisable, and (iv) determined to recommend that the stockholders of Merger Partner vote to adopt this Agreement and thereby approve the Merger and such other actions as contemplated hereby. The execution and delivery of this Agreement and the consummation of the transactions contemplated by this Agreement by Merger Partner have been duly authorized by all necessary corporate action on the part of Merger Partner, subject only to the required receipt of the Merger Partner Stockholder Approval. This Agreement has been duly executed and delivered by Merger Partner and, assuming the due execution and delivery of this Agreement by Public Company, constitutes the valid and binding obligation of Merger Partner, enforceable against such party in accordance with its terms, subject to applicable bankruptcy, insolvency, fraudulent transfer, reorganization, moratorium and similar Laws of general applicability relating to or affecting creditors’ rights and to general equity principles (the “Bankruptcy and Equity Exception”).
(b) The execution and delivery of this Agreement by Merger Partner does not, and the consummation by Merger Partner of the transactions contemplated by this Agreement shall not, (i) conflict with, or result in any violation or breach of, any provision of the certificate of incorporation or bylaws of Merger Partner, (ii) conflict with, or result in any material violation or breach of, or constitute (with or without notice or lapse of time, or both) a default (or give rise to a right of termination, cancellation or acceleration of any obligation or loss of any material benefit) under, or require a consent or waiver under, constitute a change in control under, require the payment of a penalty under or result in the imposition of any mortgage, security interest, pledge, lien, charge or encumbrance of any nature (“Liens”) on Merger Partner’s assets (including Merger Partner Intellectual Property) under any of the terms, conditions or provisions of any Contract required to be disclosed in Section 3.11(a) of the Merger Partner Disclosure Schedules, or (iii) subject to obtaining the Merger Partner Stockholder Approval and compliance with the requirements specified in clauses (i) through (iv) of Section 3.4(c), conflict with or violate any permit, concession, franchise, license, judgment, injunction, order, decree, statute, Law, ordinance, rule or regulation applicable to Merger Partner or any of its properties or assets, except in the case of clauses (ii) and (iii) of this Section 3.4(b), as would not, individually or in the aggregate, reasonably be expected to result in a Merger Partner Material Adverse Effect.
(c) No consent, approval, license, permit, order or authorization of, or registration, declaration, notice or filing with, any court, arbitrational tribunal, administrative agency or commission or other governmental or regulatory authority, agency or instrumentality (a “Governmental Entity”) is required by or with respect to Merger Partner in connection with the execution and delivery of this Agreement by Merger Partner or the consummation by Merger Partner of the transactions contemplated by this Agreement, except for (i) the filing of the Certificate of Merger with the Delaware Secretary of State and appropriate corresponding documents with the appropriate authorities of other states in which Merger Partner is qualified as a foreign corporation to transact business, (ii) the filing of the Registration Statement and the Proxy Statement/Prospectus with the U.S. Securities and Exchange Commission (the “SEC”) in accordance with the Exchange Act, (iii) such consents, approvals, orders, authorizations, registrations, declarations and filings as may be required under applicable state securities Laws and the Laws of any foreign country, (iv) such other consents, declarations, authorizations, orders, filings, approvals and registrations that, individually or in the aggregate, if not obtained or made, would not be reasonably expected to result in a Merger Partner Material Adverse Effect.
(d) The affirmative vote in favor of the Merger Partner Voting Proposal by the holders of a (i) majority of the votes represented by the outstanding shares of Merger Partner Voting Common Stock and Merger Partner Preferred Stock voting together as a single class on an as-converted to Merger Partner Voting Common Stock basis, and (ii) majority of the votes represented by the outstanding shares of Merger Partner Preferred Stock voting together as a single class on an as-converted to Merger Partner Voting Common Stock basis, including (A) at least one of (1) Petrichor Opportunities Fund I LP and Petrichor Opportunities Fund I Intermediate LP (collectively, “Petrichor”) or (2) the affirmative vote or written consent of the holders of at least seventy percent (70%) of the outstanding shares of Merger Partner Series D Preferred Stock held by stockholders of Merger Partner other than Petrichor, and (B) at least one of (1) CDK Associates L.L.C. or (2) Samsara BioCapital L.P., which is to be delivered pursuant to written consents of stockholders in lieu of a meeting, in form and substance reasonably acceptable to Public Company (collectively, the “Written Consents”), is the only vote of the holders of any class or series of Merger Partner’s capital stock or other securities necessary for the adoption of this Agreement by Merger Partner and for the consummation by Merger Partner of the other transactions contemplated by this Agreement. There are no bonds, debentures, notes or other indebtedness of Merger Partner having the right to vote (or convertible into, or exchangeable for, securities having the right to vote) on any matters on which stockholders of Merger Partner may vote.
3.5. Financial Statements; Information Provided.
(a) Merger Partner has made available to Public Company correct and complete copies of the Financial Statements. The Financial Statements (i) were prepared in accordance with United States generally accepted accounting principles (“GAAP”) applied on a consistent basis throughout the periods covered thereby (except (x) that the unaudited Financial Statements do not contain footnotes and (y) as may be indicated in the notes to such Financial Statements) and (ii) fairly present in all material respects the financial position of Merger Partner as of the dates thereof, except that the unaudited interim Financial Statements are subject to normal year-end adjustments, that are not expected to be material in amount. For purposes of this Agreement, “Financial Statements” means (A) the unaudited balance sheet as at December 31, 2023, and the related unaudited statements of income, changes in stockholders’ equity and cash flows of Merger Partner as for the period then ended, (B) the audited balance sheet as at December 31, 2022, and the related audited statements of income, changes in stockholders’ equity and cash flows of Merger Partner as for the period then ended and (C) the unaudited balance sheet as at March 31, 2024 (the “Most Recent Balance Sheet Date”), and the related audited statements of income, changes in stockholders’ equity and cash flows of Merger Partner as for the period then ended.
(b) The information to be supplied by or on behalf of Merger Partner for inclusion or incorporation by reference in the registration statement on Form S-4 to be filed by Public Company pursuant to which shares of Public Company Common Stock issued in connection with the Merger shall be registered under the Securities Act (the “Registration Statement”), or supplied by or on behalf of Merger Partner for inclusion in any filing pursuant to Rule 165 and Rule 425 under the Securities Act or Rule 14a-12 under the Exchange Act (each a “Regulation M-A Filing”), shall not at the time the Registration Statement or any such Regulation M-A Filing is filed with the SEC, at any time it is amended or supplemented or at the time the Registration Statement is declared effective by the SEC, as applicable, contain any untrue statement of a material fact or omit to state any material fact required to be stated therein or necessary in order to make the statements therein, in light of the circumstances under which they are made, not false or misleading. The information to be supplied by or on behalf of Merger Partner for inclusion in the proxy statement/prospectus/information statement (the “Proxy Statement/Prospectus”) to be sent to the stockholders of Public Company and Merger Partner in connection with the meeting of Public Company’s stockholders (the “Public Company Meeting”) to solicit the approval of the Public Company’s stockholders of (i) the Share Issuances under Nasdaq Rules or applicable NRS provisions, and (ii) the Charter Amendment Proposal (clauses (i) and (ii) collectively, the “Required Public Company Voting Proposals”) and (iii) the 2024 Equity Incentive Plan Proposal (the “Other Public Company Voting Proposal” and, collectively with the Required Public Company Stockholder Approvals, the “Public Company Stockholder Approvals”), which information shall be deemed to include all information about or relating to Merger Partner and/or the Merger Partner Voting Proposal, shall not, on the date the Proxy Statement/Prospectus is first mailed to stockholders of Public Company, or at the time of the Public Company Meeting or as of the Effective Time, contain any untrue statement that, at such time and in light of the circumstances under which it shall be made, is false or misleading with respect to any material fact, or omit to state any material fact necessary in order to make the statements made in the Proxy Statement/Prospectus not false or misleading; or omit to state any material fact necessary to correct any statement in any earlier communication with respect to the solicitation of proxies for the Public Company Meeting that has become false or misleading.
3.6. No Undisclosed Liabilities. Merger Partner does not have any material Liability, except for (a) Liabilities shown on the Most Recent Balance Sheet, (b) Liabilities of a type required to be shown on the Most Recent Balance Sheet that have arisen since the Most Recent Balance Sheet Date in the Ordinary Course of Business (and which have not resulted from a breach of contract, breach of warranty, tort, infringement or violation of Law), and (c) Liabilities for transaction expenses incurred in connection with the transactions contemplated by this Agreement.
3.7. Absence of Certain Changes or Events. During the period beginning on the Most Recent Balance Sheet Date and ending on the date hereof, Merger Partner has conducted its business only in the Ordinary Course of Business and, since such date, there has not been (a) any change, event, circumstance, development or effect that, individually or in the aggregate, has had, or is reasonably expected to have, a Merger Partner Material Adverse Effect, or (b) any other action or event that would have required the consent of Public Company pursuant to Section 5.1 had such action or event occurred after the date of this Agreement.
3.8. Taxes.
(a) Merger Partner has properly filed on a timely basis all income and other material Tax Returns that it was required to file, and all such Tax Returns are true, correct and complete in all material respects. Merger Partner has paid on a timely basis all Taxes, whether or not shown on any Tax Return, that were due and payable.
(b) Merger Partner is not, nor has it ever been a member of an affiliated group with which it has filed (or been required to file) consolidated, combined, unitary or similar U.S. federal Tax Returns, other than a group of which the common parent is Merger Partner. With the exception of customary commercial leases or contracts that are not primarily related to Taxes entered into in the Ordinary Course of Business and liabilities thereunder, Merger Partner (i) does not have any liability under Treasury Regulations Section 1.1502-6 (or any comparable or similar provision of state, local or non-U.S. Law), as a transferee or successor, pursuant to any contractual obligation, or otherwise for any Taxes of any Person other than Merger Partner, and (ii) is not a party to or bound by any Tax indemnity, Tax sharing, Tax allocation or similar agreement.
(c) All material Taxes that Merger Partner was required by Law to withhold or collect have been duly withheld or collected and, to the extent required, have been properly paid to the appropriate Governmental Entity, in each case in compliance in all material respects with applicable Law.
(d) Merger Partner has delivered or made available to Public Company (i) complete and correct copies of all income and other material Tax Returns of Merger Partner relating to Taxes for all taxable periods for which the applicable statute of limitations has not yet expired, (ii) complete and correct copies of all private letter rulings, revenue agent reports, information document requests, notices of proposed deficiencies, deficiency notices, protests, petitions, closing agreements, settlement agreements, pending ruling requests and any similar documents submitted by, received by, or agreed to by or on behalf of Merger Partner relating to Taxes for all taxable periods for which the statute of limitations has not yet expired, and (iii) complete and correct copies of all material agreements, rulings, settlements or other Tax documents with or from any Governmental Entity relating to Tax incentives of Merger Partner.
(e) No examination or audit of any Tax Return of Merger Partner by any Governmental Entity is currently in progress or, to the knowledge of Merger Partner, has been threatened by any Governmental Entity. No deficiencies for Taxes of Merger Partner have been claimed, proposed or assessed by any Governmental Entity in writing. Merger Partner has not been informed in writing by any jurisdiction in which Merger Partner does not file a Tax Return that the jurisdiction believes that Merger Partner was required to file any Tax Return that was not filed or is subject to Tax in such jurisdiction. Merger Partner has not (i) waived any statute of limitations with respect to Taxes or agreed to extend the period for assessment or collection of any Taxes (other than any automatic extension granted in the Ordinary Course of Business and consistent with past custom and practice of Merger Partner), which waiver or extension is still in effect, (ii) requested any extension of time within which to file any Tax Return (other than any extension granted in the Ordinary Course of Business and consistent with past custom and practice of Merger Partner), or (iii) executed or filed any power of attorney with any taxing authority, which is still in effect.
(f) Neither Merger Partner nor any of its subsidiaries has made any payment or provided any benefit that has resulted in, and neither the execution and delivery of this Agreement nor the consummation of the transactions contemplated hereby (either alone or in conjunction with additional or subsequent events, including any termination of employment or service), will result in, any payment or provide any benefit that may be treated as an “excess parachute payment” under Section 280G of the Code (without regard to Sections 280G(b)(4) and 280G(b)(5) of the Code).
(g) Merger Partner has not been a United States real property holding corporation within the meaning of Section 897(c)(2) of the Code during the applicable period specified in Section 897(c)(l)(A)(ii) of the Code.
(h) Merger Partner has not distributed to its stockholders or security holders stock or securities of a controlled corporation, nor has stock or securities of Merger Partner been distributed, in a transaction to which Section 355 of the Code applies in the two years prior to the date of this Agreement.
(i) There are no Liens with respect to Taxes upon any of the assets or properties of Merger Partner, other than with respect to Taxes not yet due and payable or being contested in good faith by appropriate proceedings.
(j) Merger Partner will not be required to include any material item of income in, or exclude any material item of deduction from, taxable income for any period (or any portion thereof) ending after the Closing Date as a result of any (i) adjustments under Section 481 of the Code (or any similar adjustments under any provision of corresponding non-U.S., state or local Tax Laws) for a taxable period (or portion thereof) ending on or prior to the Closing Date, (ii) installment sale or other open transaction disposition made on or prior to the Closing Date, or (iii) prepaid amount or deferred revenue received on or prior to the Closing Date outside the Ordinary Course of Business.
(k) Merger Partner has not participated in any “reportable transaction” as defined in Treasury Regulations Section 1.6011-4(b).
(l) Merger Partner (i) is not a party to any joint venture, partnership, or other arrangement that is treated as a partnership for U.S. federal income Tax purposes and (ii) since the date of its formation until May 29, 2018 it was taxed classified as a partnership for U.S. federal income tax purposes, and since May 30, 2018 has been classified as an association taxable as a C corporation.
(m) All related party transactions involving Merger Partner have been conducted at arm’s length in compliance with Section 482 of the Code and the Treasury Regulations promulgated thereunder and any comparable provisions of any other Tax Law.
(n) None of the Merger Partner Capital Stock is subject to a “substantial risk of forfeiture” within the meaning of Section 83 of the Code (or any corresponding or similar provision of state, local or non-U.S. income Tax Law). Each Person who was issued stock in Merger Partner that was subject to a “substantial risk of forfeiture” within the meaning of Section 83 of the Code made a timely election with respect to such stock pursuant to Section 83(b) of the Code (or any corresponding or similar provision of state, local or non-U.S. income Tax Law, or election).
(o) Merger Partner is not subject to income Tax in any country other than its country of incorporation, organization or formation by virtue of having employees, a permanent establishment or other fixed place of business in that country.
(p) To the knowledge of Merger Partner, neither Merger Partner nor any of its Affiliates has taken or agreed to take any action, has omitted to take any action, or has any knowledge of any fact or circumstance, the taking, omission, or existence of which, as the case may be, would reasonably be expected to prevent the Merger from constituting a transaction qualifying as a reorganization under Section 368(a) of the Code and, in the event the Control Requirement is satisfied, as a non-taxable exchange of shares of Merger Partner Capital Stock for shares of Public Company Common Stock under Section 351(a) of the Code.
(q) Merger Partner is not an investment company as defined in Section 368(a)(2)(F)(iii) and (iv) of the Code.
(r) For purposes of this Agreement, (i) “Taxes” shall mean any taxes, charges, fees, duties, contributions, levies or other similar assessments or liabilities in the nature of a tax, including, without limitation, income, gross receipts, corporation, ad valorem, premium, value-added, net worth, capital stock, capital gains, documentary, recapture, alternative or add-on minimum, disability, estimated, registration, recording, excise, real property, personal property, sales, use, license, lease, service, service use, transfer, withholding, employment, unemployment, insurance, social security, national insurance, business license, business organization, environmental, workers compensation, payroll, profits, severance, stamp, occupation, windfall profits, escheat or unclaimed property, customs duties, franchise and other taxes of any kind imposed by the United States of America or any state, local or non-U.S. government, or any agency or political subdivision thereof, and any interest, fines, penalties, assessments or additions to tax imposed with respect to such items, and (ii) “Tax Returns” shall mean any and all reports, returns (including information returns), declarations, or statements relating to Taxes, including any schedule or attachment thereto and any amendment thereof, filed with or submitted to, or required to be filed with or submitted to, a Governmental Entity in connection with the determination, assessment, collection or payment of Taxes or in connection with the administration, implementation or enforcement of or compliance with any legal requirement relating to any Tax.
3.9. Owned and Leased Real Properties.
(a) Merger Partner does not own nor has ever owned any real property.
(b) Section 3.9(b) of the Merger Partner Disclosure Schedule sets forth a complete and accurate list of all real property leased, subleased or licensed by Merger Partner as of the date of this Agreement (collectively, the “Merger Partner Leases”) and the location of the premises of such real property. Neither Merger Partner nor, to the knowledge of Merger Partner, any other party is in breach or default and no event has occurred, is pending or, to the knowledge of Merger Partner, is threatened, which, after the giving of notice, with lapse of time, or otherwise, would constitute any such breach or default under any of Merger Partner Leases, except where the existence of such breaches or defaults, individually or in the aggregate, has not had, and is not reasonably likely to result in, a Merger Partner Material Adverse Effect. Merger Partner does not lease, sublease or license any real property to any Person other than Merger Partner. Merger Partner has made available to Public Company complete and accurate copies of all Merger Partner Leases.
3.10. Intellectual Property.
(a) Section 3.10(a) of the Merger Partner Disclosure Schedule lists all Merger Partner Registrations, in each case enumerating specifically the applicable filing or registration number, title, jurisdiction in which filing was made or from which registration issued, date of filing or issuance, and names of all current applicant(s) and registered owner(s), as applicable except that, for any Merger Partner Registrations that are Internet domain names or social media accounts and identifiers, such enumeration shall be the applicable account name or number, the domain registrar or social media company and the registered owner(s). All assignments of Merger Partner Registrations to Merger Partner have been properly executed and recorded, and all issuance, renewal, maintenance and other payments that are or have become due with respect thereto have been timely paid by or on behalf of the Merger Partner. To the knowledge of Merger Partner, all Merger Partner Registrations are valid and enforceable.
(b) There are no inventorship challenges, inter partes proceedings, opposition or nullity proceedings or interferences declared, commenced or provoked, or, to the knowledge of Merger Partner, threatened, with respect to any Patent Rights included in the Merger Partner Registrations. None of the Patent Rights included in the Merger Partner Registrations have been abandoned. Merger Partner has complied with its duty of candor and disclosure to the United States Patent and Trademark Office and any relevant foreign patent office with respect to all patent and trademark applications filed by or on behalf of Merger Partner and has made no material misrepresentation in such applications. Merger Partner has no knowledge of any information that would preclude Merger Partner from having clear title to the Merger Partner Registrations.
(c) Merger Partner is the sole and exclusive owner of all Merger Partner Owned Intellectual Property, free and clear of any Liens, other than any joint owners of the Merger Partner Owned Intellectual Property that are listed in Section 3.10(c) of the Merger Partner Disclosure Schedule. None of the Merger Partner Intellectual Property is subject to any orders, decrees or injunctions.
(d) Merger Partner has taken reasonable measures to protect the proprietary nature of each item of Merger Partner Owned Intellectual Property, and to maintain in confidence all trade secrets and confidential information comprising a part thereof. To Merger Partner’s knowledge, there has been no unauthorized disclosure of any third party proprietary or confidential information in the possession, custody or control of Merger Partner.
(e) To the knowledge of Merger Partner, the operations of Merger Partner as currently conducted do not infringe, misappropriate or otherwise violate and have not in the past five years infringed, misappropriated or otherwise violated the valid and enforceable Intellectual Property rights of any individual or entity. To Merger Partner’s knowledge, no individual or entity has infringed, misappropriated or otherwise violated the Merger Partner Owned Intellectual Property or any rights under the Merger Partner Intellectual Property that are exclusively licensed to Merger Partner, and Merger Partner has not filed or threatened in writing any claims alleging that a third party or Worker has infringed, misappropriated or otherwise violated any Merger Partner Intellectual Property. No individual or entity has filed and served upon Merger Partner or, to Merger Partner’s knowledge, threatened or otherwise filed any action or proceeding alleging that Merger Partner has infringed, misappropriated or otherwise violated any individual’s or entity’s Intellectual Property rights nor has Merger Partner received any written notification that a license under any other individual’s or entity’s Intellectual Property is or may be required.
(f) Merger Partner has made available to Public Company copies of all material written complaints, claims, notices or threats, or disclosed to Public Company all material non-written complaints, claims, notices or threats, in each case, concerning the infringement, violation or other misappropriation of any Merger Partner Intellectual Property.
(g) Section 3.10(g) of the Merger Partner Disclosure Schedule identifies (i) each license or agreement pursuant to which Merger Partner has granted rights to any Merger Partner Licensed Intellectual Property, and (ii) each agreement, contract, assignment or other instrument pursuant to which Merger Partner has granted any joint ownership interest in or to each item of Merger Partner Owned Intellectual Property, in each case (i) and (ii) other than Excluded Contracts.
(h) Section 3.10(h) of the Merger Partner Disclosure Schedule identifies (i) each license or agreement pursuant to which Merger Partner has obtained rights to any Merger Partner Licensed Intellectual Property (excluding generally available, off the shelf software programs that are licensed by Merger Partner pursuant to “shrink wrap” licenses, the total fees associated with which are less than $50,000) and (ii) each agreement, contract, assignment or other instrument pursuant to which Merger Partner has obtained any joint or sole ownership interest in or to each item of Merger Partner Owned Intellectual Property, in each case (i) and (ii) excluded for Excluded Contracts.
(i) To Merger Partner’s knowledge, no Worker of Merger Partner is in default or breach of any term of any employment Contract, non-disclosure Contract, assignment of invention Contract or similar Contract between such Worker and Merger Partner, as applicable, relating to the protection, ownership, development, use, assignment or transfer of Merger Partner Intellectual Property. To the extent that any Merger Partner Owned Intellectual Property has been conceived, reduced to practice, authored, developed or created for Merger Partner by any individual while a Worker, Merger Partner has obtained the entire and unencumbered right, title and interest therein and thereto by operation of Law or by valid written assignment.
(j) The execution and delivery of this Agreement by Merger Partner does not, and the consummation by Merger Partner of the transactions contemplated by this Agreement shall not, result in (i) a material breach of or default under any agreement governing any Merger Partner Intellectual Property, (ii) the grant or transfer to any third party of any new license or other interest under, the abandonment, assignment to any third party, or modification or loss of any right with respect to, any Merger Partner Intellectual Property, (iii) the grant or transfer to any third party of any license or other interest under, or any covenant not to sue in respect of, any Public Company Intellectual Property, or (iv) Merger Partner, Public Company or any of their respective Affiliates being obligated to pay any penalty or new or increased royalty or fee to any individual or entity under any agreement governing any Merger Partner Intellectual Property.
(k) For purposes of this Agreement, the following terms shall have the following meanings:
(i) “Intellectual Property” shall mean the following subsisting throughout the world: (A) Patent Rights; (B) Trademarks and all goodwill in the Trademarks; (C) copyrights, designs, data and database rights and registrations and applications for registration thereof, including moral rights of authors; (D) mask works and registrations and applications for registration thereof and any other rights under the Laws of any jurisdiction; (E) inventions, invention disclosures, statutory invention registrations, trade secrets and confidential business information, know-how, scientific and technical information, data and technology, including medical, clinical, toxicological and other scientific data, manufacturing and product processes, algorithms, techniques and analytical methodology, research and development information, financial, marketing and business data, pricing and cost information, business and marketing plans and customer and supplier lists and information, whether patentable or nonpatentable, whether copyrightable or noncopyrightable and whether or not reduced to practice; and (F) other proprietary rights relating to any of the foregoing (including remedies against infringement thereof and rights of protection of interest therein under the Laws of all jurisdictions).
(ii) “Intellectual Property Registrations” shall mean Patent Rights, applications and registrations for Trademarks, applications and registrations for copyrights and designs, mask work registrations and applications for each of the foregoing, which are issued by, filed with, or recorded by any state, government or other public legal authority at any time in any jurisdictions, or, in the case of Internet domain names and social media accounts and identifiers, which are issued by, filed with, or recorded by any third party.
(iii) “Law” shall mean each applicable transnational, domestic or foreign federal, state or local laws (statutory, common or otherwise), order, judgment, rule, code, statute, regulation, requirement, variance, decree, writ, injunction, award, ruling, Permit or ordinance of any Governmental Entity, including any applicable stock exchange rule or requirement.
(iv) “Merger Partner Intellectual Property” shall mean the Merger Partner Owned Intellectual Property and the Merger Partner Licensed Intellectual Property.
(v) “Merger Partner Licensed Intellectual Property” shall mean all Intellectual Property that is licensed to Merger Partner by any individual or entity, excluding any Intellectual Property that is licensed to Merger Partner under Excluded Contracts.
(vi) “Merger Partner Owned Intellectual Property” shall mean all Intellectual Property owned or purported to be owned by Merger Partner, in whole or in part.
(vii) “Merger Partner Registrations” shall mean Intellectual Property Registrations that are registered or filed in the name of Merger Partner or where Merger Partner is the assignee thereof, in each case, alone or jointly with others.
(viii) “Patent Rights” shall mean all patents, patent applications, utility models, design registrations and certificates of invention and other governmental grants for the protection of inventions or industrial designs (including all related continuations, continuations-in-part, divisionals, reissues and reexaminations).
(ix) “Trademarks” shall mean all registered trademarks and service marks, logos, Internet domain names, social media accounts and identifiers, corporate names and doing business designations and all registrations and applications for registration of the foregoing, common Law trademarks and service marks and trade dress.
(x) “Worker” means any individual who is an officer, director, employee (regular, temporary, part-time or otherwise), consultant or independent contractor of Merger Partner or Public Company or any of its subsidiaries, as applicable.
3.11. Contracts.
(a) Section 3.11(a) of the Merger Partner Disclosure Schedule lists the following Contracts of Merger Partner in effect as of the date of this Agreement (in each case, excluding Excluded Contracts):
(i) any Contract (or group of related Contracts) for the purchase or sale of products or for the furnishing or receipt of services (A) which expressly requires aggregate payments by or to Merger Partner of more than $200,000 or (B) in which Merger Partner has granted manufacturing rights, “most favored nation” pricing provisions or marketing or distribution rights relating to any products or territory, or has agreed to purchase goods or services exclusively from a particular party or to a right of first offer, right of first refusal, right of first negotiation in favor of any third party;
(ii) any Contract under which Merger Partner has granted to a third party a license under, or option or covenant not to sue with respect to, any Merger Partner Intellectual Property;
(iii) any Contract under which Merger Partner is prohibited from selling, licensing or otherwise distributing any of its technology or products, or providing services to, customers or potential customers or any class of customers, in any geographic area, during any period of time or any segment of the market or line of business;
(iv) any (A) employment Contract (excluding offer letters for at-will employment that do not provide for severance or for advance notice of termination or for any change of control, transaction, retention or other special remuneration) that provides for base salary and target bonus, when taken together, of at least $200,000 and (B) individual independent contractor or consulting Contract that involves or could involve payments in excess of $150,000 within any twelve (12) month period;
(v) any Contract, plan, policy or program providing for severance, termination compensation, retention or stay pay, change in control payments or transaction-based bonuses;
(vi) any settlement Contract or settlement-related Contract (including any Contract in connection with which any employment-related claim is settled) under which either side has remaining financial obligations;
(vii) any dealer, distribution, joint marketing, joint venture, joint development, partnership, strategic alliance, collaboration, development agreement or outsourcing arrangement;
(viii) any Contract for the conduct of research studies, pre-clinical or clinical studies, manufacturing, distribution, supply, marketing or co-promotion of any products in development by or which has been or which is being marketed, distributed, supported, sold or licensed out, in each case by or on behalf of Merger Partner; and
(ix) any Contract that involved or would reasonably be expected to result in (A) the grant or transfer to any third party of any new license or other interest under, the abandonment, assignment to any third party, or modification or loss of any right with respect to, or the creation of any Lien (excluding a Permitted Lien) on any Merger Partner Intellectual Property, (B) the grant or transfer to any third party of any license or other interest under, or any covenant not to sue with respect to, any Public Company Intellectual Property; or (C) Merger Partner, Public Company or any of their respective Affiliates being obligated to pay any penalty or new or increased royalty or fee to any individual or entity under any agreement governing any Merger Partner Intellectual Property.
(b) Merger Partner has made available to Public Company a complete and accurate copy of each Contract listed in Sections 3.10(a), 3.10(g), 3.10(h), and 3.11(a) of the Merger Partner Disclosure Schedule. With respect to each Contract so listed or that should be listed: (i) the Contract is legal, valid, binding and enforceable and in full force and effect against Merger Partner, as applicable, and, to the knowledge of Merger Partner, against each other party thereto, as applicable, subject to the Bankruptcy and Equity Exception; and (ii) none of Merger Partner, nor, to the knowledge of Merger Partner, any other party, is in material breach or violation of, or default under, any such Contract, and no event has occurred, is pending or, to the knowledge of Merger Partner, is threatened, which, with or without notice or lapse of time, or both, would constitute a material breach or default by Merger Partner or, to the knowledge of Merger Partner, any other party under such Contract, except for such breaches, violations or defaults that, individually or in the aggregate, have not had, and are not reasonably likely to have, a Merger Partner Material Adverse Effect.
(c) For purposes of this Agreement, the term “Contract” shall mean, with respect to any Person, any written, oral or other agreement, contract, subcontract, lease (whether for real or personal property), mortgage, understanding, arrangement, instrument, note, option, warranty, license, sublicense, insurance policy, benefit plan or commitment or undertaking of any nature to which such Person is a party or by which such Person or any of its assets are bound under applicable Law.
3.12. Litigation. As of the date of this Agreement, there is no action, suit, proceeding, claim, arbitration or investigation before any Governmental Entity or before any arbitrator that is pending or has been threatened in writing against Merger Partner that (a) seeks either damages in excess of $100,000 or equitable relief or (b) in any manner challenges or seeks to prevent, enjoin, alter or delay the transactions contemplated by this Agreement,. There are no material judgments, orders or decrees outstanding against Merger Partner.
3.13. Environmental Matters.
(a) Except for such matters that, individually or in the aggregate, have not had, and are not reasonably likely to have, a Merger Partner Material Adverse Effect: (i) Merger Partner has complied in all material respects with all applicable Environmental Laws; (ii) the properties currently or, to the knowledge of Merger Partner, formerly owned, leased or operated by Merger Partner (including soils, groundwater, surface water, buildings or other structures) are and were not contaminated with any Hazardous Substances in a manner or amounts that would be reasonably likely to create a material liability under any Environmental Law or relating to Hazardous Substances; (iii) Merger Partner has not received written notice from any party alleging actual or potential material liability for any Hazardous Substance disposal or contamination on the property of any third party; and (iv) Merger Partner has not released any Hazardous Substance into the environment in a manner or amounts that would be reasonably likely to create a material liability under any Environmental Law or relating to Hazardous Substances.
(b) As of the date of this Agreement, Merger Partner has not received any written notice, demand, letter, claim or request for information alleging that Merger Partner or any of its subsidiaries may be in material violation of or have material liability or obligations under, any Environmental Law.
(c) Merger Partner is not subject to any orders, decrees, injunctions or other arrangements with any Governmental Entity or is subject to any indemnity or other agreement with any third party relating to any material liability under any Environmental Law or relating to Hazardous Substances.
(d) For purposes of this Agreement, the following terms shall have the following meanings:
(i) “Environmental Law” means any Law, regulation, order, decree, permit, authorization, common law or agency requirement of any jurisdiction relating to: (i) the protection, investigation or restoration of the environment, human health and safety (as it relates to exposure to Hazardous Substances) or natural resources; (ii) the handling, use, storage, treatment, presence, disposal, release or threatened release of any Hazardous Substance; or (iii) wetlands, pollution, contamination or any injury or threat of injury to persons or property.
(ii) “Hazardous Substance” means any substance that is: (i) listed, classified, regulated or which falls within the definition of a “hazardous substance,” “hazardous waste” or “hazardous material” pursuant to any Environmental Law; (ii) any petroleum product or by-product, asbestos-containing material, lead-containing paint or plumbing, polychlorinated biphenyls, radioactive materials or radon; or (iii) any other substance that is the subject of regulatory action by any Governmental Entity pursuant to any Environmental Law.
3.14. Employee Benefit Plans.
(a) Merger Partner has made available a complete and accurate copy, as of the date of this Agreement, of all written material Employee Benefit Plans sponsored, maintained, or contributed to (or required to be contributed to), by Merger Partner for the benefit of any current or former employee or other individual service provider of Merger Partner (or such employee or other individual service provider’s beneficiary) or with respect to which Merger Partner or any Merger Partner ERISA Affiliate has any liability (collectively, the “Merger Partner Employee Plans”). No Merger Partner Employee Benefit Plan is sponsored or maintained by a professional employer organization (PEO) or similar provider.
(b) Each Merger Partner Employee Benefit Plan is and has been established and administered in all material respects in accordance with ERISA, the Code, the Patient Protection and Affordable Care Act, including the Health Care and Education Reconciliation Act of 2010, as amended and including any guidance issued thereunder (“ACA”), and all other applicable Laws and the regulations thereunder and in accordance with its terms, and Merger Partner has in all material respects met its obligations with respect to such Merger Partner Employee Benefit Plan and has in all material respects made all required contributions thereto (or reserved such contributions on the Merger Partner Balance Sheet). There is no audit, investigation, litigation or other proceeding (including any voluntary correction application) pending against or involving any Merger Partner Employee Benefit Plan, and to the knowledge of Merger Partner, no such audit, investigation, litigation or other proceeding is threatened.
(c) With respect to Merger Partner Employee Benefit Plans, there are no material benefit obligations for which contributions have not been made or properly accrued and there are no material benefit obligations that have not been accounted for by reserves, or otherwise properly footnoted in accordance with GAAP, on the Financial Statements of Merger Partner.
(d) All Merger Partner Employee Benefit Plans that are intended to be qualified under Section 401(a) of the Code have received determination, advisory or opinion letters from the IRS to the effect that such Merger Partner Employee Plans are qualified and the plans and trusts related thereto are exempt from federal income taxes under Sections 401(a) and 501(a), respectively, of the Code, no such determination, advisory or opinion letter has been revoked and, to the knowledge of Merger Partner, no revocation has been threatened and no event has occurred that would reasonably be expected to adversely affect the qualified status of any such Merger Partner Employee Plan.
(e) Neither Merger Partner nor any of its ERISA Affiliates has (i) ever maintained, contributed to, or had any Liability with respect to an Employee Benefit Plan that was a defined benefit pension plan as defined in Section 3(2) and 3(35) of ERISA or ever subject to Section 412 or 430 of the Code or Section 302 or Title IV of ERISA or (ii) ever been obligated to contribute to a “multiemployer plan” (as defined in Section 3(37) or 4001(a)(3) of ERISA). No Merger Partner Employee Benefit Plan that is intended to be qualified under Section 401(a) of the Code is funded by, associated with or otherwise holds securities issued by Merger Partner. No Merger Partner Employee Benefit Plan is funded by, associated with or related to a “voluntary employees’ beneficiary association” within the meaning of Section 501(c)(9) of the Code. No Merger Partner Employee Benefit Plan is a “multiple employer plan” within the meaning of Section 413(c) of the Code or a “multiple employer welfare arrangement” as defined in Section 3(40) of ERISA.
(f) No Merger Partner Employee Benefit Plan provides, and neither Merger Partner nor any of its ERISA Affiliates has any obligation to provide, any post-termination health, disability or life insurance benefits to any individual, except as required by (i) COBRA or similar state Law or (ii) contractually required subsidies for COBRA coverage during a severance period.
(g) Each Merger Partner Employee Benefit Plan that is a group health plan under Section 733(a)(1) of ERISA has satisfied all obligations under COBRA and each applicable state Law relating to continuation of health coverage for participants and beneficiaries with respect to any qualifying event that has occurred on or before the Closing Date. Neither Merger Partner nor any of the Merger Partner Employee Benefit Plans has incurred (whether or not assessed), and are not reasonably expected to incur or to be subject to, any Tax, penalty, assessment, or other Liability that may be imposed under the ACA or Sections 4980B, 4980D, 4980H, 6721 or 6722 of the Code or with respect to any requirement to timely file ACA information returns with the IRS or provide statements to participants under Section 6056 or 6055 of the Code or state requirements as applicable, or pursuant to Sections 4976 through 4980 of the Code or Title I of ERISA with respect to any of the Merger Partner Employee Benefit Plans. No IRS Letter 226J, 5699, 5698, or IRS Notice 972CG has been issued to or with respect to Merger Partner or any Merger Partner Employee Benefit Plan. No Merger Partner Employee Benefit Plan is “self-insured medical reimbursement plan” as defined in Section 105(h) of the Code.
(h) Except as contemplated by this Agreement or set forth in Section 3.14(g) of the Public Company Disclosure Schedule, neither the execution and delivery of this Agreement nor the consummation of the transactions contemplated hereby (either alone or in conjunction with additional or subsequent events, including any termination of employment or service), will (i) result in any payment (including any severance or bonus payment) becoming due to any current or former employee or other individual service provider of Merger Partner, (ii) result in any forgiveness of indebtedness to any current or former employee or other individual service provider of Merger Partner, (iii) increase, or result in an acceleration of the time of payment or vesting of, the compensation or benefits otherwise due to any current or former employee or other individual service provider of Merger Partner, or (iv) trigger any payment or funding of any compensation or benefits under any Merger Partner Employee Benefit Plan. No Merger Partner Employee Benefit Plan provides, and neither Merger Partner nor any of its ERISA Affiliates has any obligation to provide, any gross-up, indemnification or reimbursement with respect to Taxes incurred under Code Section 4999 or 409A.
(i) Each Merger Partner Employee Benefit Plan that is a “nonqualified deferred compensation plan” (as defined in Section 409A(d)(1) of the Code) materially complies and has complied in form and operation with Section 409A of the Code and all IRS regulations and other guidance promulgated thereunder. No event has occurred that would be treated by Section 409A(b) of the Code as a transfer of property for purposes of Section 83 of the Code.
(j) No Merger Partner Employee Benefit Plan is maintained for the benefit of Merger Partner employees or an ERISA Affiliate outside of the United States or is otherwise subject to the Laws of any jurisdiction other than the United States or a political subdivision thereof.
(k) For purposes of this Agreement, the following terms shall have the following meanings:
(i) “COBRA” means the Consolidated Omnibus Budget Reconciliation Act of 1985, as amended.
(ii) “Employee Benefit Plan” means any “employee pension benefit plan” (as defined in Section 3(2) of ERISA), any “employee welfare benefit plan” (as defined in Section 3(1) of ERISA), any “employee benefit plans” (as defined in Section 3(3) of ERISA), and any other written or oral plan, agreement, program, policy or arrangement involving direct or indirect compensation or benefits, including insurance coverage (including any health, dental, vision and cafeteria plan benefits), employment agreements, severance benefits, disability benefits, fringe benefits, perquisites, change in control benefits, retention benefits, paid time off benefits, nonqualified deferred compensation, bonuses, stock options, stock purchase, phantom stock, stock appreciation or other forms of incentive compensation or post-retirement compensation and all unexpired severance agreements, including, with respect to the Merger Partner Employee Plans, any Merger Partner Stock Plan and with respect to the Public Company employees, any Public Company Stock Plan, or any plan in which a subsidiary or an ERISA Affiliate of either Merger Partner or Public Company could incur Liability that would be a Liability of either the Merger Partner or Public Company.
(iii) “ERISA” means the Employee Retirement Income Security Act of 1974, as amended.
(iv) “ERISA Affiliate” means any Person or entity (whether or not incorporated) that is, or at any applicable time was, treated as a “single employer” or under common control with Merger Partner or Public Company, as applicable, or with any of such Person’s subsidiaries, within the meaning of Section 414 of the Code or Section 4001 of ERISA.
3.15. Compliance With Laws. Except as would not reasonably be expected to result in a Merger Partner Material Adverse Effect, Merger Partner has complied with, is not in violation of, and, as of the date of this Agreement, has not received any notice alleging any violation with respect to, any applicable provisions of any Law related to the conduct of its business or the ownership or operation of its properties or assets.
3.16. Permits and Regulatory Matters.
(a) Merger Partner has all required permits, licenses, registrations, authorizations, certificates, orders, approvals, franchises, variances and other similar rights issued by or obtained from any Governmental Entity (collectively, “Permits”) that are material to the conduct of its business as currently conducted, including all such Permits required by the U.S. Food and Drug Administration (the “FDA”), such as authorization of an Investigational New Drug application (“IND”), or by any other federal, state or foreign agencies or bodies engaged in the regulation of pharmaceutical or biological products (together with the FDA, the “Regulating Authorities”).
(b) All Permits that are necessary for the conduct of the business of Merger Partner as currently conducted (“Merger Partner Authorizations”) are in full force and effect. No Merger Partner Authorization shall cease to be effective as a result of the consummation of the transactions contemplated by this Agreement. Merger Partner is in compliance in all material respects with the terms of each such Merger Partner Authorization. All applications, reports, notices and other documents required to be filed by Merger Partner with any Governmental Entity have been timely filed and are complete and correct in all material respects as of the date filed or as amended prior to the date of this Agreement. Neither Merger Partner, and to Merger Partner’s knowledge, nor any officer, employee or agent of Merger Partner has been convicted of any crime or engaged in any conduct that has previously caused or would reasonably be expected to result in (i) disqualification or debarment by the FDA under 21 U.S.C. Sections 335(a) or (b), or any similar Law of any other Governmental Entity, or (ii) exclusion under 42 U.S.C. Section 1320a-7 or any similar Law of any Governmental Entity.
(c) Merger Partner: (i) is and at all times has been in material compliance, to the extent applicable, with all Laws applicable to the ownership, testing, development, manufacture, packaging, processing, use, distribution, marketing, labeling, promotion, sale, offer for sale, storage, import, export or disposal of any pharmaceutical or biological product tested, developed, promoted, marketed, manufactured or distributed by Merger Partner; (ii) has not received any written notice or correspondence from any Governmental Entity alleging or asserting any material noncompliance with any Merger Partner Authorizations; and (iii) has not received notice that any Governmental Entity has taken or is intending to take action to limit, suspend, modify or revoke any Merger Partner Authorizations (except where such limitation, suspension, modification, or revocation would not reasonably be expected to have a Merger Partner Material Adverse Effect) and, to the knowledge of Merger Partner, there is no action or proceeding pending or threatened against Merger Partner by any Governmental Entity (including any prosecution, injunction, seizure, civil fine, suspension or recall), in each case alleging that Merger Partner is in material noncompliance with any and all applicable Laws, regulations or orders implemented by the FDA or any other Governmental Entity exercising comparable authority. Neither Merger Partner nor any of its officers, employees or, to the knowledge of Merger Partner, agents has made an untrue statement of a material fact or fraudulent statement to any Governmental Entity relating to the Merger Partner Authorizations or failed to disclose a material fact required to be disclosed to any Governmental Entity relating to the Merger Partner Authorizations.
(d) To Merger Partner’s knowledge, all studies, tests, preclinical and clinical investigations and trials sponsored or conducted by, or conducted on behalf of, Merger Partner were and, if still pending, are being conducted in compliance in all material respects with applicable Laws, including, as applicable, Good Manufacturing Practices, Good Clinical Practices and Good Laboratory Practices requirements. The descriptions of, protocols for, and material data and other results of, any such studies, tests and/or trials that have been furnished or made available to Public Company are accurate and complete in all material respects with respect to what is currently known by or available to Merger Partner. Merger Partner is not aware of any studies, tests or trials the results of which would cause Merger Partner to reasonably believe the results would have a material adverse effect on the studies, tests and trials conducted by or on behalf of Merger Partner, and Merger Partner has not received any written notices from any Governmental Entity, institutional review board, independent ethics committee, data and safety monitoring board, or other oversight body with respect to any clinical or pre-clinical studies or tests, or chemistry, manufacturing, and control quality issues, requiring the termination, suspension or material modification of any such studies or tests or chemistry, manufacturing and control activities and, to Merger Partner’s knowledge, there are no facts that would reasonably be expected to give rise to such an action (except where such material modification would not reasonably be expected to have a Merger Partner Material Adverse Effect, such as modifications that are part of routine correspondence with or sponsor-solicited feedback from any Governmental Entity).
3.17. Employees.
(a) All current and past key employees of Merger Partner have entered into confidentiality and assignment of inventions agreements with Merger Partner, a copy or form of which has previously been made available to Public Company. To the knowledge of Merger Partner, as of the date of this Agreement, no employee of Merger Partner is in violation of any term of any patent disclosure agreement, non-competition agreement, or any restrictive covenant to a former employer relating to the right of any such employee to be employed by Merger Partner because of the nature of the business currently conducted by Merger Partner or to the use of Intellectual Property of others. To the knowledge of Merger Partner, as of the date of this Agreement, no key employee or group of key employees has any plans to terminate employment with Merger Partner.
(b) Merger Partner is not nor has been a party to or otherwise bound by any collective bargaining agreement, contract or other agreement or understanding with a labor union or labor organization, nor to the knowledge of Merger Partner, have there been any labor organizing activities with respect to any employees of Merger Partner. Merger Partner is not and has not been the subject of any proceeding asserting that Merger Partner has committed an unfair labor practice or is seeking to compel it to bargain with any labor union or labor organization, nor is there or has there been pending or, to the knowledge of Merger Partner, threatened, any labor strike, dispute, walkout, work stoppage, slow-down or lockout involving Merger Partner.
(c) Except as would not reasonably be expected to result in a Merger Partner Material Adverse Effect, Merger Partner is and has been in compliance with all applicable Laws related to employment (including verification of employment eligibility), employment practices (including without limitation Laws related to discrimination, harassment, and retaliation), terms and conditions of employment and wages and hours (including, without limitation, classification of employees) with respect to any employee (as defined by, or determined in accordance with, applicable Laws). All Workers of Merger Partner are lawfully authorized to work in the United States.
(d) Merger Partner has not received written notice of any material charge or complaint pending before the Equal Employment Opportunity Commission or other Governmental Entity alleging unlawful discrimination, harassment, retaliation or any other violation of or non-compliance with applicable Law relating to the employment, treatment, or termination of any employees of Merger Partner, nor, to the knowledge of Merger Partner, has any such charge been threatened within the preceding twelve (12) months. No current or former employee of Merger Partner has, pursuant to internal complaint procedures, made a written complaint of discrimination, retaliation or harassment, nor to Merger Partner’s knowledge, has an oral complaint of any of the foregoing been made within the preceding twelve (12) months.
(e) Merger Partner has not caused a plant closing as defined in the Worker Adjustment and Retraining Notification Act (the “WARN Act”) affecting any site of employment or one or more operating units within any site of employment, or a mass layoff as defined in the WARN Act, nor have any of the foregoing been affected by any transaction or engaged in layoffs or employment terminations sufficient in number to trigger application of any similar foreign, state or local Law.
3.18. Insurance. Merger Partner maintains insurance policies (the “Merger Partner Insurance Policies”), including insurance covering directors and officers for securities Law and other customary liabilities, with reputable insurance carriers against all risks of a character and in such amounts as are usually insured against by similarly situated companies in the same or similar businesses. Each Merger Partner Insurance Policy is in full force and effect. None of the Merger Partner Insurance Policies shall terminate or lapse (or be affected in any other adverse manner) by reason of any of the transactions contemplated by this Agreement. Merger Partner has complied in all material respects with the provisions of each Merger Partner Insurance Policy under which it is the insured party. No insurer under any Merger Partner Insurance Policy has cancelled or generally disclaimed liability under any such policy or indicated any intent to do so or not to renew any such policy.
3.19. Brokers; Fees and Expenses. Except as set forth on Section 3.19 of the Merger Partner Disclosure Schedule, No agent, broker, investment banker, financial advisor or other firm or Person is or shall be entitled, as a result of any action, agreement or commitment of Merger Partner to any broker’s, finder’s, financial advisor’s or other similar fee or commission in connection with any of the transactions contemplated by this Agreement.
3.20. Certain Business Relationships with Affiliates. No Affiliate of Merger Partner (a) owns any material property or right, tangible or intangible, which is used in the business of Merger Partner, (b) has any material claim or cause of action against Merger Partner or (c) owes any material money to, or is owed any material money by, Merger Partner. Section 3.20 of the Merger Partner Disclosure Schedule describes any material Contracts between Merger Partner and any Affiliate thereof which were entered into or have been in effect at any time since January 1, 2023 other than (i) any employment or service Contracts, invention assignment agreements and other Contracts relating to or entered into in connection with any employment or service, including any Contracts relating to stock purchases and awards, stock options and other equity or equity-based incentive arrangements, in each case relating to compensation or (ii) any arms-length agreements with any portfolio company of any venture capital firm, private equity firm, angel investor, or similar investor of Merger Partner.
3.21. Controls and Procedures, Certifications and Other Matters.
(a) Merger Partner maintains accurate books and records reflecting its assets and liabilities and maintains proper and adequate internal control over financial reporting that provide assurance that (i) transactions are executed with management’s authorization, (ii) transactions are recorded as necessary to permit preparation of the Financial Statements and to maintain accountability for Merger Partner’s consolidated assets, (iii) access to assets of Merger Partner is permitted only in accordance with management’s authorization, (iv) the reporting of assets of Merger Partner is compared with existing assets at regular intervals and (v) accounts, notes and other receivables and inventory were recorded accurately, and proper and adequate procedures are implemented to effect the collection thereof on a current and timely basis.
(b) Merger Partner has not extended or maintained credit, arranged for the extension of credit, modified or renewed an extension of credit, in the form of a personal loan or otherwise, to or for any director or executive officer of Merger Partner.
(c) Merger Partner either (i) satisfies the conditions to qualification as a “smaller reporting company” set forth in 17 C.F.R. 229.10(f)(1), or (ii) if shares of Merger Partner Common Stock were traded on any regulated market or stock exchange, would qualify as a “smaller reporting company,” as defined by 17 C.F.R. 229.10(f)(1).
3.22. Books and Records. The minute books and other similar records of Merger Partner contain accurate summaries, in all material respects, of all actions taken at any meetings of Merger Partner’s stockholders, Merger Partner Board any committee thereof and of all written consents executed in lieu of the holding of any such meeting.
3.23. Ownership of Public Company Common Stock. Except as set forth on Section 3.23 of the Merger Partner Disclosure Schedule, Merger Partner does not and, to the knowledge of Merger Partner, none of Merger Partner’s directors, officers, or 5% or greater stockholders directly or indirectly “owns,” beneficially or otherwise, and at all times during the three-year period prior to the date of this Agreement, to the knowledge of Merger Partner, none of Merger Partner’s directors, officers, or 5% or greater stockholders directly or indirectly has “owned,” beneficially or otherwise, any of the outstanding Public Company Common Stock, as those terms are defined in Section 203 of the DGCL. None of Merger Partner and its “affiliates” or “associates” is, or has been at any time during the two-year period prior to the date of this Agreement, an “interested stockholder” of Public Company or a “beneficial owner” of Public Company Common Stock, as those terms are defined in NRS Chapter 78.
3.24. Privacy and Data Protection.
(a) Merger Partner has complied at all times, and currently complies, in each case, in all material respects, with any applicable data protection and privacy Law with respect to their businesses, including, as applicable, with respect to (i) requirements relating to notification and/or registration of processing of personal data with any applicable national data protection regulator, (ii) requests from data subjects under data protection and Privacy Laws, (iii) where necessary, the obtaining of consent to the processing of personal data and/or direct marketing activity, and (iv) where necessary, the obtaining of any approval, consultation and/or agreement of any applicable works councils or such similar worker representation bodies. Merger Partner has all rights necessary to process all personal data used in the business of the Merger Partner. Merger Partner has not received any written notice or complaint from any individual, third party and/or Governmental Entity, or to the knowledge of Merger Partner, has not been threatened with any written notice or complaint from any individual, third party and/or Governmental Entity (x) alleging non-compliance by Merger Partner with any applicable data protection and privacy Law (including any prohibition or restriction on the transfer of data to any jurisdiction) or (y) claiming compensation for or an injunction for non-compliance with any applicable data protection and privacy Law.
(b) Merger Partner has established and maintains commercially reasonable technical, physical and organizational controls, policies, procedures, safeguards, measures and security systems, plans and technologies in material compliance with requirements under applicable privacy and data protection Laws. Since January 1, 2020, no material breach or material security incident in relation to Merger Partner’s personal data and/or proprietary data has occurred or, to Merger Partner’s knowledge, is threatened, and there has been no actual or, to Merger Partner’s knowledge, threatened unauthorized or illegal processing of, or accidental or unlawful destruction, loss or alteration of, any of Merger Partner’s personal data and/or proprietary data.
3.25. No Other Representations or Warranties. Merger Partner hereby acknowledges and agrees that, except for the representations and warranties contained in this Agreement, none of Public Company, Merger Sub nor any other Person on behalf of Public Company or Merger Sub makes any express or implied representation or warranty with respect to Public Company or Merger Sub or their respective financial condition, business, results of operations, properties, assets, liabilities, or prospects or otherwise or with respect to any other statements made or information provided to Merger Partner or any of its Affiliates in connection with the transactions contemplated hereby, and (subject to the express representations and warranties of Public Company and Merger Sub set forth in Article IV (in each case as qualified and limited by the Public Company Disclosure Schedule) or any representations and warranties of a signatory to any Public Company Support Agreement or Lock-Up Agreements) none of Merger Partner or any of its Affiliates, stockholders, directors, officers, employees, agents, representatives or advisors, or any other Person, has relied on any representations, warranties, statements or information (including the accuracy or completeness thereof).
ARTICLE IV
REPRESENTATIONS AND WARRANTIES OF PUBLIC
COMPANY AND THE MERGER SUB
Except (a) as disclosed in the Public Company SEC Reports filed with the SEC prior to the date hereof and publicly available on the SEC’s Electronic Data Gathering Analysis and Retrieval system (but excluding any disclosures under the heading “Risk Factors” and any disclosure of risks included in any “forward looking statements” disclaimers or in any other section to the extent they are forward-looking statements or cautionary, predictive or forward-looking in nature), or (b) as expressly set forth herein or in the disclosure schedule delivered by Public Company and Merger Sub to Merger Partner on the date of this Agreement (the “Public Company Disclosure Schedule”), Public Company and Merger Sub represent and warrant, jointly and not severally, to Merger Partner as follows:
4.1. Organization, Standing and Power. Each of Public Company and Merger Sub is a corporation duly organized, validly existing and in good standing under the Laws of the jurisdiction of its incorporation, has all requisite corporate power and authority to own, lease and operate its properties and assets and to carry on its business as currently conducted, and is duly qualified to do business and is in good standing (to the extent applicable in such jurisdiction) under the Laws of all jurisdictions in which the character of the properties it owns, operates or leases or the nature of its activities makes such qualification necessary, except for such failures to be so qualified or in good standing, individually or in the aggregate, that have not had, and are not reasonably likely to have, a Public Company Material Adverse Effect. Each of Public Company and Merger Sub has made available to Merger Partner complete and accurate copies of its certificate of incorporation and bylaws, and copies of any amendments thereto, existing as of the date of this Agreement and is not in material default under or in material violation of any provision of any such documents.
4.2. Capitalization.
(a) As of the date of this Agreement, the authorized capital stock of Public Company consists of 3,750,000 shares of Public Company Common Stock and 10,000,000 shares of preferred stock, $0.001 par value per share (“Public Company Preferred Stock”). The rights and privileges of each class of Public Company’s capital stock are as set forth in Public Company’s articles of incorporation (as defined in NRS 78.010(1)(b)). As of the close of business on the Business Day immediately prior to the date of this Agreement, (i) 1,320,240 shares of Public Company Common Stock were issued or outstanding, (ii) no shares of Public Company Common Stock were held in the treasury of Public Company or by subsidiaries of Public Company, and (iii) 15,617 shares of Public Company Preferred Stock were issued or outstanding.
(b) As of the date of this Agreement, there are outstanding options to purchase 135,941 shares of Public Company Common Stock (each, a “Public Company Stock Option” and collectively, the “Public Company Stock Options”). Public Company has made available to Merger Partner complete and accurate copies of all stock or equity related plans, agreements, or arrangements of Public Company (collectively, the “Public Company Stock Plans”) and the forms of all award agreements evidencing such awards. As of the date of this Agreement, Public Company has reserved 9,375 shares of Public Company Common Stock for issuance to employees pursuant to Public Company’s 2023 Employee Stock Purchase Plan (the “Public Company ESPP”), of which 8,337 shares remain available for issuance thereunder as of the date hereof. Public Company has not granted, issued or authorized the grant or issuance of any Public Company Stock Options on the Business Day prior to the date of this Agreement or on the date of this Agreement. With respect to each Public Company Stock Option (whether outstanding or previously exercised) (i) each grant of a Public Company Stock Option was duly authorized no later than the date on which the grant of such Public Company Stock Option was by its terms to be effective by all necessary corporate action, including, as applicable, approval by the Public Company’s Board of Directors (or a duly constituted and authorized committee thereof), or a duly authorized delegate thereof, and any required stockholder approval by the necessary number of votes or written consents, (ii) each such grant was made in accordance with the terms of the applicable Public Company Stock Plan, the Securities Act, the Exchange Act, to the extent applicable, and all other applicable Laws and are not and have not been the subject of any internal investigation, review or inquiry.
(c) Section 4.2(c) of the Public Company Disclosure Schedule lists, as of the date hereof, the number of shares of Public Company Common Stock reserved for future issuance pursuant to warrants or other outstanding rights (other than Public Company Stock Options) to purchase shares of Public Company Common Stock outstanding as of the close of business on the Business Day prior to the date of this Agreement (such outstanding warrants or other rights, the “Public Company Warrants”) and the agreement or other document under which such Public Company Warrants were granted, and the exercise price, the date of grant and the expiration date thereof.
(d) Except (i) as set forth in this Section 4.2 or in Article II, (ii) as reserved for future grants under Public Company Stock Plans, outstanding as of the close of business on the Business Day prior to the date of this Agreement, (iii) as reserved for issuance and issuable upon conversion of outstanding shares of Public Company Preferred Stock, (iv) for the rights to acquire shares pursuant to the Public Company ESPP, and (v) commitments to issue shares of Public Company Common Stock pursuant to the Subscription Agreement in the Concurrent Financing, as of the date of this Agreement, (A) there are no equity securities of any class of Public Company, or any security exchangeable into or exercisable for such equity securities, issued, reserved for issuance or outstanding and (B) there are no options, warrants, equity securities, calls, rights, commitments or agreements of any character to which Public Company or any of its subsidiaries is a party or by which Public Company or any of its subsidiaries is bound obligating Public Company or any of its subsidiaries to issue, exchange, transfer, deliver or sell, or cause to be issued, exchanged, transferred, delivered or sold, additional shares of capital stock or other equity interests of Public Company or any security or rights convertible into or exchangeable or exercisable for any such shares or other equity interests, or obligating Public Company or any of its subsidiaries to grant, extend, accelerate the vesting of, otherwise modify or amend or enter into any such option, warrant, equity security, call, right, commitment or agreement. As of the date of this Agreement, other than the Public Company Support Agreement or pursuant to any Public Company Stock Plan, Public Company is not a party to or is bound by any, and to the knowledge of Public Company, there are no, agreements or understandings with respect to the voting (including voting trusts and proxies) or sale or transfer (including agreements imposing transfer restrictions) of any shares of capital stock or other equity interests of Public Company. Except as contemplated by this Agreement, the Subscription Agreement or described in this Section 4.2(d), there are no registration rights to which Public Company or any of its subsidiaries is a party or by which it or they are bound with respect to any equity security of any class of Public Company. Stockholders of Public Company are not entitled to dissenters’ or appraisal rights under applicable state Law in connection with the Merger, and the Public Company Board has not adopted or approved any resolution pursuant to the NRS or otherwise granting dissenter’s, appraisal or similar rights to any holder of shares of Public Company Common Stock or any other equity interests of or in Public Company, or to any other Person.
(e) All outstanding shares of Public Company Common Stock are, and all shares of Public Company Common Stock subject to issuance as specified in Sections 4.2(b) and 4.2(c) or pursuant to Article II, upon issuance on the terms and conditions specified in the instruments pursuant to which they are issuable, will be, duly authorized, validly issued, fully paid and nonassessable and not subject to or issued in violation of any purchase option, call option, right of first refusal, preemptive right, subscription right or any similar right under any provision of NRS Chapter 78, Public Company’s articles of incorporation or bylaws or any agreement to which Public Company is a party or is otherwise bound.
4.3. Subsidiaries.
(a) Section 4.3(a) of the Public Company Disclosure Schedule sets forth, for each subsidiary of Public Company (including Merger Sub): (i) its name; (ii) the number and type of outstanding equity securities and a list of the holders thereof; and (iii) its jurisdiction of organization.
(b) Each subsidiary of Public Company is a corporation duly organized, validly existing and in good standing under the Laws of the jurisdiction of its incorporation, has all requisite corporate power and authority to own, lease and operate its properties and assets and to carry on its business as currently conducted, and is duly qualified to do business and is in good standing as a foreign corporation in each jurisdiction where the character of its properties owned, operated or leased or the nature of its activities makes such qualification necessary, except for such failures to be so organized, qualified or in good standing, individually or in the aggregate, that have not had, and are not reasonably likely to have, a Public Company Material Adverse Effect. All of the outstanding shares of capital stock and other equity securities or interests of each subsidiary of Public Company are duly authorized, validly issued, fully paid, nonassessable and free of preemptive rights and all such shares (other than directors’ qualifying shares in the case of non-U.S. subsidiaries, all of which Public Company has the power to cause to be transferred for no or nominal consideration to Public Company or Public Company’s designee) are owned, of record and beneficially, by Public Company or another of its subsidiaries free and clear of all Liens, claims, pledges, agreements or limitations in Public Company’s voting rights. There are no outstanding or authorized options, warrants, rights, agreements or commitments to which Public Company or any of its subsidiaries is a party or which are binding on any of them providing for the issuance, disposition or acquisition of any capital stock of any subsidiary of Public Company. There are no outstanding stock appreciation, phantom stock or similar rights with respect to any subsidiary of Public Company. There are no voting trusts, proxies or other agreements or understandings with respect to the voting of any capital stock of any subsidiary of Public Company.
(c) Public Company has made available to Merger Partner complete and accurate copies of the articles of incorporation, bylaws, or other organizational documents of each subsidiary of Public Company.
(d) Except as set forth on Section 4.3(a) of the Public Company Disclosure Schedule, Public Company does not own any shares of capital stock or any interest in any other Person nor does Public Company control directly or indirectly or have any direct or indirect equity participation or similar interest in any Person which is not a subsidiary of Public Company.
4.4. Authority; No Conflict; Required Filings and Consents.
(a) Each of Public Company and Merger Sub has all requisite corporate power and authority to enter into this Agreement and, subject only to the receipt of the approval by the Public Company Stockholders of the Public Company Stockholder Approvals and the adoption of this Agreement by Public Company in its capacity as the sole stockholder of Merger Sub, to consummate the transactions contemplated by this Agreement. Without limiting the generality of the foregoing, Public Company Board, at a duly called meeting at which all directors were present, by the unanimous vote, (i) determined that the Merger is fair to, and in the best interests of Public Company and its stockholders and (ii) directed that the Public Company Stockholder Approvals be submitted to the stockholders of Public Company for their approval and resolved to recommend that the stockholders of Public Company vote in favor of the approval of Public Company Stockholder Approvals. The execution and delivery of this Agreement and the consummation of the transactions contemplated by this Agreement by Public Company and Merger Sub have been duly authorized by all necessary corporate action on the part of each of Public Company and Merger Sub, subject only to the required receipt of the Required Public Company Stockholder Approval and the adoption of this Agreement by Public Company in its capacity as the sole stockholder of Merger Sub. This Agreement has been duly executed and delivered by each of Public Company and Merger Sub and, assuming the due execution and delivery of this Agreement by Merger Partner, constitutes the valid and binding obligation of each of Public Company and Merger Sub, enforceable against Public Company and Merger Sub in accordance with its terms, subject to the Bankruptcy and Equity Exception.
(b) The execution and delivery of this Agreement by each of Public Company and Merger Sub do not, and the consummation by Public Company and Merger Sub of the transactions contemplated by this Agreement shall not, (i) conflict with, or result in any violation or breach of, any provision of the articles of incorporation or bylaws of Public Company or Merger Sub or of the certificate of incorporation or bylaws, or any other organizational document, of any other subsidiary of Public Company, (ii) conflict with, or result in any material violation or breach of, or constitute (with or without notice or lapse of time, or both) a default (or give rise to a right of termination, cancellation or acceleration of any obligation or loss of any material benefit) under, or require a consent or waiver under, constitute a change in control under, require the payment of a penalty under or result in the imposition of any Lien on Public Company’s or any of its subsidiaries’ assets under any of the terms, conditions or provisions of any Contract required to be disclosed in Section 4.11(c) of the Public Company Disclosure Schedule, or (iii) subject to obtaining the Required Public Company Stockholder Approval and compliance with the requirements specified in clauses (i) through (vii) of Section 4.4(c), conflict with or violate any permit, concession, franchise, license, judgment, injunction, order, decree, statute, Law, ordinance, rule or regulation applicable to Public Company or any of its subsidiaries or any of its or their properties or assets, except in the case of clauses (ii) and (iii) of this Section 4.4(b), as would not, individually or in the aggregate, reasonably be expected to result in a Public Company Material Adverse Effect.
(c) No consent, approval, license, permit, order or authorization of, or registration, declaration, notice or filing with, any Governmental Entity or any stock market or stock exchange on which shares of Public Company Common Stock are listed for trading is required by or with respect to Public Company or any of its subsidiaries in connection with the execution and delivery of this Agreement or the consummation by Public Company or Merger Sub of the transactions contemplated by this Agreement, except for (i) the filing of the Certificate of Merger with the Delaware Secretary of State, (ii) the filing of the Registration Statement with the SEC in accordance with the Securities Act, (iii) the filing of the Proxy Statement/Prospectus with the SEC in accordance with the Exchange Act, (iv) the filing of such reports, schedules or materials under Section 13 of or Rule 14a-12 under the Exchange Act and materials under Rule 165 and Rule 425 under the Securities Act as may be required in connection with this Agreement and the transactions contemplated hereby and thereby, (v) such consents, approvals, orders, authorizations, registrations, declarations and filings as may be required under applicable state securities Laws and the Laws of any foreign country, (vi) the filing of an initial listing application for the Public Company Common Stock on Nasdaq with respect to the shares of Public Company Common Stock to be issued pursuant to this Agreement (the “Nasdaq Listing Application”), and (vii) such other consents, authorizations, orders, filings, approvals and registrations that, individually or in the aggregate, if not obtained or made, would not be reasonably expected to result in a Public Company Material Adverse Effect.
(d) The affirmative vote in favor of the Charter Amendment Proposal (as it relates to the Public Company Charter Amendment to increase the number of shares of authorized Public Company Common Stock) by the holders of a majority of the voting power of the outstanding Public Company Common Stock is the only vote of the holders of any class or series of Public Company’s capital stock or other securities of Public Company necessary to approve the Charter Amendment Proposal. The affirmative vote in favor of the Share Issuances and the Other Public Company Voting Proposal by a majority of the votes cast for or against such proposal is the only vote of the holders of any class or series of Public Company’s capital stock or other securities of Public Company necessary to approve the Share Issuances and the Other Public Company Voting Proposal. There are no bonds, debentures, notes or other indebtedness of Public Company having the right to vote (or convertible into, or exchangeable for, securities having the right to vote) on any matters on which stockholders of Public Company may vote.
4.5. SEC Filings; Financial Statements; Information Provided.
(a) Public Company has filed or furnished all registration statements, forms, reports, certifications and other documents required to be filed or furnished by Public Company with the SEC for a period of at least twelve calendar months immediately preceding the execution of this Agreement. All such registration statements, forms, reports, certifications, and other documents, as amended prior to the date hereof, and those that Public Company may file or furnish after the date hereof until the Closing, are referred to herein as the “Public Company SEC Reports.” All of the Public Company SEC Reports (A) were or will be filed or furnished, as applicable on a timely basis, (B) at the time filed or furnished (or if amended prior to the date hereof, when so amended), complied, or will comply when filed or furnished, as to form in all material respects with the requirements of the Securities Act and the Exchange Act applicable to such Public Company SEC Reports and (C) did not or will not at the time they were filed or furnished (or if amended prior to the date hereof, when so amended) or are filed or furnished contain any untrue statement of a material fact or omit to state a material fact required to be stated in such Public Company SEC Reports or necessary in order to make the statements in such Public Company SEC Reports, in the light of the circumstances under which they were made, not misleading, in any material respect.
(b) Each of the consolidated financial statements (including, in each case, any related notes and schedules) contained or to be contained in the Public Company SEC Reports at the time filed (or if amended prior to the date hereof, when so amended) (i) complied or will comply as to form in all material respects with applicable accounting requirements and the published rules and regulations of the SEC with respect thereto, (ii) were or will be prepared in accordance with GAAP applied on a consistent basis throughout the periods involved and at the dates involved (except as may be indicated in the notes to such financial statements or, in the case of unaudited interim financial statements, as permitted by the SEC) and (iii) fairly presented or will fairly present in all material respects the consolidated financial position of Public Company and its subsidiaries as of the dates indicated and the consolidated results of its operations and cash flows for the periods indicated, except that the unaudited interim financial statements were or are subject to normal and recurring year-end adjustments that are not expected to be material in amount. The consolidated balance sheet of Public Company as of March 31, 2024 is referred to herein as the “Public Company Balance Sheet.”
(c) Since January 1, 2024 and prior to the date of this Agreement, Public Company has not received any correspondence from Nasdaq or the staff thereof relating to the delisting or maintenance of listing of the Public Company Common Stock on Nasdaq. As of the date hereof, there are no outstanding or unresolved comments in comment letters received from the SEC staff with respect to any Public Company SEC Reports.
(d) Public Company’s auditor has at all times since its engagement by Public Company been “independent” with respect to Public Company within the meaning of Regulation S-X under the Exchange Act and, to the knowledge of Public Company, in compliance with subsections (g) through (l) of Section 10A of the Exchange Act (to the extent applicable) and the related rules and regulations promulgated by the SEC and the Public Company Accounting Oversight Board thereunder.
(e) The information in the Registration Statement to be supplied by or on behalf of Public Company for inclusion or incorporation by reference in the Registration Statement or supplied by or on behalf of Public Company for inclusion in any Regulation M-A Filing, shall not at the time the Registration Statement or any such Regulation M-A filing is filed with the SEC, at any time it is amended or supplemented or at the time the Registration Statement is declared effective by the SEC, as applicable, contain any untrue statement of a material fact or omit to state any material fact required to be stated therein or necessary in order to make the statements therein in light of the circumstances under which they are made, not false or misleading. The information to be supplied by or on behalf of Public Company for inclusion in the Proxy Statement/Prospectus to be sent to the stockholders of Public Company and Merger Partner in connection with the Public Company Meeting, to solicit the approval by the Public Company’s stockholders of the Public Company Stockholder Approvals, which information shall be deemed to include all material information about or relating to Public Company, the Public Company Stockholder Approvals or the Public Company Meeting, shall not, on the date the Proxy Statement/Prospectus is first mailed to stockholders of Public Company or Merger Partner, or at the time of the Public Company Meeting, contain any untrue statement of a material fact or omit to state a material fact necessary in order to make the statements therein, in the light of the circumstances under which they were made, not misleading; provided, however, Public Company makes no representations or warranties as to the information contained in or omitted from the Registration Statement or the Proxy Statement/Prospectus in reliance upon and in conformity with information furnished in writing to Public Company by or on behalf of Merger Partner specifically for inclusion in the Registration Statement or the Proxy Statement/Prospectus which is misleading by virtue of such reliance or conformity.
4.6. No Undisclosed Liabilities. Public Company does not have any material Liability, except for (a) Liabilities shown on the Public Company Balance Sheet, (b) Liabilities of a type required to be shown on the Public Company Balance Sheet that have arisen since the date of the Public Company Balance Sheet in the Ordinary Course of Business (and which have not resulted from a breach of contract, breach of warranty, tort, infringement or violation of Law), (c) liabilities for transaction expenses incurred in connection with the transactions contemplated by this Agreement, and (d) executory performance obligations under Contracts.
4.7. Absence of Certain Changes or Events. During the period beginning on the date of the Public Company Balance Sheet and ending on the date hereof, Public Company and its subsidiaries have conducted their respective businesses only in the Ordinary Course of Business and, since such date, there has not been (a) any change, event, circumstance, development or effect that, individually or in the aggregate, has had, or is reasonably expected to have, a Public Company Material Adverse Effect or (b) any other action or event that would have required the consent of Merger Partner pursuant to Section 5.2 had such action or event occurred after the date of this Agreement.
4.8. Taxes.
(a) Except as set forth in Section 4.8(a) of the Public Company Disclosure Schedule, each of Public Company and its subsidiaries has properly filed on a timely basis all income and other material Tax Returns that it was required to file, and all such Tax Returns are true, correct and complete in all material respects. Each of Public Company and its subsidiaries has paid on a timely basis all Taxes, whether or not shown on any Tax Return, that were due and payable.
(b) Neither Public Company nor any of its subsidiaries is or has ever been a member of an affiliated group with which it has filed (or been required to file) consolidated, combined, unitary or similar U.S. federal Tax Returns, other than a group of which the common parent is Public Company. With the exception of customary commercial leases or contracts that are not primarily related to Taxes entered into in the Ordinary Course of Business and liabilities thereunder, neither Public Company nor any of its subsidiaries (i) has any liability under Treasury Regulations Section 1.1502-6 (or any comparable or similar provision of state, local or non-U.S. Law), as a transferee or successor, pursuant to any contractual obligation, or otherwise for any Taxes of any Person other than Public Company or any of its subsidiaries, or (ii) is a party to or bound by any Tax indemnity, Tax sharing, Tax allocation or similar agreement.
(c) All material Taxes that Public Company or any of its subsidiaries was required by Law to withhold or collect have been duly withheld or collected and, to the extent required, have been properly paid to the appropriate Governmental Entity, in each case in compliance in all material respects with applicable Law.
(d) Public Company has delivered or made available to Merger Partner (i) complete and correct copies of all income and other material Tax Returns of Public Company and any of its subsidiaries relating to Taxes for all taxable periods for which the applicable statute of limitations has not yet expired, (ii) complete and correct copies of all private letter rulings, revenue agent reports, information document requests, notices of proposed deficiencies, deficiency notices, protests, petitions, closing agreements, settlement agreements, pending ruling requests and any similar documents submitted by, received by, or agreed to by or on behalf of Public Company or any of its subsidiaries relating to Taxes for all taxable periods for which the statute of limitations has not yet expired, and (iii) complete and correct copies of all material agreements, rulings, settlements or other Tax documents with or from any Governmental Entity relating to Tax incentives of Public Company or any of its subsidiaries.
(e) Except as set forth in Section 4.8(e) of the Public Company Disclosure Schedule, no examination or audit of any Tax Return of Public Company or any of its subsidiaries by any Governmental Entity is currently in progress or, to the knowledge of Public Company, has been threatened by any Governmental Entity. No deficiencies for Taxes of Public Company or any of its subsidiaries have been claimed, proposed or assessed by any Governmental Entity in writing. Neither Public Company nor any of its subsidiaries has been informed in writing by any jurisdiction in which Public Company or any of its subsidiaries does not file a Tax Return that the jurisdiction believes that Public Company or any of its subsidiaries was required to file any Tax Return that was not filed or is subject to Tax in such jurisdiction. Since January 1, 2021, neither Public Company nor any of its subsidiaries has (i) waived any statute of limitations with respect to Taxes or agreed to extend the period for assessment or collection of any Taxes (other than any automatic extension granted in the Ordinary Course of Business and consistent with past custom and practice of Public Company), which waiver or extension is still in effect, (ii) requested any extension of time within which to file any Tax Return (other than any extension granted in the Ordinary Course of Business and consistent with past custom and practice of Public Company), or (iii) executed or filed any power of attorney with any taxing authority, which is still in effect.
(f) Neither Public Company nor any of its subsidiaries has made any payment or provided any benefit, is obligated to make any payment or provide any benefit, or is a party to any plan, program, policy, agreement or arrangement that could obligate it to make, and neither the execution and delivery of this Agreement nor the consummation of the transactions contemplated hereby (either alone or in conjunction with additional or subsequent events, including any termination of employment or service), will result in any payment or provide any benefit that may be treated as an “excess parachute payment” under Section 280G of the Code (without regard to Sections 280G(b)(4) and 280G(b)(5) of the Code).
(g) Neither Public Company nor any of its subsidiaries (i) is a party to any joint venture, partnership, or other arrangement that is treated as a partnership for U.S. federal income Tax purposes or (ii) since the date of its formation or the formation of any predecessor thereof, has always been, for all U.S. federal income tax purposes, classified as an association taxable as a C corporation.
(h) Neither Public Company nor any of its subsidiaries has distributed to its stockholders or security holders stock or securities of a controlled corporation, nor has stock or securities of Public Company or any of its subsidiaries been distributed, in a transaction to which Section 355 of the Code applies in the two years prior to the date of this Agreement.
(i) There are no Liens with respect to Taxes upon any of the assets or properties of Public Company or any of its subsidiaries, other than with respect to Taxes not yet due and payable or being contested in good faith by appropriate proceedings.
(j) Neither Public Company nor any of its subsidiaries will be required to include any material item of income in, or exclude any material item of deduction from, taxable income for any period (or any portion thereof) ending after the Closing Date as a result of any (i) adjustments under Section 481 of the Code (or any similar adjustments under any provision corresponding non-U.S., state or local Tax Laws) for a taxable period (or portion thereof) ending on or prior to the Closing Date, (ii) installment sale or other open transaction disposition made on or prior to the Closing Date, or (iii) prepaid amount or deferred revenue received on or prior to the Closing Date outside the Ordinary Course of Business.
(k) Neither Public Company nor any of its subsidiaries has participated in any “reportable transaction” as defined in Treasury Regulations Section 1.6011-4(b).
(l) Neither Public Company nor any of its subsidiaries is subject to income Tax in any country other than its country of incorporation, organization or formation by virtue of having employees, a permanent establishment or other fixed place of business in that country.
(m) All related party transactions involving Public Company or any of its subsidiaries have been conducted at arm’s length in compliance with Section 482 of the Code and the Treasury Regulations promulgated thereunder and any comparable provisions of any other Tax Law.
(n) Neither Public Company nor any of its Affiliates has taken or agreed to take any action, has omitted to take any action, or has any knowledge of any fact or circumstance, the taking, omission, or existence of which, as the case may be, would reasonably be expected to prevent the Merger from constituting a transaction qualifying as a reorganization under Section 368(a) of the Code and, in the event the Control Requirement is satisfied, as a non-taxable exchange of shares of Merger Partner Capital Stock for shares of Public Company Common Stock under Section 351(a) of the Code.
4.9. Owned and Leased Real Properties.
(a) Neither Public Company nor any of its subsidiaries owns or has ever owned any real property, nor is either party to any agreement to purchase or sell any real property.
(b) Neither the Public Company nor any of its subsidiaries as of the date of this Agreement leases, subleases, licenses or otherwise occupies any real property nor is party to any lease, sublease, license or any other occupancy agreement (collectively, the “Public Company Leases”) and all of its previous Public Company Leases have been terminated and neither Public Company nor any of its subsidiaries has any remaining affirmative obligations under such Public Company Leases and termination agreements. Neither the Public Company nor any of its subsidiaries is party to any agreement or subject to any claim that may require the payment of any real estate brokerage commissions. Neither Public Company nor any of its subsidiaries nor, to the knowledge of Public Company, any other party is in breach or default and no event has occurred, is pending or, to the knowledge of Public Company, is threatened, which, after the giving of notice, with lapse of time, or otherwise, would constitute any such breach or default under any of the Public Company Leases, except where the existence of such defaults, individually or in the aggregate, has not had, and is not reasonably likely to result in, the loss of a material right or in a material liability of Public Company or any of its subsidiaries. Neither Public Company nor any of its subsidiaries leases, subleases or licenses any real property to any Person other than Public Company and its subsidiaries. Public Company has made available to Merger Partner complete and accurate copies of all Public Company Leases.
4.10. Intellectual Property.
(a) Section 4.10(a) of the Public Company Disclosure Schedule lists all Public Company Registrations, in each case enumerating specifically the applicable filing or registration number, title, jurisdiction in which filing was made or from which registration issued, date of filing or issuance, and names of all current applicant(s) and registered owners(s), as applicable, except that, for any Public Company Registrations that are Internet domain names or social media accounts and identifiers, such enumeration shall be the applicable account name or number, the domain registrar or social media company and the registered owner(s). Except as set forth on Section 4.10(a) of the Public Company Disclosure Schedule, all assignments of Public Company Registrations to Public Company have been properly executed and recorded, and, to the knowledge of Public Company, all issuance, renewal, maintenance and other payments that are or have become due with respect thereto have been timely paid (other than any such payments for which a bona fide grace period applies under applicable Law) by or on behalf of the Public Company. To the knowledge of Public Company, all Public Company Registrations are valid and enforceable.
(b) There are no inventorship challenges, inter partes proceedings, opposition or nullity proceedings or interferences declared, commenced or provoked, or, to the knowledge of Public Company, threatened, with respect to any Patent Rights included in the Public Company Registrations. Except as set forth on Section 4.10(b) of the Public Company Disclosure Schedule, none of the Patent Rights included in the Public Company Registrations have been abandoned. Public Company has complied with its duty of candor and disclosure to the United States Patent and Trademark Office and any relevant foreign patent office with respect to all patent and trademark applications filed by or on behalf of Public Company and has made no material misrepresentation in such applications. Public Company has no knowledge of any information that would preclude Public Company from having clear title to the Public Company Registrations.
(c) Public Company is the sole and exclusive owner of all Public Company Owned Intellectual Property, free and clear of any Liens, other than any joint owners of the Public Company Owned Intellectual Property listed in Section 4.10(c) of the Public Company Disclosure Schedule. None of the Public Company Intellectual Property is subject to any orders, decrees or injunctions.
(d) Public Company has taken reasonable measures to protect the proprietary nature of each item of Public Company Owned Intellectual Property, and to maintain in confidence all trade secrets and confidential information comprising a part thereof. To Public Company’s knowledge, there has been no unauthorized disclosure of any third party proprietary or confidential information in the possession, custody or control of Public Company.
(e) To the knowledge of Public Company, the operations of Public Company and its subsidiaries as currently conducted do not infringe, misappropriate or otherwise violate and have not in the past five years infringed, misappropriated or otherwise violated any valid and enforceable Intellectual Property rights of any individual or entity. To Public Company’s knowledge, no individual or entity has infringed, misappropriated or otherwise violated the Public Company Owned Intellectual Property or any rights under the Public Company Licensed Intellectual Property that are exclusively licensed to Public Company or any of its subsidiaries, and neither Public Company nor any of its subsidiaries has filed or threatened in writing any claims alleging that a third party or Worker has infringed, misappropriated or otherwise violated any Public Company Intellectual Property. No individual or entity has filed and served upon Public Company or any of its subsidiaries or, to Public Company’s knowledge, threatened or otherwise filed any action or proceeding alleging that Public Company or any of its subsidiaries has infringed, misappropriated or otherwise violated any individual’s or entity’s Intellectual Property rights nor has Public Company or any of its subsidiaries received any written notification that a license under any other individual’s or entity’s Intellectual Property is or may be required.
(f) Public Company has made available to Merger Partner copies of all material written complaints, claims, notices or threats, or disclosed to Merger Partner all material non-written complaints, claims, notices or threats, in each case, concerning the infringement, violation or other misappropriation of any Public Company Intellectual Property.
(g) Section 4.10(g) of the Public Company Disclosure Schedule identifies each (i) license or other agreement pursuant to which Public Company has granted rights to any Public Company Licensed Intellectual Property, and (ii) each agreement, contract, assignment or other instrument pursuant to which Public Company has granted any joint ownership interest in or to each item of Public Company Owned Intellectual Property, in each case (i) and (ii) other than Excluded Contracts.
(h) Section 4.10(h) of the Public Company Disclosure Schedule identifies (i) each license or agreement pursuant to which Public Company has obtained rights to any Public Company Licensed Intellectual Property (excluding generally available, off the shelf software programs that are licensed by Public Company pursuant to “shrink wrap” licenses, the total fees associated with which are less than $50,000) and (ii) each agreement, contract, assignment or other instrument pursuant to which Public Company has obtained any joint or sole ownership interest in or to each item of Public Company Owned Intellectual Property, in each case (i) and (ii) other than Excluded Contracts.
(i) To Public Company’s knowledge, no Worker of Public Company or any of its subsidiaries is in default or breach of any term of any employment Contract, non-disclosure Contract, assignment of invention Contract or similar Contract between such Worker and Public Company or any of its subsidiaries, as applicable, relating to the protection, ownership, development, use, assignment or transfer of Public Company Intellectual Property. To the extent that any Public Company Owned Intellectual Property has been conceived, reduced to practice, authored, developed or created for Public Company or any of its subsidiaries by any individual while a Worker, Public Company or such subsidiary has obtained the entire and unencumbered right, title and interest therein and thereto by operation of Law or by valid written assignment.
(j) The execution and delivery of this Agreement by Public Company does not, and the consummation by Public Company of the transactions contemplated by this Agreement shall not, result in (i) a material breach of or default under any agreement governing any Public Company Intellectual Property; (ii) the grant or transfer to any third party of any new license or other interest under, the abandonment, assignment to any third party, or modification or loss of any right with respect to, any Public Company Intellectual Property; (iii) the grant or transfer to any third party of any license or other interest under, or any covenant not to sue in respect of, any Merger Partner Intellectual Property; or (iv) Merger Partner, Public Company or any of their respective Affiliates being obligated to pay any penalty or new or increased royalty or fee to any individual or entity under any agreement governing any Public Company Intellectual Property.
(k) For purposes of this Agreement, the following terms shall have the following meanings:
(i) “Public Company Intellectual Property” shall mean the Public Company Owned Intellectual Property and the Public Company Licensed Intellectual Property.
(ii) “Public Company Licensed Intellectual Property” shall mean all Intellectual Property that is licensed to Public Company or any of its subsidiaries by any individual or entity other than Public Company or any of its subsidiaries, as applicable, excluding any Intellectual Property that is licensed to Public Company or any of its subsidiaries under Excluded Contracts.
(iii) “Public Company Owned Intellectual Property” shall mean all Intellectual Property owned or purported to be owned by Public Company or any of its subsidiaries, in whole or in part.
(iv) “Public Company Registrations” shall mean Intellectual Property Registrations that are registered or filed in the name of Public Company, or where Merger Partner is the assignee thereof, in each case, alone or jointly with others.
4.11. Contracts.
(a) Section 4.11(a) of the Public Company Disclosure Schedule lists the following Contracts of Public Company and its subsidiaries in effect as of the date of this Agreement (in each case, excluding Excluded Contracts):
(i) any Contract (or group of related Contracts) for the purchase or sale of products or for the furnishing or receipt of services (A) which expressly requires future payments by or to Public Company or any of its subsidiaries in excess of $50,000 in the aggregate, or (B) in which Public Company or any of its subsidiaries has granted manufacturing rights, “most favored nation” pricing provisions or marketing or distribution rights relating to any products or territory, or has agreed to purchase goods or services exclusively from a particular party or to a right of first offer, right of first refusal, right of first negotiation in favor of any third party;
(ii) any Contract under which Public Company has granted to a third party a license under, or option or covenant not to sue with respect to, any Public Company Intellectual Property, except for any Excluded Contract;
(iii) any Contract under which Public Company or any of its subsidiaries is prohibited from selling, licensing or otherwise distributing any of its technology or products, or providing services to, customers or potential customers or any class of customers, in any geographic area, during any period of time or any segment of the market or line of business;
(iv) any (A) employment Contract (excluding offer letters for at-will employment that do not provide for severance or for advance notice of termination or for any change of control, transaction, retention or other special remuneration) that provides for base salary and target bonus, when taken together, of at least $200,000 and (B) individual independent contractor or consulting Contract that involves or could involve payments in excess of $50,000 within any twelve (12) month period;
(v) any Contract, plan, policy or program providing for severance, termination compensation, retention or stay pay, change in control payments or transaction-based bonuses;
(vi) any settlement Contract or settlement-related Contact (including any Contract in connection with which any employment-related claim is settled) under which either side has remaining financial obligations;
(vii) any dealer, distribution, joint marketing, joint venture, joint development, partnership, strategic alliance, collaboration, development agreement or outsourcing arrangement, other than Excluded Contracts;
(viii) any Contract for the conduct of research studies, pre-clinical or clinical studies, manufacturing, distribution, supply, marketing or co-promotion of any products in development or which is being marketed, distributed, supported, sold or licensed out, in each case by or on behalf of Public Company or any of its subsidiaries; and
(ix) any Contract that involved or would reasonably be expected to result in (A) the grant or transfer to any third party of any new license or other interest under, the abandonment, assignment to any third party, or modification or loss of any right with respect to, or the creation of any Lien (excluding a Permitted Lien) on any Public Company Intellectual Property, (B) the grant or transfer to any third party of any license or other interest under, or any covenant not to sue with respect to any Merger Partner Intellectual Property, or (C) Public Company or any of its subsidiaries being obligated to pay any penalty or new or increased royalty or fee to any individual or entity under any agreement governing any Public Company Intellectual Property.
(b) Public Company has made available to Merger Partner a complete and accurate copy of each Contract listed in Sections 4.10(a), 4.10(g), 4.10(h) and 4.11(a) of the Public Company Disclosure Schedule. With respect to each Contract so listed or that should be listed: (i) the Contract is legal, valid, binding and enforceable and in full force and effect against Public Company and/or its subsidiaries, as applicable, and, to the knowledge of Public Company, against each other party thereto, as applicable, subject to the Bankruptcy and Equity Exception; and (ii) none of Public Company, its subsidiaries nor, to the knowledge of Public Company, any other party, is in material breach or violation of, or default under, any such Contract no event has occurred, is pending or, to the knowledge of Public Company, is threatened, which, with or without notice or lapse of time, or both, would constitute a material breach or default by Public Company, its subsidiaries or, to the knowledge of Public Company, any other party under such Contract, except for such breaches, violations or defaults that, individually or in the aggregate, have not had, and are not reasonably likely to have, a Public Company Material Adverse Effect.
4.12. Litigation. As of the date of this Agreement, there is no action, suit, proceeding, claim, arbitration or investigation before any Governmental Entity or before any arbitrator that is pending or has been threatened in writing against Public Company or any of its subsidiaries that (a) seeks either damages in excess of $50,000 or equitable relief or (b) in any manner challenges or seeks to prevent, enjoin, alter or delay the transactions contemplated by this Agreement. There are no material judgments, orders or decrees outstanding against Public Company or any of its subsidiaries.
4.13. Environmental Matters.
(a) Except for such matters that, individually or in the aggregate, have not had, and are not reasonably likely to have, a Public Company Material Adverse Effect: (i) Public Company has complied in all material respects with all applicable Environmental Laws; (ii) the properties currently or, to the knowledge of Public Company, formerly owned, leased or operated by Public Company (including soils, groundwater, surface water, buildings or other structures) are and were not contaminated with any Hazardous Substances in a manner or amounts that would be reasonably likely to create a material liability under any Environmental Law or relating to Hazardous Substances; (iii) Public Company has not received written notice from any party alleging actual or potential material liability for any Hazardous Substance disposal or contamination on the property of any third party; and (iv) Public Company has not released any Hazardous Substance into the environment in a manner or amounts that would be reasonably likely to create a material liability under any Environmental Law or relating to Hazardous Substances.
(b) As of the date of this Agreement, neither Public Company nor any of its subsidiaries has received any written notice, demand, letter, claim or request for information alleging that Public Company or any of its subsidiaries may be in material violation of or have material liability or obligations under, any Environmental Law.
(c) Neither Public Company nor any of its subsidiaries is subject to any orders, decrees, injunctions or other arrangements with any Governmental Entity or is subject to any indemnity or other agreement with any third party relating to any material liability under any Environmental Law or relating to Hazardous Substances.
4.14. Employee Benefit Plans.
(a) Public Company has made available a complete and accurate copy, as of the date of this Agreement, of all written material Employee Benefit Plans sponsored, maintained, or contributed to (or required to be contributed to), by Public Company or any of its subsidiaries for the benefit of any current or former employee or other individual service provider of Public Company, and of its ERISA Affiliates or any of its subsidiaries (or such employee or other individual service provider’s beneficiary) or with respect to which Public Company or any of its subsidiaries have any liability (collectively, the “Public Company Employee Plans”).
(b) Each Public Company Employee Plan is and has been established and administered in all material respects in accordance with ERISA, the Code, the ACA and all other applicable Laws and the regulations thereunder and in accordance with its terms and each of Public Company and its subsidiaries has in all material respects met its obligations with respect to such Public Company Employee Plan and has in all material respects made all required contributions thereto (or reserved such contributions on the Public Company Balance Sheet). There is no audit, investigation or other proceeding (including any voluntary correction application) pending against or involving any Public Company Employee Plan, and to the knowledge of Public Company, no such audit, investigation or other proceeding is threatened.
(c) With respect to Public Company Employee Plans, there are no material benefit obligations for which contributions have not been made or properly accrued and there are no material benefit obligations that have not been accounted for by reserves, or otherwise properly footnoted in accordance with GAAP, on the financial statements of Public Company or any of its subsidiaries.
(d) All Public Company Employee Plans that are intended to be qualified under Section 401(a) of the Code have received determination or opinion letters from the IRS to the effect that such Public Company Employee Plans are qualified and the plans and trusts related thereto are exempt from federal income taxes under Sections 401(a) and 501(a), respectively, of the Code, no such determination or opinion letter has been revoked and, to the knowledge of Public Company, no revocation has been threatened and no event has occurred that would reasonably be expected to adversely affect the qualified status of any such Public Company Employee Plan.
(e) Neither Public Company nor any of its subsidiaries nor any of their respective ERISA Affiliates has (i) ever maintained, contributed to, or had any Liability with respect to an Employee Benefit Plan that is or was a defined benefit pension plan as defined in Section 3(2) and 3(35) of ERISA or was ever subject to Section 412 or 430 of the Code or Title IV of ERISA or (ii) ever been obligated to contribute to a “multiemployer plan” (as defined in Section 3(37) 4001(a)(3) of ERISA). No Public Company Employee Plan that is intended to be qualified under Section 401(a) of the Code is funded by, associated with or related to a “voluntary employees’ beneficiary association” within the meaning of Section 501(c)(9) of the Code. No Public Company Employee Plan is funded with or otherwise holds securities issued by Merger Partner or any of its subsidiaries. No Public Company Employee Plan is a “multiple employer plan” within the meaning of Section 413(c) of the Code or a “multiple employer welfare arrangement” as defined in Section 3(40) of ERISA.
(f) No Public Company Employee Plan, and neither Public Company nor any of its ERISA Affiliates has any obligation to provide, any provides post-termination health, disability or life insurance benefits to any individual, except as required by (i) COBRA or similar state Law or (ii) contractually required subsidies for COBRA coverage during a severance period.
(g) Each Public Company Employee Benefit Plan that is a group health plan under Section 733(a)(1) of ERISA has satisfied all obligations under COBRA and each applicable state Law relating to continuation of health coverage for participants and beneficiaries with respect to any qualifying event that has occurred on or before the Closing Date. Neither Public Company nor any of the Public Company Employee Benefit Plans has incurred (whether or not assessed), and are not reasonably expected to incur or to be subject to, any Tax, penalty, assessment, or other Liability that may be imposed under the ACA or Sections 4980B, 4980D, 4980H, 6721 or 6722 of the Code or with respect to any requirement to timely file ACA information returns with the IRS or provide statements to participants under Section 6056 or 6055 of the Code or state requirements as applicable, or pursuant to Sections 4976 through 4980 of the Code or Title I of ERISA with respect to any of the Public Company Employee Benefit Plans. No IRS Letter 226J, 5699, 5698, or IRS Notice 972CG has been issued to or with respect to Public Company or any Public Company Employee Benefit Plan. No Public Company Employee Benefit Plan is “self-insured medical reimbursement plan” as defined in Section 105(h) of the Code.
(h) Except as contemplated by this Agreement or set forth in Section 4.14(h) of the Public Company Disclosure Schedule, neither the execution and delivery of this Agreement nor the consummation of the transactions contemplated hereby (either alone or in conjunction with additional or subsequent events, including any termination of employment or service), will (i) result in any payment (including any severance or bonus payment) becoming due to any current or former employee or other individual service provider of Public Company or any of its subsidiaries, (ii) result in any forgiveness of indebtedness to any current or former employee or other individual service provider of Public Company or any of its subsidiaries, (iii) increase, or result in an acceleration of the time of payment or vesting of, the compensation or benefits otherwise due to any current or former employee or other individual service provider of Public Company or any of its subsidiaries, or (iv) trigger any payment or funding of any compensation or benefits under any Public Company Employee Plan. No Public Company Employee Plan provides, and neither Public Company nor any of its ERISA Affiliates has any obligation to provide, any gross-up, indemnification or reimbursement with respect to Taxes incurred under Code Section 4999 or 409A.
(i) Each Public Company Employee Plan that is a “nonqualified deferred compensation plan” (as defined in Section 409A(d)(1) of the Code) materially complies and has complied in form and operation with Section 409A of the Code and all IRS regulations and other guidance thereunder. No event has occurred that would be treated by Section 409A(b) of the Code as a transfer of property for purposes of Section 83 of the Code.
4.15. Compliance With Laws. Except as would not reasonably be expected to result in a Public Company Material Adverse Effect, Public Company and each of its subsidiaries has complied with, is not in material violation of, and, as of the date of this Agreement, has not received any notice alleging any violation with respect to, any applicable provisions of any Law related to the conduct of its business or the ownership or operation of its properties or assets.
4.16. Permits and Regulatory Matters.
(a) Public Company and each of its subsidiaries have all required Permits that are material to the conduct of their businesses as currently conducted, including all such Permits required by the FDA, such as authorization of an IND, or by any other Regulating Authorities (the “Public Company Authorizations”).
(b) All Permits that are necessary for the conduct of the business of Public Company as currently conducted are in full force and effect. No Public Company Authorization shall cease to be effective as a result of the consummation of the transactions contemplated by this Agreement. Public Company and its subsidiaries are in compliance in all material respects with the terms of each such Public Company Authorization. All applications, reports, notices and other documents required to be filed by Public Company and its subsidiaries with all Governmental Entities have been timely filed and are complete and correct in all material respects as of the date filed or as amended prior to the date of this Agreement. None of Public Company and its subsidiaries, and to Public Company’s knowledge, any officer, employee or agent of Public Company or any of its subsidiaries has been convicted of any crime or engaged in any conduct that has previously caused or would reasonably be expected to result in (i) disqualification or debarment by the FDA under 21 U.S.C. Sections 335(a) or (b), or any similar Law of any other Governmental Entity, or (ii) exclusion under 42 U.S.C. Section 1320a-7 or any similar Law of any Governmental Entity.
(c) (i) Public Company and its subsidiaries are and at all times have been in material compliance, to the extent applicable, with all Laws applicable to the ownership, testing, development, manufacture, packaging, processing, use, distribution, marketing, labeling, promotion, sale, offer for sale, storage, import, export or disposal of any pharmaceutical or biological product tested, developed, promoted, marketed, manufactured or distributed by Public Company; (ii) neither Public Company nor any of its subsidiaries has received any written notices or correspondence from any Governmental Entity alleging or asserting any material noncompliance with any Public Company Authorizations; and (iii) neither Public Company nor any of its subsidiaries has received notice that any Governmental Entity has taken or is intending to take action to limit, suspend, modify or revoke any Public Company Authorizations (except where such limitation, suspension, modification, or revocation would not reasonably be expected to have a Public Company Material Adverse Effect) and, to the knowledge of Public Company, there is no action or proceeding pending or threatened against Public Company by a Governmental Entity (including any prosecution, injunction, seizure, civil fine, suspension or recall), in each case alleging that Public Company or any of its subsidiaries is in material noncompliance with any and all applicable Laws, regulations or orders implemented by the FDA or any other Governmental Entity exercising comparable authority. Neither Public Company nor any of its subsidiaries nor any of their respective officers, employees or, to the knowledge of Public Company, agents has made an untrue statement of a material fact or fraudulent statement to any Governmental Entity relating to the Public Company Authorizations or failed to disclose a material fact required to be disclosed to any Governmental Entity relating to the Public Company Authorizations.
(d) To Public Company’s knowledge, all studies, tests, preclinical and clinical investigations and trials sponsored or conducted by, or conducted on behalf of, Public Company were and, if still pending, are being conducted in compliance in all material respects with applicable Laws, including, as applicable, Good Manufacturing Practices, Good Clinical Practices and Good Laboratory Practices requirements. The descriptions of, protocols for, and material data and other results of, any such studies, tests and/or trials that have been furnished or made available to Merger Partner are accurate and complete in all material respects with respect to what is currently known by or available to Public Company. Public Company is not aware of any studies, tests or trials the results of which would cause Public Company to reasonably believe the results would have a material adverse effect on the studies, tests and trials conducted by or on behalf of Public Company, and Public Company has not received any written notices from any Governmental Entity, institutional review board, independent ethics committee, data and safety monitoring board, or other oversight body with respect to any clinical or pre-clinical studies or tests, or chemistry, manufacturing, and control quality issues, requiring the termination, suspension or material modification of any such studies, tests or chemistry, manufacturing and control activities and, to Public Company’s knowledge, there are no facts that would reasonably be expected to give rise to such an action (except where such material modification would not reasonably be expected to have a Public Company Material Adverse Effect, such as modifications that are part of routine correspondence with or sponsor-solicited feedback from any Governmental Entity).
4.17. Employees.
(a) All current employees of Public Company have entered into confidentiality and assignment of inventions agreements with Public Company, a copy or form of which has previously been made available to Merger Partner. To the knowledge of Public Company, as of the date of this Agreement, no employee of Public Company or any subsidiary of Public Company is in violation of any term of any patent disclosure agreement, non-competition agreement, or any restrictive covenant to a former employer relating to the right of any such employee to be employed by Public Company or any of its subsidiaries because of the nature of the business currently conducted by Public Company or any of its subsidiaries or to the use of trade secrets or proprietary information of others. To the knowledge of Public Company, as of the date of this Agreement, no key employee or group of key employees has any plans to terminate employment with Public Company or its subsidiaries.
(b) Since January 1, 2020, neither Public Company nor any of its subsidiaries is or has been a party to or otherwise bound by any collective bargaining agreement, contract or other agreement or understanding with a labor union or labor organization, nor to the knowledge of Public Company and its subsidiaries, have there been any labor organizing activities with respect to any employees of Public Company or any of its subsidiaries. Neither Public Company nor any of its subsidiaries is or has been the subject of any proceeding asserting that Public Company or any of its subsidiaries has committed an unfair labor practice or is seeking to compel it to bargain with any labor union or labor organization, nor is there or has there been pending or, to the knowledge of Public Company, threatened, any labor strike, dispute, walkout, work stoppage, slow-down or lockout involving Public Company or any of its subsidiaries.
(c) Except as would not reasonably be expected to result in a Public Company Material Adverse Effect, Public Company and its subsidiaries are and have been in material compliance with all applicable Laws related to employment (including verification of employment eligibility), employment practices (including Laws related to discrimination, harassment, and retaliation), terms and conditions of employment and wages and hours (including, without limitation, classification of employees) with respect to any employee (as defined by, or determined in accordance with, applicable Laws). To the knowledge of Public Company, all Workers of Public Company and its subsidiaries are lawfully authorized to work in the United States or European Union, as applicable.
(d) Neither Public Company nor any of its subsidiaries has received written notice of any material charge or complaint pending before the Equal Employment Opportunity Commission or other Governmental Entity alleging unlawful discrimination, harassment, retaliation or any other violation of or non-compliance with applicable Law by Public Company or any of its subsidiaries relating to the employment, treatment, or termination of any employees of Public Company or any of its subsidiaries, nor, to the knowledge of Public Company, has any such charge been threatened within the preceding twelve (12) months. No current or former employee of Public Company or any of its subsidiaries has, pursuant to internal complaint procedures, made a written complaint of discrimination, retaliation or harassment, nor to Public Company’s knowledge, has an oral complaint of any of the foregoing been made within the preceding twelve (12) months.
(e) Neither Public Company nor any of its subsidiaries has caused a plant closing as defined in the WARN Act affecting any site of employment or one or more operating units within any site of employment, or a mass layoff as defined in the WARN Act, nor have any of the foregoing been affected by any transaction or engaged in layoffs or employment terminations sufficient in number to trigger application of any similar foreign, state or local Law.
4.18. Insurance. Public Company and its subsidiaries maintain insurance policies (the “Public Company Insurance Policies”), including insurance covering directors and officers for securities Law and other customary liabilities, with reputable insurance carriers against all risks of a character and in such amounts as are usually insured against by similarly situated companies in the same or similar businesses. Each Public Company Insurance Policy is in full force and effect. Except as set forth on Section 4.18 of the Public Company Disclosure Schedule, none of the Public Company Insurance Policies shall terminate or lapse (or be affected in any other adverse manner) by reason of any of the transactions contemplated by this Agreement. Public Company and each of its subsidiaries have complied in all material respects with the provisions of each Public Company Insurance Policy under which it is the insured party. No insurer under any Public Company Insurance Policy has cancelled or generally disclaimed liability under any such policy or indicated any intent to do so or not to renew any such policy. Public Company has delivered to Merger Partner accurate and complete copies of all material insurance policies and all material self-insurance programs and arrangements relating to the business, assets, liabilities and operations of Public Company and its subsidiaries.
4.19. Opinion of Financial Advisor. Prior to the execution and delivery of this Agreement, the financial advisor of Public Company, Stifel, Nicolaus & Company, Inc. (the “Public Company Financial Advisor”), has delivered to the Public Company Board an opinion to the effect that, as of the date of such opinion and subject to the assumptions, qualifications and limitations set forth therein, the aggregate consideration (i.e., the Merger Partner Merger Shares) to be paid by Public Company in the Merger pursuant to this Agreement is fair, from a financial point of view, to Public Company, a signed copy of which opinion will be provided by Public Company to Merger Partner within one Business Day following the date of this Agreement solely for informational purposes to confirm delivery of such opinion to the Public Company Board.
4.20. Valid Issuance. The Public Company Common Stock to be issued in the Share Issuances will, when issued in accordance with the provisions of this Agreement, be validly issued, fully paid and nonassessable.
4.21. Nevada “Combinations with Interested Stockholders” Statutes. Assuming the accuracy of the representations and warranties of Merger Partner in Section 3.23, Public Company Board has taken all actions so that the restrictions contained in NRS 78.411 through 78.444, inclusive, applicable to a “combination” (as defined in NRS 78.416) shall not apply to the execution, delivery or performance of this Agreement, the Public Company Support Agreement or the consummation of the Merger or the other transactions contemplated by this Agreement or the Public Company Support Agreement.
4.22. Brokers; Fees and Expenses. Except as set forth on Section 4.22 of the Public Company Disclosure Schedule (which shall include the Public Company Financial Advisor), no agent, broker, investment banker, financial advisor or other firm or Person is or shall be entitled, as a result of any action, agreement or commitment of Public Company or any of its subsidiaries, to any broker’s, finder’s, financial advisor’s or other similar fee or commission in connection with any of the transactions contemplated by this Agreement.
4.23. Operations of Merger Sub. Merger Sub was formed solely for the purpose of engaging in the transactions contemplated by this Agreement, has engaged in no other business activities and has conducted its operations only as contemplated by this Agreement. Merger Sub has no assets or liabilities other than those incident to its formation, the execution of this Agreement and the completion of the transactions hereunder.
4.24. Certain Business Relationships with Affiliates. No Affiliate of Public Company (other than a wholly owned subsidiary of Public Company) (a) owns any material property or right, tangible or intangible, which is used in the business of Public Company or any of its subsidiaries, (b) has any material claim or cause of action against Public Company or any of its subsidiaries or (c) owes any material money to, or is owed any material money by, Public Company or any of its subsidiaries. Section 4.24 of the Public Company Disclosure Schedule describes any material Contracts between Public Company and any Affiliate thereof (other than a wholly owned subsidiary of Public Company), other than (x) any employment or service Contracts, invention assignment agreements and other Contracts entered into in connection with any employment or service, including any Contracts relating to stock purchases and awards, stock options and other equity or equity-based incentive arrangements, in each case relating to compensation, or (y) any arms-length agreements with any portfolio company of any venture capital firm, private equity firm, angel investor, or similar investor of Public Company.
4.25. Controls and Procedures, Certifications and Other Matters.
(a) Public Company and each of its subsidiaries maintains accurate books and records reflecting its assets and liabilities and maintains proper and adequate internal control over financial reporting designed to provide assurance that (i) transactions are executed with management’s authorization, (ii) transactions are recorded as necessary to permit preparation of the consolidated financial statements of Public Company and to maintain accountability for Public Company’s consolidated assets, (iii) access to assets of Public Company and its subsidiaries is permitted only in accordance with management’s authorization, (iv) the reporting of assets of Public Company and its subsidiaries is compared with existing assets at regular intervals and (v) accounts, notes and other receivables and inventory were recorded accurately, and proper and adequate procedures are implemented to effect the collection thereof on a current and timely basis.
(b) Public Company maintains disclosure controls and procedures required by Rules 13a-15 or 15d-15 under the Exchange Act, and such controls and procedures are reasonably designed to ensure that all material information concerning Public Company and its subsidiaries is made known on a timely basis to the individuals responsible for the preparation of Public Company’s filings with the SEC and other public disclosure documents.
(c) Neither Public Company nor any of its subsidiaries has, since Public Company became subject to the reporting requirements of Section 13 or Section 15(d) of the Exchange Act, extended or maintained credit, arranged for the extension of credit, modified or renewed an extension of credit, in the form of a personal loan or otherwise, to or for any director or executive officer of Public Company or any of its subsidiaries. Section 4.25(c) of the Public Company Disclosure Schedule identifies any loan or extension of credit maintained by Public Company or any subsidiary to which the second sentence of Section 13(k)(1) of the Exchange Act applies.
4.26. Books and Records. The minute books and other similar records of Public Company contain accurate summaries, in all material respects, of all actions taken at any meetings of Public Company’s stockholders, the Public Company Board or any committee thereof and of all written consents executed in lieu of the holding of any such meeting since January 1, 2020.
4.27. Privacy and Data Protection.
(a) Public Company and its subsidiaries have complied at all times, and currently comply, in each case, in all material respects, with any applicable data protection and privacy Law with respect to their businesses, including, as applicable, with respect to (a) the requirements relating to notification and/or registration of processing of personal data with any applicable national data protection regulator, (b) requests from data subjects under applicable data protection and privacy Laws, (c) where necessary, the obtaining of consent to the processing of personal data and/or direct marketing activity, and (d) where necessary, the obtaining of any approval, consultation and/or agreement of any applicable works councils or such similar worker representation bodies. Public Company has all rights necessary to process all personal data used in the business of the Public Company. Neither Public Company nor any of its subsidiaries has received any written notice or complaint from any individual, third party and/or regulatory (x) authority alleging non-compliance by Public Company or any of its subsidiaries with any applicable data protection and privacy Law (including any prohibition or restriction on the transfer of data to any jurisdiction) or (y) claiming compensation for or an injunction for non-compliance with any applicable data protection and privacy Law.
(b) Public Company has established and maintains commercially reasonable technical, physical and organizational controls, policies, procedures, safeguards, measures and security systems, plans and technologies in material compliance with requirements under applicable privacy and data protection Laws. Since January 1, 2020, no material breach or material security incident in relation to Public Company’s personal data and/or proprietary data has occurred or, to the Public Company’s knowledge, is threatened, and there has been no actual or, to the Public Company’s knowledge, threatened unauthorized or illegal processing of, or accidental or unlawful destruction, loss or alteration of, any of Public Company’s personal data and/or proprietary data.
4.28. No Other Representations or Warranties. Each of Public Company and Merger Sub hereby acknowledges and agrees that, except for the representations and warranties contained in this Agreement, none of Merger Partner nor any other Person on behalf of Merger Partner makes any express or implied representation or warranty with respect to Merger Partner or its financial condition, business, results of operations, properties, assets, liabilities, or prospects or otherwise or with respect to any other statements made or information provided to Public Company, Merger Sub or any of their Affiliates in connection with the transactions contemplated hereby, and (subject to the express representations and warranties of Merger Partner set forth in Article III (in each case as qualified and limited by the Merger Partner Disclosure Schedule) or any representations and warranties of a signatory to any Merger Partner Support Agreement or Merger Partner Lock-Up Agreement) none of Public Company, Merger Sub or any of their respective Affiliates, stockholders, directors, officers, employees, agents, representatives or advisors, or any other Person, has relied on any representations, warranties, statements, or information (including the accuracy or completeness thereof).
ARTICLE V
CONDUCT OF BUSINESS
5.1. Covenants of Merger Partner. Except as set forth in Section 5.1 of the Merger Partner Disclosure Schedule, as expressly provided herein, or as consented to in writing by Public Company (which consent shall not be unreasonably withheld, conditioned or delayed), or to the extent necessary to comply with any applicable Law, or as required in connection with the Concurrent Financing, from and after the date of this Agreement until the earlier of the termination of this Agreement in accordance with its terms and the Effective Time, Merger Partner shall use commercially reasonable efforts to, act and carry on its business in the Ordinary Course of Business. Without limiting the generality of the foregoing, except as set forth in Section 5.1 of the Merger Partner Disclosure Schedule, or as expressly provided herein, or to the extent necessary to comply with any applicable Law, or as required in connection with the Concurrent Financing, from and after the date of this Agreement until the earlier of the termination of this Agreement in accordance with its terms and the Effective Time, Merger Partner shall not, directly or indirectly, do any of the following without the prior written consent of Public Company (which consent shall not be unreasonably withheld, conditioned or delayed):
(a) (i) declare, set aside or pay any dividends on, or make any other distributions (whether in cash, securities or other property) in respect of, any of its capital stock; (ii) split, combine or reclassify any of its capital stock or issue or authorize the issuance of any other securities in respect of, in lieu of or in substitution for shares of its capital stock or any of its other securities, other than the issuance of shares upon exercise or conversion of any Merger Partner Preferred Stock, Merger Partner Stock Options, or other convertible securities of Merger Partner; or (iii) purchase, redeem or otherwise acquire any shares of its capital stock or any other of its securities or any rights, warrants or options to acquire any such shares or other securities, other than, in the case of this clause (iii), from former employees, directors and consultants in accordance with Merger Partner Stock Plans, as in effect as of the date of this Agreement;
(b) issue, deliver, sell, grant, pledge or otherwise dispose of or encumber any shares of its capital stock, any other voting securities or any securities convertible into or exchangeable for, or any rights, warrants or options to acquire, any such shares, voting securities or convertible or exchangeable securities, other than (i) the issuance of shares of Merger Partner Voting Common Stock upon exercise or conversion of any Merger Partner Preferred Stock, Merger Partner Stock Options or other convertible securities of Merger Partner outstanding on the date of this Agreement, (ii) the grant of Merger Partner Stock Options in the Ordinary Course of Business in accordance with Merger Partner Stock Plans, as in effect as of the date of this Agreement and (iii) the issuance of Merger Partner Notes up to an aggregate principal amount of $20,000,000, including any Merger Partner Notes outstanding as of the date hereof;
(c) except as required to give effect to anything in contemplation of the Closing, amend its certificate of incorporation, bylaws or other comparable charter or organizational documents or effect or be a party to any merger, consolidation, share exchange, business combination, liquidation, dissolution, reorganization, statutory conversion, recapitalization, reclassification of shares, stock split or reverse stock split or form any new subsidiary or acquire any equity interest or other interest in any other Person;
(d) acquire, by merging or consolidating with, or by purchasing all or a substantial portion of the assets or any stock of, or by any other manner, any business or any corporation, partnership, joint venture, limited liability company, association or other business organization or division thereof;
(e) except in the Ordinary Course of Business, sell, lease, license, pledge, or otherwise dispose of or encumber any properties or assets material to Merger Partner;
(f) enter into any material transaction other than in the Ordinary Course of Business;
(g) license any material Intellectual Property to or from any third party;
(h) initiate, threaten, compromise or settle any litigation or arbitration proceeding (other than any litigation to enforce its rights under this Agreement) other than waivers, settlements or agreements which (i) do not exceed, in the aggregate, $200,000 more than the amounts covered by insurance policies (ii) do not impose any material restrictions on the operations or businesses of the Merger Partners(including following the Merger and/or the Surviving Company), taken as a whole, (C) do not provide for equitable relief on, or the admission of wrongdoing by Merger Partner, and (D) do not relate to claims, litigations, investigations or proceedings brought by a Governmental Entity;
(i) (i) incur or suffer to exist any indebtedness for borrowed money or guarantee any such indebtedness of another Person in excess of $200,000, (ii) issue, sell, or amend any debt securities or warrants or other rights to acquire any debt securities of Merger Partner, guarantee any debt securities of another Person, enter into any “keep well” or other agreement to maintain any financial statement condition of another Person, or enter into any arrangement having the economic effect of any of the foregoing, or (iii) make any loans, advances (other than routine advances to employees of Merger Partner in the Ordinary Course of Business) or capital contributions to, or investment in, any other Person, in each case, other than the issuance of Merger Partner Notes, up to an aggregate principal amount of $20,000,000, including any Merger Partner Notes outstanding as of the date hereof;
(j) make, change or revoke any Tax election (other than elections made in the Ordinary Course of Business), change an annual accounting period, enter into any closing agreement, waive or extend any statute of limitations with respect to Taxes (other than any automatic extension granted in the Ordinary Course of Business and consistent with past custom and practice of Merger Partner), settle or compromise any material Tax liability, claim or assessment, knowingly surrender any right to claim a refund of material Taxes, or amend any income or other material Tax Return; or
(k) authorize any of, or commit or agree, in writing or otherwise, to take any of, the foregoing actions or any action that would reasonably be expected to, individually or in the aggregate, (i) make any representation or warranty of Merger Partner in this Agreement untrue or incorrect, or (ii) impair, delay or prevent the satisfaction of any conditions in Article VII hereof.
If Merger Partner desires to take an action which would be prohibited pursuant to this Section 5.1 without the consent of Public Company, Merger Partner may request such consent (which consent shall not be unreasonably withheld, conditioned or delayed) by sending an email to both of the following individuals, which email shall be sufficient notice under this Agreement:
Tom Bures
and
Karam Hijji
5.2. Covenants of Public Company. Except as set forth in Section 5.2 of the Public Company Disclosure Schedule or as expressly provided herein or as consented to in writing by Merger Partner (which consent shall not be unreasonably withheld, conditioned or delayed) or to the extent necessary to comply with any applicable Laws, or as required in connection with the Concurrent Financing, from and after the date of this Agreement until the earlier of the termination of this Agreement in accordance with its terms and the Effective Time, Public Company shall, and shall cause each of its subsidiaries to, use commercially reasonable efforts to, act and carry on its business in the Ordinary Course of Business. Without limiting the generality of the foregoing, except as set forth in Section 5.2 of the Public Company Disclosure Schedule, as expressly provided herein, as required in connection with the Concurrent Financing, or to the extent necessary to comply with any applicable Law, from and after the date of this Agreement until the earlier of the termination of this Agreement in accordance with its terms and the Effective Time, Public Company shall not, and shall not permit any of its subsidiaries to, directly or indirectly, do any of the following without the prior written consent of Merger Partner (which consent shall not be unreasonably withheld, conditioned or delayed):
(a) (i) except as contemplated by the Closing Dividend (as defined below), declare, set aside or pay any dividends on, or make any other distributions (whether in cash, securities or other property) in respect of, any of its capital stock; (ii) split, combine or reclassify any of its capital stock or issue or authorize the issuance of any other securities in respect of, in lieu of or in substitution for shares of its capital stock or any of its other securities, other than in respect of any Public Company Preferred Stock or upon exercise or conversion of any Public Company Stock Option or Public Company Warrant, in each case, outstanding on the date of this Agreement; or (iii) purchase, redeem or otherwise acquire any shares of its capital stock or any other of its securities or any rights, warrants or options to acquire any such shares or other securities, other than, in the case of this clause (iii), from former employees, directors and consultants in accordance with Public Company Stock Plans, as in effect as of the date of this Agreement;
(b) issue, deliver, sell, grant, pledge or otherwise dispose of or encumber any shares of its capital stock, any other voting securities or any securities convertible into or exchangeable for, or any rights, warrants or options to acquire, any such shares, voting securities or convertible or exchangeable securities, in each case other than the issuance of shares of Public Company Common Stock pursuant to the Concurrent Financing, upon the exercise of Public Company Stock Options or Public Company Warrants or conversion of Public Company Preferred Stock, in each case, outstanding on the date of this Agreement in accordance with their present terms (including cashless exercises);
(c) except as required or appropriate to give effect to anything in contemplation of the Closing, amend its articles of incorporation, bylaws or other comparable charter or organizational documents or effect or be a party to any merger, consolidation, share exchange, business combination, liquidation, dissolution, reorganization, statutory conversion, recapitalization, reclassification of shares, stock split or reverse stock split or form any new subsidiary or acquire any equity interest or other interest in any other Person;
(d) acquire (i) by merging or consolidating with, or by purchasing all or a substantial portion of the assets or any stock of, or by any other manner, any business or any corporation, partnership, joint venture, limited liability company, association or other business organization or division thereof or (ii) any assets that are material, individually or in the aggregate, to Public Company and its subsidiaries, taken as a whole;
(e) sell, lease, license, pledge, or otherwise dispose of or encumber any properties or assets material to Public Company or any of its subsidiaries;
(f) enter into any material transaction;
(g) license any material Intellectual Property rights to or from any third party;
(h) (i) incur or suffer to exist any indebtedness for borrowed money or guarantee any such indebtedness of another Person, (ii) issue, sell or amend any debt securities or warrants or other rights to acquire any debt securities of Public Company or any of its subsidiaries, guarantee any debt securities of another Person, enter into any “keep well” or other agreement to maintain any financial statement condition of another Person or enter into any arrangement having the economic effect of any of the foregoing, (iii) make any loans, advances (other than routine advances to employees of Public Company in the Ordinary Course of Business) or capital contributions to, or investment in, any other Person, other than Public Company or any of its direct or indirect wholly owned subsidiaries or (iv) enter into any hedging agreement or other financial agreement or arrangement designed to protect Public Company or its subsidiaries against fluctuations in commodities prices or exchange rates;
(i) forgive any loans to any Person, including its employees, officers, directors or Affiliate;
(j) enter into any agreement to purchase or sell any interest in real property, grant any security interest in any real property, enter into any lease, sublease, license or other occupancy agreement with respect to any real property or alter, amend, modify any agreement that terminated any Public Company Lease;
(k) make (i) any capital expenditures or other expenditures with respect to property, plant or equipment or (ii) other material expenditures in excess of $50,000 in the aggregate (other than any expenditures in the Ordinary Course of Business);
(l) make any changes in accounting methods, principles or practices, except insofar as may have been required by the SEC or a change in GAAP or, except as so required, change any assumption underlying, or method of calculating, any bad debt, contingency or other reserve;
(m) except for terminations as a result of the expiration of any contract that expires in accordance with its terms, (i) modify or amend in any material respect, or terminate, any material contract or agreement to which Public Company or any of its subsidiaries is party, or (ii) knowingly waive, release or assign any material rights or claims (including any write-off or other compromise of any accounts receivable of Public Company of any of its subsidiaries);
(n) (i) enter into any contract or agreement, including those relating to the rendering of services or the distribution, sale or marketing by third parties of the products, of, or products licensed by, Public Company or any of its subsidiaries or (ii) license any Intellectual Property rights to or from any third party;
(o) except as required to comply with a Public Company Employee Plan, (i) take any action with respect to, adopt, enter into, terminate (other than terminations for cause) or amend any Public Company Employee Plan (or any other employee benefit or compensation plan, program, policy, agreement or arrangement that would have constituted a Public Company Employee Plan had it been in effect on the date of this Agreement) or any collective bargaining agreement, (ii) increase the compensation (including any compensation opportunities) or fringe benefits of, or pay any bonus or grant any bonus opportunity to, any director, officer, employee or consultant, (iii) amend or accelerate the payment, right to payment or vesting of any compensation or benefits, including any outstanding equity or equity-based incentive awards, (iv) pay any benefit not provided for as of the date of this Agreement under any benefit plan under any Public Company Employee Plan, (v) grant any awards under any Public Company Employee Plan (or under any other employee benefit or compensation plan, program, policy, agreement or arrangement that would have constituted a Public Company Employee Plan had it been in effect on the date of this Agreement), or (vi) take any action other than in the Ordinary Course of Business to fund or in any other way secure the payment of compensation or benefits under any Public Company Employee Plan (or under any other employee benefit or compensation plan, program, policy, agreement or arrangement that would have constituted a Public Company Employee Plan had it been in effect on the date of this Agreement);
(p) make, change or revoke any Tax election (other than elections made in the Ordinary Course of Business), change an annual accounting period, enter into any closing agreement, waive or extend any statute of limitations with respect to Taxes (other than any automatic extension granted in the Ordinary Course of Business and consistent with past custom and practice of Public Company), settle or compromise any material Tax liability, claim or assessment, knowingly surrender any right to claim a refund of material Taxes, or amend any income or other material Tax Return;
(q) commence any offering of shares of Public Company Common Stock, including pursuant to any employee stock purchase plan;
(r) initiate, threaten, compromise or settle any litigation or arbitration proceeding (other than any litigation to enforce its rights under this Agreement), other than a Permitted Settlement;
(s) fail to use commercially reasonable efforts to maintain insurance levels substantially comparable to levels existing as of the date of this Agreement;
(t) open or close any facility or office;
(u) delay or fail to pay accounts payable and other obligations when due; or
(v) authorize any of, or commit or agree, in writing or otherwise, to take any of, the foregoing actions or any action that would reasonably be expected to, individually or in the aggregate, make any representation or warranty of Public Company in this Agreement untrue or incorrect in any material respect, or would materially impair, delay or prevent the satisfaction of any conditions in Article VII hereof.
If Public Company desires to take an action which would be prohibited pursuant to this Section 5.2 without the consent of Merger Partner, Public Company may request such consent (which consent shall not be unreasonably withheld, conditioned or delayed) by sending an email to the following individuals, which email shall be sufficient notice under this Agreement:
Wesley H. Kaupinen
and
Kathleen A. McGowan
5.3. Confidentiality. The parties acknowledge that Public Company and Merger Partner have previously executed a letter agreement, dated as of March 22, 2024 (the “Confidentiality Agreement”), which Confidentiality Agreement shall continue in full force and effect in accordance with its terms, except as expressly modified by this Agreement.
5.4. Closing Dividend; Legacy Asset Dispositions.
(a) Prior to the Effective Time, Public Company shall declare a dividend (the “Closing Dividend”) to each holder of record of shares of Public Company Common Stock and shares of Public Company Preferred Stock entitled to such a dividend in accordance with its terms the right to receive one contingent value right (each, a “CVR”) for each outstanding share of Public Company Common Stock held by such stockholder as of such date, each representing the right to receive contingent payments upon the occurrence of certain events set forth in, and subject to and in accordance with the terms and conditions of, the Contingent Value Rights Agreement in the form attached hereto as Exhibit C (the “CVR Agreement”). The record date for the Closing Dividend shall be the close of business on the last Business Day prior to the day on which the Effective Time occurs and the payment date for which shall be three (3) Business Days after the Effective Time; provided, that the payment of such dividend may be conditioned upon the occurrence of the Effective Time. In connection with the Closing Dividend, Public Company shall cause the CVR Agreement to be duly authorized, executed and delivered by Public Company and a rights agent selected by Public Company with Merger Partner’s prior approval (such approval not to be unreasonably withheld, delayed or conditioned). Public Company shall deduct and withhold from the payment of the Closing Dividend such amount as it is required to deduct and withhold with respect to the making of such payment under the Code, the rules or regulations promulgated thereunder, any provision of applicable state, local or foreign Tax Law or any other applicable Law. To the extent that amounts are so deducted or withheld, such deducted or withheld amounts shall be treated for purposes of this Agreement and the CVR Agreement as having been paid to the Person in respect of which such deduction and withholding was made.
(b) Public Company and Merger Partner agree that Public Company may, without the prior consent of Merger Partner, sell, assign, license, transfer, or otherwise dispose of, in one or more transactions, some or all of its assets (or any assets of any of its subsidiaries) (any such assets, “Legacy Assets”) at any time at or prior to the Closing (any such transaction, a “Legacy Asset Disposition”) pursuant to any definitive agreement entered into after the date hereof and prior to the Closing that is a customary “as is / where is” sale; provided that such definitive agreement does not contain (i) any non-competition obligation affecting any of the assets of Merger Partner, Public Company or any subsidiary of Public Company or (ii) any material post-disposition obligations or Liabilities for Public Company or any of its subsidiaries following the Closing, including any indemnification obligations or Liability for any representation or warranty, other than customary obligations of confidentiality. For the avoidance of doubt, if an agreement (or amendment thereto) calling for the sale, assignment, license, or other disposition of any of the assets of the Public Company contains any of the obligations described in clause (i) or (ii) above, Public Company may pursue and negotiate such agreement after the date hereof, but shall not enter into (or amend) such agreement without the prior written consent of Merger Partner (which consent shall not be unreasonably withheld, conditioned or delayed). Public Company shall keep Merger Partner reasonably apprised of all material discussions and communications concerning any actual or potential Legacy Asset Disposition and the actual or potential transactions contemplated thereby, including sharing all material draft Contracts and other documents with respect thereto. In the event that Public Company intends to consummate a Legacy Asset Disposition, Public Company shall provide Merger Partner with prompt written notice of such intention.
ARTICLE VI
ADDITIONAL AGREEMENTS
6.1. No Solicitation.
(a) No Solicitation or Negotiation. Except as set forth in this Section 6.1, until the earlier to occur of the termination of this Agreement pursuant to Article VIII and Effective Time, each of Merger Partner, Public Company and their respective subsidiaries shall not, and each of Merger Partner and Public Company shall cause their respective directors, officers, employees and consultants not to, and shall instruct their respective attorneys and financial advisors (“Representatives”) not to, directly or indirectly:
(i) solicit, seek or initiate or knowingly take any action to facilitate or encourage any offers, inquiries or the making of any proposal or offer that constitutes, or would reasonably be expected to lead to, any Acquisition Proposal;
(ii) enter into, continue or otherwise participate or engage in any discussions or negotiations regarding any Acquisition Proposal, or furnish to any Person any non-public information or afford any Person other than Public Company or Merger Partner, as applicable, access to such party’s property, books or records (except pursuant to a request by a Governmental Entity) in connection with any offers, inquiries or the making of any proposal or offer that constitutes, or would reasonably be expected to lead to, any Acquisition Proposal;
(iii) take any action to make the provisions of any “fair price”, “business combination” or “control share acquisition” statute or other similar statute or regulation inapplicable to any transactions contemplated by an Acquisition Proposal; or
(iv) publicly propose to do any of the foregoing described in clauses (i) through (iii).
Notwithstanding the foregoing or anything to the contrary set forth in this Agreement, subject to compliance with Section 6.1(c), prior to the Specified Time, each of Public Company and Merger Partner, and their respective Representatives, may (A) furnish non-public information with respect to Public Company and its subsidiaries or Merger Partner, as the case may be, to any Qualified Person (and the Representatives of such Qualified Person), or (B) engage in discussions or negotiations (including solicitation of revised Acquisition Proposals) with any Qualified Person (and the Representatives of such Qualified Person) regarding any bona fide, unsolicited written Acquisition Proposal which such party’s board of directors determines in good faith, after consultation with such party’s financial advisors and outside legal counsel, constitutes, or is reasonably likely to result in, a Superior Proposal (and is not withdrawn); provided, (x) that either Merger Partner or Public Company, as the case may be, receives from the Qualified Person an executed confidentiality agreement on the terms not less restrictive than exist in the Confidentiality Agreement and, if entered into after the date of this Agreement, containing additional provisions that expressly permit such party to comply with this terms of this Section 6.1 (a copy of which shall be provided to the other party), (y) that the party seeking to make use of this proviso has not otherwise materially breached this Section 6.1 with respect to such Acquisition Proposal or the Person making such Acquisition Proposal, and (z) the Merger Partner Board or Public Company Board, as the case may be, has determined in good faith (after consultation with outside legal counsel) that the failure to take such actions would reasonably be expected to be inconsistent with its fiduciary duties under applicable Law. It is understood and agreed that any violation of the restrictions in this Section 6.1 (or action that, if taken by Public Company or Merger Partner, as applicable, would constitute such a violation) by any director, officer, attorney, or financial advisor of Public Company or Merger Partner shall be deemed to be a breach of this Section 6.1 by Public Company or Merger Partner, as the case may be.
(b) No Change in Recommendation or Alternative Acquisition Agreement. Prior to the Effective Time:
(i) (A) Merger Partner Board (and any committee thereof) shall not, except as set forth in this Section 6.1, (1) withhold, withdraw or modify, or publicly propose to withhold, withdraw or modify, the approval or recommendation by the Merger Partner Board with respect to the Merger, (2) fail to recommend against acceptance of a tender offer within ten (10) Business Days after commencement, or (3) propose publicly to approve, endorse, adopt or recommend, or has approved, endorsed, adopted, or recommended any Acquisition Proposal (each, a “Merger Partner Board Recommendation Change”) and (B) the Public Company Board (and any committee thereof) shall not, except as set forth in this Section 6.1, (1) fail to include its recommendation to the approval of the Required Public Company Voting Proposals in the Proxy Statement/Prospectus or shall have withdrawn or modified in a manner adverse to Merger Partner its recommendation of the Required Public Company Voting Proposals, (2) withhold, withdraw or modify, or publicly propose to withhold, withdraw or modify, the approval or recommendation by the Public Company Board with respect to the Share Issuances or the Public Company Charter Amendment, (3) after the receipt by Public Company of an Acquisition Proposal and Merger Partner requests in writing that Public Company Board reconfirm its recommendation of the Required Public Company Voting Proposals, fail to do so within ten Business Days after its receipt of Merger Partner’s request, (4) fail to recommend against acceptance of a tender offer within ten (10) Business Days after commencement or (5) propose publicly to approve, endorse, adopt or recommend, or has approved, endorsed, adopted, or recommended any Acquisition Proposal (each a “Public Company Board Recommendation Change”);
(ii) each of Public Company and Merger Partner shall not enter into any letter of intent, memorandum of understanding, agreement in principle, acquisition agreement, merger agreement or similar agreement (an “Alternative Acquisition Agreement”) providing for the consummation of a transaction contemplated by any Acquisition Proposal (other than a confidentiality agreement referred to in Section 6.1(a) entered into in the circumstances referred to in Section 6.1(a)); and
(iii) each of the Public Company Board and the Merger Partner Board, and each committee thereof, shall not, except as set forth in this Section 6.1, adopt, approve or recommend, or publicly propose to adopt, approve or recommend, any Acquisition Proposal.
Notwithstanding the foregoing or anything to the contrary set forth in this Agreement (including the provisions of this Section 6.1), at any time prior to the Specified Time, the Public Company Board or the Merger Partner Board, as the case may be, may effect a Public Company Board Recommendation Change or Merger Partner Board Recommendation Change, as the case may be, with respect to a Superior Proposal, if: (i) such board of directors shall have determined in good faith (after consultation with outside legal counsel) that the failure to effect such Public Company Board Recommendation Change or Merger Partner Board Recommendation Change, as applicable, would reasonably be expected to be inconsistent with its fiduciary duties under applicable Law; (ii) such party has provided at least four (4) Business Days prior written notice to the other party that it intends to effect a Public Company Board Recommendation Change or Merger Partner Board Recommendation Change, as the case may be, and written copies of any relevant proposed transactions agreements with any party making a potential Superior Proposal (including the identity of the Person making such Superior Proposal) (a “Recommendation Change Notice”) (it being understood that the Recommendation Change Notice shall not constitute a Public Company Board Recommendation Change or Merger Partner Board Recommendation Change for purposes of this Agreement); (iii) such party has complied in all material respects with the requirements of this Section 6.1 in connection with any potential Superior Proposal; (iv) such party has, and has caused its financial advisors and outside legal counsel to, during the four (4) Business Day period referred to in clause (ii) above, negotiate with the other party in good faith to make such adjustments to the terms and conditions of this Agreement so that such Acquisition Proposal ceases to constitute a Superior Proposal (to the extent the other party desires to negotiate); and (v) if the other party shall have delivered to such party a written, binding and irrevocable offer to alter the terms or conditions of this Agreement during the four (4) Business Day period referred to in clause (ii) above, such party’s board of directors shall have determined in good faith (after consultation with outside legal counsel), after considering the terms of such offer by the other party, that the failure to effect a Public Company Board Recommendation Change or Merger Partner Board Recommendation Change, as the case may be, would reasonably be expected to be inconsistent with its fiduciary duties under applicable Law. In the event of any material amendment to any Superior Proposal (including any revision in the amount, form or mix of consideration such party’s stockholders would receive as a result of such potential Superior Proposal), such party shall be required to provide the other party with notice of such material amendment and there shall be a new two (2) Business Day period following such notification during which the parties shall comply again with the requirements of this Section 6.1(b) and the board of directors of such party shall not make a Public Company Board Recommendation Change or Merger Partner Board Recommendation Change, as applicable, prior to the end of any such period as so extended.
(c) Notices of Proposals. Each party will as promptly as reasonably practicable (and in any event within twenty-four (24) hours after receipt) (i) notify the other party of its receipt of any Acquisition Proposal and (ii) provide to the other party a copy of such Acquisition Proposal (if written), or a summary of the material terms and conditions of such Acquisition Proposal (if oral), including the identity of the Person making such Acquisition Proposal, and copies of all written communications and materials from such Person with respect to such actual or potential Acquisition Proposal. Such party in receipt of an Acquisition Proposal shall notify the other party, in writing, of its first decision of its board of directors as to whether to consider any Acquisition Proposal or to enter into discussions or negotiations concerning any Acquisition Proposal or to provide non-public information with respect to such to any Person, which notice shall be given as promptly as practicable after such determination was reached (and in any event no later than twenty-four (24) hours after such determination was reached). Such party in receipt of an Acquisition Proposal will (A) provide the other party with written notice setting forth such information as is reasonably necessary to keep such other party reasonably informed of the material terms of any such Acquisition Proposal and of any material amendments or modifications thereto made by the Person making an Acquisition Proposal, and (B) at least two (2) business days prior to the provision of any material non-public information of such party to any such Person, provide such information to the other party (including by posting such information to an electronic data room), to the extent such information has not previously been made available the other party.
(d) Certain Permitted Disclosure. Nothing contained in this Agreement shall prohibit Merger Partner or Public Company or their respective Boards of Directors from complying with Rules 14d-9 and 14e-2(a) promulgated under the Exchange Act; provided, however, that any disclosure made by Merger Partner or Public Company or their respective Boards of Directors pursuant to Rules 14d-9 and 14e-2(a) shall be limited to a statement that Merger Partner or Public Company, as applicable, is unable to take a position with respect to the bidder’s tender offer unless the applicable Board of Directors determines after consultation with its outside legal counsel, that such statement would reasonably be expected to be inconsistent with its fiduciary duties under applicable Law; provided, further, that any such disclosures (other than a “stop, look and listen” communication or similar communication of the type contemplated by Section 14d-9(f) under the Exchange Act) shall be deemed to be a Merger Partner Board Recommendation Change or Public Company Board Recommendation Change, as applicable, unless such communication expressly reaffirms its recommendation for the Merger and the other transactions contemplated hereby in such communication.
(e) Cessation of Ongoing Discussions. Each of Public Company and Merger Partner shall, and shall direct its Representatives to, cease immediately all discussions and negotiations that commenced prior to the date of this Agreement regarding any proposal that constitutes, or would reasonably be expected to lead to, an Acquisition Proposal; provided, however, that the foregoing shall not in any way limit or modify the rights of any party hereto under the other provisions of this Section 6.1. Public Company and Merger Partner will each promptly revoke or withdraw access of any Person (other than Public Company, Merger Partner and their respective Representatives) to any data room (virtual or actual) containing any non-public information with respect to Public Company that was established or shared in connection with any potential Acquisition Proposal and request from each third party (other than Public Company, Merger Partner and their Representatives) the prompt return or destruction of all non-public information with respect to Public Company or Merger Partner, as applicable, previously provided to such Person.
(f) Definitions. For purposes of this Agreement, the following terms shall have the following meanings:
(i) “Acquisition Proposal” means, with respect to Public Company or Merger Partner, (A) any inquiry, proposal or offer for a merger, consolidation, dissolution, sale of substantial assets, recapitalization, share exchange, tender offer or other business combination involving such party and its subsidiaries (other than mergers, consolidations, recapitalizations, share exchanges or other business combinations involving solely such party and/or one or more subsidiaries of such party), (B) any proposal for the issuance by such party of 15% or more of its equity securities or (C) any proposal or offer to acquire in any manner, directly or indirectly, 15% or more of the equity securities or consolidated total assets of such party and its subsidiaries, in each case other than the transactions contemplated by this Agreement; provided, however, that no inquiry, proposal, or offer received pursuant to the terms of, (1) the Concurrent Financing or (2) any Legacy Asset Disposition, shall be an Acquisition Proposal.
(ii) “Qualified Person” means any Person making a bona fide, unsolicited written Acquisition Proposal that the Public Company Board or the Merger Partner Board, as the case may be, determines in good faith (after consultation with outside counsel and its financial advisors) is, or would reasonably be expected to lead to, a Superior Proposal, and such Acquisition Proposal has not resulted from a material breach by Public Company or Merger Partner, as the case may be, of its obligations under Section 6.1(a).
(iii) “Specified Time” means the earliest to occur of (A) the Effective Time, (B) in the case of Public Company, the date on which the stockholders of Public Company shall have approved the Required Public Company Voting Proposals, (C) in the case of Merger Partner, the date on which the stockholders of Merger Partner shall have approved the Merger Partner Voting Proposal, and (D) the time at which this Agreement is terminated in accordance with the terms hereof.
(iv) “Superior Proposal” means, with respect to Public Company or Merger Partner, any bona fide, unsolicited written Acquisition Proposal (for purpose of this definition, replacing all references in such definition to 15% with 50%), (A) on terms which the board of directors of such party determines in its good faith judgment to be more favorable to the holders of such party’s capital stock from a financial point of view than the transactions contemplated by this Agreement (after consultation with its financial and outside legal advisors), taking into account all the terms and conditions of such proposal and this Agreement (including any termination or break-up fees and conditions to consummation, as well as any written, binding offer by the other party hereto to amend the terms of this Agreement for at least four (4) Business Days) that the board of directors of such party determines to be relevant, and (B) which board of directors of such party has determined to be reasonably capable of being completed on the terms proposed, taking into account all financial, regulatory, legal and other aspects of such proposal that board of directors of such party determines to be relevant (including the likelihood and timing of consummation as compared to the transactions contemplated hereby).
6.2. Proxy Statement/Prospectus; Registration Statement.
(a) As promptly as practical after the execution of this Agreement and contingent upon receipt from Merger Partner of the information required by the following sentence, Public Company, with the cooperation of Merger Partner, shall prepare and file with the SEC the Registration Statement, in which the Proxy Statement/Prospectus will be included as a prospectus. Merger Partner, Merger Sub and Public Company shall (i) provide to the other parties as promptly as practical all information, including financial statements and descriptions of its business and financial condition, as Public Company as such other parties may reasonably request for preparation of the Registration Statement and the Proxy Statement/Prospectus and (ii) cause the timely cooperation of its independent public accountants in connection with the preparation and filing of the Registration Statement and the Proxy Statement/Prospectus, including by causing such accountants to provide a consent to the inclusion of such accountant’s reports in respect of the financial statements of the applicable party in the Registration Statement and/or in the Proxy Statement/Prospectus (as applicable) and to the reference to such accountant firm as an “expert” therein. Public Company shall respond to any comments of the SEC and shall use reasonable best efforts to have the Registration Statement declared effective under the Securities Act as promptly as practicable after such filing, and Public Company shall cause the Proxy Statement/Prospectus to be mailed to its stockholders at the earliest practicable time after the Registration Statement is declared effective under the Securities Act. Public Company shall notify Merger Partner promptly upon the receipt of any comments from the SEC or its staff and of any request by the SEC or its staff for amendments or supplements to the Registration Statement, the Proxy Statement/Prospectus or any filing pursuant to Section 6.2(b) or for additional information and shall supply Merger Partner with copies of all correspondence between Public Company or any of its representatives, on the one hand, and the SEC, or its staff, on the other hand, with respect to the Registration Statement, the Proxy Statement/Prospectus, the Merger or any filing pursuant to Section 6.2(b). Each of Public Company and Merger Partner shall use commercially reasonable efforts to cause the Registration Statement and the Proxy Statement/Prospectus to comply in all material respects with all applicable requirements of Law and the rules and regulations promulgated thereunder. Whenever either Public Company or Merger Partner shall become aware that any information contained in the Registration Statement or Proxy Statement/Prospectus shall have become false or misleading in any material respect or that the Proxy Statement/Prospectus (including the information statement contained therein), the Registration Statement or any filing pursuant to Section 6.2(b) is otherwise required to be amended or supplemented, Public Company or Merger Partner, as the case may be, shall promptly inform the other of such occurrence and cooperate in filing with the SEC or its staff, and/or mailing to stockholders of Public Company and Merger Partner, such amendment or supplement.
(b) Notwithstanding anything to the contrary stated above, prior to filing and mailing, as applicable, the Registration Statement or Proxy Statement/Prospectus (or any amendment or supplement thereto) or responding to any comments of the SEC with respect thereto, Public Company shall provide Merger Partner and its outside legal counsel a reasonable opportunity to review and comment on such document or response and shall consider in good faith any such comments proposed by Merger Partner. Public Company will advise Merger Partner, promptly after Merger Partner receives notice thereof, of the time when the Registration Statement has become effective or any supplement or amendment has been filed, of the issuance of any stop order or the suspension of the qualification of Public Company Common Stock for offering or sale in any jurisdiction, of the initiation or threat of any proceeding for any such purpose, or of any request by the SEC for the amendment or supplement of the Registration Statement or for additional information.
(c) Public Company and Merger Partner shall promptly make all necessary filings with respect to the Merger and the Share Issuances under the Securities Act, the Exchange Act, applicable state blue sky Laws and the rules and regulations thereunder.
6.3. Nasdaq Listing. Public Company shall use its commercially reasonable efforts, and shall take all reasonably necessary actions, to continue the listing of Public Company Common Stock on Nasdaq during the term of this Agreement (through and until the Effective Time) and to cause the shares of Public Company Common Stock being issued in connection with the Merger to be approved for listing (subject to notice of issuance) on Nasdaq at or prior to the Effective Time, including by filing the Nasdaq Listing Application. Merger Partner will cooperate with Public Company to cause the Nasdaq Listing Application to be conditionally approved prior to the Effective Time and shall promptly furnish to Public Company all information concerning Merger Partner and its officers, directors, and equityholders and such other matters, in each case, that may be required or reasonably requested in connection with any action contemplated by this Section 6.3.
6.4. Access to Information. Each of Public Company and Merger Partner shall (and Public Company shall cause its subsidiaries to) afford to the other party’s officers, employees, accountants, counsel and other representatives, reasonable access, during normal business hours during the period prior to the Effective Time, to all its properties, books, contracts, commitments, personnel and records and, during such period, each of Public Company and Merger Partner shall (and Public Company shall cause its subsidiaries to) furnish promptly to the other party all information concerning its business, properties, assets and personnel as the other party may reasonably request in furtherance of the consummation of the Merger, the Concurrent Financing, or the other transactions contemplated by this Agreement; provided, however, that a party may restrict the foregoing access to the extent that (a) any applicable Law requires such restriction, (b) such access would give rise to a risk of waiving any attorney-client privilege, work product doctrine or other applicable privilege, or (c) such access would be in breach of any confidentiality obligation or similar obligation. Each of Public Company and Merger Partner will (and Public Company will cause its subsidiaries to) hold any such information which is nonpublic in confidence in accordance with the Confidentiality Agreement. No information or knowledge obtained in any investigation pursuant to this Section 6.4 or otherwise shall affect or be deemed to modify any representation or warranty contained in this Agreement or the conditions to the obligations of the parties to consummate the Merger. Any information obtained pursuant to the access contemplated by this Section 6.4 shall be subject to the Confidentiality Agreement. Any access to any facilities of Merger Partner, Public Company, or any of their subsidiaries, shall be subject to the reasonable security measures and insurance requirements of Merger Partner, Public Company, or any of their subsidiaries, as applicable, and shall not include the right to perform any “invasive” testing or soil, air or groundwater sampling, including, without limitation, any Phase I or Phase II environmental assessments. Without limiting the generality of the foregoing, from the date of this Agreement until the Effective Time, each of Public Company and Merger Partner shall promptly provide the other party with copies of any material notice, report or other document received from any Governmental Entity in connection with the Merger or any of the transactions contemplated by this Agreement.
6.5. Stockholder Approval.
(a) Promptly after the Registration Statement is declared effective under the Securities Act and the Proxy Statement/Prospectus related thereto has been filed and distributed, and in any event no later than two (2) Business Days thereafter, Merger Partner shall solicit and obtain the Merger Partner Stockholder Approval by the Written Consents for the purposes of (1) evidencing the adoption of this Agreement and the approval of the Merger and the other transactions contemplated hereby, (2) acknowledging that the approval given thereby is irrevocable and that such stockholder is aware of its rights to demand appraisal for its shares pursuant to Section 262 of the DGCL, a copy of which was attached to the Written Consent, and that such stockholder has received and read a copy of Section 262 of the DGCL and (3) acknowledging that by its approval of the Merger it is not entitled to appraisal rights with respect to its shares in connection with the Merger and thereby waives any rights to receive payment in cash of the fair value of its Merger Partner Capital Stock under Section 262 the DGCL. Reasonably promptly following receipt of the Merger Partner Stockholder Approval by the Written Consents, Merger Partner shall prepare and mail a notice (the “Merger Partner Stockholder Notice”) to every stockholder of Merger Partner that did not execute the Written Consents (in form and substance reasonably acceptable to Public Company). The Merger Partner Stockholder Notice shall (i) be a statement to the effect that the Merger Partner Board determined that the Merger is advisable in accordance with Section 251(b) of the DGCL and in the best interests of the stockholders of Merger Partner and approved and adopted this Agreement, the Merger and the other transactions contemplated hereby and (ii) provide the stockholders of the Merger Partner to whom it is sent with notice of the availability of appraisal rights and notice of the actions taken in the Merger Partner’s Stockholder Approval, including the adoption and approval of this Agreement, the Merger and the other transactions contemplated hereby in accordance with Sections 228(e) and 262 of the DGCL and the certificate of incorporation and bylaws of the Merger Partner. In connection with the Merger Partner Stockholder Approval, Merger Partner shall comply with all disclosure and other obligations to its stockholders under the DGCL and any other applicable Laws. Merger Partner shall take all action that is both reasonable and lawful to obtain the Merger Partner Stockholder Approval. Without limiting the generality of the foregoing, Merger Partner agrees that its obligations under this Section 6.5(a) shall not be affected by the commencement, public proposal, public disclosure or communication to Merger Partner of any Acquisition Proposal or a Merger Partner Board Recommendation Change. Any solicitation or similar disclosure circulated to Merger Partner’s stockholders in connection with this Agreement and the Merger shall be in form and substance reasonably satisfactory to Public Company and, except in the case of a Merger Partner Board Recommendation Change, any solicitation or similar disclosure, if the Merger Partner Stockholder Approval has not already been obtained, shall include the recommendation of Merger Partner Board that Merger Partner’s stockholders consent to the adoption of this Agreement and approval of the Merger.
(b) Subject to Section 6.1(b), Public Company, acting through the Public Company Board, shall take all actions in accordance with applicable Laws, its articles of incorporation and bylaws and Nasdaq rules to duly call, give notice of, convene and hold as promptly as practicable, after the declaration of effectiveness of the Registration Statement, the Public Company Meeting for the purpose of considering and voting upon the Public Company Stockholder Approvals. Subject to Section 6.1(b), the Public Company Board shall include in the Proxy Statement/Prospectus the recommendation of the Public Company Board in favor of approval of the Public Company Stockholder Approvals. Subject to Section 6.1(b), Public Company shall take all action that is both reasonable and lawful to solicit from its stockholders proxies in favor of the Public Company Stockholder Approvals. The Public Company Meeting shall be held as promptly as practicable after the effective date of the Registration Statement (on a date selected by Public Company in consultation with Merger Partner) but in no event later than thirty-five (35) days after the effective date of the Registration Statement. If prior to the originally scheduled date of the Public Company Meeting, as set forth in the Proxy Statement/Prospectus, Public Company reasonably believes that (A) there are insufficient shares of Public Company Common Stock represented (either in person or by proxy) to constitute a quorum necessary to vote upon the Public Company Stockholder Approvals or (B) Public Company will not receive proxies sufficient to obtain the approval of the Public Company Stockholder Approvals, whether or not a quorum would be present, Public Company shall have the right to adjourn or postpone (or make one or more successive postponements or adjournments of) the Public Company Meeting to a later date, provided that no such adjournment shall exceed thirty (30) calendar days from the original date that the Public Company Meeting was scheduled.
(c) Unless the Public Company Board has effected a Public Company Board Recommendation Change in accordance with Section 6.1 and terminated this Agreement to enter into a definitive agreement with respect to a Superior Proposal pursuant to Section 8.1, Public Company’s obligation to call, give notice of and hold the Public Company Meeting in accordance with Section 6.5(b) shall not be limited or otherwise affected by the commencement, disclosure, announcement or submission of any Acquisition Proposal.
(d) Except in the case of a Public Company Board Recommendation Change made in compliance with Section 6.1, (i) Public Company shall use its reasonable best efforts to solicit from the Public Company stockholders proxies in favor of the Public Company Stockholder Approvals, (ii) Public Company shall ensure that all proxies solicited in connection with the Public Company Meeting are solicited in material compliance with all applicable Laws and (iii) Public Company, in its capacity as the sole stockholder of Merger Sub, shall approve the Merger.
(e) Notwithstanding the foregoing, nothing herein shall limit a party’s right to terminate this Agreement pursuant to Section 8.1.
6.6. Legal Conditions to Merger.
(a) Subject to the terms hereof, including Section 6.6(b), Merger Partner and Public Company shall each use reasonable best efforts to (i) take, or cause to be taken, all actions, and do, or cause to be done, and to assist and cooperate with the other parties in doing, all things necessary, proper or advisable to consummate and make effective the transactions contemplated hereby as promptly as practicable, (ii) as promptly as practicable, obtain from any Governmental Entity or any other third party any consents, licenses, permits, waivers, approvals, authorizations, or orders required to be obtained or made by Merger Partner or Public Company or any of their subsidiaries in connection with the authorization, execution and delivery of this Agreement and the consummation of the transactions contemplated hereby, (iii) as promptly as practicable, make all necessary filings, and thereafter make any other required submissions, with respect to this Agreement and the Merger required under (A) the Securities Act and the Exchange Act, and any other applicable federal or state securities Laws, and (B) any other applicable Laws, and (iv) execute or deliver any additional instruments necessary to consummate the transactions contemplated by, and to fully carry out the purposes of, this Agreement. Merger Partner and Public Company shall reasonably cooperate with each other in connection with the making of all such filings. Merger Partner and Public Company shall use their respective reasonable best efforts to furnish to each other all information required for any application or other filing to be made pursuant to the rules and regulations of any applicable Law (including all information required to be included in the Proxy Statement/Prospectus and the Registration Statement) in connection with the transactions contemplated by this Agreement. For the avoidance of doubt, Public Company and Merger Partner agree that nothing contained in this Section 6.6(a) shall modify or affect their respective rights and responsibilities under Section 6.6(b).
(b) Each of Merger Partner and Public Company shall use reasonable best efforts to give (or shall cause their respective subsidiaries to give) any notices to third parties, and use, and cause their respective subsidiaries to use, their reasonable best efforts to obtain any third party consents related to or required in connection with the Merger that are (i) necessary to consummate the transactions contemplated hereby, (ii) disclosed or required to be disclosed in the Merger Partner Disclosure Schedule or the Public Company Disclosure Schedule, as the case may be, or (iii) required to prevent the occurrence of an event that may have a Merger Partner Material Adverse Effect or a Public Company Material Adverse Effect, as the case may be, from occurring prior to or after the Effective Time. Notwithstanding the foregoing, upon request of Merger Partner, Public Company will provide a guaranty of any Merger Partner Leases requested by a lessor thereunder to the extent such guaranty is conditioned on the occurrence of the Closing and effective at or after the Effective Time.
(c) Subject to the terms hereof, Public Company and Merger Partner agree, and shall cause each of their respective subsidiaries, to (i) cooperate and to use their respective commercially reasonable efforts to obtain any required government clearances or approvals under any other federal, state or foreign Law or, regulation or decree designed to prohibit, restrict or regulate actions for the purpose or effect of monopolization or restraint of trade (“Antitrust Laws”), and (ii) respond to any government requests for information under any Antitrust Law. The parties hereto will consult and cooperate with one another, and consider in good faith the views of one another, in connection with any analyses, appearances, presentations, memoranda, briefs, arguments, opinions and proposals made or submitted by or on behalf of any party hereto in connection with proceedings under or relating to any Antitrust Law.
6.7. Public Disclosure. The initial press release announcing the execution of this Agreement shall be issued only in such form as shall be mutually agreed upon by Public Company and Merger Partner. Neither party shall issue any other press release or otherwise make any public statement with respect to the Merger or this Agreement unless required by applicable Law or stock exchange rule, in which case the party required to make such disclosure shall use commercially reasonable efforts to consult with the other party before making any such press release or public statement; provided that Public Company may comply with SEC requirements under the Securities Act or the Exchange Act that require any disclosure, without the consent of Merger Partner. Without limiting the foregoing, Public Company shall, by 9:00 a.m. Eastern Time, on the first Business Day immediately following the date hereof, file with the SEC a Current Report on Form 8-K (the “Signing 8-K”) in form and substance as reasonably approved by Merger Partner (which approval shall not be unreasonably withheld, conditioned or delayed).
6.8. Section 368(a) Reorganization; Section 351 Exchange.
(a) The parties intend that the Merger (i) shall be treated as a transaction that qualifies as a “reorganization” within the meaning of Section 368(a) of the Code, and (ii) in the event that the Control Requirement is satisfied shall also qualify as a non-taxable exchange of shares of Merger Partner Capital Stock for shares of Public Company Common Stock within the meaning of Section 351(a) of the Code. Other than transactions or actions contemplated by this Agreement, each of Public Company, Merger Sub and Merger Partner shall use reasonable best efforts to cause the Merger to qualify, and agree not to, and not to permit or cause any of their Affiliates to, take any action or cause any action to be taken which to its knowledge would reasonably be expected to prevent or impede the Merger from qualifying, as a “reorganization” within the meaning of Section 368(a) of the Code and, in the event the Control Requirement is satisfied, as a non-taxable exchange of shares of Merger Partner Capital Stock for shares of Public Company Common Stock within the meaning of Section 351(a) of the Code (the “U.S. Tax Treatment”). This Agreement is intended to constitute, and the parties hereto hereby adopt this Agreement as, a “plan of reorganization” within the meaning of Treasury Regulation Sections 1.368-2(g) and 1.368-3(a). Each of Public Company, Merger Sub and Merger Partner shall report the Merger as a “reorganization” within the meaning of Section 368(a) of the Code unless otherwise required pursuant to a “determination” within the meaning of Section 1313(a) of the Code, including attaching the statement described in Treasury Regulations Section 1.368-3(a) on or with its Tax Return for the taxable year of the Merger.
(b) If, in connection with the preparation and filing of the Registration Statement or any other filing required by applicable Law or the SEC’s review thereof, the SEC requests or requires that a tax opinion with respect to the U.S. federal income tax consequences of the Merger and the intended U.S. Tax Treatment be prepared and submitted (a “Tax Opinion”), (i) Public Company and Merger Partner shall each use their respective reasonable best efforts to deliver to Mintz, Levin, Cohn, Ferris, Glovsky, and Popeo, P.C., counsel to Public Company, and to Troutman Pepper Hamilton Sanders, LLP, counsel to Merger Partner, customary Tax representation letters satisfactory to each such counsel, dated and executed as of the date such relevant filing shall have been declared effective by the SEC and such other date(s) as determined to be reasonably necessary by each such counsel in connection with the preparation and filing of such Registration Statement or any other filing required by applicable Law, (ii) Public Company shall use its reasonable best efforts to cause Mintz, Levin, Cohn, Ferris, Glovsky, and Popeo, P.C. to furnish a Tax Opinion addressed to Public Company, subject to customary assumptions and limitations, satisfactory to the SEC and (iii) Merger Partner shall use its reasonable best efforts to cause Troutman Pepper Hamilton Sanders, LLP to furnish a Tax Opinion addressed to Merger Partner, subject to customary assumptions and limitations, satisfactory to the SEC.
6.9. Affiliate Legends. Section 6.9 of the Merger Partner Disclosure Schedule sets forth a list of those Persons who are, in Merger Partner’s reasonable judgment, “affiliates” of Merger Partner within the meaning of Rule 145 promulgated under the Securities Act (“Rule 145 Affiliates”). Merger Partner shall notify Public Company in writing regarding any change in the identity of its Rule 145 Affiliates prior to the Closing Date. Public Company shall be entitled to place appropriate legends on the certificates evidencing any shares of Public Company Common Stock to be received by Rule 145 Affiliates of Merger Partner in the Merger reflecting the restrictions set forth in Rule 145 promulgated under the Securities Act and to issue appropriate stop transfer instructions to the transfer agent for Public Company Common Stock.
6.10. D&O Indemnification.
(a) From the Effective Time through the sixth (6th) anniversary of the date on which the Effective Time occurs, each of Public Company and the Surviving Corporation shall, jointly and severally, indemnify and hold harmless each Person who is now, or has been at any time prior to the date hereof, or who becomes prior to the Effective Time, a director or officer of Merger Partner, Public Company or any of their respective subsidiaries (the “Indemnified Persons”), against all claims, losses, liabilities, damages, judgments, fines and reasonable fees, costs and expenses, including attorneys’ fees and disbursements, incurred in connection with any claim, action, suit, proceeding or investigation, whether civil, criminal, administrative or investigative, arising out of or pertaining to the fact that the Indemnified Person is or was an officer, director, employee or agent of Merger Partner, Public Company or any of their respective subsidiaries, or, while a director or officer of Merger Partner, Public Company or any of their respective subsidiaries, is or was serving at the request of Merger Partner, Public Company or any of their respective subsidiaries as a director, officer, employee or agent of another Person, whether asserted or claimed prior to, at or after the Effective Time, to the extent permitted under the applicable certificate or articles of incorporation and bylaws. Each Indemnified Person will be entitled to advancement of expenses (including reasonable attorneys’ fees) incurred in the defense of any such claim, action, suit, proceeding or investigation from each of Public Company and the Surviving Corporation following receipt by Public Company or the Surviving Corporation from the Indemnified Party of a request therefor; provided, that any Person to whom expenses are advanced provides an undertaking, to the extent then required by the DGCL or the NRS, as applicable, to repay such advances if it is ultimately determined that such Person is not entitled to indemnification. From the Effective Time through the sixth (6th) anniversary of the date on which the Effective Time occurs, the certificate of incorporation and bylaws of the Surviving Corporation will contain provisions at least as favorable as the provisions relating to the indemnification, advance of expenses and elimination of liability for monetary damages set forth in the certificate or articles of incorporation and bylaws of Merger Partner and Public Company immediately prior to the Effective Time.
(b) Prior to the Effective Time, Public Company shall purchase a six (6)-year prepaid “D&O tail policy” (the “D&O Public Company Tail Policy”) for the non-cancellable extension of the directors’ and officers’ liability coverage of Public Company’s existing directors’ and officers’ insurance policies for a claims reporting or discovery period of at least six (6) years from and after the Effective Time with respect to any claim related to any period of time at or prior to the Effective Time with terms, conditions, retentions and limits of liability to be mutually agreed by Public Company and Merger Partner prior to the Closing (which approval will not be unreasonably withheld, conditioned or delayed), but that are no less favorable than the coverage provided under Public Company’s existing policies as of the date of this Agreement with respect to coverage of any actual or alleged error, misstatement, misleading statement, act, omission, neglect, breach of duty or any matter claimed against a director or officer of Public Company by reason of him or her serving in such capacity that existed or occurred at or prior to the Effective Time (including in connection with this Agreement or the Merger).
(c) Prior to the Effective Time, Merger Partner shall purchase a six (6)-year prepaid “D&O tail policy” (the “Merger Partner Tail Policy”) for the non-cancellable extension of the directors’ and officers’ liability coverage of Merger Partner’s existing directors’ and officers’ insurance policies for a claims reporting or discovery period of at least six (6) years from and after the Effective Time with respect to any claim related to any period of time at or prior to the Effective Time with terms, conditions, retentions and limits of liability that are no less favorable than the coverage provided under Merger Partner’s existing policies as of the date of this Agreement with respect to any actual or alleged error, misstatement, misleading statement, act, omission, neglect, breach of duty or any matter claimed against a director or officer of Merger Partner by reason of him or her serving in such capacity that existed or occurred at or prior to the Effective Time (including in connection with this Agreement or the Merger).
(d) Public Company shall pay all expenses, including reasonable attorneys’ fees, that may be incurred by a Person in successfully enforcing such Person’s rights provided in this Section 6.10.
(e) Public Company and Merger Partner agree that all rights to exculpation, indemnification and advancement of expenses for acts or omissions occurring at or prior to the Effective Time, whether asserted or claimed prior to, at or after the Effective Time, now existing in favor of the current or former directors, officers or employees, as the case may be, of Public Company, Merger Partner or any of their respective subsidiaries as provided in their respective certificates of incorporation or bylaws or other organization documents or in any agreement in existence immediately prior to the Effective Time shall survive the Merger and shall continue in full force and effect. The provisions of this Section 6.10 are intended to be in addition to the rights otherwise available to the current officers and directors of Public Company, Merger Partner or any of their respective subsidiaries by Law, charter, statute, bylaw or agreement, and shall operate for the benefit of, and shall be enforceable by, each of the Indemnified Persons, their heirs and their representatives. The obligations set forth in this Section 6.10 shall not be terminated, amended or otherwise modified in any manner that adversely affects any Indemnified Person, or any Person who is a beneficiary under the policies referred to in this Section 6.10 and their heirs and representatives, without the prior written consent of such affected Indemnified Person or other Person.
(f) If the Surviving Corporation or Public Company or any of their respective successors or assigns shall (i) consolidate with or merge into any other Person and shall not be the continuing or surviving corporation or entity of such consolidation or merger, or (ii) transfer all or substantially all of its properties and assets to any Person, then, and in each such case, proper provisions shall be made so that the successors and assigns of such Person shall assume all of the obligations of such Person set forth in this Section 6.10.
(g) Nothing in this Agreement is intended to, shall be construed to or shall release, waive or impair any rights to directors’ and officers’ insurance claims under any policy that is or has been in existence with respect to Merger Partner, Public Company or any of their respective subsidiaries for any of their respective directors, officers or other employees, it being understood and agreed that the indemnification provided for in this Section 6.10 is not prior to or in substitution for any such claims under such policies.
6.11. Notification of Certain Matters. Public Company shall give prompt notice to Merger Partner, and Merger Partner shall give prompt notice to Public Company, upon becoming aware of the occurrence, or failure to occur, of any event, which occurrence or failure to occur would be reasonably likely to cause (a) any representation or warranty of such party contained in this Agreement to be untrue or inaccurate in a manner that would reasonably be expected to cause the failure of a condition set forth in Article VII, in each case, at any time from and after the date of this Agreement until the Effective Time, or (b) any material failure of Public Company and Merger Sub or Merger Partner, as the case may be, or of any officer, director, employee or agent thereof, to comply with or satisfy any covenant, condition or agreement to be complied with or satisfied by it under this Agreement.
6.12. Employee Communications. Public Company and Merger Partner will use reasonable best efforts to consult with each other, and will consider in good faith each other’s advice, prior to sending any formal written notices or other formal written communication materials to all or a material portion of its employees or other individual service providers regarding this Agreement, the Merger or the effects thereof on the employment or service, compensation or benefits of its employees or other individual service providers.
6.13. FIRPTA Tax Certificates. On or prior to the Closing, Merger Partner shall deliver to Public Company a properly executed certification that shares of Merger Partner Capital Stock are not “United States real property interests” in accordance with the Treasury Regulations under Sections 897 and 1445 of the Code, together with a notice to the IRS (which shall be filed by Public Company with the IRS following the Closing) in accordance with the provisions of Treasury Regulations Section 1.897-2(h)(2) of the Treasury Regulations.
6.14. State Takeover Laws. If any “fair price,” “business combination” or “control share acquisition” statute or other similar statute or regulation (or any similar anti-takeover provision of Merger Partner’s or Public Company’s governing documents) is or may become applicable to any of the transactions contemplated by this Agreement, the parties hereto shall use their respective commercially reasonable efforts to (a) take such actions as are reasonably necessary so that the transactions contemplated hereunder may be consummated as promptly as practicable on the terms contemplated hereby and (b) otherwise take all such actions as are reasonably necessary to eliminate or minimize the effects of any such statute or regulation on such transactions.
6.15. Security Holder Litigation. Notwithstanding anything to the contrary herein, (a) Public Company shall have the right to control the defense and settlement of any litigation related to this Agreement (“Security Holder Litigation”), the Merger or the other transactions contemplated by this Agreement brought by any stockholder or any holder of other securities of Public Company, and whether a direct or derivative claim, against Public Company, any subsidiary of Public Company and/or any of their respective directors or officers; provided, that Public Company shall give Merger Partner the opportunity to participate in the defense of any such Security Holder Litigation and shall not settle any such Security Holder Litigation without the prior written consent of Merger Partner (which consent shall not be unreasonably withheld, conditioned or delayed), and (b) Merger Partner shall have the right to control the defense and settlement of any Security Holder Litigation, the Merger or the other transactions contemplated by this Agreement brought by any stockholder or any holder of other securities of Merger Partner against Merger Partner and/or its directors or officers; provided, that Merger Partner shall give Public Company the opportunity to participate in the defense of any such Security Holder Litigation and shall not settle any such Security Holder Litigation without the prior written consent of Public Company (which consent shall not be unreasonably withheld, conditioned or delayed).
6.16. Section 16 Matters. Prior to the Effective Time, Public Company shall take all such steps as may be required (to the extent permitted under applicable Law and no-action letters issued by the SEC) to cause any acquisitions of Public Company Common Stock (and any options to purchase the same) in connection with this Agreement and the transactions contemplated hereby, by each individual who is reasonably expected to become subject to the reporting requirements of Section 16(a) of the Exchange Act with respect to Public Company following the Merger, to be exempt under Rule 16b-3 promulgated under the Exchange Act; provided, that Merger Partner has provided Public Company with information concerning directors, officers and/or equityholders of Merger Partner sufficient for Public Company to take such steps.
6.17. Calculation of Public Company Net Cash and Exchange Ratio.
(a) Not less than ten (10) Business Days prior to the anticipated date for Closing (the “Anticipated Closing Date”), Public Company shall deliver to Merger Partner a draft schedule (the “Draft Public Company Net Cash Schedule”) setting forth, in reasonable detail, Public Company’s good faith, estimated calculation of (i) Public Company Net Cash, and (ii) the Exchange Ratio, in each case, as of the Anticipated Closing Date; provided, that Merger Partner shall make available to Public Company the Merger Partner capitalization information required to calculate the Exchange Ratio, or any such additional information of Merger Partner as Public Company may reasonably request. Public Company shall make available to Merger Partner the work papers and back-up materials used in or reasonably relevant to preparing the Draft Public Company Net Cash Schedule and, if reasonably requested by Merger Partner, Public Company’s accountants and counsel at reasonable times and upon reasonable advance notice. Public Company shall consider in good faith any comments provided by Merger Partner within four (4) Business Days of delivery of the Draft Public Company Net Cash Schedule.
(b) Not less than four (4) (but no more than six (6)) Business Days prior to the Anticipated Closing Date, Public Company will deliver to Merger Partner a proposed final schedule (the “Final Public Company Net Cash Schedule”) setting forth, in reasonable detail, Public Company’s good faith, estimated calculation of (i) Public Company Net Cash, and (ii) the Exchange Ratio, prepared and certified by Public Company’s Chief Financial Officer (or if there is no Chief Financial Officer, the Chief Executive Officer of Public Company), which shall be subject to the agreement and consent of Merger Partner. Public Company shall make available to Merger Partner, as requested by Merger Partner, the work papers and back-up materials used in or reasonably relevant to preparing the Final Public Company Net Cash Schedule and, if reasonably requested by Merger Partner, Public Company’s accountants and counsel at reasonable times and upon reasonable advance notice.
(c) On the fourth (4th) calendar day following Public Company’s delivery of the Final Public Company Net Cash Schedule pursuant to Section 6.17(b), the Final Public Company Net Cash Schedule shall become final and binding on all parties to this Agreement and to represent Public Company Net Cash on the Closing Date (the “Final Public Company Net Cash”) unless on or prior to the third (3rd) calendar day following such delivery Merger Partner disputes the Final Public Company Net Cash and/or the Exchange Ratio set forth on the Final Public Company Net Cash Schedule by delivering a written notice to Public Company describing in reasonable detail each item in dispute and Merger Partner’s proposed revisions to the Final Public Company Net Cash Schedule (a “Dispute Notice”).
(d) If Merger Partner timely delivers a Dispute Notice to Public Company, then Representatives of Public Company and Merger Partner shall promptly meet and attempt in good faith to resolve any disputes between them regarding the Final Public Company Net Cash Schedule and negotiate an agreed-upon determination of Public Company Net Cash, which agreed upon Public Company Net Cash amount shall be deemed to have been finally determined for purposes of this Agreement and to represent the Final Public Company Net Cash at the Cash Determination Time for all purposes under this Agreement. If prior to the date that is two (2) Business Days prior to the Anticipated Closing Date, Representatives of Public Company and Merger Partner are unable to negotiate an agreed-upon determination of any disputed items in the Final Public Company Net Cash Schedule prior to the Anticipated Closing Date, then any such disputed items shall be referred to an independent auditor of recognized national standing jointly selected by Public Company and Merger Partner (the “Accounting Firm”). At the Accounting Firm’s request, (1) Public Company shall promptly make available or deliver to the Accounting Firm the work papers and back-up materials used by Public Company in preparing the Final Public Company Net Cash Schedule, and (2) Merger Partner shall promptly make available or deliver to the Accounting Firm the work papers and back-up materials used by Merger Partner in disputing the Final Public Company Net Cash Schedule. Public Company and Merger Partner shall use commercially reasonable best efforts to cause the Accounting Firm to resolve all remaining matters in dispute within five (5) Business Days of accepting its selection. Merger Partner and Public Company shall be afforded the opportunity to present to the Accounting Firm any material related to the unresolved disputes and to discuss the issues with the Accounting Firm; provided, however, that no such presentation or discussion shall occur without the presence of a Representative of each of Merger Partner and Public Company. The determination of the Accounting Firm shall be limited to the items in dispute submitted to the Accounting Firm. The Accounting Firm shall act as an expert and not as an arbitrator and shall be instructed to resolve only such matters relating to the Final Public Company Net Cash Schedule as are then in dispute. Any determination of the amount of Final Public Company Net Cash and the Exchange Ratio made by the Accounting Firm shall be made in writing and delivered to each of Public Company and Merger Partner, shall be final and binding on the parties hereto, and shall be deemed to have been finally determined for purposes of this Agreement and to represent the Final Public Company Net Cash and the Exchange Ratio set forth on the Final Public Company Net Cash Schedule for all purposes of this Agreement. The parties shall delay the Closing until the resolution of the matters described in this Section 6.17(d). The fees and expenses of the Accounting Firm shall be allocated between Public Company and Merger Partner in the same proportion that the disputed amount of Final Public Company Net Cash that was unsuccessfully disputed by such party (as finally determined by the Accounting Firm) bears to the total disputed amount of Final Public Company Net Cash. If this Section 6.17(d) applies as to the determination of Final Public Company Net Cash, upon resolution of the matter in accordance with this Section 6.17(d), the parties shall not be required to determine Final Public Company Net Cash again even though the Closing Date may occur later than the Anticipated Closing Date, except that either Public Company or Merger Partner may request a redetermination of Final Public Company Net Cash if the Closing Date is more than five (5) Business Days after the Anticipated Closing Date, in which case the procedures contained in this Section 6.17 shall be repeated, starting with the delivery of a new Draft Public Company Net Cash Schedule.
(e) For purposes of this Agreement, the following terms shall have the following meanings:
(i) “Cash Determination Time” means the close of business as of the Business Day immediately prior to the Anticipated Closing Date.
(ii) “Indebtedness” shall mean any liabilities of Public Company or its subsidiaries (A) for borrowed money, (B) evidenced by bonds, debentures, notes or similar instruments, (C) upon which interest charges are customarily paid (other than obligations accepted in connection with the purchase of products or services in the ordinary course of business), (D) in respect of liabilities of others that are secured by (or which the holder of such liabilities has an existing right, contingent or otherwise, to be secured by) any Lien or security interest on property owned or acquired by the Person in question whether or not the obligations secured thereby have been assumed, (E) under leases required to be accounted for as capital leases under GAAP, or (F) guarantees relating to any such liabilities.
(iii) “Public Company Net Cash” shall mean as of the Cash Determination Time (except as otherwise provided herein) and without duplication, and calculated as if the Merger were consummated as of the Cash Determination Time:
(A) the sum of:
(1) the unrestricted cash, cash equivalents, and marketable securities of Public Company and its subsidiaries;
(2) accounts receivable that would be required to be set forth in a balance sheet prepared in accordance with GAAP and deposits (to the extent refundable or applicable to Merger Partner, Public Company or any subsidiary of Public Company following the Effective Time);
(3) expenses paid, or Liabilities incurred, prior to Closing, that are approved in writing to be covered and reimbursed by the D&O Public Company Tail Policy in excess of the deductible and within overall policy limits;
(4) any amounts owed to Public Company or any of its subsidiaries in respect of a Legacy Asset Disposition, which amounts have not been received as of the Closing Date (in each case, net of any expenses, fees, or Taxes payable by Public Company or its subsidiaries in respect of such Legacy Asset Disposition), if the proceeds of such Legacy Asset Disposition would be reflected as a bona fide account receivable in accordance with GAAP;
(5) the amount of any Tax refund or credit (including, for the avoidance of doubt, the German R&D Tax Credit) (i) received by Public Company or any of its Affiliates or (ii) awarded to Public Company or any of its Affiliates, whether or not such amount is pending, due and owning, or unpaid as of the Closing, if such Tax refund would be reflected as a bona fide account receivable in accordance with GAAP and approved by Merger Partner in its reasonable discretion;
(6) all prepaid expenses and deposits of Public Company set forth on Section 6.17(e)(iii) usable by Merger Partner, Public Company or any subsidiary of Public Company following the Effective Time, or any other prepaid expenses or deposits of Public Company or any of its Affiliates mutually agreed upon by Public Company and Merger Partner;
(7) fifty percent (50%) of the amount of any costs or expenses (including attorney’s fees, settlement amounts or judgments) incurred or paid in connection with any Security Holder Litigation (including all amounts paid or payable up to the retention amount of any insurance policy that covers or may cover such costs or expenses or amounts not covered by any such insurance policy);
(8) fifty percent (50%) of any fees or expenses incurred by Public Company prior to the Effective Time in connection with filing the Nasdaq Listing Application; and
(9) fifty percent (50%) of any fees and expenses incurred by Public Company prior to the Effective Time in connection with Nasdaq, proxy solicitation, exchange agent services, and any and all SEC matters, including, without limitation, the printing, filing and mailing of the Proxy Statement/Prospectus (including any related preliminary materials) and the Registration Statement and any amendments or supplements thereto.
Minus
(B) the sum of:
(1) accrued and unpaid accounts payable and other accrued and unpaid expenses of Public Company and its subsidiaries, other than Transaction Expenses, including legal settlements that are not covered by a director and officer insurance policy and the cost of the D&O Public Company Tail Policy, without duplication, any accrued but unpaid Taxes related to or attributable to all taxable periods, or portions of taxable periods, ending on or before the Closing Date, including, for the avoidance of doubt, any Tax liabilities relating to the disposition of any Legacy Assets after taking into account any available net operating loss carryforwards or other tax attributes of Public Company and any foreign tax credits which foreign tax credits are directly related to the disposition of the Legacy Assets (if any)) and any employer portion employment Taxes incurred in connection with any compensatory payments made to employees and any and all other bona fide current and long-term liabilities payable in cash that would be required to be set forth in a balance sheet prepared in accordance with GAAP;
(2) any unpaid Transaction Expenses of Public Company or its subsidiaries;
(3) any change of control, bonus, deferred compensation, severance (voluntary or otherwise) (including a reasonable estimate of payment or reimbursement for continued coverage under any employee benefit plan), retention or similar payments (whether “single trigger” or “double trigger”) that become due and payable by Public Company or any of its subsidiaries pursuant to Contracts entered into at or prior to the Effective Time as a result of or in connection with the Merger or any Legacy Asset Disposition and remain unpaid as of the Effective Time;
(4) the amount of any notice payments, fines or other payments to be made by Public Company in order to terminate any existing agreement to which Public Company is a party and to wind down any current and future clinical trial obligations, in each case, that remain unpaid as of the Effective Time;
(5) fifty percent (50%) of the amount of any costs or expenses (including attorney’s fees, settlement amounts or judgments) incurred or paid in connection with any Security Holder Litigation (including all amounts paid or payable up to the retention amount of any insurance policy that covers or may cover such costs or expenses or amounts not covered by any such insurance policy) that remain unpaid as of the Effective Time.
Each component of Public Company Net Cash shall be determined in accordance with GAAP applied on a basis consistent with the application of GAAP in the preparation of Public Company’s most recent audited financial statements. For purposes of determining Public Company Net Cash, the Surviving Corporation and its subsidiaries shall not constitute subsidiaries of Public Company. A sample calculation of Public Company Net Cash and its components is set forth in Annex B for illustrative purposes only.
(iv) “Transaction Expenses” shall mean, with respect to Public Company and its subsidiaries as of the Cash Determination Time and without duplication, and calculated as if the Merger were consummated as of the Cash Determination Time, the sum of (A) all premiums, underwriting costs, brokerage commissions, costs, expenses, and other amounts in respect of the D&O Public Company Tail Policy, and (B) all costs, fees and expenses incurred by Public Company or its subsidiaries at or prior to the Effective Time in connection with the negotiation, preparation and execution of this Agreement or any agreements, documents, certificates, opinions or other items contemplated hereby and the consummation of the Merger or the other transactions contemplated hereby or any Legacy Asset Disposition, in each case, that are unpaid as of the Effective Time, including brokerage fees and commissions, finders’ fees or financial advisory fees payable by such Person at or prior to the Effective Time.
6.18. Termination of Section 401(k) Plans. Effective as of no later than the day immediately preceding the Closing Date, the Public Company shall terminate any and all Public Company Employee Plans intended to include a Code Section 401(k) arrangement (each, a “401(k) Plan”). The Public Company shall provide Merger Partner with evidence that each 401(k) Plan has been terminated (effective as of no later than the day immediately preceding the Closing Date) pursuant to resolutions of the Public Company Board as the case may be. The form and substance of such resolutions shall be subject to review and approval of Merger Partner.
6.19. Merger Partner Financials. As promptly as reasonably practicable following the date hereof, Merger Partner shall deliver to Public Company any audited or unaudited consolidated balance sheets and the related audited or unaudited consolidated statements of operations and comprehensive loss, and stockholders’ deficit and cash flows of Merger Partner as of and for a year-to-date period ended as of the end of any other different fiscal quarter (and as of and for the same period from the previous fiscal year) or fiscal year (and as of and for the prior fiscal quarter), as applicable that is required to be included in the Registration Statement or Proxy Statement/Prospectus. All such financial statements, together with any audited or unaudited consolidated balance sheet and the related audited or unaudited consolidated statements of operations and comprehensive loss, and stockholders’ deficit and cash flows of Merger Partner as of and for a year-to-date period ended as of the end of a different fiscal quarter (and as of and for the same period from the previous fiscal year) or fiscal year (and as of and for the prior fiscal quarter) that is required to be included in the Registration Statement or Proxy Statement/Prospectus (A) will fairly present in all material respects the financial position of Merger Partner as of the date thereof, and the results of its operations, stockholders’ equity and cash flows for the respective periods then ended (subject, in the case of any unaudited interim financial statements, to normal year-end audit adjustments, none of which is expected to be material), (B) will be prepared in conformity with GAAP applied on a consistent basis during the periods involved (except, in the case of any audited financial statements, as may be indicated in the notes thereto and subject, in the case of any unaudited financial statements, to normal year-end audit adjustments, none of which is expected to be material), (C) in the case of any audited financial statements, will be audited in accordance with the standards of the PCAOB and contain an unqualified report of Merger Partner’s auditor and (D) will comply in all respects with the applicable accounting requirements and with the rules and regulations of the SEC, the Exchange Act and the Securities Act in effect as of the respective dates thereof (including Regulation S-X or Regulation S-K, as applicable).
6.20. New Public Company Equity Plan; Termination of Public Company ESPP.
(a) Prior to the Effective Time, the Public Company Board will adopt the 2024 Equity Incentive Plan, subject to the Closing and effective as of the Effective Time, and will include provisions in the Proxy Statement for the stockholders of Public Company to approve the 2024 Equity Incentive Plan Proposal.
(b) Public Company will use commercially reasonable efforts to ensure that (i) no new purchase periods under the Public Company’s 2023 Employee Stock Purchase Plan (the “Public Company ESPP”) will commence during the period from the date of this Agreement through the Closing Date; (ii) there will be no increase in the amount of payroll deductions (or other contributions) permitted to be made by participants under the Public Company ESPP during any current purchase periods, except those made in accordance with payroll deduction elections that are in effect as of the date of this Agreement; (iii) there will be no grants of new purchase rights under the Public Company ESPP from the date of this Agreement through the Closing Date; and (iv) no individuals shall commence participation in the Public Company ESPP during the period from the date of this Agreement through the Closing Date. To the extent applicable, no later than ten (10) Business Days prior to the Closing Date, in the case of any outstanding purchase rights under the Public Company ESPP, any then-current purchase period under the Public Company ESPP shall end and each participant’s accumulated payroll deductions shall be used to purchase shares of Public Company Common Stock (rounded down to the nearest whole share) in accordance with the terms of the Public Company ESPP. Prior to the Effective Time, Public Company will use commercially reasonable efforts to terminate the Public Company ESPP as of immediately prior to the Effective Time but contingent upon the occurrence of the Closing. Any accumulated contributions of each participant under the Public Company ESPP, to the extent not used to purchase shares in accordance with the terms of the Public Company ESPP as of the final purchase thereunder, shall be refunded to the applicable participant as promptly as practicable following such final purchase.
ARTICLE VII
CONDITIONS TO MERGER
7.1. Conditions to Each Party’s Obligation to Effect the Merger. The respective obligations of each party to this Agreement to effect the Merger shall be subject to the satisfaction prior to the Closing Date of the following conditions:
(a) Stockholder Approvals. The Merger Partner Voting Proposal shall have been approved by means of the Written Consents by the requisite vote of the stockholders of Merger Partner under applicable Law and Merger Partner’s certificate of incorporation. The Required Public Company Voting Proposals shall have been approved at the Public Company Meeting (or adjournment or postponement thereof), at which a quorum is present, by the requisite vote of the stockholders of Public Company under applicable Law.
(b) Registration Statement; Proxy Statement/Prospectus. The Registration Statement shall have become effective under the Securities Act and no stop order suspending the effectiveness of the Registration Statement shall have been issued and no proceeding for that purpose, and no similar proceeding with respect to the Proxy Statement/Prospectus, shall have been initiated or threatened in writing by the SEC or its staff.
(c) No Injunctions. No Governmental Entity of competent jurisdiction shall have enacted, issued, promulgated, enforced or entered any order, executive order, stay, decree, judgment or injunction (preliminary or permanent) or statute, rule or regulation which is in effect and which has the effect of making the Merger illegal or otherwise prohibiting consummation of the Merger.
(d) Nasdaq Notification. (i) The Nasdaq Listing Application shall have been approved and any notification required by Nasdaq shall have been made, and (ii) the shares of the Public Company Common Stock to be issued pursuant to the Share Issuances shall have been approved for listing (subject to official notice of issuance) on Nasdaq.
(e) Concurrent Financing. The Concurrent Financing shall have been consummated or will be consummated concurrently with the Closing or immediately following the Closing in accordance with the terms of the Subscription Agreement.
(f) Public Company Net Cash. Public Company Net Cash shall have been finally determined in accordance with Section 6.17.
7.2. Additional Conditions to the Obligations of Public Company and Merger Sub. The obligations of Public Company and Merger Sub to effect the Merger shall be subject to the satisfaction on or prior to the Closing Date of each of the following additional conditions, any of which may be waived in writing exclusively by Public Company and Merger Sub:
(a) Representations and Warranties. The representations and warranties of Merger Partner set forth in Article III (in each case as qualified and limited by the Merger Partner Disclosure Schedule) and in any certificate or other writing delivered by Merger Partner pursuant hereto shall be true and correct (i) as of the date of this Agreement (except in the case of this clause (i), (A) to the extent such representations and warranties are specifically made as of a particular date, in which case such representations and warranties shall be true and correct as of such date, and (B) where the failure to be true and correct (without regard to any materiality or Merger Partner Material Adverse Effect qualifications contained therein), individually or in the aggregate, has not had, and is not reasonably likely to have, a Merger Partner Material Adverse Effect) and (ii) as of the Closing Date as though made on and as of the Closing Date (except in the case of this clause (ii), (A) to the extent such representations and warranties are specifically made as of a particular date, in which case such representations and warranties shall be true and correct as of such date, (B) for changes expressly provided for in this Agreement, and (C) where the failure to be true and correct (without regard to any materiality or Merger Partner Material Adverse Effect qualifications contained therein), individually or in the aggregate, has not had, and is not reasonably likely to have, a Merger Partner Material Adverse Effect); provided, however, that the representations and warranties made by Merger Partner in Sections 3.1, 3.2, 3.4(a), 3.4(b), and 3.7(a) shall not be subject to the qualifications set forth in clauses (i)(B) and (ii)(C) above and instead shall be true and correct except where the failure to be true and correct (with regard to any materiality or Merger Partner Material Adverse Effect qualifications contained therein), individually or in the aggregate is not material to Merger Partner.
(b) Performance of Obligations of Merger Partner. Merger Partner shall have performed in all material respects all obligations required to be performed by it under this Agreement on or prior to the Closing Date.
(c) No Merger Partner Material Adverse Effect. No Merger Partner Material Adverse Effect shall have occurred since the date of this Agreement and be continuing.
(d) Officers’ Certificate. Public Company shall have received an officers’ certificate duly executed by the Chief Executive Officer of Merger Partner to the effect that the conditions of Sections 7.2(a), (b) and (c) have been satisfied.
(e) Termination of Investor Agreements. The agreements listed in Section 7.2(e) of the Merger Partner Disclosure Schedule shall have been terminated.
(f) Third Party Consents. Merger Partner shall have obtained (i) all consents and approvals of third parties listed in Section 7.2(f) of the Merger Partner Disclosure Schedule and (ii) any other consent or approval of any third party (other than a Governmental Entity) the failure of which to obtain, individually or in the aggregate, is reasonably likely to have a Merger Partner Material Adverse Effect.
7.3. Additional Conditions to the Obligations of Merger Partner. The obligation of Merger Partner to effect the Merger shall be subject to the satisfaction on or prior to the Closing Date of each of the following additional conditions, any of which may be waived, in writing, exclusively by Merger Partner:
(a) Representations and Warranties. The representations and warranties of Public Company and Merger Sub set forth in Article IV (in each case as qualified and limited by the Public Company Disclosure Schedule) and in any certificate or other writing delivered by Public Company or Merger Sub pursuant hereto shall be true and correct (i) as of the date of this Agreement (except in the case of this clause (i), (A) to the extent such representations and warranties are specifically made as of a particular date, in which case such representations and warranties shall be true and correct as of such date and (B) where the failure to be true and correct (without regard to any materiality or Public Company Material Adverse Effect qualifications contained therein), individually or in the aggregate, has not had, and is not reasonably likely to have, a Public Company Material Adverse Effect) and (ii) as of the Closing Date as though made on and as of the Closing Date (except in the case of this clause (ii), (A) to the extent such representations and warranties are specifically made as of a particular date, in which case such representations and warranties shall be true and correct as of such date, (B) for changes contemplated by this Agreement and (C) where the failure to be true and correct (without regard to any materiality or Public Company Material Adverse Effect qualifications contained therein), individually or in the aggregate, has not had, and is not reasonably likely to have, a Public Company Material Adverse Effect); provided, however, that the representations and warranties made by Public Company and Merger Sub in Sections 4.1, 4.2, 4.4(a), 4.4(d), and 4.7(a), shall not be subject to the qualifications set forth in clauses (i)(B) and (ii)(C) above and instead shall be true and correct except where the failure to be true and correct (with regard to any materiality or Public Company Material Adverse Effect qualifications contained therein), individually or in the aggregate is not material to Public Company.
(b) Performance of Obligations of Public Company and Merger Sub. Public Company and Merger Sub shall have performed in all material respects all obligations required to be performed by them under this Agreement on or prior to the Closing Date.
(c) No Public Company Material Adverse Effect. No Public Company Material Adverse Effect shall have occurred since the date of this Agreement and be continuing.
(d) Resignations. Merger Partner shall have received copies of the resignations, effective as of the Effective Time, of each director and officer (for such officers, limited to the offices held by such officers and not to such officer’s employment) of Public Company and its subsidiaries, other than a resignation from the individual designated a director to Public Company Board by the Public Company in compliance with Section 1.5(a).
(e) Third Party Consents. Public Company shall have obtained (i) all consents and approvals of third parties listed in Section 7.3(e) of the Public Company Disclosure Schedule and (ii) any other consent or approval of any third party (other than a Governmental Entity) the failure of which to obtain, individually or in the aggregate, is reasonably likely to have a Public Company Material Adverse Effect.
(f) Officers’ Certificate. Merger Partner shall have received an officers’ certificate duly executed by the Chief Executive Officer of Public Company to the effect that the conditions of Sections 7.3(a), (b), and (c) have been satisfied.
ARTICLE VIII
TERMINATION AND AMENDMENT
8.1. Termination. This Agreement may be terminated at any time prior to the Effective Time (with respect to Sections 8.1(b) through 8.1(k), by written notice by the terminating party to the other party), whether before or, subject to the terms hereof, after approval of the Merger Partner Voting Proposal by the stockholders of Merger Partner or approval of the Required Public Company Voting Proposals by the stockholders of Public Company:
(a) by mutual written consent of Public Company and Merger Partner;
(b) by either Public Company or Merger Partner if the Merger shall not have been consummated by March 31, 2025 (the “Outside Date”) (provided that the right to terminate this Agreement under this Section 8.1(b) shall not be available to Public Company or Merger Partner if such party’s (or in the case of Public Company, Merger Sub’s) failure to fulfill any obligation under this Agreement has been a principal cause of the failure of the Merger to occur on or before the Outside Date); provided, that, if as of such date all conditions set forth in Article VII (other than the condition set forth in Section 7.1(f)) have been satisfied or waived, the Outside Date shall automatically be extended until the date that is two (2) Business Days following the final determination of Final Public Company Net Cash in accordance with Section 6.17;
(c) by either Public Company or Merger Partner if a Governmental Entity of competent jurisdiction shall have issued a nonappealable final order, decree or ruling or taken any other nonappealable final action, in each case having the effect of permanently restraining, enjoining or otherwise prohibiting the Merger; provided, that a party hereto shall not be permitted to terminate this Agreement pursuant to this Section 8.1(c) if the issuance of any such order, decree, ruling or other action is principally attributable to the failure of such party (or any Affiliate of such party) to perform in any material respect any covenant in this Agreement required to be performed by such party (or any Affiliate of such party) at or prior to the Effective Time;
(d) by either Public Company or Merger Partner if (i) the Public Company Meeting (including, if applicable, following adjournments or postponements thereof as permitted or required pursuant to Section 6.5(b)) shall have been held and completed and the stockholders of Public Company shall have taken a final vote on the Required Public Company Voting Proposals and (ii) the Required Public Company Voting Proposals shall not have been approved at the Public Company Meeting (or at any adjournment or postponement thereof) by the requisite vote of the stockholders of Public Company under applicable Law; provided, that the right to terminate under this Section 8.1(d) shall not be available to Public Company where Public Company’s failure to fulfill any obligation under this Agreement has been the principal cause of the failure to obtain the requisite vote of the stockholders of Public Company;
(e) by Public Company, if at any time prior to the receipt of the Merger Partner Stockholder Approval: (i) the Merger Partner Board shall have effected a Merger Partner Board Recommendation Change, or (ii) Merger Partner shall have materially breached its obligations under Section 6.1 or Section 6.5(a);
(f) by Merger Partner, at any time prior to the receipt of the Required Public Company Stockholder Approval, if: (i) the Public Company Board shall have effected a Public Company Board Recommendation Change, or (ii) Public Company shall have materially breached its obligations under Section 6.1 or Section 6.5(b);
(g) by Public Company, if there has been a breach of or failure to perform any representation, warranty, covenant or agreement set forth in this Agreement on the part of Merger Partner, which breach would cause the conditions set forth in Section 7.2(a) or Section 7.2(b) not to be satisfied; provided, that Public Company is not then in material breach of any representation, warranty or covenant under this Agreement; and provided, further, that if such breach or failure to perform is curable by Merger Partner, as applicable, then this Agreement shall not terminate pursuant to this Section 8.1(g) as a result of such particular breach or failure until the expiration of a thirty (30)-calendar day period commencing upon delivery of written notice from Public Company to Merger Partner of such breach or failure and it being understood that this Agreement shall not terminate pursuant to this Section 8.1(g) as a result of such particular breach or failure if such breach or failure is cured prior to such termination becoming effective;
(h) by Merger Partner, if there has been a breach of or failure to perform any representation, warranty, covenant or agreement set forth in this Agreement on the part of Public Company, which breach would cause the conditions set forth in Section 7.3(a) or Section 7.3(b) not to be satisfied; provided, that Merger Partner is not then in material breach of any representation, warranty or covenant under this Agreement; and provided, further, that if such breach or failure to perform is curable by Public Company or Merger Sub, then this Agreement shall not terminate pursuant to this Section 8.1(h) as a result of such particular breach or failure until the expiration of a thirty (30)-calendar day period commencing upon delivery of written notice from Merger Partner to Public Company of such breach or failure and it being understood that this Agreement shall not terminate pursuant to this Section 8.1(h) as a result of such particular breach or failure if such breach or failure is cured prior to such termination becoming effective;
(i) by Public Company, if the Merger Partner Stockholder Approval is not obtained by delivery of the Written Consents on or prior to 5:00 p.m., New York City time, on the date that is two (2) Business Days after the Registration Statement is declared effective under the Securities Act;
(j) by Merger Partner if, at any time prior to the receipt of the Merger Partner Stockholder Approval, each of the following occur: (i) Merger Partner shall have received a Superior Proposal; (ii) Merger Partner shall have complied in all material respects with its obligations under Section 6.1 with respect to such Superior Proposal, including with respect to making a Merger Partner Board Recommendation Change with respect to such Superior Proposal; (iii) the Merger Partner Board approves, and Merger Partner substantially concurrently with the termination of this Agreement enters into, a definitive agreement with respect to such Superior Proposal; and (iv) prior to or concurrently with such termination, Merger Partner pays to the Public Company the amount contemplated by Section 8.3(b); or
(k) by Public Company if, at any time prior to the receipt of the Required Public Company Stockholder Approval, each of the following occur: (i) Public Company shall have received a Superior Proposal; (ii) Public Company shall have complied in all material respects with its obligations under Section 6.1 with respect to such Superior Proposal, including with respect to making a Public Company Board Recommendation Change with respect to such Superior Proposal; (iii) the Public Company Board approves, and Public Company substantially concurrently with the termination of this Agreement enters into, a definitive agreement with respect to such Superior Proposal; and (iv) prior to or concurrently with such termination, Public Company pays to Merger Partner the amount contemplated by Section 8.3(c).
8.2. Effect of Termination. In the event of termination of this Agreement as provided in Section 8.1, this Agreement shall immediately become void and there shall be no liability or obligation on the part of Public Company, Merger Partner, Merger Sub or their respective officers, directors, stockholders or Affiliates; provided, that (a) any such termination shall not relieve any party from liability for any material and willful breach of this Agreement, fraud or intentional misconduct and (b) the provisions of Section 5.3 (Confidentiality), this Section 8.2 (Effect of Termination), Section 8.3 (Fees and Expenses) and Article IX (Miscellaneous) (other than Section 9.14) and the Confidentiality Agreement shall remain in full force and effect and survive any termination of this Agreement. A “material and willful breach” by a party of a provision of this Agreement means that the party knowingly undertook an action, or failed to undertake an action, with the understanding that the action, or failure to act, was a material breach by such party of the applicable provisions of this Agreement. For purposes of this Agreement, the failure to consummate the Closing pursuant to, and when required by, the terms of this Agreement shall constitute a material and willful breach hereunder.
8.3. Fees and Expenses.
(a) Except as set forth in this Section 8.3 or expressly set forth in this Agreement, all fees and expenses incurred in connection with this Agreement and the transactions contemplated hereby shall be paid by the party incurring such expenses, whether or not the Merger is consummated; provided, however, that Merger Partner and Public Company shall share equally (i) all fees and expenses of the Exchange Agent, (ii) all fees and expenses, other than accountant’s and attorneys’ fees, incurred with respect to the printing, filing and mailing of the Proxy Statement/Prospectus (including any related preliminary materials) and the Registration Statement and any amendments or supplements thereto, and (iii) all fees and expenses incurred in connection with the engagement of any proxy soliciting firms.
(b) Merger Partner shall pay Public Company a termination fee of Two Million dollars ($2,000,000) (the “Merger Partner Termination Fee”) in the event of the termination of this Agreement:
(i) by Public Company pursuant to Section 8.1(e);
(ii) by Merger Partner pursuant to Section 8.1(j); or
(iii) by Public Company or Merger Partner, as applicable, pursuant to Section 8.1(b) or Section 8.1(g) or Section 8.1(i) so long as (A) prior to the termination of this Agreement, any Person makes an Acquisition Proposal or amends an Acquisition Proposal made prior to the date of this Agreement with respect to Merger Partner; and (B) within twelve (12) months after such termination Merger Partner enters into a definitive agreement to consummate (which is consummated, whether or not within or after the twelve (12)-month period), or consummates, any Acquisition Proposal (regardless of whether made before or after the termination of this Agreement); provided, that for purposes of this Section 8.3(b)(iii), the references to 15% in the definition of Acquisition Proposal shall be deemed to be 50%.
(c) Public Company shall pay Merger Partner a termination fee of One Million dollars ($1,000,000) (the “Public Company Termination Fee”) in the event of the termination of this Agreement:
(i) by Merger Partner pursuant to Section 8.1(f);
(ii) by Public Company pursuant to Section 8.1(k); or
(iii) by Public Company or Merger Partner, as applicable, pursuant to Section 8.1(b), Section 8.1(d) or Section 8.1(h), so long (A) prior to the termination of this Agreement, any Person makes an Acquisition Proposal or amends an Acquisition Proposal made prior to the date of this Agreement with respect to Public Company; and (B) within twelve (12) months after such termination Public Company enters into a definitive agreement to consummate (which is consummated, whether or not within or after the twelve (12)-month period), or consummates, any Acquisition Proposal (regardless of whether made before or after the termination of this Agreement); provided, that for purposes of this Section 8.3(c)(iii), the references to 15% in the definition of Acquisition Proposal shall be deemed to be 50%.
(d) If this Agreement is terminated by Merger Partner pursuant to Section 8.1(d), Public Company shall reimburse Merger Partner for all reasonable out of pocket fees and expenses (the “Third Party Expenses”) incurred by Merger Partner in connection with this Agreement and the consummation of the transactions contemplated hereby, up to a maximum of Five Hundred Thousand Dollars ($500,000), by wire transfer of same day funds within five (5) Business Days following the date on which Merger Partner submits to Public Company true and correct copies of reasonable documentation supporting such expenses.
(e) If this Agreement is terminated by Public Company pursuant to Section 8.1(i), Merger Partner shall reimburse Public Company for all Third Party Expenses incurred by Public Company in connection with this Agreement and the consummation of the transactions contemplated hereby, up to a maximum of Five Hundred Thousand Dollars ($500,000), by wire transfer of same day funds within five (5) Business Days following the date on which Public Company submits to Merger Partner true and correct copies of reasonable documentation supporting such expenses.
(f) Any fee due under Section 8.3(b)(i) or 8.3(c)(i) shall be paid by wire transfer of same day funds concurrently with the termination of this Agreement (and shall be a condition to the effectiveness of such termination). Any fee due under Section 8.3(b)(ii) or 8.3(c)(ii) shall be paid by wire transfer of same day funds on the date of termination of this Agreement (and shall be a condition to the effectiveness of such termination). Any fee due under Section 8.3(b)(iii) or 8.3(c)(iii) shall be paid by wire transfer of same-day funds within two (2) Business Days after the date on which the transaction referenced in clause (B) of such Section 8.3(b)(iii) or 8.3(c)(iii), as applicable, is consummated. If one party fails to promptly pay to the other any fee due pursuant to this Section 8.3, the defaulting party shall pay the costs and expenses (including legal fees and expenses) in connection with any action, including the filing of any lawsuit or other legal action, taken to collect payment, together with interest on the amount of any unpaid fee at the publicly announced prime rate of Bank of America, N.A. plus five percent (5%) per annum, compounded quarterly, from the date such fee was required to be paid.
(g) The parties hereto acknowledge that the agreements contained in this Section 8.3 are an integral part of the transactions contemplated by this Agreement, and that, without these agreements, the parties hereto would not enter into this Agreement. Notwithstanding Section 8.2 or any other provision of this Agreement, payment of the termination fees described in, and under the circumstances provided for in, this Section 8.3 shall constitute the sole and exclusive remedy of Public Company or Merger Partner, as applicable in connection with any termination of this Agreement in the circumstances in which such fees became payable. In the event that Public Company or Merger Partner shall receive the payment of a termination fee under the circumstances provided for in this Section 8.3, the receipt of such fee shall be deemed to be liquidated damages for any and all losses or damages suffered or incurred by Public Company and any of its Affiliates or Merger Partner and any of its Affiliates, as applicable, or any other Person in connection with this Agreement (and the termination hereof), the transactions contemplated hereby (and the abandonment thereof) or any matter forming the basis for such termination, and none of the Public Company, Merger Sub any of their respective Affiliates or Merger Partner or any of its Affiliates, as applicable, or any other Person, shall be entitled to bring or maintain any other claim, action or proceeding against Public Company or Merger Partner, as applicable, or any of their respective Affiliates arising out of this Agreement, any of the transactions contemplated hereby or any matters forming the basis for such termination.
(h) The parties hereto acknowledge and agree that in no event shall Merger Partner be required to pay the Merger Partner Termination Fee or any expenses referred to in Section 8.3(e) on more than one occasion, nor shall Public Company be required to pay the Public Company Termination Fee or any expenses referred to in Section 8.3(d) on more than one occasion, in each case, whether or not such fee or expenses may be payable under more than one provision of this Agreement at the same or at different times and the occurrence of different events. For the avoidance of doubt, to the extent any Third Party Expenses are paid, such amounts shall be credited against any Merger Partner Termination Fee or Public Company Termination Fee, as applicable, which becomes payable thereafter.
ARTICLE IX
MISCELLANEOUS
9.1. Non-survival of Representations, Warranties and Agreements. None of the representations, warranties, covenants or agreements in this Agreement or in any instrument delivered pursuant to this Agreement shall survive the Effective Time, other than those covenants or agreements of the parties which by their terms apply, or are to be performed in whole or in part, after the Effective Time.
9.2. Notices. All notices and other communications hereunder shall be in writing and shall be deemed duly delivered (i) three (3) Business Days after being sent by registered or certified mail, return receipt requested, postage prepaid, or (ii) one (1) Business Day after being sent for next Business Day delivery, fees prepaid, via a reputable overnight courier service, in each case to the intended recipient as set forth below:
(a) if to Public Company or Merger Sub, to:
Pieris Pharmaceuticals, Inc.
225 Franklin Street, 26th Floor
Boston, MA 02110
Attention: Stephen Yoder, Tom Bures, Karam Hijji
with a copy (which shall not constitute notice) to:
Mintz, Levin, Cohn, Ferris, Glovsky and Popeo, P.C.
One Financial Center
Boston, MA 02111
Attention: William C. Hicks, Marc D. Mantell, Scott Dunberg
(b) if to Merger Partner, to:
Palvella Therapeutics, Inc.
125 Strafford Ave, Suite 360
Wayne, PA 19087
Attention: Chief Executive Officer
with a copy (which shall not constitute notice) to:
Troutman Pepper Hamilton Sanders, LLP
400 Berwyn Park
899 Cassatt Road
Berwyn, PA 19312
Attention: Christopher S. Miller
Any party to this Agreement may give any notice or other communication hereunder using any other means (including personal delivery, messenger service, telecopy, ordinary mail or electronic mail), but no such notice or other communication shall be deemed to have been duly given unless and until it actually is received by the party for whom it is intended. Any party to this Agreement may change the address to which notices and other communications hereunder are to be delivered by giving the other parties to this Agreement notice in the manner set forth in this Section 9.2.
9.3. Entire Agreement. This Agreement (including the Schedules, Annexes and Exhibits hereto and the documents and instruments referred to herein that are to be delivered at the Closing) constitutes the entire agreement among the parties to this Agreement and supersedes any prior understandings, agreements or representations by or among the parties hereto, or any of them, written or oral, with respect to the subject matter hereof and the parties hereto expressly disclaim reliance on any such prior understandings, agreements or representations to the extent not embodied in this Agreement. Notwithstanding the foregoing, the Confidentiality Agreement shall remain in effect in accordance with its terms.
9.4. Amendment. Subject to applicable Law, and Section 6.10(e) (D&O Indemnification), this Agreement may be amended with the approval of the Public Company Board and the Merger Partner Board, at any time prior to the Effective Time (whether before or after obtaining the Required Public Company Stockholder Approval or the Written Consents); provided, however, that after the Required Public Company Stockholder Approval or the Written Consents have been obtained, no amendment to this Agreement may be made without the further approval of the stockholders of Public Company or Merger Partner, as applicable, if such further approval is required by Law. This Agreement may not be amended except by an instrument in writing signed on behalf of each of Public Company and Merger Partner.
9.5. Extension; Waiver. Public Company may (a) extend the time for the performance of any of the obligations or other acts of Merger Partner set forth herein, (b) waive any inaccuracies in the representations and warranties of Merger Partner set forth herein or (c) waive compliance by Merger Partner with any of the agreements or conditions set forth herein. Merger Partner may (i) extend the time for the performance of any of the obligations or other acts of Public Company or any of its subsidiaries, set forth herein, (ii) waive any inaccuracies in the representations and warranties of Public Company or any of its subsidiaries set forth herein or (iii) waive compliance by Public Company or any of its subsidiaries with any of the agreements or conditions set forth herein. Any agreement on the part of any such party to any such extension or waiver shall be valid only if set forth in a written instrument signed on behalf of such party. Any waiver of any term or condition shall not be construed as a waiver of any subsequent breach or a subsequent waiver of the same term or condition, or a waiver of any other term or condition of this Agreement. The failure of any party to assert any of its rights hereunder shall not constitute a waiver of such rights.
9.6. Procedure for Termination, Amendment, Extension or Waiver. A termination of this Agreement pursuant to Section 8.1, an amendment, modification or supplement of this Agreement pursuant to Section 9.4 or an extension or waiver of this Agreement pursuant to Section 9.5 shall, in order to be effective, require action by the respective boards of directors of the applicable parties.
9.7. No Third Party Beneficiaries. This Agreement is not intended to, and shall not, confer upon any other Person any rights or remedies hereunder, except as set forth in or contemplated by the terms and provisions of Section 6.10.
9.8. Assignment. No party may assign any of its rights or delegate any of its performance obligations under this Agreement, in whole or in part, by operation of Law or otherwise without the prior written consent of the other parties, and any such assignment without such prior written consent shall be null and void. Subject to the preceding sentence, this Agreement shall be binding upon, inure to the benefit of, and be enforceable by, the parties hereto and their respective successors and permitted assigns. Any purported assignment of rights or delegation of performance obligations in violation of this Section 9.8 is void.
9.9. Severability. Any term or provision of this Agreement that is invalid or unenforceable in any situation in any jurisdiction shall not affect the validity or enforceability of the remaining terms and provisions hereof or the validity or enforceability of the offending term or provision in any other situation or in any other jurisdiction. If the final judgment of a court of competent jurisdiction declares that any term or provision hereof is invalid or unenforceable, the parties hereto agree that the court making such determination shall have the power to limit the term or provision, to delete specific words or phrases, or to replace any invalid or unenforceable term or provision with a term or provision that is valid and enforceable and that comes closest to expressing the intention of the invalid or unenforceable term or provision, and this Agreement shall be enforceable as so modified. In the event such court does not exercise the power granted to it in the prior sentence, the parties hereto agree to replace such invalid or unenforceable term or provision with a valid and enforceable term or provision that will achieve, to the extent possible, the economic, business and other purposes of such invalid or unenforceable term.
9.10. Counterparts and Signature. This Agreement and any signed agreement or instrument entered into in connection with this Agreement may be executed in two or more counterparts (including by facsimile or by an electronic scan delivered by electronic mail), each of which shall be deemed an original but all of which together shall be considered one and the same agreement and shall become effective when counterparts have been signed by each of the parties hereto and delivered to the other parties, it being understood that all parties need not sign the same counterpart. To the extent signed and delivered by means of a facsimile machine or telecopy, by email delivery of a “.pdf” or “.jpg” format data file or by any electronic signature complying with the U.S. federal ESIGN Act of 2000, this Agreement shall be treated in all manner and respects as an original agreement or instrument and shall be considered to have the same binding legal effect as if it were the original signed version thereof delivered in Person. No party hereto or to any such agreement or instrument shall raise the use of a facsimile machine or telecopy, email delivery of a “.pdf” or “.jpg” format data file or electronic signature complying with the U.S. federal ESIGN Act of 2000 to deliver a signature to this Agreement or any amendment hereto or the fact that any signature or agreement or instrument was transmitted or communicated through the use of a facsimile machine or telecopy, email delivery of a “.pdf” or “.jpg” format data file or by any electronic signature complying with the U.S. federal ESIGN Act of 2000 as a defense to the formation of a contract and each party hereto forever waives any such defense.
9.11. Interpretation. When reference is made in this Agreement to an Article or a Section, such reference shall be to an Article or Section of this Agreement, unless otherwise indicated. The table of contents, table of defined terms and headings contained in this Agreement are for convenience of reference only and shall not affect in any way the meaning or interpretation of this Agreement. The language used in this Agreement shall be deemed to be the language chosen by the parties hereto to express their mutual intent, and no rule of strict construction shall be applied against any party. Whenever the context may require, any pronouns used in this Agreement shall include the corresponding masculine, feminine or neuter forms, and the singular form of nouns and pronouns shall include the plural, and vice versa. Any reference to any federal, state, local or foreign statute or Law shall be deemed also to refer to all rules and regulations promulgated thereunder, unless the context requires otherwise. Whenever the words “include,” “includes” or “including” are used in this Agreement, they shall be deemed to be followed by the words “without limitation.” The word “or” is used in the inclusive sense of “and/or.” The terms “or,” “any” and “either” are not exclusive. When used herein, the phrase “to the extent” shall be deemed to be followed by the words “but only to the extent.” The word “extent” in the phrase “to the extent” means the degree to which a subject or other thing extends, and such phrase shall not mean simply “if”. Where this Agreement refers to information that was “made available”, that means that such information was either (i) provided directly to the Public Company or Merger Partner, as applicable, by the other party, with confirmation of receipt, (ii) included in the virtual data rooms established by Public Company and Merger Partner created for the purposes of providing information to the other party in connection with this Agreement at least one (1) Business Day prior to the execution and delivery of this Agreement or (iii) solely with respect to information made available by Public Company, filed with and publicly available on the SEC’s EDGAR prior to the date of this Agreement. When used in the Agreement, “Person” shall mean any natural person, corporation, exempted company, limited liability company, partnership, exempted limited partnership, association, joint venture, trust, or other entity or business association. No summary of this Agreement prepared by any party shall affect the meaning or interpretation of this Agreement. For the avoidance of doubt, the parties agree that the terms “material,” “materially” and “materiality” as used in this Agreement with an initial lower case “m” shall have their respective customary and ordinary meanings, without regard to the meanings ascribed to Merger Partner Material Adverse Effect or Public Company Material Adverse Effect, in each case as defined in this Agreement.
9.12. Governing Law. This Agreement and all matters, claims, counterclaims, or causes of action (whether in contract, tort, statute, or otherwise) arising out of or relating to this Agreement and the transactions contemplated hereby (including its interpretation, construction, performance and enforcement), or the actions of any party in the negotiation, administration, performance, or enforcement of this Agreement (collectively, “Relevant Matters”) shall be governed by and construed in accordance with the internal Laws of the State of Delaware without giving effect to any choice or conflict of Law provision or rule (whether of the State of Delaware or any other jurisdiction) that would cause the application of Laws of any jurisdictions other than those of the State of Delaware. For the avoidance of doubt, all matters relating to the internal affairs of Public Company (including the fiduciary duties of its directors and officers) shall be governed by the internal Laws of the State of Nevada without giving effect to any choice or conflict of Law provision or rule (whether of the State of Nevada or any other jurisdiction) that would cause the application of Laws of any jurisdictions other than those of the State of Nevada.
9.13. Remedies. Except as otherwise provided herein, any and all remedies herein expressly conferred upon a party will be deemed cumulative with and not exclusive of any other remedy conferred hereby, or by Law or equity upon such party, and the exercise by a party of any one remedy will not preclude the exercise of any other remedy. The parties hereto agree that irreparable damage would occur in the event that any of the provisions of this Agreement were not performed in accordance with their specific terms or were otherwise breached. It is accordingly agreed that the parties shall be entitled to an injunction or injunctions to prevent breaches of this Agreement and to enforce specifically the terms and provisions of this Agreement, this being in addition to any other remedy to which they are entitled at Law or in equity.
9.14. Submission to Jurisdiction. Each of the parties to this Agreement (a) consents to submit itself to the exclusive personal jurisdiction of the Court of Chancery of the State of Delaware, New Castle County, or, if that court does not have jurisdiction, a state or federal court sitting in Wilmington, Delaware in any action or proceeding arising out of or relating any Relevant Matter, (b) agrees that all claims in respect of such action or proceeding shall be heard and determined in any such court, (c) agrees that it shall not attempt to deny or defeat such personal jurisdiction by motion or other request for leave from any such court and (d) agrees not to bring any action or proceeding arising out of or relating to any Relevant Matter in any other court. Each of the parties hereto waives any defense of inconvenient forum to the maintenance of any action or proceeding so brought and waives any bond, surety or other security that might be required of any other party with respect thereto. Any party may make service on another party by sending or delivering a copy of the process to the party to be served at the address and in the manner provided for the giving of notices in Section 9.2. Nothing in this Section 9.14, however, shall affect the right of any party to serve legal process in any other manner permitted by Law.
9.15. WAIVER OF JURY TRIAL. EACH OF PUBLIC COMPANY, THE MERGER SUB AND MERGER PARTNER HEREBY IRREVOCABLY WAIVES ALL RIGHT TO TRIAL BY JURY IN ANY ACTION OR PROCEEDING ARISING OUT OF OR RELATING TO ANY RELEVANT MATTER.
9.16. Disclosure Schedule. Each of the Merger Partner Disclosure Schedule and the Public Company Disclosure Schedule shall be arranged in sections corresponding to the numbered sections contained in this Agreement, and the disclosure in any section shall qualify only (a) the corresponding section of this Agreement and (b) the other sections of this Agreement, to the extent that it is reasonably apparent from a reading of such disclosure that it also qualifies or applies to such other sections. The inclusion of any information in the Merger Partner Disclosure Schedule or the Public Company Disclosure Schedule, as applicable, shall not be deemed to be an admission or acknowledgment, in and of itself, that such information is required by the terms hereof to be disclosed, is material, has resulted in or would result in a Merger Partner Material Adverse Effect or a Public Company Material Adverse Effect, as applicable, or is outside the Ordinary Course of Business.
9.17. Certain Defined Terms. For purposes of this Agreement:
(a) “2024 Equity Plan means the 2024 equity incentive plan of Public Company in form and substance as determined by Merger Company (subject to Public Company’s approval, not to be unreasonably withheld, conditioned or delayed.
(b) “Excluded Contracts” means (i) Contracts solely concerning non-exclusive rights granted to Merger Partner or Public Company (as applicable) that are not material to the business of such Person, including any Contract solely for the license of “off-the-shelf” software that is available on standard commercial terms, (ii) Contracts the terms of which are solely focused on obligations relating to non-disclosure or confidentiality or assignments of Intellectual Property (to the extent in customary form and copies of which forms have been made available to Public Company or Merger Partner, as applicable), in each case entered into in the Ordinary Course of Business, (iii) statements of work, works orders, project annexes, purchase orders and associated terms and conditions to the extent the Contract accompanying such statements of work, works orders, project annexes, purchase orders, and associated terms and conditions has been made available to Public Company or Merger Partner, as applicable, (iv) agreements with clinical trial sites, (v) solely with respect to Public Company, Contracts related to Intellectual Property that has been abandoned as of the date hereof, including those identified on Section 9.17(b) of the of the Public Company Disclosure Schedule, and (vi) solely with respect to Public Company, Contracts entered into in connection with any Legacy Asset Disposition.
(c) “German R&D Tax Credit” means any tax refund or credit under the Research Allowance Act (called research allowance or Forschungszulage).
(d) “Good Clinical Practices” means applicable ethical and quality standards and rules for designing, conducting, monitoring, recording, reporting, auditing, and analyses of trials that involve the participation of human subjects, including without limitation 21 CFR Parts 11, 50, 54, 56, and 312, and 45 CFR 46.
(e) “Good Laboratory Practices” means applicable standards, rules and criteria, including 21 CFR Part 58, relating to a quality system and controls concerned with the organizational process, the conditions under which non-clinical health and environmental safety studies are planned, performed, monitored, recorded, reported and archived, and the integrity of data collected in such studies.
(f) “Good Manufacturing Practices” means applicable standards, quality management system regulations, and rules for ensuring that products are consistently produced and controlled according to quality standards, including without limitation requirements for methods, facilities, and controls in manufacturing, processing, packing, storing, labeling, and monitoring of the identity, strength, quality, and purity of drug products, including without limitation 21 CFR Parts 210, 211, 314, and 600, as applicable.
(g) “knowledge of Merger Partner” and similar expressions mean the actual knowledge of the individuals identified on Schedule K of the Merger Partner Disclosure Schedule for this purpose.
(h) “knowledge of Public Company” and similar expressions mean the actual knowledge of the individuals identified on Schedule K of the Public Company Disclosure Schedule for this purpose.
(i) “Liability” means, with respect to any Person, any and all liabilities, obligations, claims, and deficiencies of any kind (whether known or unknown, contingent, accrued, due or to become due, secured or unsecured, matured or otherwise), including accounts payable, all liabilities, obligations, claims, and deficiencies related to Indebtedness or guarantees, costs, expenses, royalties payable, and other reserves, termination payment obligations, and all other liabilities, obligations, claims, and deficiencies of such Person or any of its subsidiaries or Affiliates, in each case, regardless of whether or not such liabilities, obligations, claims, and deficiencies are required to be reflected on a balance sheet in accordance with GAAP.
(j) “Merger Partner Material Adverse Effect” means any change, effect, event, circumstance or development (an “Effect”) that, individually or in the aggregate with all other Effects that have occurred through the date of determination, has had, or is reasonably likely to have, a material adverse effect on the business, assets and liabilities, financial condition or results of operations of Merger Partner, taken as a whole; provided, however, that no Effect, to the extent resulting from or arising out of any of the following, shall be deemed to be a Merger Partner Material Adverse Effect or be taken into account for purposes of determining whether a Merger Partner Material Adverse Effect has occurred or is reasonably likely to occur: (i) changes after the date of this Agreement in prevailing economic or market conditions in the United States or any other jurisdiction (except to the extent those changes have a disproportionate effect on Merger Partner relative to the other participants in the industry or industries in which Merger Partner operates), (ii) changes or events after the date of this Agreement affecting the industry or industries in which Merger Partner operates generally (except to the extent those changes or events have a disproportionate effect on Merger Partner relative to the other participants in the industry or industries in which Merger Partner operates), (iii) changes after the date of this Agreement in generally accepted accounting principles or requirements or the interpretation thereof (except to the extent those changes have a disproportionate effect on Merger Partner relative to the other participants in the industry or industries in which Merger Partner operates), (iv) changes after the date of this Agreement in Laws, rules or regulations of general applicability or interpretations thereof by any Governmental Entity (except to the extent those changes have a disproportionate effect on Merger Partner relative to the other participants in the industry or industries in which Merger Partner operates), (v) any natural disaster, epidemic, pandemic or other disease outbreak (including the COVID-19 pandemic) or any outbreak of major hostilities or any act of terrorism (except to the extent those changes or events have a disproportionate effect on Merger Partner relative to the other participants in the industry or industries in which Merger Partner operates), (vi) the execution, delivery, announcement or performance of the obligations under this Agreement or the announcement, pendency or anticipated consummation of the transactions contemplated by this Agreement, (vii) any failure by Merger Partner to meet any internal guidance, budgets, plans or forecasts of its revenues, earnings or other financial performance or results of operations (but not, in the case of this clause (vii), the underlying cause of such changes or failures, unless such changes or failures would otherwise be excepted from this definition), or (viii) any equityholder or derivative litigation arising from or relating to this Agreement or the transactions contemplated by the Agreement.
(k) “Ordinary Course of Business” means, with respect to a Person, in the ordinary course of business consistent in all material respects with past practice of such Person; provided that, with respect to Public Company, during the period prior to the Closing, the Ordinary Course of Business of Public Company shall also be deemed to include any reasonable actions taken to effect the winding down of its prior research and development activities, but only if such actions do not and would not reasonably be expected to result in any Liability to Public Company following the Closing.
(l) “Permitted Liens” means (A) Liens of landlords, carriers, warehousemen, mechanics, vendors, materialmen or other Persons securing obligations arising in the Ordinary Course of Business that are not yet due and payable, (B) Liens incurred in the Ordinary Course of Business in connection with workers’ compensation, unemployment insurance and other types of social security, (C) Liens incurred to secure the performance of tenders, statutory obligations, surety and appeal bonds, bids, leases, government contracts, performance and return of money bonds and similar obligations in the Ordinary Course of Business, (D) Liens for Taxes (1) not yet due, or (2) being contested through appropriate proceedings and for which adequate reserves are reflected on the Merger Partner Balance Sheet, (E) Liens expressly set forth in Excluded Contracts, and (F) other Liens that are not material.
(m) “Permitted Settlement” means, with respect to any Person, a settlement by such Person of any pending legal proceeding that: (i) provides for the payment by such Person of money damages and no other relief of any nature; and (ii) includes an unconditional release and wavier of future claims by all plaintiffs in favor of such Person.
(n) “Pre-Closing Tax Period” shall mean any taxable period ending on or prior to the Closing Date.
(o) “Public Company Charter Amendments” means the amendments to the Public Company’s articles of incorporation to (i) increase the number of shares of authorized Public Company Common Stock from 3,750,000 to 200,000,000 and (ii) change the name of Public Company to “Palvella Therapeutics, Inc.”
(p) “Public Company Material Adverse Effect” means any Effect that, individually or in the aggregate with all other Effects that have occurred through the date of determination, has had, or is reasonably likely to have, a material adverse effect on the business, assets and liabilities, financial condition or results of operations of Public Company and its subsidiaries, taken as a whole; provided, however, that no Effect, to the extent resulting from or arising out of any of the following, shall be deemed to be a Public Company Material Adverse Effect or be taken into account for purposes of determining whether a Public Company Material Adverse Effect has occurred or is reasonably likely to occur: (i) changes after the date of this Agreement in prevailing economic or market conditions in the United States or any other jurisdiction (except to the extent those changes have a disproportionate effect on Public Company and its subsidiaries relative to the other participants in the industry or industries in which Public Company and its subsidiaries operate), (ii) changes or events after the date of this Agreement affecting the industry or industries in which Public Company and its subsidiaries operate generally (except to the extent those changes or events have a disproportionate effect on Public Company and its subsidiaries relative to the other participants in the industry or industries in which Public Company and its subsidiaries operate), (iii) changes after the date of this Agreement in generally accepted accounting principles or requirements or the interpretation thereof (except to the extent those changes have a disproportionate effect on Public Company and its subsidiaries relative to the other participants in the industry or industries in which Public Company and its subsidiaries operate), (iv) changes after the date of this Agreement in Laws, rules or regulations of general applicability or interpretations thereof by any Governmental Entity (except to the extent those changes have a disproportionate effect on Public Company and its subsidiaries relative to the other participants in the industry or industries in which Public Company and its subsidiaries operate), (v) any natural disaster, epidemic, pandemic or other disease outbreak (including the COVID-19 pandemic) or any outbreak of major hostilities or any act of terrorism (except to the extent those changes or events have a disproportionate effect on Public Company and its subsidiaries relative to the other participants in the industry or industries in which Public Company and its subsidiaries operate), (vi) a change in the public trading price of Public Company Common Stock or the implications hereof (it being understood that any Effect causing or giving rise to any such change shall be taken into account for purposes of determining whether a Public Company Material Adverse Effect has occurred or is reasonably likely to occur), (vii) the execution, delivery, announcement or performance of the obligations under this Agreement or the announcement, pendency or anticipated consummation of the transactions contemplated by this Agreement, (viii) any failure by Public Company or any of its subsidiaries to meet any public estimates or expectations of such Person’s revenue, earnings or other financial performance or results of operations for any period, (ix) any failure by Public Company or any of its subsidiaries to meet any internal guidance, budgets, plans or forecasts of such Person’s revenues, earnings or other financial performance or results of operations, (x) any changes in or affecting research and development, preclinical studies, clinical trials or other drug development activities (including the failure to obtain positive results from clinical trials, the occurrence of adverse events or serious adverse events in any clinical trial, development activities or favorable responses from any applicable Governmental Entity) conducted by or on behalf of Public Company or any of its subsidiaries or licensees in respect of such Person’s products or product candidates, (xi) any rejection or non-acceptance by a Governmental Entity of a registration or filing by Public Company or any of its subsidiaries relating to any IP Rights of Public Company or any of its subsidiaries, (xii) regulatory approval of, or regulatory action or announcement with respect to, any product, or product candidates, of a third party that are similar to, or expected to compete against, any of Public Company’s or any of its subsidiaries’ product candidates, including product candidates licensed out to the third parties, (xiii) any stockholder or derivative litigation arising from or relating to this Agreement or the transactions contemplated by the Agreement, (xiv) any Legacy Asset Disposition, or (xv) any of the matters set forth on Section 9.17(m) of the Public Company Disclosure Schedule (but in the case of clauses (vi), (vii), (viii), or (ix), the underlying cause of such changes or failures shall be taken into account for purposes of determining whether a Public Company Material Adverse Effect has occurred or is reasonably likely to occur, unless such changes or failures would otherwise be excepted from this definition).
[Remainder of Page Intentionally Left Blank]
IN WITNESS WHEREOF, the parties have executed this Agreement as of the date first written above.
|
|
PIERIS PHARMACEUTICALS, INC. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
By: |
/s/ Stephen S. Yoder |
|
|
|
Name: Stephen S. Yoder Title: Chief Executive Officer |
|
|
|
|
|
|
|
| POLO MERGER SUB, INC. | |||
| By: | /s/ Thomas Bures | ||
|
Name: Thomas Bures Title: President |
|||
| PALVELLA THERAPEUTICS, INC. | |||
| By: | /s/ Wesley H. Kaupinen | ||
|
Name: Wesley H. Kaupinen Title: Chief Executive Officer |
|||
ANNEX B
Execution Version
This SECURITIES PURCHASE AGREEMENT (this “Agreement”) is dated as of July 23, 2024, by and among Pieris Pharmaceuticals, Inc., a Nevada corporation (the “Company”), and each purchaser identified on Exhibit A hereto and each Convertible Note Purchaser (as defined below) who becomes a party to this Agreement in accordance with Section 6.1 below (each, including its successors and assigns, a “Purchaser” and collectively, the “Purchasers”).
RECITALS
A. Concurrently with the execution of this Agreement, the Company has entered into that certain Agreement and Plan of Merger (the “Merger Agreement”) dated July 23, 2024, by and among the Company, Polo Merger Sub, Inc., a Delaware corporation and a wholly-owned subsidiary of the Company (the “Merger Sub”), and Palvella Therapeutics, Inc. a Delaware corporation (“Merger Partner”), pursuant to which Merger Sub will be merged with and into Merger Partner (Merger Partner as the surviving corporation following the Merger (as defined in the Merger Agreement) is sometimes referred to herein as the “Surviving Corporation”) and the separate existence of Merger Sub shall cease;.
B. The Company and each Purchaser are executing and delivering this Agreement in reliance upon the exemption from securities registration afforded by Section 4(a)(2) of the Securities Act of 1933, as amended, and the rules and regulations thereunder (the “Securities Act”).
C. Each Purchaser, severally and not jointly, wishes to purchase, and the Company wishes to issue and sell, upon the terms and conditions stated in this Agreement, for an aggregate purchase amount of $60,000,000 (plus the Outstanding Amounts (as defined below) of the Convertible Notes (as defined below) in the principal amount not to exceed $20,000,000 in the aggregate) at a price per share equal to (i) $13.7299 multiplied by (ii) (x) 0.315478 divided by (y) the Exchange Ratio (as defined in the Merger Agreement) (as may be adjusted, the “Per Share Purchase Price”) (subject to adjustment for any stock split, reverse stock split or similar recapitalization transaction effected after the date hereof and prior to Closing) shares (the “Shares”) of Common Stock of the Company, par value $0.001 per share (“Common Stock”) and/or pre-funded warrants, in substantially the form attached hereto as Exhibit B (each a “Pre-Funded Warrant” and collectively, the “Pre-Funded Warrants”), to purchase shares (the “Warrant Shares”) of Common Stock, as more fully described in this Agreement (the shares of Common Stock, Pre-Funded Warrants and Warrant Shares are collectively referred to herein as the “Securities”).
D. Merger Partner has engaged TD Securities (USA) LLC and Cantor Fitzgerald & Co. as its exclusive placement agents (the “Placement Agents”) for the offering of the Securities on a “best efforts” basis.
E. Prior to the Closing (as defined below), the parties hereto shall execute and deliver a Registration Rights Agreement, substantially in the form attached hereto as Exhibit C (the “Registration Rights Agreement”), pursuant to which, among other things, the Company will agree to provide certain registration rights with respect to the Shares and Warrant Shares under the Securities Act and the rules and regulations promulgated thereunder and applicable state securities laws.
NOW, THEREFORE, in consideration of the mutual covenants contained in this Agreement, and for other good and valuable consideration, the receipt and adequacy of which are hereby acknowledged, the Company and each Purchaser, severally and not jointly, hereby agree as follows:
ARTICLE I
DEFINITIONS
1.1. Definitions. In addition to the terms defined elsewhere in this Agreement, for all purposes of this Agreement, the following terms shall have the meanings indicated in this Section 1.1:
“Acquiring Person” has the meaning set forth in Section 4.5.
“Action” means any Proceeding pending or, to the Company’s Knowledge, threatened against the Company, its Subsidiaries or any of their respective properties, or any officer, director or employee of the Company or any of its Subsidiaries acting in his or her capacity as an officer, director or employee, before or by any federal, state, county, local or foreign court, arbitrator, governmental or administrative agency, regulatory authority, stock market, stock exchange or trading facility.
“Affiliate” when used with respect to any party shall mean any Person who is an “affiliate” of that party within the meaning of Rule 405 promulgated under the Securities Act.
“Agreement” has the meaning set forth in the Preamble.
“Attribution Parties” means, collectively, the following Persons: (i) any direct or indirect Affiliates of the Purchaser, (ii) any Person acting or who could be deemed to be acting as a group together with the Purchaser or any of the foregoing and (iii) any other Persons whose beneficial ownership of Common Stock would or could be aggregated with the Purchaser’s and the other Attribution Parties for purposes of Section 13(d) or Section 16 of the Exchange Act. For clarity, the purpose of the foregoing is to subject collectively the Purchaser and all other Attribution Parties to the Beneficial Ownership Limitation indicated on each such Purchaser’s signature page attached hereto.
“Audited Balance Sheet” means the consolidated balance sheets of the Company and its Subsidiaries as of December 31, 2023 included in the Company’s Annual Report on Form 10-K filed with the Commission on March 29, 2024.
“Board of Directors” means the board of directors of the Company.
“Business Day” means any day other than a Saturday, Sunday or other day on which commercial banking institutions in New York, New York, Las Vegas, Nevada or Wilmington, Delaware are required or permitted by Law to be closed or other day on which the Delaware Secretary of State is closed.
“Closing” has the meaning set forth in Section 2.2(a).
“Closing Date” has the meaning set forth in Section 2.2(a).
“Code” means the Internal Revenue Code of 1986, as amended.
“Commission” means the United States Securities and Exchange Commission.
“Common Stock” has the meaning set forth in the Recitals.
“Company” has the meaning set forth in the Preamble.
“Company Authorized Stock Increase” means, collectively, the approval of the Charter Amendment Proposal (as defined in the Merger Agreement) and the effectiveness of the amendment to the Articles of Incorporation (as defined below) to effectuate the increase in the number of authorized shares of Common Stock approved in such proposal.
“Company Counsel” means Mintz, Levin, Cohn, Ferris, Glovsky and Popeo, P.C. with offices at One Financial Center, Boston, MA 02111.
“Company Deliverables” has the meaning set forth in Section 2.3(a).
“Company’s Knowledge” means with respect to any statement made to the Company’s Knowledge, that the statement is based upon the actual knowledge, or knowledge that would have been acquired after reasonable inquiry, of the executive officers or directors of the Company having responsibility for the matter or matters that are the subject of the statement. With respect to any matters relating to Intellectual Property, such awareness or reasonable expectation to have knowledge does not require any such individual to conduct or have conducted or obtain or have obtained any freedom to operate opinions of counsel or any Intellectual Property rights clearance searches.
“Contract” means, with respect to any Person, any written, oral or other agreement, contract, subcontract, lease (whether for real or personal property), mortgage, understanding, arrangement, instrument, note, option, warranty, license, sublicense, insurance policy, benefit plan or commitment or undertaking of any nature to which such Person is a party or by which such person or any of its assets are bound under applicable Law.
“Control” (including the terms “controlling”, “controlled by” or “under common control with”) means the possession, direct or indirect, of the power to direct or cause the direction of the management and policies of a Person, whether through the ownership of voting securities, by Contract or otherwise.
“Effective Date” means the date on which the initial registration statement required by Section 2(a) of the Registration Rights Agreement is first declared effective by the Commission.
“Employee Plan” means any “employee benefit plan” as defined in Section 3(3) of ERISA and any other pension, retirement, deferred compensation, excess benefit, profit-sharing, bonus, incentive, equity or equity-based, phantom equity, employment, consulting, severance, change-of-control, retention, health, life, disability, group insurance, paid time off, holiday, welfare and fringe benefit plan, program, agreement, Contract, or arrangement (whether written or unwritten, qualified or nonqualified, funded or unfunded, subject or not subject to ERISA and including any that have been frozen) that the Company or any of its Subsidiaries (i) sponsors, maintains, administers, or contributes to, (ii) provides benefits under or through, (iii) has any obligation to contribute to or provide benefits under or through, (iv) with respect to which have any liability, or (v) utilizes to provide benefits to or otherwise cover any current or former employee, officer, director or other service provider of the Company or any of its Subsidiaries (or their spouses, dependents, or beneficiaries).
“Encumbrance” means any lien, pledge, hypothecation, charge, mortgage, security interest, lease, exclusive license, option, easement, reservation, servitude, adverse title, claim, infringement, interference, option, right of first refusal, preemptive right, community property interest or restriction or encumbrance of any nature (including any restriction on the voting of any security, any restriction on the transfer of any security or other asset, any restriction on the receipt of any income derived from any asset, any restriction on the use of any asset and any restriction on the possession, exercise or transfer of any other attribute of ownership of any asset).
“Environmental Laws” has the meaning set forth in Section 3.1(bb).
“ERISA” means the Employee Retirement Income Security Act of 1974, as amended.
“Exchange Act” means the Securities Exchange Act of 1934, as amended, or any successor statute, and the rules and regulations promulgated thereunder.
“GAAP” means generally accepted accounting principles and practices in effect from time to time within the United States applied consistently throughout the period involved.
“Governmental Authority” means any: (a) nation, state, commonwealth, province, territory, county, municipality, district or other jurisdiction of any nature, (b) federal, state, local, municipal, foreign, supra-national or other government, (c) governmental or quasi-governmental authority of any nature (including any governmental division, department, agency, commission, bureau, instrumentality, official, ministry, fund, foundation, center, organization, unit, body or entity and any court or other tribunal, and any taxing authority) or (d) self-regulatory organization (including Nasdaq).
“Hazardous Materials” means any pollutant, chemical, substance and any toxic, infectious, carcinogenic, reactive, corrosive, ignitable or flammable chemical, or chemical compound, or hazardous substance, material or waste, whether solid, liquid or gas, that is subject to regulation, control or remediation under any Environmental Law, including without limitation, crude oil or any fraction thereof, and petroleum products or by-products.
“Irrevocable Transfer Agent Instructions” means, with respect to the Company, the Irrevocable Transfer Agent Instructions, in substantially the form of Exhibit D, executed by the Company and delivered to the Transfer Agent.
“Law” means each applicable transnational, domestic or foreign federal, state or local laws (statutory, common or otherwise), order, judgment, rule, code, statute, regulation, requirement, variance, decree, writ, injunction, award, ruling, Permit or ordinance of any Governmental Authority, including any applicable stock exchange rule or requirement.
“Material Adverse Effect” shall mean any “Public Company Material Adverse Effect” as such term is defined in the Merger Agreement.
“Material Contract” means any Contract to which the Company or any Subsidiary is a party or by which either is bound that is material to the business of the Company, including those that have been filed as an exhibit to the SEC Reports pursuant to Item 601(b)(10) of Regulation S-K.
“Nasdaq” means The Nasdaq Stock Market.
“Nevada Counsel” means Brownstein Hyatt Farber Schreck, LLP, with offices at 100 N. City Pkwy, Suite 1600, Las Vegas, NV 89106.
“New York Courts” means the state and federal courts sitting in the City of New York, Borough of Manhattan.
“Outstanding Amount” has the meaning set forth in Section 2.1.
“Per Share Purchase Price” has the meaning set forth in the Recitals.
“Permit” means all required permits, licenses, registrations, authorizations, certificates, orders, approvals, franchises, variances and other similar rights issued by or obtained from any Governmental Authority.
“Permitted Liens” means: (a) Liens of landlords, carriers, warehousemen, mechanics, vendors, materialmen or other Persons securing obligations arising in the ordinary course of business that are not yet due and payable, (b) Liens incurred in the ordinary course of business in connection with workers’ compensation, unemployment insurance and other types of social security, (c) Liens incurred to secure the performance of tenders, statutory obligations, surety and appeal bonds, bids, leases, government contracts, performance and return of money bonds and similar obligations in the ordinary course of business, (d) Liens for Taxes (i) not yet due, or (ii) being contested through appropriate proceedings and for which adequate reserves are reflected on the Audited Balance Sheet, (e) Liens expressly set forth in any Contract that is not a Material Contract, and (f) other Liens that are not material.
“Person” means an individual or a corporation, partnership, trust, incorporated or unincorporated association, joint venture, limited liability company, joint stock company, government (or an agency or subdivision thereof) or other entity of any kind.
“Placement Agents” has the meaning set forth in the Recitals.
“Pre-Funded Warrants” has the meaning set forth in the Recitals.
“Press Release” has the meaning set forth in Section 4.4.
“Principal Trading Market” means the Trading Market on which the Common Stock is primarily listed on and quoted for trading, which, as of the date of this Agreement and the Closing Date, shall be the Nasdaq Capital Market.
“Proceeding” means an action, claim, suit, investigation or proceeding (including, without limitation, an investigation or partial proceeding, such as a deposition), whether commenced or threatened.
“Purchaser” or “Purchasers” has the meaning set forth in the Preamble.
“Purchaser Deliverables” has the meaning set forth in Section 2.3(b).
“Registrable Securities” has the meaning set forth in the Registration Rights Agreement.
“Registration Rights Agreement” has the meaning set forth in the Recitals.
“Rule 144” means Rule 144 promulgated by the Commission pursuant to the Securities Act, as such Rule may be amended from time to time, or any similar rule or regulation hereafter adopted by the Commission having substantially the same effect as such Rule.
“SEC Reports” has the meaning set forth in Section 3.1(g).
“Secretary’s Certificate” has the meaning set forth in paragraph 2.3(a)(vi).
“Securities” has the meaning set forth in the Recitals.
“Securities Act” has the meaning set forth in the Recitals.
“Shares” has the meaning set forth in the Recitals.
“Short Sales” include, without limitation, (i) all “short sales” as defined in Rule 200 promulgated under Regulation SHO under the Exchange Act, whether or not against the box, and all types of direct and indirect stock pledges, forward sale contracts, options, puts, calls, short sales, swaps, “put equivalent positions” (as defined in Rule 16a-1(h) under the Exchange Act) and similar arrangements (including on a total return basis), and (ii) sales and other transactions through non-U.S. broker dealers or non-U.S. regulated brokers (but shall not be deemed to include the location and/or reservation of borrowable shares of Common Stock).
“Subscription Amount” means, with respect to each Purchaser, the aggregate amount to be paid for the Shares and/or Prefunded Warrants purchased hereunder by such Purchaser set forth in the “Aggregate Cash Purchase Price” column opposite such Purchaser’s name in the table set forth on Exhibit A, in United States dollars and in immediately available funds.
“Subsidiary” means any material subsidiary of the Company, which for the avoidance of doubt shall include Merger Partner as of the Closing.
“Taxes” shall mean any taxes, charges, fees, duties, contributions, levies or other similar assessments or liabilities in the nature of a tax, including, without limitation, income, gross receipts, corporation, ad valorem, premium, value-added, net worth, capital stock, capital gains, documentary, recapture, alternative or add-on minimum, disability, estimated, registration, recording, excise, real property, personal property, sales, use, license, lease, service, service use, transfer, withholding, employment, unemployment, insurance, social security, national insurance, business license, business organization, environmental, workers compensation, payroll, profits, severance, stamp, occupation, windfall profits, escheat or unclaimed property, customs duties, franchise and other taxes of any kind imposed by the United States of America or any state, local or non-U.S. government, or any agency or political subdivision thereof, and any interest, fines, penalties, assessments or additions to tax imposed with respect to such items.
“Trading Day” means a day on which the Principal Trading Market is open for business.
“Trading Market” means any of the following markets or exchanges on which the Common Stock is listed or quoted for trading on the date in question: the NYSE American, the Nasdaq Capital Market, the Nasdaq Global Market, the Nasdaq Global Select Market, or the New York Stock Exchange (or any successors to any of the foregoing).
“Transaction Documents” means this Agreement, the schedules and exhibits attached hereto, the Pre-Funded Warrant, Registration Rights Agreement, the Irrevocable Transfer Agent Instructions and any other documents or agreements explicitly contemplated hereunder.
“Transfer Agent” means Computershare Trust Company, N.A., the current transfer agent of the Company, or any successor transfer agent for the Company.
“Warrant Shares” has the meaning set forth in the Recitals.
ARTICLE II
PURCHASE AND SALE
2.1. Purchase and Sale. On the Closing Date, upon the terms and subject to the conditions set forth herein, the Company will issue and sell to the Purchasers, and the Purchasers will purchase, severally and not jointly, (i) a number of Shares equal to (x) in the case of Shares being acquired for cash, the amount of the cash payment set forth opposite the names of each such Purchaser under the heading “Aggregate Cash Purchase Price” on Exhibit A divided by the Per Share Purchase Price and/or (y) in the case of Shares being acquired in exchange for and in consideration of the termination and cancellation of the Convertible Notes, the principal amount of such Convertible Notes set forth opposite the names of each such Purchaser under the heading “Convertible Note Principal Amount” on Exhibit A plus accrued interest thereunder (such amounts, the “Outstanding Amount”) divided by the Per Share Purchase Price, in each case, up to the number of Shares such that each such Purchaser (together with the other Attribution Parties) would not beneficially own a number of shares of Common Stock in excess of the Beneficial Ownership Limitation as indicated on each such Purchaser’s signature page attached hereto, if applicable, and (ii) the number of Pre-Funded Warrants equal to the balance of the Aggregate Cash Purchase Price and/or the Outstanding Amount in excess of the consideration paid for the Shares issued pursuant to clause (i) above divided by the Per Share Purchase Price minus $0.001. The aggregate amount of cash and/or Outstanding Amount of Convertible Notes exchanged by each Purchaser for Shares and/or Pre-Funded Warrants under this Agreement shall constitute the total consideration (as such term is used in Nevada Revised Statutes 78.211) due to the Company in respect of the issuance by the Company of such Shares and/or Pre-Funded Warrants.
2.2. Closing.
(a) Closing. Upon the satisfaction or waiver of the conditions set forth in Article V, the closing of the purchase and sale of the Shares and/or Pre-Funded Warrants (the “Closing”) shall take place remotely via exchange of executed documents and funds immediately following the Effective Time (as defined in the Merger Agreement) on the date on which the closing of the Merger occurs (the “Closing Date”).
(b) Payment. On or before the Closing Date, each Purchaser shall deliver to the Company (i) the Subscription Amount in full by wire transfer of immediately available funds to an account designated in writing by the Company, (ii) such Purchaser’s Convertible Note(s) (as defined below) in accordance with Section 2.2(c) below, or (iii) by any combination of such methods. At the Closing, the Company shall deliver to such Purchaser against payment therefor (x) a book-entry statement from the Transfer Agent evidencing the number of shares of Common Stock calculated in accordance with Section 2.1, registered in the name of such Purchaser (or its nominee in accordance with its delivery instructions), free and clear of any liens or restrictions (other than those arising under state and federal securities laws) and bearing the legend set forth in Section 4.1(b) and/or (y) electronic copies of the Pre-Funded Warrants, if applicable, executed by the Company and registered in the name of such Purchaser, pursuant to which such Purchaser shall have the right to acquire such number of shares of Common Stock calculated in accordance with Section 2.1, on the terms set forth therein, with the original Pre-Funded Warrants, delivered within five (5) Business Days after the Closing Date. If a Purchaser has delivered the Subscription Amount by wire transfer pursuant to clause (i) of this Section 2.2(b) prior to the Closing Date, and the Closing does not occur for any reason on or prior to the fifth (5th) Business Day following the expected Closing Date, the Company shall promptly (but not later than one (1) Business Day thereafter) return the Subscription Amount to such Purchasers by wire transfer of United States dollars in immediately available funds to the account specified by such Purchaser, and any book entries for the Shares shall be deemed cancelled; provided that, unless this Agreement has been terminated pursuant to Section 6.19, such return of funds shall not terminate this Agreement or relieve the Purchasers of their respective obligations to purchase the Shares and/or Pre-Funded Warrants at the Closing. Notwithstanding anything in this Agreement to the contrary and as may be agreed to among the Company and one or more Purchasers, a Purchaser that is a mutual fund and subject to regulations related to the timing of funding and the issuance of securities thereunder or a Purchaser that has internal policies and/or procedures relating to the timing of funding and issuance of securities thereafter shall not be required to wire its respective portion of the Subscription Amount as set forth on Exhibit A until it confirms receipt of a book-entry statement from the Transfer Agent evidencing the issuance of the Shares and/or Pre-Funded Warrants to such Purchaser on and as of the Closing Date.
(c) Conversion and Cancellation of Merger Partner Convertible Notes. By executing and delivering this Agreement, each Purchaser holding one or more convertible promissory notes issued by the Merger Partner (each, a “Convertible Note” and, collectively, the “Convertible Notes”), whether such Convertible Note is outstanding as of the date hereof or issued by the Merger Partner prior to Closing, hereby irrevocably acknowledges and agrees that:
(i) Such Purchaser is the sole owner of all right, title and interest in and to the Convertible Note(s) corresponding to the principal amounts shown opposite such Purchaser’s name on Exhibit A;
(ii) At the Closing, all of such Purchaser’s Convertible Note(s) will automatically and without any action on the part of such Purchaser be exchanged for that number of Shares and/or Pre-Funded Warrants equal to the Outstanding Amount divided by the Per Share Purchase Price, rounded down to the nearest whole share, regardless of whether any such Convertible Notes is actually delivered in original or other form; and
(iii) As to such Purchaser, upon the issuance of the Shares and/or Prefunded Warrants to such Purchaser pursuant to Section 2.2(c)(ii), such Purchaser’s Convertible Note(s) shall be automatically terminated and cancelled and shall have no further force or effect, and all obligations (including outstanding principal, interest and other amounts) under such Purchaser’s Convertible Note(s), shall be deemed fully paid and shall be automatically, irrevocably and unconditionally satisfied, released and discharged in full.
(iv) No fractional shares of the Shares a shall be created or issuable to the Purchasers upon the issuance of Shares upon the exchange, termination and cancellation of the Convertible Note(s) pursuant to this Section 2.2.
2.3. Closing Deliverables.
(a) On or prior to the Closing, the Company shall issue, deliver or cause to be delivered to each Purchaser the following (the “Company Deliverables”):
(i) evidence of the issuance of the Shares in the names of the Purchasers by book-entry statement from the Transfer Agent (in the name of such Purchaser as set forth on the Stock Issuance Questionnaire included as Exhibit E hereto);
(ii) if applicable, for each Purchaser of Pre-Funded Warrants pursuant to Section 2.1, electronic copies of the Pre-Funded Warrants, if applicable, executed by the Company and registered in the name of such Purchaser, pursuant to which such Purchaser shall have the right to acquire such number of Warrant Shares calculated in accordance with Section 2.1, on the terms set forth therein, with the original Pre-Funded Warrants, delivered within five (5) Business Days after the Closing Date;
(iii) a legal opinion of Company Counsel and/or Nevada Counsel, dated as of the Closing Date and in form and substance reasonably satisfactory to the Placement Agents, executed by such counsel and addressed to the Purchasers and the Placement Agents;
(iv) the Registration Rights Agreement, duly executed by the Company;
(v) duly executed Irrevocable Transfer Agent Instructions instructing the Transfer Agent to deliver, on an expedited basis, of the issuance of the Shares to be issued to each Purchaser, registered in the name of such Purchaser (or its nominee, as directed by the Purchaser);
(vi) a copy of the Notification Form: Listing of Additional Shares for the listing of the Shares and Warrant Shares filed by the Company with Nasdaq;
(vii) a certificate of the Secretary of the Company (the “Secretary’s Certificate”), dated as of the Closing Date, (A) certifying the resolutions adopted by the Board of Directors or a duly authorized committee thereof approving the transactions contemplated by this Agreement, the other Transaction Documents and the issuance of the Securities, (B) certifying the current versions of the Articles of Incorporation and Bylaws (as each term is defined below) and (C) certifying as to the signatures and authority of persons signing the Transaction Documents and related documents on behalf of the Company, in substantially the form attached hereto as Exhibit F;
(viii) the Compliance Certificate referred to in Section 5.1(i).
(ix) a certificate evidencing the good standing of the Company issued by the Secretary of State of the State of Nevada, as of a date within three (3) Business Days of the Closing Date; and
(x) a certificate evidencing the Company’s qualification as a foreign corporation and good standing issued by the Secretary of State (or comparable office) of each jurisdiction in which the Company is qualified to do business as a foreign corporation, as of a date within three (3) Business Days of the Closing Date.
(b) On or prior to the Closing, each Purchaser shall deliver or cause to be delivered to the Company the following (the “Purchaser Deliverables”):
(i) this Agreement, duly executed by such Purchaser;
(ii) its Subscription Amount in accordance with Section 2.2(b), if applicable;
(iii) its Convertible Note(s) in accordance with Section 2.2(c), if applicable;
(iv) the Registration Rights Agreement, duly executed by such Purchaser; and
(v) a fully completed Stock Issuance Questionnaire in the form attached hereto as Exhibit D.
ARTICLE III
REPRESENTATIONS AND WARRANTIES
3.1. Representations and Warranties of the Company. Except as previously disclosed in the SEC Reports, the Company hereby represents and warrants to each of the Purchasers and to the Placement Agents as follows:
(a) Due Organization; Subsidiaries. Each of the Company and its Subsidiaries is a corporation or limited liability company duly incorporated or formed, validly existing and in good standing under the Laws of the jurisdiction of its incorporation or organization and has all necessary corporate power and authority: (i) to conduct its business in the manner in which its business is currently being conducted and as proposed to be conducted as described in the SEC Reports, (ii) to own or lease and use its property and assets in the manner in which its property and assets are currently owned or leased and used and (iii) to perform its obligations under all Contracts by which it is bound. All of the Subsidiaries are wholly owned by the Company. Each of the Company and the Subsidiaries is licensed and qualified to do business, and is in good standing (to the extent applicable in such jurisdiction), under the Laws of all jurisdictions where the nature of its business or the manner in which its business is currently being conducted requires such licensing or qualification other than in jurisdictions where the failure to be so qualified individually or in the aggregate would not have or reasonably be expected to have a Material Adverse Effect.
(b) Authorization; Enforcement; Validity. The Company has the requisite corporate power and authority to enter into the Transaction Documents, to perform its obligations hereunder and thereunder and, upon the Company Authorized Stock Increase, to consummate the transactions contemplated hereby or thereby. All corporate action on the part of the Company, its directors and stockholders necessary for the authorization, execution, sale, issuance and delivery of the Securities (including the Company Authorized Stock Increase) has been or will be taken prior to the Closing. Each of the Transaction Documents have been (or upon delivery will have been) duly executed and delivered by the Company and is, or when delivered in accordance with the terms hereof or thereof, will constitute the legal, valid and binding obligation of the Company enforceable against the Company in accordance with its respective terms, except (i) as such enforceability may be limited by applicable bankruptcy, examinership, insolvency, reorganization, moratorium, liquidation or similar laws relating to, or affecting generally the enforcement of, creditors’ rights and remedies or by other equitable principles of general application, (ii) as limited by Laws relating to the availability of specific performance, injunctive relief or other equitable remedies and (iii) insofar as indemnification and contribution provisions may be limited by applicable law.
(c) No Conflicts. The execution, delivery and performance by the Company of the Transaction Documents and the issuance, sale and delivery of the securities to be sold by the Company under the Transaction Documents (including the issuance of the Securities), the performance by the Company of its obligations under the Transaction Documents and the consummation of the transactions contemplated hereby or thereby (including without limitation, the issuance of the Securities) do not and will not conflict with, result in the breach or violation of, or constitute (with or without the giving of notice or the passage of time or both) a violation of, or default under, (i) any bond, debenture, note or other evidence of indebtedness, or under any lease, license, franchise, permit, indenture, mortgage, deed of trust, loan agreement, joint venture or other Contract, agreement or instrument to which the Company or any of its Subsidiaries is a party or by which it or its properties may be bound or affected, (ii) the Company’s articles of incorporation (as amended, the “Articles of Incorporation”) (upon the Company Authorized Stock Increase), the Company’s bylaws (as amended, the “Bylaws”), or the equivalent document with respect to any of the Company’s Subsidiaries, as amended and as in effect on the date hereof, or (iii) any statute or Law (upon the Company Authorized Stock Increase), judgment, decree, rule, regulation, ordinance or order of any Governmental Authority applicable to the Company, any of its Subsidiaries or their respective properties, except in the case of clauses (i) and (iii) for such conflicts, breaches, violations or defaults that would not reasonably be expected to have, individually or in the aggregate, a Material Adverse Effect.
(d) Filings, Consents and Approvals. Except for any Current Report on Form 8-K to be filed by the Company in connection with the transaction contemplated hereby, any required filing with Nasdaq, the Public Stockholder Approvals (as defined in the Merger Agreement), the Public Company Charter Amendment relating to the Company Authorized Stock Increase, and the registration statement required to be filed by the Registration Rights Agreement, neither the Company nor any of its Subsidiaries is required to give any notice to, or make any filings with, or obtain any authorization, consent, or approval of any Governmental Authority in order to consummate the transactions contemplated by the Transaction Documents. Assuming the accuracy of the representations of the Purchasers in Section 3.2, no consent, approval, authorization or other order of, or registration, qualification or filing (other than the Public Company Charter Amendment relating to the Company Authorized Stock Increase) with, any Governmental Authority is required for the execution and delivery of the Transaction Documents, the valid issuance, sale and delivery of the Securities to be sold pursuant to the Transaction Documents other than such as have been or will be made or obtained, or for any securities filings required to be made under federal or state securities laws applicable to the offering of the Securities (other than filings that have been made, or will be made, pursuant to the rules and regulations of Nasdaq). The Company and its Subsidiaries are unaware of any facts or circumstances that might prevent the Company from obtaining or effecting any of the registration, application or filings pursuant to this Section 3.1(d).
(e) Issuance of the Securities. The issuance of the Shares has been duly authorized, and the Shares, when issued and paid for in accordance with the terms of the Transaction Documents (including the Company Authorized Stock Increase), will be duly and validly issued, fully paid and nonassessable and free and clear of any Encumbrances, preemptive rights or restrictions (other than any restrictions on transfer generally imposed under applicable securities laws). The issuance of the Pre-Funded Warrants has been duly authorized, and the Pre-Funded Warrants, when issued and paid for in accordance with the terms of the Transaction Documents, will constitute the legal, valid and binding obligation of the Company enforceable against the Company in accordance with its respective terms, except (i) as such enforceability may be limited by applicable bankruptcy, examinership, insolvency, reorganization, moratorium, liquidation or similar laws relating to, or affecting generally the enforcement of, creditors’ rights and remedies or by other equitable principles of general application, (ii) as limited by Laws relating to the availability of specific performance, injunctive relief or other equitable remedies and (iii) insofar as indemnification and contribution provisions may be limited by applicable law. The issuance of the Warrant Shares has been duly authorized, and the Warrant Shares, when issued and paid for in accordance with the terms of the Pre-Funded Warrants (including the Company Authorized Stock Increase), will be duly and validly issued, fully paid and nonassessable and free and clear of any Encumbrances, preemptive rights or restrictions (other than any restrictions on transfer generally imposed under applicable securities laws).
(f) Capitalization. As of immediately prior to the Closing, the Company will have an authorized capitalization as set forth in the SEC Reports. All outstanding shares of capital stock of the Company have been duly authorized and validly issued and are fully paid and non-assessable. None of the outstanding shares of capital stock of the Company were issued in violation of the preemptive or other similar rights of any securityholder of the Company which have not been waived. There are no securities or instruments issued by or to which the Company is a party containing anti-dilution or similar provisions that will be triggered by the issuance of the Securities pursuant to this Agreement.
(g) SEC Reports; Disclosure Materials. The Company has filed or furnished, as applicable, on a timely basis all forms, statements, schedules, certifications, reports and other documents required to be filed or furnished by it with the Commission under the Exchange Act or the Securities Act since January 1, 2022 (collectively, and in each case including all exhibits and schedules thereto and documents incorporated by reference therein, the “SEC Reports”). As of the time it was filed with the Commission (or, if amended or superseded by a filing prior to the Closing Date, then on the date of such filing), each of the SEC Reports complied in all material respects with the applicable requirements of the Securities Act or the Exchange Act (as the case may be) and as of the time they were filed, none of the SEC Reports contained any untrue statement of a material fact or omitted to state a material fact required to be stated therein or necessary in order to make the statements therein, in the light of the circumstances under which they were made, not misleading. There are no material outstanding or unresolved comments in comment letters from the staff of the Division of Corporation Finance of the Commission with respect to any of the SEC Reports.
(h) Financial Statements. As of their respective filing dates, the financial statements (including any related notes) contained or incorporated by reference in the SEC Reports (i) complied as to form in all material respects with the Securities Act and the Exchange Act, as applicable, and the published rules and regulations of the Commission applicable thereto, (ii) were prepared in accordance with GAAP (except as may be indicated in the notes to such financial statements or, in the case of unaudited financial statements, as permitted by Form 10-Q of the Commission, and except that the unaudited financial statements may not contain footnotes and are subject to normal and recurring year-end adjustments that are not reasonably expected to be material in amount) applied on a consistent basis unless otherwise noted therein throughout the periods indicated and (iii) fairly present, in all material respects, the consolidated financial position of the Company as of the respective dates thereof and the results of operations and cash flows of the Company for the periods covered thereby. Other than as expressly disclosed in the SEC Reports filed at least one (1) Business Day prior to the date hereof, there has been no material change in the Company’s accounting methods or principles that would be required to be disclosed in the Company’s financial statements in accordance with GAAP. Except as set forth in the consolidated financial statements of the Company included in the SEC Reports filed at least one (1) Business Day prior to the date hereof, the Company has not incurred any liabilities, contingent or otherwise, except those incurred in the ordinary course of business, consistent (as to amount and nature) with past practices since the date of such financial statements, and those incurred in connection with the transactions contemplated hereby and by the Merger Agreement, none of which, individually or in the aggregate, have had or would reasonably be expected to have a Material Adverse Effect. The books of account and other financial records of the Company and each of its Subsidiaries are true and complete in all material respects.
(i) Independent Accountants. Ernst & Young LLP, who have certified certain financial statements of the Company and delivered their report with respect to the audited financial statements included in the SEC Reports, have at all times since the date of enactment of the Sarbanes-Oxley Act been (i) a registered public accounting firm (as defined in Section 2(a)(12) of the Sarbanes-Oxley Act), (ii) to the Company’s Knowledge, “independent” with respect to the Company within the meaning of Regulation S-X under the Exchange Act and (iii) to the Company’s Knowledge, in compliance with subsections (g) through (l) of Section 10A of the Exchange Act and the rules and regulations promulgated by the Commission and the Public Accounting Oversight Board thereunder.
(j) Absence of Certain Changes. Since the date of the Audited Balance Sheet, there has been (i) no material adverse change to, and no material adverse development in, the assets, liabilities, business, properties, operations, condition (financial or otherwise), results of operations or prospects of the Company or its Subsidiaries, (ii) no Material Adverse Effect, (iii) no satisfaction or discharge of any material lien, claim or encumbrance or payment of any obligation by the Company, except in the ordinary course of business and with respect to payments under the CVRs (as defined in the Merger Agreement) and (iv) no waiver, not in the ordinary course of business, by the Company or any Subsidiary of a material right or of a material debt owed to it. Since the date of the Audited Balance Sheet, neither the Company nor any of its Subsidiaries has (i) purchased any of its outstanding Common Stock (other than from its employees or other service providers in connection with the termination of their service pursuant to the terms of its equity compensation plans or agreements) or declared or paid any dividends or distributions, other than payments under the CVRs, (ii) sold any material assets, individually or in the aggregate, outside of the ordinary course of business, (iii) made any material change or material amendment to, or waiver of any material right, or termination of, any Material Contract, (iv) entered into any material transaction or made any material capital expenditures, individually or in the aggregate, outside of the ordinary course of business or (v) experienced any loss of services of any executive officer (as defined in Rule 405 under the Securities Act), other than as disclosed in the SEC Reports prior to the date hereof. Neither the Company nor any of its Subsidiaries has taken any steps to seek protection pursuant to any bankruptcy law, nor does the Company have any knowledge or reason to believe that its creditors (if any) intend to initiate involuntary bankruptcy proceedings or any actual knowledge of any fact that would reasonably lead any such creditor to do so. The Company and its Subsidiaries, individually and on a consolidated basis, are not as of the date hereof, and after giving effect to the transactions contemplated hereby to occur at the Closing, will not be Insolvent (as defined below). For purposes of this Section 3.1(j), “Insolvent” means, with respect to any Person, (i) the present fair saleable value of such Person’s assets is less than the amount required to pay such Person’s total indebtedness, (ii) such Person is unable to pay its debts and liabilities, subordinated, contingent or otherwise, as such debts and liabilities become absolute and matured, (iii) such Person intends to incur or believes that it will incur debts that would be beyond its ability to pay as such debts mature or (iv) such Person has unreasonably small capital with which to conduct the business in which it is engaged as such business is now conducted and is proposed to be conducted.
(k) Litigation. There is no action, suit, proceeding or investigation pending or, to the Company’s Knowledge, currently threatened against the Company, any of its Subsidiaries or any of their respective directors and officers that questions the validity of the Transaction Documents or the right of the Company to enter into the Transaction Documents or to consummate the transactions contemplated hereby and thereby. There is no action, suit, proceeding or investigation pending or, to the Company’s Knowledge, currently threatened against the Company or any Subsidiary or any of their respective directors and officers which would, if there were an unfavorable decision, have or reasonably be expected to have, either individually or in the aggregate, a Material Adverse Effect.
(l) Employment Matters. No material labor dispute exists or, to the Company’s Knowledge, is threatened with respect to any of the employees of the Company or any of its Subsidiaries which would have or would reasonably be expected to result in a Material Adverse Effect. None of the Company’s or any Subsidiary’s employees is a member of a labor union that relates to such employee’s relationship with the Company, and neither the Company nor any of its Subsidiaries is a party to a collective bargaining agreement. To the Company’s Knowledge, no executive officer or key employee of the Company or any Subsidiary, is, or is now expected to be, in violation of any material term of any employment Contract, confidentiality, disclosure or proprietary information agreement or non-competition agreement, or any other Contract or agreement or any restrictive covenant in favor of any third party, and to the Company’s Knowledge, the continued employment of each such executive officer or key employee does not subject the Company or any Subsidiary to any liability with respect to any of the foregoing matters, except, in each case, matters that, individually or in the aggregate, would not reasonably be expected to result in a Material Adverse Effect. The Company is in compliance in all material respects with all U.S. federal, state, local and foreign Laws and regulations relating to employment and employment practices, terms and conditions of employment and wages and hours. Any Employee Plans that are “employee pension benefit plans” within the meaning of Section 3(2) of ERISA and which are intended to meet the qualification requirements of Section 401(a) of the Code have received determination or opinion letters from the Internal Revenue Service on which they may currently rely to the effect that such plans are qualified under Section 401(a) of the Code and the related trusts are exempt from federal income Taxes under Section 501(a) of the Code, respectively, and nothing has occurred that would reasonably be expected to materially adversely affect the qualification of such Employee Plan or the tax exempt status of the related trust.
(m) Conduct of Business; Regulatory Permits. Neither the Company nor any of its Subsidiaries is in violation of any term of or in default under its Articles of Incorporation or the Bylaws or their organizational charter or bylaws, respectively. Neither the Company nor any of its Subsidiaries (i) is in default of or in violation of, nor has the Company or any of its Subsidiaries received notice of a claim that it is in default under or that it is in violation of, any Material Contract (whether or not such default or violation has been waived), or (ii) is in violation of any judgment, decree or order or any statute, ordinance, rule or regulation applicable to the Company or its Subsidiaries, and neither the Company nor any of its Subsidiaries will conduct its business in violation of any of the foregoing, except for possible violations which would not, individually or in the aggregate, have or reasonably be expected to have a Material Adverse Effect. Without limiting the generality of the foregoing, the Company is not in violation of any of the rules, regulations or requirements of the Nasdaq and, to the Company’s Knowledge, there exist no facts or circumstances that would reasonably lead to delisting or suspension of the Common Stock by Nasdaq in the foreseeable future. The Company and its Subsidiaries possess all certificates, authorizations and permits issued by the appropriate regulatory authorities necessary to conduct their respective businesses as currently conducted and as proposed to be conducted, except where the failure to possess such certificates, authorizations or permits would not reasonably be expected to have, individually or in the aggregate, a Material Adverse Effect, and neither the Company nor any such Subsidiary has received any written notice of proceedings relating to the revocation or modification of any such certificate, authorization or permit.
(n) Title to Properties and Assets. None of the Company or its Subsidiaries owns, or has ever owned, any real property. The Company’s or its Subsidiaries’ possession, occupancy, lease, use and/or operation of each such leased property conforms to all applicable Laws in all material respects, and the Company or its Subsidiary, as applicable, has exclusive possession of each such leased property and leasehold interest and has not granted any occupancy rights to tenants or licensees with respect to such leased property or leasehold interest. In addition, each such leased property and leasehold interest is free and clear of all Encumbrances other than Permitted Liens. The Company and each of its Subsidiaries owns, and has good and marketable title to, or, in the case of leased properties and assets, valid leasehold interests in, all tangible properties or tangible assets and equipment used or held for use in their respective business or operations or purported to be owned by any of them, including: (a) all tangible assets reflected on the Audited Balance Sheet and (b) all other tangible assets reflected in the books and records of the Company as being owned by the Company or any of its Subsidiaries. All of such assets are owned or, in the case of leased assets, leased by the Company or any of its Subsidiaries free and clear of any Encumbrances, other than Permitted Liens.
(o) Intellectual Property Rights. The Company and its Subsidiaries own, or have obtained valid and enforceable licenses for, the inventions, patent applications, patents, trademarks, trade names, service names, copyrights, trade secrets and other intellectual property described in the SEC Reports as being owned or licensed by them, other than intellectual property of the Company no longer used that has been abandoned or granted back to the licensor, or which are necessary for the conduct of their respective businesses as currently conducted or as currently proposed to be conducted (collectively, “Intellectual Property”). To the Company’s Knowledge, the operation of the business of the Company and its Subsidiaries, as now conducted or as proposed to be conducted in the SEC Reports, together with the Company’s use of the Company’s Intellectual Property, does not conflict with, infringe, misappropriate or otherwise violate the Intellectual Property of any third party. No actions, suits, claims or proceedings have been asserted, or, to the Company’s Knowledge, threatened against the Company or any of its Subsidiaries alleging any of the foregoing or seeking to challenge, deny or restrict the operation of the business of the Company or its Subsidiaries, and the Company is unaware of any facts which would form a reasonable basis for any such claim. None of the Company or any of its Subsidiaries has received any notice of a claim of infringement, misappropriation or conflict with Intellectual Property rights of others, except for such claims that would not, individually or the in aggregate, have or reasonably be expected to have a Material Adverse Effect. The Intellectual Property rights owned by the Company and its Subsidiaries and, to the Company’s Knowledge, any Intellectual Property rights licensed to the Company or its Subsidiaries, have not been adjudged invalid or unenforceable, in whole or in part, and there is no pending or, to the Company’s Knowledge, threatened action, suit, proceeding or claim by others challenging the validity or scope of any such Intellectual Property rights, and the Company is unaware of any facts which would form a reasonable basis for any such challenge, except for such actions, suits, proceedings, or claims that would not, individually or the in aggregate, be reasonably expected to have a Material Adverse Effect. None of the Company or any of its Subsidiaries is a party to or bound by any options, licenses or agreements with respect to the Intellectual Property rights of any other person or entity that are required to be set forth in the SEC Reports. None of the technology or Intellectual Property used by the Company or its Subsidiaries in their respective businesses has been obtained or is being used by the Company in violation of any contractual obligation binding on the Company or any of its Subsidiaries or, to the Company’s Knowledge, any of their respective officers, directors or employees or otherwise in violation of the rights of any persons, except as would not reasonably be expected to result in a Material Adverse Effect.
(p) Insurance. Each of the Company and its Subsidiaries are insured by recognized, financially sound and reputable institutions with policies in such amounts and with such deductibles and covering such risks as are generally deemed adequate and customary for their businesses including, but not limited to, policies covering real and personal property owned or leased by the Company and its Subsidiaries against theft, damage, destruction, acts of vandalism and earthquakes and policies covering the Company and its Subsidiaries for product liability claims and clinical trial liability claims. The Company has no reason to believe that it or any of its Subsidiaries will not be able to (i) renew its existing insurance coverage as and when such policies expire or (ii) obtain comparable coverage from similar institutions as may be necessary or appropriate to conduct its business as now conducted and at a cost that could not reasonably be expected to result in a Material Adverse Effect. Neither the Company nor any of its Subsidiaries has been denied any insurance coverage which it has sought or for which it has applied.
(q) Transactions with Affiliates and Employees. Since the date of the Company’s Annual Report on Form 10-K filed with the Commission on March 29, 2024, no event has occurred that is material to the Company and that would be required to be reported by the Company pursuant to Item 404 of Regulation S-K promulgated by the SEC.
(r) Company’s Accounting System. The Company and each of its Subsidiaries makes and keeps accurate books and records and maintains a system of internal control over financial reporting (as defined in Rules 13a-15 and 15d-15 under the Exchange Act) sufficient to provide reasonable assurance that: (i) transactions are executed in accordance with management’s general or specific authorization; (ii) transactions are recorded as necessary to permit preparation of financial statements in conformity with generally accepted accounting principles as applied in the United States and to maintain accountability for assets; (iii) access to assets is permitted only in accordance with management’s general or specific authorization; (iv) the recorded accountability for assets is compared with existing assets at reasonable intervals and appropriate action is taken with respect to any differences; and (v) the interactive data in eXtensible Business Reporting Language included or incorporated by reference in the SEC Reports fairly presents the information called for in all material respects and is prepared in accordance with the Commission’s rules and guidelines applicable thereto, except as would not, individually or the in aggregate, be reasonably expected to have a Material Adverse Effect. Since January 1, 2022, neither the Company nor any Subsidiary nor, to the Company’s Knowledge, any director, officer, employee, auditor, accountant or representative of the Company or any Subsidiary has received or otherwise had or obtained knowledge of any material complaint, allegation, assertion or claim, whether written or oral, regarding the accounting or auditing practices, procedures, methodologies or methods of the Company or any Subsidiary or their respective internal accounting controls, including any material complaint, allegation, assertion or claim that the Company or any Subsidiary has engaged in questionable accounting or auditing practices.
(s) Sarbanes-Oxley; Disclosure Controls. The Company is in compliance in all material respects with the applicable provisions of the Sarbanes-Oxley Act of 2002 and the rules and regulations promulgated thereunder. The Company maintains a system of internal accounting controls designed to ensure that (a) material information relating to the Company and its Subsidiaries, is made known to the Company’s principal executive officer and its principal financial officer by others within those entities and (b) that information required to be disclosed by the Company in reports that it files, furnishes or submits under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the Commission’s rules and forms, including controls and procedures designed to ensure that such information is accumulated and communicated to the Company’s management as appropriate to allow timely decisions regarding required disclosure. The Company has established and maintains disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) for the Company and designed such disclosure controls and procedures to ensure that material information relating to the Company is made known to the certifying officers by others within those entities, particularly during the period in which the Company’s most recently filed periodic report under the Exchange Act, as the case may be, is being prepared. The Company has established internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with GAAP. The Company’s certifying officers have evaluated the effectiveness of the Company’s disclosure controls and procedures and the Company’s internal control over financial reporting (collectively, “internal controls”) as of the end of the period covered by the most recently filed periodic report under the Exchange Act (such date, the “Evaluation Date”). The Company presented in its most recently filed periodic report under the Exchange Act the conclusions of the certifying officers about the effectiveness of such internal controls based on their evaluations as of the Evaluation Date. Since the Evaluation Date, there have been no significant changes in the Company’s internal controls or, to the Company’s Knowledge, in other factors that could materially affect the Company’s internal controls and there have been no material weaknesses in the Company’s internal control over financial reporting (whether or not remediated). The Company maintains and will continue to maintain a standard system of accounting established and administered in accordance with GAAP and the applicable requirements of the Exchange Act.
(t) Certain Fees. No person or entity will have, as a result of the transactions contemplated by this Agreement, any valid right, interest or claim against or upon the Company or a Purchaser for any commission, fee or other compensation pursuant to any agreement, arrangement or understanding entered into by or on behalf of the Company, other than the Placement Agents with respect to the offer and sale of the Securities (which placement agent fees are being paid by the Company). The Purchasers shall have no obligation with respect to any fees or with respect to any claims made by or on behalf of other Persons for fees of a type contemplated in this Section 3.1(t) that may be due in connection with the transactions contemplated by the Transaction Documents. The Company shall indemnify, pay, and hold each Purchaser harmless against, any liability, loss or expense (including, without limitation, attorneys’ fees and out-of-pocket expenses) arising in connection with any such right, interest or claim.
(u) Private Placement. Assuming the accuracy of the Purchasers’ representations and warranties set forth in Section 3.2 of this Agreement, no registration under the Securities Act is required for the offer and sale of the Securities by the Company to the Purchasers under the Transaction Documents. The issuance and sale of the Securities hereunder does not contravene the rules and regulations of the Principal Trading Market.
(v) Company Not an “Investment Company”. The Company is not, and will not be, immediately after receipt of payment for the Securities, required to register as an “investment company” under the Investment Company Act of 1940, as amended. As long as the Pre-Funded Warrants remain outstanding, the Company shall use its commercially reasonable efforts to conduct its business in a manner so that it will not become an “investment company” subject to registration under the Investment Company Act of 1940, as amended.
(w) Registration Rights. Other than each of the Purchasers pursuant to the Registration Rights Agreement, no Person has any right to cause the Company to effect the registration under the Securities Act of the offer and sale of any securities of the Company other than those offers and sales which are currently registered on an effective registration statement on file with the Commission.
(x) Listing and Maintenance Requirements. The Company’s Common Stock is registered pursuant to Section 12(b) or Section 12(g) of the Exchange Act, and the Company has taken no action designed to terminate the registration of the Common Stock under the Exchange Act, nor has the Company received any notification that the Commission or the Principal Trading Market is contemplating terminating such registration or listing. The Company is, and immediately following the Closing will be, in compliance with all applicable listing requirements of the Principal Trading Market.
(y) Reservation of Common Stock. Upon the Company Authorized Stock Increase, the Company shall have reserved and the Company shall thereafter continue to reserve and keep available at all times, free of preemptive rights, a sufficient number of shares of Common Stock for the purpose of enabling the Company to issue shares of Common Stock pursuant to any exercise of the Pre-Funded Warrants.
(z) Disclosure. The Company confirms that it has not provided, and to the Company’s Knowledge, none of its officers or directors nor any other Person acting on its or their behalf (including, without limitation, the Placement Agents) has provided, and it has not authorized the Placement Agents to provide, any Purchaser or its respective agents or counsel with any information that it believes constitutes material, non-public information except insofar as the existence, provisions and terms of the Transaction Documents and the proposed transactions hereunder and thereunder may constitute such information, all of which will be disclosed by the Company in the Press Release as contemplated by Section 4.4 hereof. The Company understands and confirms that the Purchasers will rely on the foregoing representations in effecting transactions in securities of the Company.
(aa) No Integrated Offering. Assuming the accuracy of the Purchasers’ representations and warranties set forth in Section 3.2, none of the Company, its Subsidiaries nor, to the Company’s Knowledge, any of its Affiliates or any Person acting on its behalf has, directly or indirectly, at any time within the past six (6) months, made any offers or sales of any Company security or solicited any offers to buy any security under circumstances that would (i) eliminate the availability of the exemption from registration under the Securities Act in connection with the offer and sale by the Company of the Securities as contemplated hereby or (ii) cause the offering of the Securities pursuant to the Transaction Documents to be integrated with prior offerings by the Company for purposes of any applicable law, regulation or stockholder approval provisions, including, without limitation, under the rules and regulations of any Trading Market on which any of the securities of the Company are listed or designated.
(bb) Tax Matters. The Company and each of its Subsidiaries has timely filed all income Tax returns and all other material Tax returns that were required to be filed by or with respect to it under applicable Law. All such Tax returns were correct and complete in all material respects and have been prepared in material compliance with all applicable Law. Subject to exceptions as would not be material, no claim has ever been made by a Governmental Authority in a jurisdiction where the Company or any of its Subsidiaries does not file Tax returns that the Company or any of its Subsidiaries is subject to Taxation by that jurisdiction. All material amounts of Taxes due and owing by the Company and each of its Subsidiaries (whether or not shown on any Tax return) have been timely paid. The unpaid Taxes of the Company and each of its Subsidiaries for periods (or portions thereof) ending on or prior to the date of the Audited Balance Sheet do not materially exceed the accruals for current Taxes set forth on the Audited Balance Sheet. Since the date of the Audited Balance Sheet, neither the Company nor any of its Subsidiaries has incurred any material liability for Taxes outside the ordinary course of business or otherwise inconsistent with past custom and practice. The Company is classified as a Subchapter C corporation for U.S. federal Tax purposes.
(cc) Compliance with Environmental Laws. Since January 1, 2022, the Company and each of its Subsidiaries has complied with all applicable federal, state, local or foreign Law relating to pollution or protection of human health or the environment (including ambient air, surface water, ground water, land surface or subsurface strata), including any law or regulation relating to emissions, discharges, releases or threatened releases of Hazardous Materials, or otherwise relating to the manufacture, processing, distribution, use, treatment, storage, disposal, transport or handling of Hazardous Materials (“Environmental Laws”), which compliance includes the possession by the Company of all permits and other governmental authorizations required under applicable Environmental Laws and compliance with the terms and conditions thereof, except for any failure to be in compliance that, individually or in the aggregate, would not result in or reasonably be expected to result in a Material Adverse Effect. Neither the Company nor any of its Subsidiaries has received since January 1, 2022, any written notice or other communication (in writing or otherwise), whether from a Governmental Authority, citizens group, employee or otherwise, that alleges that the Company or any of its Subsidiaries is not in compliance with any Environmental Law, and, to the Company’s Knowledge, there are no circumstances that may prevent or interfere with the Company’s or any of its Subsidiaries’ compliance with any Environmental Law in the future, except where such failure to comply would not reasonably be expected to have a Material Adverse Effect. To the Company’s Knowledge: (i) no current or prior owner of any property leased or controlled by the Company or any of its Subsidiaries has received, since January 1, 2022, any written notice or other communication relating to property owned or leased at any time by the Company or any of its Subsidiaries, whether from a Governmental Authority, citizens group, employee or otherwise, that alleges that such current or prior owner or the Company or any of its Subsidiaries is not in compliance with or violated any Environmental Law relating to such property and neither the Company nor any of its Subsidiaries has any material liability under any Environmental Law.
(dd) No General Solicitation. Neither the Company nor, to the Company’s Knowledge, any person acting on behalf of the Company has, directly or indirectly, offered or sold any of the Securities, or solicited any offers to buy any Securities, under any circumstances that would require registration under the Securities Act of the Securities, including by any form of general solicitation or general advertising.
(ee) Anti-Corruption and Anti-Bribery Laws. Neither the Company nor any of its Subsidiaries nor any director, officer, or employee of the Company or any of its Subsidiaries, nor to the Company’s Knowledge, any agent, Affiliate or other person acting on behalf of the Company or any of its Subsidiaries has, in the course of its actions for, or on behalf of, the Company or any of its Subsidiaries (i) used any corporate funds for any unlawful contribution, gift, entertainment or other unlawful expenses relating to political activity; (ii) made or taken any act in furtherance of an offer, promise, or authorization of any direct or indirect unlawful payment or benefit to any non-U.S. or domestic government official or employee, including of any government-owned or controlled entity or public international organization, or any political party, party official, or candidate for political office; (iii) violated or is in violation of any provision of the U.S. Foreign Corrupt Practices Act of 1977, as amended (the “FCPA”), the UK Bribery Act 2010, or any other applicable anti-bribery or anti-corruption Law; or (iv) made, offered, authorized, requested, or taken an act in furtherance of any unlawful bribe, rebate, payoff, influence payment, kickback or other unlawful payment or benefit. The Company and its Subsidiaries and, to the Company’s Knowledge, the Company’s Affiliates have conducted their respective businesses in compliance with the FCPA and have instituted and maintain policies and procedures designed to ensure, and which are reasonably expected to continue to ensure, continued compliance therewith.
(ff) Money Laundering Laws. The operations of the Company and its Subsidiaries are and have been conducted at all times in compliance with applicable financial recordkeeping and reporting requirements of the USA Patriot Act, the Bank Secrecy Act of 1970, as amended, the money laundering statutes of all jurisdictions, the rules and regulations thereunder and any related or similar rules, regulations or guidelines, issued, administered or enforced by any governmental agency (collectively, the “Money Laundering Laws”); and no action, suit or proceeding by or before any court or governmental agency, authority or body or any arbitrator or non-governmental authority involving the Company or its Subsidiaries with respect to the Money Laundering Laws is pending or, to the Company’s Knowledge, threatened.
(gg) OFAC. Neither the Company nor its Subsidiaries nor any of their respective Affiliates, directors, officers, nor to the Company’s Knowledge, any agent or employee of the Company or its Subsidiaries is subject to any sanctions administered or enforced by the Office of Foreign Assets Control (“OFAC”) of the United States Treasury Department, the U.S. Department of State, the United Nations Security Council, the European Union, His Majesty’s Treasury or any other relevant sanctions authority; and the Company will not directly or indirectly use the proceeds of the offering of the securities contemplated hereby, or lend, contribute or otherwise make available such proceeds to any subsidiary, joint venture partner or other person or entity for the purpose of financing the activities of any person that is the target of sanctions administered or enforced by such authorities or in connection with any country or territory that is the target of country- or territory-wide OFAC sanctions (currently, Iran, Syria, Cuba, North Korea, the Crimea, so-called Donetsk People’s Republic, and so-called Luhansk People’s Republic regions of Ukraine).
(hh) Off Balance Sheet Arrangements. There is no transaction, arrangement, or other relationship between the Company (or any Subsidiary) and an unconsolidated or other off balance sheet entity that is required to be disclosed by the Company in SEC Reports and is not so disclosed and would have or reasonably be expected to result in a Material Adverse Effect.
(ii) Acknowledgment Regarding Purchaser’s Purchase of Securities. The Company acknowledges and agrees that each Purchaser is acting solely in the capacity of an arm’s length purchaser with respect to this Agreement and the other Transaction Documents and the transactions contemplated hereby and thereby, and that the obligations of each Purchaser under this Agreement and the other Transaction Documents are several and not joint. The Company further acknowledges that no Purchaser is acting as a financial advisor or fiduciary of the Company or any of its Subsidiaries (or in any similar capacity) with respect to this Agreement and the other Transaction Documents and the transactions contemplated hereby and thereby, and any advice given by a Purchaser or any of its representatives or agents in connection with this Agreement and the other Transaction Documents and the transactions contemplated hereby and thereby is merely incidental to such Purchaser’s purchase of the Securities. The Company further represents to each Purchaser that the Company’s decision to enter into the Transaction Documents has been based solely on the independent evaluation by the Company and its representatives.
(jj) No Price Stabilization or Manipulation; Compliance with Regulation M. Neither the Company nor any of its Subsidiaries has taken, directly or indirectly, any action designed to or that might cause or result in stabilization or manipulation of the price of any security of the Company to facilitate the sale or resale of the Securities or otherwise, and has taken no action which would directly or indirectly violate Regulation M under the Exchange Act.
(kk) Clinical Data and Regulatory Compliance. The preclinical tests, clinical trials and other studies (collectively, “studies”) that are described in, or the results of which are referred to in, the SEC Reports were and, if still pending, are being conducted in all material respects in accordance with the protocols, procedures and controls designed and approved for such studies and with standard medical and scientific research procedures. Each description of the results of such studies is accurate and complete in all material respects and fairly presents the data derived from such studies, and the Company and its Subsidiaries have no knowledge of any other studies the results of which are inconsistent with, or otherwise call into question, the results described or referred to in the SEC Reports. The Company and its Subsidiaries have made all such filings and obtained all such approvals as may be required by the Food and Drug Administration of the U.S. Department of Health and Human Services or any committee thereof or from any other U.S. or non-U.S. government or drug or medical device regulatory agency, or health care facility Institutional Review Board (collectively, the “Regulatory Agencies”). Neither the Company nor any of its Subsidiaries has received any notice of, or correspondence from, any Regulatory Agency requiring the termination, suspension or modification of any clinical trials that are described or referred to in the SEC Reports and are currently being conducted. The Company and its Subsidiaries have each operated and currently are in compliance in all material respects with all applicable rules, regulations and policies of the Regulatory Agencies.
(ll) No Additional Agreements. The Company does not have any agreement or understanding (including side letters) with any Purchaser with respect to the transactions contemplated by the Transaction Documents other than as specified in the Transaction Documents.
(mm) Security. Except as would not reasonably be expected to have a Material Adverse Effect, the Company and its Subsidiaries’ information technology assets and equipment, computers, systems, networks, hardware, software, websites, applications, and databases (collectively, “IT Systems”) are adequate for, and operate and perform in all respects as required in connection with the operation of the business of the Company and its Subsidiaries as currently conducted, and are free and clear of all material Trojan horses, time bombs, malware and other malicious code. The Company and its Subsidiaries have implemented and maintained commercially reasonable physical, technical and administrative controls designed to maintain and protect the confidentiality, integrity, availability, privacy and security of all sensitive, confidential or regulated data (“Confidential Data”) used or maintained in connection with their businesses and Personal Data, and the integrity, availability continuous operation, redundancy and security of all IT Systems. “Personal Data” means the following data used in connection with the Company’s and its Subsidiaries’ businesses and in their possession or control: (i) a natural person’s name, street address, telephone number, e-mail address, photograph, social security number or other tax identification number, driver’s license number, passport number, credit card number, bank information, or customer or account number; (ii) information that identifies, relates to, or may reasonably be used to identify an individual; (iii) any information regarding an individual’s medical history, mental or physical condition, or medical treatment or diagnosis by a health care professional; (iv) an individual’s health insurance policy number or subscriber identification number, any unique identifier used by a health insurer to identify the individual, or any information in an individual’s application and claims history; (v) any information which would qualify as “protected health information” under the Health Insurance Portability and Accountability Act of 1996, as amended by the Health Information Technology for Economic and Clinical Health Act (collectively, “HIPAA”); (vi) any information which would qualify as “personal data,” “personal information” (or similar term) under the Privacy Laws (as defined below); and (vii) any other piece of information that alone, or combined with other information, allows the identification of such natural person, or his or her family, or permits the collection or analysis of any data related to an identified person’s health or sexual orientation. To the Company’s Knowledge, there have been no breaches, outages or unauthorized uses of or accesses to the IT Systems, Confidential Data, and Personal Data. The Company and its Subsidiaries are presently, and at all prior times were, in material compliance with all applicable laws or statutes and all judgments and orders binding on the Company, applicable binding rules and regulations of any court or arbitrator or governmental or regulatory authority, and their internal policies and contractual obligations, each relating to the Processing, privacy and security of Personal Data and Confidential Data, the privacy and security of IT Systems and the protection of such IT Systems, Confidential Data, and Personal Data from unauthorized use, access, misappropriation or modification.
(nn) Compliance with Data Privacy Laws. The Company and its Subsidiaries are, and at all prior times were, in material compliance with all applicable state and federal data privacy and security Laws and regulations regarding the collection, use, storage, retention, disclosure, transfer, disposal, or any other processing (collectively “Process” or “Processing”) of Personal Data, including HIPAA, the California Consumer Privacy Act, and the European Union General Data Protection Regulation (EU 2016/679) (collectively, the “Privacy Laws”). To ensure compliance with the Privacy Laws, the Company and its Subsidiaries have in place, comply with, and take all appropriate steps necessary to ensure compliance in all material respects with their policies and procedures relating to data privacy and security, and the Processing of Personal Data and Confidential Data (the “Privacy Statements”). The Company and its Subsidiaries have, except as would not reasonably be expected, individually or in the aggregate, to have a Material Adverse Effect, at all times since January 1, 2022 provided accurate notice of its Privacy Statements then in effect to its clients, employees, third party vendors and representatives. None of such disclosures made or contained in any Privacy Statements have been materially inaccurate, misleading, incomplete, or in material violation of any Privacy Laws. The Company further certifies that neither it nor any of its Subsidiaries: (i) has received notice of any actual or potential claim, complaint, proceeding, regulatory proceeding or liability under or relating to, or actual or potential violation of, any of the Privacy Laws, contracts related to the Processing of Personal Data or Confidential Data, or Privacy Statements, and has no knowledge of any event or condition that would reasonably be expected to result in any such notice; (ii) is currently conducting or paying for, in whole or in part, any investigation, remediation, or other corrective action pursuant to any Privacy Law or contract; or (iii) is a party to any order, decree, or agreement that imposes any obligation or liability under any Privacy Law.
3.2. Representations and Warranties of the Purchasers. Each Purchaser hereby, for itself and for no other Purchaser, represents and warrants to the Company and the Placement Agents as follows:
(a) Organization; Authority. Such Purchaser is an entity duly organized, validly existing and in good standing under the Laws of the jurisdiction of its organization with the requisite corporate or, if such Purchaser is not a corporation, such partnership, limited liability company or other applicable power and authority to enter into and to consummate the transactions contemplated by the applicable Transaction Documents and otherwise to carry out its obligations hereunder and thereunder. The execution and delivery of this Agreement by such Purchaser and performance by such Purchaser of the transactions contemplated by this Agreement have been duly authorized by all necessary corporate or, if such Purchaser is not a corporation, such partnership, limited liability company or other applicable like action, on the part of such Purchaser. Each Transaction Document to which it is a party has been duly executed by such Purchaser, and when delivered by such Purchaser in accordance with the terms hereof, will constitute the valid and legally binding obligation of such Purchaser, enforceable against it in accordance with its terms, except as such enforceability may be limited by applicable bankruptcy, examinership, insolvency, reorganization, moratorium, liquidation or similar laws relating to, or affecting generally the enforcement of, creditors’ rights and remedies or by other equitable principles of general application.
(b) Convertible Notes. To the extent a Purchaser is surrendering such Purchaser’s Convertible Note(s) pursuant to Section 2.2(c), such Convertible Note(s) is free and clear of any lien, encumbrance or other adverse claim.
(c) No Conflicts. The execution, delivery and performance by such Purchaser of this Agreement and the Registration Rights Agreement and the consummation by such Purchaser of the transactions contemplated hereby and thereby will not (i) result in a violation of the organizational documents of such Purchaser, (ii) conflict with, or constitute a default (or an event which with notice or lapse of time or both would become a default) under, or give to others any rights of termination, amendment, acceleration or cancellation of, any agreement, indenture or instrument to which such Purchaser is a party, or (iii) result in a violation of any Law, rule, regulation, order, judgment or decree (including U.S. federal and state securities laws) applicable to such Purchaser, except in the case of clauses (ii) and (iii) above, for such conflicts, defaults, rights or violations which would not, individually or in the aggregate, reasonably be expected to have a material adverse effect on the ability of such Purchaser to perform its obligations hereunder.
(d) Investment Intent. Such Purchaser understands that the Securities are “restricted securities” and the offer and sale thereof have not been registered under the Securities Act or any applicable U.S. state securities law and is acquiring the Securities as principal for its own account and not with a view to, or for distributing or reselling such Securities or any part thereof in violation of the Securities Act or any applicable U.S. state or other securities laws, provided, however, that by making the representations herein, such Purchaser does not agree to hold any of the Securities for any minimum period of time and reserves the right, subject to the provisions of this Agreement and the Registration Rights Agreement, at all times to sell or otherwise dispose of all or any part of such Securities pursuant to an effective registration statement under the Securities Act or under an exemption from such registration and in compliance with applicable U.S. federal, state and other securities laws. Such Purchaser is acquiring the Securities hereunder in the ordinary course of its business. Such Purchaser does not presently have any agreement, plan or understanding, directly or indirectly, with any Person to distribute or effect any distribution of any of the Securities (or any securities which are derivatives thereof) to or through any person or entity in violation of federal securities law; such Purchaser is not a registered broker-dealer under Section 15 of the Exchange Act or an entity engaged in a business that would require it to be so registered as a broker-dealer.
(e) Purchaser Status. At the time such Purchaser was offered the Securities, it was, and at the date hereof it is, and on each date on which it exercises any Pre-Funded Warrants, it will be, an “accredited investor” as defined in Rule 501(a) under the Securities Act.
(f) General Solicitation. Such Purchaser is not purchasing the Securities as a result of any advertisement, article, notice or other communication regarding the Securities published in any newspaper, magazine or similar media or broadcast over television or radio or presented at any seminar or any other general advertisement. The purchase of the Securities by such Purchaser has not been solicited by or through anyone other than the Merger Partner or, on the Merger Partner’s behalf, the Placement Agents.
(g) Experience of Such Purchaser. Such Purchaser, either alone or together with its representatives, has such knowledge, sophistication and experience in business and financial matters so as to be capable of evaluating the merits and risks of the prospective investment in the Securities, and has so evaluated the merits and risks of such investment. Such Purchaser is able to bear the economic risk of an investment in the Securities.
(h) Access to Information. Such Purchaser acknowledges that it has had the opportunity to review the SEC Reports and has been afforded (i) the opportunity to ask such questions as it has deemed necessary of, and to receive answers from, representatives of the Company or Merger Partner concerning the terms and conditions of the offering of the Securities and the merits and risks of investing in the Securities; (ii) access to information about the Company and the Subsidiaries, the Merger Partner and their respective financial condition, results of operations, business, properties, management and prospects sufficient to enable it to evaluate its investment; and (iii) the opportunity to obtain such additional information that the Company possesses or can acquire without unreasonable effort or expense that is necessary to make an informed investment decision with respect to the investment. Neither such inquiries nor any other investigation conducted by or on behalf of such Purchaser or its representatives or counsel shall modify, amend or affect such Purchaser’s right to rely on the truth, accuracy and completeness of the SEC Reports and the Company’s representations and warranties contained in the Transaction Documents. Such Purchaser has sought such accounting, legal and tax advice as it has considered necessary to make an informed decision with respect to its acquisition of the Securities.
(i) Certain Trading Activities. Other than with respect to the transactions contemplated herein, since the time that such Purchaser was first contacted by the Company, the Placement Agents or any other Person regarding the transactions contemplated hereby, the Purchaser has not, directly or indirectly, effected or agreed to effect any Short Sales. Notwithstanding the foregoing, (i) in the case of a Purchaser that is a multi-managed investment vehicle whereby separate portfolio managers manage separate portions of such Purchaser’s assets, the foregoing representation shall only apply with respect to the portion of assets managed by the portfolio manager that made the investment decision to purchase the Securities covered by this Agreement and (ii) and in the case of a Purchaser whose investment adviser utilized an information barrier with respect to the information regarding the transactions contemplated hereunder after first being contacted by the Company or such other Person representing the Company, the representation set forth above shall only apply after the point in time when the portfolio manager who manages such Purchaser’s assets was informed of the information regarding the transactions contemplated hereunder and, with respect to the Purchaser’s investment adviser, the representation set forth above shall only apply with respect to any purchases or sales, including Short Sales, of the securities of the Company on behalf of other funds or investment vehicles for which the Purchaser’s investment adviser is also an investment adviser or sub-adviser after the point in time when the portfolio manager who manages the assets of such other funds or investment vehicles for which the Purchaser’s investment adviser is also an investment adviser or sub-adviser was informed of the information regarding the transactions contemplated hereunder. Other than to other Persons party to this Agreement and to the Purchaser’s representatives or agents, including, but not limited to, the Purchaser’s legal, tax and investment advisors, such Purchaser has maintained the confidentiality of all disclosures made to it in connection with this transaction (including the existence and terms of this transaction). Notwithstanding the foregoing, for avoidance of doubt, nothing contained herein shall constitute a representation or warranty, or preclude any actions, with respect to the identification of the availability of, or securing of, available shares to borrow in order to effect Short Sales or similar transactions in the future.
(j) Brokers and Finders. No Person will have, as a result of the transactions contemplated by this Agreement, any valid right, interest or claim against or upon the Purchaser for any commission, fee or other compensation pursuant to any agreement, arrangement or understanding entered into by or on behalf of the Purchaser. No Purchaser shall have any obligation with respect to any fees, or with respect to any claims made by or on behalf of other Persons for fees, in each case of the type contemplated by this Section 3.2(j) that may be due in connection with the transactions contemplated by this Agreement or the Transaction Documents.
(k) Independent Investment Decision. Such Purchaser has independently evaluated the merits of its decision to purchase Securities pursuant to the Transaction Documents, and such Purchaser confirms that it has not relied on the advice of any other Purchaser’s business and/or legal counsel in making such decision. Such Purchaser understands that nothing in this Agreement or any other materials presented by or on behalf of the Company to the Purchaser in connection with the purchase of the Securities constitutes legal, tax or investment advice. Such Purchaser has consulted such legal, tax and investment advisors as it, in its sole discretion, has deemed necessary or appropriate in connection with its purchase of the Securities. Such Purchaser understands that the Placement Agents have acted solely as the agent of the Company in this placement of the Securities and such Purchaser has not relied on the business or legal advice of the Placement Agents or any of their agents, counsel or Affiliates in making its investment decision hereunder, and confirms that none of such Persons has made any representations or warranties to such Purchaser in connection with the transactions contemplated by the Transaction Documents.
(l) Reliance on Exemptions. Such Purchaser understands that the Securities are being offered and sold to it in reliance on specific exemptions from the registration requirements of United States federal and state securities laws and that the Company is relying in part upon the truth and accuracy of, and such Purchaser’s compliance with, the representations, warranties, agreements, acknowledgements and understandings of such Purchaser set forth herein in order to determine the availability of such exemptions and the eligibility of such Purchaser to acquire the Securities.
(m) No Governmental Review. Such Purchaser understands that no United States federal or state agency or any other government or governmental agency has passed on or made any recommendation or endorsement of the Securities or the fairness or suitability of the investment in the Securities nor have such authorities passed upon or endorsed the merits of the offering of the Securities.
(n) Regulation M. Such Purchaser is aware that the anti-manipulation rules of Regulation M under the Exchange Act may apply to sales of Securities and other activities with respect to the Securities by the Purchasers.
(o) Beneficial Ownership. The purchase by such Purchaser of the Securities issuable to it at the Closing will not result in such Purchaser (individually or together with any other Person with whom such Purchaser has identified, or will have identified, itself as part of a “group” in a public filing made with the Commission involving the Company’s securities) acquiring, or obtaining the right to acquire, beneficial ownership in excess of 19.9% of the outstanding shares of Common Stock or the voting power of the Company on a post transaction basis that assumes that such Closing shall have occurred. Such Purchaser does not presently intend to, alone or together with others, make a public filing with the Commission to disclose that it has (or that it together with such other Persons have) acquired, or obtained the right to acquire, as a result of such Closing when added to any other securities of the Company that it or they then own or have the right to acquire, beneficial ownership in excess of 19.9% of the outstanding shares of Common Stock or the voting power of the Company on a post transaction basis that assumes that such Closing shall have occurred.
(p) Residency. Such Purchaser’s residence (if an individual) or offices in which its investment decision with respect to the Securities was made (if an entity) are located at the address set forth under such Purchaser’s name on Exhibit A hereto or as otherwise specified below its address on Exhibit A hereto.
The Company and each of the Purchasers acknowledge and agree that no party to this Agreement has made or makes any representations or warranties with respect to the transactions contemplated hereby other than those specifically set forth in this Article III and the Transaction Documents.
ARTICLE IV
OTHER AGREEMENTS OF THE PARTIES
4.1. Transfer Restrictions.
(a) Compliance with Laws. Notwithstanding any other provision of this Article IV, each Purchaser covenants that the Securities may be disposed of only pursuant to an effective registration statement under, and in compliance with the requirements of, the Securities Act, or pursuant to an available exemption from, or in a transaction not subject to, the registration requirements of the Securities Act, and in compliance with any applicable U.S. state and federal securities laws. In connection with any transfer of the Securities other than (i) pursuant to an effective registration statement, (ii) to the Company, (iii) pursuant to Rule 144 (provided that the Purchaser provides the Company with reasonable assurances (in the form of seller and, if applicable, broker representation letters) that the securities may be sold pursuant to such rule), or (iv) in connection with a bona fide pledge as contemplated in Section 4.1(b), the Company may require the transferor thereof to provide to the Company an opinion of counsel selected by the transferor and reasonably acceptable to the Company, the form and substance of which opinion shall be reasonably satisfactory to the Company, to the effect that such transfer does not require registration of such transferred Securities under the Securities Act and, as a condition of transfer, any such transferee shall agree in writing to be bound by the terms of this Agreement and the Registration Rights Agreement and shall have the rights of a Purchaser under this Agreement and the Registration Rights Agreement with respect to such transferred Securities.
(b) Legends. Book-entry statements evidencing the Shares and Warrant Shares and the certificate representing the Pre-Funded Warrants shall bear any legend as required by the “blue sky” Laws of any state and a restrictive legend in substantially the following form:
“THE OFFER AND SALE OF THE SECURITIES REPRESENTED BY THIS CERTIFICATE TO WHICH THIS CONFIRMATION RELATES OR THE SECURITIES INTO WHICH THIS SECURITY IS EXERCISABLE HAVE NOT BEEN REGISTERED UNDER THE SECURITIES ACT OF 1933, AS AMENDED, OR THE SECURITIES LAWS OF ANY STATE OF THE UNITED STATES. THE SECURITIES MAY NOT BE SOLD, OFFERED FOR SALE, PLEDGED, HYPOTHECATED, TRANSFERRED OR ASSIGNED IN THE ABSENCE OF AN EFFECTIVE REGISTRATION STATEMENT FOR THE SECURITIES UNDER APPLICABLE SECURITIES LAWS, OR UNLESS OFFERED, SOLD, PLEDGED, HYPOTHECATED OR TRANSFERRED PURSUANT TO AN AVAILABLE EXEMPTION FROM THE REGISTRATION REQUIREMENTS OF THOSE LAWS. THE COMPANY AND ITS TRANSFER AGENT SHALL BE ENTITLED TO REQUIRE AN OPINION OF COUNSEL SATISFACTORY TO THE COMPANY AND THE TRANSFER AGENT THAT SUCH REGISTRATION IS NOT REQUIRED. NOTWITHSTANDING THE FOREGOING, THE SECURITIES MAY BE PLEDGED IN CONNECTION WITH A BONA FIDE MARGIN ACCOUNT OR OTHER LOAN OR FINANCING ARRANGEMENT SECURED BY THE SECURITIES.”
The Company acknowledges and agrees that a Purchaser may from time to time pledge, and/or grant a security interest in, some or all of the legended Securities in connection with applicable securities laws, pursuant to a bona fide margin agreement in compliance with a bona fide margin loan. Such a pledge would not be subject to approval or consent of the Company and no legal opinion of legal counsel to the pledgee, secured party or pledgor shall be required in connection with the pledge, but such legal opinion shall be required in connection with a subsequent transfer or foreclosure following default by the Purchaser transferee of the pledge. No notice shall be required of such pledge, but Purchaser’s transferee shall promptly notify the Company of any such subsequent transfer or foreclosure. Each Purchaser acknowledges that the Company shall not be responsible for any pledges relating to, or the grant of any security interest in, any of the Securities or for any agreement, understanding or arrangement between any Purchaser and its pledgee or secured party. At the appropriate Purchaser’s expense, the Company will execute and deliver such reasonable documentation as a pledgee or secured party of Securities may reasonably request in connection with a pledge or transfer of the Securities, including the preparation and filing of any required prospectus supplement under Rule 424(b)(3) of the Securities Act or other applicable provision of the Securities Act to appropriately amend the list of selling stockholders thereunder. Each Purchaser acknowledges and agrees that any Securities subject to a pledge or security interest as contemplated by this Section 4.1(b) shall continue to bear the legend set forth in this Section 4.1(b) and be subject to the restrictions on transfer set forth in Section 4.1(b).
(c) Removal of Legends. Once a registration statement covering the resale of the Shares and the Warrant Shares is declared effective, the Company shall remove all restrictive legends, including the legend set forth in Section 4.1(b) above, and the Company shall, provide the Transfer Agent with a blanket opinion of counsel permitting such removal. Further, the Company shall remove all restrictive legends, including the legend set forth in Section 4.1(b) above, (i) following any sale of such Shares or Warrant Shares pursuant to Rule 144 (assuming cashless exercise of the Pre-Funded Warrants) or any other applicable exemption from the registration requirements of the Securities Act, or (ii) if such Shares or Warrant Shares are eligible for resale under Rule 144(b)(1) or any successor provision (assuming cashless exercise of the Pre-Funded Warrants). Without limiting the foregoing, upon request of the Purchaser, subject to receipt by the Company of an opinion of counsel reasonably satisfactory to the Company to the effect that such legend is no longer required under the Securities Act and applicable state securities laws.
(d) Irrevocable Transfer Agent Instructions. The Company shall issue the Irrevocable Transfer Agent Instructions. The Company represents and warrants that no instruction other than the Irrevocable Transfer Agent Instructions referred to in this Section 4.1(d) (or instructions that are consistent therewith) will be given by the Company to its Transfer Agent in connection with this Agreement, and that the Shares and Warrant Shares shall otherwise be freely transferable on the books and records of the Company as and to the extent provided in this Agreement and the other Transaction Documents and applicable Law. The Company acknowledges that a breach by it of its obligations under this Section 4.1(d) will cause irreparable harm to a Purchaser. Accordingly, the Company acknowledges that the remedy at law for a breach of its obligations under this Section 4.1(d) may be inadequate and agrees, in the event of a breach or threatened breach by the Company of the provisions of this Section 4.1(d) that a Purchaser shall be entitled, in addition to all other available remedies, to an order and/or injunction restraining any breach and requiring immediate issuance and transfer, without the necessity of showing irreparable harm or economic loss and without any bond or other security being required.
(e) Acknowledgement. Each Purchaser hereunder acknowledges its primary responsibilities under the Securities Act and accordingly will not sell or otherwise transfer the Securities or any interest therein without complying with the requirements of the Securities Act.
4.2. Furnishing of Information. In order to enable the Purchasers to sell the Shares and Warrant Shares under Rule 144, until such time as Purchaser may sell the Shares and Warrant Shares (assuming cashless exercise of the Pre-Funded Warrants) without limitation under Rule 144, the Company shall use its commercially reasonable efforts to timely file (or obtain extensions in respect thereof and file within the applicable grace period) all reports required to be filed by the Company after the date hereof pursuant to the Exchange Act and, if during such period, the Company is not required to file reports pursuant to the Exchange Act, it will prepare and furnish to the Purchasers and make publicly available in accordance with Rule 144(c) such information as is required for the Purchasers to sell the Shares and Warrant Shares under Rule 144.
4.3. Integration. The Company shall not, and shall use its commercially reasonable efforts to ensure that no Affiliate of the Company shall, sell, offer for sale or solicit offers to buy or otherwise negotiate in respect of any security (as defined in Section 2 of the Securities Act) that will be integrated with the offer or sale of the Securities in a manner that would require the registration under the Securities Act of the sale of the Securities to the Purchasers, or that will be integrated with the offer or sale of the Securities for purposes of the rules and regulations of any Trading Market such that it would require stockholder approval prior to the closing of such other transaction unless stockholder approval is obtained before the closing of such subsequent transaction.
4.4. Securities Laws Disclosure; Publicity. By no later than 9:00 A.M., New York City time, on the Trading Day immediately following the date hereof (provided that, if this Agreement is executed between midnight and 9:00 A.M., New York City time on any Trading Day, no later than 5:29 P.M. on the date hereof), the Company shall (a) issue a press release (the “Press Release”) reasonably acceptable to the Placement Agents disclosing all material terms of the transactions contemplated hereby and (b) file a Current Report on Form 8-K with the Commission describing the terms of the Transaction Documents (and including as exhibits to such Current Report on Form 8-K the material Transaction Documents (including, without limitation, this Agreement, the Registration Rights Agreement)); provided that the Press Release shall not publicly disclose the name of any Purchaser or investment adviser of any Purchaser or an Affiliate of any Purchaser without the prior written consent of such Purchaser, unless required by the rules and regulations of the Commission or Trading Market. In addition, notwithstanding the foregoing, the Company shall not publicly disclose the name of any Purchaser or investment adviser of any Purchaser, or include the name of any Purchaser or an Affiliate of any Purchaser without the prior written consent of such Purchaser (i) in any press release or marketing materials or (ii) in any filing with the Commission or any regulatory agency or Trading Market, except as required by U.S. federal securities law (A) in connection with any registration statement contemplated by the Registration Rights Agreement (which shall be subject to review and comment of the Purchasers pursuant to the terms of the Registration Rights Agreement) or the filing of final Transaction Documents (including signature pages thereto) with the Commission and (B) to the extent such disclosure is required by Law, request of the Commission’s staff or Trading Market regulations, in which case the Company shall provide the Purchasers with prior written notice of such disclosure permitted under this subclause (ii). From and after the issuance of the Press Release, no Purchaser shall be in possession of any material, non-public information received from the Company, any Subsidiary or any of their respective officers, directors, employees or agents, that is not disclosed in the Press Release. Each Purchaser, severally and not jointly with the other Purchasers, covenants that until such time as the transactions contemplated by this Agreement are publicly disclosed by the Company pursuant to the Press Release, such Purchaser will maintain the confidentiality of the existence and terms of this transaction and the information provided in connection therewith; provided, however, that any disclosure may be made by the Purchaser to the Purchaser’s representatives or agents, including, but not limited to, the Purchaser’s legal, tax and investment advisors.
4.5. Shareholder Rights Plan. No claim will be made or enforced by the Company or, with the consent of the Company, any other Person, that any Purchaser is an “Acquiring Person” under any control share acquisition, business combination, poison pill (including any distribution under a rights agreement) or similar anti-takeover plan or arrangement or Law in effect or hereafter adopted by the Company, or that any Purchaser could be deemed to trigger the provisions of any such plan or arrangement, in either case solely by virtue of receiving Securities under the Transaction Documents.
4.6. Non-Public Information. Except with respect to the material terms and conditions of the transactions contemplated by the Transaction Documents, including this Agreement, or as expressly required by any applicable securities law, the Company covenants and agrees that neither it, nor any other Person acting on its behalf, will provide any Purchaser or its agents or counsel with any information regarding the Company that the Company believes constitutes material non-public information without the express written (email being sufficient) consent of such Purchaser, (i) unless prior thereto such Purchaser shall have committed to customary obligations regarding the confidentiality and use of such information and (ii) except in the case of material, nonpublic information provided to an observer of the Board of Directors or member of the Board of Directors who is affiliated with such Purchaser. The Company understands and confirms that each Purchaser shall be relying on the foregoing covenant in effecting transactions in securities of the Company.
4.7. Use of Proceeds. The Company shall use the net proceeds from the sale of the Securities hereunder for working capital and general corporate purposes.
4.8. Principal Trading Market Listing. The Company shall use its reasonable best efforts to take all steps necessary to cause the Shares and Warrant Shares to be approved for listing on the Principal Trading Market as promptly as possible.
4.9. Blue Sky. The Company, on or before the Closing Date, shall take such action as the Company shall reasonably determine is necessary in order to obtain an exemption for or to qualify the Securities for sale to the Purchasers under applicable securities or “blue sky” Laws of the states of the United States (or to obtain an exemption from such qualification) and shall provide evidence of such actions promptly upon the written request of any Purchaser.
4.10. Short Sales After the Date Hereof. Such Purchaser shall not engage, directly or indirectly, in any transactions in the Company’s securities (including, without limitation, any Short Sales involving the Company’s securities) during the period from the date hereof until the earlier of such time as (i) the transactions contemplated by this Agreement are first publicly announced as required by and described in Section 4.4 or (ii) this Agreement is terminated in full pursuant to Section 6.19.
Notwithstanding the foregoing, no Purchaser makes any representation, warranty or covenant hereby that it will not engage in Short Sales in the securities of the Company after the time that the transactions contemplated by this Agreement are first publicly announced as described in Section 4.4; provided, however, each Purchaser agrees, severally and not jointly with any Purchasers, that they will not enter into any Net Short Sales (as hereinafter defined) from the period commencing on the Closing Date and ending on the earliest of (x) the Effective Date, (y) the twenty-four (24) month anniversary of the Closing Date or (z) the date that such Purchaser no longer holds any Securities. For purposes of this Section 4.10, a “Net Short Sale” by any Purchaser shall mean a sale of Common Stock by such Purchaser that is marked by such Purchaser as a short sale and that is made at a time when there is no equivalent offsetting long position in Common Stock held by such Purchaser. Notwithstanding the foregoing, in the event that a Purchaser is a multi-managed investment vehicle whereby separate portfolio managers manage separate portions of such Purchaser’s assets and the portfolio managers have no direct knowledge of the investment decisions made by the portfolio managers managing other portions of such Purchaser’s assets, the representation set forth above shall apply only with respect to the portion of assets managed by the portfolio manager that have knowledge about the financing transaction contemplated by this Agreement.
4.11. Indemnification of Purchasers. Subject to the provisions of this Section 4.11, the Company will indemnify and hold each Purchaser and its directors, officers, shareholders, members, partners, employees, investment advisers and agents (and any other Persons with a functionally equivalent role of a Person holding such titles notwithstanding a lack of such title or any other title), each Person who controls such Purchaser (within the meaning of Section 15 of the Securities Act and Section 20 of the Exchange Act), and the directors, officers, shareholders, agents, members, partners, investment advisers or employees (and any other Persons with a functionally equivalent role of a Person holding such titles notwithstanding a lack of such title or any other title) of such controlling persons (each, a “Purchaser Party”) harmless from any and all losses, liabilities, obligations, claims, contingencies, damages, costs and expenses, including all judgments, amounts paid in settlements, court costs and reasonable attorneys’ fees and costs of investigation that any such Purchaser Party may suffer or incur as a result of or relating to (i) any breach of any of the representations, warranties, covenants or agreements made by the Company in this Agreement or in the other Transaction Documents or (ii) any Action instituted against a Purchaser in any capacity, or any Purchaser Party, by any stockholder of the Company who is not an Affiliate of such Purchaser seeking indemnification, with respect to any of the transactions contemplated by the Transaction Documents (unless such Action is based upon a breach of such Purchaser’s representations, warranties or covenants under the Transaction Documents, or any agreements or understandings such Purchaser may have with any such stockholder or any violations by the Purchaser of state or federal securities Laws or any conduct by such Purchaser which constitutes fraud, gross negligence, willful misconduct or malfeasance). Promptly after receipt by any such Person (the “Indemnified Person”) of notice of any demand, claim or circumstances that would or may give rise to a claim or the commencement of any Proceeding or investigation in respect of which indemnity may be sought pursuant to this Section 4.11, such Indemnified Person shall promptly notify the Company in writing and the Company shall assume the defense thereof, including the employment of counsel reasonably satisfactory to such Indemnified Person, and shall assume the payment of all fees and expenses relating to such Proceeding or investigation; provided, however, that the failure of any Indemnified Person so to notify the Company shall not relieve the Company of its obligations hereunder except to the extent that the Company is actually and materially prejudiced by such failure to notify. In any such proceeding, any Indemnified Person shall have the right to retain its own counsel, but the fees and expenses of such counsel shall be at the expense of such Indemnified Person unless: (i) the Company and the Indemnified Person shall have mutually agreed to the retention of such counsel; (ii) the Company shall have failed promptly to assume the defense of such proceeding and to employ counsel reasonably satisfactory to such Indemnified Person in such proceeding; or (iii) in the reasonable judgment of counsel to such Indemnified Person, representation of both parties by the same counsel would be inappropriate due to actual or potential differing interests between them. In the event of the circumstances described in the foregoing clause (iii), if the Indemnified Person notifies the Company in writing that such Indemnified Person elects to employ separate counsel at the expense of the Company, then the Company shall not have the right to assume the defense of such claim on behalf of such Indemnified Person. The Company shall not be liable for any settlement of any proceeding effected without its prior written consent, which consent shall not be unreasonably withheld, delayed or conditioned or to the extent fees or costs incurred pursuant to this Section 4.11 are attributable to the Indemnified Person’s breach of any of the representations, warranties, covenants or agreements made by the Purchasers in this Agreement or the other Transaction Documents. Without the prior written consent of the Indemnified Person, which consent shall not be unreasonably withheld, delayed or conditioned, the Company shall not effect any settlement of any pending or threatened proceeding in respect of which any Indemnified Person is or could have been a party and indemnity could have been sought hereunder by such Indemnified Person, unless such settlement includes an unconditional release of such Indemnified Person from all liability arising out of such proceeding.
4.12. Equal Treatment of Purchasers. No consideration shall be offered or paid to any Purchaser to amend or consent to a waiver or modification of any provision of this Agreement unless the same consideration is also offered to all of the Purchasers. For clarification purposes, this provision constitutes a separate right granted to each Purchaser by the Company and negotiated separately by each Purchaser and shall not in any way be construed as the Purchasers acting in concert or as a group with respect to the purchase, disposition or voting of shares of Common Stock or otherwise.
4.13. Acknowledgements Regarding Placement Agents.
(a) Each Purchaser acknowledges that each of the Placement Agents is acting as a placement agent on a “best efforts” basis for the Securities being offered hereby and will be compensated by the Company for acting in such capacity. Each Purchaser represents that such Purchaser was contacted regarding the sale of the Securities by a Placement Agent or Merger Partner(or an authorized agent or representative thereof) with whom the Purchaser entered into a verbal or written confidentiality agreement.
(b) Each Purchaser represents that it is making this investment based on the results of its own due diligence investigation of the Company, and has not relied on any information or advice furnished by or on behalf of either of the Placement Agents in connection with the transactions contemplated hereby. Each Purchaser acknowledges that neither of the Placement Agents has made, and will not make, any representations and warranties with respect to the Company or the transactions contemplated hereby, and the Purchaser will not rely on any statements made by either of the Placement Agents, orally or in writing, to the contrary.
4.14 Lock-Up Agreements. The Company shall not consent or agree to amend, alter, waive or otherwise modify the terms of any of the Lock-Up Agreements (as defined in the Merger Agreement) without the consent of the Placement Agents.
ARTICLE V
CONDITIONS PRECEDENT TO CLOSING
5.1. Conditions Precedent to the Obligations of the Purchasers to Purchase Shares and/or Pre-Funded Warrants. The obligation of each Purchaser to acquire Shares and/or Pre-Funded Warrants at the Closing is subject to the fulfillment to such Purchaser’s satisfaction, on or prior to the Closing Date, of each of the following conditions, any of which may be waived by such Purchaser (as to itself only):
(a) Representations and Warranties. The representations and warranties of the Company contained herein shall be (i) true and correct in all material respects as of the date hereof and (ii) true and correct in all material respects as of the Closing Date (except in the case of this clause (ii), where the failure to be true and correct in all material respects (without regard to any materiality or Material Adverse Effect qualifications contained therein), individually or in the aggregate, has not had, and is not reasonably likely to have, a Material Adverse Effect), as though made on and as of such date, except for such representations and warranties that speak as of a specific date, which shall be true and correct in all material respects as of such date.
(b) Performance. The Company shall have performed, satisfied and complied in all material respects with all covenants, agreements and conditions required by the Transaction Documents to be performed, satisfied or complied with by it at or prior to the Closing.
(c) No Injunction. No statute, rule, regulation, order, executive order, decree, judgment, writ, order, ruling or injunction shall have been enacted, entered, promulgated, issued or endorsed by any court of competent jurisdiction or any Governmental Authority that enjoins, prevents or prohibits the consummation of any of the transactions contemplated by the Transaction Documents.
(d) Closing of Merger. All conditions precedent to the consummation of the Merger as set forth in the Merger Agreement shall have been satisfied or waived by the party entitled to the benefit thereof under the Merger Agreement, but subject to satisfaction or waiver by such party of such conditions as of the consummation of the Merger, and the Company Authorized Stock Increase shall have occurred.
(e) Consents. The Company shall have obtained in a timely fashion any and all consents, permits, approvals, registrations and waivers necessary for consummation of the purchase and sale of the Securities, all of which shall be and remain so long as necessary in full force and effect.
(f) Adverse Changes. Since the date hereof, no event or series of events shall have occurred that has had or would reasonably be expected to have a Material Adverse Effect.
(g) No Suspensions of Trading in Common Stock; No Stop Orders; Listing. The Common Stock shall not have been suspended by the Commission or the Principal Trading Market from trading on the Principal Trading Market nor shall suspension by the Commission or the Principal Trading Market have been threatened, either (i) in writing by the Commission or the Principal Trading Market or (ii) by falling below the minimum listing maintenance requirements of the Principal Trading Market. No stop order shall have been imposed by Nasdaq, the Commission or any other Governmental Authority or regulatory body with respect to public trading in the Common Stock. The Company shall have filed the Notification Form: Listing of Additional Shares for the listing of the Shares and Warrant Shares with Nasdaq, and Nasdaq shall have raised no objection to such notice and the transactions contemplated hereby.
(h) Company Deliverables. The Company shall have delivered the Company Deliverables in accordance with Section 2.3(a).
(i) Compliance Certificate. The Company shall have delivered the Placement Agents, on behalf of each Purchaser, a certificate, dated as of the Closing Date and signed by its Chief Executive Officer and its Chief Financial Officer, certifying to the fulfillment of the conditions specified in Sections 5.1(a), 5.1(b), 5.1(e) and 5.1(f) in the form attached hereto as Exhibit G.
(j) Termination. This Agreement shall not have been terminated as to such Purchaser in accordance with Section 6.19 herein.
5.2. Conditions Precedent to the Obligations of the Company to issue Shares and/or Pre-Funded Warrants. The Company’s obligation to issue the Shares and/or Pre-Funded Warrants at the Closing to each Purchaser is subject to the fulfillment to the satisfaction of the Company on or prior to the Closing Date of the following conditions, any of which may be waived by the Company:
(a) Representations and Warranties. The representations and warranties made by each Purchaser in Section 3.2 hereof shall be true and correct in all material respects (except for those representations and warranties which are qualified as to materiality, in which case such representations and warranties shall be true and correct in all respects) as of the date when made, and as of the Closing Date as though made on and as of such date, except for representations and warranties that speak as of a specific date, which shall be true and correct in all material respects (except for those representations and warranties which are qualified as to materiality, which representations and warranties shall be true and correct in all material respects) as of such date.
(b) Performance. Such Purchaser shall have performed, satisfied and complied in all material respects with all covenants, agreements and conditions required by the Transaction Documents to be performed, satisfied or complied with by such Purchaser at or prior to the Closing Date.
(c) No Injunction. No statute, rule, regulation, order, executive order, decree, judgment, writ, order, ruling or injunction shall have been enacted, entered, promulgated, issued or endorsed by any court of competent jurisdiction or any Governmental Authority that enjoins, prevents or prohibits the consummation of any of the transactions contemplated by the Transaction Documents.
(d) Purchasers Deliverables. Such Purchaser shall have delivered its Purchaser Deliverables in accordance with Section 2.3(b).
(e) Closing of Merger. All conditions precedent to the consummation of the Merger as set forth in the Merger Agreement shall have been satisfied or waived by the party entitled to the benefit thereof under the Merger Agreement (other than the conditions in Section 7.3(e) of the Merger Agreement and those conditions that may only be satisfied at the consummation of the Merger, but subject to satisfaction or waiver by such party of such conditions as of the consummation of the Merger) and the Company Authorized Stock Increase shall have occurred.
(f) Termination. This Agreement shall not have been terminated as to such Purchaser in accordance with Section 6.19 herein.
ARTICLE VI
MISCELLANEOUS
6.1. Convertible Note Purchasers. At any time prior to the Closing Date, each of the holders of Convertible Notes (in the principal amount not to exceed $20,000,000 in the aggregate) (each, a “Convertible Note Purchaser”) may become a party to this Agreement and the Company shall sell any such Shares and/or Pre-Funded Warrants on the same terms and conditions as those contained in this Agreement, provided that each Convertible Note Purchaser becomes a party to this Agreement by executing and delivering a counterpart signature page. Notwithstanding anything to the contrary set forth herein, any representation, warranty or covenant of the Purchasers that speaks to the date hereof, shall be deemed to speak as of the date that each Convertible Note Purchaser becomes a party to this Agreement with respect to each such Convertible Note Purchaser. Exhibit A to this Agreement shall be updated accordingly, which update shall not be deemed to be an amendment to this Agreement.
6.2. Fees and Expenses. The Company and the Purchasers shall each pay the fees and expenses of their respective advisers, counsel, accountants and other experts, if any, and all other expenses incurred by such party in connection with the negotiation, preparation, execution, delivery and performance of this Agreement. The Company shall pay all Transfer Agent fees, stamp Taxes and other Taxes and duties levied in connection with the issuance and sale of the Shares and/or Pre-Funded Warrants to the Purchasers.
6.3. Entire Agreement. The Transaction Documents, together with the exhibits and schedules thereto, contain the entire understanding of the parties with respect to the subject matter hereof and thereof and supersede all prior agreements, understandings, discussions and representations, oral or written, with respect to such matters, which the parties acknowledge have been merged into such documents, exhibits and schedules. Before or at the Closing, the Company and the Purchasers will execute and deliver to the other such further documents as may be reasonably requested in order to give practical effect to the intention of the parties under the Transaction Documents.
6.4. Notices. Any and all notices or other communications or deliveries required or permitted to be provided hereunder shall be in writing and shall be deemed given and effective on the earliest of (a) the date of transmission, if such notice or communication is delivered via electronic mail at the e-mail address specified in this Section 6.4 prior to 5:00 P.M., New York City time, on a Trading Day, (b) the next Trading Day after the date of transmission, if such notice or communication is delivered via electronic mail at the e-mail address specified in this Section 6.4 on a day that is not a Trading Day or later than 5:00 P.M., New York City time, on any Trading Day, (c) the Trading Day following the date of mailing, if sent by U.S. nationally recognized overnight courier service with next day delivery specified, or (d) upon actual receipt by the party to whom such notice is required to be given if delivered personally or if sent by U.S. certified or registered mail, return receipt requested; provided, in the case of clauses (a) and (b), that notice shall not be deemed given or effective if the sender receives an automatic system-generated response that such electronic mail was undeliverable. The address for such notices and communications shall be as follows:
|
If to the Company: |
Pieris Pharmaceuticals, Inc. 255 Franklin Street, 26th Floor Boston, MA 02110 Attention: Stephen Yoder, Tom Bures, Karam Hijji |
|
With a copy (which shall not constitute notice) to: |
Mintz, Levin, Cohn, Ferris, Glovsky and Popeo, P.C. One Financial Center Boston, MA 02111 Attention: William C. Hicks, Marc D. Mantell, Scott Dunberg |
|
If to a Purchaser: |
To the address set forth under such Purchaser’s name on Exhibit A hereto; |
|
or such other address as may be designated in writing hereafter, in the same manner, by such Person. |
6.5. Amendments; Waivers; No Additional Consideration. No provision of this Agreement may be waived, modified, supplemented or amended except in a written instrument signed, in the case of an amendment, by the Company and the Purchasers who collectively have subscribed to purchase at least a majority in interest of the Shares and Warrant Shares, provided that (i) no amendment to Section 4.4, Section 4.6, Section 4.10, Section 4.11, Section 4.12, this Section 6.5 or Section 6.19 may be made without the consent of each Purchaser, or, in the case of a waiver, by the party against whom enforcement of any such waiver is sought and (ii) any proposed amendment that would, by its terms, have a disproportionate and materially adverse effect on any Purchaser shall require the consent of such Purchaser(s). No waiver of any default with respect to any provision, condition or requirement of this Agreement shall be deemed to be a continuing waiver in the future or a waiver of any subsequent default or a waiver of any other provision, condition or requirement hereof, nor shall any delay or omission of either party to exercise any right hereunder in any manner impair the exercise of any such right. No consideration shall be offered or paid to any Purchaser to amend or consent to a waiver or modification of any provision of any Transaction Document unless the same consideration is also offered to all Purchasers who then hold Securities.
6.6. Construction. The headings herein are for convenience only, do not constitute a part of this Agreement and shall not be deemed to limit or affect any of the provisions hereof. The language used in this Agreement will be deemed to be the language chosen by the parties to express their mutual intent, and no rules of strict construction will be applied against any party. This Agreement shall be construed as if drafted jointly by the parties, and no presumption or burden of proof shall arise favoring or disfavoring any party by virtue of the authorship of any provisions of this Agreement or any of the Transaction Documents. As used in this Agreement, the words “include” and “including,” and variations thereof, shall not be deemed to be terms of limitation, but rather shall be deemed to be followed by the words “without limitation.”
6.7. Successors and Assigns. The provisions of this Agreement shall inure to the benefit of and be binding upon the parties and their successors and permitted assigns. This Agreement, or any rights or obligations hereunder, may not be assigned by the Company without the prior written consent of each Purchaser. Any Purchaser may assign its rights hereunder in whole or in part to any Person to whom such Purchaser assigns or transfers any Securities in compliance with the Transaction Documents and applicable Law, provided such transferee shall agree in writing to be bound, with respect to the transferred Securities, by the terms and conditions of this Agreement that apply to the Purchasers.
6.8. No Third-Party Beneficiaries. This Agreement is intended for the benefit of the parties hereto and their respective successors and permitted assigns and is not for the benefit of, nor may any provision hereof be enforced by, any other Person, except (i) the Placement Agents are intended third-party beneficiaries of the representations and warranties in Article III and Article IV, and of this Section 6.8 and Section 6.20 and (ii) the Purchaser Parties are intended beneficiaries of Section 4.11.
6.9. Governing Law. All questions concerning the construction, validity, enforcement and interpretation of this Agreement shall be governed by and construed and enforced in accordance with the internal laws of the State of New York, without regard to the principles of conflicts of law thereof. Each party agrees that all Proceedings concerning the interpretations, enforcement and defense of the transactions contemplated by this Agreement and any other Transaction Documents (whether brought against a party hereto or its respective Affiliates, employees or agents) shall be commenced exclusively in the New York Courts. Each party hereto hereby irrevocably submits to the exclusive jurisdiction of the New York Courts for the adjudication of any dispute hereunder or in connection herewith or with any transaction contemplated hereby or discussed herein (including with respect to the enforcement of any of the Transaction Documents), and hereby irrevocably waives, and agrees not to assert in any Proceeding, any claim that it is not personally subject to the jurisdiction of any such New York Court, or that such Proceeding has been commenced in an improper or inconvenient forum. Each party hereto hereby irrevocably waives personal service of process and consents to process being served in any such Proceeding by mailing a copy thereof via registered or certified mail or overnight delivery (with evidence of delivery) to such party at the address in effect for notices to it under this Agreement and agrees that such service shall constitute good and sufficient service of process and notice thereof. Nothing contained herein shall be deemed to limit in any way any right to serve process in any manner permitted by law. EACH PARTY HERETO HEREBY IRREVOCABLY WAIVES, TO THE FULLEST EXTENT PERMITTED BY APPLICABLE LAW, ANY AND ALL RIGHT TO TRIAL BY JURY IN ANY LEGAL PROCEEDING ARISING OUT OF OR RELATING TO THIS AGREEMENT OR THE TRANSACTIONS CONTEMPLATED HEREBY.
6.10. Survival. Subject to applicable statute of limitations, the representations, warranties, agreements and covenants contained herein shall survive the Closing and the delivery of the Shares and/or Pre-Funded Warrants.
6.11. Execution. This Agreement may be executed in two or more counterparts, all of which when taken together shall be considered one and the same agreement and shall become effective when counterparts have been signed by each party and delivered to the other party, it being understood that both parties need not sign the same counterpart. In the event that any signature is delivered by facsimile transmission, or by e-mail delivery of a “.pdf” format data file, or by any electronic signature complying with the U.S. ESIGN Act of 2000 or the New York Electronic Signatures and Records Act, such signature shall create a legally valid and binding obligation of the party executing (or on whose behalf such signature is executed) with the same force and effect as if such facsimile or “.pdf” signature page were an original thereof.
6.12. Severability. If any provision of this Agreement is held to be invalid or unenforceable in any respect, the validity and enforceability of the remaining terms and provisions of this Agreement shall not in any way be affected or impaired thereby and the parties will attempt to agree upon a valid and enforceable provision that is a reasonable substitute therefor, and upon so agreeing, shall incorporate such substitute provision in this Agreement.
6.13. Rescission and Withdrawal Right. Notwithstanding anything to the contrary contained in (and without limiting any similar provisions of) the Transaction Documents, whenever any Purchaser exercises a right, election, demand or option under a Transaction Document and the Company does not timely perform its related obligations within the periods therein provided, then such Purchaser may rescind or withdraw, in its sole discretion from time to time upon written notice to the Company, any relevant notice, demand or election in whole or in part without prejudice to its future actions and rights.
6.14. Replacement of Securities. If any certificate or instrument evidencing any Shares Securities is mutilated, lost, stolen or destroyed, the Company may issue or cause to be issued in exchange and substitution for and upon cancellation thereof, or in lieu of and substitution therefor, a new certificate or instrument, but only upon receipt of evidence reasonably satisfactory to the Company and the Transfer Agent of such loss, theft or destruction and the execution by the holder thereof of a customary lost certificate affidavit of that fact and an agreement to indemnify and hold harmless the Company and the Transfer Agent for any losses in connection therewith or, if required by the Transfer Agent, a bond in such form and amount as is required by the Transfer Agent. The applicants for a new certificate or instrument under such circumstances shall also pay any reasonable third-party costs associated with the issuance of such replacement Securities. If a replacement certificate or instrument evidencing any Securities is requested due to a mutilation thereof, the Company may require delivery of such mutilated certificate or instrument as a condition precedent to any issuance of a replacement.
6.15. Remedies. In addition to being entitled to exercise all rights provided herein or granted by Law, including recovery of damages, each of the Purchasers and the Company will be entitled to specific performance under the Transaction Documents, without the requirement of posting a bond. The parties agree that monetary damages may not be adequate compensation for any loss incurred by reason of any breach of obligations described in the foregoing sentence and hereby agree to waive in any action for specific performance of any such obligation (other than in connection with any action for a temporary restraining order) the defense that a remedy at law would be adequate.
6.16. Payment Set Aside. To the extent that the Company makes a payment or payments to any Purchaser pursuant to any Transaction Document or a Purchaser enforces or exercises its rights thereunder, and such payment or payments or the proceeds of such enforcement or exercise or any part thereof are subsequently invalidated, declared to be fraudulent or preferential, set aside, recovered from, disgorged by or are required to be refunded, repaid or otherwise restored to the Company, a trustee, receiver or any other person under any Law (including, without limitation, any bankruptcy law, state or federal law, common law or equitable cause of action), then to the extent of any such restoration the obligation or part thereof originally intended to be satisfied shall be revived and continued in full force and effect as if such payment had not been made or such enforcement or setoff had not occurred.
6.17. Adjustments in Share Numbers and Prices. In the event of any stock split, subdivision, dividend or distribution payable in shares of Common Stock (or other securities or rights convertible into, or entitling the holder thereof to receive directly or indirectly shares of Common Stock), combination or other similar recapitalization or event occurring after the date hereof and prior to the Closing, each reference in any Transaction Document to a number of shares or a price per share shall be deemed to be amended to appropriately account for such event.
6.18. Independent Nature of Purchasers’ Obligations and Rights. The obligations of each Purchaser under any Transaction Document are several and not joint with the obligations of any other Purchaser, and no Purchaser shall be responsible in any way for the performance of the obligations of any other Purchaser under any Transaction Document. The decision of each Purchaser to purchase Securities pursuant to the Transaction Documents has been made by such Purchaser independently of any other Purchaser and independently of any information, materials, statements or opinions as to the business, affairs, operations, assets, properties, liabilities, results of operations, condition (financial or otherwise) or prospects of the Company or any Subsidiary which may have been made or given by any other Purchaser or by any agent or employee of any other Purchaser, and no Purchaser and any of its agents or employees shall have any liability to any other Purchaser (or any other Person) relating to or arising from any such information, materials, statement or opinions. Nothing contained herein or in any Transaction Document, and no action taken by any Purchaser pursuant thereto, shall be deemed to constitute the Purchasers as a partnership, an association, a joint venture or any other kind of entity, or create a presumption that the Purchasers are in any way acting in concert or as a group (including, without limitation, a “group” within the meaning of Section 13(d)(3) of the Exchange Act) with respect to such obligations or the transactions contemplated by the Transaction Documents. Each Purchaser acknowledges that no other Purchaser has acted as agent for such Purchaser in connection with making its investment hereunder and that no Purchaser will be acting as agent of such Purchaser in connection with monitoring its investment in the Securities or enforcing its rights under the Transaction Documents. Each Purchaser shall be entitled to independently protect and enforce its rights, including without limitation the rights arising out of this Agreement or out of the other Transaction Documents, and it shall not be necessary for any other Purchaser to be joined as an additional party in any proceeding for such purpose. Each Purchaser has been represented by its own separate legal counsel in its review and negotiation of the Transaction Documents. For reasons of administrative convenience only, Purchasers and their respective counsels may choose to communicate with the Company through Cooley LLP, counsel to the Placement Agents. Each Purchaser acknowledges that Cooley LLP has rendered legal advice to the Placement Agents and not to such Purchaser in connection with the transactions contemplated hereby, and that each such Purchaser has relied for such matters on the advice of its own respective counsel. The Company has elected to provide all Purchasers with the same terms and Transaction Documents for the convenience of the Company and not because it was required or requested to do so by any Purchaser. It is expressly understood that each provision contained in this Agreement is between the Company and a Purchaser, solely, and not between the Company and the Purchasers collectively and not between and among the Purchasers.
6.19. Termination. This Agreement may be terminated and the sale and purchase of the Securities abandoned at any time prior to the Closing (i) automatically if the Closing has not been consummated on or prior to 5:00 P.M., New York City time, on April 30, 2025, (ii) automatically if the Merger Agreement is terminated in accordance with its terms without the Merger being consummated or (iii) by a Purchaser (with respect to itself) if any of the conditions set forth in Section 5.1 shall have become incapable of fulfillment, and shall not have been waived by such Purchaser; provided, however, that the right to terminate this Agreement under clause (ii) shall not be available to any Person whose failure to comply with its obligations under this Agreement has been the cause of or resulted in the failure of the Closing to occur on or before such time. Nothing in this Section 6.19 shall be deemed to release any party from any liability for any breach by such party of the terms and provisions of this Agreement or the other Transaction Documents or to impair the right of any party to compel specific performance by any other party of its obligations under this Agreement or the other Transaction Documents. In the event of a termination pursuant to this Section 6.19, the Company shall promptly notify all non-terminating Purchasers. Upon a termination in accordance with this Section 6.19, the Company and the terminating Purchaser(s) shall not have any further obligation or liability (including arising from such termination) to the other, and no Purchaser will have any liability to any other Purchaser under the Transaction Documents as a result therefrom.
6.20. Exculpation of the Placement Agents. Each party hereto agrees for the express benefit of the Placement Agents, their Affiliates and their representatives that:
(a) Each Placement Agent is acting as placement agent for the Merger Partner solely in connection with the sale of the Securities and is not acting in any other capacity and is not and shall not be construed as a fiduciary for any Purchaser, or any other person or entity in connection with the sale of Securities.
(b) None of the Placement Agents nor any of their Affiliates or any of their respective representatives (i) shall be liable for any improper payment made in accordance with the information provided by the Company; (ii) has made or will make any representation or warranty, express or implied, of any kind or character, and has not provided any recommendation in connection with the purchase or sale of the Securities; (iii) has any responsibilities as to the validity, accuracy, completeness, value or genuineness, as of any date, of any information, certificates or documentation delivered by or on behalf of the Company pursuant to this Agreement, the other Transaction Documents, or in connection with any of the transactions contemplated by such agreements; or (iv) shall be liable or have any obligation (including, without limitation, for or with respect to any losses, claims, damages, obligations, penalties, judgments, awards, liabilities, costs, expenses or disbursements incurred by any Purchaser, the Company or any other Person or entity), whether in contract, tort or otherwise to any Purchaser or to any person claiming through such Purchaser, (A) for any action taken, suffered or omitted by any of them in good faith and reasonably believed to be authorized or within the discretion or rights or powers conferred upon it by this Agreement, any other Transaction Document, (B) for anything which any of them may do or refrain from doing in connection with this Agreement, any other Transaction Document, or (C) for anything otherwise in connection with the purchase and sale of the Securities, except in each case for such party’s own gross negligence or willful misconduct.
6.21. Waivers of Conflicts. Each party to this Agreement acknowledges that Company Counsel and/or Nevada Counsel may have in the past performed and may continue to perform legal services to one or more Purchasers or their affiliates in matters unrelated to the transactions described in this Agreement, including the representation of one or more Purchasers or their affiliates in matters of a similar nature to such transactions. Accordingly, except as otherwise agreed between the Company and a Purchaser, each party to this Agreement hereby (a) acknowledges that they have had an opportunity to ask for information relevant to this disclosure; and (b) gives its informed consent to representation by Company Counsel and/or Nevada Counsel of (i) one or more Purchasers or their affiliates in such unrelated matters and (ii) the Company in connection with this Agreement and the transactions contemplated hereby.
6.22. Arm’s Length Transaction. The Company acknowledges and agrees that (i) the transactions described in this Agreement are an arm’s-length commercial transaction between the parties, (ii) the Purchasers have not assumed nor will they assume an advisory or fiduciary responsibility in the Company’s favor with respect to any of the transactions contemplated by this Agreement or the process leading thereto, and the Purchasers have no obligation to the Company with respect to the transactions contemplated by this Agreement except those obligations expressly set forth in this Agreement or the other Transaction Documents to which they are a party, and (iii) the Company’s decision to enter into the Transaction Documents has been based solely on the independent evaluation by the Company and its representatives.
[REMAINDER OF PAGE INTENTIONALLY LEFT BLANK]
IN WITNESS WHEREOF, the parties hereto have caused this Securities Purchase Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
|
|
PIERIS PHARMACEUTICALS, INC. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
By: |
/s/ Stephen S. Yoder |
|
|
|
Name: Stephen S. Yoder Title: Chief Executive Officer |
|
|
IN WITNESS WHEREOF, the parties hereto have caused this Securities Purchase Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
|
|
PURCHASER:
Frazier Life Sciences Public Fund, L.P. |
|
|
|
|
|
|
|
| By: | FHMLSP, L.P. | ||
| Its: | General Partner | ||
| By: | FHMLSP, L.L.C. | ||
| Its: | General Partner | ||
|
|
|
|
|
|
|
By: |
/s/ Albert Cha |
|
|
|
Name: Albert Cha Title: Managing Director
Beneficial Ownership Limitation: 4.99% |
|
|
IN WITNESS WHEREOF, the parties hereto have caused this Securities Purchase Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
|
|
PURCHASER:
Frazier Life Sciences XI, L.P. |
|
|
|
|
|
|
|
| By: | FHMLS XI, L.P. | ||
| Its: | General Partner | ||
| By: | FHMLS XI, L.L.C. | ||
| Its: | General Partner | ||
|
|
|
|
|
|
|
By: |
/s/ James N. Topper |
|
|
|
Name: James N. Topper Title: Managing Director |
|
|
|
|
|
|
|
| Beneficial Ownership Limitation: 4.99% | |||
IN WITNESS WHEREOF, the parties hereto have caused this Securities Purchase Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
|
|
PURCHASER:
Frazier Life Sciences X, L.P. |
|
|
|
|
|
|
|
| By: | FHMLS X, L.P. | ||
| Its: | General Partner | ||
| By: | FHMLS X, L.L.C. | ||
| Its: | General Partner | ||
|
|
|
|
|
|
|
By: |
/s/ James N. Topper |
|
|
|
Name: James N. Topper Title: Managing Director |
|
|
|
|
|
|
|
| Beneficial Ownership Limitation: 4.99% | |||
IN WITNESS WHEREOF, the parties hereto have caused this Securities Purchase Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
|
|
PURCHASER:
Frazier Life Sciences Public Overage Fund, L.P. |
|
|
|
|
|
|
|
| By: | FHMLSP Overage, L.P. | ||
| Its: | General Partner | ||
| By: | FHMLSP Overage, L.L.C. | ||
| Its: | General Partner | ||
|
|
|
|
|
|
|
By: |
/s/ Albert Cha |
|
|
|
Name: Albert Cha Title: Managing Director |
|
|
|
|
|
|
|
| Beneficial Ownership Limitation: 4.99% | |||
IN WITNESS WHEREOF, the parties hereto have caused this Securities Purchase Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
|
|
PURCHASER:
Biotechnology Value Fund, L.P. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
By: |
/s/ Mark Lampert |
|
|
|
Name: Mark Lampert Title: Chief Executive Officer of BVF I GP LLC, General Partner of Biotechnology Value Fund, L.P. |
|
|
|
|
|
|
|
| Beneficial Ownership Limitation: 9.99% | |||
IN WITNESS WHEREOF, the parties hereto have caused this Securities Purchase Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
|
|
PURCHASER:
Biotechnology Value Fund II, L.P. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
By: |
/s/ Mark Lampert |
|
|
|
Name: Mark Lampert Title: Chief Executive Officer of BVF II GP LLC, General Partner of Biotechnology Value Fund II, L.P. |
|
|
|
|
|
|
|
| Beneficial Ownership Limitation: 9.99% | |||
IN WITNESS WHEREOF, the parties hereto have caused this Securities Purchase Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
|
|
PURCHASER:
Biotechnology Value Trading Fund OS LP |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
By: |
/s/ Mark Lampert |
|
|
|
Name: Mark Lampert Title: President of BVF Inc., General Partner of BVF Partners L.P., Sole Member of BVF Partners OS Ltd., General Partner of Biotechnology Value Trading Fund OS LP |
|
|
|
|
|
|
|
| Beneficial Ownership Limitation: 9.99% | |||
IN WITNESS WHEREOF, the parties hereto have caused this Securities Purchase Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
|
|
PURCHASER:
MSI BVF SPV, LLC |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
By: |
/s/ Mark Lampert |
|
|
|
Name: Mark Lampert Title: President of BVF Inc., General Partner of BVF Partners L.P., Attorney in Fact of MSI BVF SPV, LLC |
|
|
|
|
|
|
|
| Beneficial Ownership Limitation: 9.99% | |||
IN WITNESS WHEREOF, the parties hereto have caused this Securities Purchase Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
|
|
PURCHASER:
Averill Master Fund, Ltd. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
By: |
/s/ Andrew Nathanson |
|
|
|
Name: Andrew Nathanson Title: Authorized Signatory |
|
|
|
|
|
|
|
| Beneficial Ownership Limitation: 9.99% | |||
IN WITNESS WHEREOF, the parties hereto have caused this Securities Purchase Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
|
|
PURCHASER:
BLUE OWL HEALTHCARE OPPORTUNITIES IV PUBLIC INVESTMENTS LP |
|
|
|
|
|
|
|
| By: Blue Owl Healthcare Opportunities GP IV LLC, its general partner | |||
|
|
|
|
|
|
|
By: |
/s/ Kevin Raidy |
|
|
|
Name: Kevin Raidy Title: Authorized Signatory |
|
|
|
|
|
|
|
| Beneficial Ownership Limitation (Select One): | |||
| ___ 4.99% | |||
| ___ 9.99% | |||
| X N/A | |||
IN WITNESS WHEREOF, the parties hereto have caused this Securities Purchase Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
|
|
PURCHASER:
NCP RFM LP |
|
|
|
|
|
|
|
| By: Nantahala Capital Management, LLC, | |||
| Its Investment Manager | |||
|
|
|
|
|
|
|
By: |
/s/ Wilmot Harkey |
|
|
|
Name: Wilmot Harkey Title: Manager |
|
|
|
|
|
|
|
| Beneficial Ownership Limitation (Select One): | |||
| ___ 4.99% | |||
| X 9.99% | |||
| N/A | |||
|
|
BLACKWELL PARTNERS LLC - SERIES A, solely with respect to the portion of its assets for which Nantahala Capital Management, LLC acts as its Investment Manager |
|
|
|
|
|
|
|
| By: Nantahala Capital Management, LLC, | |||
| Its Investment Manager | |||
|
|
|
|
|
|
|
By: |
/s/ Wilmot Harkey |
|
|
|
Name: Wilmot Harkey Title: Manager |
|
|
|
|
|
|
|
| Beneficial Ownership Limitation (Select One): | |||
| ___ 4.99% | |||
| X 9.99% | |||
| N/A | |||
|
|
NANTAHALA CAPITAL PARTNERS LIMITED PARTNERSHIP |
|
|
|
|
|
|
|
| By: Nantahala Capital Management, LLC, | |||
| Its Investment Manager | |||
|
|
|
|
|
|
|
By: |
/s/ Wilmot Harkey |
|
|
|
Name: Wilmot Harkey Title: Manager |
|
|
|
|
|
|
|
| Beneficial Ownership Limitation (Select One): | |||
| ___ 4.99% | |||
| X 9.99% | |||
| N/A | |||
IN WITNESS WHEREOF, the parties hereto have caused this Securities Purchase Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
|
|
PURCHASER:
SAMSARA BIOCAPITAL, L.P. |
|
|
|
|
|
|
|
|
|
By: |
/s/ Srinivas Akkaraju, MD, PhD |
|
|
|
Name: Srinivas Akkaraju, MD, PhD Title: Managing Director of Samsara BioCapital GP, LLC, General Partner of Samsara BioCapital, L.P. |
|
|
|
|
|
|
|
|
Beneficial Ownership Limitation 9.99% |
|||
IN WITNESS WHEREOF, the parties hereto have caused this Securities Purchase Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
|
|
PURCHASER:
CDK ASSOCIATES, L.L.C. |
|
|
|
|
|
|
|
|
|
By: |
/s/ Karen Cross |
|
|
|
Name: Karen Cross Title: Treasurer |
|
|
|
|
|
|
|
|
Beneficial Ownership Limitation 4.99% |
|||
IN WITNESS WHEREOF, the parties hereto have caused this Securities Purchase Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
|
|
PURCHASER:
THIRD STREET HOLDINGS LLC |
|
|
|
|
|
|
|
|
|
By: |
/s/ Peter P. D’Angelo |
|
|
|
Name: Peter P. D’Angelo Title: Manager of Managing Member |
|
|
|
|
|
|
|
|
Beneficial Ownership Limitation (Select One): 4.99% |
|||
IN WITNESS WHEREOF, the parties hereto have caused this Securities Purchase Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
|
|
PURCHASER:
ADARI PARTNERS, LP |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
By: |
/s/ Daniel Schneeberger |
|
|
|
Name: Daniel Schneeberger Title: General Partner |
|
|
|
|
|
|
|
| Beneficial Ownership Limitation (Select One): | |||
| X 4.99% | |||
| ___ 9.99% | |||
| N/A | |||
IN WITNESS WHEREOF, the parties hereto have caused this Securities Purchase Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
|
|
PURCHASER:
SPEARHEAD INSURANCE SOLUTIONS IDF, LLC – SERIES ADAR1 |
|
|
|
By: Spearhead IDF Partners, LLC, the Manager of Spearhead Insurance Solutions IDF, LLC – Series ADAR1 |
|||
|
|
|
|
|
|
|
By: |
/s/ Ken Foley |
|
|
|
Name: Ken Foley Title: Manager of Spearhead IDF Partners, LLC |
|
|
|
|
|
|
|
| Beneficial Ownership Limitation (Select One): | |||
| X 4.99% | |||
| ___ 9.99% | |||
| N/A | |||
IN WITNESS WHEREOF, the parties hereto have caused this Securities Purchase Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
|
|
PURCHASER:
DAFNA LIFESCIENCE LP |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
By: |
/s/ Fariba Ghodsian |
|
|
|
Name: Fariba Ghodsian Title: C.I.O. |
|
|
|
|
|
|
|
|
Beneficial Ownership Limitation
4.99% |
|||
IN WITNESS WHEREOF, the parties hereto have caused this Securities Purchase Agreement to be duly executed by their respective authorized signatories as of the date first indicated above.
|
|
PURCHASER:
DAFNA LIFESCIENCE SELECT LP |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
By: |
/s/ Fariba Ghodsian |
|
|
|
Name: Fariba Ghodsian Title: C.I.O. |
|
|
|
|
|
|
|
|
Beneficial Ownership Limitation
4.99% |
|||
EXHIBIT A
SCHEDULE OF PURCHASERS
|
Purchaser Name |
Shares |
Warrant Shares |
Aggregate Purchase Price |
Aggregate Cash Purchase Price |
Convertible Note Principal Amount |
|
Frazier Life Sciences Public Fund, L.P. |
245,565 |
1,147,500 |
$19,125,495.64 |
$19,125,495.64 |
- |
|
Frazier Life Sciences XI, L.P. |
1,541 |
7,200 |
$120,005.86 |
$120,005.86 |
- |
|
Frazier Life Sciences X, L.P. |
1,444 |
6,750 |
$112,496.05 |
$112,496.05 |
- |
|
Frazier Life Sciences Public Overage Fund, L.P. |
72,450 |
338,550 |
$5,642,650.35 |
$5,642,650.35 |
- |
|
Biotechnology Value Fund, L.P. |
- |
572,438 |
$7,858,944.06 |
$7,858,944.06 |
- |
|
Biotechnology Value Fund II, L.P. |
- |
454,472 |
$6,239,400.64 |
$6,239,400.64 |
- |
|
Biotechnology Value Trading Fund OS LP |
- |
45,643 |
$626,628.18 |
$626,628.18 |
- |
|
MSI BVF SPV, LLC |
- |
20,032 |
$275,017.32 |
$275,017.32 |
- |
|
Averill Master Fund, Ltd. |
728,337 |
- |
$9,999,994.18 |
$9,999,994.18 |
- |
|
BlueOwlHealthCareOpportunities IV PublicInvestments LP |
254,918 |
- |
$3,499,998.65 |
$3,499,998.65 |
- |
|
NCP RFM LP |
44,049 |
- |
$604,788.37 |
$604,788.37 |
- |
|
Blackwell Partners LLC - Series A |
156,087 |
- |
$2,143,058.90 |
$2,143,058.90 |
- |
|
Nantahala Capital Partners Limited Partnership |
54,782 |
- |
$752,151.38 |
$752,151.38 |
- |
|
Samsara BioCapital, L.P. |
36,416 |
- |
$499,988.04 |
$499,988.04 |
- |
|
CDK Associates, L.L.C. |
34,231 |
- |
$469,988.21 |
$469,988.21 |
- |
|
Third Street Holdings LLC |
2,185 |
- |
$29,999.83 |
$29,999.83 |
- |
|
ADAR1 Partners, LP |
65,250 |
- |
$895,875.98 |
$895,875.98 |
- |
|
Spearhead Insurance Solutions IDF, LLC - Series ADAR1 |
7,590 |
- |
$104,209.94 |
$104,209.94 |
- |
|
Dafna Lifescience LP |
52,080 |
- |
$715,053.19 |
$715,053.19 |
- |
|
Dafna Lifescience Select LP |
20,760 |
- |
$285,032.72 |
$285,032.72 |
- |
EXHIBIT B
PRE-FUNDED WARRANT
(See Attached)
EXHIBIT C
REGISTRATION RIGHTS AGREEMENT
(See Attached)
EXHIBIT D
STOCK ISSUANCE QUESTIONNAIRE
(See Attached)
EXHIBIT E
IRREVOCABLE TRANSFER AGENT INSTRUCTIONS
(See Attached)
EXHIBIT F
SECRETARY’S CERTIFICATE
(See Attached)
EXHIBIT G
COMPLIANCE CERTIFICATE
(See Attached)
July 23, 2024
Board of Directors
Pieris Pharmaceuticals, Inc.
225 Franklin Street, 26th Floor
Boston, MA 02110
Members of the Board:
Stifel, Nicolaus & Company, Incorporated (“Stifel” or “we”) has been advised that Pieris Pharmaceuticals, Inc. (the “Company” or “Pieris” or “Public Company”) is considering entering into an Agreement and Plan of Merger (the “Merger Agreement”) with Palvella Therapeutics (“Palvella” or “Merger Partner”) and Polo Merger Sub, Inc. (“Merger Sub”), which is a wholly-owned subsidiary of the Company, pursuant to which Merger Agreement Merger Sub will be merged with and into Palvella (the “Merger”), with Palvella continuing as the surviving corporation and as a wholly-owned subsidiary of the Company, and each share (other than to-be-cancelled shares held in treasury or owned by the Company, Merger Sub or any other subsidiary of the Company and any Dissenting Shares (as defined in the Merger Agreement)) of voting common stock, par value $0.0001 per share of Palvella, non-voting common stock, $0.001 per share of Palvella and preferred stock, $0.001 par value per share, of Palvella (such voting common stock, non-voting common stock and preferred stock, “Palvella Capital Stock”) issued and outstanding immediately prior to the Effective Time (as defined in the Merger Agreement) will be converted into the right to receive a number of shares of common stock, $0.001 par value per share, of Pieris (“Pieris Common Stock”) equal to the quotient obtained by dividing (a) the number of Merger Partner Merger Shares (as defined in the Merger Agreement), which will be derived from an allocation percentage determined by dividing (i) the Merger Partner Valuation (as defined in the Merger Agreement) for Palvella of $95,000,000 by (ii) the sum of the Merger Partner Valuation and the Public Company Valuation (as defined in the Merger Agreement) for Pieris of $21,000,000, subject to adjustment in the case of the Public Company Valuation (as to which adjustment we express no opinion) to reflect the difference on a dollar-for-dollar basis between the closing net cash position of Pieris and a specified target amount of $11,000,000, by (b) the number of Merger Partner Outstanding Shares (as defined in the Merger Agreement). At the direction of the Company and without independent verification, we have relied upon and assumed for purposes of our analyses and this Opinion (as defined below), that, based on the unadjusted Public Company Valuation (reflecting the estimated closing net cash position of Pieris provided to us by the Company) and also Public Company Outstanding Shares (as defined in the Merger Agreement) of 1,529,509 shares of Pieris Common Stock if the Public Company Closing Price (as defined in the Merger Agreement) were equal to the closing price per share of Pieris Common Stock on July 19, 2024, the aggregate consideration (i.e., the Merger Partner Merger Shares) to be paid by the Company in the Merger pursuant to the Merger Agreement will be 6,919,206 shares of Pieris Common Stock (the “Aggregate Merger Consideration”), representing approximately 81.8966% of the equity of the Company, on a fully-diluted and as-converted basis (before giving effect to the Concurrent Financing (as defined below)), at the Effective Time. The terms and conditions of the Merger are more fully set forth in the Merger Agreement. The Merger Agreement further provides that, (1) as a closing condition of the Merger, participating investors will have purchased newly-issued shares of Pieris Common Stock (including upon conversion of certain issued or to be issued convertible notes of Palvella (the “Palvella Notes”)) or will purchase such shares concurrently with or immediately following the closing of the Merger in accordance with the terms of a subscription agreement executed and delivered concurrently with the execution and delivery of the Merger Agreement (the “Concurrent Financing”), and (2) prior to the Effective Time, the Company will declare a dividend (the “Closing Dividend”) to its stockholders as of the close of business on the last business day prior to the day on which the Effective Time occurs of contingent value rights (the “Legacy CVRs”) representing the right to receive contingent payments upon the future occurrence of certain events relating to the legacy programs and assets of Pieris. At the direction of the Company and without independent verification, we have relied upon and assumed for purposes of our analyses and this Opinion, that, in connection with the Concurrent Financing, 4,370,270 additional shares of Pieris Common Stock will be issued and sold for $60,000,777 in cash and 1,376,556 additional shares of Pieris Common Stock will be issued upon conversion of the Palvella Notes.
The Board of Directors of the Company (the “Board”) has requested Stifel’s opinion, as investment bankers, as to the fairness, from a financial point of view, to the Company of the Aggregate Merger Consideration to be paid by the Company to the holders of Palvella Capital Stock in the Merger pursuant to the Merger Agreement (the “Opinion”).
In rendering our Opinion, we have, among other things:
|
(i) |
discussed the Merger and related matters with the Company’s counsel and reviewed the financial terms of the Merger contained in an execution version of the Merger Agreement, which is the most recent version of the Merger Agreement provided to Stifel; |
|
(ii) |
reviewed the audited consolidated financial statements of Pieris contained in its Annual Reports on Form 10-K for the three years ended December 31, 2023 and unaudited consolidated financial statements of Pieris contained in its Quarterly Report on Form 10-Q for the quarter ended March 31, 2024; |
|
(iii) |
reviewed the audited financial statements of Palvella for the year ended December 31, 2022, unaudited financial statements of Palvella for the year ended December 31, 2023 and unaudited financial statements of Palvella for the quarter ended March 31, 2024; |
|
(iv) |
reviewed and discussed with the Company’s management certain other publicly available information concerning the Company; |
|
(v) |
reviewed certain non-publicly available information concerning Pieris and Palvella, including the estimated closing net cash position of Pieris provided by the Company and internal financial analyses and forecasts for Palvella prepared by its management and reflecting the probabilities of technical success determined by Palvella’s management, and held discussions with the Company’s and Palvella’s respective senior managements regarding recent developments; |
|
(vi) |
reviewed and analyzed certain publicly available financial and stock market data relating to selected public companies that we deemed relevant to our analysis of Palvella; |
|
(vii) |
reviewed and analyzed certain publicly available information concerning the terms of selected initial public offerings that we considered relevant to our analysis; |
|
(viii) |
participated in certain discussions and negotiations between representatives of the Company and Palvella; |
|
(ix) |
reviewed the reported prices and trading activity of Pieris Common Stock; |
|
(x) |
considered the results of the Company’s strategic review process publicly announced in July 2023, including the Company’s efforts, with our assistance, to solicit indications of interest from selected third parties with respect to a merger or other transaction with Pieris; |
|
(xi) |
conducted such other financial studies, analyses and investigations and considered such other information as we deemed necessary or appropriate for purposes of our opinion; and |
|
(xii) |
took into account our assessment of general economic, market and financial conditions and our experience in other transactions, as well as our experience in securities valuations and our knowledge of Pieris’ and Palvella’s industry generally. |
The Company has not provided us with any internal financial analyses or forecasts for Pieris (other than working capital adjustments and operating and Merger-related costs estimates and other cash forecasts used to calculate the estimated closing net cash position of Pieris provided by the Company). For purposes of evaluating the Aggregate Merger Consideration, we have relied on the implied value of Pieris based on the estimated closing net cash position of Pieris provided by the Company and have not performed any financial analyses of Pieris or its non-cash assets, including, without limitation, the legacy programs and assets to which the Legacy CVRs relate. At the direction of the Company, we have relied upon and assumed, without independent verification, that the value of the Legacy CVRs will be substantially equivalent to the value of the existing business of Pieris. Furthermore, we have not performed any separate financial analysis of any particular class of Palvella Capital Stock or the Palvella Notes.
In rendering our Opinion, we have relied upon and assumed, without independent verification, the accuracy and completeness of all of the financial and other information that was provided to Stifel by or on behalf of the Company or Palvella, or that was otherwise reviewed by Stifel, and have not assumed any responsibility for independently verifying any of such information. With respect to the financial forecasts (including probabilities of technical success reflected therein) supplied to us by Palvella, we have assumed, at the direction of the Company, that they were reasonably prepared on the basis reflecting the best currently available estimates and judgments of the management of Palvella as to the future operating and financial performance of Palvella and that they provide a reasonable basis upon which we can form our opinion. Such forecasts were not prepared with the expectation of public disclosure. All such projected financial information is based on numerous variables and assumptions that are inherently uncertain, including, without limitation, factors related to general economic and competitive conditions and the widespread disruption, extraordinary uncertainty and unusual volatility arising from global tensions and political unrest, economic uncertainty, inflation, prolonged higher interest rates, the COVID-19 pandemic, and also including, without limitation, the effect of evolving governmental interventions and non-interventions. Accordingly, actual results could vary significantly from those set forth in such projected financial information. Stifel has relied on this projected information without independent verification or analyses and does not in any respect assume any responsibility for the accuracy or completeness thereof.
We also assumed that there were no material changes in the assets, liabilities, financial condition, results of operations, business or prospects of either Pieris or Palvella since the date of the last financial statements of each company made available to us. We did not make or obtain any independent evaluation, appraisal or physical inspection of either Pieris’ or the Palvella’s assets or liabilities, nor have we been furnished with any such evaluation or appraisal. Estimates of values of companies and assets do not purport to be appraisals or necessarily reflect the prices at which companies or assets may actually be sold. Such estimates are inherently subject to uncertainty and should not be taken as our view of the actual value of any companies or assets.
We have assumed, with your consent, that there are no factors that would delay or subject to any adverse conditions any necessary regulatory or governmental approval and that all conditions to the Merger will be satisfied and not waived. In addition, we have assumed that the definitive Merger Agreement will not differ materially from the execution version we reviewed. We have also assumed that the representations and warranties of the parties set forth in the Merger Agreement are and will be true and correct and that the Merger and related transactions (including, without limitation, the Concurrent Financing and the Closing Dividend) will be consummated substantially on the terms and conditions described in the Merger Agreement, without any waiver of material terms or conditions by the Company or any other party and without any anti-dilution or other adjustment to the Aggregate Merger Consideration (whether due to changes in the Public Company Net Cash (as defined in the Merger Agreement) or otherwise), and that obtaining any necessary regulatory approvals or satisfying any other conditions for consummation of the Merger and related transactions will not have an adverse effect on the Company, Palvella or the Merger. We have assumed that the Merger and related transactions will be consummated in a manner that complies with the applicable provisions of the Securities Act of 1933, as amended, the Securities Exchange Act of 1934, as amended, and all other applicable federal and state statutes, rules and regulations. We have further assumed that the Company has relied upon the advice of its counsel, independent accountants and other advisors (other than Stifel) as to all legal, financial reporting, tax, accounting and regulatory matters with respect to the Company, Palvella, the Merger and related transactions and the Merger Agreement.
Our Opinion is limited to whether the Aggregate Merger Consideration to be paid by the Company to the holders of Palvella Capital Stock is fair to the Company, from a financial point of view, and does not address any other terms, aspects or implications of the Merger or any related transaction (including the Concurrent Financing, the Closing Dividend and any potential sale by the Company of some or all of its assets at or prior to the closing of the Merger (a “Legacy Asset Disposition”)), including, without limitation, the form or structure of the Merger or any related transaction, the allocation of the Aggregate Merger Consideration among the different classes of Palvella Capital Stock, the prices at which shares of Pieris Common Stock will be issued in the Concurrent Financing or assets of the Company will be sold in any Legacy Asset Disposition, any consequences of the Merger or any related transaction on the Company, its shareholders, creditors or otherwise, or any terms, aspects or implications of the Legacy CVRs, the related contingent value rights agreements or any lock-up, voting, support, stockholder or other agreements, arrangements or understandings contemplated or entered into in connection with the Merger or any related transaction or otherwise. Our Opinion also does not consider, address or include: (i) any other strategic alternatives currently (or which have been or may be) contemplated by the Board or the Company; (ii) the legal, tax or accounting consequences of the Merger or any related transaction on the Company (including, without limitation, whether or not the Merger will qualify as a tax-free reorganization pursuant to Section 368 of the Internal Revenue Code or also as a non-taxable exchange of shares of Merger Partner Capital Stock for shares of Pieris Common Stock within the meaning of Section 351(a) of the Internal Revenue Code); (iii) the fairness of the amount or nature of any compensation to any of the Company’s officers, directors or employees, or class of such persons, relative to the compensation to the holders of the Company’s securities or relative to the Aggregate Merger Consideration; (iv) the effect of the Merger or any related transaction on, or the fairness of the consideration to be received by, holders of any class of securities of the Company, Palvella or any other party to any transaction contemplated by the Merger Agreement; or (v) any advice or opinions provided by any other advisor to the Company or Palvella. Furthermore, we are not expressing any opinion herein as to the Merger Partner Valuation, the Public Company Valuation, the actual value of Pieris Common Stock and the Legacy CVRs to be issued in connection with the Merger, the Concurrent Financing and the Closing Dividend, or the prices, trading range or volume at which Pieris Common Stock, the Legacy CVRs or any other securities of the Company will trade following public announcement or consummation of the Merger or any related transaction.
Our Opinion is necessarily based on economic, market, financial and other conditions as they exist on, and on the information made available to us by or on behalf of the Company, Palvella or their respective advisors, or information otherwise reviewed by Stifel, as of the date of this Opinion. It is understood that subsequent developments may affect the conclusion reached in this Opinion and that Stifel does not have any obligation to update, revise or reaffirm this Opinion. Further, as the Board is aware, the credit, financial and stock markets have been experiencing unusual volatility and we express no opinion or view as to any potential effects of such volatility on the Company, Palvella or the Merger. In addition, we express no view or opinion as to any changes after the date hereof that would result in the actual number of Merger Partner Merger Shares pursuant to the Merger Agreement being different from the amount of the Aggregate Merger Consideration that we have been directed to assume for purposes of our analyses and this Opinion.
Our Opinion is for the information of, and directed to, the Board (in its capacity as such) for its information and assistance in connection with its consideration of the financial terms of the Merger. Our Opinion does not constitute a recommendation to the Board as to how the Board should vote on the Merger or to any shareholder of the Company or stockholder of Palvella as to how any such shareholder or stockholder should vote at any shareholders’ or stockholders’ meeting at which the Merger is considered, or whether or not any shareholder or stockholder should enter into a voting, shareholders’, or affiliates’ agreement with respect to the Merger, or exercise any dissenters’ or appraisal rights that may be available to such shareholder or stockholder. In addition, this Opinion does not compare the relative merits of the Merger with any other alternative transactions or business strategies which may have been available to the Company and does not address the underlying business decision of the Board or the Company to proceed with or effect the Merger.
We are not legal, tax, regulatory or bankruptcy advisors. We have not considered any potential legislative or regulatory changes currently being considered or recently enacted by the United States Congress, the various federal banking agencies, the Securities and Exchange Commission (the “SEC”), or any other regulatory bodies, or any changes in accounting methods or generally accepted accounting principles that may be adopted by the SEC or the Financial Accounting Standards Board, or any changes in regulatory accounting principles that may be adopted by any or all of the federal banking agencies. Our Opinion is not a solvency opinion and does not in any way address the solvency or financial condition of the Company, Palvella or any other party.
Stifel, as part of its investment banking services, is regularly engaged in the independent valuation of businesses and securities in connection with mergers, acquisitions, underwritings, sales and distributions of listed and unlisted securities, private placements and valuations for estate, corporate and other purposes. We have acted as financial advisor to the Company in connection with the Merger and will receive a fee for our services, a substantial portion of which is contingent upon the completion of the Merger. We have also acted as financial advisor to the Board and will receive a fee upon the delivery of this Opinion that is not contingent upon consummation of the Merger. We will not receive any other significant payment or compensation contingent upon the successful consummation of the Merger. In addition, The Company has agreed to indemnify us for certain liabilities arising out of our engagement. There are no material relationships that existed during the two years prior to the date of this Opinion or that are mutually understood to be contemplated in which any compensation was received or is intended to be received as a result of the relationship between Stifel and the Company or Palvella. Stifel may seek to provide investment banking services to the Company or Palvella or their affiliates in the future, for which we would seek customary compensation. In the ordinary course of business, Stifel and our clients may transact in the equity securities of the Company and may at any time hold a long or short position in such securities.
Stifel’s Fairness Opinion Committee has approved the issuance of this Opinion. Our Opinion may not be published or otherwise used or referred to, nor shall any public reference to Stifel be made, without our prior written consent, except in accordance with the terms and conditions of Stifel’s engagement letter agreement with the Company.
Based upon and subject to the foregoing, we are of the opinion that, as of the date hereof, the Aggregate Merger Consideration to be paid by the Company to the holders of Palvella Capital Stock in the Merger pursuant to the Merger Agreement is fair to the Company, from a financial point of view.
Very truly yours,
STIFEL, NICOLAUS & COMPANY, INCORPORATED
Annex D
Final Form
FORM OF
CONTINGENT VALUE RIGHTS AGREEMENT
by and among
PIERIS PHARMACEUTICALS, INC.,
SHAREHOLDER REPRESENTATIVE SERVICES LLC,
as the Holder Representative, and
COMPUTERSHARE INC. and COMPUTERSHARE TRUST COMPANY, N.A.,
acting jointly as Rights Agent
Dated as of [●], 2024
TABLE OF CONTENTS
|
Page |
||
|
ARTICLE I DEFINITIONS |
1 |
|
|
Section 1.1 |
Definitions |
1 |
|
Section 1.2 |
Other Definitional Provisions |
3 |
|
ARTICLE II CONTINGENT VALUE RIGHTS |
4 |
|
|
Section 2.1 |
CVRs |
4 |
|
Section 2.2 |
Nontransferable |
4 |
|
Section 2.3 |
No Certificate; Registration; Registration of Transfer; Change of Address |
4 |
|
Section 2.4 |
Payment Procedures |
5 |
|
Section 2.5 |
No Voting, Dividends or Interest; No Equity or Ownership Interest in Public Company |
7 |
|
Section 2.6 |
Ability to Abandon CVR |
8 |
|
Section 2.7 |
Grant of Security |
8 |
|
ARTICLE III THE RIGHTS AGENT |
8 |
|
|
Section 3.1 |
Appointment of Rights Agent |
8 |
|
Section 3.2 |
Certain Duties and Responsibilities |
8 |
|
Section 3.3 |
Certain Rights of the Rights Agent |
8 |
|
Section 3.4 |
Resignation and Removal; Appointment of Successor |
11 |
|
Section 3.5 |
Acceptance of Appointment by Successor |
12 |
|
ARTICLE IV COVENANTS |
12 |
|
|
Section 4.1 |
List of Holders |
12 |
|
Section 4.2 |
Payment of CVR Event Payments |
12 |
|
Section 4.3 |
Access; Books and Records; Audit and Information Rights |
13 |
|
Section 4.4 |
Further Assurances |
14 |
|
Section 4.5 |
Covenants. |
14 |
|
Section 4.6 |
No Conflict |
15 |
|
ARTICLE V AMENDMENTS |
15 |
|
|
Section 5.1 |
Amendments without Consent of the Holder Representative |
15 |
|
Section 5.2 |
Amendments with Consent of the Holder Representative |
16 |
|
Section 5.3 |
Execution of Amendments |
16 |
|
Section 5.4 |
Effect of Amendments |
17 |
|
ARTICLE VI THE HOLDER REPRESENTATIVE |
17 |
|
|
Section 6.1 |
Appointment |
17 |
|
Section 6.2 |
Actions of Holder Representative |
17 |
|
Section 6.3 |
Removal; Appointment of Successor |
18 |
|
Section 6.4 |
Grant of Authority |
19 |
|
ARTICLE VII MISCELLANEOUS AND GENERAL |
19 |
|
|
Section 7.1 |
Termination |
19 |
|
Section 7.2 |
Notices to Public Company, the Rights Agent and the Holder Representative |
20 |
|
Section 7.3 |
Notice to Holders |
20 |
|
Section 7.4 |
Governing Law |
20 |
|
Section 7.5 |
Specific Enforcement; Jurisdiction |
21 |
|
Section 7.6 |
WAIVER OF JURY TRIAL |
21 |
|
Section 7.7 |
Other Remedies |
22 |
|
Section 7.8 |
Entire Agreement; Counterparts |
22 |
|
Section 7.9 |
Third-Party Beneficiaries; Action by the Holder Representative |
22 |
|
Section 7.10 |
Severability |
23 |
|
Section 7.11 |
Assignment |
23 |
|
Section 7.12 |
Confidentiality |
24 |
CONTINGENT VALUE RIGHTS AGREEMENT
This CONTINGENT VALUE RIGHTS AGREEMENT, dated as of [●], 2024 (this “Agreement”), is by and among Pieris Pharmaceuticals, Inc., a Nevada corporation (“Public Company”), Computershare Inc., a Delaware corporation (“Computershare”) and its affiliate Computershare Trust Company, N.A., a federally chartered trust company (together with Computershare, the “Rights Agent”), and Shareholder Representative Services LLC, a Colorado limited liability company (the “Holder Representative”), acting solely in its capacity as the representative, agent and attorney-in-fact of the Holders (as defined below), in favor of each Person who from time to time holds one or more contingent value rights to receive the Payments (as defined below) upon the occurrence of one or more CVR Events (as defined below) (each such contingent value right, a “CVR”), subject to the terms and conditions set forth herein. Each of the parties hereto shall be referred to as a “Party” and, collectively, as the “Parties.”
RECITALS
WHEREAS, this Agreement is entered into pursuant to that certain Agreement and Plan of Merger, dated July 23, 2024 (the “Merger Agreement”), by and among Public Company, Polo Merger Sub, Inc., a Delaware corporation and a wholly owned subsidiary of Public Company (“Merger Sub”) and Palvella Therapeutics, Inc., a Delaware corporation (the “Company”), pursuant to which Merger Sub will merge with and into the Company, with the Company surviving such merger as a wholly-owned subsidiary of Public Company on the terms and subject to the conditions set forth therein;
WHEREAS, pursuant to the terms of the Merger Agreement, and in accordance with the terms and conditions thereof, Public Company has agreed to provide, by way of dividend, to the Holders (as defined herein), which Holders shall initially be Persons who are stockholders of Public Company as of the close of business on the last Business Day prior to the day on which the Effective Time occurs, CVRs as hereinafter described, by way of a dividend or distribution consistent with the Merger Agreement; and
WHEREAS, the Parties have done all things necessary to make the CVRs, when issued pursuant to the Merger Agreement and hereunder, the valid obligations of Public Company and to make this Agreement a valid and binding agreement of Public Company, in accordance with its terms.
NOW, THEREFORE, in consideration of the foregoing and the consummation of the transactions referred to above, Public Company, the Holder Representative, and the Rights Agent agree, for the equal and proportionate benefit of all Holders, as follows:
ARTICLE I
DEFINITIONS
Section 1.1 Definitions. Capitalized terms used in this Agreement and not otherwise defined shall have the meanings assigned to them in the Merger Agreement. For purposes of this Agreement, the following terms shall have the following meanings:
“CVR Event” means from and after the Effective Time, each of the Strategic Partner Payments and the R&D Tax Credit Approval, as applicable. For the avoidance of doubt, more than one CVR Event may occur from time to time under this Agreement.
“CVR Event Payment” means, with respect to any CVR Event, an amount equal to the Net Proceeds received by Public Company or any of its Affiliates in respect of such CVR Event.
“Holder” means a Person in whose name a CVR is registered in the CVR Register at the applicable time.
“Independent Accountant” means an independent certified public accounting firm of nationally recognized standing designated either (a) jointly by the Holder Representative and Public Company, or (b) if the Holder Representative and Public Company fail to make a designation, jointly by an independent public accounting firm selected by Public Company and an independent public accounting firm selected by the Holder Representative.
“Majority Holders” means, at the time of determination, Holders of at least a majority of the outstanding CVRs as set forth in the CVR Register.
“Net Proceeds” means with respect to each CVR Event, an aggregate amount of payments received by Public Company or any of its Affiliates after the Effective Time equal to the sum of: (i) the aggregate amount actually paid to Public Company or any of its Affiliates in connection with such CVR Event, less (ii) the sum of (collectively, “CVR Expenses”), without duplication: (A) all reasonable and documented out-of-pocket expenses incurred or payable by Public Company or any of its Affiliates after the Effective Time to Third Parties in connection with such CVR Event, if any, including any attorney’s fees, (B) all reasonable out-of-pocket costs and expenses incurred or payable by Public Company or any of its Affiliates after the Effective Time to the Rights Agent and/or Holder Representative in connection with any of the obligations of Public Company or any of its Affiliates pursuant to this Agreement, if any, including any attorney’s fees, and (C) any applicable sales, income and other taxes in respect of the CVR Event that are incurred or payable by the Public Company after the Effective Time after taking into account any available net operating loss carryforwards or other tax attributes of Public Company or any of its Affiliates and any foreign tax credits which foreign tax credits are directly related to the applicable CVR Event, if any. Amounts placed in escrow or earnout or other contingent payments in connection with a CVR Event will not be considered Net Proceeds unless and until (and only to the extent that) such amounts are released from escrow or otherwise paid to the Public Company. For the avoidance of doubt, the calculation of CVR Expenses, with respect to any CVR Event, shall be without duplication and “CVR Expenses” shall not include (x) any amount deducted from the Public Company Expense Fund to pay Public Company CVR Expenses and (y) any amount offset against a CVR Event Payment to pay Public Company CVR Expenses.
“Officer’s Certificate” means a certificate signed by an authorized officer of Public Company, in his or her capacity as such an officer, and delivered to the Holder Representative and the Rights Agent.
“Permitted Transfer” means a transfer of a CVR (a) upon death of a Holder by will or intestacy, (b) by instrument to an inter vivos or testamentary trust in which the CVRs are to be passed to beneficiaries upon the death of the trustee, (c) pursuant to a court order, (d) by operation of Law (including by consolidation or merger) or without consideration in connection with the dissolution, liquidation or termination of any corporation, limited liability company, partnership or other entity, (e) in the case of CVRs held in book-entry or other similar nominee form, from a nominee to a beneficial owner (and, if applicable, through an intermediary) or from such nominee to another nominee for the same beneficial owner, in each case to the extent allowable by The Depository Trust Company (“DTC”) or (f) as permitted by Section 2.6.
“Person” means any natural person, corporation, exempted company, limited liability company, partnership, exempted limited partnership, association, trust or other entity, including a Governmental Entity, as applicable.
“Proceeding” means any private, governmental, or administrative claim, counterclaim, proceeding, suit, arbitration, hearing, litigation, audit, inquiry or investigation in each case whether civil, criminal, administrative, judicial or investigative, or any appeal therefrom.
“R&D Tax Credit Approval” means the approval by the relevant German Governmental Entity of the R&D tax credit (called research allowance or Forschungszulage) applied for by Public Company or any of its subsidiaries prior to the Effective Time (as may be amended or supplemented from time to time, the “R&D Tax Credit Application”).
“Rights Agent” means the Rights Agent named in the first paragraph of this Agreement, until a successor Rights Agent becomes such pursuant to the applicable provisions of this Agreement, and thereafter “Rights Agent” shall mean such successor Rights Agent.
“Strategic Partner Agreements” means each of (a) that certain Amended and Restated License and Collaboration Agreement by and among Public Company, Pieris Pharmaceuticals GmbH and Pfizer (formerly Seagen, Inc.), dated March 24, 2021, as amended or restated from time to time, (b) that certain Non-Exclusive Anticalin Platform Technology License Agreement by and among Public Company, Pieris Pharmaceuticals GmbH and Pfizer (formerly Seagen, Inc.), dated February 8, 2018, as amended or restated from time to time, (c) that certain Exclusive Product License Agreement, dated April 24, 2021, by and among Public Company, Pieris Pharmaceuticals GmbH, and BP Asset XII, Inc., as amended or restated from time to time, and (d) any license, collaboration or similar agreement by and among Public Company, Pieris Pharmaceuticals GmbH or any of their respective Affiliates and a third party involving any Legacy Asset, including PRS-400 and PRS-220, that is in effect as of the date hereof. Each of the parties to the Strategic Partners Agreements, other than Public Company and Pieris Pharmaceuticals GmbH, are referred to herein, as the “Strategic Partners.”
“Strategic Partner Payments” means the receipt by Public Company or any of its Affiliates of any milestone, royalty, license, or any similar cash payment, fee or amount pursuant to any of the Strategic Partner Agreements.
“Third Party” means any Person other than Public Company, the Holder Representative, or the Rights Agent or their respective Affiliates.
“TUM Agreement” means that certain Research and Licensing Agreement by and between Public Company and Technische Universität München, dated as of July 26, 2007, as amended or restated from time to time.
Section 1.2 Other Definitional Provisions. The headings contained in this Agreement and in the table of contents to this Agreement are for reference purposes only and shall not affect in any way the meaning or interpretation of this Agreement. The definitions of terms in this Agreement shall apply equally to the singular and plural forms of the terms defined. Whenever the context may require, any pronoun shall include the corresponding masculine, feminine and neuter forms. The word “will” shall be construed to have the same meaning as the word “shall.” The words “include,” “includes” and “including” shall be deemed, in each case, to be followed by the phrase “without limitation.” The word “extent” in the phrase “to the extent” shall mean the degree to which a subject or other thing extends, and such phrase shall not mean simply “if.” All references to “dollars” or “$” shall refer to the lawful currency of the United States. Unless the context requires otherwise (i) any reference in this Agreement to any “Person” shall be construed to include such Person’s successors and permitted assigns, (ii) the words “herein,” “hereof” and “hereunder,” and words of similar import, shall be construed to refer to this Agreement in its entirety and not to any particular provision hereof, (iii) all references in this Agreement to Articles, Sections and Exhibits shall be construed to refer to Articles and Sections of, and Exhibits to, this Agreement, unless otherwise indicated, (iv) references to clauses without a cross-reference to a Section or subsection are references to clauses within the same Section or, if more specific, subsection and (v) references from or through any date shall mean, unless otherwise specified, from and including or through and including, respectively. This Agreement shall be construed without regard to any presumption or rule requiring construction or interpretation against the party hereto drafting or causing any instrument to be drafted. The parties hereto have participated jointly in the negotiation and drafting of this Agreement and, in the event an ambiguity or question of intent or interpretation arises, this Agreement shall be construed as jointly drafted by the parties hereto and no presumption or burden of proof shall arise favoring or disfavoring any party hereto by virtue of the authorship of any provision of this Agreement. Unless indicated otherwise, (i) any action required to be taken by or on a day or Business Day may be taken until 11:59 p.m. Eastern Time on such day or Business Day, (ii) all references to “days” shall be to calendar days unless otherwise indicated as a “Business Day” and (iii) all days, Business Days, times and time periods contemplated by this Agreement will be determined by reference to Eastern Time. Unless indicated otherwise, all mathematical calculations contemplated by this Agreement shall be rounded to the fourth decimal place, except in respect of payments, which shall be rounded down to the nearest whole United States cent.
ARTICLE II
CONTINGENT VALUE RIGHTS
Section 2.1 CVRs. CVRs shall be issued and distributed by Public Company in the form of a dividend, in connection with the Merger, to the Persons who, as of the close of business on the last Business Day prior to the day on which the Effective Time occurs, are a record holder of shares of Public Company Common Stock or shares of Public Company Preferred Stock entitled to receive such dividend in accordance with the terms of such Public Company Preferred Stock. Furthermore, to the extent a holder of a Public Company Warrant outstanding as of the date hereof exercises such Public Company Warrant after the date hereof (whether before or after the Closing), a number of CVRs equal to the number of shares of Public Company Common Stock issued to such holder in connection with such exercise shall be issued to such holder, subject to and in accordance with the terms and conditions of such Public Company Warrant, as applicable, and this Agreement. Notwithstanding anything to the contrary, this Agreement shall only become effective as of, and contingent upon, the Closing and shall be void ab initio and of no effect upon the valid termination of the Merger Agreement.
Section 2.2 Nontransferable. The CVRs may not be sold, assigned, transferred, pledged, encumbered or in any other manner transferred or disposed of, in whole or in part, other than through a Permitted Transfer. Any attempted sale, assignment, transfer, pledge, encumbrance or disposition of the CVRs, in whole or in part, that is not a Permitted Transfer, will be null and void ab initio and of no effect. CVRs will not be listed on any quotation system or traded on any securities exchange.
Section 2.3 No Certificate; Registration; Registration of Transfer; Change of Address.
(a) The Holders’ rights and obligations in respect of CVRs derive solely from this Agreement; CVRs shall not be evidenced by a certificate or other instrument.
(b) Subject to the receipt by the Rights Agent of the information and instructions described in Section 4.1, the Rights Agent shall keep an up-to-date register (the “CVR Register”) for the purpose of: (i) identifying the Holders of CVRs and (ii) registering CVRs and Permitted Transfers thereof. The CVRs shall initially, in the case of the holders of shares of Public Company Common Stock and shares of Public Company Preferred Stock entitled to receive the dividend of CVRs in accordance with the terms of such Public Company Preferred Stock outstanding as of the as of the close of business on the last Business Day prior to the day on which the Effective Time occurs, be registered in the names and addresses of the respective holders as set forth in the form Public Company furnishes or causes to be furnished to the Rights Agent pursuant to Section 4.1. The CVR Register will initially show one position for Cede & Co. representing all of the shares of Public Company Common Stock held by DTC on behalf of the street holders of the shares of Public Company Common Stock held by such holders as of immediately prior to the Effective Time. The Rights Agent will have no responsibility whatsoever directly to the street name holders with respect to transfers of CVRs. With respect to any payments to be made under Section 2.4(d) below, the Rights Agent will accomplish the payment to any former street name holders of shares of Public Company Common Stock by sending one lump payment to DTC. The Rights Agent will have no responsibilities whatsoever with regard to the distribution of payments by DTC to such street name holders. In addition, upon notification by the Public Company pursuant to Section 4.1, the Rights Agent shall reflect in the CVR Register the additional CVRs issued to the holders of Public Company Warrants who exercise such Public Company Warrants following the Closing.
(c) Subject to the restrictions on transferability set forth in Section 2.2, every request made to transfer a CVR must be in writing and accompanied by a written instrument of transfer and other requested documentation in form reasonably satisfactory to the Rights Agent pursuant to its customary policies and guidelines, which may include a guaranty of signature by an “eligible guarantor institution” that is a member or participant in the Securities Transfer Agents Medallion Program, duly executed by the Holder thereof, the Holder’s attorney duly authorized in writing, the Holder’s personal representative or the Holder’s survivor, as applicable, and setting forth in reasonable detail the circumstances relating to the transfer. Upon receipt of such written notice, the Rights Agent shall, subject to its reasonable determination that the transfer instrument is in proper form and the transfer otherwise complies with the other terms and conditions of this Agreement (including the provisions of Section 2.2), register the transfer of such CVR in the CVR Register. Any transfer of CVRs will be without charge to the applicable Holder; provided, however, that Public Company, its Affiliates, and the Rights Agent may require payment of a sum sufficient to cover any stamp or other Tax or governmental charge that is imposed in connection with any such registration or transfer. The Rights Agent shall have no duty or obligation to take any action under any section of this Agreement that requires the payment by a Holder of applicable Taxes or charges unless and until the Rights Agent is satisfied that all such Taxes or charges have been paid. All duly transferred CVRs registered in the CVR Register shall be the valid obligations of Public Company and shall entitle the transferee to the same benefits and rights under this Agreement as those held immediately prior to the transfer by the transferor. No transfer of a CVR shall be valid until registered in the CVR Register in accordance with this Agreement.
(d) A Holder may make a written request to the Rights Agent to change such Holder’s address of record in the CVR Register. The written request must be duly executed by the Holder. Upon receipt of such written notice, the Rights Agent shall promptly record the change of address in the CVR Register.
Section 2.4 Payment Procedures.
(a) Upon the occurrence of a CVR Event, as promptly as practicable (and in any event within forty-five (45) days after Public Company has received Net Proceeds from such CVR Event), Public Company shall (i) deliver (or cause to be delivered) to the Holder Representative and the Rights Agent an Officer’s Certificate (a “CVR Event Notice”): (A) certifying that a CVR Event has occurred and providing a reasonably detailed description of such CVR Event, including the date of the occurrence of such CVR Event, (B) certifying that each Holder is entitled to receive the CVR Payment Amount applicable to such Holder, and (C) setting forth Public Company’s calculation of the CVR Event Payment, and (ii) deliver (or cause to be delivered) to the Rights Agent any letter of instruction reasonably required by the Rights Agent.
(b) If (i) the Holder Representative does not object to any determination or calculation set forth in the CVR Event Notice by delivery of a written notice thereof to Public Company (with a copy to the Rights Agent) setting forth in reasonable detail such objection, together with reasonable supporting documentation (an “Objection Notice”) within thirty (30) calendar days following receipt of the applicable CVR Event Notice (the “Objection Period”), or (ii) the Holder Representative delivers a written notice to Public Company (with a copy to the Rights Agent) indicating that it has no objections to the calculations set forth in the CVR Event Notice, Public Company’s calculation of any CVR Event Payment, as applicable, shall be final and binding on all parties and the Holders. If the Holder Representative has not timely delivered an Objection Notice to Public Company during the Objection Period, the Holder Representative shall be deemed to have accepted the determination or calculation set forth in the CVR Event Notice.
(c) If the Holder Representative timely delivers to Public Company (with a copy to the Rights Agent) an Objection Notice, Public Company and the Holder Representative shall attempt in good faith to resolve such matters within thirty (30) calendar days after receipt of the same by Public Company, and if unable to do so, Public Company and the Holder Representative shall resolve any unresolved disputed in accordance with the provisions of Section 7, which decision will be final and binding on the parties.
(d) Within ten (10) Business Days of the final determination of a CVR Event Payment in accordance with the terms of this Agreement, Public Company shall deliver (or cause to be delivered) to the Rights Agent (i) any letter of instruction reasonably required by the Rights Agent, and (ii) cash, by wire transfer of immediately available funds to an account designated by the Rights Agent, in an amount equal to the applicable CVR Event Payment due to all Holders pursuant to Section 4.2, as finally determined pursuant to this Section 2.4. The Rights Agent will promptly, and in any event within ten (10) Business Days of receipt of such CVR Event Payment and any letter of instruction reasonably required by the Rights Agent, pay to each Holder in accordance with the corresponding letter of instruction, an amount equal to the product determined by multiplying (i) the quotient of: (A) the applicable CVR Event Payment divided by (B) the sum of: (x) the total number of CVRs registered in the CVR Register as of the date of such CVR Event, plus (y) the total number of CVRs that would be issued to the holders of Public Company Warrants outstanding as of the date of such CVR Event assuming the full exercise of such Public Company Warrants by the holders thereof (which number shall be determined by the Company and provided to the Rights Agent), by (ii) the number of CVRs registered to such Holder in the CVR Register at such time (each such amount, a “CVR Payment Amount”) (1) by check mailed to the address of such Holder, reflected in the CVR Register as of 5:00 p.m. New York City time on the last Business Day before such payment is made or (2) with respect to any such Holder who has provided the Rights Agent wiring instructions in writing as of the close of business on the last Business Day before such payment is made, by wire transfer of immediately available funds to the account specified on such instructions. Concurrently with the payment of the CVR Payment Amounts pursuant to the preceding sentence, Public Company shall deposit with the Rights Agent, for the benefit of holders of any Public Company Warrants that remain outstanding and unexercised, an amount equal to the aggregate CVR Payment Amounts that would be due with respect to the CVRs issuable to the holders of such Public Company Warrants if such Public Company Warrants had been exercised and the corresponding CVRs had been issued in respect of Public Company Common Stock issuable in connection with such exercise. The applicable portion of such CVR Payment Amounts deposited for the benefit of such holders of Public Company Warrants shall be (i) upon exercise of a Public Company Warrant and issuance of CVRs in respect of Public Company Common Stock issuable in connection with such exercise, paid out to the Holder of the CVRs so issued as a result of such exercise within ten (10) Business Days after such exercise, and (ii) upon expiration or termination of a Public Company Warrant, paid out as additional CVR Event Payments to the existing Holders within ten (10) Business Days after such expiration or termination.
(e) Each of Public Company, the Rights Agent and each of their respective Affiliates shall be entitled to deduct and withhold from the amounts otherwise payable pursuant to this Agreement such amounts as it is required to deduct or withhold therefrom under applicable Law. Prior to making any such Tax withholdings or causing any such Tax withholdings to be made with respect to any Holder, the Rights Agent shall, to the extent practicable, provide notice to the Holder of such potential withholding and, if applicable, a reasonable opportunity for the Holder to provide any necessary Tax forms in order to avoid or reduce such withholding amounts; provided, that the time period for payment of a CVR Payment Amount by the Rights Agent to such Holder set forth in this Section 2.4 shall be extended by a period equal to any delay caused by such Holder in providing such forms. Any such amounts deducted or withheld and remitted to the appropriate Governmental Entity in accordance with applicable Law shall be treated for all purposes under this Agreement and the Merger Agreement as having been paid to the Holder to whom such amounts would otherwise have been paid.
(f) Any cash deposited with the Rights Agent pursuant to Section 2.4(d) (and any interest or other income earned thereon) that remains undistributed as of the date such CVR Payment Amount could properly be delivered to a public official pursuant to applicable abandoned, property, escheat, or similar applicable Law (including by means of invalid addresses on the CVR Register) shall be delivered by the Rights Agent to Public Company or its designated Affiliate, upon demand, and any Holder entitled to his, her or its CVR Payment Amount hereunder shall thereafter look to Public Company or any successor-in-interest of Public Company for payment of its claim for such CVR Payment Amount (subject to applicable abandoned property, escheat and other similar Law), without interest, but such Holder will have no greater rights against Public Company than those accorded to general unsecured creditors of Public Company under applicable Law. In addition to and not in limitation of any other indemnity obligation herein, Public Company agrees to indemnify and hold harmless the Rights Agent with respect to any liability, penalty, cost or expense the Rights Agent may incur or be subject to in connection with transferring such property to Public Company.
(g) The right of the Holders to receive CVR Payment Amounts payable in respect of the CVRs hereunder shall rank equal in seniority with, or have priority over, the payment of any dividends on Public Company Common Stock whether in liquidation or otherwise.
(h) Except to the extent otherwise required pursuant to a change in applicable Law after the Closing Date, the Parties shall treat, for all Tax purposes, the issuance of the CVRs in accordance with the Agreed Tax Treatment as determined by Public Company, upon the advice and recommendation of its tax advisor PricewaterhouseCoopers LLP, after Public Company’s good faith consultation with its legal counsel and Merger Partner. The Agreed Tax Treatment shall be the treatment of the issuance of the CVRs, for Tax purposes, that is determined in accordance with the previous sentence, and shall be determined at least two (2) Business Days prior to the filing of the initial Registration Statement. The Parties will not take any position contrary to the Agreed Tax Treatment on any Tax Return or for other Tax purposes, except as required by a change in applicable Law after the Closing Date.
Section 2.5 No Voting, Dividends or Interest; No Equity or Ownership Interest in Public Company.
(a) The CVRs will not have any voting or dividend rights, and interest will not accrue on any amounts payable on the CVRs to any Holder.
(b) The CVRs will not represent any equity or ownership interest in Public Company, any constituent company to the transactions contemplated by the Merger Agreement, or any of their respective Subsidiaries or Affiliates. The rights of the Holders and the obligations of Public Company are contract rights limited to those expressly set forth in this Agreement, and such Holders’ sole right to receive property hereunder is the right to receive cash from Public Company, if any, through the Rights Agent in accordance with the terms hereof.
(c) It is hereby acknowledged and agreed that the CVRs and the possibility of any payment hereunder with respect thereto are highly speculative, and it is highly possible that Holders will not receive any payments under this Agreement or in connection with the CVRs. It is highly possible that no CVR Event will occur and accordingly it is highly possible that there will not be any CVR Payment. It is acknowledged and agreed that this Section 2.5(c) is an essential and material term of this Agreement.
Section 2.6 Ability to Abandon CVR. A Holder may, at any time and at such Holder’s option, abandon all of such Holder’s remaining rights in a CVR by transferring such CVR to Public Company without consideration therefor, which a Holder may affect by delivery of a written notice of such abandonment to Public Company and the Rights Agent, which abandonment notice, if given, shall be irrevocable. Nothing in this Agreement shall prohibit Public Company or any of its Affiliates from offering to acquire or acquiring any CVRs for consideration from the Holders, in private transactions or otherwise, in its sole discretion. Any CVRs acquired by Public Company or any of its Affiliates shall be automatically deemed extinguished and no longer outstanding for purposes of the definition of Majority Holders, Section 2.4, Article V and Section 7.9. The Rights Agent shall update the CVR Register to reflect any abandonment or acquisition of CVRs described in this Section 2.6.
Section 2.7 Grant of Security. To secure all obligations owed to the Holders and the Holder Representative hereunder, including payment of the CVR Event Payments, Public Company hereby grants to the Holder Representative, for the benefit of the Holders, a first priority security interest in and continuing lien on all of the Public Company’s right, title and interest in, to and under the Strategic Partner Agreements, the TUM Agreement, and any payments to Public Company or any of its Affiliates associated therewith (net of any CVR Expenses incurred), including the right to receive any payments under such agreements, in each case whether now existing or hereafter acquired or arising, and any proceeds thereof
ARTICLE III
THE RIGHTS AGENT
Section 3.1 Appointment of Rights Agent. Public Company hereby appoints the Rights Agent to act as rights agent in accordance with the express terms and conditions set forth in this Agreement, and the Rights Agent hereby accepts such appointment.
Section 3.2 Certain Duties and Responsibilities. The Rights Agent shall not have any liability for any actions taken, suffered or omitted to be taken in connection with this Agreement, except to the extent of its gross negligence, bad faith or willful misconduct (each as determined by a final non-appealable order, judgment, decree or ruling of a court of competent jurisdiction)of the Rights Agent.
Section 3.3 Certain Rights of the Rights Agent. The Rights Agent undertakes to perform such duties and only such duties as are specifically set forth in this Agreement, and no implied covenants or obligations shall be read into this Agreement against the Rights Agent. In addition:
(a) the Rights Agent may rely and shall be protected and held harmless by Public Company in acting or refraining from acting upon any resolution, certificate, statement, instrument, opinion, report, notice, request, direction, consent, order or other paper or document reasonably believed by it in the absence of bad faith to be genuine and to have been signed or presented by the proper Party or Parties;
(b) whenever the Rights Agent shall deem it desirable that a matter be proved or established prior to taking, suffering or omitting any action hereunder, the Rights Agent may rely upon an Officer’s Certificate, which certificate shall be deemed full authorization and protection to the Rights Agent, and the Rights Agent shall, in the absence of bad faith, incur no liability and be held harmless by Public Company for or in respect of any action taken, suffered or omitted to be taken by it under the provisions of this Agreement in the absence of bad faith reliance upon such certificate;
(c) the Rights Agent may engage and consult with counsel of its selection and the written advice of such counsel or any opinion of counsel shall be full and complete authorization and protection to the Rights Agent, and the Rights Agent shall be held harmless by Public Company in respect of any action taken, suffered or omitted by it hereunder in the absence of bad faith and in reliance thereon;
(d) the permissive rights of the Rights Agent to do things enumerated in this Agreement shall not be construed as a duty;
(e) the Rights Agent shall not be required to give any note or surety in respect of the execution of such powers;
(f) the Rights Agent shall not be liable for or by reason of, and shall be held harmless by Public Company with respect to any of the statements of fact or recitals contained in this Agreement or be required to verify the same, but all such statements and recitals are and shall be deemed to have been made by Public Company only;
(g) the Rights Agent shall have no liability and shall be held harmless by Public Company in respect of the validity of this Agreement or the execution and delivery hereof (except the due execution and delivery hereof by the Rights Agent and the enforceability of this Agreement against the Rights Agent, assuming the due execution and delivery hereof by Public Company), nor shall it be responsible for any breach by Public Company of any covenant or condition contained in this Agreement;
(h) Public Company agrees to indemnify the Rights Agent for, and hold the Rights Agent harmless against, any loss, liability, damage, judgment, fine, penalty, claim, demand, suit, settlement, cost or expense for any action taken, suffered or omitted to be taken by the Rights Agent arising out of or in connection with the execution, acceptance, administration, exercise and performance by the Rights Agent of its duties under this Agreement, including the reasonable and documented out-of-pocket costs and expenses (including counsel expenses) of defending the Rights Agent against any loss, liability, damage, judgment, fine, penalty, claim, demands, suits or expense arising therefrom, directly or indirectly, or enforcement of its rights hereunder incurred without gross negligence, bad faith, or willful misconduct (each as determined by a final non-appealable order, judgment, decree or ruling of a court of competent jurisdiction) on the part of the Rights Agent;
(i) notwithstanding anything to the contrary herein, in no event shall the Rights Agent be liable for any special, punitive, indirect, consequential or incidental loss or damage of any kind whatsoever (including but not limited to lost profits) arising out of any act or failure to act hereunder, even if the Rights Agent has been advised of the likelihood of such loss or damage or has foreseen the possibility or likelihood of such damages.
(j) Notwithstanding anything to the contrary contained herein, the aggregate liability of the Rights Agent arising in connection with this Agreement, whether in contract, or in tort, or otherwise, is limited to, and shall not exceed the amounts paid or payable hereunder by Public Company to the Rights Agent as fees and charges during the twelve (12) months immediately preceding the event for which recovery from the Rights Agent is being sought;
(k) Public Company agrees (i) to pay the fees and expenses of the Rights Agent in connection with this Agreement as set forth on Schedule 1 hereto and (ii) to reimburse the Rights Agent for (x) all Taxes other than Taxes imposed on or measured by the Rights Agent’s net income and franchise or similar Taxes imposed on it (in lieu of net income Taxes) and (y) governmental charges, reasonable and documented out-of-pocket expenses and other charges of any kind and nature incurred by the Rights Agent in the execution of this Agreement (other than Taxes). The Rights Agent will also be entitled to reimbursement from Public Company for all reasonable, documented and necessary out-of-pocket expenses paid or incurred by it in connection with the administration by the Rights Agent of its duties hereunder;
(l) no provision of this Agreement shall require the Rights Agent to expend or risk its own funds or otherwise incur any financial liability in the performance of any of its duties hereunder or in the exercise of its rights if there shall be reasonable grounds for believing that repayment of such funds or adequate indemnification against such risk or liability is not reasonably assured to it;
(m) the Rights Agent shall not be deemed to have knowledge of any event of which it was supposed to receive notice thereof hereunder, and the Rights Agent shall be fully protected and shall incur no liability for failing to take action in connection therewith, in each case, unless and until such notice has been given in accordance with Section 7.2;
(n) unless otherwise specifically prohibited by the terms of this Agreement, the Rights Agent and any stockholder, affiliate, member, director, officer, agent, representative or employee of the Rights Agent may buy, sell or deal in any of the securities of Public Company or become pecuniarily interested in any transaction in which Public Company may be interested, or contract with or lend money to Parent or otherwise act as fully and freely as though it were not the Rights Agent under this Agreement. Nothing herein shall preclude the Rights Agent or any such stockholder, affiliate, director, member, officer, agent, representative or employee from acting in any other capacity for Public Company or for any other Person;
(o) the Rights Agent may perform any and all of its duties (i) itself (through its directors, officers, or employees) or (ii) through its agents, representatives, attorneys, custodians and/or nominees and the Rights Agent shall not be answerable or accountable for any act, default, neglect or misconduct of any such agents, representatives, attorneys, custodians and/or nominees, absent their gross negligence, bad faith or willful misconduct (each as determined by a final non-appealable judgment of a court of competent jurisdiction) in the selection and continued employment thereof;
(p) the Rights Agent shall act hereunder solely as agent for Public Company and it shall not assume any obligations or relationship of agency or trust with any of the Holders;
(q) the Rights Agent shall not have any duty or responsibility with respect to any action or default by Public Company, including, without limiting the generality of the foregoing, any duty or responsibility to initiate or attempt to initiate any proceedings at law or otherwise or to make any demand upon Public Company;
(r) all funds received by Computershare under this Agreement that are to be distributed or applied by Computershare in the performance of services hereunder (the “Funds”) shall be held by Computershare as agent for Public Company and deposited in one or more bank accounts to be maintained by Computershare in its name as agent for Public Company. Until paid pursuant to the terms of this Agreement, Computershare will hold the Funds through such accounts in: deposit accounts of commercial banks with Tier 1 capital exceeding $1 billion or with an average rating above investment grade by S&P (LT Local Issuer Credit Rating), Moody’s (Long Term Rating) and Fitch Ratings, Inc. (LT Issuer Default Rating) (each as reported by Bloomberg Finance L.P.). The Rights Agent shall have no responsibility or liability for any diminution of the Funds that may result from any deposit made by Computershare in accordance with this paragraph, including any losses resulting from a default by any bank, financial institution or other third party. Computershare may from time to time receive interest, dividends or other earnings in connection with such deposits. Computershare shall not be obligated to pay such interest, dividends or earnings to Public Company, any Holder or any other party;
(s) other than for guaranty of signature as provided in Section 2.3(c), no Holder shall be obligated to indemnify the Rights Agent for, or hold the Rights Agent harmless against, any loss, liability, claim, demand, suit or expense arising out of or in connection with the Rights Agent’s duties under this Agreement or to pay or reimburse the Rights Agent for any fees, costs or expenses incurred by the Rights Agent in connection with this Agreement or the administration of its duties hereunder, and the Rights Agent shall not be entitled to deduct any amount from any CVR Event Payment in any circumstance except as provided in Section 2.4(e); and
(t) the provisions of this Section 3.3 shall survive the termination of this Agreement and the resignation, replacement or removal of the Rights Agent.
Section 3.4 Resignation and Removal; Appointment of Successor.
(a) The Rights Agent may resign at any time by giving written notice thereof to Public Company and the Holder Representative specifying a date when such resignation shall take effect, which notice shall be sent at least thirty (30) calendar days prior to the date so specified, and such resignation become effective on the earlier of (i) the date so specified and (ii) the appointment of a successor Rights Agent. Public Company has the right to remove the Rights Agent at any time by specifying a date when such removal shall take effect. Notice of such removal shall be given by Public Company to the Holder Representative and the Rights Agent, which notice shall be sent at least thirty (30) calendar days prior to the date so specified.
(b) If the Rights Agent provides notice of its intent to resign, is removed pursuant to Section 3.4(a) or becomes incapable of acting, Public Company shall, as soon as is reasonably possible, appoint a qualified successor Rights Agent who shall be a stock transfer agent of national reputation or the corporate trust department of a commercial bank. The successor Rights Agent so appointed shall, forthwith upon its acceptance of such appointment in accordance with Section 3.5, become the successor Rights Agent. Notwithstanding the foregoing, if Public Company fails to make such appointment within a period of thirty (30) calendar days after giving notice of such removal or after it has been notified in writing of such resignation or incapacity by the resigning or incapacitated Rights Agent, then the incumbent Rights Agent may apply to any court of competent jurisdiction for the appointment of a new Rights Agent. The successor Rights Agent so appointed shall, forthwith upon its acceptance of such appointment in accordance with Section 3.4, become the successor Rights Agent.
(c) Public Company shall give notice of each resignation and each removal of a Rights Agent and each appointment of a successor Rights Agent by mailing written notice of such event by first-class mail or the facilities of DTC, as applicable, to the Holder Representative and to the Holders as their names and addresses appear in the CVR Register. Each notice shall include the name and address of the successor Rights Agent. If Public Company fails to send such notice within thirty (30) calendar days after acceptance of appointment by a successor Rights Agent, the successor Rights Agent shall cause the notice to be mailed at the expense of Public Company. Failure to give any notice provided for in this Section 3.4, however, shall not affect the legality or validity of the resignation or removal of the Rights Agent or the appointment of the successor Rights Agent, as the case may be.
(d) The Rights Agent will reasonably cooperate with Public Company and any successor Rights Agent in connection with the transition of the duties and responsibilities of the Rights Agent to the successor Rights Agent, including the transfer of all relevant data, including the CVR Register, to the successor Rights Agent, but such predecessor Rights Agent shall not be required to make any additional expenditure or assume any additional liability in connection with the foregoing.
Section 3.5 Acceptance of Appointment by Successor. Every successor Rights Agent appointed hereunder shall execute, acknowledge and deliver to Public Company, the Holder Representative and to the predecessor Rights Agent an instrument accepting such appointment and a counterpart of this Agreement, and thereupon such successor Rights Agent, without any further act, deed or conveyance, shall become vested with all the rights, powers, liabilities, and duties of the predecessor Rights Agent. On request of Public Company, the Holder Representative or the successor Rights Agent, the predecessor Rights Agent shall execute and deliver an instrument transferring to the successor Rights Agent all the rights, powers and trusts of the predecessor Rights Agent, except such rights which survive its resignation or removal under the terms hereunder.
ARTICLE IV
COVENANTS
Section 4.1 List of Holders. Public Company shall furnish or cause to be furnished to the Rights Agent and the Holder Representative, promptly after the Effective Time and in no event later than thirty (30) calendar days following the Effective Time, in such form as Public Company receives from Public Company’s transfer agent (or other agent performing similar services for Public Company), the names and addresses of the Holders. The Rights Agent will reflect all such names and addresses on the CVR Register and confirm the write up of the CVR Register and list of initial Holders to Public Company and the Holder Representative promptly thereafter and, in any event, within thirty (30) calendar days of the receipt of such names and addresses from Public Company’s transfer agent, as the case may be. Public Company shall promptly provide written notice to the Rights Agent and the Holder Representative of the (a) exercise of any Public Company Warrants and the issuance of any additional CVRs in connection therewith and provide to the Rights Agent and the Holder Representative the names and addresses of the Holders of such additional CVRs and the respective numbers of CVRs acquired by them pursuant to such exercise, and (b) expiration or termination of any Public Company Warrants including the number of CVRs that were subject to such expired or terminated Public Company Warrants and are therefore no longer to be included in the calculation of CVR Event Payments. The Rights Agent shall not be deemed to have any knowledge of the exercise or expiration of any Public Company Warrant or any CVRs to be issued or terminated in connection therewith, nor shall the Rights Agent be required to investigate or verify any calculation or adjustment made in any such notice.
Section 4.2 Payment of CVR Event Payments. Public Company will cause to be deposited with the Rights Agent, for payment to the Holders, when payable in accordance with the terms of this Agreement, the CVR Event Payments in accordance with Section 2.4(b) hereof.
Section 4.3 Access; Books and Records; Audit and Information Rights.
(a) Upon reasonable notice, Public Company shall, and shall cause its Affiliates to, provide the Holder Representative and its representatives with reasonable access during normal business hours to the books, records, work papers, other information, facilities and employees of Public Company and its Affiliates as the Holder Representative may reasonably request for purposes of performing its duties and exercising its rights hereunder, including determining compliance with the terms of this Agreement, the amounts payable hereunder and compliance by Public Company, its Affiliates, the Strategic Partners and each of their respective successors and assigns, with and under the Strategic Partner Agreements; provided that (x) the Holder Representative and such representative, as applicable, enters into customary and reasonable confidentiality agreements reasonably satisfactory to Public Company with respect to the confidential information of Public Company or its Affiliates to be furnished pursuant to this Section 4.3 (a “Confidentiality Agreement”) and (y) such access does not unreasonably interfere with the conduct of the business of Public Company or any of its Affiliates. Notwithstanding anything herein to the contrary, following the Closing, the Holder Representative shall be permitted to disclose information as required by Law or to representatives and advisors of the Holder Representative and to the Holders, in each case who have a need to know such information; provided that such persons are subject to confidentiality obligations with respect thereto. Without limiting the generality of the foregoing, (i) within ten (10) Business Days of Public Company or any of its Affiliates receiving any notices, records, files, documents, reports, correspondence, studies, or other information (collectively, “Strategic Partner Information”) from a Strategic Partner or any of such Strategic Partner’s Affiliates related to a Strategic Partner Agreement or Strategic Partner Payment, Public Company shall (or shall cause its Affiliates to) deliver such Strategic Partner Information to the Holder Representative, and (ii) promptly (an in any event within ten (10) Business Days) upon becoming aware of any material development under any Strategic Partner Agreement, Public Company or one of its Affiliates shall deliver to the Holder Representative notice of such material development. For the avoidance of doubt, to the extent reasonably requested by the Holder Representative, Public Company and each of its Affiliates shall exercise such Person’s rights under the relevant Strategic Partner Agreement in order to obtain Strategic Partner Information.
(b) Public Company shall maintain for a period of seven (7) years, and will not permit any other Person to destroy or dispose of, the books and records of Public Company and its Affiliates, or any portions thereof, without first giving reasonable prior notice to the Holder Representative and offering (at Public Company’s sole cost and expense) to surrender to the Holder Representative (for the benefit of the Holders) such books and records or such portions thereof; provided, any books and records related to the Strategic Partner Agreements shall be maintained, and not destroyed or disposed of, until the later of: (a) the termination of this Agreement in accordance with its terms, and (b) the date on which there are no further milestone, royalty, license, or any similar payments payable to Public Company, any of its Affiliates, or their respective successors or assigns, under any Strategic Partner Agreement.
(c) Upon the written request of the Holder Representative provided to Public Company not less than thirty (30) days in advance (such request not to be made more than once in any twelve (12) month period), Public Company shall permit, and shall cause its Affiliates to permit, the Independent Accountant to have access during normal business hours to such of the records of Public Company or any of its Affiliates as may be reasonably necessary to determine whether a CVR Event has occurred and the accuracy of any CVR Event Notice delivered hereunder, including the amount of the Net Proceeds reported by Public Company. Public Company shall, and shall cause its Affiliates to, furnish to the Independent Accountant such access, work papers and other documents and information reasonably necessary for the Independent Accountant to confirm compliance with this Agreement, including the calculation of any Net Proceeds; provided that Public Company may, and may cause its Affiliates to, redact documents and information not relevant for such purposes. The Independent Accountant shall disclose to Public Company and the Holder Representative any matters directly related to its findings to the extent reasonably necessary to verify compliance with the terms of this Agreement. Notwithstanding anything to the contrary, including Section 2.4, if the Independent Accountant concludes that a CVR Event Payment that was properly due was not paid to the Rights Agent, or that any CVR Event Payment made was in an amount less than the amount due, Public Company shall pay the CVR Event Payment or underpayment thereof to the Rights Agent for further distribution to the Holders (such amount being the “CVR Event Payment Shortfall”). The CVR Event Payment Shortfall shall be paid within ten (10) Business Days after the date the Independent Accountant delivers to Public Company and the Holder Representative the Independent Accountant’s written report. The decision of the Independent Accountant shall be final, conclusive and binding on Public Company and the Holders, shall be non-appealable and shall not be subject to further review. The fees charged by the Independent Accountant shall be paid by the Holder Representative (on behalf of the Holders from the Holder Representative Expense Fund, to the extent such funds are available). Each Person seeking to receive information from Public Company or any of its Affiliates in connection with a review pursuant to this Section 4.3 shall enter into a reasonable and mutually satisfactory confidentiality agreement with Public Company or any controlled Affiliate obligating such party to retain all such information disclosed to such party in confidence pursuant to such confidentiality agreement.
Section 4.4 Further Assurances. Public Company hereby agrees that it will perform, execute, acknowledge and deliver or cause to be performed, executed, acknowledged and delivered, all such further and other acts, instruments and assurances as may reasonably be required by the Rights Agent for the carrying out or performing by the Rights Agent of the provisions of this Agreement.
Section 4.5 Covenants.
(a) From and after the Effective Time, except as the Holder Representative shall otherwise consent in writing (in the Holder’s Representative’s sole discretion), Public Company shall, and shall cause each of its Affiliates to maintain, enforce Public Company’s and such Affiliate’s rights under, and comply with all of Public Company’s or such Affiliate’s contractual obligations under, the Strategic Partner Agreements and the TUM Agreement.
(b) From and after the Effective Time, except as the Holder Representative shall otherwise consent in writing (in the Holder’s Representative’s sole discretion), Public Company shall not, and shall cause each of its Affiliates not to:
(1) adopt a plan of, or undertake, any complete or partial liquidation, dissolution, wind down or termination, of Public Company or Pieris Pharmaceuticals GmbH, or otherwise fail to maintain in good standing Public Company and Pieris Pharmaceuticals GmbH;
(2) terminate, assign, or transfer any Strategic Partner Agreement or the TUM Agreement;
(3) amend or modify, unless any such amendment or modification does not adversely affect the interests of the Holders, any Strategic Partner Agreement or the TUM Agreement; or
(4) (i) commence a voluntary case under any foreign, federal or state bankruptcy, insolvency or other similar Law now or hereafter in effect of Public Company or Pieris Pharmaceuticals GmbH, (ii) consent to the entry of an order for relief in an involuntary bankruptcy or similar case, or to the conversion of an involuntary case to a voluntary case, under any such Law of Public Company or Pieris Pharmaceuticals GmbH, (iii) the consent to the appointment of, or the taking of possession by, a receiver, trustee or other custodian for all or a substantial part of its properties of Public Company or Pieris Pharmaceuticals GmbH, or (iv) make any assignment for the benefit of creditors of Public Company or Pieris Pharmaceuticals GmbH.
(c) From and after the Effective Time, except as the Holder Representative shall otherwise consent in writing (in the Holder’s Representative’s sole discretion), Public Company shall, and shall cause each of its Affiliates to, take the steps set forth on Schedule 4.5(c) to pursue the R&D Tax Credit Approval.
(d) Prior to the Effective Time, Public Company will wire on behalf of the Holders US$600,000 (the “Public Company Expense Fund”) to an account established by Public Company, which will be used to pay any reasonable and documented out-of-pocket expenses incurred by Public Company or any of its Affiliates (with the prior written approval of the Holder Representative) in connection with the following (to the extent not taken into account in the calculation of the Exchange Ratio under the Merger Agreement), including without limitation, the expenses set forth on Schedule 4.5(d) (the “Public Company CVR Expenses”): (i) performing its obligations under Section 4.5 of this Agreement, (ii) performing its obligations and enforcing its rights under each Strategic Partner Agreement and the TUM Agreement, and (iii) as reimbursement for indemnification paid to the Holder Representative pursuant to Section 6.2(c) of this Agreement. Public Company will hold these funds separate from its corporate funds and will not voluntarily make these funds available to its creditors in the event of bankruptcy. Public Company may, upon five (5) Business Days’ prior written notice to the Holder Representative, offset Public Company CVR Expenses against any CVR Event Payments then owed, or that may become due and owing, to the Holders hereunder; provided, that such prior written notice shall be delivered to the Holder Representative at least five (5) Business Days’ prior to any such offset and contain a detailed accounting of the Public Company CVR Expenses for which the CVR Event Payments are being offset, along with written documentation evidencing such Public Company CVR Expenses and the payment of such Public Company CVR Expenses by the Company or any of its Affiliates. As soon as practicable following the completion of Public Company’s responsibilities hereunder, Public Company will deliver any remaining balance of the Public Company Expense Fund to the Rights Agent (for pro rata distribution to the Holders).
Section 4.6 No Conflict. Public Company will not, and will cause each of its Subsidiaries not to, enter into any agreement with any Person that is, or otherwise take any actions or inactions, in conflict with this Agreement in any material respect or materially adversely affect the performance of its obligations under this Agreement.
ARTICLE V
AMENDMENTS
Section 5.1 Amendments without Consent of the Holder Representative.
(a) Without the consent of any Holders, the Holder Representative or Public Company, at any time and from time to time, may enter into one or more amendments hereto with the Rights Agent, for any of the following purposes:
(1) to evidence the succession of another Person as a successor Rights Agent and the assumption by any such successor of the covenants and obligations of the Rights Agent herein in compliance with the terms hereof;
(2) to add to the covenants of Public Company such further covenants, restrictions, conditions or provisions as Public Company shall consider to be for the protection of the Holders; provided that, in each case, such provisions do not adversely affect the interests of the Holders;
(3) to cure any ambiguity, to correct or supplement any provision herein that may be defective or inconsistent with any other provision herein, or to make any other provisions with respect to matters or questions arising under this Agreement; provided that, in each case, such provisions do not adversely affect the interests of the Holders;
(4) as may be necessary or appropriate to ensure that the CVRs are not subject to registration under the Securities Act, the Exchange Act or any applicable state securities or “blue sky” Laws; provided that, such amendments do not adversely affect the interests of the Holders;
(5) subject to Section 7.11, to evidence the succession of another Person to Public Company and the assumption by any such successor of the covenants of such party contained herein;
(6) to evidence the assignment of this Agreement by Public Company as provided in Section 7.11; or
(7) any other amendment to this Agreement that would provide any additional rights or benefits to the Holders or that does not adversely affect the rights under this Agreement of any such Holder.
(b) Promptly after the execution and delivery by Public Company and the Rights Agent of any amendment pursuant to the provisions of this Section 5.1, Public Company shall transmit (or cause the Rights Agent to transmit) (i) a notice thereof through the facilities of DTC in accordance with DTC’s procedures and/or by first class mail to the Holders at their addresses as they appear on the CVR Register, and (ii) a notice thereof to the Holder Representative, in each case, setting forth such amendment. Notwithstanding anything to the contrary in this Section 5.1, Public Company may not enter into any amendment to this Agreement affecting the rights or obligations of the Holder Representative without the Holder Representative’s prior written consent.
Section 5.2 Amendments with Consent of the Holder Representative.
(a) Subject to Section 5.1 (which amendments pursuant to Section 5.1 may be made without the consent of the Holders or the Holder Representative), Public Company, the Holder Representative, and the Rights Agent may enter into one or more amendments hereto for the purpose of adding, eliminating or changing any provisions of this Agreement, even if such addition, elimination or change is materially adverse to the interest of the Holders.
(b) Promptly after the execution and delivery by Public Company, the Holder Representative, and the Rights Agent of any amendment pursuant to the provisions of this Section 5.2, Public Company shall transmit (or cause the Rights Agent to transmit) a notice thereof through the facilities of DTC in accordance with DTC’s procedures and/or by first class mail to the Holders at their addresses as they appear on the CVR Register, setting forth such amendment.
Section 5.3 Execution of Amendments. In executing any amendment permitted by this Article V, the Rights Agent shall be entitled to receive, and shall be fully protected in relying upon, an opinion of counsel stating that the execution of such amendment is authorized or permitted by this Agreement. Each amendment to this Agreement shall be evidenced by a writing signed by the Rights Agent. Notwithstanding anything in this Agreement to the contrary, the Rights Agent shall not be required to enter into any such amendment that it has reasonably determined adversely affects its own rights, privileges, covenants or duties under this Agreement or otherwise, and the Rights Agent shall not be bound by amendments not executed by it.
Section 5.4 Effect of Amendments. Upon the execution of any amendment in compliance with this Article V, this Agreement shall be modified in accordance therewith, such amendment shall form a part of this Agreement for all purposes and every Holder shall be bound thereby.
ARTICLE VI
THE HOLDER REPRESENTATIVE
Section 6.1 Appointment. Effective upon the Closing and the issuance of the CVRs under this terms of this Agreement, and without any further act of any of Holders, the Holder Representative is appointed as the representative of the Holders and as the attorney-in-fact and agent for and on behalf of each Holder for purposes of this Agreement and will take such actions to be taken by the Holder Representative under this Agreement and such other actions on behalf of such Holders as it may deem necessary or appropriate in connection with or to consummate the transactions contemplated hereby, including (i) executing and delivering this Agreement and any other ancillary documents and negotiating and executing any amendments, modifications, waivers or changes thereto as to which the Holder Representative, in its sole discretion, has consented (provided that any waiver or amendment that adversely and disproportionately affects the rights or obligations of one or more Holders as compared to other Holders will require the prior written consent of a majority in interest of the disproportionately affected Holders), (ii) agreeing to, negotiating, entering into settlements and compromises of, complying with orders of courts with respect to, and otherwise administering and handling any claims under this Agreement on behalf of such Holders, and (iii) taking all other actions that are either necessary or appropriate in the judgment of the Holder Representative for the accomplishment of the foregoing or contemplated by the terms of this Agreement. The Holder Representative hereby accepts such appointment as of the Closing. The appointment of the Holder Representative as each Holder’s attorney-in-fact revokes any power of attorney heretofore granted that authorized any other Person to represent such Holder with regard to this Agreement and any other agreements or documents executed or delivered in connection with this Agreement. The Holder Representative is the sole and exclusive representative of each of the Holders for any purpose provided for by this Agreement.
Section 6.2 Actions of Holder Representative.
(a) A decision, act, consent or instruction of the Holder Representative hereunder will constitute a decision, act, consent or instruction of all Holders and will be final, binding and conclusive upon each such Holder, and Public Company and the Rights Agent may rely upon any such decision, act, consent or instruction of the Holder Representative as being the decision, act, consent or instruction of each and every such Holder. Public Company and the Rights Agent will be relieved from any liability to any Person for any acts done by them in accordance with such decision, act, consent or instruction of the Holder Representative.
(b) The Holder Representative will incur no liability in connection with its services pursuant to this Agreement and any related agreements except to the extent resulting from its gross negligence, bad faith or willful misconduct. The Holder Representative shall not be liable for any action or omission pursuant to the advice of counsel.
(c) Public Company will indemnify the Holder Representative against any reasonable, documented, and out-of-pocket losses, liabilities and expenses (“Representative Losses”) arising out of or in connection with this Agreement and any related agreements, in each case as such Representative Loss is suffered or incurred; provided, that in the event that any such Representative Loss is finally adjudicated to have been caused by the gross negligence, bad faith or willful misconduct of the Holder Representative, the Holder Representative will reimburse Public Company the amount of such indemnified Representative Loss to the extent attributable to such gross negligence, bad faith or willful misconduct. Representative Losses shall be recovered by the Holder Representative from (i) the funds in the Expense Fund and (ii) any CVR Event Payments owed, or that may become due and owing, to the Holders hereunder; provided, that while the Holder Representative may be paid from the aforementioned sources of funds, this does not relieve Public Company from its obligation to promptly pay such Representative Losses as they are suffered or incurred. In no event will the Holder Representative be required to advance its own funds on behalf of Public Company, the Holders or otherwise. Notwithstanding anything in this Agreement to the contrary, any restrictions or limitations on liability or indemnification obligations of, or provisions limiting the recourse against non-parties otherwise applicable to, the Holders or Public Company set forth elsewhere in this Agreement are not intended to be applicable to the indemnities provided to the Holder Representative hereunder. The foregoing indemnities will survive the Closing, the resignation or removal of the Holder Representative or the termination of this Agreement.
(d) At the Effective Time, Public Company will wire US$250,000 (the “Holder Representative Expense Fund”) to the Holder Representative, which will be used for any expenses incurred by the Holder Representative. Neither Public Company nor the Holders will receive any interest or earnings on the Holder Representative Expense Fund and irrevocably transfer and assign to the Holder Representative any ownership right that they may otherwise have had in any such interest or earnings. The Holder Representative will hold these funds separate from its corporate funds and will not voluntarily make these funds available to its creditors in the event of bankruptcy. As soon as practicable following the completion of the Holder Representative’s responsibilities hereunder, the Holder Representative will deliver any remaining balance of the Holder Representative Expense Fund to the Rights Agent (for pro rata distribution to the Holders). For tax purposes, the Holder Representative Expense Fund will be treated as having been received and voluntarily set aside by the Holders at the time of Closing.
(e) Public Company shall deliver a copy of any and all notices, written instruments and any other information delivered to the Rights Agent hereunder to the Holder Representative concurrently with such delivery to the Rights Agent.
Section 6.3 Resignation and Removal; Appointment of Successor.
(a) The Holder Representative may resign at any time by giving written notice thereof to Public Company and the Rights Agent specifying a date when such resignation shall take effect, which notice shall be sent at least thirty (30) calendar days prior to the date so specified.
(b) At any time the Majority Holders may remove the Holder Representative by specifying a date when such removal shall take effect, but no such removal shall become effective until a successor Holder Representative has been appointed. Notice of such removal shall be given by the Majority Holders to Public Company and the Rights Agent, which notice shall be sent at least thirty (30) calendar days prior to the date so specified.
(c) If the Holder Representative provides notice of its intent to resign pursuant to Section 6.3(a), is removed pursuant to Section 6.3(b) or becomes incapable of acting, the Majority Holders shall, as soon as is reasonably possible, appoint a qualified successor Holder Representative. Notwithstanding the foregoing, if the Majority Holders fail to make such appointment within a period of thirty (30) calendar days after giving notice of such removal or after it has been notified in writing of such resignation or incapacity by the resigning or incapacitated Holder Representative, then the Majority Holders may apply to any court of competent jurisdiction for the appointment of a new Holder Representative. The successor Holder Representative so appointed shall, forthwith upon its acceptance of such appointment in accordance with Section 6.3, become the successor Holder Representative.
(d) The Majority Holders shall give notice of each resignation and each removal of a Holder Representative and each appointment of a successor Holder Representative by mailing written notice of such event by first-class mail to Public company, the Rights Agent and to the Holders as their names and addresses appear in the CVR Register. Each notice shall include the name and address of the successor Holder Representative. If the Majority Holders fail to send such notice within thirty (30) calendar days after acceptance of appointment by a successor Holder Representative, the successor Holder Representative shall cause the notice to be mailed.
(e) The Rights Agent and Public Company will reasonably cooperate with any successor Holder Representative in connection with the transition of the duties and responsibilities of the Holder Representative to the successor Holder Representative but the Rights Agent shall not be required to make any additional expenditure or assume any additional liability in connection with the foregoing.
Section 6.4 Grant of Authority. The grant of authority provided for in this Article VI is coupled with an interest and will be irrevocable and survive the death, incompetency, bankruptcy or liquidation of any Holder. The provisions of this Article VI will be binding upon the executors, heirs, legal representatives, successors and assigns of each Holder, and any references in this Agreement to any Holder or the Holders will mean and include the successors to such Holder’s rights hereunder, whether pursuant to testamentary disposition, the Laws of descent and distribution or otherwise.
ARTICLE VII
MISCELLANEOUS AND GENERAL
Section 7.1 Termination. This Agreement will be terminated and of no force or effect, the Parties will have no liability hereunder (other than with respect to monies due and owing by Public Company to the Rights Agent and the obligations that expressly survive the termination or expiration of this Agreement) and no payments will be required to be made, upon the earlier to occur of: (a) the date on which no Strategic Partner Agreement is in effect and no Strategic Partner Payments are payable under any Strategic Partner Agreement and (b) the delivery of a written notice of termination duly executed by Public Company and the Holder Representative. For the avoidance of doubt, the termination of this Agreement will not affect or limit the right to receive the CVR Event Payments under Section 2.4 to the extent earned prior to termination of this Agreement and the provisions applicable thereto will survive the expiration or termination of this Agreement. The termination of this Agreement will not affect or limit the rights to indemnification in favor of the Holder Representative pursuant to Section 6.2(c) hereof and the protections and immunities of the Rights Agent pursuant to Section 3.3 hereof.
Section 7.2 Notices to Public Company, the Rights Agent and the Holder Representative. Any notice, request, or demand desired or required to be given hereunder will be in writing and will be given by personal delivery, email delivery, or overnight courier service, in each case addressed as respectively set forth below or to such other address as any party hereto will have previously designated by such a notice. The effective date of any notice, request, or demand will be the date of personal delivery, the date on which email is sent (provided that the sender of such email does not receive an automatic and contemporaneous written notification of delivery failure) or one day after it is delivered to a reputable overnight courier service, as the case may be, in each case properly addressed as provided in this Agreement and with all charges prepaid. Any notice, request, or demand delivered by Public Company to the Rights Agent hereunder shall be delivered concurrently to the Holder Representative.
If to Public Company:
Pieris Pharmaceuticals, Inc.
225 Franklin Street, 26th Floor
Boston, MA 02110
Attention: Stephen Yoder, Tom Bures, Karam Hijji
With a copy to (which shall not constitute notice):
Mintz, Levin, Cohn, Ferris, Glovsky and Popeo, P.C.
One Financial Center
Boston, MA 02111
Attention: William C. Hicks, Marc D. Mantell, Scott Dunberg
If to the Rights Agent:
Computershare Inc. and Computershare Trust Company, N.A.
150 Royall St.
Canton, MA 02021
Attention: Relationship Manager
If to the Holder Representative:
Shareholder Representative Services LLC
950 17th Street, Suite 1400
Denver, CO 80202
Attention: Managing Director
Telephone: (303) 648-4085
Section 7.3 Notice to Holders. Where this Agreement provides for notice to Holders, such notice shall be sufficiently given (unless otherwise herein expressly provided) if in writing and transmitted through the facilities of DTC in accordance with DTC’s procedures or mailed, first-class postage prepaid, to each Holder affected by such event, at the Holder’s address as it appears in the CVR Register, not later than the latest date, and not earlier than the earliest date, if any, prescribed for the giving of such notice. In any case where notice to Holders is given by mail, neither the failure to mail such notice, nor any defect in any notice so mailed, to any particular Holder shall affect the sufficiency of such notice with respect to other Holders.
Section 7.4 Governing Law. This Agreement shall be governed by, and construed in accordance with, the Laws of the State of Delaware, regardless of the Laws that might otherwise govern under applicable principles of conflicts of Laws thereof.
Section 7.5 Specific Enforcement; Jurisdiction.
(a) Public Company and Holder Representative acknowledge and agree that irreparable damage would occur in the event that any of the provisions of this Agreement were not performed in accordance with its specific terms or were otherwise breached, and that monetary damages, even if available, would not be an adequate remedy therefor. It is accordingly agreed that Public Company and Holder Representative shall be entitled to an injunction or injunctions, or any other appropriate form of equitable relief, to prevent breaches of this Agreement and to enforce specifically the performance of the terms and provisions of this Agreement in any court referred to in Section 7.5(b), without proof of damages or otherwise (and each party hereto hereby waives any requirement for the securing or posting of any bond in connection with such remedy), this being in addition to any other remedy to which they are entitled at Law or in equity. Public Company and Holder Representative further agree not to assert that a remedy of specific enforcement is unenforceable, invalid, contrary to Law or inequitable for any reason, nor to assert that a remedy of monetary damages would provide an adequate remedy. Public Company and Holder Representative acknowledge and agree that the right of specific enforcement is an integral part of this Agreement and without such right, none of the parties hereto would have entered into this Agreement.
(b) Each of the parties hereto hereby irrevocably submits to the exclusive jurisdiction of the courts of the State of Delaware and to the jurisdiction of the United States District Court for the State of Delaware, for the purpose of any Proceeding arising out of or relating to this Agreement or the actions of the parties hereto in the negotiation, administration, performance and enforcement thereof, and each of the parties hereto hereby irrevocably agrees that all claims with respect to such Proceeding may be heard and determined exclusively in the Delaware Court of Chancery or, solely if the Delaware Court of Chancery does not have subject matter jurisdiction thereof, any other court of the State of Delaware or any federal court sitting in the State of Delaware. Each of the parties hereto (i) consents to submit itself to the personal jurisdiction of the Delaware Court of Chancery, any other court of the State of Delaware and any federal court sitting in the State of Delaware in the event any Proceeding arises out of this Agreement, (ii) agrees that it will not attempt to deny or defeat such personal jurisdiction by motion or other request for leave from any such court, (iii) irrevocably consents to the service of process in any Proceeding arising out of or relating to this Agreement, on behalf of itself or its property, by U.S. registered mail to such party’s respective address set forth in Section 7.2 (provided that nothing in this Section 7.5(b) shall affect the right of any party hereto to serve legal process in any other manner permitted by Law) and (iv) agrees that it will not bring any Proceeding relating to this Agreement in any court other than the Delaware Court of Chancery (or, solely if the Delaware Court of Chancery shall be unavailable, any other court of the State of Delaware or any federal court sitting in the State of Delaware). The parties hereto agree that a final trial court judgment in any such Proceeding shall be conclusive and may be enforced in other jurisdictions by suit on the judgment or in any other manner provided by Law; provided that nothing in the foregoing shall restrict any party’s rights to seek any post-judgment relief regarding, or any appeal from, such final trial court judgment.
Section 7.6 WAIVER OF JURY TRIAL. EACH PARTY HERETO HEREBY WAIVES, TO THE FULLEST EXTENT PERMITTED BY APPLICABLE LAW, ANY RIGHT IT MAY HAVE TO A TRIAL BY JURY IN RESPECT OF ANY PROCEEDING ARISING OUT OF THIS AGREEMENT. EACH PARTY HERETO (A) CERTIFIES THAT NO REPRESENTATIVE, AGENT OR ATTORNEY OF ANY OTHER PARTY HAS REPRESENTED, EXPRESSLY OR OTHERWISE, THAT SUCH PARTY WOULD NOT, IN THE EVENT OF ANY PROCEEDING, SEEK TO ENFORCE THE FOREGOING WAIVER AND (B) ACKNOWLEDGES THAT IT AND THE OTHER PARTIES HERETO HAVE BEEN INDUCED TO ENTER INTO THIS AGREEMENT BY, AMONG OTHER THINGS, THE MUTUAL WAIVER AND CERTIFICATIONS IN THIS SECTION 7.6.
Section 7.7 Other Remedies. Except as otherwise provided herein, any and all remedies herein expressly conferred upon a Party will be deemed cumulative with and not exclusive of any other remedy conferred hereby, or by Law or equity upon such Party, and the exercise by a Party of any one remedy will not preclude the exercise of any other remedy.
Section 7.8 Entire Agreement; Counterparts. As between the Public Company and Holder Representative, this Agreement, the Merger Agreement and the other agreements, exhibits, annexes and schedules referred to herein constitute the entire agreement and supersede all prior agreements and understandings, both written and oral, between such parties, with respect to the subject matter hereof and thereof. As among the Public Company, Holder Representative, and the Rights Agent, this Agreement and any schedule or exhibit attached hereto contains the entire understanding of such parties with reference to the transactions and matters contemplated hereby and supersedes all prior agreements, written or oral, among the parties with respect hereto and thereto. If and to the extent that any provision of this Agreement is inconsistent or conflicts with the Merger Agreement with respect to the rights, duties, protections and liabilities of the Rights Agent, this Agreement will govern and be controlling. This Agreement and any signed agreement or instrument entered into in connection with this Agreement may be executed in two or more counterparts (including by facsimile or by an electronic scan delivered by electronic mail), each of which shall be deemed an original but all of which together shall be considered one and the same agreement and shall become effective when counterparts have been signed by each of the parties hereto and delivered to the other parties, it being understood that all parties need not sign the same counterpart. To the extent signed and delivered by means of a facsimile machine or telecopy, by email delivery of a “.pdf” or “.jpg” format data file or by any electronic signature complying with the U.S. federal ESIGN Act of 2000, this Agreement shall be treated in all manner and respects as an original agreement or instrument and shall be considered to have the same binding legal effect as if it were the original signed version thereof delivered in person. No party hereto or to any such agreement or instrument shall raise the use of a facsimile machine or telecopy, email delivery of a “.pdf” or “.jpg” format data file or electronic signature complying with the U.S. federal ESIGN Act of 2000 to deliver a signature to this Agreement or any amendment hereto or the fact that any signature or agreement or instrument was transmitted or communicated through the use of a facsimile machine or telecopy, email delivery of a “.pdf” or “.jpg” format data file or by any electronic signature complying with the U.S. federal ESIGN Act of 2000 as a defense to the formation of a contract and each party hereto forever waives any such defense.
Section 7.9 Third-Party Beneficiaries; Action by the Holder Representative. Public Company, the Holder Representative, and the Rights Agent hereby agree that the respective covenants and agreements set forth herein are intended to be for the benefit of all Holders, who are intended third-party beneficiaries hereof, and shall be enforceable by the Holder Representative on their behalf. Public Company, the Holder Representative, and the Rights Agent further agree that this Agreement and their respective covenants and agreements set forth herein are solely for the benefit of Public Company, the Rights Agent, the Holder Representative, and the Holders and their respective permitted successors and assigns hereunder in accordance with and subject to the terms of this Agreement, and nothing in this Agreement, express or implied, will confer upon any Person other than Public Company, the Rights Agent, the Holder Representative, the Holders and their respective permitted successors and assigns hereunder any benefit or any legal or equitable right, remedy or claim hereunder. The rights of Holders and their successors and assigns pursuant to Permitted Transfers are limited to those expressly provided in this Agreement and the Merger Agreement. Notwithstanding anything to the contrary contained herein, any Holder or Holder’s successor or assign pursuant to a Permitted Transfer may agree to renounce, in whole or in part, its rights under this Agreement by written notice to the Holder Representative, Rights Agent and Public Company, which notice, if given, shall be irrevocable. The Holder Representative will have the right, on behalf of all Holders, by virtue of or under any provision of this Agreement, to institute a Proceeding with respect to this Agreement and no individual Holder or other group of Holders, nor the Rights Agent on behalf thereof, will be entitled to exercise such rights. In the event of an insolvency proceeding of Public Company or any of its Affiliates, individual Holders shall be entitled to assert claims in such insolvency proceeding and take related actions in pursuit of such claims with respect to any payment that may be claimed by or on behalf of Public Company or by any creditor of Public Company, as applicable. Except for the right of the Rights Agent set forth in this Agreement or as set forth in the preceding sentence, the Holder Representative will have the sole right, on behalf of all Holders, by virtue of or under any provision of this Agreement, to institute any action or Proceeding at Law or in equity or in bankruptcy or otherwise upon or under or with respect to this Agreement, and no individual Holder or other group of Holders will be entitled to exercise such rights. Notwithstanding any other provision in this Agreement, the right of any Holder to receive payment of the applicable CVR Payment Amounts upon the occurrence of a CVR Event shall not be impaired or adversely affected without the consent of such Holder.
Section 7.10 Severability. Any term or provision of this Agreement that is invalid or unenforceable in any situation in any jurisdiction shall not affect the validity or enforceability of the remaining terms and provisions of this Agreement or the validity or enforceability of the offending term or provision in any other situation or in any other jurisdiction. If a final judgment of a court of competent jurisdiction declares that any term or provision of this Agreement is invalid or unenforceable, the Parties hereto agree that the court making such determination shall have the power to limit such term or provision, to delete specific words or phrases or to replace such term or provision with a term or provision that is valid and enforceable and that comes closest to expressing the intention of the invalid or unenforceable term or provision, and this Agreement shall be valid and enforceable as so modified. In the event such court does not exercise the power granted to it in the prior sentence, the Parties hereto agree to replace such invalid or unenforceable term or provision with a valid and enforceable term or provision that will achieve, to the extent possible, the economic, business and other purposes of such invalid or unenforceable term or provision. Notwithstanding anything to the contrary herein, if an excluded provision shall affect the rights, immunities, liabilities, duties or obligations of the Rights Agent, the Rights Agent shall be entitled to resign immediately upon written notice to Public Company.
Section 7.11 Assignment. This Agreement shall not be assignable without the prior written consent of each of the other Parties to this Agreement; provided, however, (a) Public Company may assign any or all of its rights, interests and obligations under this Agreement to a Person that is a controlled Affiliate of Public Company without the consent of any other party; provided that Public Company remains jointly and severally liable for all of the obligations of Public Company under this Agreement, (b) Public Company may assign any or all of its rights, interests and obligations under this Agreement in connection with a sale of all or substantially all of the assets of Public Company; provided that the successor in any such sale transaction expressly assumes all of the obligations of Public Company under this Agreement, (c) Public Company may assign any or all of its rights, interests and obligations under this Agreement to any Person with the prior written consent of the Holder Representative, and (d) the Rights Agent may, without the consent of any other party, assign this Agreement to a successor Rights Agent appointed in compliance with Section 3.4. The Rights Agent, Public Company and the Holder Representative agree that all books, records, information and data pertaining to the business of the other party, including inter alia, personal, non-public Holder information, which are exchanged or received pursuant to the negotiation or the carrying out of this Agreement including the fees for services set forth in the attached schedule shall remain confidential, and shall not be voluntarily disclosed to any other person, except as may be required by law, including, without limitation, pursuant to subpoenas from state or federal government authorities (e.g., in divorce and criminal actions).
Section 7.12 Confidentiality. The Rights Agent, Public Company and the Holder Representative agree that all books, records, information and data pertaining to the business of the other parties, including inter alia, personal, non-public Holder information, and the fees for the services of the Rights Agent and the Holder Representative, which are exchanged or received pursuant to the negotiation or the carrying out of this Agreement shall be confidential and shall not be used for any purpose other than carrying out their respective duties under this Agreement and shall not be voluntarily disclosed to any other Person, including any Holder, except as may be required by a valid order of any Governmental Entity of competent jurisdiction or is otherwise required by applicable Law, the rules and regulations of the Securities and Exchange Commission or any stock exchange on which the securities of the disclosing party are listed, or pursuant to subpoenas from state or federal Governmental Entities (subject to (x) the disclosing party notifying, to the extent practicable, the other parties hereto of such potential disclosure reasonably in advance of such disclosure, or (y) cooperating with the other parties, at the expense of the other parties, in any effort to restrict disclosure of such book, records, information). Notwithstanding anything herein or in Section 7.11 to the contrary, following Closing, the Holder Representative shall be permitted to disclose information to advisors and representatives of the Holder Representative and to the Holders, in each case who have a need to know such information, provided that such persons are subject to confidentiality obligations with respect thereto.
Section 7.13 No Obligation. Except as expressly set forth in this Agreement, in no event shall Public Company or any of its Affiliates be required to undertake any level of efforts, or employ any level of resources, to ensure that a CVR Event Payment occurs; provided, that none of Public Company, its Affiliates or any of their respective successors and assigns shall take any action or fail to take any action in bad faith for the purpose of avoiding the occurrence of any CVR Event or the payment of (or minimizing or reducing the amount of) any CVR Event Payment.
Section 7.14 Force Majeure. Notwithstanding anything to the contrary contained herein, no party hereto will have any liability for not performing, or a delay in the performance of, any act, duty, obligation or responsibility by reason of any occurrence beyond the reasonable control of such party (including any act or provision or any present or future law or regulation or governmental authority, any act of God, epidemics, pandemics, war, civil or military disobedience or disorder, riot, rebellion, terrorism, insurrection, fire, earthquake, storm, flood, strike, work stoppage, interruptions or malfunctions of computer facilities, loss of data due to power failures or mechanical difficulties, labor dispute, accident or failure or malfunction of any utilities communication or computer services or similar occurrence).
[Remainder of Page Intentionally Left Blank; Signature Page Follows]
IN WITNESS WHEREOF, this Agreement has been duly executed and delivered by the duly authorized officers of the Parties as of the date first written above.
|
|
PIERIS PHARMACEUTICALS, INC. |
|
|
|
|
|
|
|
|
|
|
|
By: |
|
|
|
Name: |
|
|
|
Title: |
Chief Executive Officer |
|
|
POLO MERGER SUB, INC. |
|
|
|
|
|
|
|
|
|
|
|
By: |
|
|
|
Name: |
|
|
|
Title: |
|
|
|
COMPUTERSHARE INC. and COMPUTERSHARE TRUST COMPANY, N.A., jointly as Rights Agent |
|
|
|
|
|
|
|
|
|
|
|
By: |
|
|
|
Name: |
|
|
|
Title: |
|
|
|
SHAREHOLDER REPRESENTATIVE SERVICES LLC |
|
|
|
|
|
|
|
|
|
|
|
By: |
|
|
|
Name: |
|
|
|
Title: |
|
Schedule 4.5(c)
R&D Tax Credit Actions
Schedule 4.5(d)
Public Company CVR Expenses
Schedule 1
Rights Agent Fees and Expenses
ANNEX E
FORM OF PIERIS SUPPORT AGREEMENT
This Support Agreement (this “Agreement”) is made and entered into as of July 23, 2024, by and among Palvella Therapeutics, Inc. a Delaware corporation (“Merger Partner”), Pieris Pharmaceuticals, Inc., a Nevada corporation (“Public Company”), and the undersigned stockholder (the “Stockholder”) of Public Company.
RECITALS
WHEREAS, concurrently with or following the execution and delivery hereof, Public Company, Merger Partner and Polo Merger Sub, Inc., a Delaware corporation and a wholly owned subsidiary of Public Company (the “Merger Sub”), have entered into an agreement and plan of merger (as such agreement may be amended or supplemented from time to time pursuant to the terms thereof, the “Merger Agreement”), pursuant to which Merger Sub will merge with and into Merger Partner, with Merger Partner surviving the merger as the surviving corporation and a wholly owned subsidiary of Public Company (the “Merger”).
WHEREAS, as of the date hereof, the Stockholder is the beneficial owner (as defined in Rule 13d-1 under the Exchange Act) of such number of shares of Public Company Common Stock as indicated in Appendix A.
WHEREAS, as an inducement to the willingness of Merger Partner to enter into the Merger Agreement, Merger Partner has required that the Stockholder enter into this Agreement.
NOW, THEREFORE, intending to be legally bound, the parties hereby agree as follows:
1. Certain Definitions. Capitalized terms used but not otherwise defined herein shall have the meanings ascribed thereto in the Merger Agreement. For all purposes of this Agreement, the following terms shall have the following respective meanings:
(a) “Constructive Sale” means, with respect to any security, a short sale with respect to such security, entering into or acquiring a derivative contract with respect to such security, entering into or acquiring a futures or forward contract to deliver such security or entering into any other hedging or other derivative transaction that has the effect of either directly or indirectly materially changing the economic benefits or risks of ownership of such security.
(b) “Shares” means (i) all shares of Public Company Common Stock and Public Company Preferred Stock owned, beneficially or of record, by the Stockholder as of the date hereof, and (ii) all additional shares of Public Company Common Stock and Public Company Preferred Stock acquired by the Stockholder, beneficially or of record, during the period commencing with the execution and delivery of this Agreement and expiring on the Closing Date.
(c) “Transfer” or “Transferred” means, with respect to any security, the direct or indirect assignment, sale, transfer, tender, exchange, pledge or hypothecation, or the grant, creation or suffrage of a lien, security interest or encumbrance in or upon, or the gift, grant or placement in trust, or the Constructive Sale or other disposition of such security (including transfers by testamentary or intestate succession, by domestic relations order or other court order, or otherwise by operation of law) or any right, title or interest therein (including any right or power to vote to which the holder thereof may be entitled, whether such right or power is granted by proxy or otherwise), or the record or beneficial ownership thereof, the offer to make such a sale, transfer, Constructive Sale or other disposition, and each agreement, arrangement or understanding, whether or not in writing, to effect any of the foregoing.
2. Transfer and Voting Restrictions. The Stockholder covenants to Merger Partner and Public Company as follows:
(a) Except as otherwise permitted by Section 2(c), during the period commencing with the execution and delivery of this Agreement and expiring on the Expiration Date (as defined below), the Stockholder shall not Transfer any of the Stockholder’s Shares, or publicly announce its intention to Transfer any of its Shares.
(b) Except as otherwise permitted by this Agreement or by order of a court of competent jurisdiction or a Governmental Entity, the Stockholder will not commit any act that would restrict the Stockholder’s legal power, authority and right to vote all of the Shares held by the Stockholder or otherwise prevent or disable the Stockholder from performing any of his, her or its obligations under this Agreement. Without limiting the generality of the foregoing, except for this Agreement and as otherwise permitted by this Agreement, the Stockholder shall not enter into any voting agreement with any person or entity with respect to any of the Stockholder’s Shares, grant any person or entity any proxy (revocable or irrevocable) or power of attorney with respect to any of the Shares, deposit any Shares in a voting trust or otherwise enter into any agreement or arrangement with any person or entity limiting or affecting the Stockholder’s legal power, authority or right to vote the Stockholder’s Shares in favor of the Required Public Company Voting Proposal and the Other Public Company Voting Proposals.
(c) Notwithstanding anything else herein to the contrary, the Stockholder may, at any time, Transfer the Stockholder’s Shares (i) by will or other testamentary document or by intestacy, (ii) to any Affiliate of Stockholder or any investment fund or other entity controlled or managed by the Stockholder or a controlling Affiliate of Stockholder, (iii) to any member of the Stockholder’s immediate family, (iv) in the case of a Stockholder who is not a natural person, by pro rata distributions from the Stockholder to its members, partners, or shareholders pursuant to the Stockholder’s organizational documents, (v) to any trust for the direct or indirect benefit of the Stockholder or the immediate family of the Stockholder or otherwise for tax or estate planning purposes, or (vi) by operation of law; provided, that (x) such Transferred Shares shall continue to be bound by this Agreement and (y) the applicable transferee shall have executed and delivered to Public Company and Merger Partner a support agreement substantially similar to this Agreement upon consummation of such Transfer.
3. No Obligation to Exercise. Notwithstanding anything to the contrary herein, nothing in this Agreement shall obligate the Stockholder to exercise any option or any other right to acquire any shares of Public Company Common Stock.
4. Agreement to Vote Shares. The Stockholder covenants to Merger Partner as follows:
(a) Until the Expiration Date, at any meeting of the stockholders of Public Company, however called, and at every adjournment or postponement thereof, and on every action or approval by written consent of the stockholders of Public Company, the Stockholder shall be present (in person or by proxy) and vote, or exercise its right to consent with respect to, all Shares held by the Stockholder (i) in favor of the Required Public Company Voting Proposals and the Other Public Company Voting Proposals and (ii) against any Acquisition Proposal.
(b) If the Stockholder is the beneficial owner, but not the record holder, of Shares, the Stockholder shall cause the record holder and any nominees to be present (in person or by proxy) and vote all the Stockholder’s Shares in accordance with this Section 4.
(c) In the event of a stock split, stock dividend or distribution, or any change in the capital stock of Public Company by reason of any split-up, reverse stock split, recapitalization, combination, reclassification, reincorporation, exchange of shares or the like, the term “Shares” shall be deemed to refer to and include such shares as well as all such stock dividends and distributions and any securities into which or for which any or all of such shares may be changed or exchanged or which are received in such transaction.
5. Action in Stockholder Capacity Only. The Stockholder is entering into this Agreement solely in the Stockholder’s capacity as a record holder and beneficial owner, as applicable, of its Shares and not in the Stockholder’s capacity as a director or officer of Public Company. Nothing herein shall limit or affect the Stockholder’s ability to act as an officer or director of Public Company.
6. Documentation and Information. The Stockholder shall permit and hereby authorizes Public Company and Merger Partner to publish and disclose in all documents and schedules filed with the SEC, and any press release or other disclosure document that Public Company or Merger Partner reasonably determines to be necessary in connection with the transactions contemplated by the Merger Agreement, such Stockholder’s identity and ownership of the Shares and the nature of such Stockholder’s commitments and obligations under this Agreement.
7. Irrevocable Proxy. The Stockholder hereby revokes (or agrees to cause to be revoked) any proxies that the Stockholder has heretofore granted with respect to its Shares. In the event and to the extent that the Stockholder fails to vote the Shares in accordance with Section 4 at any applicable meeting of the stockholders of Public Company or pursuant to any applicable written consent of the stockholders of Public Company, the Stockholder shall, solely with respect to the matters described in Section 4, be deemed to have irrevocably granted to, and appointed, Merger Partner, and any individual designated in writing by Merger Partner, and each of them individually, as his, her or its proxy and attorney-in-fact (with full power of substitution), for and in its name, place and stead, to vote his, her or its Shares in any action by written consent of Public Company stockholders or at any meeting of the Public Company stockholders called with respect to any of the matters specified in, and in accordance and consistent with, Section 4 of this Agreement. Merger Partner agrees not to exercise the proxy granted herein for any purpose other than the purposes described in this Agreement. Except as otherwise provided for herein, the Stockholder hereby affirms that the irrevocable proxy is coupled with an interest and may under no circumstances be revoked and that such irrevocable proxy is executed and intended to be irrevocable. Notwithstanding any other provisions of this Agreement, the irrevocable proxy granted hereunder shall automatically terminate upon the termination of this Agreement.
8. Representations and Warranties.
(a) Representations and Warranties of the Stockholder. The Stockholder hereby represents and warrants to Merger Partner and Public Company as follows:
(i) The Stockholder (A) is the beneficial or record owner of the shares of Public Company Common Stock and Public Company Preferred Stock, as applicable, indicated in Appendix A (each of which shall be deemed to be “held” by the Stockholder for purposes of Section 4 unless otherwise expressly stated with respect to any shares in Appendix A), free and clear of any and all Liens (other than any Liens that may exist pursuant to applicable securities laws) and (B) does not beneficially own any securities of Public Company other than the shares of Public Company Common Stock and Public Company Preferred Stock and rights to purchase shares of Public Company Common Stock and Public Company Preferred Stock set forth in Appendix A.
(ii) Except as otherwise provided in this Agreement, the Stockholder has full power and authority to (A) make, enter into and carry out the terms of this Agreement and (B) vote all of its Shares in the manner set forth in this Agreement without the consent or approval of, or any other action on the part of, any other person or entity (including any Governmental Entity). Without limiting the generality of the foregoing, the Stockholder has not entered into any voting agreement (other than this Agreement) with any person with respect to any of the Stockholder’s Shares, granted any person any proxy (revocable or irrevocable) or power of attorney with respect to any of the Stockholder’s Shares, pledged or disposed of any of the Stockholder’s Shares, deposited any of the Stockholder’s Shares in a voting trust or entered into any arrangement or agreement with any person limiting or affecting the Stockholder’s legal power, authority or right to vote the Stockholder’s Shares on any matter.
(iii) This Agreement has been duly and validly executed and delivered by the Stockholder and (assuming the due authorization, execution and delivery by the other parties hereto) constitutes a valid and binding agreement of the Stockholder enforceable against the Stockholder in accordance with its terms, subject to the Bankruptcy and Equity Exception. If (i) the Stockholder is a corporation, partnership or other investment or business entity, the Stockholder is duly organized, validly existing and in good standing under the Laws of the jurisdiction in which it is organized, and the execution, delivery and performance of this Agreement by the Stockholder and the consummation by the Stockholder of the transactions contemplated hereby are within the corporate power and capacity of the Stockholder and have been duly authorized by all necessary corporate action of the Stockholder and (ii) the Stockholder is an individual, the signature on this Agreement is genuine, and the Stockholder has full power and authority and legal competence and capacity to execute this Agreement, to perform fully the Stockholder’s obligations hereunder, and to consummate the transactions contemplated hereby. The execution and delivery of this Agreement by the Stockholder and the performance by the Stockholder of the agreements and obligations hereunder will not result in any breach or violation of or be in conflict with or constitute a default under any term of any Contract or if applicable any provision of an organizational document (including a certificate of incorporation) to or by which the Stockholder is a party or bound, or any applicable law to which the Stockholder is subject or bound, except for any such breach, violation, conflict or default which, individually or in the aggregate, would not reasonably be expected to materially impair or adversely affect the Stockholder’s ability to perform its obligations under this Agreement.
(iv) The Stockholder has had the opportunity to discuss the Merger Agreement, this Agreement and the transactions contemplated hereby and thereby with the Stockholder’s legal counsel. The Stockholder understands and acknowledges that Merger Partner is entering into the Merger Agreement in reliance upon the Stockholder’s execution, delivery and performance of this Agreement.
(v) The execution, delivery and performance of this Agreement by the Stockholder do not and will not require any consent, approval, authorization or permit of, action by, filing with or notification to, any Governmental Entity, except for any such consent, approval, authorization, permit, action, filing or notification the failure of which to make or obtain, individually or in the aggregate, has not and would not materially impair the Stockholder’s ability to perform its obligations under this Agreement.
(vi) As of the date hereof, there is no action, suit, investigation or proceeding (whether judicial, arbitral, administrative, or otherwise) pending against, or, to the knowledge of the Stockholder, threatened against, the Stockholder or any of the Stockholder’s properties or assets (including the Shares) that would reasonably be expected to prevent or materially delay or impair the ability of the Stockholder to perform its obligations hereunder or to consummate the transactions contemplated hereby.
(vii) Neither the Stockholder nor any of its Representatives or Affiliates (excluding, for the avoidance of doubt, the Public Company) has employed or made any agreement with any broker, finder or similar agent or any Person which will result in the obligation of such Stockholder, Public Company, Merger Partner, or any of their respective Affiliates to pay any finder’s fee, brokerage fees or commission or similar payment in connection with the transactions contemplated hereby.
(b) Representations of the Merger Partner. The Merger Partner hereby represents and warrants to the Stockholder as follows: (i) it is a corporation duly organized, validly existing and in good standing under the Laws of the jurisdiction in which it is organized (in the case of good standing, to the extent the concept is recognized by such jurisdiction); (ii) it has all requisite corporate power and authority to enter into and deliver this Agreement and to perform its obligations hereunder; (iii) the execution and delivery by it of this Agreement and the consummation of the transactions contemplated hereby have been duly authorized by all necessary corporate action on the part of the Merger Partner; and (d) this Agreement constitutes a legal, valid and binding obligation of the Merger Partner, enforceable against it in accordance with its terms (except insofar as such enforceability may be limited by the Bankruptcy and Equity Exception).
(c) Representations of the Public Company. The Public Company hereby represents and warrants to the Stockholder as follows: (i) it is a corporation duly organized, validly existing and in good standing under the Laws of the jurisdiction in which it is organized (in the case of good standing, to the extent the concept is recognized by such jurisdiction); (ii) it has all requisite corporate power and authority to enter into and deliver this Agreement and to perform its obligations hereunder; (iii) the execution and delivery by it of this Agreement and the consummation of the transactions contemplated hereby have been duly authorized by all necessary corporate action on the part of the Public Company; and (d) this Agreement constitutes a legal, valid and binding obligation of the Public Company, enforceable against it in accordance with its terms (except insofar as such enforceability may be limited by the Bankruptcy and Equity Exception).
9. Termination. This Agreement shall terminate and shall cease to be of any further force or effect as of the earlier of (a) such date and time as the Merger Agreement shall have been terminated pursuant to the terms thereof, (b) the Effective Time, or (c) the date a Public Company Board Recommendation Change or a Merger Partner Board Recommendation Change is made (the “Expiration Date”); provided, however, that (x) Section 10 shall survive the termination of this Agreement, and (y) the termination of this Agreement shall not relieve any party hereto from any liability for any material and willful breach of this Agreement prior to the Expiration Date. A “material and willful breach” by a party of a provision of this Agreement means that the party knowingly undertook an action, or knowingly failed to undertake an action, with the understanding that the action, or failure to act, was a material breach by such party of the applicable provisions of this Agreement.
10. Miscellaneous Provisions.
(a) Amendments. No amendment of this Agreement shall be effective against any party unless it shall be in writing and signed by each of the parties hereto. In the event Merger Partner agrees to amend or waive the terms and conditions of any Support Agreement it has entered into with any other stockholder of the Public Company, the result of which would make the terms and conditions of such Support Agreement more favorable to such stockholder than the terms and conditions hereof are to the Stockholder, then the Merger Partner and Public Company will offer to amend or waive the terms and conditions of this Agreement so they are no less favorable to the Stockholder than the terms and conditions of such other Support Agreement are to such other stockholder.
(b) Entire Agreement. This Agreement constitutes the entire agreement between the parties to this Agreement and supersedes all other prior agreements, arrangements and understandings, both written and oral, among the parties with respect to the subject matter hereof.
(c) Governing Law. This Agreement and all matters, claims, counterclaims, or causes of action (whether in contract, tort, statute, or otherwise) arising out of or relating to this Agreement and the transactions contemplated hereby (including its interpretation, construction, performance and enforcement), or the actions of any party in the negotiation, administration, performance, or enforcement of this Agreement (collectively, the “Relevant Matters”) shall be governed by and construed in accordance with the internal laws of the State of Delaware without giving effect to any choice or conflict of Law provision or rule (whether of the State of Delaware or any other jurisdiction) that would cause the application of laws of any jurisdictions other than those of the State of Delaware. For the avoidance of doubt, all matters relating to the internal affairs of Public Company (including the fiduciary duties of its directors and officers) shall be governed by the internal Laws of the State of Nevada without giving effect to any choice or conflict of Law provision or rule (whether of the State of Nevada or any other jurisdiction) that would cause the application of Laws of any jurisdictions other than those of the State of Nevada.
(d) Jurisdiction. Each of the parties to this Agreement (i) consents to submit itself to the exclusive personal jurisdiction of the Court of Chancery of the State of Delaware, New Castle County, or, if that court does not have jurisdiction, a state or federal court sitting in Wilmington, Delaware in any action or proceeding arising out of or relating to any Relevant Matter, (ii) agrees that all claims in respect of such action or proceeding shall be heard and determined in any such court, (iii) agrees that it shall not attempt to deny or defeat such personal jurisdiction by motion or other request for leave from any such court and (iv) agrees not to bring any action or proceeding arising out of or relating to any Relevant Matter in any other court. Each of the parties hereto waives any defense of inconvenient forum to the maintenance of any action or proceeding so brought and waives any bond, surety or other security that might be required of any other party with respect thereto. Any party may make service on another party by sending or delivering a copy of the process to the party to be served at the address and in the manner provided for the giving of notices in Section 10(j). Nothing in this Section 10(d), however, shall affect the right of any party to serve legal process in any other manner permitted by law.
(e) WAIVER OF JURY TRIAL. EACH OF THE PARTIES TO THIS AGREEMENT HEREBY IRREVOCABLY WAIVES ALL RIGHT TO TRIAL BY JURY IN ANY ACTION OR PROCEEDING ARISING OUT OF OR RELATING TO ANY RELEVANT MATTER.
(f) Assignment. Except as otherwise provided in Section 2(c) hereof, no party may assign any of its rights or delegate any of its performance obligations under this Agreement, in whole or in part, by operation of law or otherwise, without the prior written consent of the other parties hereto, and any such assignment without such prior written consent shall be null and void. Subject to the preceding sentence, this Agreement shall be binding upon, inure to the benefit of, and be enforceable by, the parties hereto and their respective successors and permitted assigns. Any purported assignment of rights or delegation of performance obligations in violation of this Section 10(f) is void.
(g) No Third Party Rights. This Agreement is not intended to, and shall not, confer upon any other person any rights or remedies hereunder other than the parties hereto to the extent expressly set forth herein.
(h) Severability. Any term or provision of this Agreement that is invalid or unenforceable in any situation in any jurisdiction shall not affect the validity or enforceability of the remaining terms and provisions hereof or the validity or enforceability of the offending term or provision in any other situation or in any other jurisdiction. If the final judgment of a court of competent jurisdiction declares that any term or provision hereof is invalid or unenforceable, the parties hereto agree that the court making such determination shall have the power to limit the term or provision, to delete specific words or phrases, or to replace any invalid or unenforceable term or provision with a term or provision that is valid and enforceable and that comes closest to expressing the intention of the invalid or unenforceable term or provision, and this Agreement shall be enforceable as so modified. In the event such court does not exercise the power granted to it in the prior sentence, the parties hereto agree to replace such invalid or unenforceable term or provision with a valid and enforceable term or provision that will achieve, to the extent possible, the economic, business and other purposes of such invalid or unenforceable term.
(i) Specific Performance. The parties hereto agree that irreparable damage would occur in the event that any of the provisions of this Agreement were not performed in accordance with their specific terms or were otherwise breached. It is accordingly agreed that the parties shall be entitled to an injunction or injunctions to prevent breaches of this Agreement and to enforce specifically the terms and provisions of this Agreement, this being in addition to any other remedy to which they are entitled at law or in equity.
(j) Notices. All notices and other communications hereunder shall be in writing and shall be deemed duly delivered (i) three Business Days after being sent by registered or certified mail, return receipt requested, postage prepaid, or (ii) one Business Day after being sent for next Business Day delivery, fees prepaid, via a reputable overnight courier service, in each case to the intended recipient as follows: (A) if to Merger Partner or Public Company, to the address, electronic mail address or facsimile provided in the Merger Agreement, including to the persons designated therein to receive copies; and/or (B) if to the Stockholder, to the Stockholder’s address, electronic mail address or facsimile shown below Stockholder’s signature to this Agreement.
(k) Counterparts. This Agreement may be executed in two or more counterparts (including by facsimile, by an electronic scan delivered by electronic mail or any electronic signature), each of which shall be deemed an original but all of which together shall be considered one and the same agreement and shall become effective when counterparts have been signed by each of the parties hereto and delivered to the other parties, it being understood that all parties need not sign the same counterpart. This Agreement may be executed and delivered by facsimile, by an electronic scan delivered by electronic mail or by delivery of any electronic signature.
(l) Confidentiality. Except to the extent required by applicable Law or regulation, the Stockholder shall hold any non-public information regarding this Agreement, the Merger Agreement and the Merger in strict confidence and shall not divulge any such information to any third person until Public Company has publicly disclosed its entry into the Merger Agreement and this Agreement; provided, however, that the Stockholder may disclose such information to its Affiliates, partners, members, stockholders, parents, subsidiaries, attorneys, accountants, consultants, trustees, beneficiaries and other representatives (provided that such Persons are subject to confidentiality obligations at least as restrictive as those contained herein). Neither the Stockholder nor any of its Affiliates (other than Public Company, whose actions shall be governed by the Merger Agreement), shall issue or cause the publication of any press release or other public announcement with respect to this Agreement, the Merger, the Merger Agreement or the other transactions contemplated hereby or thereby without the prior written consent of the Merger Partner and Public Company, except (i) as may be required by applicable Law in which circumstance such announcing party shall make reasonable efforts to consult with Merger Partner and Public Company to the extent practicable or (ii) for any amendments to the Schedule 13D of the Stockholder required by virtue of this Agreement.
(m) Interpretation. When reference is made in this Agreement to a Section or Appendix, such reference shall be to a Section of or Appendix to this Agreement, unless otherwise indicated. The headings contained in this Agreement are for convenience of reference only and shall not affect in any way the meaning or interpretation of this Agreement. The language used in this Agreement shall be deemed to be the language chosen by the parties hereto to express their mutual intent, and no rule of strict construction shall be applied against any party. Whenever the context may require, any pronouns used in this Agreement shall include the corresponding masculine, feminine or neuter forms, and the singular form of nouns and pronouns shall include the plural, and vice versa. Any reference to any federal, state, local or foreign statute or law shall be deemed also to refer to all rules and regulations promulgated thereunder, unless the context requires otherwise. Whenever the words “include,” “includes” or “including” are used in this Agreement, they shall be deemed to be followed by the words “without limitation.”
(n) Compliance with Governmental Entities. Notwithstanding anything to the contrary in this Agreement, if at any time following the date hereof and prior to the Expiration Date a Governmental Entity enters an order restraining, enjoining or otherwise prohibiting the Stockholder from taking any action pursuant to Section 4 of this Agreement, then the obligations of the Stockholder set forth in Section 4 of this Agreement shall be of no force and effect for so long as such order is in effect solely to the extent such order restrains, enjoins or otherwise prohibits the Stockholder from taking any such action.
[Remainder of Page Left Intentionally Blank]
IN WITNESS WHEREOF, the undersigned have caused this Agreement to be duly executed as of the date first above written.
MERGER PARTNER:
PALVELLA THERAPEUTICS, INC.
By:
Name: Wesley H. Kaupinen
Title: Chief Executive Officer
PUBLIC COMPANY:
PIERIS PHARMACEUTICALS, INC.
By:
Name: Stephen S. Yoder
Title: Chief Executive Officer
[STOCKHOLDER],
in his/her/its capacity as the Stockholder:
Signature: _______________________________
Address:
Appendix A
Public Company Common Stock:
Public Company Preferred Stock:
ANNEX F
Final Form
FORM OF PALVELLA SUPPORT AGREEMENT
This Support Agreement (this “Agreement”) is made and entered into as of July 23, 2024, by and among Palvella Therapeutics, Inc. a Delaware corporation (“Merger Partner”), Pieris Pharmaceuticals, Inc., a Nevada corporation (“Public Company”), and the undersigned stockholder (the “Stockholder”) of Merger Partner.
RECITALS
WHEREAS, concurrently with or following the execution and delivery hereof, Public Company, Merger Partner and Polo Merger Sub, Inc., a Delaware corporation and a wholly owned subsidiary of Public Company (the “Merger Sub”), have entered into an agreement and plan of merger (as such agreement may be amended or supplemented from time to time pursuant to the terms thereof, the “Merger Agreement”), pursuant to which Merger Sub will merge with and into Merger Partner, with Merger Partner surviving the merger as the surviving corporation and a wholly owned subsidiary of Public Company (the “Merger”).
WHEREAS, as of the date hereof, the Stockholder is the beneficial owner (as defined in Rule 13d-1 under the Exchange Act) of such number of shares of Merger Partner Capital Stock as indicated in Appendix A.
WHEREAS, as an inducement to the willingness of Public Company to enter into the Merger Agreement, Public Company has required that the Stockholder enter into this Agreement.
NOW, THEREFORE, intending to be legally bound, the parties hereby agree as follows:
1. Certain Definitions. Capitalized terms used but not otherwise defined herein shall have the meanings ascribed thereto in the Merger Agreement. For all purposes of this Agreement, the following terms shall have the following respective meanings:
(a) “Constructive Sale” means, with respect to any security, a short sale with respect to such security, entering into or acquiring a derivative contract with respect to such security, entering into or acquiring a futures or forward contract to deliver such security or entering into any other hedging or other derivative transaction that has the effect of either directly or indirectly materially changing the economic benefits or risks of ownership of such security.
(b) “Shares” means (i) all shares of Merger Partner Capital Stock owned, beneficially or of record, by the Stockholder as of the date hereof, and (ii) all additional shares of Merger Partner Capital Stock acquired by the Stockholder, beneficially or of record, during the period commencing with the execution and delivery of this Agreement and expiring on the Closing Date.
(c) “Transfer” or “Transferred” means, with respect to any security, the direct or indirect assignment, sale, transfer, tender, exchange, pledge or hypothecation, or the grant, creation or suffrage of a lien, security interest or encumbrance in or upon, or the gift, grant or placement in trust, or the Constructive Sale or other disposition of such security (including transfers by testamentary or intestate succession, by domestic relations order or other court order, or otherwise by operation of law) or any right, title or interest therein (including any right or power to vote to which the holder thereof may be entitled, whether such right or power is granted by proxy or otherwise), or the record or beneficial ownership thereof, the offer to make such a sale, transfer, Constructive Sale or other disposition, and each agreement, arrangement or understanding, whether or not in writing, to effect any of the foregoing.
2. Transfer and Voting Restrictions. The Stockholder covenants to Merger Partner and Public Company as follows:
(a) Except as otherwise permitted by Section 2(c), during the period commencing with the execution and delivery of this Agreement and expiring on the Expiration Date (as defined below), the Stockholder shall not Transfer any of the Stockholder’s Shares, or publicly announce its intention to Transfer any of its Shares.
(b) Except as otherwise permitted by this Agreement or by order of a court of competent jurisdiction or a Governmental Entity, the Stockholder will not commit any act that would restrict the Stockholder’s legal power, authority and right to vote all of the Shares held by the Stockholder or otherwise prevent or disable the Stockholder from performing any of his, her or its obligations under this Agreement. Without limiting the generality of the foregoing, except for this Agreement and as contemplated by or related to the Palvella Therapeutics, Inc. Third Amended and Restated Voting Agreement, dated December 16, 2022 (the “Voting Agreement”, and together with Palvella Therapeutics, Inc. Third Amended and Restated Investors’ Rights Agreement, dated December 16, 2022, and Palvella Therapeutics, Inc. Third Amended and Restated Right of First Refusal and Co-Sale Agreement, dated December 16, 2022, the “Shareholder Agreements”) and as otherwise permitted by this Agreement, the Stockholder shall not enter into any voting agreement with any person or entity with respect to any of the Stockholder’s Shares, grant any person or entity any proxy (revocable or irrevocable) or power of attorney with respect to any of the Shares, deposit any Shares in a voting trust or otherwise enter into any agreement or arrangement with any person or entity limiting or affecting the Stockholder’s legal power, authority or right to vote the Stockholder’s Shares in favor of the Merger Partner Voting Proposal.
(c) Notwithstanding anything else herein to the contrary, the Stockholder may, at any time, Transfer the Stockholder’s Shares (i) by will or other testamentary document or by intestacy, (ii) to any Affiliate of Stockholder or any investment fund or other entity controlled or managed by the Stockholder or a controlling Affiliate of Stockholder, (iii) to any member of the Stockholder’s immediate family, (iv) in the case of a Stockholder who is not a natural person, by pro rata distributions from the Stockholder to its members, partners, or shareholders pursuant to the Stockholder’s organizational documents, (v) to any trust for the direct or indirect benefit of the Stockholder or the immediate family of the Stockholder or otherwise for tax or estate planning purposes, or (vi) by operation of law; provided, that (x) such Transferred Shares shall continue to be bound by this Agreement and (y) the applicable transferee shall have executed and delivered to Public Company and Merger Partner a support agreement substantially similar to this Agreement upon consummation of such Transfer.
3. No Obligation to Exercise. Notwithstanding anything to the contrary herein, nothing in this Agreement shall obligate the Stockholder to exercise any option or any other right to acquire any shares of Merger Partner Capital Stock.
4. Agreement to Vote Shares. The Stockholder covenants to Merger Partner and Public Company as follows:
(a) Until the Expiration Date, at any meeting of the stockholders of Merger Partner, however called, and at every adjournment or postponement thereof, and on every action or approval by written consent of the stockholders of Merger Partner, the Stockholder shall be present (in person or by proxy) and vote, or exercise its right to consent with respect to, all Shares held by the Stockholder (i) in favor of the Merger Partner Voting Proposal and (ii) against any Acquisition Proposal.
(b) If the Stockholder is the beneficial owner, but not the record holder, of Shares, the Stockholder shall cause the record holder and any nominees to be present (in person or by proxy) and vote all the Stockholder’s Shares in accordance with this Section 4.
(c) In the event of a stock split, stock dividend or distribution, or any change in the capital stock of Public Company by reason of any split-up, reverse stock split, recapitalization, combination, reclassification, reincorporation, exchange of shares or the like, the term “Shares” shall be deemed to refer to and include such shares as well as all such stock dividends and distributions and any securities into which or for which any or all of such shares may be changed or exchanged or which are received in such transaction.
(d) The Stockholder hereby waives and agrees not to exercise any rights of appraisal or any dissenters’ rights (including under Section 262 of the DGCL) that the Stockholder may have (whether under applicable law or otherwise) or could potentially have or acquire in connection with the Merger.
5. Action in Stockholder Capacity Only. The Stockholder is entering into this Agreement solely in the Stockholder’s capacity as a record holder and beneficial owner, as applicable, of its Shares and not in the Stockholder’s capacity as a director or officer of Merger Partner. Nothing herein shall limit or affect the Stockholder’s ability to act as an officer or director of Merger Partner.
6. Documentation and Information. The Stockholder shall permit and hereby authorizes Public Company and Merger Partner to publish and disclose in all documents and schedules filed with the SEC, and any press release or other disclosure document that Public Company or Merger Partner reasonably determines to be necessary in connection with the transactions contemplated by the Merger Agreement, such Stockholder’s identity and ownership of the Shares and the nature of such Stockholder’s commitments and obligations under this Agreement.
7. Irrevocable Proxy. The Stockholder hereby revokes (or agrees to cause to be revoked) any proxies that the Stockholder has heretofore granted with respect to its Shares. In the event and to the extent that the Stockholder fails to vote the Shares in accordance with Section 4 at any applicable meeting of the stockholders of Merger Partner or pursuant to any applicable written consent of the stockholders of Merger Partner, the Stockholder shall, solely with respect to the matters described in Section 4, be deemed to have irrevocably granted to, and appointed, Public Company, and any individual designated in writing by Public Company, and each of them individually, as his, her or its proxy and attorney-in-fact (with full power of substitution), for and in its name, place and stead, to vote his, her or its Shares in any action by written consent of Merger Partner stockholders or at any meeting of the Merger Partner stockholders called with respect to any of the matters specified in, and in accordance and consistent with, Section 4 of this Agreement. Public Company agrees not to exercise the proxy granted herein for any purpose other than the purposes described in this Agreement. Except as otherwise provided for herein, the Stockholder hereby affirms that the irrevocable proxy is coupled with an interest and may under no circumstances be revoked and that such irrevocable proxy is executed and intended to be irrevocable. Notwithstanding any other provisions of this Agreement, the irrevocable proxy granted hereunder shall automatically terminate upon the termination of this Agreement. For the avoidance of doubt, this Agreement does not, and is not an agreement to, revoke or otherwise terminate any proxy granted by the Stockholder pursuant to the Voting Agreement.
8. No Exercise of Appraisal Rights; Waivers. The Stockholder hereby irrevocably and unconditionally (a) waives, and agrees to cause to be waived and to prevent the exercise of, any rights of appraisal, any dissenters’ rights and any similar rights (including any notice requirements related thereto) relating to the Merger that Stockholder may have by virtue of, or with respect to, any Shares (including all rights under Section 262 of the DGCL) and (b) agrees that the Stockholder will not bring, commence, institute, maintain, prosecute or voluntarily aid or participate in any action, claim, suit or cause of action, in law or in equity, in any court or before any Governmental Entity, which (i) challenges the validity of or seeks to enjoin the operation of any provision of this Agreement or (ii) alleges that the execution and delivery of this Agreement by the Stockholder breaches any duty that such Stockholder has (or may be alleged to have) to the Company or to the other Company stockholders; provided, that (x) the Stockholder may defend against, contest or settle any such action, claim, suit or cause of action brought against the Stockholder that relates solely to the Stockholder’s capacity as a director, officer or securityholder of the Company and (y) the foregoing shall not limit or restrict in any manner the Stockholder from enforcing the Stockholder’s rights under this Agreement and the other agreements entered into by the Stockholder in connection herewith, or otherwise in connection with the Merger, including the Stockholder’s right to receive the Merger Consideration pursuant to the terms of the Merger Agreement.
9. Representations and Warranties.
(a) Representations and Warranties of the Stockholder. The Stockholder hereby represents and warrants to Merger Partner and Public Company as follows:
(i) The Stockholder (A) is the beneficial or record owner of the shares of Merger Partner Capital Stock indicated in Appendix A (each of which shall be deemed to be “held” by the Stockholder for purposes of Section 4 unless otherwise expressly stated with respect to any shares in Appendix A), free and clear of any and all Liens (other than any Liens that may exist pursuant to the Shareholder Agreements or applicable securities laws) and (B) does not beneficially own any securities of Merger Partner other than the shares of Merger Partner Capital Stock and rights to purchase shares of Merger Partner Capital Stock set forth in Appendix A.
(ii) Except as otherwise provided in this Agreement, the Stockholder has full power and authority to (A) make, enter into and carry out the terms of this Agreement and (B) vote all of its Shares in the manner set forth in this Agreement without the consent or approval of, or any other action on the part of, any other person or entity (including any Governmental Entity). Without limiting the generality of the foregoing, the Stockholder has not entered into any voting agreement (other than this Agreement and the Voting Agreement) with any person with respect to any of the Stockholder’s Shares, granted any person any proxy (revocable or irrevocable) or power of attorney with respect to any of the Stockholder’s Shares, pledged or disposed of any of the Stockholder’s Shares, deposited any of the Stockholder’s Shares in a voting trust or entered into any arrangement or agreement with any person limiting or affecting the Stockholder’s legal power, authority or right to vote the Stockholder’s Shares on any matter.
(iii) This Agreement has been duly and validly executed and delivered by the Stockholder and (assuming the due authorization, execution and delivery by the other parties hereto) constitutes a valid and binding agreement of the Stockholder enforceable against the Stockholder in accordance with its terms, subject to the Bankruptcy and Equity Exception. If (i) the Stockholder is a corporation, partnership or other investment or business entity, the Stockholder is duly organized, validly existing and in good standing under the Laws of the jurisdiction in which it is organized, and the execution, delivery and performance of this Agreement by the Stockholder and the consummation by the Stockholder of the transactions contemplated hereby are within the corporate power and capacity of the Stockholder and have been duly authorized by all necessary corporate action of the Stockholder and (ii) the Stockholder is an individual, the signature on this Agreement is genuine, and the Stockholder has full power and authority and legal competence and capacity to execute this Agreement, to perform fully the Stockholder’s obligations hereunder, and to consummate the transactions contemplated hereby. The execution and delivery of this Agreement by the Stockholder and the performance by the Stockholder of the agreements and obligations hereunder will not result in any breach or violation of or be in conflict with or constitute a default under any term of any Contract or if applicable any provision of an organizational document (including a certificate of incorporation) to or by which the Stockholder is a party or bound, or any applicable law to which the Stockholder is subject or bound, except for any such breach, violation, conflict or default which, individually or in the aggregate, would not reasonably be expected to materially impair or adversely affect the Stockholder’s ability to perform its obligations under this Agreement.
(iv) The Stockholder has had the opportunity to discuss the Merger Agreement, this Agreement and the transactions contemplated hereby and thereby with the Stockholder’s legal counsel. The Stockholder understands and acknowledges that Public Company is entering into the Merger Agreement in reliance upon the Stockholder’s execution, delivery and performance of this Agreement.
(v) The execution, delivery and performance of this Agreement by the Stockholder do not and will not require any consent, approval, authorization or permit of, action by, filing with or notification to, any Governmental Entity, except for any such consent, approval, authorization, permit, action, filing or notification the failure of which to make or obtain, individually or in the aggregate, has not and would not materially impair the Stockholder’s ability to perform its obligations under this Agreement.
(vi) As of the date hereof, there is no action, suit, investigation or proceeding (whether judicial, arbitral, administrative, or otherwise) pending against, or, to the knowledge of the Stockholder, threatened against, the Stockholder or any of the Stockholder’s properties or assets (including the Shares) that would reasonably be expected to prevent or materially delay or impair the ability of the Stockholder to perform its obligations hereunder or to consummate the transactions contemplated hereby.
(vii) Neither the Stockholder nor any of its Representatives or Affiliates (excluding, for the avoidance of doubt, Merger Partner) has employed or made any agreement with any broker, finder or similar agent or any Person which will result in the obligation of such Stockholder, Public Company, Merger Partner, or any of their respective Affiliates to pay any finder’s fee, brokerage fees or commission or similar payment in connection with the transactions contemplated hereby.
(b) Representations of the Merger Partner. The Merger Partner hereby represents and warrants to the Stockholder as follows: (i) it is a corporation duly organized, validly existing and in good standing under the Laws of the jurisdiction in which it is organized (in the case of good standing, to the extent the concept is recognized by such jurisdiction); (ii) it has all requisite corporate power and authority to enter into and deliver this Agreement and to perform its obligations hereunder; (iii) the execution and delivery by it of this Agreement and the consummation of the transactions contemplated hereby have been duly authorized by all necessary corporate action on the part of the Merger Partner; and (iv) this Agreement constitutes a legal, valid and binding obligation of the Merger Partner, enforceable against it in accordance with its terms (except insofar as such enforceability may be limited by the Bankruptcy and Equity Exception).
(c) Representations of the Public Company. The Public Company hereby represents and warrants to the Stockholder as follows: (i) it is a corporation duly organized, validly existing and in good standing under the Laws of the jurisdiction in which it is organized (in the case of good standing, to the extent the concept is recognized by such jurisdiction); (ii) it has all requisite corporate power and authority to enter into and deliver this Agreement and to perform its obligations hereunder; (iii) the execution and delivery by it of this Agreement and the consummation of the transactions contemplated hereby have been duly authorized by all necessary corporate action on the part of the Public Company; and (iv) this Agreement constitutes a legal, valid and binding obligation of the Public Company, enforceable against it in accordance with its terms (except insofar as such enforceability may be limited by the Bankruptcy and Equity Exception).
10. Termination. This Agreement shall terminate and shall cease to be of any further force or effect as of the earlier of (a) such date and time as the Merger Agreement shall have been terminated pursuant to the terms thereof, (b) the Effective Time, or (c) the date a Public Company Board Recommendation Change or a Merger Partner Board Recommendation Change is made (the “Expiration Date”); provided, however, that (x) Section 11 shall survive the termination of this Agreement, and (y) the termination of this Agreement shall not relieve any party hereto from any liability for any material and willful breach of this Agreement prior to the Expiration Date. A “material and willful breach” by a party of a provision of this Agreement means that the party knowingly undertook an action, or knowingly failed to undertake an action, with the understanding that the action, or failure to act, was a material breach by such party of the applicable provisions of this Agreement.
11. Miscellaneous Provisions.
(a) Amendments. No amendment of this Agreement shall be effective against any party unless it shall be in writing and signed by each of the parties hereto. In the event Merger Partner agrees to amend or waive the terms and conditions of any Support Agreement it has entered into with any other stockholder of the Public Company, the result of which would make the terms and conditions of such Support Agreement more favorable to such stockholder than the terms and conditions hereof are to the Stockholder, then the Merger Partner and Public Company will offer to amend or waive the terms and conditions of this Agreement so they are no less favorable to the Stockholder than the terms and conditions of such other Support Agreement are to such other stockholder.
(b) Entire Agreement. This Agreement constitutes the entire agreement between the parties to this Agreement and supersedes all other prior agreements, arrangements and understandings, both written and oral, among the parties with respect to the subject matter hereof.
(c) Governing Law. This Agreement and all matters, claims, counterclaims, or causes of action (whether in contract, tort, statute, or otherwise) arising out of or relating to this Agreement and the transactions contemplated hereby (including its interpretation, construction, performance and enforcement), or the actions of any party in the negotiation, administration, performance, or enforcement of this Agreement (collectively, the “Relevant Matters”) shall be governed by and construed in accordance with the internal laws of the State of Delaware without giving effect to any choice or conflict of Law provision or rule (whether of the State of Delaware or any other jurisdiction) that would cause the application of laws of any jurisdictions other than those of the State of Delaware. For the avoidance of doubt, all matters relating to the internal affairs of Public Company (including the fiduciary duties of its directors and officers) shall be governed by the internal Laws of the State of Nevada without giving effect to any choice or conflict of Law provision or rule (whether of the State of Nevada or any other jurisdiction) that would cause the application of Laws of any jurisdictions other than those of the State of Nevada.
(d) Jurisdiction. Each of the parties to this Agreement (i) consents to submit itself to the exclusive personal jurisdiction of the Court of Chancery of the State of Delaware, New Castle County, or, if that court does not have jurisdiction, a state or federal court sitting in Wilmington, Delaware in any action or proceeding arising out of or relating to any Relevant Matter, (ii) agrees that all claims in respect of such action or proceeding shall be heard and determined in any such court, (iii) agrees that it shall not attempt to deny or defeat such personal jurisdiction by motion or other request for leave from any such court and (iv) agrees not to bring any action or proceeding arising out of or relating to any Relevant Matter in any other court. Each of the parties hereto waives any defense of inconvenient forum to the maintenance of any action or proceeding so brought and waives any bond, surety or other security that might be required of any other party with respect thereto. Any party may make service on another party by sending or delivering a copy of the process to the party to be served at the address and in the manner provided for the giving of notices in Section 11(j). Nothing in this Section 11(d), however, shall affect the right of any party to serve legal process in any other manner permitted by law.
(e) WAIVER OF JURY TRIAL. EACH OF THE PARTIES TO THIS AGREEMENT HEREBY IRREVOCABLY WAIVES ALL RIGHT TO TRIAL BY JURY IN ANY ACTION OR PROCEEDING ARISING OUT OF OR RELATING TO ANY RELEVANT MATTER.
(f) Assignment. Except as otherwise provided in Section 2(c) hereof, no party may assign any of its rights or delegate any of its performance obligations under this Agreement, in whole or in part, by operation of law or otherwise, without the prior written consent of the other parties hereto, and any such assignment without such prior written consent shall be null and void. Subject to the preceding sentence, this Agreement shall be binding upon, inure to the benefit of, and be enforceable by, the parties hereto and their respective successors and permitted assigns. Any purported assignment of rights or delegation of performance obligations in violation of this Section 11(f) is void.
(g) No Third Party Rights. This Agreement is not intended to, and shall not, confer upon any other person any rights or remedies hereunder other than the parties hereto to the extent expressly set forth herein.
(h) Severability. Any term or provision of this Agreement that is invalid or unenforceable in any situation in any jurisdiction shall not affect the validity or enforceability of the remaining terms and provisions hereof or the validity or enforceability of the offending term or provision in any other situation or in any other jurisdiction. If the final judgment of a court of competent jurisdiction declares that any term or provision hereof is invalid or unenforceable, the parties hereto agree that the court making such determination shall have the power to limit the term or provision, to delete specific words or phrases, or to replace any invalid or unenforceable term or provision with a term or provision that is valid and enforceable and that comes closest to expressing the intention of the invalid or unenforceable term or provision, and this Agreement shall be enforceable as so modified. In the event such court does not exercise the power granted to it in the prior sentence, the parties hereto agree to replace such invalid or unenforceable term or provision with a valid and enforceable term or provision that will achieve, to the extent possible, the economic, business and other purposes of such invalid or unenforceable term.
(i) Specific Performance. The parties hereto agree that irreparable damage would occur in the event that any of the provisions of this Agreement were not performed in accordance with their specific terms or were otherwise breached. It is accordingly agreed that the parties shall be entitled to an injunction or injunctions to prevent breaches of this Agreement and to enforce specifically the terms and provisions of this Agreement, this being in addition to any other remedy to which they are entitled at law or in equity.
(j) Notices. All notices and other communications hereunder shall be in writing and shall be deemed duly delivered (i) three Business Days after being sent by registered or certified mail, return receipt requested, postage prepaid, or (ii) one Business Day after being sent for next Business Day delivery, fees prepaid, via a reputable overnight courier service, in each case to the intended recipient as follows: (A) if to Merger Partner or Public Company, to the address, electronic mail address or facsimile provided in the Merger Agreement, including to the persons designated therein to receive copies; and/or (B) if to the Stockholder, to the Stockholder’s address, electronic mail address or facsimile shown below Stockholder’s signature to this Agreement.
(k) Counterparts. This Agreement may be executed in two or more counterparts (including by facsimile, by an electronic scan delivered by electronic mail or any electronic signature), each of which shall be deemed an original but all of which together shall be considered one and the same agreement and shall become effective when counterparts have been signed by each of the parties hereto and delivered to the other parties, it being understood that all parties need not sign the same counterpart. This Agreement may be executed and delivered by facsimile, by an electronic scan delivered by electronic mail or by delivery of any electronic signature.
(l) Confidentiality. Except to the extent required by applicable Law or regulation, the Stockholder shall hold any non-public information regarding this Agreement, the Merger Agreement and the Merger in strict confidence and shall not divulge any such information to any third person until Merger Partner has publicly disclosed its entry into the Merger Agreement and this Agreement; provided, however, that the Stockholder may disclose such information to its Affiliates, partners, members, stockholders, parents, subsidiaries, attorneys, accountants, consultants, trustees, beneficiaries and other representatives (provided that such Persons are subject to confidentiality obligations at least as restrictive as those contained herein). Neither the Stockholder nor any of its Affiliates (other than Merger Partner, whose actions shall be governed by the Merger Agreement), shall issue or cause the publication of any press release or other public announcement with respect to this Agreement, the Merger, the Merger Agreement or the other transactions contemplated hereby or thereby without the prior written consent of the Merger Partner and Public Company, except (i) as may be required by applicable Law in which circumstance such announcing party shall make reasonable efforts to consult with Merger Partner and Public Company to the extent practicable or (ii) for any amendments to the Schedule 13D of the Stockholder required by virtue of this Agreement.
(m) Interpretation. When reference is made in this Agreement to a Section or Appendix, such reference shall be to a Section of or Appendix to this Agreement, unless otherwise indicated. The headings contained in this Agreement are for convenience of reference only and shall not affect in any way the meaning or interpretation of this Agreement. The language used in this Agreement shall be deemed to be the language chosen by the parties hereto to express their mutual intent, and no rule of strict construction shall be applied against any party. Whenever the context may require, any pronouns used in this Agreement shall include the corresponding masculine, feminine or neuter forms, and the singular form of nouns and pronouns shall include the plural, and vice versa. Any reference to any federal, state, local or foreign statute or law shall be deemed also to refer to all rules and regulations promulgated thereunder, unless the context requires otherwise. Whenever the words “include,” “includes” or “including” are used in this Agreement, they shall be deemed to be followed by the words “without limitation.”
(n) Compliance with Governmental Entities. Notwithstanding anything to the contrary in this Agreement, if at any time following the date hereof and prior to the Expiration Date a Governmental Entity enters an order restraining, enjoining or otherwise prohibiting the Stockholder from taking any action pursuant to Section 4 of this Agreement, then the obligations of the Stockholder set forth in Section 4 of this Agreement shall be of no force and effect for so long as such order is in effect solely to the extent such order restrains, enjoins or otherwise prohibits the Stockholder from taking any such action.
[Remainder of Page Left Intentionally Blank]
IN WITNESS WHEREOF, the undersigned have caused this Agreement to be duly executed as of the date first above written.
MERGER PARTNER:
PALVELLA THERAPEUTICS, INC.
By:
Name: Wesley H. Kaupinen
Title: Chief Executive Officer
PUBLIC COMPANY:
PIERIS PHARMACEUTICALS, INC.
By:
Name: Stephen S. Yoder
Title: Chief Executive Officer
[STOCKHOLDER],
in his/her/its capacity as the Stockholder:
Signature: _______________________________
Address:
_______________________
_______________________
_______________________
Appendix A
Merger Partner Capital Stock
|
● |
Non-Voting Common Stock: |
|
● |
Voting Common Stock: |
|
● |
Series A-1 Preferred Stock: |
|
● |
Series A-2 Preferred Stock: |
|
● |
Series B Preferred Stock: |
|
● |
Series C Preferred Stock: |
|
● |
Series D Preferred Stock: |
ANNEX G
July 23, 2024
Pieris Pharmaceuticals, Inc.
225 Franklin Street, 26th Floor
Boston, MA 02110
Palvella Therapeutics, Inc.
125 Strafford Ave.
Wayne, PA 19087
Ladies and Gentlemen:
The undersigned signatory of this lock-up agreement (this “Lock-Up Agreement”) understands that Pieris Pharmaceuticals, Inc., a Nevada corporation (“Public Company”), has entered into an Agreement and Plan of Merger, dated as of July 23, 2024 (as the same may be amended from time to time, the “Merger Agreement”) with Polo Merger Sub, Inc., a Delaware corporation and a wholly-owned subsidiary of Public Company, and Palvella Therapeutics, Inc. a Delaware corporation, a Delaware corporation (“Merger Partner”). Capitalized terms used but not otherwise defined herein shall have the respective meanings ascribed to such terms in the Merger Agreement.
As a condition and inducement to Public Company and Merger Partner to enter into the Merger Agreement and to consummate the transactions contemplated thereby, and for other good and valuable consideration, the receipt and sufficiency of which are hereby acknowledged, the undersigned hereby irrevocably agrees that, subject to the exceptions set forth herein, without the prior written consent of Public Company and Merger Partner, the undersigned will not, during the period commencing upon the Closing and ending on the date that is 180 days after the Closing Date (the “Restricted Period”):
(1) offer, pledge, sell, contract to sell, sell any option, warrant or contract to purchase, purchase any option, warrant or contract to sell, grant any option, right or warrant to purchase, grant any proxies or powers of attorney, or otherwise transfer or dispose of (or enter into any transaction that is designed to, or could reasonably be expected to, result in the disposition (whether by actual disposition or effective economic disposition due to cash settlement or otherwise) by the undersigned or any affiliate of the undersigned or any Person in privity with the undersigned or any affiliate of the undersigned), or establish or increase a put equivalent position or liquidate or decrease a call equivalent position within the meaning of Section 16 of the Exchange Act, directly or indirectly, any shares of Public Company Common Stock or any securities convertible into or exercisable or exchangeable for Public Company Common Stock (including without limitation, Public Company Common Stock or such other securities which may be deemed to be beneficially owned by the undersigned in accordance with the rules and regulations of the SEC and securities of Public Company which may be issued upon exercise or vesting, as applicable, of an option, warrant, restricted stock award or restricted stock unit, in each case to purchase, receive in the future or otherwise acquire Public Company Common Stock (collectively, “Public Company Equity Rights”)) that are currently or hereafter owned by the undersigned (collectively, the “Undersigned’s Shares”);
(2) enter into any swap, short sale, hedge or other agreement that transfers, in whole or in part, any of the economic consequences of ownership of the Undersigned’s Shares regardless of whether any such transaction described in clause (1) above or this clause (2) is to be settled by delivery of Public Company Common Stock or other securities, in cash or otherwise;
(3) make any demand for, or exercise any right with respect to, the registration of any shares of Public Company Common Stock or Public Company Equity Rights (other than (i) such rights set forth in the Merger Agreement and (ii) the exercise of piggyback registration rights in connection with any secondary underwritten public offering of the Public Company Common Stock); or
(4) publicly disclose the intention to do any of the foregoing.
The restrictions and obligations contemplated by this Lock-Up Agreement shall not apply to:
(a) transfers of the Undersigned’s Shares:
(1) if the undersigned is a natural person, (A) to any person related to the undersigned by blood or adoption who is an immediate family member of the undersigned, or by marriage or domestic partnership (a “Family Member”), or to a trust formed for the benefit of the undersigned or any of the undersigned’s Family Members, (B) to the undersigned’s estate, following the death of the undersigned, by will, intestacy or other operation of Law, (C) as a bona fide gift or a charitable contribution, (D) by operation of Law pursuant to a qualified domestic order or in connection with a divorce settlement or (E) to any partnership, corporation or limited liability company which is controlled by the undersigned and/or by any such Family Member(s);
(2) if the undersigned is a corporation, partnership, limited liability company or other entity, (A) to another corporation, partnership, or other entity that is a direct or indirect affiliate (as defined under Rule 12b-2 of the Exchange Act) of the undersigned, including investment funds or other entities that control or manage, are under common control or management with, or are controlled or managed by the undersigned (including, for the avoidance of doubt, a fund managed by the same manager or managing member or general partner or management company or by an entity controlling, controlled by or under common control with such manager or managing member or general partner or management company as the undersigned), (B) as a distribution or dividend to equity holders, current or former general or limited partners, members or managers (or to the estates of any of the foregoing), as applicable, of the undersigned (including upon the liquidation and dissolution of the undersigned pursuant to a plan of liquidation approved by the undersigned’s equity holders), (C) as a bona fide gift or a charitable contribution or (D) transfers or dispositions not involving a change in beneficial ownership; or
(3) if the undersigned is a trust, to any grantors or beneficiaries of the trust;
provided that, in the case of any transfer or distribution pursuant to this clause (a), such transfer is not for value and each donee, heir, beneficiary or other transferee or distributee shall sign and deliver to Public Company a lock-up agreement in the form of this Lock-Up Agreement with respect to the shares of Public Company Common Stock or Public Company Equity Rights;
(b) the exercise or settlement of any Public Company Equity Rights (including a net or cashless exercise), and any related transfer of shares of Public Company Common Stock to Public Company for the purpose of paying the exercise price of such Public Company Equity Rights or for paying taxes (including estimated taxes or tax withholding obligations) due as a result of such exercise; provided that, for the avoidance of doubt, the underlying shares of Public Company Common Stock shall continue to be subject to the restrictions on transfer set forth in this Lock-Up Agreement;
(c) the establishment of a trading plan pursuant to Rule 10b5-1 under the Exchange Act for the transfer of Public Company Common Stock; provided that such plan does not provide for any transfers of Public Company Common Stock during the Restricted Period;
(d) transfers by the undersigned of shares of Public Company Common Stock purchased by the undersigned on the open market or in a public offering by Public Company, in each case following the date of the Merger Agreement, provided that no filing by any party (including any donor, donee, transferor, transferee, distributor or distributee) under the Exchange Act or other public announcement shall be required or shall be made voluntarily in connection with such transfer or disposition (whether on Form 4, Form 5 or otherwise) during the Restricted Period;
(e) pursuant to a bona-fide third party tender offer, merger, consolidation or other similar transaction made to all holders of Public Company’s capital stock involving a change of control of Public Company, provided that in the event that such tender offer, merger, consolidation or other such transaction is not completed, the Undersigned’s Shares shall remain subject to the restrictions contained in this Lock-Up Agreement;
(f) pursuant to an order of a Governmental Entity, provided that no filing by any party under the Exchange Act shall be voluntarily made or other public announcement shall be required or shall be made voluntarily in connection with such transfer or disposition during the Restricted Period and in respect of a required filing, the undersigned shall include a statement in such required filing to the effect that the disposition relates to the circumstances described in this paragraph (f);
or
(h) consented to by the Merger Partner and Public Company.
and provided, further, that, with respect to each of (a), (b), and (c), above, no filing by any party (including any donor, donee, transferor, transferee, distributor or distributee) under the Exchange Act or other public announcement shall be required or shall be made voluntarily in connection with such transfer or disposition during the Restricted Period (other than (x) any exit filings or public announcements that may be required under applicable federal and state securities Laws, (y) in respect of a required filing under the Exchange Act in connection with the exercise or the net settlement of any Public Company Equity Right, settled in Public Company Common Stock, that would otherwise expire during the Restricted Period, provided that reasonable notice shall be provided to Public Company prior to any such filing or (z) in respect of a required filing under the Exchange Act regarding the establishment or existence of a 10b5-1 plan, provided that such required filing shall include a statement that sales under such 10b5-1 plan will not occur until after the expiration of the Restricted Period) and provided further that the undersigned shall include a statement in such required filing to the effect that the disposition relates to the exercise or vesting, as applicable, of an option, warrant, restricted stock award or restricted stock unit, and that the shares of Common Stock received upon exercise or vesting are subject to the restrictions of this Agreement.
Any attempted transfer in violation of this Lock-Up Agreement will be of no effect and null and void, regardless of whether the purported transferee has any actual or constructive knowledge of the transfer restrictions set forth in this Lock-Up Agreement, and will not be recorded on the share register of Public Company. In furtherance of the foregoing, the undersigned agrees that Public Company and any duly appointed transfer agent for the registration or transfer of the securities described herein are hereby authorized to decline to make any transfer of securities if such transfer would constitute a violation or breach of this Lock-Up Agreement. Public Company may cause the legend set forth below, or a legend substantially equivalent thereto, to be placed upon any certificate(s) or other documents, ledgers or instruments evidencing the undersigned’s ownership of Public Company Common Stock:
THE SHARES REPRESENTED BY THIS CERTIFICATE ARE
SUBJECT TO AND MAY ONLY BE TRANSFERRED IN
COMPLIANCE WITH A LOCK-UP AGREEMENT, A COPY
OF WHICH IS ON FILE AT THE PRINCIPAL OFFICE OF THE COMPANY.
The undersigned hereby represents and warrants that the undersigned has full power, capacity and authority to enter into this Lock-Up Agreement. All authority herein conferred or agreed to be conferred and any obligations of the undersigned shall be binding upon the successors, assigns, heirs or personal representatives of the undersigned.
The undersigned understands that if the Merger Agreement is terminated for any reason, this Lock-Up Agreement will automatically terminate, and the undersigned shall be released from all of his, her or its obligations under this Lock-Up Agreement. The undersigned understands that Public Company is proceeding with the transactions contemplated by the Merger Agreement in reliance upon this Lock-Up Agreement. This Lock-Up Agreement will automatically terminate, and the undersigned will be released from all of his, her or its obligations hereunder, upon March 31, 2025 in the event that transactions contemplated by the Merger Agreement have not been consummated by such date.
Any and all remedies herein expressly conferred upon Public Company will be deemed cumulative with and not exclusive of any other remedy conferred hereby, or by Law or equity, and the exercise by Public Company of any one remedy will not preclude the exercise of any other remedy. The undersigned agrees that irreparable damage would occur to Public Company in the event that any provision of this Lock-Up Agreement was not performed in accordance with its specific terms or were otherwise breached. It is accordingly agreed that Public Company shall be entitled to an injunction or injunctions to prevent breaches of this Lock-Up Agreement and to enforce specifically the terms and provisions hereof in any court of the United States or any state having jurisdiction, this being in addition to any other remedy to which Public Company is entitled at Law or in equity, and the undersigned waives any bond, surety or other security that might be required of Public Company with respect thereto.
In the event that any holder of Public Company’s securities that are subject to a substantially similar agreement entered into by such holder, other than the undersigned, is permitted by Public Company to sell or otherwise transfer or dispose of shares of Public Company Common Stock for value other than as permitted by this or a substantially similar agreement entered into by such holder, the same percentage of shares of Public Company Common Stock held by the undersigned on the date of such release or waiver as the percentage of the total number of outstanding shares of Public Company Common Stock held by such holder on the date of such release or waiver that are subject of such release or waiver shall be immediately and fully released on the same terms from any remaining restrictions set forth herein (the “Pro-Rata Release”); provided, however, that such Pro-Rata Release shall not be applied unless and until permission has been granted by Public Company to an equity holder or equity holders to sell or otherwise transfer or dispose of all or a portion of such equity holders shares of Public Company Common Stock in an aggregate amount in excess of 1% of the total number of shares of Public Company Common Stock originally subject to a substantially similar agreement. The Public Company shall notify the undersigned within two (2) business days prior to the effective date of a release of any holder of Public Company Common Stock of such holder’s obligations under a lock-up or substantially similar agreement that gives rise to a Pro-Rata Release.
Upon the release of any of the Undersigned’s Shares from this Lock-Up Agreement, Public Company will cooperate with the undersigned to facilitate the timely preparation and delivery of certificates representing the Undersigned Shares without the restrictive legend above or the withdrawal of any stop transfer instructions by virtue of this Lock-Up Agreement.
This Lock-Up Agreement and all matters, claims, counterclaims, or causes of action (whether in contract, tort, statute, or otherwise) arising out of or relating to this Lock-Up Agreement and the transactions contemplated hereby (including its interpretation, construction, performance and enforcement), or the actions of any party in the negotiation, administration, performance, or enforcement of this Lock-Up Agreement (collectively, “Relevant Matters”) shall be governed by and construed in accordance with the Laws of the state of Delaware, without regard to the conflict of Laws principles (whether of the State of Delaware or any other jurisdiction) that would cause the application of Laws of any jurisdictions other than those of the State of Delaware. For the avoidance of doubt, all matters relating to the internal affairs of Public Company (including the fiduciary duties of its directors and officers) shall be governed by the internal Laws of the State of Nevada without giving effect to any choice or conflict of Law provision or rule (whether of the State of Nevada or any other jurisdiction) that would cause the application of Laws of any jurisdictions other than those of the State of Nevada.
EACH OF THE PARTIES TO THIS LOCK-UP AGREEMENT HEREBY IRREVOCABLY WAIVES ALL RIGHT TO TRIAL BY JURY IN ANY ACTION OR PROCEEDING ARISING OUT OF OR RELATING TO ANY RELEVANT MATTER.
Each of the parties to this Lock-Up Agreement (a) consents to submit itself to the exclusive personal jurisdiction of the Court of Chancery of the State of Delaware, New Castle County, or, if that court does not have jurisdiction, a state or federal court sitting in Wilmington, Delaware in any action or proceeding arising out of or relating any Relevant Matter, (b) agrees that all claims in respect of such action or proceeding shall be heard and determined in any such court, (c) agrees that it shall not attempt to deny or defeat such personal jurisdiction by motion or other request for leave from any such court and (d) agrees not to bring any action or proceeding arising out of or relating to any Relevant Matter in any other court. Each of the parties hereto waives any defense of inconvenient forum to the maintenance of any action or proceeding so brought and waives any bond, surety or other security that might be required of any other party with respect thereto. Any party may make service on another party by sending or delivering a copy of the process to the party to be served at the address and in the manner provided for the giving of notices in the paragraph immediately below. Nothing in this paragraph, however, shall affect the right of any party to serve legal process in any other manner permitted by Law.
All notices and other communications hereunder shall be in writing and shall be deemed duly delivered (i) (i) three Business Days after being sent by registered or certified mail, return receipt requested, postage prepaid, or (ii) one Business Day after being sent for next Business Day delivery, fees prepaid, via a reputable overnight courier service, in each case to the intended recipient as follows: (A) if to Public Company, to the address, electronic mail address or facsimile provided in the Merger Agreement, including to the persons designated therein to receive copies; and/or (B) if to the Stockholder, to the Stockholder’s address, electronic mail address or facsimile shown below Stockholder’s signature to this Lock-Up Agreement.
Any term or provision of this Lock-Up Agreement that is invalid or unenforceable in any situation in any jurisdiction shall not affect the validity or enforceability of the remaining terms and provisions hereof or the validity or enforceability of the offending term or provision in any other situation or in any other jurisdiction. If the final judgment of a court of competent jurisdiction declares that any term or provision hereof is invalid or unenforceable, the parties hereto agree that the court making such determination shall have the power to limit the term or provision, to delete specific words or phrases, or to replace any invalid or unenforceable term or provision with a term or provision that is valid and enforceable and that comes closest to expressing the intention of the invalid or unenforceable term or provision, and this Lock-Up Agreement shall be enforceable as so modified. In the event such court does not exercise the power granted to it in the prior sentence, the parties hereto agree to replace such invalid or unenforceable term or provision with a valid and enforceable term or provision that will achieve, to the extent possible, the economic, business and other purposes of such invalid or unenforceable term.
This Lock-Up Agreement may be executed in several counterparts, each of which shall be deemed an original and all of which shall constitute one and the same instrument. The exchange of a fully executed Lock-Up Agreement (in counterparts or otherwise) by Public Company and the undersigned by facsimile or electronic transmission in .pdf format shall be sufficient to bind such parties to the terms and conditions of this Lock-Up Agreement.
[Remainder of Page Intentionally Left Blank; Signature Page Follows]
|
|
Very truly yours,
Print Name of Stockholder: |
|||
| Signature (for individuals): | ||||
| Signature (for entities): | ||||
|
|
|
|
|
|
|
|
By: |
|
|
|
|
|
Name: |
|
|
|
|
|
Title: |
|
|
|
Accepted and Agreed
by Pieris Pharmaceuticals, Inc.
By:
Name: Stephen S. Yoder
Title: Chief Executive Officer
[Signature Page to Lock-Up Agreement]
ANNEX H
2024 EQUITY INCENTIVE PLAN
Section 1. Purpose; Definitions. The purposes of the Palvella Therapeutics, Inc. 2024 Equity Incentive Plan (as amended from time to time, the “Plan”) are to: (a) enable Palvella Therapeutics, Inc. (the “Company”) and its affiliated companies to recruit and retain highly qualified employees, directors and consultants; (b) provide those employees, directors and consultants with an incentive for productivity; and (c) provide those employees, directors and consultants with an opportunity to share in the growth and value of the Company. Upon the Effective Date, no further awards shall be made under the Prior Plan (as defined below).
For purposes of the Plan, the following terms will have the meanings defined below, unless the context clearly requires a different meaning:
(a) “Affiliate” means, with respect to a Person, a Person that directly or indirectly controls, is controlled by, or is under common control with such Person.
(b) “Applicable Law” means the legal requirements relating to the administration of and issuance of securities under stock incentive plans, including, without limitation, the requirements of state corporations law, federal, state and foreign securities law, federal, state and foreign tax law, and the requirements of any stock exchange or quotation system upon which the Shares may then be listed or quoted.
(c) “Award” means an award of Options, Stock Appreciation Rights, Restricted Stock, Restricted Stock Units or Cash or Other Stock Based Awards made under this Plan.
(d) “Award Agreement” means, with respect to any particular Award, the written document that sets forth the terms of that particular Award.
(e) “Board” means the Board of Directors of the Company, as constituted from time to time.
(f) “Cash or Other Stock Based Award” means an award that is granted under Section 10.
(g) “Cause” has the meaning ascribed to such term in any written agreement between the Participant and the Company defining such term and, in the absence of such agreement, such term means, with respect to a Participant, the occurrence of any of the following events: (i) a Participant’s commission of any felony or any crime involving fraud, dishonesty or moral turpitude under the laws of the United States or any state thereof; (ii) a Participant’s attempted commission of, or participation in, a fraud or act of dishonesty against the Company; (iii) a Participant’s intentional, material violation of any contract or agreement between the Participant and the Company or of any statutory duty owed to the Company; (iv) a Participant’s unauthorized use or disclosure of the Company’s confidential information, intellectual property or trade secrets; (v) a Participant’s refusal to comply with any lawful directive or policy of the Company which refusal is not cured by the Participant within ten (10) days of such written notice from the Company; or (vi) such Participant’s gross misconduct. The determination that a termination of the Participant’s service is either for Cause or without Cause will be made by the Board or Committee, as applicable, in its sole and exclusive judgment and discretion. Any determination by the Company that the service of a Participant was terminated with or without Cause for the purposes of outstanding Awards held by such Participant will have no effect upon any determination of the rights or obligations of the Company or such Participant for any other purpose.
(h) “Change in Control” means the occurrence of any of the following events: (i) any “person” (as such term is used in Sections 13(d) and 14(d) of the Exchange Act) is or becomes a “beneficial owner” (as defined in Rule 13d-3 under the Exchange Act), directly or indirectly, of securities of the Company representing 50% or more of the total power to vote for the election of directors of the Company; (ii) during any twelve month period, individuals who at the beginning of such period constitute the Board and any new director (other than a director designated by a person who has entered into an agreement with the Company to effect a transaction described in Section 1(h)(i), Section 1(h)(iii), Section 1(h)(iv) or Section 1(h)(v) hereof) whose election by the Board or nomination for election by the Company’s stockholders was approved by a vote of at least a majority of the directors then still in office who either were directors at the beginning of the period of whose election or nomination for election was previously approved, cease for any reason to constitute a majority thereof; (iii) the merger or consolidation of the Company with another corporation other than a merger or consolidation which would result in the voting securities of the Company outstanding immediately prior to the merger or consolidation, continuing to represent (either by remaining outstanding or by being converted into voting securities of the surviving entity or the parent of such entity) more than 50% of the total voting power represented by the voting securities of the Company or such surviving entity or parent of such corporation, as the case may be, outstanding immediately after such merger or consolidation; (iv) the sale or other disposition of all or substantially all of the assets of the Company; (v) a liquidation or dissolution of the Company; or (vi) such other event deemed to constitute a “Change in Control” by the Board.
Notwithstanding anything in the Plan or an Award Agreement to the contrary, to the extent necessary to comply with Section 409A of the Code, no event that, but for the application of this paragraph, would be a Change in Control as defined in the Plan or the Award Agreement, as applicable, shall be a Change in Control unless such event is also a “change in control event” as defined in Section 409A of the Code.
(i) “Code” means the Internal Revenue Code of 1986, as amended from time to time, and any successor thereto.
(j) “Committee” means the committee designated by the Board to administer the Plan under Section 2. To the extent required under Applicable Law, the Committee shall have at least two members and each member of the Committee shall be a Non-Employee Director. Unless otherwise determined by the Board, the Compensation Committee of the Board will serve as the Committee.
(k) “Director” means a member of the Board.
(l) “Disability” means a condition rendering a Participant Disabled.
(m) “Disabled” will have the same meaning as set forth in Section 22(e)(3) of the Code. Notwithstanding anything in the Plan or an Award Agreement to the contrary, to the extent necessary to comply with Section 409A of the Code, a Participant will not be deemed “Disabled” unless the Participant is considered Disabled within the meaning of Section 409A of the Code.
(n) “Effective Date” has the meaning defined below in Section 18.
(o) “Exchange Act” means the Securities Exchange Act of 1934, as amended.
(p) “Fair Market Value” means, as of any date, the value of a Share determined as follows: (i) if the Shares are listed on any established stock exchange or a national market system, including, without limitation, the Nasdaq Global Market, the Fair Market Value of a Share will be the closing sales price for such stock as quoted on that system or exchange (or the system or exchange with the greatest volume of trading in Shares) at the close of regular hours trading on the day of determination; (ii) if the Shares are regularly quoted by recognized securities dealers but selling prices are not reported, the Fair Market Value of a Share will be the mean between the high bid and low asked prices for Shares at the close of regular hours trading on the day of determination; or (iii) if Shares are not traded as set forth above, the Fair Market Value will be determined in good faith by the Committee taking into consideration such factors as the Committee considers appropriate, such determination by the Committee to be final, conclusive and binding. Notwithstanding the foregoing, in connection with a Change in Control, Fair Market Value shall be determined in good faith by the Committee, such determination by the Committee to be final conclusive and binding.
(q) “Incentive Stock Option” means any Option intended to be an “Incentive Stock Option” within the meaning of Section 422 of the Code.
(r) “Non-Employee Director” has the meaning set forth in Rule 16b-3(b)(3)(i) promulgated by the Securities and Exchange Commission under the Exchange Act, or any successor definition adopted by the Securities and Exchange Commission.
(s) “Non-Qualified Stock Option” means any Option that is not an Incentive Stock Option.
(t) “Option” means any option to purchase Shares (including an option to purchase Restricted Stock, if the Committee so determines) granted pursuant to Section 5 hereof.
(u) “Parent” means, in respect of the Company, a “parent corporation” as defined in Section 424(e) of the Code.
(v) “Participant” means an employee, consultant, Director, or other service provider of or to the Company or any of its Affiliates to whom an Award is granted.
(w) “Person” means an individual, partnership, corporation, limited liability company, trust, joint venture, unincorporated association, or other entity or association.
(x) “Prior Plan” means each of the Pieris Pharmaceuticals, Inc. (i) 2014 Employee, Director and Consultant Equity Incentive Plan, the 2016 Employee, Director and Consultant Equity Incentive Plan, the 2018 Employee, Director and Consultant Equity Incentive Plan, the 2019 Employee, Director and Consultant Equity Incentive Plan, and the 2020 Employee, Director and Consultant Equity Incentive Plan, as amended.
(y) “Prior Plan Awards” mean awards granted under the Prior Plan.
(z) “Restricted Stock” means Shares that are subject to restrictions pursuant to Section 8 hereof.
(aa) “Restricted Stock Unit” means a right granted under and subject to restrictions pursuant to Section 9 hereof.
(bb) “Shares” means shares of the Company’s common stock, par value $.001, subject to substitution or adjustment as provided in Section 3(e) hereof.
(cc) “Stock Appreciation Right” means a right granted under and subject to Section 6 hereof.
(dd) “Subsidiary” means, in respect of the Company, a subsidiary company as defined in Sections 424(f) and (g) of the Code.
Section 2. Administration. The Plan shall be administered by the Committee; provided that, notwithstanding anything to the contrary herein, in its sole discretion, the Board may at any time and from time-to-time exercise any and all rights and duties of the Committee under the Plan except with respect to matters which under Applicable Law are required to be determined in the sole discretion of the Committee. Any action of the Committee in administering the Plan shall be final, conclusive and binding on all persons, including the Company, its Subsidiaries, Affiliates, their respective employees, the Participants, persons claiming rights from or through Participants and stockholders of the Company.
The Committee will have full authority to grant Awards under this Plan and determine the terms of such Awards. Such authority will include the right to:
(a) select the individuals to whom Awards are granted (consistent with the eligibility conditions set forth in Section 4);
(b) determine the type of Award to be granted;
(c) determine the number of Shares, if any, to be covered by each Award;
(d) establish the other terms and conditions of each Award;
(e) approve forms of agreements (including Award Agreements) for use under the Plan; and
(f) modify or amend each Award, subject to the Participant’s consent if such modification or amendment would materially impair such Participant’s rights.
The Committee will have the authority to adopt, alter and repeal such administrative rules, guidelines and practices governing the Plan as it, from time to time, deems advisable; to interpret the terms and provisions of the Plan and any Award issued under the Plan (and any Award Agreement); and to otherwise take any action that may be necessary or desirable to facilitate the administration of the Plan. The Committee may correct any defect, supply any omission or reconcile any inconsistency in the Plan or in any Award Agreement in the manner and to the extent it deems necessary to carry out the intent of the Plan.
To the extent permitted by Applicable Law and the Company’s governing documents, the Committee may delegate its authority as identified herein to any individual or committee of individuals (who may, but need not be, one or more Directors or Non-Employee Directors), including without limitation the authority to grant Awards hereunder. To the extent that the Committee delegates its authority to make Awards hereunder, applicable references in the Plan to the Committee’s authority to make Awards and determinations with respect thereto shall be deemed to include the Committee’s delegate, but the authority to administer the Plan will otherwise remain with the Committee. The Committee may revoke any such delegation at any time for any reason with or without prior notice.
No Director will be liable for any good faith determination, act or omission in connection with the Plan or any Award.
Section 3. Shares Subject to the Plan.
(a) Shares Subject to the Plan. Subject to adjustment as provided in this Section 3(a), Section 3(d), and in Section 3(e) of the Plan, the maximum number of Shares that may be issued in respect of Awards under the Plan is the sum of: (i) 3,340,639 Shares, minus (ii) the number of Shares subject to Prior Plan Awards granted between September 12, 2024 and the Effective Date, plus (iii) up to 115,294 additional Shares subject to Prior Plan Awards to the extent they become available for reissuance pursuant to Section 3(c) hereof. Any Shares issued hereunder may consist, in whole or in part, of authorized and unissued Shares or treasury shares.
(b) Substitute Awards. Notwithstanding the foregoing, any Shares issued in respect of Awards granted in substitution for equity-based awards of an entity acquired by the Company or a Subsidiary, or with which the Company or a Subsidiary combines, will not be counted against the number of Shares available for issuance hereunder.
(c) Share Recycling. If and to the extent that an Award or a Prior Plan Award terminates, expires, is canceled or is forfeited for any reason on or after the Effective Date, the Shares associated with that Award or Prior Plan Award will become available (or again be available) for grant under the Plan. Shares withheld on or after the Effective Date in settlement of a tax withholding obligation associated with an Award or a Prior Plan Award, or in satisfaction of the exercise price payable upon exercise of an Award or Prior Plan Award, will again be available for grant under the Plan.
(d) Incentive Stock Option Limit. Subject to adjustment as provided in Section 3(e) of the Plan, the maximum aggregate number of Shares that may be issued under the Plan in respect of Incentive Stock Options is 10,367,799.
(e) Other Adjustment. In the event of any corporate event or transaction such as a merger, consolidation, reorganization, recapitalization, stock split, reverse stock split, split up, spin-off, combination of shares, exchange of shares, stock dividend, dividend in kind, or other like change in capital structure (other than ordinary cash dividends), or other similar corporate event or transaction affecting the Shares, the Committee, to prevent dilution or enlargement of Participants’ rights under the Plan, shall, in such manner as it deems equitable, substitute or adjust, in its sole discretion, the number and kind of shares that may be issued under the Plan or under any outstanding Awards, the number and kind of shares subject to outstanding Awards, the exercise price, grant price or purchase price applicable to outstanding Awards, and/or any other affected terms and conditions of this Plan or outstanding Awards.
(f) Change in Control. Notwithstanding anything to the contrary set forth in the Plan, upon or in anticipation of any Change in Control, the Committee may, in its sole and absolute discretion and without the need for the consent of any Participant, take one or more of the following actions contingent upon the occurrence of that Change in Control:
(i) cause any or all outstanding Awards to become vested and immediately exercisable (as applicable), in whole or in part;
(ii) cause any outstanding Option or Stock Appreciation Right to become exercisable for a reasonable period in advance of the Change in Control and, to the extent not exercised prior to that Change in Control, cancel that Option or Stock Appreciation Right upon closing of the Change in Control;
(iii) cancel any unvested Award or unvested portion thereof, with or without consideration;
(iv) cancel any Award in exchange for a substitute award;
(v) redeem any Restricted Stock or Restricted Stock Unit for cash and/or other substitute consideration with value equal to the Fair Market Value of an unrestricted Share on the date of the Change in Control;
(vi) cancel any Option or Stock Appreciation Right in exchange for cash and/or other substitute consideration with a value equal to: (A) the number of Shares subject to that Option or Stock Appreciation Right, multiplied by (B) the difference, if any, between the Fair Market Value on the date of the Change in Control and the exercise price of that Option or the base price of the Stock Appreciation Right; provided, that if the Fair Market Value on the date of the Change in Control does not exceed the exercise price of any such Option or the base price of any such Stock Appreciation Right, the Committee may cancel that Option or Stock Appreciation Right without any payment of consideration therefor; and/or
(vii) take such other action as the Committee determines to be appropriate under the circumstances.
In the discretion of the Committee, any cash or substitute consideration payable upon cancellation of an Award may be subjected to (i) vesting terms substantially identical to those that applied to the cancelled Award immediately prior to the Change in Control, or (ii) earn-out, escrow, holdback or similar arrangements, to the extent such arrangements are applicable to any consideration paid to stockholders in connection with the Change in Control.
Notwithstanding any provision of this Section 3(f), in the case of any Award subject to Section 409A of the Code, the Committee shall only be permitted to take actions under this Section 3(f) to the extent that such actions would be consistent with the intended treatment of such Award under Section 409A of the Code.
(g) Foreign Holders. Notwithstanding any provision of the Plan to the contrary, in order to comply with the laws in countries other than the United States in which the Company and its Subsidiaries operate or have employees, directors and consultants, or in order to comply with the requirements of any foreign securities exchange or other Applicable Law, the Committee, in its sole discretion, shall have the power and authority to: (i) modify the terms and conditions of any Award granted to employees, directors and consultants outside the United States to comply with Applicable Law (including, without limitation, applicable foreign laws or listing requirements of any foreign securities exchange); (ii) establish subplans and modify exercise procedures and other terms and procedures, to the extent such actions may be necessary or advisable; provided, however, that no such subplans and/or modifications shall increase the share limitations contained in Section 3; and (iii) take any action, before or after an Award is made, that it deems advisable to obtain approval or comply with any necessary local governmental regulatory exemptions or approvals or listing requirements of any foreign securities exchange.
(h) Annual Compensation Limitations for Non-Employee Directors. Beginning with the fiscal year following the year in which the Effective Date occurs, the aggregate amount of equity and cash compensation (collectively “Compensation”) payable to a Non-Employee Director with respect to a fiscal year, whether under the Plan or otherwise, for services as a Non-Employee Director, shall not exceed $750,000; provided however, that such amount shall be $1,000,000 for the fiscal year in which the applicable Non-Employee Director is initially elected or appointed to the Board (collectively, the “Director Limit”). Equity incentive awards shall be counted towards the Director Limit in the fiscal year in which they are granted, based on the grant date fair value of such awards for financial reporting purposes (but excluding the impact of estimated forfeitures related to service-based vesting provisions). Cash fees shall be counted towards the Director Limit in the fiscal year for which they are reported as compensation in the Company’s director compensation disclosures pursuant to Item 402 of Regulation S-K under the US Securities Act of 1933, or a successor provision. The Director Limit shall not apply to (i) Compensation earned by a Non-Employee Director solely in his or her capacity as chairperson of the Board or lead independent director; (ii) Compensation earned with respect to services a Non-Employee Director provides in a capacity other than as a Non-Employee Director, such as an advisor or consultant to the Company; and (iii) Compensation awarded by the Board to a Non-Employee Director in extraordinary circumstances, as determined by the Board in its discretion, in each case provided that the Non-Employee Director receiving such additional Compensation does not participate in the decision to award such Compensation.
Section 4. Eligibility. Employees, Directors, consultants, and other individuals who provide services to the Company or its Affiliates are eligible to be granted Awards under the Plan; provided, however, that only employees of the Company, any Parent or a Subsidiary are eligible to be granted Incentive Stock Options.
Section 5. Options. Options granted under the Plan may be of two types: (i) Incentive Stock Options or (ii) Non-Qualified Stock Options. The Award Agreement shall state whether such grant is an Incentive Stock Option or a Non-Qualified Stock Option.
The Award Agreement evidencing any Option will incorporate the following terms and conditions and will contain such additional terms and conditions, not inconsistent with the terms of the Plan, as the Committee deems appropriate in its sole and absolute discretion:
(a) Option Price. The exercise price per Share under an Option will be determined by the Committee and will not be less than 100% of the Fair Market Value on the date of the grant. However, any Incentive Stock Option granted to any Participant who, at the time the Option is granted, owns, either directly and/or within the meaning of the attribution rules contained in Section 424(d) of the Code, stock possessing more than 10% of the total combined voting power of all classes of stock of the Company, will have an exercise price per Share of not less than 110% of Fair Market Value on the date of the grant.
(b) Option Term. The term of each Option will be fixed by the Committee, but no Option will be exercisable more than 10 years after the date the Option is granted. However, any Incentive Stock Option granted to any Participant who, at the time such Option is granted, owns, either directly and/or within the meaning of the attribution rules contained in Section 424(d) of the Code, stock possessing more than 10% of the total combined voting power of all classes of stock of the Company, may not have a term of more than 5 years. No Option may be exercised by any Person after expiration of the term of the Option.
(c) Exercisability. Options will vest and be exercisable at such time or times and subject to such terms and conditions as determined by the Committee. Such terms and conditions may include the continued employment or service of the Participant, the attainment of specified individual or corporate performance goals, or such other factors as the Committee may determine in its sole discretion (the “Vesting Conditions”). The Committee may provide in the terms of an Award Agreement that the Participant may exercise the unvested portion of an Option in whole or in part in exchange for shares of Restricted Stock subject to the same vesting terms as the portion of the Option so exercised. Restricted Stock acquired upon the exercise of an unvested Option shall be subject to such additional terms and conditions as determined by the Committee.
(d) Method of Exercise. Subject to the terms of the applicable Award Agreement, the exercisability provisions of Section 5(c) and the termination provisions of Section 7, Options may be exercised in whole or in part from time to time during their term by the delivery of written notice to the Company specifying the number of Shares to be purchased. Such notice will be accompanied by payment in full of the purchase price and any taxes required to be withheld in connection with such exercise, either by certified or bank check, or such other means as the Committee may accept. The Committee may, in its sole discretion, permit payment of the exercise price of an Option in the form of previously acquired Shares based on the Fair Market Value of the Shares on the date the Option is exercised or by means of a “net settlement,” whereby the Option exercise price will not be due in cash and where the number of Shares issued upon such exercise will be equal to: (A) the product of (i) the number of Shares as to which the Option is then being exercised, and (ii) the excess, if any, of (a) the then current Fair Market Value over (b) the Option exercise price, divided by (B) the then current Fair Market Value.
An Option will not confer upon the Participant any of the rights or privileges of a stockholder in the Company unless and until the Participant exercises the Option in accordance with the paragraph above and is issued Shares pursuant to such exercise. For avoidance of doubt, dividends, dividend equivalents and other distributions shall not accrue on any Shares subject to an Option.
(e) Incentive Stock Option Limitations. In the case of an Incentive Stock Option, the aggregate Fair Market Value (determined as of the time of grant) of the Shares with respect to which Incentive Stock Options are exercisable for the first time by the Participant during any calendar year under the Plan and/or any other plan of the Company, its Parent or any Subsidiary will not exceed $100,000. For purposes of applying the foregoing limitation, Incentive Stock Options will be taken into account in the order granted. To the extent any Option does not meet such limitation, that Option will be treated for all purposes as a Non-Qualified Stock Option.
(f) Termination of Service. Unless otherwise specified in the applicable Award Agreement or as otherwise provided by the Committee at or after the time of grant, Options will be subject to the terms of Section 7 with respect to exercise upon or following termination of employment or other service.
Section 6. Stock Appreciation Right. Subject to the other terms of the Plan, the Committee may grant Stock Appreciation Rights to eligible individuals. Each Stock Appreciation Right shall represent the right to receive, upon exercise, an amount equal to the number of Shares subject to the Award that is being exercised multiplied by the excess of (i) the Fair Market Value on the date the Award is exercised, over (ii) the base price specified in the applicable Award Agreement. Distributions may be made in cash, Shares, or a combination of both, at the discretion of the Committee. The Award Agreement evidencing each Stock Appreciation Right shall indicate the base price, the term and the Vesting Conditions for such Award. A Stock Appreciation Right base price may never be less than the Fair Market Value of the underlying common stock of the Company on the date of grant of such Stock Appreciation Right. The term of each Stock Appreciation Right will be fixed by the Committee, but no Stock Appreciation Right will be exercisable more than 10 years after the date the Stock Appreciation Right is granted. Subject to the terms and conditions of the applicable Award Agreement, Stock Appreciation Rights may be exercised in whole or in part from time to time during their term by the delivery of written notice to the Company specifying the portion of the Award to be exercised. Unless otherwise specified in the applicable Award Agreement or as otherwise provided by the Committee at or after the time of grant, Stock Appreciation Rights will be subject to the terms of Section 7 with respect to exercise upon or following termination of employment or other service. For avoidance of doubt, dividends, dividend equivalents and other distributions shall not accrue on any Shares subject to a Stock Appreciation Right.
Section 7. Termination of Service. Unless otherwise specified with respect to a particular Option or Stock Appreciation Right in the applicable Award Agreement or otherwise determined by the Committee, any portion of an Option or Stock Appreciation Right that is not exercisable upon termination of service will expire immediately and automatically upon such termination and any portion of an Option or Stock Appreciation Right that is exercisable upon termination of service will expire on the date it ceases to be exercisable in accordance with this Section 7.
(a) Termination by Reason of Death. If a Participant’s service with the Company or any Affiliate terminates by reason of death, any Option or Stock Appreciation Right held by such Participant may thereafter be exercised, to the extent it was exercisable at the time of his or her death or on such accelerated basis as the Committee may determine at or after grant, by the legal representative of the estate or by the legatee of the Participant, for a period expiring (i) at such time as may be specified by the Committee at or after grant, or (ii) if not specified by the Committee, then 12 months from the date of death, or (iii) if sooner than the applicable period specified under (i) or (ii) above, upon the expiration of the stated term of such Option or Stock Appreciation Right.
(b) Termination by Reason of Disability. If a Participant’s service with the Company or any Affiliate terminates by reason of Disability, any Option or Stock Appreciation Right held by such Participant may thereafter be exercised by the Participant or his or her personal representative, to the extent it was exercisable at the time of termination, or on such accelerated basis as the Committee may determine at or after grant, for a period expiring (i) at such time as may be specified by the Committee at or after grant, or (ii) if not specified by the Committee, then 12 months from the date of termination of service, or (iii) if sooner than the applicable period specified under (i) or (ii) above, upon the expiration of the stated term of such Option or Stock Appreciation Right.
(c) Cause. If a Participant’s service with the Company or any Affiliate is terminated for Cause or if a Participant resigns at a time that there was a Cause basis for such Participant’s termination: (i) any Option or Stock Appreciation Right, or portion thereof, not already exercised will be immediately and automatically forfeited as of the date of such termination, and (ii) any Shares for which the Company has not yet delivered share certificates will be immediately and automatically forfeited and the Company will refund to the Participant the Option exercise price paid for such Shares, if any.
(d) Other Termination. If a Participant’s service with the Company or any Affiliate terminates for any reason other than death, Disability or Cause, any Option or Stock Appreciation Right held by such Participant may thereafter be exercised by the Participant, to the extent it was exercisable at the time of such termination, or on such accelerated basis as the Committee may determine at or after grant, for a period expiring (i) at such time as may be specified by the Committee at or after grant, or (ii) if not specified by the Committee, then 90 days from the date of termination of service, or (iii) if sooner than the applicable period specified under (i) or (ii) above, upon the expiration of the stated term of such Option or Stock Appreciation Right.
Section 8. Restricted Stock.
(a) Issuance. Restricted Stock may be issued either alone or in conjunction with other Awards. The Committee will determine the time or times within which Restricted Stock may be subject to forfeiture, and all other conditions of such Awards. The purchase price for Restricted Stock may, but need not, be zero.
(b) Certificates. Upon the Award of Restricted Stock, the Committee may direct that a certificate or certificates representing the number of Shares subject to such Award be issued to the Participant or placed in a restricted stock account (including an electronic account) with the transfer agent and in either case designating the Participant as the registered owner. The certificate(s), if any, representing such shares shall be physically or electronically legended, as applicable, as to sale, transfer, assignment, pledge or other encumbrances during the Restriction Period. If physical certificates are issued, they will be held in escrow by the Company or its designee during the Restriction Period. As a condition to any Award of Restricted Stock, the Participant may be required to deliver to the Company a share power, endorsed in blank, relating to the Shares covered by such Award.
(c) Restrictions and Conditions. The Award Agreement evidencing the grant of any Restricted Stock will incorporate the following terms and conditions and such additional terms and conditions, not inconsistent with the terms of the Plan, as the Committee deems appropriate in its sole and absolute discretion:
(i) During a period commencing with the date of an Award of Restricted Stock and ending at such time or times as specified by the Committee (the “Restriction Period”), the Participant will not be permitted to sell, transfer, pledge, assign or otherwise encumber Restricted Stock awarded under the Plan. The Committee may condition the lapse of restrictions on Restricted Stock upon one or more Vesting Conditions.
(ii) While any Share of Restricted Stock remains subject to restriction, the Participant will have, with respect to the Restricted Stock, the right to vote the Shares. If any cash distributions or dividends are payable with respect to the Restricted Stock, the Committee, in its sole discretion, may require the cash distributions or dividends to be subjected to the same Restriction Period as is applicable to the Restricted Stock with respect to which such amounts are paid, or, if the Committee so determines, reinvested in additional Restricted Stock to the extent Shares are available under Section 3 of the Plan. A Participant shall not be entitled to interest with respect to any dividends or distributions subjected to the Restriction Period. Any distributions or dividends paid in the form of securities with respect to Restricted Stock will be subject to the same terms and conditions as the Restricted Stock with respect to which they were paid, including, without limitation, the same Restriction Period.
(iii) Subject to the provisions of the applicable Award Agreement or as otherwise determined by the Committee, if a Participant’s service with the Company and its Affiliates terminates prior to the expiration of the applicable Restriction Period, the Participant’s Restricted Stock that then remains subject to forfeiture will then be forfeited automatically.
Section 9. Restricted Stock Units. Subject to the other terms of the Plan, the Committee may grant Restricted Stock Units to eligible individuals and may impose one or more Vesting Conditions on such units. Each Restricted Stock Unit will represent a right to receive from the Company, upon fulfillment of any applicable conditions, an amount equal to the Fair Market Value (at the time of the distribution) of one Share. Distributions may be made in cash, Shares, or a combination of both, at the discretion of the Committee. The Award Agreement evidencing a Restricted Stock Unit shall set forth the Vesting Conditions and time and form of payment with respect to such Award. The Participant shall not have any stockholder rights with respect to the Shares subject to a Restricted Stock Unit Award until that Award vests and the Shares are actually issued thereunder; provided, however, that an Award Agreement may provide for the inclusion of dividend equivalent payments or unit credits with respect to the Award in the discretion of the Committee. Subject to the provisions of the applicable Award Agreement or as otherwise determined by the Committee, if a Participant’s service with the Company terminates prior to the Restricted Stock Unit Award vesting in full, any portion of the Participant’s Restricted Stock Units that then remain subject to forfeiture will then be forfeited automatically.
Section 10. Cash or Other Stock Based Awards. Subject to the other terms of the Plan, the Committee may grant Cash or Other Stock Based Awards (including Awards to receive unrestricted Shares or immediate cash payments) to eligible individuals. The Award Agreement evidencing a Cash or Other Stock Based Award shall set forth the terms and conditions of such Cash or Other Stock Based Award, including, as applicable, the term, any exercise or purchase price, performance goals, Vesting Conditions and other terms and conditions. Payment in respect of a Cash or Other Stock Based Award may be made in cash, Shares, or a combination of cash and Shares, as determined by the Committee.
Section 11. Amendments and Termination. Subject to any stockholder approval that may be required under Applicable Law, the Plan may be amended or terminated at any time or from time to time by the Board.
Section 12. Repricing. The Committee and the Board may not reprice any outstanding Options or Stock Appreciation Rights without stockholder approval, whether such repricing is accomplished by (i) implementing any cancellation/re-grant program pursuant to which outstanding Options or Stock Appreciation Rights under the Plan are cancelled and new Options or Stock Appreciation Rights are granted in replacement with a lower exercise or base price per share, (ii) cancelling outstanding Options or Stock Appreciation Rights under the Plan with exercise prices or base prices per share in excess of the then current Fair Market Value for consideration payable in equity securities of the Company or cash or (iii) otherwise directly or indirectly reducing the exercise price or base price in effect for outstanding Options or Stock Appreciation Rights under the Plan.
Section 13. Conditions Upon Grant of Awards and Issuance of Shares.
(a) The implementation of the Plan, the grant of any Award and the issuance of Shares in connection with the issuance, exercise or vesting of any Award made under the Plan shall be subject to the Company’s procurement of all approvals and permits required by regulatory authorities having jurisdiction over the Plan, the Awards made under the Plan and the Shares issuable pursuant to those Awards.
(b) No Shares or other assets shall be issued or delivered under the Plan unless and until there shall have been compliance with all applicable requirements of Applicable Law.
Section 14. Limits on Transferability; Beneficiaries. No Award or other right or interest of a Participant under the Plan shall be pledged, encumbered, or hypothecated to, or in favor of, or subject to any lien, obligation, or liability of such Participant to, any party, other than the Company, any Subsidiary or Affiliate, or assigned or transferred by such Participant other than by will or the laws of descent and distribution, and such Awards and rights shall be exercisable during the lifetime of the Participant only by the Participant or his or her guardian or legal representative. Notwithstanding the foregoing, the Committee may, in its discretion, provide that Awards or other rights or interests of a Participant granted pursuant to the Plan (other than an Incentive Stock Option) be transferable, without consideration, to immediate family members (i.e., children, grandchildren or spouse), to trusts for the benefit of such immediate family members, to partnerships in which such family members are the only partners, to other similar estate planning vehicles, or to such other transferees as the Committee permits (taking into account the restrictions or requirements of applicable tax, securities and other laws). The Committee may attach to such transferability feature such terms and conditions as it deems advisable. In addition, a Participant may, in the manner established by the Committee, designate a beneficiary (which may be a person or a trust) to exercise the rights of the Participant, and to receive any distribution, with respect to any Award upon the death of the Participant. A beneficiary, guardian, legal representative or other person claiming any rights under the Plan from or through any Participant shall be subject to all terms and conditions of the Plan and any Award Agreement applicable to such Participant, except as otherwise determined by the Committee, and to any additional restrictions deemed necessary or appropriate by the Committee.
Section 15. Withholding of Taxes. No later than the date as of which an amount first becomes includible in the gross income of the Participant for federal income tax purposes with respect to any Award under the Plan, the Participant will pay to the Company, or make arrangements satisfactory to the Company regarding the payment of, any federal, state or local taxes of any kind required by law to be withheld with respect to such amount. To the extent authorized by the Committee, the required tax withholding may be satisfied by the withholding of Shares subject to the Award based on the fair market value of those Shares on the date of withholding, but in any case not in excess of the amount determined based on the maximum statutory tax rate in the applicable jurisdiction. The obligations of the Company under the Plan will be conditioned on such payment or arrangements and the Company will have the right to deduct any such taxes from any payment of any kind otherwise due to the Participant.
Section 16. Liability of Company.
(a) Inability to Obtain Authority. If the Company cannot, by the exercise of commercially reasonable efforts, obtain authority from any regulatory body having jurisdiction for the sale of any Shares under this Plan, and such authority is deemed by the Company’s counsel to be necessary to the lawful issuance of those Shares, the Company will be relieved of any liability for failing to issue or sell those Shares.
(b) Rights of Participants and Beneficiaries. The Company will pay all amounts payable under this Plan only to the applicable Participant, or beneficiaries entitled thereto pursuant to this Plan. The Company will not be liable for the debts, contracts, or engagements of any Participant or his or her beneficiaries, and rights to cash payments under this Plan may not be taken in execution by attachment or garnishment, or by any other legal or equitable proceeding while in the hands of the Company.
Section 17. General Provisions.
(a) The Committee may require each Participant to represent to and agree with the Company in writing that the Participant is acquiring securities of the Company for investment purposes and without a view to distribution thereof and as to such other matters as the Committee believes are appropriate.
(b) The Awards, and any Shares associated therewith, shall be subject to the Company’s stock ownership, securities trading, anti-hedging and other similar policies, as in effect from time to time.
(c) All certificates for Shares or other securities delivered under the Plan, if any, will be subject to such stop-transfer orders and other restrictions as the Board may deem advisable under the rules, regulations and other requirements of the Securities Act, the Exchange Act, any stock exchange upon which the Shares are then listed, and any other Applicable Law, and the Board may cause Shares or other securities to be legended to reflect those restrictions.
(d) Nothing contained in the Plan will prevent the Company from adopting other or additional compensation arrangements, subject to stockholder approval if such approval is required. Similarly, the grant of any Award will not in any way affect the right or power of the Company to make adjustments, reclassification or changes in its capital or business structure or to merge, consolidate, dissolve, liquidate, sell or transfer all or any part of its business or assets.
(e) Neither the adoption of the Plan nor the execution of any document in connection with the Plan will: (i) confer upon any employee or other service provider of the Company or an Affiliate any right to continued employment or engagement with the Company or such Affiliate, or (ii) interfere in any way with the right of the Company or such Affiliate to terminate the employment or engagement of any of its employees or other service providers at any time.
Section 18. Term of Plan. The Plan was adopted by the Board on September 12, 2024 (the “Adoption Date”), subject to approval by the Company’s stockholders (the date of such approval, the “Effective Date”). Unless the Plan shall theretofore have been terminated in accordance with Section 11, the Plan shall terminate on September 12, 2034, which is the 10-year anniversary of the Adoption Date, and no Awards shall thereafter be granted under the Plan.
Section 19. Clawbacks. The Awards (whether vested or unvested) shall be subject to rescission, cancellation or recoupment, in whole or in part, under any current or future “clawback” or similar policy of the Company that is applicable to the Participant. Notwithstanding any other provisions in this Plan, any Award which is subject to recovery under any law, government regulation or stock exchange listing requirement, will be subject to such deductions and clawback as may be required to be made pursuant to such law, government regulation or stock exchange listing requirement.
Section 20. Section 409A. All Awards are intended to be exempt from or comply with the requirements of Section 409A of the Code (“Section 409A”) and should be interpreted accordingly. Nonetheless, the Company does not guaranty any particular tax treatment for any Award. For any Award that is non-qualified deferred compensation subject to Section 409A, the Committee may elect to liquidate such Award at any time in a manner intended to comply with Treas. Reg. § 1.409A-3(j)(4)(ix) or any successor provision. Notwithstanding anything to the contrary in the Plan or an Award, if at the time of a Participant’s separation from service, such Participant is a “specified employee" (within the meaning of Section 409A), then any amounts payable under the Plan on account of such separation from service that would (but for this provision) be payable within six (6) months following the date of the separation from service shall instead be paid on the next business day following the expiration of such six (6) month period or, if earlier, upon the Participant’s death, to the extent compliance with the requirements of Treas. Reg. § 1.409A‑3(i)(2) (or any successor provision) is necessary to avoid the application of an additional tax under Section 409A to such amounts. Notwithstanding the foregoing, neither the Company nor the Committee shall have any obligation to take any action to prevent the assessment of any excise tax or penalty on any Participant under Section 409A and neither the Company nor the Committee will have any liability to any Participant for such tax or penalty.
Section 21. Invalid Provisions. In the event that any provision of this Plan is found to be invalid or otherwise unenforceable under any Applicable Law, such invalidity or unenforceability will not be construed as rendering any other provisions contained herein as invalid or unenforceable, and all such other provisions will be given full force and effect to the same extent as though the invalid or unenforceable provision was not contained herein.
Section 22. Governing Law. The Plan and all Awards granted hereunder will be governed by and construed in accordance with the laws and judicial decisions of the State of Delaware, without regard to the application of the principles of conflicts of laws.
Section 23. Notices. Any notice to be given to the Company pursuant to the provisions of this Plan must be given in writing and addressed, if to the Company, to its principal executive office to the attention of its Chief Financial Officer (or such other Person as the Company may designate in writing from time to time), and, if to a Participant, to the address contained in the Company’s personnel files, or at such other address as that Participant may hereafter designate in writing to the Company. Any such notice will be deemed duly given: if delivered personally or via recognized overnight delivery service, on the date and at the time so delivered; if sent via telecopier or email, on the date and at the time telecopied or emailed with confirmation of delivery; or, if mailed, five (5) days after the date of mailing by registered or certified mail.




THE OFFER AND SALE OF THE SECURITIES REPRESENTED BY THIS CERTIFICATE TO WHICH THIS CONFIRMATION RELATES OR THE SECURITIES INTO WHICH THIS SECURITY IS EXERCISABLE HAVE NOT BEEN REGISTERED UNDER THE SECURITIES ACT OF 1933, AS AMENDED, OR THE SECURITIES LAWS OF ANY STATE OF THE UNITED STATES. THE SECURITIES MAY NOT BE SOLD, OFFERED FOR SALE, PLEDGED, HYPOTHECATED, TRANSFERRED OR ASSIGNED IN THE ABSENCE OF AN EFFECTIVE REGISTRATION STATEMENT FOR THE SECURITIES UNDER APPLICABLE SECURITIES LAWS, OR UNLESS OFFERED, SOLD, PLEDGED, HYPOTHECATED OR TRANSFERRED PURSUANT TO AN AVAILABLE EXEMPTION FROM THE REGISTRATION REQUIREMENTS OF THOSE LAWS. THE COMPANY AND ITS TRANSFER AGENT SHALL BE ENTITLED TO REQUIRE AN OPINION OF COUNSEL SATISFACTORY TO THE COMPANY AND THE TRANSFER AGENT THAT SUCH REGISTRATION IS NOT REQUIRED. NOTWITHSTANDING THE FOREGOING, THE SECURITIES MAY BE PLEDGED IN CONNECTION WITH A BONA FIDE MARGIN ACCOUNT OR OTHER LOAN OR FINANCING ARRANGEMENT SECURED BY THE SECURITIES.
PIERIS PHARMACEUTICALS, INC.
FORM OF PRE-FUNDED WARRANT TO PURCHASE COMMON STOCK
|
Warrant No. [•] |
Number of Shares: [•] |
|
(subject to adjustment) |
|
|
Original Issue Date: [•] |
|
|
Beneficial Ownership Limitation: [•]% |
Pieris Pharmaceuticals, Inc., a Nevada corporation (the “Company”), hereby certifies that, for good and valuable consideration, the receipt and sufficiency of which are hereby acknowledged, [•] or its permitted registered assigns (the “Holder”), is entitled, subject to the terms set forth below, to purchase from the Company up to a total of [•] shares of common stock, $0.001 par value per share (the “Common Stock”), of the Company (each such share, a “Warrant Share” and all such shares, the “Warrant Shares”) at an exercise price per share equal to $0.001 per share (as adjusted from time to time as provided in Section 9 herein, the “Exercise Price”), upon surrender of this Warrant to Purchase Common Stock (including any Warrants to Purchase Common Stock issued in exchange, transfer or replacement hereof, the “Warrant”), and subject to the following terms and conditions:
1. Definitions. For purposes of this Warrant, the following terms shall have the following meanings:
(a) “Affiliate” when used with respect to any party shall mean any Person who is an “affiliate” of that party within the meaning of Rule 405 promulgated under the Securities Act.
(b) “Attribution Parties” means, collectively, the following Persons: (i) any direct or indirect Affiliates of the Holder, (ii) any Person acting or who could be deemed to be acting as a group together with the Holder or any of the foregoing and (iii) any other Persons whose beneficial ownership of the Company’s Common Stock would or could be aggregated with the Holder’s and the other Attribution Parties for purposes of Section 13(d) or Section 16 of the Exchange Act. For clarity, the purpose of the foregoing is to subject collectively the Holder and all other Attribution Parties to the Beneficial Ownership Limitation (as defined below).
(c) “Closing Sale Price” means, for any security as of any date, the last trade price for such security on the Principal Trading Market for such security, as reported by Bloomberg Financial Markets, or, if such Principal Trading Market begins to operate on an extended hours basis and does not designate the last trade price, then the last trade price of such security prior to 4:00 P.M., New York City time, as reported by Bloomberg Financial Markets, or if the foregoing do not apply, the last trade price of such security in the over-the-counter market on the electronic bulletin board for such security as reported by Bloomberg Financial Markets, or, if no last trade price is reported for such security by Bloomberg Financial Markets, the average of the bid and ask prices, of any market makers for such security as reported in the “pink sheets” by Pink Sheets LLC. If the Closing Sale Price cannot be calculated for a security on a particular date on any of the foregoing bases, the Closing Sale Price of such security on such date shall be the fair market value as mutually determined by the Company and the Holder. If the Company and the Holder are unable to agree upon the fair market value of such security, then the Board of Directors of the Company shall use its good faith judgment to determine the fair market value. The determination of the Board of Directors of the Company shall be binding upon all parties absent demonstrable error. All such determinations shall be appropriately adjusted for any stock dividend, stock split, stock combination or other similar transaction during the applicable calculation period.
(d) “Exchange Act” means the Securities Exchange Act of 1934, as amended.
(e) “Person” means an individual or a corporation, partnership, trust, incorporated or unincorporated association, joint venture, limited liability company, joint stock company, government (or an agency or subdivision thereof) or other entity of any kind.
(f) “Principal Trading Market” means the trading market on which the Common Stock is primarily listed on and quoted for trading, which, as of the Original Issue Date shall be the Nasdaq Global Market.
(g) “SEC” means the United States Securities and Exchange Commission.
(h) “Securities Act” means the Securities Act of 1933, as amended.
(i) “Trading Day” means a day on which the Principal Trading Market is open for business.
(j) “Transfer Agent” means Computershare Trust Company, N.A., the current transfer agent of the Company, or any successor transfer agent for the Company.
2. Registration of Warrants. The Company shall register this Warrant, upon records to be maintained by the Company for that purpose (the “Warrant Register”), in the name of the record Holder (which shall include the initial Holder or, as the case may be, any registered assignee to which this Warrant is permissibly assigned hereunder) from time to time. The Company may deem and treat the registered Holder of this Warrant as the absolute owner hereof for the purpose of any exercise hereof or any distribution to the Holder, and for all other purposes, absent actual notice to the contrary.
3. Registration of Transfers. Subject to compliance with all applicable securities laws, the Company shall, or will cause its Transfer Agent to, register the transfer of all or any portion of this Warrant in the Warrant Register, upon surrender of this Warrant, together with a written assignment of this Warrant substantially in the form attached hereto as Schedule 2, duly executed by the Holderand payment for all applicable transfer taxes. Upon any such registration or transfer, a new warrant to purchase Common Stock in substantially the form of this Warrant (any such new warrant, a “New Warrant”) evidencing the portion of this Warrant so transferred shall be issued to the transferee, and a New Warrant evidencing the remaining portion of this Warrant not so transferred, if any, shall be issued to the transferring Holder. The acceptance of the New Warrant by the transferee thereof shall be deemed the acceptance by such transferee of all of the rights and obligations in respect of the New Warrant that the Holder has in respect of this Warrant. The Company shall, or will cause its Transfer Agent to, prepare, issue and deliver at the Company’s own expense any New Warrant under this Section 3. Until due presentment for registration of transfer, the Company may treat the registered Holder hereof as the owner and holder for all purposes, and the Company shall not be affected by any notice to the contrary.
4. Exercise of Warrants.
(a) All or any part of this Warrant shall be exercisable by the registered Holder in any manner permitted by Section 10 of this Warrant at any time and from time to time on or after the Original Issue Date.
(b) The Holder may exercise this Warrant by delivering to the Company (i) an exercise notice, in the form attached as Schedule 1 hereto (the “Exercise Notice”), completed and duly signed, and (ii) payment of the Exercise Price for the number of Warrant Shares as to which this Warrant is being exercised (which may take the form of a “cashless exercise” if so indicated in the Exercise Notice pursuant to Section 10 below), and the date on which the last of such items is delivered to the Company (as determined in accordance with the notice provisions hereof) is an “Exercise Date.” The Holder shall not be required to deliver the original Warrant in order to effect an exercise hereunder. Execution and delivery of the Exercise Notice shall have the same effect as cancellation of the original Warrant and issuance of a New Warrant evidencing the right to purchase the remaining number of Warrant Shares, if any.
5. Delivery of Warrant Shares.
(a) Upon exercise of this Warrant, the Company shall promptly (but in no event later than two (2) Trading Days after the Exercise Date), upon the request of the Holder, credit such aggregate number of shares of Common Stock to which the Holder is entitled pursuant to such exercise to the Holder’s or its designee’s balance account with The Depository Trust Company (“DTC”) through its Deposit Withdrawal Agent Commission system, or if the Transfer Agent is not participating in the Fast Automated Securities Transfer Program (FAST) or if the certificates are required to bear a legend regarding restriction on transferability, issue and dispatch by overnight courier to the address as specified in the Exercise Notice, a certificate, registered in the Company’s share register in the name of the Holder or its designee, for the number of shares of Common Stock to which the Holder is entitled pursuant to such exercise. The Holder, or any Person permissibly so designated by the Holder to receive Warrant Shares, shall be deemed to have become the holder of record of such Warrant Shares as of the Exercise Date, irrespective of the date such Warrant Shares are credited to the Holder’s or its designee’s DTC account or the date of delivery of the certificates evidencing such Warrant Shares, as the case may be.
(b) If by the close of the second (2nd) Trading Day after the Exercise Date, the Company fails to deliver to the Holder a certificate representing the required number of Warrant Shares in the manner required pursuant to Section 5(a) or fails to credit the Holder’s or its designee’s balance account with DTC for such number of Warrant Shares to which the Holder is entitled, and if after such second (2nd) Trading Day and prior to the receipt of such Warrant Shares, the Holder purchases (in an open market transaction or otherwise) shares of Common Stock to deliver in satisfaction of a sale by the Holder of the Warrant Shares which the Holder anticipated receiving upon such exercise (a “Buy-In”), then the Company shall, within two (2) Trading Days after the Holder’s request and in the Holder’s sole discretion, either (1) pay in cash to the Holder an amount equal to the Holder’s total purchase price (including brokerage commissions, if any) for the shares of Common Stock so purchased, at which point the Company’s obligation to deliver such certificate (and to issue such Warrant Shares) shall terminate or (2) promptly honor its obligation to deliver to the Holder a certificate or certificates representing such Warrant Shares or credit the Holder’s or its designee’s balance account with DTC for such Warrant Shares and pay cash to the Holder in an amount equal to the excess (if any) of Holder’s total purchase price (including brokerage commissions, if any) for the shares of Common Stock so purchased in the Buy-In over the product of (A) the number of shares of Common Stock purchased in the Buy-In, times (B) the Closing Sale Price of a share of Common Stock on the Exercise Date.
(c) To the extent permitted by law and subject to Section 5(b), the Company’s obligations to issue and deliver Warrant Shares in accordance with and subject to the terms hereof (including the limitations set forth in Section 11 below) are absolute and unconditional, irrespective of any action or inaction by the Holder to enforce the same, any waiver or consent with respect to any provision hereof, the recovery of any judgment against any Person or any action to enforce the same, or any setoff, counterclaim, recoupment, limitation or termination, or any breach or alleged breach by the Holder or any other Person of any obligation to the Company or any violation or alleged violation of law by the Holder or any other Person, and irrespective of any other circumstance that might otherwise limit such obligation of the Company to the Holder in connection with the issuance of Warrant Shares. Subject to Section 5(b), nothing herein shall limit the Holder’s right to pursue any other remedies available to it hereunder, at law or in equity including, without limitation, a decree of specific performance and/or injunctive relief with respect to the Company’s failure to timely deliver certificates representing shares of Common Stock upon exercise of the Warrant as required pursuant to the terms hereof.
6. Charges, Taxes and Expenses. Issuance and delivery of certificates for shares of Common Stock upon exercise of this Warrant shall be made without charge to the Holder for any issue or transfer tax, transfer agent fee or other incidental tax or expense (excluding any applicable stamp duties) in respect of the issuance of such certificates, all of which taxes and expenses shall be paid by the Company; provided, however, that the Company shall not be required to pay any tax that may be payable in respect of any transfer involved in the registration of any certificates for Warrant Shares or the Warrants in a name other than that of the Holder or an Affiliate thereof. The Holder shall be responsible for all other tax liability that may arise as a result of holding or transferring this Warrant or receiving Warrant Shares upon exercise hereof.
7. Replacement of Warrant. If this Warrant is mutilated, lost, stolen or destroyed, the Company shall issue or cause to be issued in exchange and substitution for and upon cancellation hereof, or in lieu of and substitution for this Warrant, a New Warrant, but only upon receipt of evidence reasonably satisfactory to the Company of such loss, theft or destruction (in such case) and, in each case, a customary and reasonable indemnity, if requested by the Company, but without any requirement that a surety bond be procured, provided or posted. Applicants for a New Warrant under such circumstances shall also comply with such other reasonable regulations and procedures as the Company may prescribe. If a New Warrant is requested as a result of a mutilation of this Warrant, then the Holder shall deliver such mutilated Warrant to the Company as a condition precedent to the Company’s obligation to issue the New Warrant.
8. Reservation of Warrant Shares. The Company covenants that it will, at all times while this Warrant is outstanding, reserve and keep available out of the aggregate of its authorized but unissued and otherwise unreserved Common Stock, solely for the purpose of enabling it to issue Warrant Shares upon exercise of this Warrant as herein provided, the number of Warrant Shares that are initially issuable and deliverable upon the exercise of this entire Warrant, free from preemptive rights or any other contingent purchase rights of persons other than the Holder (taking into account the adjustments and restrictions of Section 9). The failure of the Company to reserve and keep available out of the aggregate of its authorized but unissued and otherwise unreserved Common Stock a sufficient number of shares of Common Stock to enable it to issue Warrant Shares upon exercise of this Warrant as herein provided is referred to herein as an “Authorized Share Failure.” The Company covenants that all Warrant Shares so issuable and deliverable shall, upon issuance and the payment of the applicable Exercise Price in accordance with the terms hereof, be duly and validly authorized, issued and fully paid and non-assessable. The Company will take all commercially reasonable actions as may be necessary to assure that such shares of Common Stock may be issued as provided herein without violation of any applicable law or regulation, or of any requirements of any securities exchange or automated quotation system upon which the Common Stock may be listed. The Company further covenants that it will not, without the prior written consent of the Holder, take any actions to increase the par value of the Common Stock at any time while this Warrant is outstanding.
9. Certain Adjustments. The Exercise Price and number of Warrant Shares issuable upon exercise of this Warrant are subject to adjustment from time to time as set forth in this Section 9.
(a) Stock Dividends and Splits. If the Company, at any time while this Warrant is outstanding, (i) pays a stock dividend on its Common Stock or otherwise makes a distribution on any class of capital stock issued and outstanding on the Original Issue Date that is payable in shares of Common Stock, (ii) subdivides its outstanding shares of Common Stock into a larger number of shares of Common Stock, (iii) combines its outstanding shares of Common Stock into a smaller number of shares of Common Stock or (iv) issues by reclassification of shares of capital stock any additional shares of Common Stock of the Company, then in each such case the Exercise Price shall be multiplied by a fraction, the numerator of which shall be the number of shares of Common Stock outstanding immediately before such event and the denominator of which shall be the number of shares of Common Stock outstanding immediately after such event. Any adjustment made pursuant to clause (i) of this paragraph shall become effective immediately after the record date for the determination of stockholders entitled to receive such dividend or distribution, provided, however, that if such record date shall have been fixed and such dividend is not fully paid on the date fixed therefor, the Exercise Price shall be recomputed accordingly as of the close of business on such record date and thereafter the Exercise Price shall be adjusted pursuant to this paragraph as of the time of actual payment of such dividends. Any adjustment pursuant to clause (ii) or (iii) of this paragraph shall become effective immediately after the effective date of such subdivision or combination.
(b) Pro Rata Distributions. In addition to any adjustments pursuant to the other subsections of this Section 9, if, on or after the Original Issue Date, the Company shall declare or make any dividend or other pro rata distribution of its assets (or rights to acquire its assets) to holders of shares of Common Stock, by way of return of capital or otherwise (including, without limitation, any distribution of cash, stock or other securities, property, options, evidence of indebtedness or any other assets by way of a dividend, spin off, reclassification, corporate rearrangement, scheme of arrangement or other similar transaction) (a “Distribution”), at any time after the Original Issue Date, then, in each such case, the Holder shall be entitled to participate in such Distribution to the same extent that the Holder would have participated therein if the Holder had held the number of shares of Common Stock acquirable upon complete exercise of this Warrant (without regard to any limitations or restrictions on exercise of this Warrant, including without limitation, the Beneficial Ownership Limitation (as defined below)) immediately before the date on which a record is taken for such Distribution, or, if no such record is taken, the date as of which the record holders of shares of Common Stock are to be determined for the participation in such Distribution (provided, that to the extent that the Holder’s right to participate in any such Distribution would result in the Holder and the other Attribution Parties exceeding the Beneficial Ownership Limitation, then the Holder shall not be entitled to participate in such Distribution to such extent (and shall not be entitled to beneficial ownership of such shares of Common Stock as a result of such Distribution (and beneficial ownership) to such extent) and the portion of such Distribution shall be held in abeyance for the benefit of the Holder until such time or times as its right thereto would not result in the Holder and the other Attribution Parties exceeding the Beneficial Ownership Limitation, at which time or times the Holder shall be granted such Distribution (and any Distributions declared or made on such initial Distribution or on any subsequent Distribution held similarly in abeyance) to the same extent as if there had been no such limitation).
(c) Purchase Rights. In addition to any adjustments pursuant to the other subsections of this Section 9, if at any time on or after the Original Issue Date, the Company grants, issues or sells any Options, Convertible Securities or rights to purchase stock, warrants, securities or other property, in each case pro rata to the record holders of any class of Common Stock (the “Purchase Rights”), then the Holder will be entitled to acquire, upon the terms applicable to such Purchase Rights, the aggregate Purchase Rights which the Holder could have acquired if the Holder had held the number of shares of Common Stock acquirable upon complete exercise of this Warrant (without regard to any limitations or restrictions on exercise of this Warrant, including without limitation, the Beneficial Ownership Limitation) immediately before the date on which a record is taken for the grant, issuance or sale of such Purchase Rights, or, if no such record is taken, the date as of which the record holders of Common Stock are to be determined for the grant, issuance or sale of such Purchase Rights (provided, that to the extent that the Holder’s right to participate in any such Purchase Right would result in the Holder and the other Attribution Parties exceeding the Beneficial Ownership Limitation, then the Holder shall not be entitled to participate in such Purchase Right to such extent (and shall not be entitled to beneficial ownership of such Common Stock as a result of such Purchase Right (and beneficial ownership) to such extent) and such Purchase Right to such extent shall be held in abeyance for the benefit of the Holder until such time or times as its right thereto would not result in the Holder and the other Attribution Parties exceeding the Beneficial Ownership Limitation, at which time or times the Holder shall be granted such right (and any Purchase Right granted, issued or sold on such initial Purchase Right or on any subsequent Purchase Right to be held similarly in abeyance) to the same extent as if there had been no such limitation). As used in this Section 9(c), (i) “Options” means any rights, warrants or options to subscribe for or purchase shares of Common Stock or Convertible Securities and (ii) “Convertible Securities” mean any stock or securities (other than Options) directly or indirectly convertible into or exercisable or exchangeable for shares of Common Stock.
(d) Fundamental Transactions. If, at any time while this Warrant is outstanding (i) the Company effects any merger or consolidation of the Company with or into another Person, in which the Company is not the surviving entity or the stockholders of the Company immediately prior to such merger or consolidation do not own, directly or indirectly, at least 50% of the voting power of the surviving entity immediately after such merger or consolidation, (ii) the Company effects any sale to another Person of all or substantially all of its assets in one or a series of related transactions, (iii) pursuant to any tender offer or exchange offer (whether by the Company or another Person), holders of capital stock who tender shares representing more than 50% of the voting power of the capital stock of the Company and the Company or such other Person, as applicable, accepts such tender for payment, (iv) the Company consummates a stock purchase agreement or other business combination (including, without limitation, a reorganization, recapitalization, spin-off or scheme of arrangement) with another Person whereby such other Person acquires more than the 50% of the voting power of the capital stock of the Company or (v) the Company effects any reclassification of the Common Stock or any compulsory share exchange pursuant to which the Common Stock is effectively converted into or exchanged for other securities, cash or property (other than as a result of a subdivision or combination of shares of Common Stock covered by Section 9(a) above) (in any such case, a “Fundamental Transaction”), then following such Fundamental Transaction the Holder shall have the right to receive, upon exercise of this Warrant, the same amount and kind of securities, cash or property as it would have been entitled to receive upon the occurrence of such Fundamental Transaction if it had been, immediately prior to such Fundamental Transaction, the holder of the number of Warrant Shares then issuable upon exercise in full of this Warrant without regard to any limitations on exercise contained herein (the “Alternate Consideration”). The Company shall not effect any Fundamental Transaction in which the Company is not the surviving entity or the Alternate Consideration includes securities of another Person unless (i) the Alternate Consideration is solely cash and the Company provides for the simultaneous “cashless exercise” of this Warrant pursuant to Section 10 below or (ii) prior to or simultaneously with the consummation thereof, any successor to the Company, surviving entity or other Person (including any purchaser of assets of the Company) shall assume the obligation to deliver to the Holder such Alternate Consideration as, in accordance with the foregoing provisions, the Holder may be entitled to receive, and the other obligations under this Warrant. The provisions of this paragraph (d) shall similarly apply to subsequent transactions analogous of a Fundamental Transaction type.
(e) Number of Warrant Shares. Simultaneously with any adjustment to the Exercise Price pursuant to paragraph (a) of this Section 9, the number of Warrant Shares that may be purchased upon exercise of this Warrant shall be increased or decreased proportionately, so that after such adjustment the aggregate Exercise Price payable hereunder for the increased or decreased number of Warrant Shares shall be the same as the aggregate Exercise Price in effect immediately prior to such adjustment.
(f) Calculations. All calculations under this Section 9 shall be made to the nearest one tenth of one cent or the nearest share, as applicable.
(g) Notice of Adjustments. Upon the occurrence of each adjustment pursuant to this Section 9, the Company at its expense will, at the written request of the Holder, promptly compute such adjustment, in good faith, in accordance with the terms of this Warrant and prepare a certificate setting forth such adjustment, including a statement of the adjusted Exercise Price and adjusted number or type of Warrant Shares or other securities issuable upon exercise of this Warrant (as applicable), describing the transactions giving rise to such adjustments and showing in detail the facts upon which such adjustment is based. Upon written request, the Company will promptly deliver a copy of each such certificate to the Holder and to the Company’s transfer agent.
(h) Notice of Corporate Events. If, while this Warrant is outstanding, the Company (i) declares a dividend or any other pro rata distribution of cash, securities or other property in respect of its Common Stock, including, without limitation, any granting of rights or warrants to subscribe for or purchase any capital stock of the Company or any subsidiary, (ii) authorizes or approves, enters into any agreement contemplating or solicits stockholder approval for any Fundamental Transaction or (iii) authorizes the voluntary dissolution, liquidation or winding up of the affairs of the Company, then the Company shall deliver to the Holder a notice of such transaction at least ten (10) days prior to the applicable record or effective date on which a Person would need to hold Common Stock in order to participate in or vote with respect to such transaction; provided, however, that the failure to deliver such notice or any defect therein shall not affect the validity of the corporate action required to be described in such notice. In addition, if while this Warrant is outstanding, the Company authorizes or approves, enters into any agreement contemplating or solicits stockholder approval for any Fundamental Transaction contemplated by Section 9(d), other than a Fundamental Transaction under clause (iii) of Section 9(d), the Company shall deliver to the Holder a notice of such Fundamental Transaction at least thirty (30) days prior to the date such Fundamental Transaction is consummated. The Holder agrees to maintain any information disclosed pursuant to this Section 9(h) in confidence until such information is publicly available, and shall comply with applicable law with respect to trading in the Company’s securities following receipt of any such information.
10. Payment of Exercise Price. Notwithstanding anything contained herein to the contrary, the Holder may, in its sole discretion, satisfy its obligation to pay the Exercise Price through a “cashless exercise,” in which event the Company shall issue to the Holder the number of Warrant Shares in an exchange of securities effected pursuant to Section 3(a)(9) of the Securities Act, determined as follows:
X = Y [(A-B)/A]
where:
“X” equals the number of Warrant Shares to be issued to the Holder;
“Y” equals the total number of Warrant Shares with respect to which this Warrant is then being exercised;
“A” equals the Closing Sale Price of the shares of Common Stock (as reported by Bloomberg Financial Markets) as of the Trading Day on the date immediately preceding the Exercise Date; and
“B” equals the Exercise Price then in effect for the applicable Warrant Shares at the time of such exercise.
For purposes of Rule 144 promulgated under the Securities Act, it is intended, understood and acknowledged that the Warrant Shares issued in a “cashless exercise” transaction shall be deemed to have been acquired by the Holder, and the holding period for the Warrant Shares shall be deemed to have commenced, on the date this Warrant was originally issued (provided that the SEC continues to take the position that such treatment is proper at the time of such exercise).
In the event that a registration statement registering the issuance of the Warrant Shares is, for any reason, not effective at the time of exercise of this Warrant, then the Warrant may only be exercised through a cashless exercise, as set forth in this Section 10. If Warrant Shares are issued in such a cashless exercise, the Company acknowledges and agrees that in accordance with Section 3(a)(9) of the Securities Act of 1933, as amended, the Warrant Shares shall take on the registered characteristics of the Warrants being exercised, and the holding period of the Warrants being exercised may be tacked on to the holding period of the Warrant Shares.
11. Limitations on Exercise.
(a) Notwithstanding anything to the contrary contained herein, the Company shall not effect the exercise of any portion of this Warrant, and the Holder shall not have the right to exercise any portion of this Warrant, pursuant to the terms and conditions of this Warrant and any such exercise shall be null and void and treated as if never made, to the extent that immediately prior to or after giving effect to such exercise, the Holder together with the other Attribution Parties collectively would beneficially own in excess of the percentage indicated above (the “Beneficial Ownership Limitation”) of the number of shares of Common Stock outstanding immediately after giving effect to such exercise. For purposes of the foregoing sentence, the aggregate number of shares of Common Stock beneficially owned by the Holder and the other Attribution Parties shall include the number of shares of Common Stock held by the Holder and all other Attribution Parties plus the number of shares of Common Stock issuable upon exercise of this Warrant with respect to which the determination of such sentence is being made, but shall exclude the number of shares of Common Stock which would be issuable upon (A) exercise of the remaining, unexercised portion of this Warrant beneficially owned by the Holder or any of the other Attribution Parties and (B) exercise or conversion of the unexercised or unconverted portion of any other securities of the Company (including, without limitation, any convertible notes or convertible preferred stock or warrants, including the other Warrants) beneficially owned by the Holder or any other Attribution Party subject to a limitation on conversion or exercise analogous to the limitation contained in this Section 11(a). For purposes of this Section 11(a), beneficial ownership shall be calculated in accordance with Section 13(d) of the Exchange Act, it being acknowledged by the Holder that the Company is not representing to the Holder that such calculation is in compliance with Section 13(d) of the Exchange Act and the Holder is solely responsible for any schedules required to be filed in accordance therewith. In addition, a determination as to any group status as contemplated above shall be determined in accordance with Section 13(d) of the Exchange Act and the rules and regulations promulgated thereunder. For purposes of this Warrant, in determining the number of outstanding shares of Common Stock the Holder may acquire upon the exercise of this Warrant without exceeding the Beneficial Ownership Limitation, the Holder may rely on the number of outstanding shares of Common Stock as reflected in (x) the Company’s most recent Annual Report on Form 10-K, Quarterly Report on Form 10-Q and Current Reports on Form 8-K or other public filing with the SEC, as the case may be, (y) a more recent public announcement by the Company or (z) any other written notice by the Company setting forth the number of shares of Common Stock outstanding (the “Reported Outstanding Share Number”). If the Company receives an Exercise Notice from the Holder at a time when the actual number of outstanding shares of Common Stock is less than the Reported Outstanding Share Number, the Company shall (i) notify the Holder in writing of the number of shares of Common Stock then outstanding and, to the extent that such Exercise Notice would otherwise cause the Holder’s beneficial ownership, as determined pursuant to this Section 11(a), to exceed the Beneficial Ownership Limitation, the Holder must notify the Company of a reduced number of Warrant Shares to be purchased pursuant to such Exercise Notice (the number of shares by which such purchase is reduced, the “Reduction Shares”) and (ii) as soon as reasonably practicable, the Company shall return to the Holder any exercise price paid by the Holder for the Reduction Shares. For any reason at any time, upon the written or oral request of the Holder, the Company shall within one (1) business day confirm orally and in writing or by electronic mail to the Holder the number of shares of Common Stock then outstanding. In any case, the number of outstanding shares of Common Stock shall be determined after giving effect to the conversion or exercise of securities of the Company, including this Warrant, by the Holder and any other Attribution Party since the date as of which the Reported Outstanding Share Number was reported. In the event that the issuance of Common Stock to the Holder upon exercise of this Warrant results in the Holder and the other Attribution Parties being deemed to beneficially own, in the aggregate, more than the Beneficial Ownership Limitation of the number of outstanding shares of Common Stock (as determined under Section 13(d) of the Exchange Act), the number of shares so issued by which the Holder’s and the other Attribution Parties’ aggregate beneficial ownership exceeds the Beneficial Ownership Limitation (the “Excess Shares”) shall be deemed null and void and shall be cancelled ab initio, and the Holder shall not have the power to vote or to transfer the Excess Shares. As soon as reasonably practicable after the issuance of the Excess Shares has been deemed null and void, the Company shall return to the Holder the exercise price paid by the Holder for the Excess Shares. Upon delivery of a written notice to the Company, the Holder may from time to time increase or decrease the Beneficial Ownership Limitation to any other percentage (not in excess of 19.99% of the issued and outstanding shares of Common Stock immediately after giving effect to the issuance of the shares of Common Stock issuable upon exercise of this Warrant if exceeding that limit would result in a change of control under Nasdaq Listing Rule 5636(b) or any successor rule) as specified in such notice; provided that (i) any such increase in the Beneficial Ownership Limitation will not be effective until the sixty-first (61st) day after such notice is delivered to the Company and (ii) any such increase or decrease will apply only to the Holder and the other Attribution Parties and not to any other holder of Warrants that is not an Attribution Party of the Holder, and (iii) no such decrease shall affect the validity of any prior exercise of Warrants by Holder or any Attribution Party. For purposes of clarity, the shares of Common Stock issuable pursuant to the terms of this Warrant in excess of the Beneficial Ownership Limitation shall not be deemed to be beneficially owned by the Holder for any purpose including for purposes of Section 13(d) or Rule 16a-1(a)(1) of the Exchange Act. No prior inability to exercise this Warrant pursuant to this paragraph shall have any effect on the applicability of the provisions of this paragraph with respect to any subsequent determination of exercisability. The provisions of this paragraph shall be construed and implemented in a manner otherwise than in strict conformity with the terms of this Section 11(a) to the extent necessary to correct this paragraph or any portion of this paragraph which may be defective or inconsistent with the intended beneficial ownership limitation contained in this Section 11(a) or to make changes or supplements necessary or desirable to properly give effect to such limitation. The limitation contained in this paragraph may not be waived and shall apply to a successor holder of this Warrant.
(b) This Section 11 shall not restrict the number of shares of Common Stock which a Holder may receive or beneficially own in order to determine the amount of securities or other consideration that such Holder may receive in the event of a Fundamental Transaction as contemplated in Section 9(d) of this Warrant.
12. No Fractional Shares. No fractional Warrant Shares will be issued in connection with any exercise of this Warrant. In lieu of any fractional shares that would otherwise be issuable, the number of Warrant Shares to be issued shall be rounded down to the next whole number and the Company shall pay the Holder in cash the fair market value (based on the Closing Sale Price) for any such fractional shares.
13. Notices. Any and all notices or other communications or deliveries hereunder (including, without limitation, any Exercise Notice) shall be in writing and shall be deemed given and effective on the earliest of (i) the date of transmission, if such notice or communication is delivered via facsimile or confirmed e-mail at the facsimile number or e-mail address specified in the books and records of the Transfer Agent prior to 5:30 P.M., New York City time, on a Trading Day, (ii) the next Trading Day after the date of transmission, if such notice or communication is delivered via facsimile or confirmed e-mail at the facsimile number or e-mail address specified in the books and records of the Transfer Agent on a day that is not a Trading Day or later than 5:30 P.M., New York City time, on any Trading Day, (iii) the Trading Day following the date of mailing, if sent by nationally recognized overnight courier service specifying next business day delivery, or (iv) upon actual receipt by the Person to whom such notice is required to be given, if by hand delivery.
14. Warrant Agent. The Company shall initially serve as warrant agent under this Warrant. Upon thirty (30) days’ notice to the Holder, the Company may appoint a new warrant agent. Any corporation into which the Company or any new warrant agent may be merged or any corporation resulting from any consolidation to which the Company or any new warrant agent shall be a party or any corporation to which the Company or any new warrant agent transfers substantially all of its corporate trust or shareholders services business shall be a successor warrant agent under this Warrant without any further act. Any such successor warrant agent shall promptly cause notice of its succession as warrant agent to be mailed (by first class mail, postage prepaid) to the Holder at the Holder’s last address as shown on the Warrant Register.
15. Miscellaneous.
(a) No Rights as a Stockholder. Except as otherwise set forth in this Warrant, the Holder, solely in such Person’s capacity as a holder of this Warrant, shall not be entitled to vote or receive dividends or be deemed the holder of share capital of the Company for any purpose, nor shall anything contained in this Warrant be construed to confer upon the Holder, solely in such Person’s capacity as the Holder of this Warrant, any of the rights of a stockholder of the Company or any right to vote, give or withhold consent to any corporate action (whether any reorganization, issue of stock, reclassification of stock, consolidation, merger, amalgamation, conveyance or otherwise), receive notice of meetings, receive dividends or subscription rights, or otherwise, prior to the issuance to the Holder of the Warrant Shares which such Person is then entitled to receive upon the due exercise of this Warrant. In addition, nothing contained in this Warrant shall be construed as imposing any liabilities on the Holder to purchase any securities (upon exercise of this Warrant or otherwise) or as a stockholder of the Company, whether such liabilities are asserted by the Company or by creditors of the Company.
(b) Authorized Shares.
|
(1) |
Except and to the extent as waived or consented to by the Holder, the Company shall not by any action, including, without limitation, amending its certificate or articles of incorporation or through any reorganization, transfer of assets, consolidation, merger, dissolution, issue or sale of securities or any other voluntary action, avoid or seek to avoid the observance or performance of any of the terms of this Warrant, but will at all times in good faith assist in the carrying out of all such terms and in the taking of all such actions as may be necessary or appropriate to protect the rights of Holder as set forth in this Warrant against impairment. Without limiting the generality of the foregoing, the Company will (a) not increase the par value of any Warrant Shares above the amount payable therefor upon such exercise immediately prior to such increase in par value, (b) take all such action as may be necessary or appropriate in order that the Company may validly and legally issue fully paid and nonassessable Warrant Shares upon the exercise of this Warrant, and (c) use commercially reasonable efforts to obtain all such authorizations, exemptions or consents from any public regulatory body having jurisdiction thereof as may be necessary to enable the Company to perform its obligations under this Warrant. |
|
(2) |
Before taking any action which would result in an adjustment in the number of Warrant Shares for which this Warrant is exercisable or in the Exercise Price, the Company shall obtain all such authorizations or exemptions thereof, or consents thereto, as may be necessary from any public regulatory body or bodies having jurisdiction thereof. |
(c) Successors and Assigns. Subject to compliance with applicable securities laws, this Warrant may be transferred or assigned by the Holder. This Warrant may not be assigned by the Company without the written consent of the Holder except to a successor in the event of a Fundamental Transaction. This Warrant shall be binding on and inure to the benefit of the Company and the Holder and their respective successors and assigns. Subject to the preceding sentence, nothing in this Warrant shall be construed to give to any Person other than the Company and the Holder any legal or equitable right, remedy or cause of action under this Warrant. This Warrant may be amended only in writing signed by the Company and the Holder, or their successors and assigns.
(d) Amendment and Waiver. Except as otherwise provided herein, the provisions of the Warrants may be amended and the Company may take any action herein prohibited, or omit to perform any act herein required to be performed by it, only if the Company has obtained the written consent of the Holder.
(e) Acceptance. Receipt of this Warrant by the Holder shall constitute acceptance of and agreement to all of the terms and conditions contained herein.
(f) Governing Law; Jurisdiction. ALL QUESTIONS CONCERNING THE CONSTRUCTION, VALIDITY, ENFORCEMENT AND INTERPRETATION OF THIS WARRANT SHALL BE GOVERNED BY AND CONSTRUED AND ENFORCED IN ACCORDANCE WITH THE LAWS OF THE STATE OF DELAWARE WITHOUT REGARD TO THE PRINCIPLES OF CONFLICTS OF LAW THEREOF. EACH OF THE COMPANY AND THE HOLDER HEREBY IRREVOCABLY SUBMITS TO THE EXCLUSIVE JURISDICTION OF THE STATE AND FEDERAL COURTS SITTING IN THE STATE OF DELAWARE, FOR THE ADJUDICATION OF ANY DISPUTE HEREUNDER OR IN CONNECTION HEREWITH OR WITH ANY TRANSACTION CONTEMPLATED HEREBY OR DISCUSSED HEREIN (INCLUDING WITH RESPECT TO THE ENFORCEMENT OF ANY OF THE TRANSACTION DOCUMENTS), AND HEREBY IRREVOCABLY WAIVES, AND AGREES NOT TO ASSERT IN ANY SUIT, ACTION OR PROCEEDING, ANY CLAIM THAT IT IS NOT PERSONALLY SUBJECT TO THE JURISDICTION OF ANY SUCH COURT. EACH OF THE COMPANY AND THE HOLDER HEREBY IRREVOCABLY WAIVES PERSONAL SERVICE OF PROCESS AND CONSENTS TO PROCESS BEING SERVED IN ANY SUCH SUIT, ACTION OR PROCEEDING BY MAILING A COPY THEREOF VIA REGISTERED OR CERTIFIED MAIL OR OVERNIGHT DELIVERY (WITH EVIDENCE OF DELIVERY) TO SUCH PERSON AT THE ADDRESS IN EFFECT FOR NOTICES TO IT AND AGREES THAT SUCH SERVICE SHALL CONSTITUTE GOOD AND SUFFICIENT SERVICE OF PROCESS AND NOTICE THEREOF. NOTHING CONTAINED HEREIN SHALL BE DEEMED TO LIMIT IN ANY WAY ANY RIGHT TO SERVE PROCESS IN ANY MANNER PERMITTED BY LAW. EACH OF THE COMPANY AND THE HOLDER HEREBY WAIVES ALL RIGHTS TO A TRIAL BY JURY.
(g) Headings. The headings herein are for convenience only, do not constitute a part of this Warrant and shall not be deemed to limit or affect any of the provisions hereof.
(h) Severability. In case any one or more of the provisions of this Warrant shall be invalid or unenforceable in any respect, the validity and enforceability of the remaining terms and provisions of this Warrant shall not in any way be affected or impaired thereby, and the Company and the Holder will attempt in good faith to agree upon a valid and enforceable provision which shall be a commercially reasonable substitute therefor, and upon so agreeing, shall incorporate such substitute provision in this Warrant.
[remainder of page intentionally left blank]
IN WITNESS WHEREOF, the Company has caused this Pre-Funded Warrant to be duly executed as of the Original Issue Date set out above.
PIERIS PHARMACEUTICALS, INC.
By:
Name:
Title:
SCHEDULE 1
FORM OF EXERCISE NOTICE
(To be executed by the Holder to purchase shares of Common Stock under the Warrant)
Ladies and Gentlemen:
|
(1) |
The undersigned is the Holder of Warrant No. [•] (the “Warrant”) issued by Pieris Pharmaceuticals, Inc., a Nevada corproation. Capitalized terms used herein and not otherwise defined herein have the respective meanings set forth in the Warrant. |
|
(2) |
The undersigned hereby exercises its right to purchase Warrant Shares pursuant to the Warrant. |
|
(3) |
The Holder intends that payment of the Exercise Price shall be made as (check one): |
|
☐ |
Cash Exercise |
|
☐ |
“Cashless Exercise” under Section 10 of the Warrant |
|
(4) |
If the Holder has elected a Cash Exercise, the Holder shall pay the sum of $[•] in immediately available funds to the Company in accordance with the terms of the Warrant. |
|
(5) |
Pursuant to this Exercise Notice, the Company shall deliver to the Holder Warrant Shares determined in accordance with the terms of the Warrant. |
|
(6) |
By its delivery of this Exercise Notice, the undersigned represents and warrants to the Company that in giving effect to the exercise evidenced hereby the Holder will not beneficially own in excess of the number of shares of Common Stock (as determined in accordance with Section 13(d) of the Securities Exchange Act of 1934, as amended) permitted to be owned under Section 11(a) or Section 11(b), as applicable, of the Warrant to which this notice relates. |
Dated:
Name of Holder:
|
By: |
||
|
Name: |
||
|
Title: |
||
(Signature must conform in all respects to name of Holder as specified on the face of the Warrant)
ASSIGNMENT FORM
(To assign the foregoing Warrant, execute this form and supply required information. Do not use this form to purchase shares.)
FOR VALUE RECEIVED, the foregoing Warrant and all rights evidenced thereby are hereby assigned to:
|
Name:
Address:
|
|
|
Phone Number: |
|
|
Email Address: |
|
|
Dated: |
|
|
Holder’s Signature: |
|
|
Holder’s Address: |
Annex L
Execution Version
This Registration Rights Agreement (this “Agreement”) is dated as of July [•], 2024, by and among Pieris Pharmaceuticals, Inc., a Nevada corporation (the “Company”), and the several purchasers signatory hereto (each, including its successors and assigns, a “Purchaser” and collectively, the “Purchasers”).
This Agreement is made pursuant to the Securities Purchase Agreement, dated as of July 23, 2024, between the Company and each Purchaser (the “Purchase Agreement”).
NOW, THEREFORE, IN CONSIDERATION of the mutual covenants contained in this Agreement, and for other good and valuable consideration, the receipt and adequacy of which are hereby acknowledged, the Company and each of the Purchasers agree as follows:
1. Definitions. Capitalized terms used and not otherwise defined herein that are defined in the Purchase Agreement shall have the meanings given such terms in the Purchase Agreement. As used in this Agreement, the following terms shall have the following meanings:
“Advice” has the meaning set forth in Section 6(d).
“Affiliate” when used with respect to any party shall mean any Person who is an “affiliate” of that party within the meaning of Rule 405 promulgated under the Securities Act.
“Agreement” has the meaning set forth in the Preamble.
“Allowed Suspension” has the meaning set forth in Section 6(d).
“Business Day” means any day other than a Saturday, Sunday or other day on which commercial banking institutions in New York, New York, Boston, Massachusetts, Philadelphia, Pennsylvania or Wilmington, Delaware are required or permitted by Law to be closed or other day on which the Delaware Secretary of State is closed.
“Common Stock” means the Company’s common stock, par value $0.001 per share, and stock of any other class of securities into which such securities may hereafter be reclassified or changed.
“Company” has the meaning set forth in the Preamble.
“Effective Date” means the date that the Registration Statement filed pursuant to Section 2(a) is first declared effective by the Commission.
“Effectiveness Deadline” means, with respect to the Initial Registration Statement or the New Registration Statement, the ninetieth (90th) calendar day following the Closing Date (or, in the event the Commission reviews and has written comments to the Initial Registration Statement or the New Registration Statement, the one hundred twentieth (120th) calendar day following the Closing Date); provided, however, that (i) if the Company is notified by the Commission that the Initial Registration Statement or the New Registration Statement will not be reviewed or is no longer subject to further review and comments, the Effectiveness Deadline as to such Registration Statement shall be the fifth (5th) Trading Day following the date on which the Company is so notified if such date precedes the dates otherwise required above; and (ii) if the Effectiveness Deadline falls on a Saturday, Sunday or other day that the Commission is closed for business, the Effectiveness Deadline shall be extended to the next Business Day on which the Commission is open for business.
“Effectiveness Period” has the meaning set forth in Section 2(b).
“Exchange Act” means the Securities Exchange Act of 1934, as amended, and the rules and regulations promulgated thereunder.
”Filing Deadline” means, with respect to the Initial Registration Statement required to be filed pursuant to Section 2(a), the thirtieth (30th) calendar day following the Closing Date, provided, however, that if the Filing Deadline falls on a Saturday, Sunday or other day that the Commission is closed for business, the Filing Deadline shall be extended to the next Business Day on which the Commission is open for business.
“FINRA” has the meaning set forth in Section 3(i).
“Holder” or “Holders” means the holder or holders, as the case may be, from time to time of Registrable Securities.
“Indemnified Party” has the meaning set forth in Section 5(c).
“Indemnifying Party” has the meaning set forth in Section 5(c).
“Initial Registration Statement” has the meaning set forth in Section 2(a).
“Losses” has the meaning set forth in Section 5(a).
“New Registration Statement” has the meaning set forth in Section 2(a).
“Person” means an individual or a corporation, partnership, trust, incorporated or unincorporated association, joint venture, limited liability company, joint stock company, government (or an agency or subdivision thereof) or other entity of any kind.
“Principal Market” means the Trading Market on which the Common Stock are primarily listed on and quoted for trading.
“Proceeding” means an action, claim, suit, investigation or proceeding (including, without limitation, an investigation or partial proceeding, such as a deposition), whether commenced or threatened.
“Prospectus” means the prospectus included in a Registration Statement (including, without limitation, a prospectus that includes any information previously omitted from a prospectus filed as part of an effective registration statement in reliance upon Rule 430B promulgated under the Securities Act), as amended or supplemented by any prospectus supplement, with respect to the terms of the offering of any portion of the Registrable Securities covered by a Registration Statement, and all other amendments and supplements to the Prospectus, including post-effective amendments, and all material incorporated by reference or deemed to be incorporated by reference in such Prospectus.
“Purchase Agreement” has the meaning set forth in the Recitals.
“Purchaser” or “Purchasers” has the meaning set forth in the Preamble.
“Registrable Securities” means all of (i) the Shares, (ii) all Warrant Shares then issued or issuable upon exercise of the Pre-Funded Warrants (assuming on such date the Pre-Funded Warrants are exercised in full without regard to any exercise limitations therein) , and (ii) any securities issued or issuable upon any stock split, dividend or other distribution, recapitalization or similar event with respect to the foregoing, provided that the Holder has completed and delivered to the Company a Selling Stockholder Questionnaire; and provided, further, that, with respect to a particular Holder, such Holder’s Registrable Securities shall cease to be Registrable Securities upon the earlier to occur of the following: (A) a sale pursuant to a Registration Statement or Rule 144 under the Securities Act (in which case, only such security sold by the Holder shall cease to be a Registrable Security); and (B) such Registrable Securities become eligible for resale by the Holder under Rule 144 without the requirement for the Company to be in compliance with the current public information required thereunder and without volume or manner-of-sale restrictions (and in the case of the Pre-Funded Warrant Shares, assuming the cashless exercise of the Pre-Funded Warrants), pursuant to a written opinion letter of counsel for the Company to such effect, addressed, delivered and reasonably acceptable to the Transfer Agent.
“Registration Statements” means any one or more registration statements of the Company filed under the Securities Act that cover the resale of any of the Registrable Securities pursuant to the provisions of this Agreement (including without limitation the Initial Registration Statement, any New Registration Statement and any Remainder Registration Statements), amendments and supplements to such Registration Statements, including post-effective amendments, all exhibits and all material incorporated by reference or deemed to be incorporated by reference in such Registration Statements.
“Remainder Registration Statement” has the meaning set forth in Section 2(a).
“Rule 144” means Rule 144 promulgated by the Commission pursuant to the Securities Act, as such Rule may be amended from time to time, or any similar rule or regulation hereafter adopted by the Commission having substantially the same effect as such Rule.
“Rule 172” means Rule 172 promulgated by the Commission pursuant to the Securities Act, as such Rule may be amended from time to time, or any similar rule or regulation hereafter adopted by the Commission having substantially the same effect as such Rule.
“Rule 415” means Rule 415 promulgated by the Commission pursuant to the Securities Act, as such Rule may be amended from time to time, or any similar rule or regulation hereafter adopted by the Commission having substantially the same effect as such Rule.
“Rule 424” means Rule 424 promulgated by the Commission pursuant to the Securities Act, as such Rule may be amended from time to time, or any similar rule or regulation hereafter adopted by the Commission having substantially the same effect as such Rule.
“Rule 461” means Rule 461 promulgated by the Commission pursuant to the Securities Act, as such Rule may be amended from time to time, or any similar rule or regulation hereafter adopted by the Commission having substantially the same effect as such Rule.
“SEC Guidance” means (i) any publicly-available written or oral guidance, comments, requirements or requests of the Commission staff; provided, that any such oral guidance, comments, requirements or requests are reduced to writing by the Commission and (ii) the Securities Act.
“Securities Act” means the Securities Act of 1933, as amended, and the rules and regulations promulgated thereunder.
“Selling Stockholder Questionnaire” means a questionnaire in the form attached as Annex B hereto, or such other form of questionnaire or information provided to the Company in connection with the preparation of a Registration Statement hereunder.
“Shares” means the shares of Common Stock issued to the Purchasers pursuant to the Purchase Agreement.
“Trading Day” means a day on which the Principal Market is open for business.
“Trading Market” means any of the following markets or exchanges on which the Common Stock is listed or quoted for trading on the date in question: the NYSE American, the Nasdaq Capital Market, the Nasdaq Global Market, the Nasdaq Global Select Market, or the New York Stock Exchange (or any successors to any of the foregoing).
|
2. |
Registration. |
(a) On or prior to the Filing Deadline, the Company shall prepare and file with the Commission a Registration Statement covering the resale of all of the Registrable Securities not then registered on an existing and effective Registration Statement for an offering to be made on a continuous basis pursuant to Rule 415 or, if Rule 415 is not available for offers and sales of the Registrable Securities, by such other means of distribution of Registrable Securities as the Holders may reasonably specify (the “Initial Registration Statement”). The Initial Registration Statement shall be on Form S-1 subject to the provisions of Section 2(a) and shall contain (except if otherwise required pursuant to written comments received from the Commission upon a review of such Registration Statement) the “Plan of Distribution” section substantially in the form attached hereto as Annex A (which may be modified to respond to comments, if any, provided by the Commission). Notwithstanding the registration obligations set forth in this Section 2, in the event the Commission informs the Company that all of the Registrable Securities cannot, as a result of the application of Rule 415, be registered for resale as a secondary offering on a single registration statement, the Company agrees to promptly (i) inform each of the Holders thereof and use its commercially reasonable efforts to file amendments to the Initial Registration Statement as required by the Commission and/or (ii) withdraw the Initial Registration Statement and file a new registration statement (a “New Registration Statement”), in either case covering the maximum number of Registrable Securities permitted to be registered by the Commission, on Form S-1; provided, however, that prior to filing such amendment or New Registration Statement, the Company shall be obligated to use its commercially reasonable efforts to advocate with the Commission for the registration of all of the Registrable Securities in accordance with the SEC Guidance, including without limitation, the Securities Act Rules Compliance and Disclosure Interpretations Question 612.09. Notwithstanding any other provision of this Agreement, if the Commission or any SEC Guidance sets forth a limitation of the number of Registrable Securities permitted to be registered on a particular Registration Statement as a secondary offering (and notwithstanding that the Company used diligent efforts to advocate with the Commission for the registration of all or a greater number of Registrable Securities), unless otherwise directed in writing by a Holder as to its Registrable Securities, the number of Registrable Securities to be registered on such Registration Statement will first be reduced by Registrable Securities not acquired pursuant to the Purchase Agreement (whether pursuant to registration rights or otherwise), and second by Registrable Securities represented by Registrable Securities applied to the Holders on a pro rata basis based on the total number of Registrable Securities held by such Holders, subject to a determination by the Commission that certain Holders must be reduced first based on the number of Registrable Securities held by such Holders. In the event of a cutback hereunder, the Company shall give the Holder at least one (1) Trading Day prior notice along with the calculations as to such Holder’s allotment. In the event the Company amends the Initial Registration Statement or files a New Registration Statement, as the case may be, in accordance with the foregoing, the Company will use its commercially reasonable efforts to file with the Commission, as promptly as allowed by Commission or SEC Guidance provided to the Company or to registrants of securities in general, one or more registration statements on Form S-1 or such other form available to register for resale those Registrable Securities that were not registered for resale on the Initial Registration Statement, as amended, or the New Registration Statement (the “Remainder Registration Statements”). No Holder shall be named as an “underwriter” in any Registration Statement without such Holder’s prior written consent.
(b) The Company shall use its commercially reasonable efforts to cause each Registration Statement to be declared effective by the Commission as soon as practicable and, with respect to the Initial Registration Statement or the New Registration Statement, as applicable, no later than the Effectiveness Deadline (including filing with the Commission a request for acceleration of effectiveness in accordance with Rule 461 promulgated under the Securities Act), and shall use its commercially reasonable efforts to keep each Registration Statement continuously effective under the Securities Act until the earlier of (i) such time as all of the Registrable Securities covered by such Registration Statement have been publicly sold by the Holders; or (ii) the date that all Registrable Securities covered by such Registration Statement may be sold by non-affiliates without volume or manner-of-sale restrictions pursuant to Rule 144, without the requirement for the Company to be in compliance with the current public information requirement under Rule 144, in each case, including pursuant to a cashless exercise of the Pre-Funded Warrants (the “Effectiveness Period”). The Company shall request effectiveness of a Registration Statement as of 4:00 P.M. New York City time on a Trading Day. The Company shall promptly notify the Holders via e-mail of the effectiveness of a Registration Statement or any post-effective amendment thereto on the same Trading Day that the Company telephonically confirms effectiveness with the Commission, which date of confirmation shall initially be the date requested for effectiveness of such Registration Statement. The Company shall, by 9:30 A.M. New York City time on the first Trading Day after the Effective date, file a final Prospectus with the Commission, as required by Rule 424(b) and shall provide the Purchasers with copies of the final Prospectus to be used in connection with the sale or other disposition of the securities covered thereby. The Company shall promptly inform each Holder in writing if, at any time during the Effectiveness Period, the Company does not satisfy the conditions specified in Rule 172 and, as a result thereof, the Holder is required to deliver a Prospectus in connection with any disposition of Registrable Securities.
(c) Each Holder of Registrable Securities to be sold agrees to furnish to the Company a completed Selling Stockholder Questionnaire not more than five (5) Trading Days following the date of this Agreement. At least 10 Trading Days prior to the first anticipated filing date of a Registration Statement for any registration under this Agreement, the Company will notify each Holder of the information the Company requires from that Holder for inclusion in the Registration Statement other than the information contained in the Selling Stockholder Questionnaire, if any, which shall be completed and delivered to the Company promptly upon request and, in any event, within three (3) Trading Days prior to the applicable anticipated filing date. Each Holder further agrees that it shall not be entitled to be named as a selling securityholder in the Registration Statement or use the Prospectus for offers and resales of Registrable Securities at any time, unless such Holder has provided such information to the Company and responded to any reasonable requests for further information as described in the previous sentence. Each Holder acknowledges and agrees that the information in the Selling Stockholder Questionnaire or request for further information as described in this Section 2(c) will be used by the Company in the preparation of the Registration Statement and hereby consents to the inclusion of such information in the Registration Statement (subject to such Holder’s right to timely review the Registration Statement as set forth herein).
(d) The Company undertakes to register the Registrable Securities on Form S-3 promptly after such form is available, provided that the Company shall maintain the effectiveness of the Registration Statement then in effect until such time as a Registration Statement on Form S-3 covering the Registrable Securities has been declared effective by the Commission.
(e) (i) If a Registration Statement covering the Registrable Securities is not filed with the Commission on or prior to the Filing Deadline (a “Registration Failure”), then, in addition to any other rights the Purchasers may have hereunder or under applicable law, the Company will make pro rata payments to each Purchaser of then outstanding Registrable Securities, as liquidated damages and not as a penalty (the “Registration Liquidated Damages”), in an amount equal to one percent (1.0%) of the aggregate amount invested by such Purchaser for the Registrable Securities then held by such Purchaser for the initial day of a Registration Failure and for each thirty (30) day period (or pro rata portion thereof with respect to a final period, if any) thereafter until the Registration Failure is cured. The Registration Liquidated Damages shall be paid monthly within ten (10) Business Days of the date of such Registration Failure and the end of each subsequent thirty (30)-day period (or portion thereof with respect to a final period, if any) thereafter until the Registration Failure is cured. Such payments shall be made in cash to each Purchaser then holding Registrable Securities. Interest shall accrue at the rate of one percent (1%) per month on any such liquidated damages payments that shall not be paid by the applicable payment date until such amount is paid in full.
(i) If (A) a Registration Statement covering the Registrable Securities is not declared effective by the Commission by the Effectiveness Deadline or (B) after a Registration Statement has been declared effective by the Commission or otherwise becomes effective, sales cannot be made pursuant to such Registration Statement for any reason (including, without limitation, by reason of a stop order or the Company’s failure to update such Registration Statement) (each of (A) and (B), a “Maintenance Failure”), then the Company will make pro rata payments to each Purchaser then holding Registrable Securities, as liquidated damages and not as a penalty (the “Effectiveness Liquidated Damages” and together with the Registration Liquidated Damages, the “Liquidated Damages”), in an amount equal to one percent (1.0%) of the aggregate amount invested by such Purchaser for the Registrable Securities then held by such Purchaser for the initial day of a Maintenance Failure and for each thirty (30)-day period (pro rata for any portion thereof) thereafter until the Maintenance Failure is cured. The Effectiveness Liquidated Damages shall be paid monthly within ten (10) Business Days of the end of the date of such Maintenance Failure and each subsequent thirty (30)-day period (pro rata for any portion thereof). Such payments shall be made to each Purchaser then holding Registrable Securities in cash. Interest shall accrue at the rate of one percent (1.0%) per month on any such liquidated damages payments that shall not be paid by the applicable payment date until such amount is paid in full.
(ii) Notwithstanding the foregoing, (A) no Liquidated Damages shall be payable with respect to any period after the expiration of the Effectiveness Period (it being understood that this sentence shall not relieve the Company of any Liquidated Damages accruing prior to the expiration of the Effectiveness Period), (B) in no event shall the aggregate amount of Liquidated Damages payable to a Purchaser exceed, in the aggregate, five percent (5.0%) of the aggregate purchase price paid by such Purchaser pursuant to the Purchase Agreement, (C) no Liquidated Damages shall accrue or be payable with respect to any reduction in the number of Registrable Securities to be included in a Registration Statement due to the application of Rule 415 as set forth in Section 2(a) and (D) no Liquidated Damages shall accrue or be payable with respect to any Allowed Suspension or a suspension as described in the last sentence of Section 3(h).
|
3. |
Registration Procedures. |
In connection with the Company’s registration obligations hereunder, the Company shall:
(a) Not less than five (5) Trading Days prior to the filing of each Registration Statement and not less than two (2) Trading Days prior to the filing of any related Prospectus or any amendment or supplement thereto (except for Annual Reports on Form 10-K, and Quarterly Reports on Form 10-Q and Current Reports on Form 8-K and any similar or successor reports), (i) furnish to each Holder copies of such Registration Statement, Prospectus or amendment or supplement thereto, as proposed to be filed, which documents will be subject to the review of such Holder (it being acknowledged and agreed that if a Holder does not object to or comment on the aforementioned documents within such five (5) Trading Day or two (2) Trading Day period, as the case may be, then the Holder shall be deemed to have consented to and approved the use of such documents) and (ii) to the extent that a Holder is identified in the Registration Statement as an “underwriter” (as defined in the Securities Act), use commercially reasonable efforts to cause its officers and directors, counsel and independent registered public accountants to respond to such inquiries as shall be necessary, in the reasonable opinion of respective counsel to each Holder, to conduct a reasonable investigation within the meaning of the Securities Act. The Company shall not file any Registration Statement or amendment or supplement thereto in a form to which a Holder reasonably objects in good faith, provided that, such Holder notifies the Company of such objection in writing within the five (5) Trading Day or two (2) Trading Day periods described above, as applicable.
(b) (i) Prepare and file with the Commission such amendments (including post-effective amendments) and supplements, to each Registration Statement and the Prospectus used in connection therewith as may be necessary to keep such Registration Statement continuously effective as to the applicable Registrable Securities for its Effectiveness Period and prepare and file with the Commission such additional Registration Statements in order to register for resale under the Securities Act all of the Registrable Securities; (ii) cause the related Prospectus to be amended or supplemented by any required Prospectus supplement (subject to the terms of this Agreement), and, as so supplemented or amended, to be filed pursuant to Rule 424; (iii) respond as promptly as reasonably practicable to any comments received from the Commission with respect to each Registration Statement or any amendment thereto and, as promptly as reasonably possible, provide the Holders true and complete copies of all correspondence from and to the Commission relating to such Registration Statement that pertains to the Holders as “Selling Stockholders” but not any comments that would result in the disclosure to the Holders of material and non-public information concerning the Company (unless such Holder consents to receive such material and non-public information); and (iv) comply with the provisions of the Securities Act and the Exchange Act with respect to the disposition of all Registrable Securities covered by a Registration Statement until such time as all of such Registrable Securities shall have been disposed of (subject to the terms of this Agreement) in accordance with the intended methods of disposition by the Holders thereof as set forth in such Registration Statement as so amended or in such Prospectus as so supplemented; provided, however, that each Purchaser shall be responsible for the delivery of the Prospectus to the Persons to whom such Purchaser sells any of the Registrable Securities (including in accordance with Rule 172 under the Securities Act) to the extent required under the Securities Act, and each Purchaser agrees to dispose of Registrable Securities in compliance with the “Plan of Distribution” described in the Registration Statement and otherwise in compliance with applicable federal and state securities laws. In the case of amendments and supplements to a Registration Statement which are required to be filed pursuant to this Agreement (including pursuant to this Section 3(b)) by reason of the Company filing a report on Form 10-K, Form 10-Q or Form 8-K or any analogous report under the Exchange Act, the Company shall have incorporated such report by reference into such Registration Statement, if applicable, or shall file such amendments or supplements with the Commission on the same day on which the Exchange Act report which created the requirement for the Company to amend or supplement such Registration Statement was filed.
(c) Notify the Holders of Registrable Securities to be sold (which notice shall, if given pursuant to clauses (iii) through (vi) hereof, be accompanied by an instruction to suspend the use of the Prospectus until the requisite changes have been made, provided that the Company shall omit any material, non-public information relating to the Company and/or any of its subsidiaries) as promptly as reasonably practicable (and, in the case of (i) (A) below, not less than one (1) Trading Day prior to such filing) and (if requested by any such Person) confirm such notice in writing no later than one (1) Trading Day following the day: (i)(A) when a Prospectus or any Prospectus supplement or post-effective amendment to a Registration Statement is proposed to be filed; (B) when the Commission notifies the Company whether there will be a “review” of such Registration Statement and whenever the Commission comments in writing on any Registration Statement (in which case the Company shall provide to each of the Holders true and complete copies of all comments that pertain to the Holders as a “Selling Stockholder” or to the “Plan of Distribution” and all written responses thereto, but not information that the Company believes would constitute material and non-public information); and (C) with respect to each Registration Statement or any post-effective amendment, when the same has become effective; (ii) of any request by the Commission or any other Federal or state governmental authority for amendments or supplements to a Registration Statement or Prospectus or for additional information that pertains to the Holders as “Selling Stockholders” or the “Plan of Distribution”; (iii) of the issuance by the Commission or any other federal or state governmental authority of any stop order suspending the effectiveness of a Registration Statement covering any or all of the Registrable Securities or the initiation of any Proceedings for that purpose; (iv) of the receipt by the Company of any notification with respect to the suspension of the qualification or exemption from qualification of any of the Registrable Securities for sale in any jurisdiction, or the initiation or threatening of any Proceeding for such purpose; (v) of the occurrence of any event or passage of time that makes the financial statements included or incorporated by reference in a Registration Statement ineligible for inclusion or incorporation by reference therein or any statement made in such Registration Statement or Prospectus or any document incorporated or deemed to be incorporated therein by reference untrue in any material respect or that requires any revisions to such Registration Statement, Prospectus or other documents so that, in the case of such Registration Statement or the Prospectus, as the case may be, it will not contain any untrue statement of a material fact or omit to state any material fact required to be stated therein or necessary to make the statements therein (in the case of any Prospectus, form of prospectus or supplement thereto, in light of the circumstances under which they were made), not misleading and (vi) of the occurrence or existence of any pending corporate development with respect to the Company that, upon the advice of legal counsel, the Company’s board of directors reasonably believes may be material and that would require additional disclosure by the Company in the Registration Statement of such material information that the Company has a bona fide business purpose for keeping confidential and the non-disclosure of which in the Registration Statement would be expected, in the reasonable determination of the Company’s board of directors, upon the advice of legal counsel, to cause the Registration Statement to fail to comply with applicable disclosure requirements, provided that, any and all such information shall remain confidential to each Holder until such information otherwise becomes public, unless disclosure by a Holder is required by law; and provided, further, that notwithstanding each Holder’s agreement to keep such information confidential, each such Holder makes no acknowledgement that any such information is material, non-public information.
(d) Use commercially reasonable efforts to avoid the issuance of, or, if issued, obtain the withdrawal of (i) any order stopping or suspending the effectiveness of a Registration Statement, or (ii) any suspension of the qualification (or exemption from qualification) of any of the Registrable Securities for sale in any jurisdiction, as soon as practicable.
(e) If requested by a Holder, furnish to such Holder, without charge, at least one conformed copy of each Registration Statement and each amendment thereto, including financial statements and schedules, all documents incorporated or deemed to be incorporated therein by reference to the extent requested by such Person, and all exhibits to the extent requested by such Person (including those previously furnished or incorporated by reference) promptly after the filing of such documents with the Commission; provided, that the Company shall have no obligation to provide any document pursuant to this clause that is available on the Commission’s EDGAR system.
(f) Prior to any resale of Registrable Securities by a Holder, use its commercially reasonable efforts to register or qualify or cooperate with the selling Holders in connection with the registration or qualification (or exemption from the registration or qualification) of the resale of such Registrable Securities (or, in the case of qualification, of such Registrable Securities for the resale) by the Holder under the securities or blue sky laws of such jurisdictions within the United States as any Holder reasonably requests in writing, to keep each registration or qualification (or exemption therefrom) effective during the Effectiveness Period and to do any and all other acts or things reasonably necessary to enable the disposition in such jurisdictions of the Registrable Securities covered by each Registration Statement; provided, that the Company shall not be required to qualify generally to do business in any jurisdiction where it is not then so qualified, subject the Company to any material tax in any such jurisdiction where it is not then so subject or file a general consent to service of process in any such jurisdiction.
(g) Cooperate with such Holder to facilitate the timely preparation and delivery of certificates or book entry statements, as applicable, representing Registrable Securities to be delivered to a transferee pursuant to the Registration Statement, which certificates or statements shall be free, to the extent permitted by the Purchase Agreement and under law, of all restrictive legends, and to enable such Registrable Securities to be in such denominations and registered in such names as any such Holders may reasonably request.
(h) Following the occurrence of any event contemplated by Section 3(c), as promptly as reasonably practicable (taking into account the Company’s good faith assessment of any adverse consequences to the Company and its stockholders of the premature disclosure of such event), prepare a supplement or amendment, including a post-effective amendment, to the affected Registration Statements or a supplement to the related Prospectus or any document incorporated or deemed to be incorporated therein by reference, and file any other required document so that, as thereafter delivered, no Registration Statement nor any Prospectus will contain an untrue statement of a material fact or omit to state a material fact required to be stated therein or necessary to make the statements therein (in the case of any Prospectus, form of prospectus or supplement thereto, in light of the circumstances under which they were made), not misleading. If the Company notifies the Holders in accordance with clauses (iii) through (vi) of Section 3(c) above to suspend the use of any Prospectus until the requisite changes to such Prospectus have been made, then the Holders shall suspend use of such Prospectus. The Company will use its commercially reasonable efforts to ensure that the use of the Prospectus may be resumed as promptly as is practicable. The Company shall be entitled to exercise its right under this Section 3(h) to suspend the availability of a Registration Statement and Prospectus in accordance with the time periods set forth in Section 6(d), which may be extended only in accordance with Section 3(h). For the avoidance of doubt, the Company’s rights under this Section 3(h) shall include suspensions of availability arising from the filing of a post-effective amendment to a Registration Statement to update the Prospectus therein to include the information contained in the Company’s Annual Report on Form 10-K, which suspensions may extend for the amount of time reasonably required to respond to any comments of the staff of the Commission on such amendment and which, for the avoidance of doubt, shall be in accordance with the time periods set forth in Section 6(d), which may be extended only in accordance with Section 6(f).
(i) The Company may require each selling Holder to furnish to the Company a certified statement as to (i) the number of shares of Common Stock beneficially owned by such Holder and any Affiliate thereof, (ii) any Financial Industry Regulatory Authority (“FINRA”) affiliations, (iii) any natural persons who have the power to vote or dispose of the Common Stock and (iv) any other information as may be requested by the Commission, FINRA or any state securities commission.
(j) The Company shall cooperate with any registered broker through which a Holder proposes to resell its Registrable Securities in effecting a filing with FINRA pursuant to FINRA Rule 5110 as requested by any such Holder and the Company shall pay the filing fee required for the first such filing within two (2) Business Days of the request therefor.
(k) If at any time the Commission takes the position that the offering of some or all of the Registrable Securities in a Registration Statement is not eligible to be made on a delayed or continuous basis under the provisions of Rule 415 under the Securities Act or requires any Holder to be named as an “underwriter,” the Company shall use commercially reasonable efforts to persuade the Commission that the offering contemplated by such Registration Statement is a valid secondary offering and not an offering “by or on behalf of the issuer” as defined in Rule 415 and that none of the Holders is an “underwriter”.
4. Registration Expenses. All fees and expenses incident to the Company’s performance of or compliance with its obligations under this Agreement (excluding any underwriting discounts and selling commissions and all legal fees and expenses of legal counsel for any Holder) shall be borne by the Company whether or not any Registrable Securities are sold pursuant to a Registration Statement. The fees and expenses referred to in the foregoing sentence shall include, without limitation, (i) all registration and filing fees (including, without limitation, fees and expenses of the Company’s counsel and independent registered public accountants (A) with respect to filings required to be made with any Trading Market on which the Common Stock are then listed for trading, (B) with respect to compliance with applicable state securities or blue sky laws (including, without limitation, fees and disbursements of counsel for the Company in connection with blue sky qualifications or exemptions of the Registrable Securities and determination of the eligibility of the Registrable Securities for investment under the laws of such jurisdictions as requested by the Holders) and (C) if not previously paid by the Company in connection with Section 3(j) above, with respect to any filing that may be required to be made by any broker through which a Holder intends to make sales of Registrable Securities with FINRA pursuant to the FINRA Rule 5110, so long as the broker is receiving no more than a customary brokerage commission in connection with such sale), (ii) printing expenses (including, without limitation, expenses of printing certificates for Registrable Securities and of printing prospectuses if the printing of prospectuses is reasonably requested by the Holders of a majority of the Registrable Securities included in the Registration Statement), (iii) messenger, telephone and delivery expenses, (iv) fees, expenses and disbursements of counsel for the Company, (v) Securities Act liability insurance, if the Company so desires such insurance, and (vi) fees and expenses of all other Persons retained by the Company in connection with the registrations and consummation of the transactions contemplated by this Agreement. In addition, the Company shall be responsible for all of its internal expenses incurred in connection with the consummation of the transactions contemplated by this Agreement (including, without limitation, all salaries and expenses of its officers and employees performing legal or accounting duties), the expense of any annual audit and the fees and expenses incurred in connection with the listing of the Registrable Securities on any securities exchange as required hereunder. In no event shall the Company be responsible for any underwriting, broker or similar fees or commissions of any Holder or, except to the extent provided for in the Transaction Documents, any legal fees or other costs of the Holders.
|
5. |
Indemnification. |
(a) Indemnification by the Company. The Company shall, notwithstanding any termination of this Agreement, indemnify, defend and hold harmless each Holder and each of their respective officers, directors, agents, partners, members, managers, stockholders, Affiliates, investment advisers and employees of each of them, each Person who controls any such Holder (within the meaning of Section 15 of the Securities Act or Section 20 of the Exchange Act) and the officers, directors, partners, members, managers, stockholders, agents, investment advisers and employees of each such controlling Person, to the fullest extent permitted by applicable law, from and against any and all losses, claims, damages, liabilities, costs (including, without limitation, reasonable costs of preparation and investigation and reasonable attorneys’ fees), expenses and disbursements (collectively, “Losses”), as incurred, that arise out of or are based upon (i) any untrue or alleged untrue statement of a material fact contained in any Registration Statement, any Prospectus or any form of prospectus or in any amendment or supplement thereto or in any preliminary prospectus, or arising out of or relating to any omission or alleged omission to state a material fact required to be stated therein or necessary to make the statements therein (in the case of any Prospectus or form of prospectus or supplement thereto, in light of the circumstances under which they were made) not misleading, or (ii) any violation or alleged violation by the Company or its agents of the Securities Act, the Exchange Act or any state securities law or any rule or regulation thereunder, in connection with the performance of its obligations under this Agreement or any action or inaction required of the Company in connection with any registration, except to the extent, but only to the extent, that (A) such untrue statements, alleged untrue statements, omissions or alleged omissions are based solely upon information regarding such Holder furnished in writing to the Company by such Holder expressly for use therein, or to the extent that such information relates to such Holder or such Holder’s proposed method of distribution of Registrable Securities and was reviewed and approved in writing by such Holder expressly for use in the Registration Statement, such Prospectus or such form of Prospectus or in any amendment or supplement thereto (it being understood that each Holder has approved Annex A hereto for this purpose), (B) in the case of an occurrence of an event of the type specified in Section 3(c)(iii)-(vi), related to the use by a Holder of an outdated or defective Prospectus after the Company has notified such Holder in writing that the Prospectus is outdated or defective and prior to the receipt by such Holder of the Advice contemplated and defined in Section 6(d) below, to the extent that following the receipt of the Advice the misstatement or omission giving rise to such Loss would have been corrected or (C) to the extent that any such Losses arise out of the Purchaser’s (or any other indemnified Person’s) failure to send or give a copy of the Prospectus or supplement (as then amended or supplemented), if required, pursuant to Rule 172 under the Securities Act (or any successor rule) to the Persons asserting an untrue statement or alleged untrue statement or alleged untrue statement or omission or alleged omission at or prior to the written confirmation of the sale of Registrable Securities to such Person if such statement or omission was corrected in such Prospectus or supplement. The Company shall notify the Holders promptly of the institution, threat or assertion of any Proceeding arising from or in connection with the transactions contemplated by this Agreement of which the Company is aware. Such indemnity shall remain in full force and effect regardless of any investigation made by or on behalf of an Indemnified Party (as defined in Section 5(c) and shall survive the transfer of the Registrable Securities by the Holders.
(b) Indemnification by Holders. Each Holder shall, severally and not jointly, indemnify and hold harmless the Company, its directors, officers, agents and employees, each Person who controls the Company (within the meaning of Section 15 of the Securities Act and Section 20 of the Exchange Act), and the directors, officers, agents or employees of such controlling Persons, to the fullest extent permitted by applicable law, from and against all Losses, as incurred, arising out of or are based solely upon any untrue or alleged untrue statement of a material fact contained in any Registration Statement, any Prospectus, or any form of prospectus, or in any amendment or supplement thereto or in any preliminary prospectus, or arising out of or relating to any omission or alleged omission of a material fact required to be stated therein or necessary to make the statements therein (in the case of any Prospectus, or any form of prospectus or supplement thereto, in light of the circumstances under which they were made) not misleading (i) to the extent that such untrue statements or omissions are based solely upon information regarding such Holder furnished in writing to the Company by such Holder expressly for use therein or (ii) to the extent that such information relates to such Holder or such Holder’s proposed method of distribution of Registrable Securities and was reviewed and approved in writing by such Holder expressly for use in a Registration Statement (it being understood that the Holder has approved Annex A hereto for this purpose), such Prospectus or such form of Prospectus or in any amendment or supplement thereto or (iii) in the case of an occurrence of an event of the type specified in Section 3(c)(iii)-(vi), to the extent related to the use by such Holder of an outdated or defective Prospectus after the Company has notified such Holder in writing that the Prospectus is outdated or defective and prior to the receipt by such Holder of the Advice contemplated in Section 6(d). In no event shall the liability of any selling Holder hereunder be greater in amount than the dollar amount of the net proceeds received by such Holder upon the sale of the Registrable Securities giving rise to such indemnification obligation.
(c) Conduct of Indemnification Proceedings. If any Proceeding shall be brought or asserted against any Person entitled to indemnity hereunder (an “Indemnified Party”), such Indemnified Party shall promptly notify the Person from whom indemnity is sought (the “Indemnifying Party”) in writing, and the Indemnifying Party shall have the right to assume the defense thereof, including the employment of counsel reasonably satisfactory to the Indemnified Party and the payment of all reasonable fees, expenses and disbursements incurred in connection with defense thereof; provided, that the failure of any Indemnified Party to give such notice shall not relieve the Indemnifying Party of its obligations or liabilities pursuant to this Agreement, except (and only) to the extent that it shall be finally determined by a court of competent jurisdiction (which determination is not subject to appeal or further review) that such failure shall have materially and adversely prejudiced the Indemnifying Party.
An Indemnified Party shall have the right to employ separate counsel in any such Proceeding and to participate in the defense thereof, but the fees, expenses and disbursements of such counsel shall be at the expense of such Indemnified Party or Indemnified Parties unless: (1) the Indemnifying Party has agreed in writing to pay such fees, expenses and amounts; (2) the Indemnifying Party shall have failed promptly to assume the defense of such Proceeding and to employ counsel reasonably satisfactory to such Indemnified Party in any such Proceeding; or (3) the named parties to any such Proceeding (including any impleaded parties) include both such Indemnified Party and the Indemnifying Party, and such Indemnified Party shall have been advised by counsel that a conflict of interest exists if the same counsel were to represent such Indemnified Party and the Indemnifying Party (in which case, if such Indemnified Party notifies the Indemnifying Party in writing that it elects to employ separate counsel at the expense of the Indemnifying Party, the Indemnifying Party shall not have the right to assume the defense thereof and such counsel shall be at the expense of the Indemnifying Party); provided, that the Indemnifying Party shall not be liable for the fees, expenses and disbursements of more than one separate firm of attorneys at any time for all Indemnified Parties. The Indemnifying Party shall not be liable for any settlement of any such Proceeding effected without its written consent, which consent shall not be unreasonably withheld, delayed or conditioned. No Indemnifying Party shall, without the prior written consent of the Indemnified Party, effect any settlement of any pending Proceeding in respect of which any Indemnified Party is a party, unless such settlement includes an unconditional release of such Indemnified Party from all liability on claims that are the subject matter of such Proceeding.
Subject to the terms of this Agreement, all fees, expenses and disbursements of the Indemnified Party (including reasonable fees, expenses and disbursements to the extent incurred in connection with investigating or preparing to defend such Proceeding in a manner not inconsistent with this Section 5) shall be paid to the Indemnified Party, as incurred, within 20 Trading Days of written notice thereof to the Indemnifying Party; provided, that the Indemnified Party shall promptly reimburse the Indemnifying Party for that portion of such fees, expenses and disbursements applicable to such actions for which such Indemnified Party is finally judicially determined to not be entitled to indemnification hereunder. The failure to deliver written notice to the Indemnifying Party within a reasonable time of the commencement of any such action shall not relieve such Indemnifying Party of any liability to the Indemnified Party under this Section 5, except to the extent that the Indemnifying Party is materially and adversely prejudiced in its ability to defend such action.
(d) Contribution. If a claim for indemnification under Section 5(a) or 5(b) is unavailable to an Indemnified Party or insufficient to hold an Indemnified Party harmless for any Losses, then each Indemnifying Party, in lieu of indemnifying such Indemnified Party, shall contribute to the amount paid or payable by such Indemnified Party as a result of such Losses, in such proportion as is appropriate to reflect the relative fault of the Indemnifying Party and Indemnified Party in connection with the actions, statements or omissions that resulted in such Losses as well as any other relevant equitable considerations. The relative fault of such Indemnifying Party and Indemnified Party shall be determined by reference to, among other things, whether any action in question, including any untrue or alleged untrue statement of a material fact or omission or alleged omission of a material fact, has been taken or made by, or relates to information supplied by, such Indemnifying Party or Indemnified Party, and the parties’ relative intent, knowledge, access to information and opportunity to correct or prevent such action, statement or omission. The amount paid or payable by a party as a result of any Losses shall be deemed to include, subject to the limitations set forth in this Agreement, any reasonable attorneys’ or other reasonable fees, expenses or disbursements incurred by such party in connection with any Proceeding to the extent such party would have been indemnified for such fees, expenses or disbursements if the indemnification provided for in this Section 5 was available to such party in accordance with its terms.
The parties hereto agree that it would not be just and equitable if contribution pursuant to this Section 5(d) were determined by pro rata allocation or by any other method of allocation that does not take into account the equitable considerations referred to in the immediately preceding paragraph. Notwithstanding the provisions of this Section 5(d), (A) no Holder shall be required to contribute, in the aggregate with any other amounts payable by it under this Section 5, any amount in excess of the net proceeds actually received by such Holder from the sale of the Registrable Securities giving rise to such contribution obligation and (B) no contribution will be made under circumstances where the maker of such contribution would not have been required to indemnify the Indemnified Party under the fault standards set forth in this Section 5. No person guilty of fraudulent misrepresentation (within the meaning of Section 11(f) of the Securities Act) shall be entitled to contribution from any Person who was not guilty of such fraudulent misrepresentation.
The indemnity and contribution agreements contained in this Section 5 are in addition to any liability that the Indemnifying Parties may have to the Indemnified Parties and are not in diminution or limitation of the indemnification provisions under the Purchase Agreement.
|
6. |
Miscellaneous. |
(a) Remedies. In the event of a breach by the Company or by a Holder of any of their obligations under this Agreement, each Holder or the Company, as the case may be, in addition to being entitled to exercise all rights granted by law and under this Agreement, including recovery of damages, will be entitled to seek specific performance of its rights under this Agreement, without the requirement of posting a bond. The Company and each Holder agree that monetary damages would not provide adequate compensation for any losses incurred by reason of a breach by it of any of the provisions of this Agreement and hereby further agrees that, in the event of any action for specific performance in respect of such breach, it shall waive the defense that a remedy at law would be adequate.
(b) No Piggyback on Registrations; Prohibition on Filing Other Registration Statements. Except and to the extent specified in the Purchase Agreement, neither the Company nor any of its security holders (other than the Holders in such capacity pursuant hereto) may include securities of the Company in a Registration Statement other than the Registrable Securities and the Company shall not prior to the Effective Date enter into any agreement providing any such right to any of its security holders. For the avoidance of doubt, the provisions of this Agreement shall not impact the terms of any lock-up agreement entered into by Purchaser for the benefit of the Company on or about the date hereof.
(c) Compliance. Each Holder covenants and agrees that it will comply with the prospectus delivery requirements of the Securities Act as applicable to it (unless an exemption therefrom is available) in connection with sales of Registrable Securities pursuant to the Registration Statement and shall sell the Registrable Securities only in accordance with a method of distribution described in the Registration Statement, or pursuant to an exemption from, or in a transaction not subject to, the registration requirements of the Securities Act.
(d) Discontinued Disposition. By its acquisition of Registrable Securities, each Holder agrees that, upon receipt of a notice from the Company of the occurrence of any event of the kind described in Section 3(c)(iii)-(vi), such Holder will forthwith discontinue disposition of such Registrable Securities under a Registration Statement until it is advised in writing (the “Advice”) by the Company that the use of the applicable Prospectus (as it may have been supplemented or amended) may be resumed. The Company will use its commercially reasonable efforts to ensure that the use of the Prospectus may be resumed as promptly as is practicable. Notwithstanding anything herein to the contrary, no Holder shall be required to discontinue disposition of Registrable Securities under a Registration Statement by virtue of the delivery by the Company of a notice of the occurrence of any event of the kind described in Section 3(c)(v) or Section 3(c)(vi) on more than two occasions or for more than 90 total calendar days, in each case during any twelve-month period, or for more than 45 calendar days during any 90-day period (an “Allowed Suspension”).
(e) No Inconsistent Agreements. Neither the Company nor any of its subsidiaries has entered, as of the date hereof, nor shall the Company or any of its subsidiaries, on or after the date hereof, enter into any agreement with respect to its securities, that would have the effect of impairing the rights granted to the Holders in this Agreement or otherwise conflicts with the provisions hereof.
(f) Amendments and Waivers. The provisions of this Agreement, including the provisions of this sentence, may not be amended, modified or supplemented, or waived unless the same shall be in writing and signed by the Company and Holders holding no less than a majority of the then outstanding Registrable Securities (for purposes of clarification, this includes any Registrable Securities issuable upon exercise or conversion of the Pre-Funded Warrants), provided that (i) any party may give a waiver as to itself and (ii) any proposed amendment that would, by its terms, have a disproportionate and materially adverse effect on any Holder shall require the consent of such Holder(s). Notwithstanding the foregoing, (1) a waiver or consent to depart from the provisions hereof with respect to a matter that relates exclusively to the rights of Holders and that does not directly or indirectly affect the rights of other Holders may be given by Holders of all of the Registrable Securities to which such waiver or consent relates and (2) none of the definitions of Filing Deadline, Effectiveness Deadline or Effectiveness Period, Section 2(e), Section 3(c), Section 5, Section 6(d), or the provisions of this sentence, may be amended, modified, or supplemented except with the consent of each Holder.
(g) Notices. Any and all notices or other communications or deliveries required or permitted to be provided hereunder shall be delivered as set forth in the Purchase Agreement.
(h) Successors and Assigns. This Agreement shall inure to the benefit of and be binding upon the successors and permitted assigns of each of the parties and shall inure to the benefit of each Holder. Nothing in this Agreement, express or implied, is intended to confer upon any party other than the parties hereto or their respective successors and assigns any rights, remedies, obligations, or liabilities under or by reason of this Agreement, except as expressly provided in this Agreement. The Company may not assign its rights (except by merger or in connection with another entity acquiring all or substantially all of the Company’s assets) or obligations hereunder without the prior written consent of all the Holders of the then outstanding Registrable Securities. Each Holder may assign its respective rights hereunder in the manner and to the Persons as permitted under the Purchase Agreement; provided in each case that (i) the Holder agrees in writing with the transferee or assignee to assign such rights and related obligations under this Agreement, and for the transferee or assignee to assume such obligations, and a copy of such agreement is furnished to the Company within a reasonable time after such assignment, (ii) the Company is, within a reasonable time after such transfer or assignment, furnished with written notice of the name and address of such transferee or assignee and the securities with respect to which such registration rights are being transferred or assigned, (iii) at or before the time the Company received the written notice contemplated by clause (ii) of this sentence, the transferee or assignee agrees in writing with the Company to be bound by all of the provisions contained herein and (iv) the transferee is an “accredited investor,” as that term is defined in Rule 501 of Regulation D.
(i) Execution and Counterparts. This Agreement may be executed in two or more counterparts, each of which when so executed shall be deemed to be an original and, all of which taken together shall constitute one and the same Agreement and shall become effective when counterparts have been signed by each party and delivered to the other party, it being understood that both parties need not sign the same counterpart. Counterparts may be delivered via facsimile, electronic mail (including any electronic signature covered by the U.S. federal ESIGN Act of 2000, Uniform Electronic Transactions Act, the Electronic Signatures and Records Act or other applicable law, e.g., www.docusign.com) or other transmission method and any counterpart so delivered shall be deemed to have been duly and validly delivered and be valid and effective for all purposes.
(j) Governing Law. All questions concerning the construction, validity, enforcement and interpretation of this Agreement shall be determined in accordance with the provisions of the Purchase Agreement.
(k) Cumulative Remedies. The remedies provided herein are cumulative and not exclusive of any other remedies provided by law.
(l) Severability. If any term, provision, covenant or restriction of this Agreement is held by a court of competent jurisdiction to be invalid, illegal, void or unenforceable, the remainder of the terms, provisions, covenants and restrictions set forth herein shall remain in full force and effect and shall in no way be affected, impaired or invalidated, and the parties hereto shall use their good faith reasonable efforts to find and employ an alternative means to achieve the same or substantially the same result as that contemplated by such term, provision, covenant or restriction. It is hereby stipulated and declared to be the intention of the parties that they would have executed the remaining terms, provisions, covenants and restrictions without including any of such that may be hereafter declared invalid, illegal, void or unenforceable.
(m) Headings. The headings in this Agreement are for convenience only and shall not limit or otherwise affect the meaning hereof.
(n) Independent Nature of Purchasers’ Obligations and Rights. The obligations of each Purchaser under this Agreement are several and not joint with the obligations of any other Purchaser hereunder, and no Purchaser shall be responsible in any way for the performance of the obligations of any other Purchaser hereunder. The decision of each Purchaser to purchase Securities pursuant to the Transaction Documents has been made by such Purchaser independently of any other Purchaser and independently of any information, materials, statements or opinions as to the business, affairs, operations, assets, properties, liabilities, results of operations, condition (financial or otherwise) or prospects of the Company or any Subsidiary which may have been made or given by any other Purchaser or by any agent or employee of any other Purchaser, and no Purchaser and any of its agents or employees shall have any liability to any other Purchaser (or any other Person) relating to or arising from any such information, materials, statement or opinions. Nothing contained herein or in any Transaction Document, and no action taken by any Purchaser pursuant hereto or thereto, shall be deemed to constitute the Purchasers as a partnership, an association, a joint venture or any other kind of entity, or create a presumption that the Purchasers are in any way acting in concert or as a group (including, without limitation, a “group” within the meaning of Section 13(d)(3) of the Exchange Act) with respect to such obligations or the transactions contemplated by the Transaction Documents. Each Purchaser acknowledges that no other Purchaser has acted as agent for such Purchaser in connection with making its investment hereunder and that no Purchaser will be acting as agent of such Purchaser in connection with monitoring its investment in the securities or enforcing its rights under the Transaction Documents. Each Purchaser shall be entitled to independently protect and enforce its rights, including, without limitation, the rights arising out of this Agreement or out of the other Transaction Documents, and it shall not be necessary for any other Purchaser to be joined as an additional party in any Proceeding for such purpose. The Company acknowledges that each of the Purchasers has been provided with the same Registration Rights Agreement for the purpose of closing a transaction with multiple Purchasers and not because it was required or requested to do so by any Purchaser. It is expressly understood that each provision contained in this Agreement is between the Company and a Purchaser, solely, and not between the Company and the Purchasers collectively and not between and among the Purchasers.
(o) Current Public Information. With a view to making available to the Holders the benefits of Rule 144 (or its successor rule) and any other rule or regulation of the Commission that may at any time permit the Holders to sell shares of Common Stock to the public without registration, for so long as the Registrable Securities remain outstanding, the Company covenants and agrees to use commercially reasonable efforts to: (i) make and keep adequate current public information available, as those terms are understood and defined in Rule 144, until such date on which the Holders no longer hold any Registrable Securities; and (ii) file with the Commission in a timely manner all reports and other documents required of the Company under the Exchange Act.
[REMAINDER OF PAGE INTENTIONALLY LEFT BLANK]
IN WITNESS WHEREOF, the parties have executed this Registration Rights Agreement as of the date first written above.
|
|
PIERIS PHARMACEUTICALS, INC. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
By: |
|
|
|
|
Name: Stephen S. Yoder Title: Chief Executive Officer |
|
|
|
|
|
|
|
IN WITNESS WHEREOF, the parties have executed this Registration Rights Agreement as of the date first written above.
|
|
[NAME OF INVESTING ENTITY] |
|
|
|
|
|
|
|
|
|
By: |
|
|
|
|
Name: Title: |
|
|
Annex A
PLAN OF DISTRIBUTION
We are registering the shares of common stock issued to the Selling Stockholders to permit the sale, transfer or other disposition of these shares by the Selling Stockholders or their donees, pledgees, transferees or other successors-in-interest from time to time after the date of this prospectus. We will not receive any of the proceeds from the sale by the Selling Stockholders of the shares of common stock. We will, or will procure to, bear all fees and expenses incident to our obligation to register the shares of common stock.
The Selling Stockholders may sell all or a portion of the shares of common stock beneficially owned by them and offered hereby from time to time directly or through one or more underwriters, broker-dealers or agents. If the shares of common stock are sold through underwriters or broker-dealers, the Selling Stockholders will be responsible for underwriting discounts (it being understood that the Selling Stockholders shall not be deemed to be underwriters solely as a result of their participation in this offering) or commissions or agent’s commissions. The shares of common stock may be sold on any national securities exchange or quotation service on which the securities may be listed or quoted at the time of sale, in the over-the-counter market or in transactions otherwise than on these exchanges or systems or in the over-the-counter market and in one or more transactions at fixed prices, at prevailing market prices at the time of the sale, at varying prices determined at the time of sale, or at negotiated prices. These sales may be effected in transactions, which may involve crosses or block transactions. The Selling Stockholders may use any one or more of the following methods when selling shares of common stock:
|
● |
ordinary brokerage transactions and transactions in which the broker-dealer solicits purchasers |
|
● |
block trades in which the broker-dealer will attempt to sell the shares as agent but may position and resell a portion of the block as principal to facilitate the transaction; |
|
● |
to or through underwriters or purchases by a broker-dealer as principal and resale by the broker-dealer for its account; |
|
● |
an exchange distribution in accordance with the rules of the applicable exchange; |
|
● |
privately negotiated transactions; |
|
● |
settlement of short sales entered into after the effective date of the registration statement of which this prospectus is a part; |
|
● |
broker-dealers may agree with the Selling Stockholders to sell a specified number of such shares at a stipulated price per share; |
|
● |
through the writing or settlement of options or other hedging transactions, whether such options are listed on an options exchange or otherwise; |
|
● |
a combination of any such methods of sale; and |
|
● |
any other method permitted pursuant to applicable law. |
The Selling Stockholders also may resell all or a portion of the shares of common stock in open market transactions in reliance upon Rule 144, as permitted by that rule, or Section 4(a)(1) under the Securities Act, if available, rather than under this prospectus, provided that they meet the criteria and conform to the requirements of those provisions.
Broker-dealers engaged by the Selling Stockholders may arrange for other broker-dealers to participate in sales. If the Selling Stockholders effect such transactions by selling shares of common stock shares of common stock to or through underwriters, broker-dealers or agents, such underwriters, broker-dealers or agents may receive commissions in the form of discounts, concessions or commissions from the Selling Stockholders or commissions from purchasers of the shares of common stock for whom they may act as agent or to whom they may sell as principal. Such commissions will be in amounts to be negotiated, but, except as set forth in a supplement to this prospectus, in the case of an agency transaction will not be in excess of a customary brokerage commission in compliance with FINRA Rule 2121; and in the case of a principal transaction a markup or markdown in compliance with FINRA IM-2121.01.
In connection with sales of the shares of common stock or otherwise, the Selling Stockholders may enter into hedging transactions with broker-dealers or other financial institutions, which may in turn engage in short sales of the shares of common stock in the course of hedging in positions they assume. The Selling Stockholders may also sell shares of common stock short and if such short sale takes place after the date that this registration statement is declared effective by the SEC, the Selling Stockholders may deliver shares of common stock covered by this prospectus to close out short positions and to return borrowed shares of common stock in connection with such short sales. The Selling Stockholders may also loan or pledge shares of common stock s to broker-dealers that in turn may sell such shares, to the extent permitted by applicable law. The Selling Stockholders may also enter into option or other transactions with broker-dealers or other financial institutions or the creation of one or more derivative securities which require the delivery to such broker-dealer or other financial institution of shares offered by this prospectus, which shares such broker-dealer or other financial institution may resell pursuant to this prospectus (as supplemented or amended to reflect such transaction). Notwithstanding the foregoing, the Selling Stockholders have been advised that they may not use shares of common stock the resale of which has been registered on this registration statement to cover short sales of our common stock made prior to the date the registration statement, of which this prospectus forms a part, has been declared effective by the SEC.
The Selling Stockholders may, from time to time, pledge or grant a security interest in some or all of the shares of common stock owned by them and, if they default in the performance of their secured obligations, the pledgees or secured parties may offer and sell the shares of common stock from time to time pursuant to this prospectus or any amendment to this prospectus under Rule 424(b)(3) or other applicable provision of the Securities Act, amending, if necessary, the list of Selling Stockholders to include the pledgee, transferee or other successors in interest as Selling Stockholders under this prospectus. The Selling Stockholders also may transfer and donate the shares of common stock in other circumstances in which case the transferees, donees, pledgees or other successors in interest will be the selling beneficial owners for purposes of this prospectus.
The Selling Stockholders and any broker-dealer or agents participating in the distribution of the shares of common stock may be deemed to be “underwriters” within the meaning of Section 2(11) of the Securities Act in connection with such sales. In such event, any commissions paid, or any discounts or concessions allowed to, any such broker-dealer or agent and any profit on the resale of the shares purchased by them may be deemed to be underwriting commissions or discounts under the Securities Act. Selling Stockholders who are “underwriters” within the meaning of Section 2(11) of the Securities Act will be subject to the applicable prospectus delivery requirements of the Securities Act including Rule 172 thereunder and may be subject to certain statutory liabilities of, including but not limited to, Sections 11, 12 and 17 of the Securities Act and Rule 10b-5 under the Exchange Act.
Each Selling Stockholder has informed us that it is not a registered broker-dealer and does not have any written or oral agreement or understanding, directly or indirectly, with any person to distribute the shares of common stock. Upon us being notified in writing by a Selling Stockholder that any material arrangement has been entered into with a broker-dealer for the sale of common stock through a block trade, special offering, exchange distribution or secondary distribution or a purchase by a broker or dealer, a supplement to this prospectus will be filed, if required, pursuant to Rule 424(b) under the Securities Act, disclosing (i) the name of each such Selling Stockholder and of the participating broker-dealer(s), (ii) the number of shares of common stock involved, (iii) the price at which such the shares of common stock were sold, (iv) the commissions paid or discounts or concessions allowed to such broker-dealer(s), where applicable, (v) that such broker-dealer(s) did not conduct any investigation to verify the information set out in this prospectus, and (vi) other facts material to the transaction. In no event shall any broker-dealer receive fees, commissions and markups, which, in the aggregate, would exceed eight percent (8.0%).
Under the securities laws of some U.S. states, the shares of common stock may be sold in such states only through registered or licensed brokers or dealers. In addition, in some U.S. states the shares of common stock may not be sold unless such shares have been registered or qualified for sale in such state or an exemption from registration or qualification is available and is complied with.
There can be no assurance that any Selling Stockholder will sell any or all of the shares of common stock registered pursuant to the shelf registration statement, of which this prospectus forms a part.
Each Selling Stockholder and any other person participating in such distribution will be subject to applicable provisions of the Exchange Act and the rules and regulations thereunder, including, without limitation, to the extent applicable, Regulation M of the Exchange Act, which may limit the timing of purchases and sales of any of the shares of common stock by the Selling Stockholder and any other participating person. To the extent applicable, Regulation M may also restrict the ability of any person engaged in the distribution of the shares of common stock to engage in market-making activities with respect to the shares of common stock. All of the foregoing may affect the marketability of the shares of common stock and the ability of any person or entity to engage in market-making activities with respect to the shares of common stock.
We will pay all expenses of the registration of the shares of common stock set forth in the registration rights agreements, including, without limitation, SEC filing fees and expenses of compliance with state securities or “blue sky” laws; provided, however, that each Selling Stockholder will pay all underwriting discounts and selling commissions, if any and any related legal expenses incurred by it. We will indemnify the Selling Stockholders against certain liabilities, including some liabilities under the Securities Act, in accordance with the registration rights agreement, or the Selling Stockholders will be entitled to contribution. We may be indemnified by the Selling Stockholders against certain civil liabilities set forth in the registration rights agreement, including liabilities under the Securities Act, that may arise from any written information furnished to us by the Selling Stockholders specifically for use in this prospectus, in accordance with the related registration rights agreements, or we may be entitled to contribution.
Annex B
Pieris Pharmaceuticals, Inc.
Selling Stockholder Notice and Questionnaire
The undersigned beneficial owner of shares of common stock (the “Registrable Securities”) of Pieris Pharmaceuticals, Inc. (the “Company”), understands that the Company has filed or intends to file with the Securities and Exchange Commission (the “Commission”) a registration statement (the “Registration Statement”) for the registration and resale under Rule 415 of the Securities Act of 1933, as amended (the “Securities Act”), of the Registrable Securities issued to the undersigned in connection with its execution of the Common Stock Purchase Agreement, dated July 23, 2024 (the “Purchase Agreement”). All capitalized terms not otherwise defined herein shall have the meanings given to them in the Purchase Agreement
This Questionnaire will assist the Company in gathering information needed to include you as a selling stockholder in the Registration Statement; information you provide may be included in the Registration Statement and related prospectus.
Certain legal consequences arise from being named as a selling stockholder in the Registration Statement and the related prospectus. Accordingly, holders and beneficial owners of Registrable Securities are advised to consult their own securities law counsel regarding the consequences of being named or not being named as a selling stockholder in the Registration Statement and the related prospectus.
NOTICE
The undersigned beneficial owner (the “Selling Stockholder”) of Registrable Securities hereby elects to include the Registrable Securities owned by it in the Registration Statement.
The undersigned hereby provides the following information to the Company and represents and warrants that such information is accurate:
QUESTIONNAIRE
|
1. |
Name. |
|
(a) |
Full Legal Name of Selling Stockholder |
|
(b) |
Full Legal Name of Registered Holder (if not the same as (a) above) through which Registrable Securities are held: |
|
© |
Full Legal Name of Natural Control Person (which means a natural person who directly or indirectly alone or with others has power to vote or dispose of the securities covered by this Questionnaire): |
2. Address for Notices to Selling Stockholder:
|
Telephone:
|
|
Fax:
|
|
Contact Person:
|
3. Broker-Dealer Status:
|
(a) |
Are you a broker-dealer? |
Yes ☐ No ☐
|
(b) |
If “yes” to Section 3(a), did you receive your Registrable Securities as compensation for investment banking services to the Company? |
Yes ☐ No ☐
|
Note: |
If “no” to Section 3(b), the Commission’s staff has indicated that you should be identified as an underwriter in the Registration Statement. |
|
(c) |
Are you an affiliate of a broker-dealer? |
Yes ☐ No ☐
|
(d) |
If you are an affiliate of a broker-dealer, do you certify that you purchased the Registrable Securities in the ordinary course of business, and at the time of the purchase of the Registrable Securities to be resold, you had no agreements or understandings, directly or indirectly, with any person to distribute the Registrable Securities? |
Yes ☐ No ☐
|
Note: |
If “no” to Section 3(d), the Commission’s staff has indicated that you should be identified as an underwriter in the Registration Statement. |
4. Beneficial Ownership of Securities of the Company Owned by the Selling Stockholder.
Except as set forth below in this Item 4, the undersigned is not the beneficial or registered owner of any securities of the Company other than the securities issuable pursuant to the Purchase Agreement.
|
(a) |
Type and Amount of other securities beneficially owned by the Selling Stockholder: |
|
5. Relationships with the Company:
Except as set forth below, neither the undersigned nor any of its affiliates, officers, directors, or principal equity holders (owners of 5% of more of the equity securities of the undersigned) has held any position or office or has had any other material relationship with the Company (or its predecessors or affiliates) during the past three years.
State any exceptions here:
The undersigned agrees to promptly notify the Company of any material inaccuracies or changes in the information provided herein that may occur after the date of this questionnaire at any time while the Registration Statement remains effective; provided, that the undersigned shall not be required to notify the Company of any changes to the number of securities held or owned by the undersigned or its affiliates.
By signing below, the undersigned consents to the disclosure of the information contained herein in its answers to Items 1 through 5 and the inclusion of such information in the Registration Statement and the related prospectus and any amendments or supplements thereto. The undersigned understands that such information will be relied upon by the Company in connection with the preparation or amendment of the Registration Statement and the related prospectus and any amendments or supplements thereto.
IN WITNESS WHEREOF the undersigned, by authority duly given, has caused this Notice and Questionnaire to be executed and delivered either in person or by its duly authorized agent.
|
Date: _________________ |
[NAME OF INVESTING ENTITY] |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
By: |
|
|
|
|
|
Name: |
|
|
|
|
Title: |
|
INFORMATION NOT REQUIRED IN PROXY
STATEMENT/PROSPECTUS
Item 20. Indemnification of Directors and Officers
The Registrant is a Nevada corporation and is generally governed by the NRS.
The Registrant’s amended and restated articles of incorporation and amended and restated bylaws, as amended, provide that each person who was or is made a party or is threatened to be made a party to or is otherwise involved (including, without limitation, as a witness) in any action, suit or proceeding, whether civil, criminal, administrative or investigative, by reason of the fact that he or she is or was one of the Registrant’s directors or officers or is or was serving at its request as a director, officer or trustee of another corporation, partnership, joint venture, trust or other enterprise, including service with respect to an employee benefit plan, whether the basis of such action, suit or proceeding is alleged action in an official capacity as a director, officer or trustee or in any other capacity while serving as a director, officer or trustee, shall be indemnified and held harmless by us to the fullest extent authorized by NRS against all expense, liability and loss (including attorneys’ fees and amounts paid in settlement) reasonably incurred or suffered by such indemnitee in connection therewith.
NRS 78.7502 permits a corporation to indemnify, pursuant to that statutory provision, a present or former director, officer, employee or agent of the corporation, or of another entity or enterprise (including as a manager of a limited liability company), for which such person is or was serving in such capacity at the request of the corporation, who was or is a party or is threatened to be made a party to any threatened, pending or completed action, suit or proceeding, except an action by or in the right of the corporation, against expenses, including attorneys’ fees, judgments, fines and amounts paid in settlement actually and reasonably incurred in connection therewith, arising by reason of such person’s service in such capacity if such person (i) is not liable pursuant to NRS 78.138, or (ii) acted in good faith and in a manner which he or she reasonably believed to be in or not opposed to the best interests of the corporation and, with respect to a criminal action or proceeding, had no reasonable cause to believe his or her conduct was unlawful. In the case of actions brought by or in the right of the corporation, however, no indemnification pursuant to NRS 78.7502 may be made for any claim, issue or matter as to which such person has been adjudged by a court of competent jurisdiction, after exhaustion of all appeals therefrom, to be liable to the corporation or for amounts paid in settlement to the corporation, unless and only to the extent that the court in which the action or suit was brought or other court of competent jurisdiction determines upon application that in view of all the circumstances of the case, such person is fairly and reasonably entitled to indemnity for such expenses as the court deems proper.
Any discretionary indemnification pursuant to the statutory mechanism provided under NRS 78.7502, unless ordered by a court or advanced to a director or officer by the corporation in accordance with the NRS, may be made by a corporation only as authorized in each specific case upon a determination that indemnification of the director, officer, employee or agent is proper in the circumstances. Such determination must be made (1) by the stockholders, (2) by the board of directors by majority vote of a quorum consisting of directors who were not parties to the action, suit or proceeding, (3) if a majority vote of a quorum consisting of directors who were not parties to the action, suit or proceeding so orders, by independent legal counsel in a written opinion, or (4) if a quorum consisting of directors who were not parties to the action, suit or proceeding cannot be obtained, by independent legal counsel in a written opinion.
NRS 78.751 further provides that indemnification pursuant to the statutory mechanism provided under NRS 78.7502 does not exclude any other rights to which a person seeking indemnification or advancement of expenses may be entitled under the Registrant’s amended and restated articles of incorporation, or any bylaw, agreement, vote of stockholders or disinterested directors or otherwise, for either an action in the person’s official capacity or an action in another capacity while holding office, except that indemnification, unless ordered by a court pursuant to NRS 78.7502 or for the advancement of expenses, may not be made to or on behalf of any director or officer finally adjudged by a court of competent jurisdiction, after exhaustion of any appeals, to be liable for intentional misconduct, fraud or a knowing violation of law, and such misconduct, fraud or violation was material to the cause of action.
The Registrant’s amended and restated articles of incorporation provide that the liability of its directors and officers shall be eliminated or limited to the fullest extent permitted by the NRS. NRS 78.138(7) provides that, subject to limited statutory exceptions and unless the articles of incorporation or an amendment thereto (in each case filed on or after October 1, 2003) provide for greater individual liability, a director or officer is not individually liable to a corporation or its stockholders or creditors for any damages as a result of any act or failure to act in his or her capacity as a director or officer unless it is proven that: (i) the act or failure to act constituted a breach of his or her fiduciary duties as a director or officer and (ii) the breach of those duties involved intentional misconduct, fraud or a knowing violation of law.
The Registrant has entered into indemnification agreements with its directors and certain officers, in addition to the indemnification provided in the NRS, its amended and restated articles of incorporation and amended and restated bylaws, as amended, and intends to enter into indemnification agreements with any new directors and officers in the future. The Registrant has purchased and intends to maintain insurance on behalf of any person who is or was a director or officer against any loss arising from any claim asserted against him or her and incurred by him or her in any such capacity, subject to certain exclusions.
The foregoing discussion of the Registrant’s amended and restated articles of incorporation, amended and restated bylaws, as amended, indemnification agreements, and Nevada law is not intended to be exhaustive and is qualified in its entirety by such amended and restated articles of incorporation, amended and restated bylaws, as amended, indemnification agreements, or law.
Item 21. Exhibits and Financial Statement Schedules
(a) Exhibit Index
A list of exhibits filed with this Registration Statement on Form S-4 is set forth on the Exhibit Index and is incorporated herein by reference.
(b) Financial Statements
The financial statements filed with this Registration Statement on Form S-4 are set forth on the indices to the section titled “Financial Statements and Supplementary Data” and are incorporated herein by reference.
Item 22. Undertakings
(a) The undersigned registrant hereby undertakes as follows:
|
(1) |
That prior to any public reoffering of the securities registered hereunder through use of a prospectus which is a part of this registration statement, by any person or party who is deemed to be an underwriter within the meaning of Rule 145(c), the issuer undertakes that such reoffering proxy statement/prospectus will contain the information called for by the applicable registration form with respect to reofferings by persons who may be deemed underwriters, in addition to the information called for by the other items of the applicable form. |
|
(2) |
That every prospectus (i) that is filed pursuant to paragraph (a)(2) immediately preceding, or (ii) that purports to meet the requirements of Section 10(a)(3) of the Securities Act and is used in connection with an offering of securities subject to Rule 415, will be filed as a part of an amendment to the registration statement and will not be used until such amendment is effective, and that, for purposes of determining any liability under the Securities Act, each such post-effective amendment shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. |
(b) To respond to requests for information that is incorporated by reference into the prospectus pursuant to Items 4, 10(b), 11, or 13 of this Form, within one business day of receipt of such request, and to send the incorporated documents by first class mail or other equally prompt means. This includes information contained in documents filed subsequent to the effective date of the registration statement through the date of responding to the request.
(c) To supply by means of a post-effective amendment all information concerning a transaction, and the company being acquired involved therein, that was not the subject of and included in the registration statement when it became effective.
Insofar as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers, and controlling persons pursuant to the foregoing provisions, or otherwise, the registrant has been advised that, in the opinion of the SEC, such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment of expenses incurred or paid by a director, officer or controlling person in a successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to the court of appropriate jurisdiction the question whether such indemnification by the registrant is against public policy as expressed in the Securities Act and will be governed by the final adjudication of such issue.
EXHIBIT INDEX
|
Incorporated by Reference (if applicable) |
||||||||||
|
Exhibit Number |
Exhibit Description |
Form |
File No. |
Exhibit |
Filing Date |
|||||
|
2.1 |
8-K |
001-37471 |
2.1 |
July 24, 2024 |
||||||
|
2.2 |
8-K |
001-37471 |
10.1 |
July 24, 2024 |
||||||
|
2.3 |
8-K |
001-37471 |
10.2 |
July 24, 2024 |
||||||
|
2.4 |
8-K |
001-37471 |
10.3 |
July 24, 2024 |
||||||
|
2.5 |
8-K |
001-37471 |
10.4 |
July 24, 2022 |
||||||
|
3.1 |
Amended and Restated Articles of Incorporation of Pieris Pharmaceuticals, Inc. |
8-K |
333-190728 |
3.1 |
December 18, 2014 |
|||||
|
3.2 |
Certificate of Designation of Series A Convertible Preferred Stock |
10-Q |
001-37471 |
3.1 |
August 11, 2016 |
|||||
|
3.3 |
Certificate of Designation of Series B Convertible Preferred Stock |
8-K |
001-37471 |
3.1 |
February 4, 2019 |
|||||
|
3.4 |
Certificate of Designation of Series C Convertible Preferred Stock |
8-K |
001-37471 |
3.1 |
November 4, 2019 |
|||||
|
3.5 |
Certificate of Designation of Series D Convertible Preferred Stock |
8-K |
001-37471 |
3.1 |
April 6, 2020 |
|||||
|
3.6 |
Certificate of Designation of Series E Convertible Preferred Stock |
8-K |
001-37471 |
3.1 |
May 21, 2021 |
|||||
|
3.7 |
8-K | 001-37471 | 3.1 | August 8, 2024 | ||||||
|
3.8 |
Certificate of Change to Articles of Incorporation of Pieris Pharmaceuticals, Inc. |
8-K |
001-37471 |
3.1 |
April 18, 2024 |
|||||
|
3.9 |
8-K |
333-190728 |
3.2 |
December 18, 2014 |
||||||
|
3.10 |
Amendment to the Amended and Restated Bylaws of the Registrant |
8-K |
001-37471 |
3.1 |
September 3, 2019 |
|||||
|
4.1 |
10-K |
001-37471 |
4.2 |
March 23, 2016 |
||||||
|
5.1* |
Opinion of Brownstein Hyatt Farber Schreck, LLP (previously filed) | |||||||||
|
8.1* |
||||||||||
|
8.2* |
||||||||||
|
10.1 |
8-K |
333-190728 |
10.10 |
December 18, 2014 |
||||||
|
10.2 |
Pieris Pharmaceuticals, Inc. 2018 Employee Stock Purchase Plan |
8-K |
001-37471 |
10.2 |
July 26, 2018 |
|||||
|
10.3 |
Pieris Pharmaceuticals, Inc. 2023 Employee Stock Purchase Plan |
10-Q |
001-37471 |
10.2 |
August 10, 2023 |
|||||
|
10.4 |
Pieris Pharmaceuticals, Inc. 2019 Employee, Director and Consultant Equity Incentive Plan |
8-K |
001-37471 |
10.1 |
July 31, 2019 |
|||||
|
10.5 |
Pieris Pharmaceuticals, Inc. 2020 Employee, Director and Consultant Equity Incentive Plan |
8-K |
001-37471 |
10.1 |
June 29, 2020 |
|||||
|
10.6 |
8-K |
001-37471 |
10.1 |
June 29, 2021 |
||||||
|
10.7 |
8-K |
001-37471 |
10.1 |
June 27, 2022 |
||||||
| 10.8 |
8-K |
0001-37471 |
10.1 |
June 26, 2023 |
||||||
|
10.9* |
Palvella Therapeutics, Inc. 2019 Equity Incentive Plan (previously filed) |
|||||||||
|
10.10* |
||||||||||
|
10.11* |
||||||||||
|
10.12* |
Amendment No. 1 to Palvella Therapeutics, Inc. 2019 Equity Incentive Plan (previously filed) |
|||||||||
|
10.13* |
||||||||||
|
10.14* |
||||||||||
|
10.15* |
Form of 2024 Convertible Note Purchase Agreement (previously filed) |
|||||||||
|
10.16* |
||||||||||
|
10.17* |
||||||||||
|
10.18*^ |
|
* |
Previously filed. |
|
** |
Filed herewith. |
|
+ |
Portions of this exhibit have been redacted in compliance with Regulation S-K Item 601(a)(6). |
|
^ |
Certain confidential information contained in this exhibit, marked by brackets, has been omitted pursuant to Item 601(b)(10)(iv) because the information (i) is not material and (ii) is the type of information that Palvella both customarily and actually treats as private and confidential. |
SIGNATURES
Pursuant to the requirements of the Securities Act, the registrant has duly caused this registration statement to be signed on its behalf by the undersigned, thereunto duly authorized, in the City of Boston, Commonwealth of Massachusetts, on October 29, 2024.
| PIERIS PHARMACEUTICALS, INC. | |||
| By: | /s/ Stephen S. Yoder | ||
| Name: | Stephen S. Yoder | ||
| Title: | President and Chief Executive Officer | ||
| (principal executive officer) | |||
Pursuant to the requirements of the Securities Act, this registration statement has been signed below by the following persons in the capacities and on the dates indicated.
|
Signature |
Title |
Date |
||
|
/s/ Stephen S. Yoder |
President and Chief Executive Officer (Principal Executive Officer) |
October 29, 2024 | ||
|
Stephen S. Yoder |
||||
|
/s/ Thomas Bures |
Chief Financial Officer (Principal Financial Officer and Principal Accounting Officer) |
October 29, 2024 | ||
|
Thomas Bures |
||||
|
* |
Chairman of the Board of Directors |
October 29, 2024 | ||
|
James Geraghty |
||||
|
* |
Director |
October 29, 2024 | ||
|
Ann Barbier |
||||
|
* |
Director |
October 29, 2024 | ||
|
Peter Kiener |
||||
|
* |
Director |
October 29, 2024 | ||
|
Christopher Kiritsy |
||||
|
* |
Director |
October 29, 2024 | ||
|
Michael Richman |
||||
|
* |
Director |
October 29, 2024 | ||
| Maya R. Said | ||||
| * | Director | October 29, 2024 | ||
| Matthew L. Sherman |
|
*By: /s/ Stephen S. Yoder |
|
Stephen S. Yoder |
|
Attorney-in-fact |